Submitted:
26 December 2025
Posted:
29 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
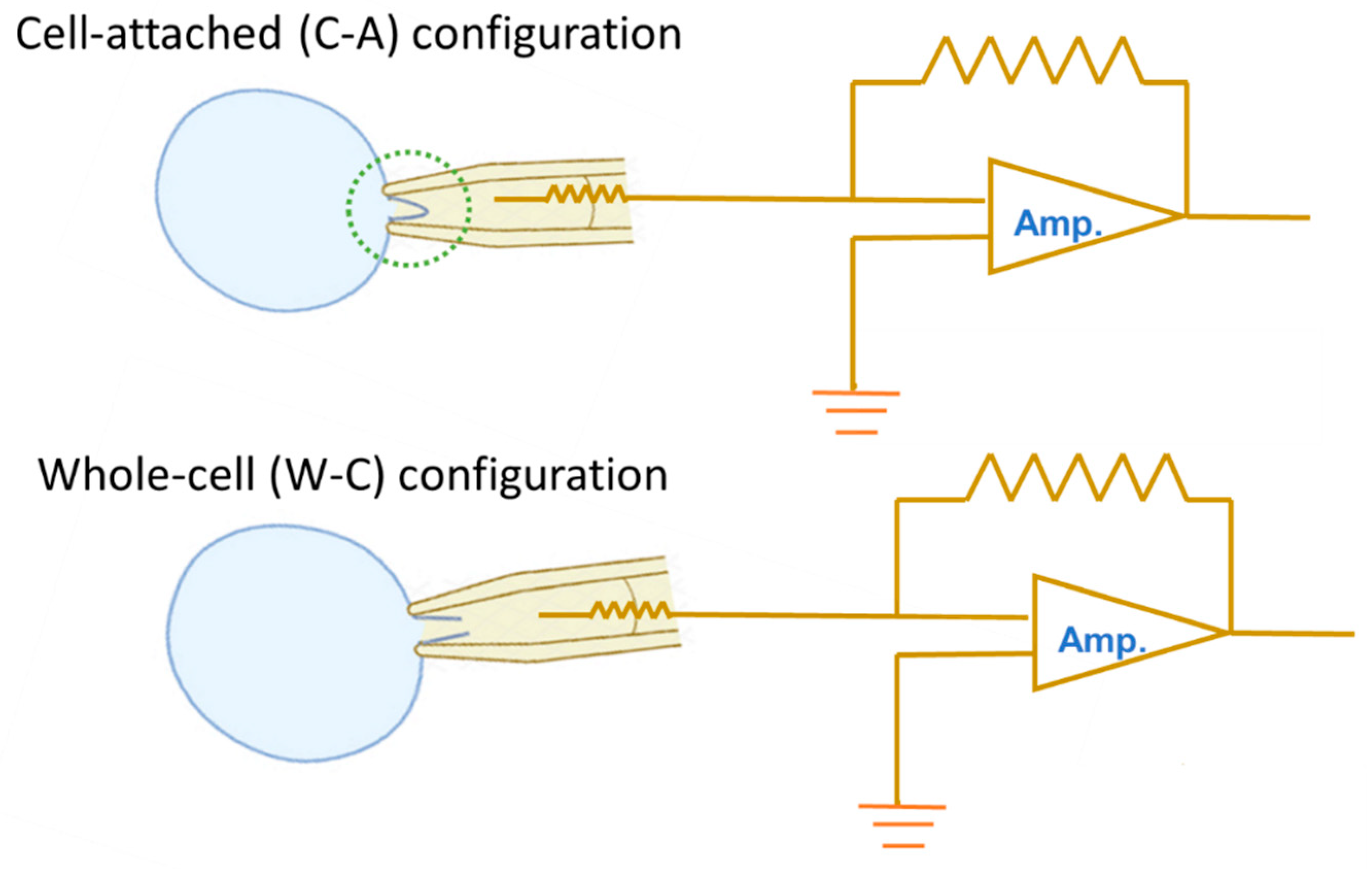
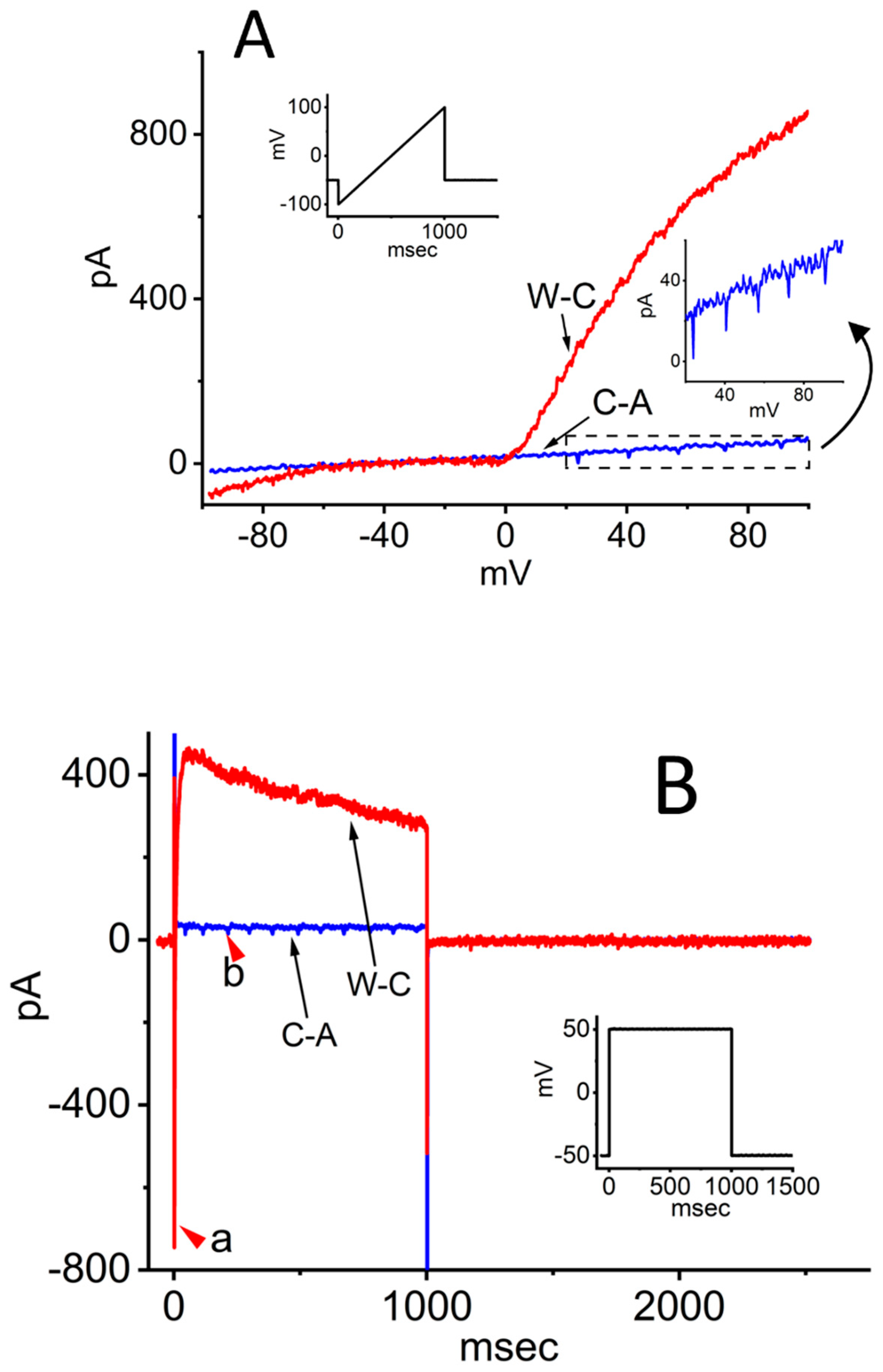
2. Ionic Currents Evoked by Ramp or Rectangular Pulse Recorded Under Cell-Attached (C-A) and Whole-Cell (W-C) Voltage-Clamp Configurations
3. Measurement of Resting Membrane Potential Under C-A Mode
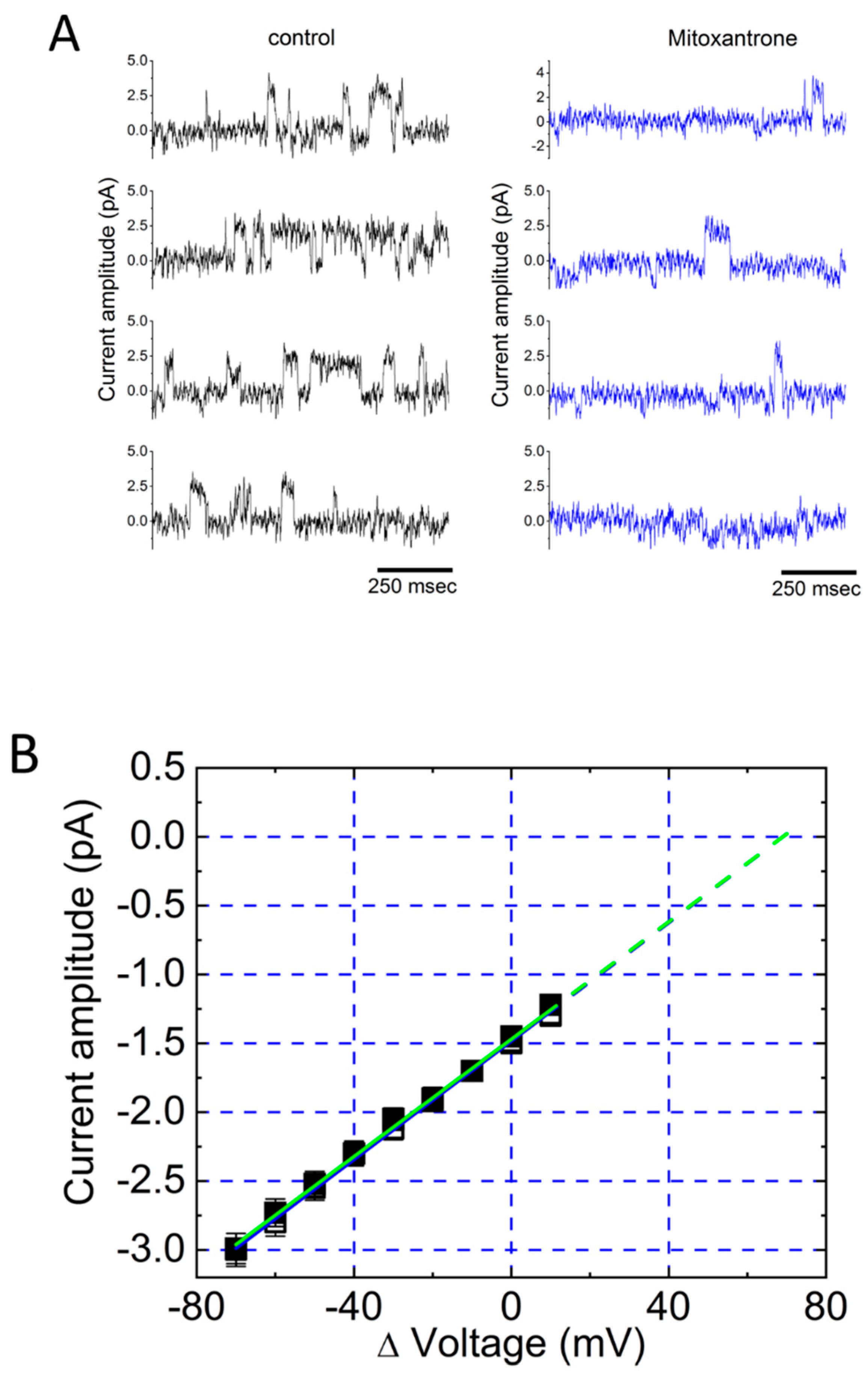
4. Differences Between ATP-Sensitive K+ (KATP) Channels and Large-Conductance Ca2+-Activated K+ (BKCa) Channels in the C-A Configuration
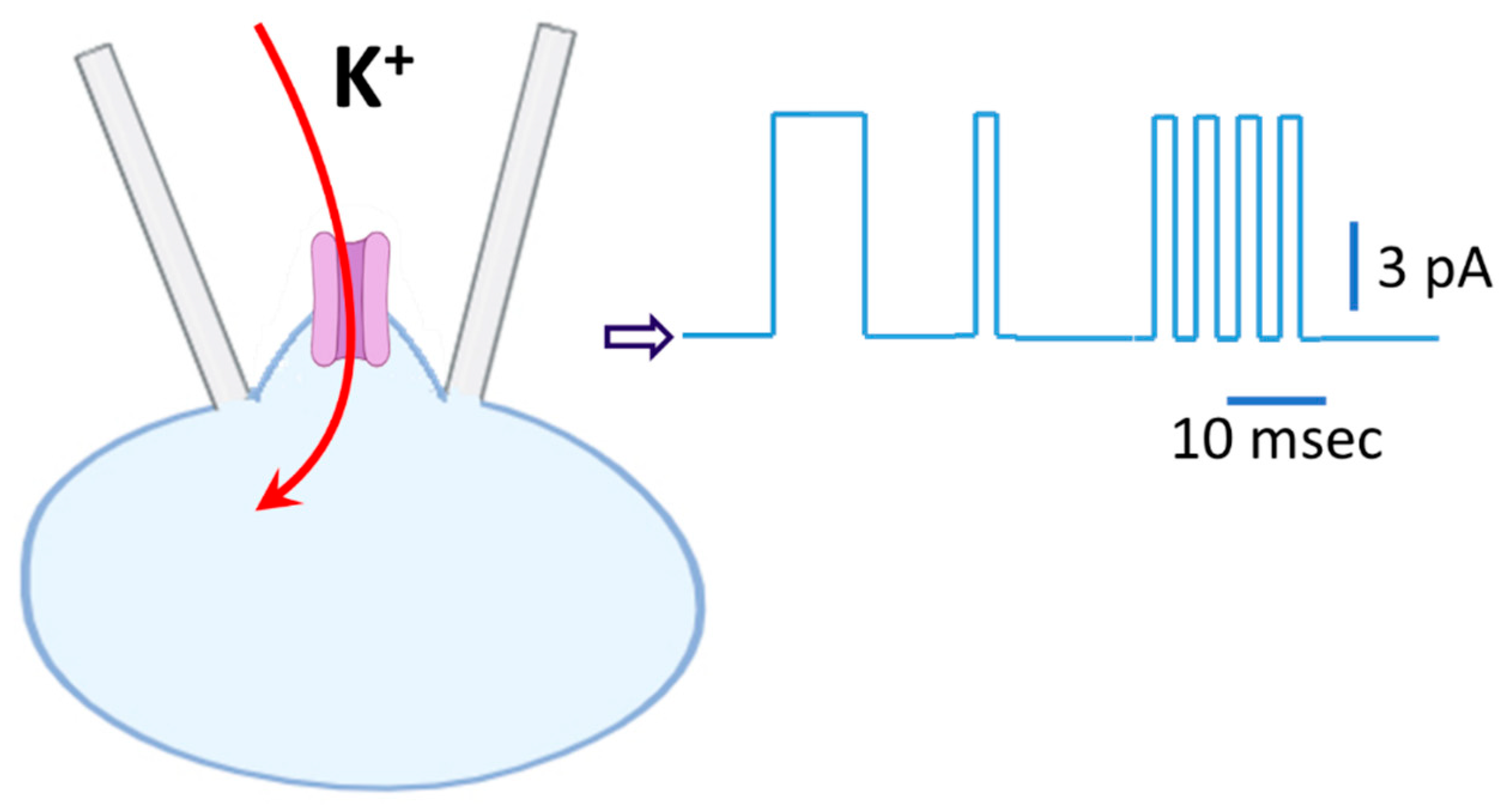
5. Interference of Single-Channel Activity by Action Currents (ACs) in C-A Current Recordings
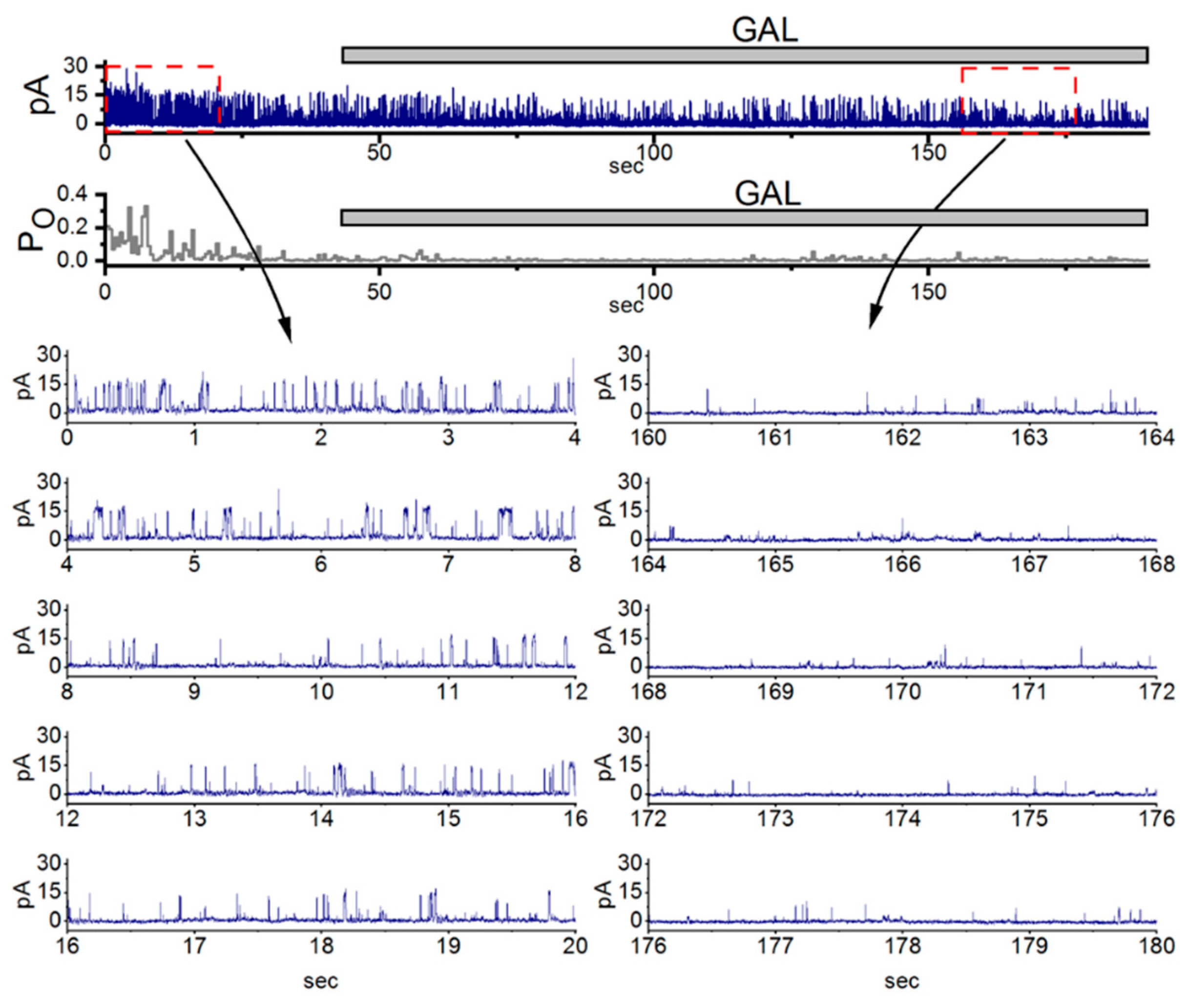
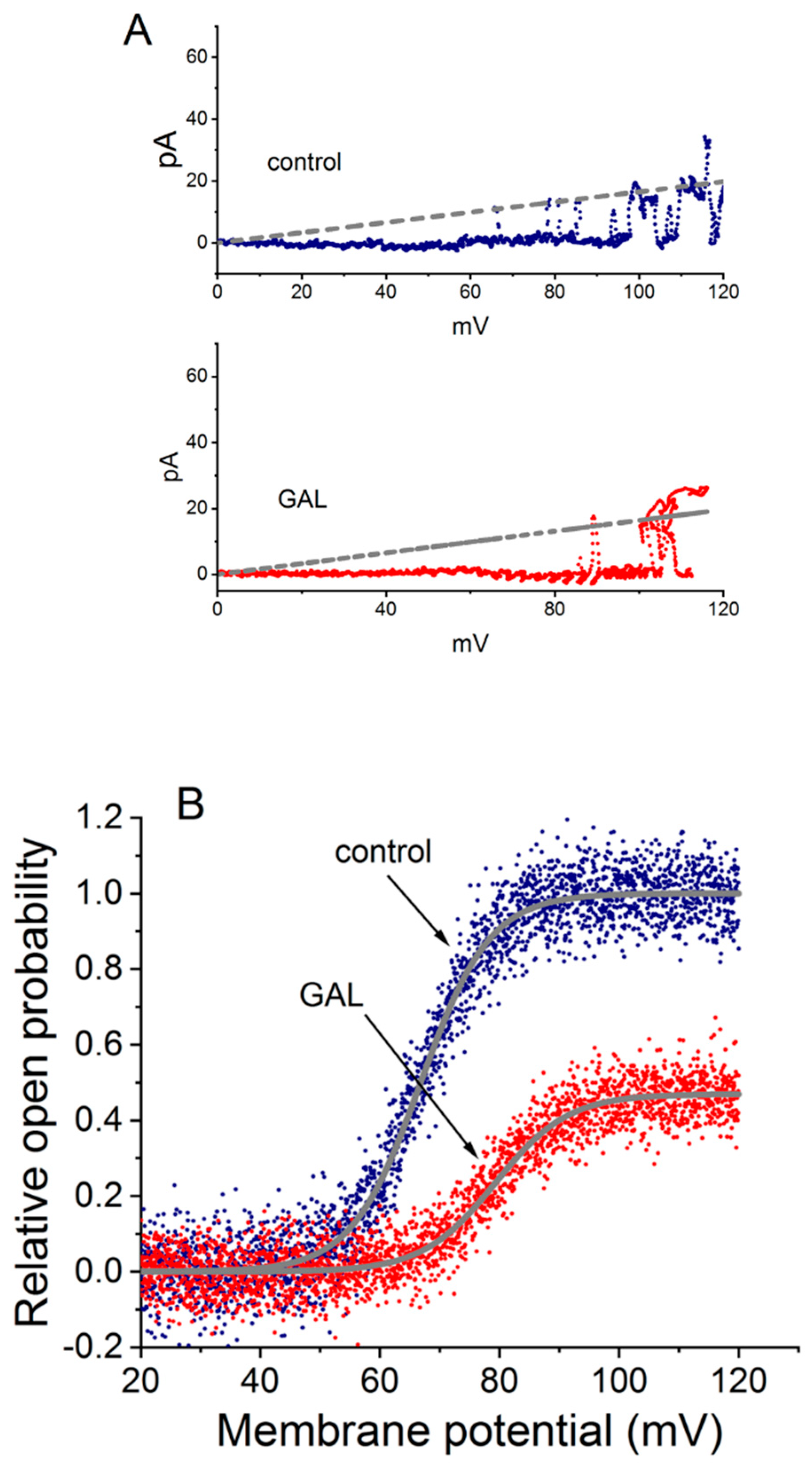
6. Modification of BKCa-Channel Activity by GAL-021 in GH3 Cells
7. Brain Slice Patch-Clamp Recordings
8. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Alcami, P; Franconville, R; Llano, I; Marty, A. Measuring the firing rate of high-resistance neurons with cell-attached recording. J Neurosci 2012, 32(9), 3118–30. [Google Scholar] [CrossRef]
- Ashcroft, FM; Ashcroft, SJ; Harrison, DE. Effects of 2-ketoisocaproate on insulin release and single potassium channel activity in dispersed rat pancreatic beta-cells. J Physiol 1987, 385, 517–29. [Google Scholar] [CrossRef] [PubMed]
- Axmacher, N; Miles, R. Intrinsic cellular currents and the temporal precision of EPSP-action potential coupling in CA1 pyramidal cells. J Physiol 2004, 555 Pt 3, 713–25. [Google Scholar] [CrossRef]
- Bak, A; Koch, H; van Loo, KMJ; Schmied, K; Gittel, B; Weber, Y; Ort, J; Schwarz, N; Tauber, SC; Wuttke, TV; Delev, D. Human organotypic brain slice cultures: a detailed and improved protocol for preparation and long-term maintenance. J Neurosci Methods 2024, 404, 110055. [Google Scholar] [CrossRef]
- Clark, RB; Nakajima, T; Giles, W; Kanai, K; Momose, Y; Szabo, G. Two distinct types of inwardly rectifying K+ channels in bull-frog atrial myocytes. J Physiol 1990, 424, 229–51. [Google Scholar] [CrossRef]
- Dalla Costa, G; Comi, G. Updates on immunosuppressant safety and malignancy risk in patients with multiple sclerosis. Expert Opin Drug Saf 2025, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Fenwick, EM; Marty, A; Neher, E. A patch-clamp study of bovine chromaffin cells and of their sensitivity to acetylcholine. J Physiol 1982, 331, 577–97. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Chiappe, F; Muraro, NI. Patch-Clamping Fly Brain Neurons. Cold Spring Harb Protoc 2022, 2022(8), Pdb.top107796. [Google Scholar] [CrossRef]
- Fricker, D; Verheugen, JA; Miles, R. Cell-attached measurements of the firing threshold of rat hippocampal neurones. J Physiol 1999, 517 Pt 3)(Pt 3, 791–804. [Google Scholar] [CrossRef]
- Hallermann, S; de Kock, CP; Stuart, GJ; Kole, MH. State and location dependence of action potential metabolic cost in cortical pyramidal neurons. Nat Neurosci 2012, 15(7), 1007–14. [Google Scholar] [CrossRef]
- Hamill, OP; Marty, A; Neher, E; Sakmann, B; Sigworth, FJ. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflugers Arch 1981, 391(2), 85–100. [Google Scholar] [CrossRef] [PubMed]
- Haug, TM; Hafting, T; Sand, O. Inhibition of BK channels contributes to the second phase of the response to TRH in clonal rat anterior pituitary cells. Acta Physiol Scand 2004, 180(4), 347–57. [Google Scholar] [CrossRef]
- He, LP; Mears, D; Atwater, I; Kitasato, H. Glucagon induces suppression of ATP-sensitive K+ channel activity through a Ca2+/calmodulin-dependent pathway in mouse pancreatic beta-cells. J Membr Biol 1998, 166(3), 237–44. [Google Scholar] [CrossRef]
- Hibino, H; Inanobe, A; Furutani, K; Murakami, S; Findlay, I; Kurachi, Y. Inwardly rectifying potassium channels: their structure, function, and physiological roles. Physiol Rev 2010, 90(1), 291–366. [Google Scholar] [CrossRef]
- Huang, CW; Huang, CC; Cheng, JT; Tsai, JJ; Wu, SN. Glucose and hippocampal neuronal excitability: role of ATP-sensitive potassium channels. J Neurosci Res 2007, 85(7), 1468–77. [Google Scholar] [CrossRef]
- Huang, HY; Huang, YB; Wu, CL; Wu, SN. Modulatory impact of tefluthrin, telmisartan, and KB-R7943 on voltage-gated Na+ currents. Biophysica 2024, 4(4), 488–506. [Google Scholar] [CrossRef]
- Hung, TY; Huang, HI; Wu, SN; Huang, CW. Depressive effectiveness of vigabatrin (γ-vinyl-GABA), an antiepileptic drug, in intermediate-conductance calcium-activated potassium channels in human glioma cells. BMC Pharmacol Toxicol 2021, 22(1), 6. [Google Scholar] [CrossRef] [PubMed]
- Kusunoki, M; Hirota, K; Shoji, T; Uba, T; Matsuo, Y; Hayashi, M. Dexmedetomidine suppresses glucose-stimulated insulin secretion in pancreatic β-cells. FEBS Open Bio 2025, 15(4), 634–646. [Google Scholar] [CrossRef]
- Lee, DS; Chen, JH; Lundy, DJ; Liu, CH; Hwang, SM; Pabon, L; Shieh, RC; Chen, CC; Wu, SN; Yan, YT; Lee, ST; Chiang, PM; Chien, S; Murry, CE; Hsieh, PC. Defined MicroRNAs Induce Aspects of Maturation in Mouse and Human Embryonic-Stem-Cell-Derived Cardiomyocytes. Cell Rep 2015, 12(12), 1960–7. [Google Scholar] [CrossRef]
- Lieste, JR; Koopman, WJ; Reynen, VC; Scheenen, WJ; Jenks, BG; Roubos, EW. Action currents generate stepwise intracellular Ca2+ patterns in a neuroendocrine cell. J Biol Chem 1998, 273(40), 25686–94. [Google Scholar] [CrossRef]
- Liu, YW; Fang, YH; Su, CT; Hwang, SM; Liu, PY; Wu, SN. The biochemical and electrophysiological profiles of amniotic fluid-derived stem cells following Wnt signaling modulation cardiac differentiation. Cell Death Discov 2019, 5, 59. [Google Scholar] [CrossRef]
- Lu, TL; Gao, ZH; Li, SW; Wu, SN. High Efficacy by GAL-021: A Known Intravenous Peripheral Chemoreceptor Modulator that Suppresses BKCa-Channel Activity and Inhibits IK(M) or Ih. Biomolecules 2020, 10(2), 188. [Google Scholar] [CrossRef]
- Lo, YC; Tseng, YT; Liu, CM; Wu, BN; Wu, SN. Actions of KMUP-1, a xanthine and piperazine derivative, on voltage-gated Na+ and Ca2+ -activated K+ currents in GH3 pituitary tumour cells. Br J Pharmacol 2015, 172(21), 5110–22. [Google Scholar] [CrossRef]
- Mahon, S; Casassus, G; Mulle, C; Charpier, S. Spike-dependent intrinsic plasticity increases firing probability in rat striatal neurons in vivo. J Physiol 2003, 550 Pt 3, 947–59. [Google Scholar] [CrossRef] [PubMed]
- Mason, MJ; Simpson, AK; Mahaut-Smith, MP; Robinson, HP. The interpretation of current-clamp recordings in the cell-attached patch-clamp configuration. Biophys J 2005, 88(1), 739–50. [Google Scholar] [CrossRef]
- McLeod, JF; Leempoels, JM; Peng, SX; Dax, SL; Myers, LJ; Golder, FJ. GAL-021, a new intravenous BKCa-channel blocker, is well tolerated and stimulates ventilation in healthy volunteers. Br J Anaesth 2014, 113(5), 875–83. [Google Scholar] [CrossRef] [PubMed]
- Montague, T; Drummond, J; Ng, K; Parratt, J. Advancements in multiple sclerosis. Intern Med J 2025, 55(6), 895–904. [Google Scholar] [CrossRef]
- Podcast, Neuroverse. The patch-clamp technique explained [Audio podcast episode]. Neuroverse Podcast. Neuroverse Media. 10 October 2025. Available online: http://neuroversepod.com.
- O'Neill, RJ; Tung, L. Cell-attached patch clamp study of the electropermeabilization of amphibian cardiac cells. Biophys J 1991, 59(5), 1028–39. [Google Scholar] [CrossRef]
- Parker, M; Yadavalli, NS; Peranidze, K; Boland, E; Reukov, V; Minko, S. Insulin-producing INS-1 cell cultures on biomimetic 3D scaffolds. J Mater Chem B 2025, 13(38), 12134–12145. [Google Scholar] [CrossRef] [PubMed]
- Perkins, KL. Cell-attached voltage-clamp and current-clamp recording and stimulation techniques in brain slices. J Neurosci Methods 2006, 154(1-2), 1–18. [Google Scholar] [CrossRef]
- Qiu, J; Zhao, Q; Li, R; Liu, Y; Ma, B; Zhao, X. Robotic Fast Patch Clamp in Brain Slices Based on Stepwise Micropipette Navigation and Gigaseal Formation Control. Sensors 2025, 25(4), 1128. [Google Scholar] [CrossRef]
- Robinson, K; Giles, W. A data acquisition, display and plotting program for the IBM PC. Comput Methods Programs Biomed 1986, 23(3), 319–27. [Google Scholar] [CrossRef]
- So, EC; Lo, YC; Chen, LT; Kao, CA; Wu, SN. High effectiveness of triptolide, an active diterpenoid triepoxide, in suppressing Kir-channel currents from human glioma cells. Eur J Pharmacol 2014, 738, 332–41. [Google Scholar] [CrossRef]
- Tsai, KL; Chang, HF; Wu, SN. The inhibition of inwardly rectifying K+ channels by memantine in macrophages and microglial cells. Cell Physiol Biochem 2013, 31(6), 938–51. [Google Scholar] [CrossRef]
- Tyzio, R; Ivanov, A; Bernard, C; Holmes, GL; Ben-Ari, Y; Khazipov, R. Membrane potential of CA3 hippocampal pyramidal cells during postnatal development. J Neurophysiol 2003, 90(5), 2964–72. [Google Scholar] [CrossRef]
- Wang, CL; Tsai, ML; Wu, SN. Evidence for mitoxantrone-induced block of inwardly rectifying K+ channels expressed in the osteoclast precursor RAW 264.7 cells differentiated with lipopolysaccharide. Cell Physiol Biochem 2012, 30(3), 687–701. [Google Scholar] [CrossRef]
- Wang, YJ; Chen, YH; Hwang, E; Yeh, CJ; Liu, YX; Chen, HH; Wu, SN. Functional Expression of NMDA Receptors in SH-SY5Y Neuroblastoma Cells Following Long-Term RA/BDNF-Induced Differentiation. NeuroSci 2025, 6(2), 47. [Google Scholar] [CrossRef] [PubMed]
- Wilson, JR; Clark, RB; Banderali, U; Giles, WR. Measurement of the membrane potential in small cells using patch clamp methods. Channels (Austin) 2011, 5(6), 530–7. [Google Scholar] [CrossRef] [PubMed]
- Wu, AZ; Ohn, TL; Shei, RJ; Wu, HF; Chen, YC; Lee, HC; Dai, DF; Wu, SN. Permissive Modulation of Sphingosine-1-Phosphate-Enhanced Intracellular Calcium on BKCa Channel of Chromaffin Cells. Int J Mol Sci. 2021, 22(4), 2175. [Google Scholar] [CrossRef] [PubMed]
- Wu, SN; Huang, YM; Kao, CA; Chen, BS; Lo, YC. Investigations on contribution of glial inwardly-rectifying K+ current to membrane potential and ion flux: an experimental and theoretical study. Kaohsiung J Med Sci 2015, 31(1), 9–17. [Google Scholar] [CrossRef]
- Wu, SN; Li, HF; Chiang, HT. Characterization of ATP-sensitive potassium channels functionally expressed in pituitary GH3 cells. J Membr Biol 2000, 178(3), 205–14. [Google Scholar] [CrossRef]
- Wu, SN; Liu, SI; Hwang, TL. Activation of muscarinic K+ channels by extracellular ATP and UTP in rat atrial myocytes. J Cardiovasc Pharmacol 1998, 31(2), 203–11. [Google Scholar] [CrossRef]
- Wu, SN; Wu, AZ; Lin, MW. Pharmacological roles of the large-conductance calcium-activated potassium channel. Curr Top Med Chem 2006, 6(10), 1025–30. [Google Scholar] [CrossRef]
- Wu, SN; Wu, AZ; Sung, RJ. Identification of two types of ATP-sensitive K+ channels in rat ventricular myocytes. Life Sci 2007, 80(4), 378–87. [Google Scholar] [CrossRef]
- Wu, SN; Wu, YH; Chen, BS; Lo, YC; Liu, YC. Underlying mechanism of actions of tefluthrin, a pyrethroid insecticide, on voltage-gated ion currents and on action currents in pituitary tumor (GH3) cells and GnRH-secreting (GT1-7) neurons. Toxicology 2009, 258(1), 70–7. [Google Scholar] [CrossRef] [PubMed]
- Wu, SN; Wang, YJ; Gao, ZH; Liutkevičienė, R; Rovite, V. Recent Advances in Ionic Mechanisms in Pituitary Cells: Implications for Electrophysiological and Electropharmacological Research. J Clin Med 2025, 14(9), 3117. [Google Scholar] [CrossRef] [PubMed]
- Zhao, HB; Liu, LM; Lu, X; Quinonez, AT; Roberts, RA; Zhai, TY; Liang, C. Prevention and treatment of noise-induced hearing loss and cochlear synapse degeneration by potassium channel blockers in vivo. Hear Res 2025, 464, 109319. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



