Submitted:
22 December 2025
Posted:
24 December 2025
You are already at the latest version
Abstract
Keywords:
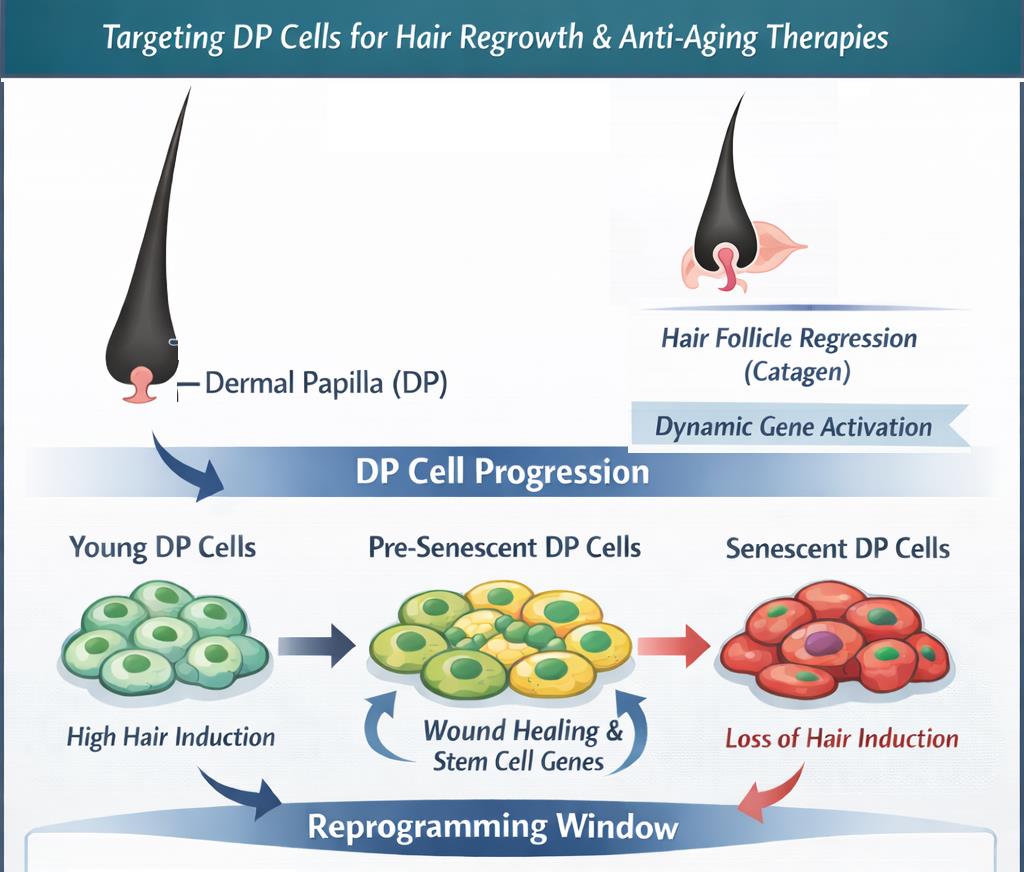
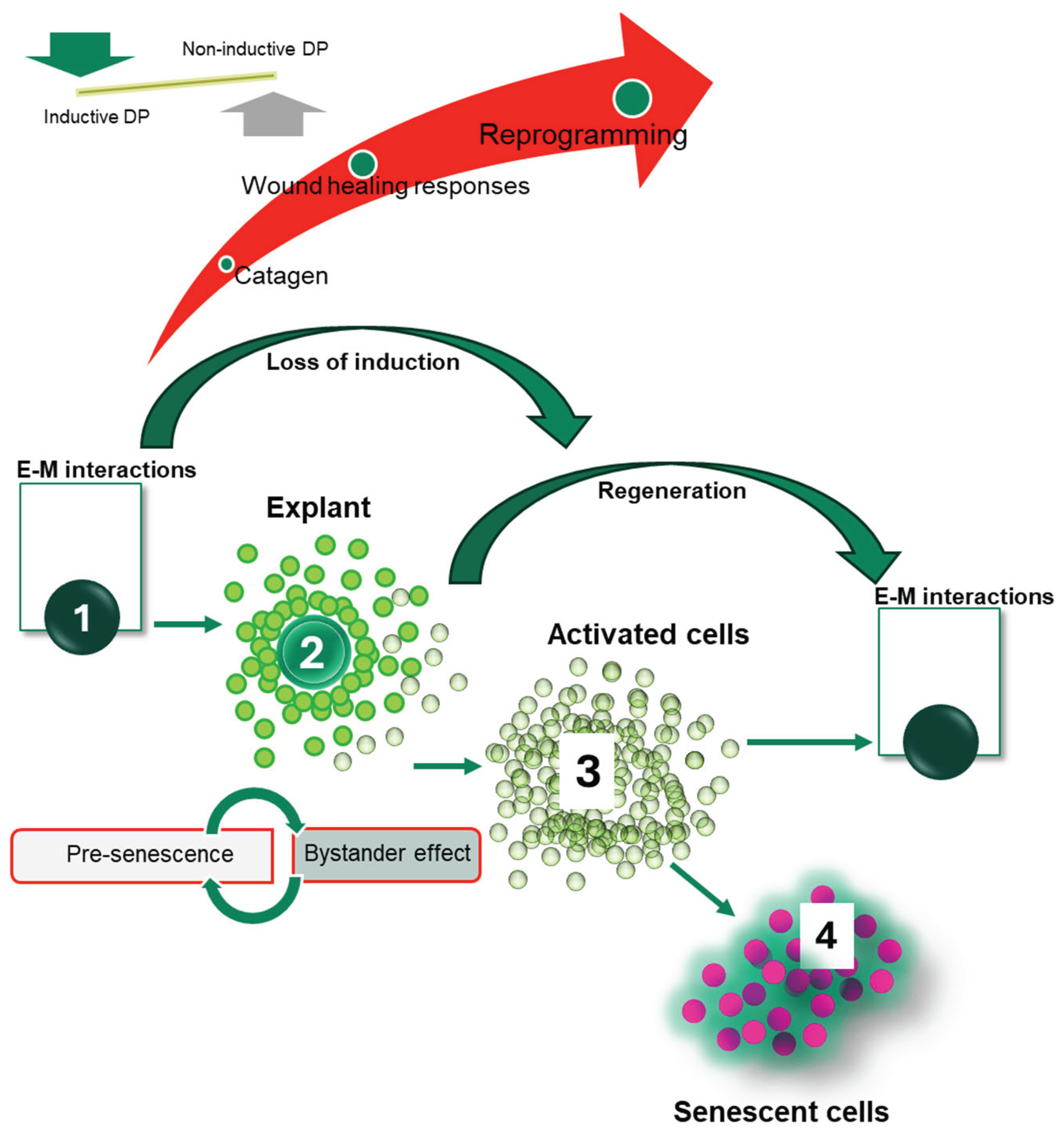
1. Introduction
2. Materials and Methods
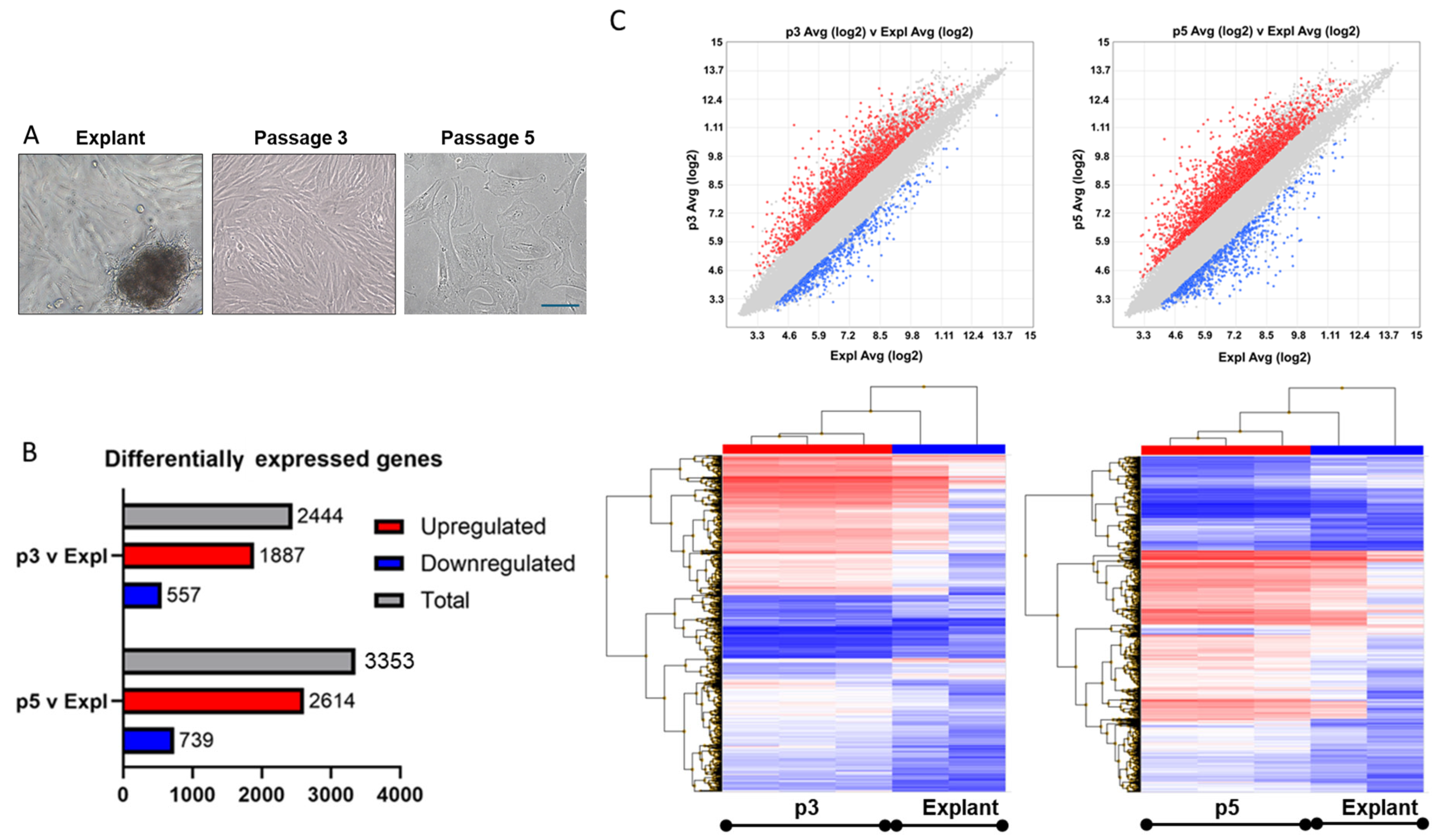
2.1. Dermal Papilla Cultures
2.2. Microarray Analysis
2.3. Transcriptome Analysis Console
2.4. String Gene Clustering Analysis
2.5. Construction of Gene Networks in Cytoscape
2.6. Topology of the Nodes with Variable Significance
2.7. PCR Analysis
3. Results
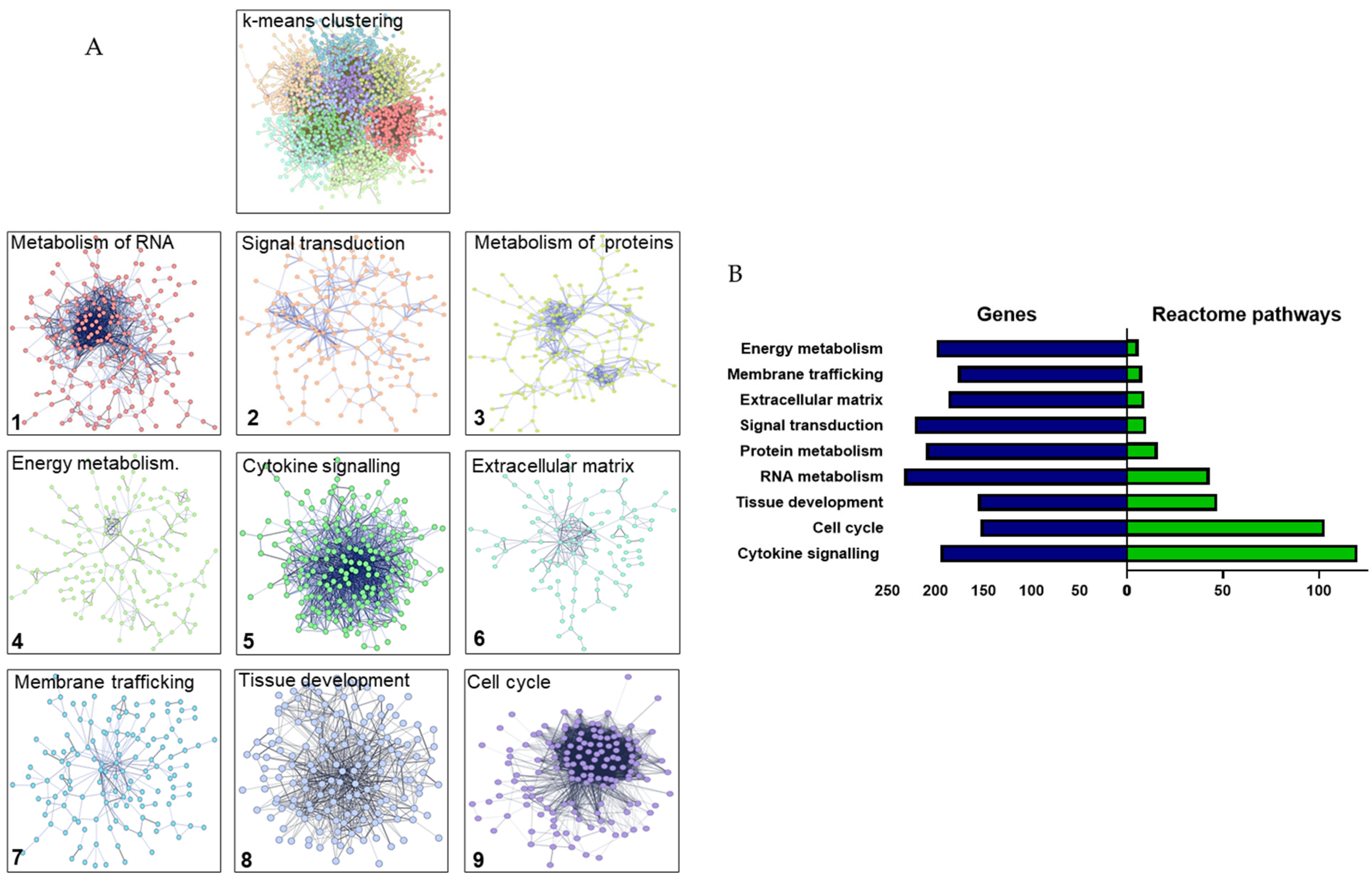
3.1. Construction of the Functional Gene Networks
3.2. Gene Interaction Networks Reveal Discrete Clusters Relevant to Response to Injury and Tissue Regeneration in Cultured DP Cells
3.3. Cultured DP Cells Transiently Express Stem Cell Markers Indicative of Tissue Regenerative Capacity upon Loss of Hair Induction
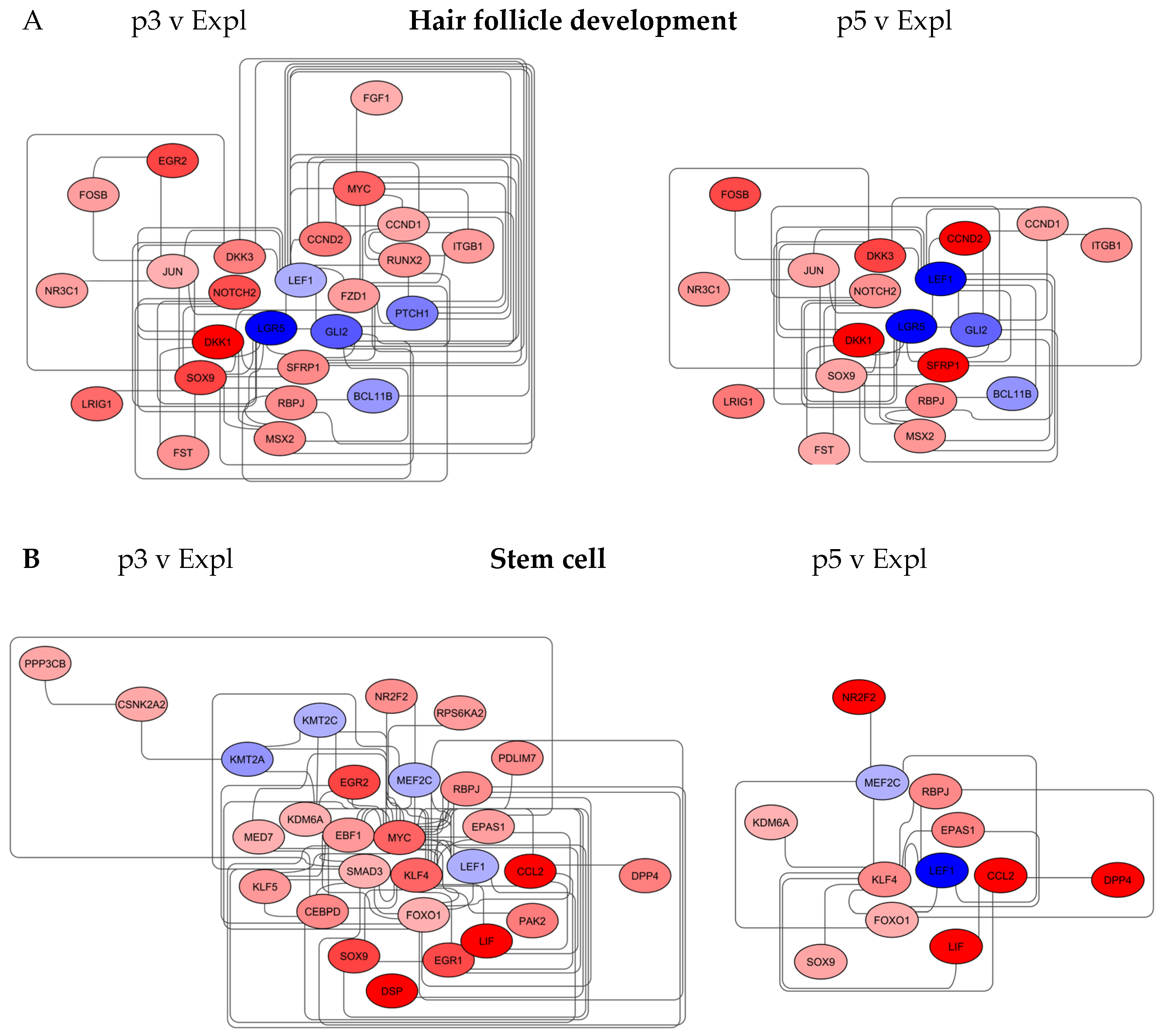
3.3.1. Hair Follicle Development Network
3.3.2. Stem Cell Network
3.4. Expression Patterns of the Inflammation and Senescence Markers Become Uncoupled in Cultured Young and Aged DP Cells
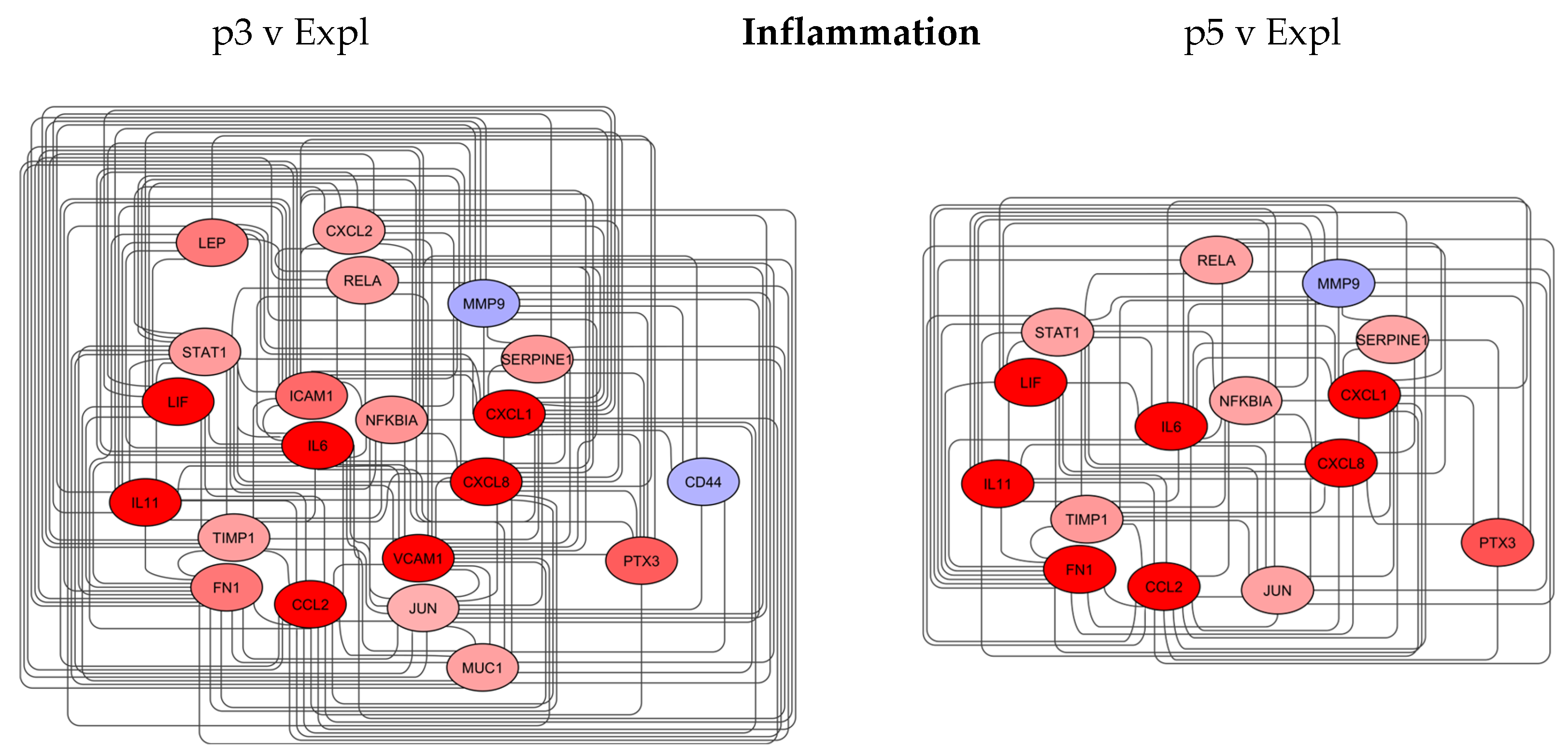
3.4.1. Inflammation Network
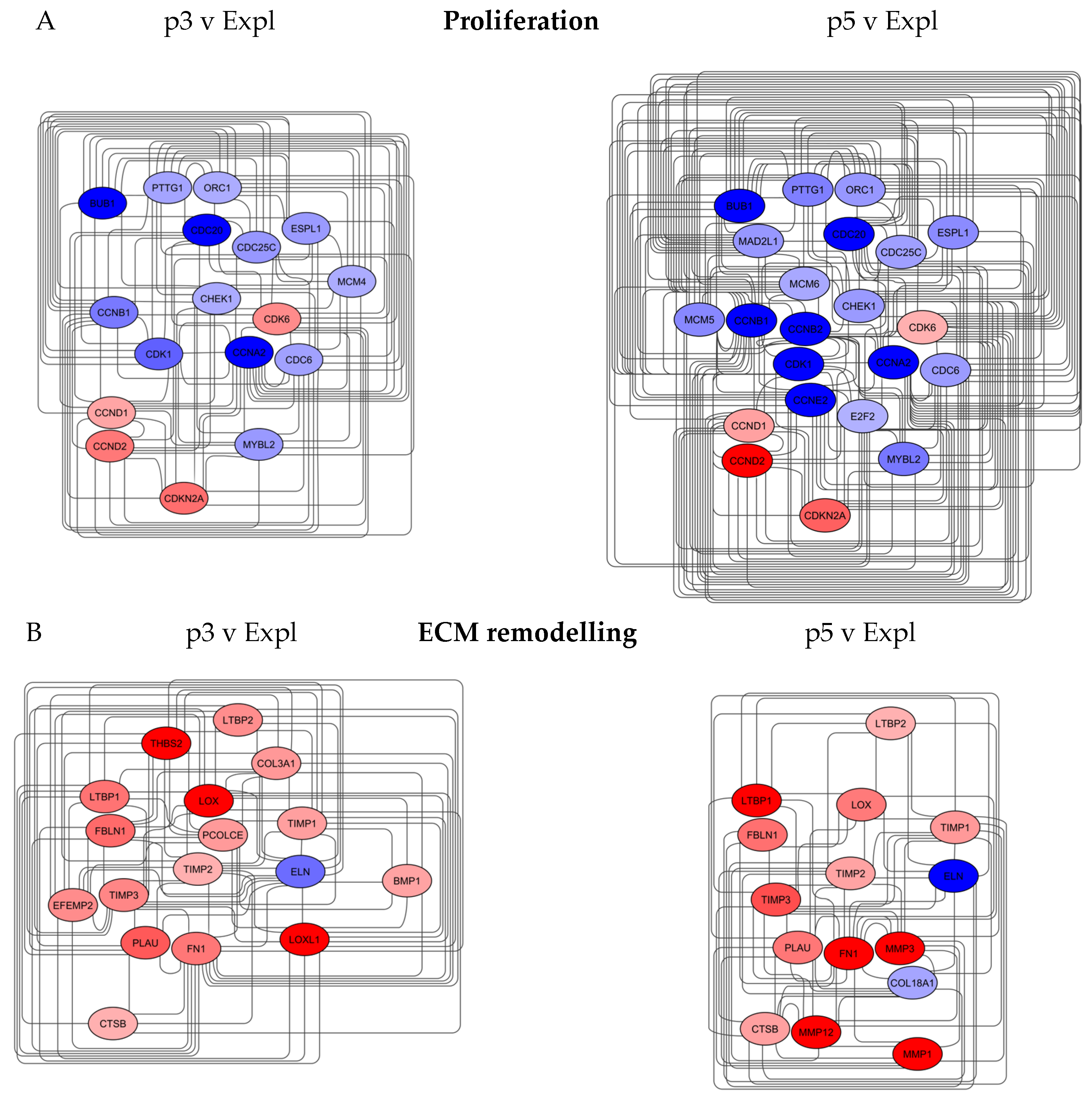
3.4.2. Proliferation and Extracellular Matrix Networks
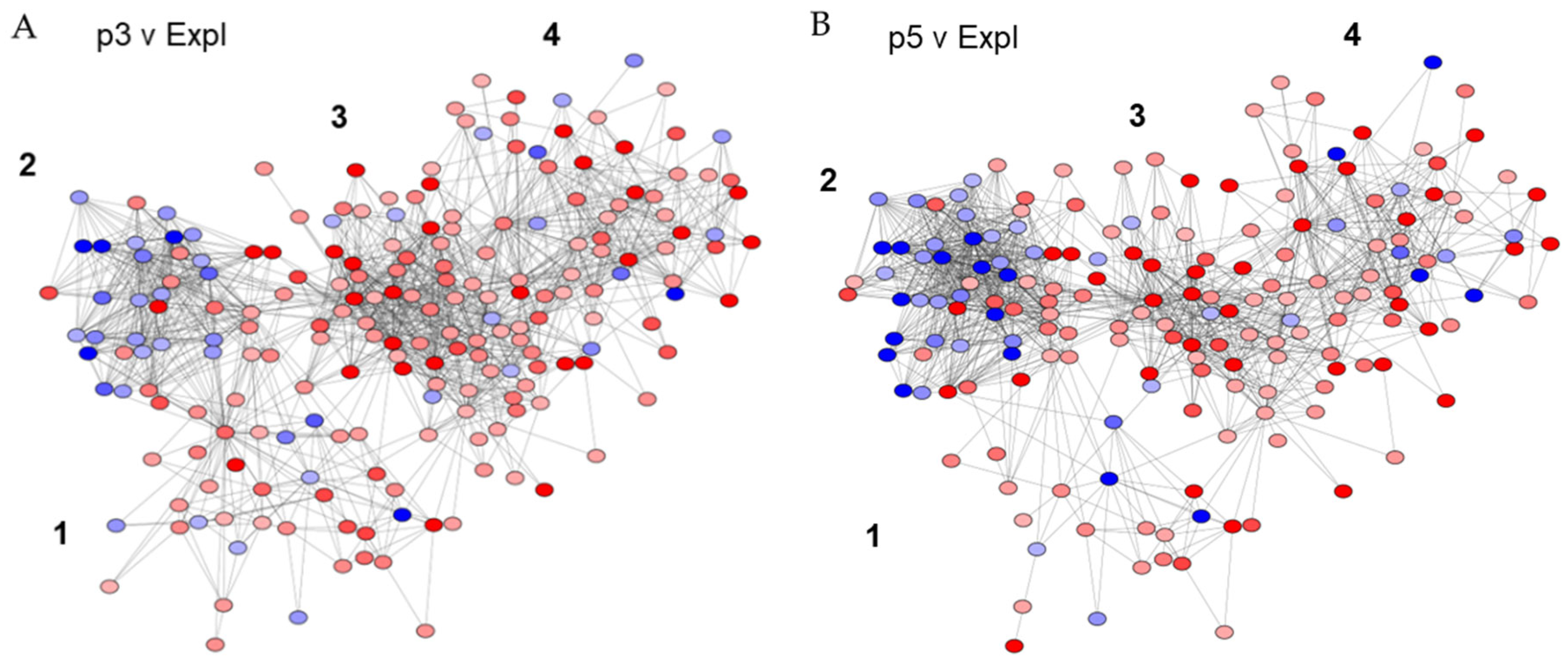
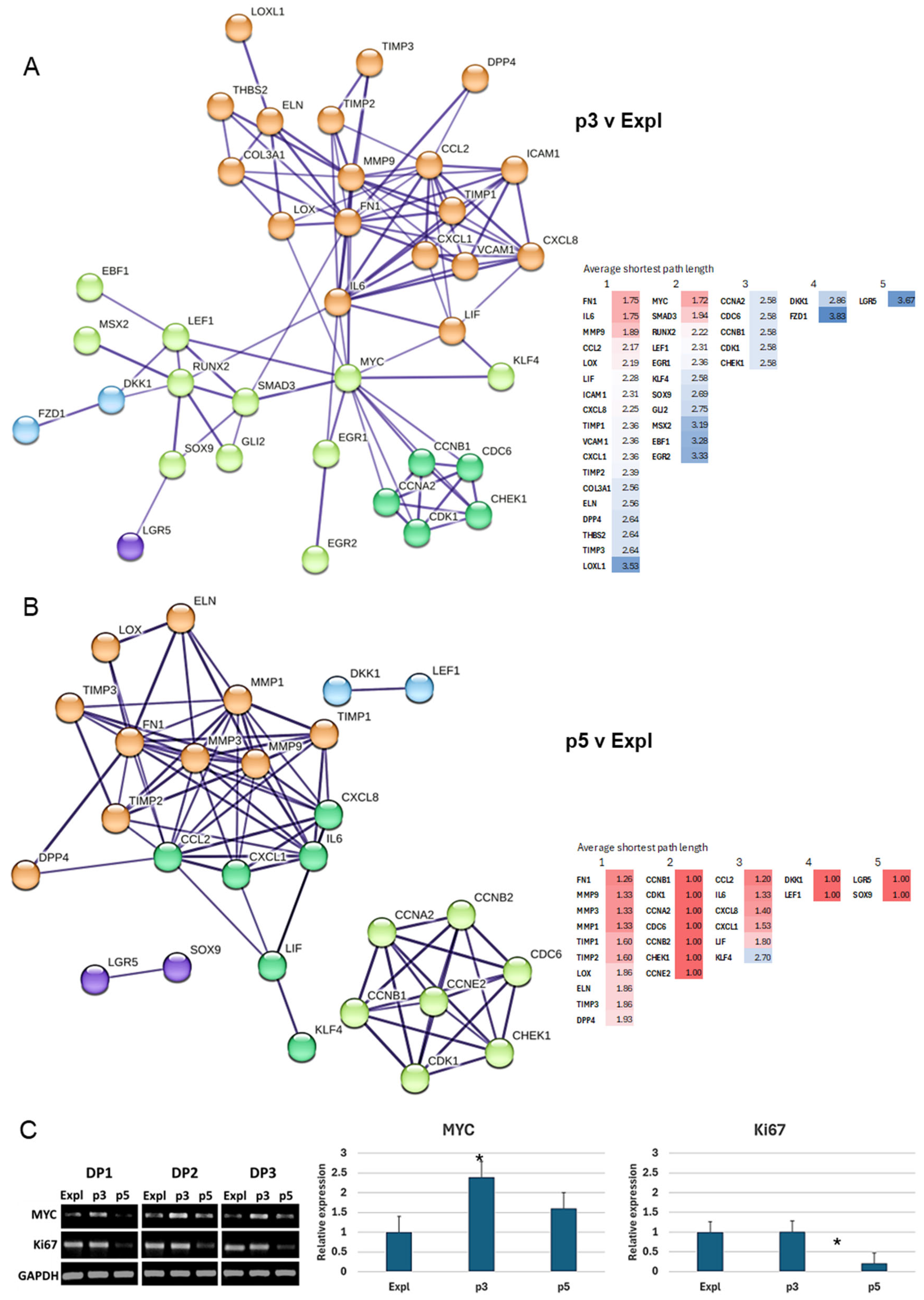
3.5. Network Analysis of the Summative Biomarkers Points to Cell Reprogramming at the Interface Between Inflammation and Senescence
3.5.1. p3 v Expl Network
3.5.2. p5 v Expl Network
3.5.3. Validation of the Most Central Node in the p3 v Expl Network by PCR
4. Discussion
5. Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, M.; Li, Y.; Wang, H.; Meng, L.; et al. Corneal regeneration strategies: From stem cell therapy to tissue engineered stem cell scaffolds. Biomed. Pharmacother. 2023, 165, 115206. [Google Scholar] [CrossRef]
- Grove, J.E.; Bruscia, E.; Krause, D.S. Plasticity of bone marrow-derived stem cells. Stem Cells 2004, 22, 487–500. [Google Scholar] [CrossRef]
- Zeng, C.W. Advancing spinal cord injury treatment through stem cell therapy: A comprehensive review of cell types, challenges, and emerging technologies in regenerative medicine. Int. J. Mol. Sci. 2023, 24, 14349. [Google Scholar] [CrossRef]
- Wang, M.; Yuan, Z.; Ma, N.; Hao, C.; et al. Advances and prospects in stem cells for cartilage regeneration. Stem Cells Int. 2017, 2017, 4130607. [Google Scholar] [CrossRef]
- Wang, B.; Liu, X.M.; Liu, Z.N.; Wang, Y.; et al. Human hair follicle-derived mesenchymal stem cells: Isolation, expansion, and differentiation. World J. Stem Cells 2020, 12, 462–470. [Google Scholar] [CrossRef]
- Ichim, T.E.; O’Heeron, P.; Kesari, S. Fibroblasts as a practical alternative to mesenchymal stem cells. J. Transl. Med. 2018, 16, 212. [Google Scholar] [CrossRef]
- Salzer, M.C.; Lafzi, A.; Berenguer-Llergo, A.; Youssif, C.; et al. Identity noise and adipogenic traits characterize dermal fibroblast aging. Cell 2018, 175, 1575–1590.e22. [Google Scholar] [CrossRef]
- Tai, C.; Xie, Z.; Li, Y.; Feng, Y.; et al. Human skin dermis-derived fibroblasts are functional mesenchymal stromal cells. Cell Biosci. 2022, 12, 105. [Google Scholar] [CrossRef]
- Sowa, Y.; Kishida, T.; Louis, F.; Sawai, S.; et al. Direct conversion of human fibroblasts into adipocytes using a novel small molecular compound. Cells 2021, 10, 605. [Google Scholar] [CrossRef]
- Cayami, F.K.; Claeys, L.; de Ruiter, R.; Smilde, B.J.; et al. Osteogenic transdifferentiation of primary human fibroblasts to osteoblast-like cells with human platelet lysate. Sci. Rep. 2022, 12, 14686. [Google Scholar] [CrossRef]
- Abdel-Raouf, K.M.A.; Rezgui, R.; Stefanini, C.; Teo, J.C.M.; Christoforou, N. Transdifferentiation of human fibroblasts into skeletal muscle cells. Biology 2021, 10, 539. [Google Scholar] [CrossRef]
- Takahashi, K.; Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef]
- Smith, D.K.; He, M.; Zhang, C.L.; Zheng, J.C. Therapeutic potential of cell identity reprogramming for aging-related neurodegenerative disorders. Prog. Neurobiol. 2017, 157, 212–229. [Google Scholar] [CrossRef]
- Gong, L.; Yan, Q.; Zhang, Y.; et al. Cancer cell reprogramming: A promising therapy converting malignancy to benignity. Cancer Commun. 2019, 39, 48. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Kong, L.; Zhu, S. Reprogramming cell fates by small molecules. Protein Cell 2017, 8, 328–348. [Google Scholar] [CrossRef]
- Mahmoudi, S.; Mancini, E.; Xu, L.; Moore, A.; et al. Heterogeneity in old fibroblasts is linked to variability in reprogramming and wound healing. Nature 2019, 574, 553–558. [Google Scholar] [CrossRef]
- Matsuzaki, T.; Yoshizato, K. Role of hair papilla cells in induction and regeneration of hair follicles. Wound Repair Regen. 1998, 6, 524–530. [Google Scholar] [CrossRef]
- Martino, P.A.; Heitman, N.; Rendl, M. The dermal sheath: An emerging component of the hair follicle stem cell niche. Exp. Dermatol. 2021, 30, 512–521. [Google Scholar] [CrossRef]
- Jahoda, C.A. Cell movement in the hair follicle dermis—More than a two-way street? J. Invest. Dermatol. 2003, 121, ix–xi. [Google Scholar] [CrossRef]
- Zhou, S.; Li, Z.; Li, X.; Ye, Y.; et al. Crosstalk between endothelial cells and dermal papilla entails hair regeneration and angiogenesis during aging. J. Adv. Res. 2025, 70, 339–353. [Google Scholar] [CrossRef]
- Leirós, G.J.; Kusinsky, A.G.; Drago, H.; Bossi, S.; et al. Dermal papilla cells improve the wound healing process and generate hair bud-like structures in grafted skin substitutes using hair follicle stem cells. Stem Cells Transl. Med. 2014, 3, 1209–1219. [Google Scholar] [CrossRef]
- Morgan, B.A. The dermal papilla: An instructive niche for epithelial stem and progenitor cells in development and regeneration of the hair follicle. Cold Spring Harb. Perspect. Med. 2014, 4, a015180. [Google Scholar] [CrossRef]
- Kishimoto, J.; Burgeson, R.E.; Morgan, B.A. Wnt signaling maintains the hair-inducing activity of the dermal papilla. Genes Dev. 2000, 14, 1181–1185. [Google Scholar] [CrossRef]
- O’Shaughnessy, R.F.L.; Yeo, W.; Gautier, J.; et al. The WNT signalling modulator, Wise, is expressed in an interaction-dependent manner during hair-follicle cycling. J. Invest. Dermatol. 2004, 123, 613–621. [Google Scholar] [CrossRef]
- Ohyama, M.; Kobayashi, T.; Sasaki, T.; Shimizu, A.; Amagai, M. Restoration of the intrinsic properties of human dermal papilla in vitro. J. Cell Sci. 2012, 125, 4114–4125. [Google Scholar] [CrossRef]
- Abreu, C.M.; Cerqueira, M.T.; Pirraco, R.P.; Gasperini, L.; et al. Rescuing key native traits in cultured dermal papilla cells for human hair regeneration. J. Adv. Res. 2021, 30, 103–112. [Google Scholar] [CrossRef]
- Li, Y.; Lin, C.M.; Cai, X.N.; Li, G.Q. Reconstruction of human hair dermal papilla with microencapsulation in vitro. J. Dermatol. Sci. 2005, 38, 107–109. [Google Scholar] [CrossRef]
- Iyer, V.R.; Eisen, M.B.; Ross, D.T.; Schuler, G.; et al. The transcriptional program in the response of human fibroblasts to serum. Science 1999, 283, 83–87. [Google Scholar] [CrossRef]
- Li, J.; Zhao, B.; Yao, S.; Dai, Y.; et al. Dermal papilla cell-derived exosomes regulate hair follicle stem cell proliferation via LEF1. Int. J. Mol. Sci. 2023, 24, 3961. [Google Scholar] [CrossRef]
- Mill, P.; Mo, R.; Fu, H.; Grachtchouk, M.; et al. Sonic hedgehog-dependent activation of Gli2 is essential for embryonic hair follicle development. Genes Dev. 2003, 17, 282–294. [Google Scholar] [CrossRef]
- Jaks, V.; Barker, N.; Kasper, M.; et al. Lgr5 marks cycling, yet long-lived, hair follicle stem cells. Nat. Genet. 2008, 40, 1291–1299. [Google Scholar] [CrossRef]
- Kwack, M.H.; Kim, M.K.; Kim, J.C.; Sung, Y.K. Dickkopf 1 promotes regression of hair follicles. J. Invest. Dermatol. 2012, 132, 1554–1560. [Google Scholar] [CrossRef]
- Hawkshaw, N.J.; Hardman, J.A.; Haslam, I.S.; Shahmalak, A.; et al. Cyclosporine A suppresses the Wnt inhibitor SFRP1 in the dermal papilla of human scalp hair follicles. PLoS Biol. 2018, 16, e2003705. [Google Scholar] [CrossRef]
- Kadaja, M.; Keyes, B.E.; Lin, M.; Pasolli, H.A.; et al. SOX9 is a stem cell transcriptional regulator of secreted niche signaling factors. Genes Dev. 2014, 28, 328–341. [Google Scholar] [CrossRef]
- Wang, X.; Liu, Y.; He, J.; Wang, J.; et al. Regulation of signaling pathways in hair follicle stem cells. Burns Trauma 2022, 10, tkac022. [Google Scholar] [CrossRef]
- Reddy, S.T.; Andl, T.; Lu, M.M.; Morrisey, E.E.; Millar, S.E. Expression of Frizzled genes in developing and postnatal hair follicles. J. Invest. Dermatol. 2004, 123, 275–282. [Google Scholar] [CrossRef]
- Glotzer, D.J.; Zelzer, E.; Olsen, B.R. Impaired skin and hair follicle development in Runx2-deficient mice. Dev. Biol. 2008, 315, 459–473. [Google Scholar] [CrossRef]
- Merrill, B.J.; Gat, U.; DasGupta, R.; Fuchs, E. Tcf3 and Lef1 regulate lineage differentiation of multipotent stem cells in skin. Genes Dev. 2001, 15, 1688–1705. [Google Scholar] [CrossRef]
- Fujimaki, S.; Seko, D.; Kitajima, Y.; Yoshioka, K.; et al. Notch1 and Notch2 coordinately regulate stem cell function in muscle satellite cells. Stem Cells 2018, 36, 278–285. [Google Scholar] [CrossRef]
- Nabhan, A.N.; Webster, J.D.; Adams, J.J.; Blazer, L.; et al. Targeted alveolar regeneration with Frizzled-specific agonists. Cell 2023, 186, 2995–3012.e15. [Google Scholar] [CrossRef]
- Ma, L.; Liu, J.; Wu, T.; Plikus, M.; et al. Cyclic alopecia in Msx2 mutants. Development 2003, 130, 379–389. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, H.; Liu, C.; Wu, Q.; et al. MSX2 initiates mesenchymal stem cell specification of hPSCs. Stem Cell Rep. 2018, 11, 497–513. [Google Scholar] [CrossRef]
- Wang, N.; Yang, T.; Li, J.; Lei, M.; et al. Expression and role of c-Myc in mouse hair follicle morphogenesis. Acta Histochem. 2012, 114, 199–206. [Google Scholar] [CrossRef]
- Chappell, J.; Dalton, S. Roles for MYC in pluripotency. Cold Spring Harb. Perspect. Med. 2013, 3, a014381. [Google Scholar] [CrossRef]
- Ge, W.; Tan, S.J.; Wang, S.H.; Li, L.; et al. Single-cell transcriptome profiling of embryonic hair follicle development. Theranostics 2020, 10, 7581–7598. [Google Scholar] [CrossRef]
- Wang, S.S.; Huang, H.Y.; Chen, S.Z.; Li, X.; et al. Early growth response 2 plays opposing roles in stem cell commitment. Int. J. Biochem. Cell Biol. 2013, 45, 1825–1832. [Google Scholar] [CrossRef]
- Muchkaeva, I.A.; Dashinimaev, E.B.; Artyuhov, A.S.; et al. Generation of iPS cells from human hair follicle dermal papilla cells. Acta Naturae 2014, 6, 45–53. [Google Scholar] [CrossRef]
- Binder, P.; Wang, S.; Radu, M.; Zin, M.; et al. Pak2 as a therapeutic target for cardioprotective ER stress response. Circ. Res. 2019, 124, 696–711. [Google Scholar] [CrossRef]
- Min, I.M.; Pietramaggiori, G.; Kim, F.S.; Passegué, E.; et al. EGR1 controls proliferation and localization of hematopoietic stem cells. Cell Stem Cell 2008, 2, 380–391. [Google Scholar] [CrossRef]
- Li, S.N.; Wu, J.F. TGF-β/SMAD signaling in adipocyte commitment. Stem Cell Res. Ther. 2020, 11, 41. [Google Scholar] [CrossRef]
- Phan, Q.M.; Salz, L.; Kindl, S.S.; Lopez, J.S.; et al. Lineage commitment of dermal fibroblast progenitors. EMBO J. 2023, 42, e113880. [Google Scholar] [CrossRef]
- Takeda, T.; Yokoyama, Y.; Takahashi, H.; et al. KLF5 regulates CCAT1 via genome structure. Br. J. Cancer 2022, 126, 109–119. [Google Scholar] [CrossRef]
- Whelan, D.S.; Caplice, N.M.; Clover, A.J.P. Mesenchymal stromal cell-derived CCL2 accelerates wound healing. Sci. Rep. 2020, 10, 2642. [Google Scholar] [CrossRef]
- Hirai, H.; Karian, P.; Kikyo, N. Regulation of embryonic stem cell pluripotency by LIF. Biochem. J. 2011, 438, 11–23. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Wang, S.; Cao, X.; Yuan, Z.; et al. Effect of EGR1 on dermal papilla cell proliferation. Genes 2022, 13, 1242. [Google Scholar] [CrossRef] [PubMed]
- Oshimori, N.; Fuchs, E. Paracrine TGF-β signaling in hair follicle stem cell activation. Cell Stem Cell 2012, 10, 63–75. [Google Scholar] [CrossRef]
- Hochfeld, L.M.; Bertolini, M.; Broadley, D.; Botchkareva, N.V.; et al. Interaction of WNT10A and EBF1 in male-pattern baldness. PLoS ONE 2021, 16, e0256846. [Google Scholar] [CrossRef]
- Wu, W.; Zhou, W.; Jiang, J.; Wang, M.; et al. Mechanical stimuli-induced CCL2 restores hair follicle regeneration. Mol. Ther. Nucleic Acids 2023, 32, 94–110. [Google Scholar] [CrossRef]
- Han, H.; Qin, H.; Yang, Y.; et al. Effect of KLF4 overexpression on hair follicle development. Dev. Genes Evol. 2013, 233, 137–145. [Google Scholar] [CrossRef]
- Helm, M.; Schmidt, M.; Del Duca, E.; Liu, Y.; et al. Repurposing DPP4 inhibition for hair follicle regeneration. J. Invest. Dermatol. 2023, 143, 2132–2144.e15. [Google Scholar] [CrossRef]
- Ridiandries, A.; Tan, J.T.M.; Bursill, C.A. Chemokines in wound healing. Int. J. Mol. Sci. 2018, 19, 3217. [Google Scholar] [CrossRef]
- Wood, S.; Jayaraman, V.; Huelsmann, E.J.; Bonish, B.; et al. Pro-inflammatory chemokine CCL2 promotes healing in diabetic wounds. PLoS ONE 2014, 9, e91574. [Google Scholar] [CrossRef]
- Iyer, R.P.; Jung, M.; Lindsey, M.L. MMP-9 signaling following myocardial infarction. Am. J. Physiol. Heart Circ. Physiol. 2016, 311, H190–H198. [Google Scholar] [CrossRef]
- Kechagia, J.; Ezra, D.; Burton, M.; et al. Fibroblast profiling in scarring trachoma identifies IL-6 feedback loops. Sci. Rep. 2016, 6, 28261. [Google Scholar] [CrossRef]
- Bui, T.M.; Wiesolek, H.L.; Sumagin, R. ICAM-1 as a master regulator in inflammation. J. Leukoc. Biol. 2020, 108, 787–799. [Google Scholar] [CrossRef]
- Olivares-Silva, F.; Landaeta, R.; Aránguiz, P.; Bolivar, S.; et al. Heparan sulfate enhances leukocyte adhesion on cardiac fibroblasts. Biochim. Biophys. Acta Mol. Basis Dis. 2018, 1864, 831–842. [Google Scholar] [CrossRef]
- Jian, C.; Feng, Z.X.; Zhang, D.C.; Guo, Y.L.; et al. Dermal white adipose tissue modulates hair follicle aging. Front. Cell Dev. Biol. 2021, 9, 728188. [Google Scholar] [CrossRef]
- Narla, S.; Azzam, M.; Townsend, S.; Vellaichamy, G.; et al. Immune system targets in hidradenitis suppurativa. Br. J. Dermatol. 2021, 184, 1004–1013. [Google Scholar] [CrossRef]
- Hou, C.; Miao, Y.; Wang, X.; Chen, C. Expression of matrix metalloproteinases in the hair cycle. Exp. Ther. Med. 2016, 12, 231–237. [Google Scholar] [CrossRef]
- Kwack, M.H.; Ahn, J.S.; Kim, M.K.; Kim, J.C.; Sung, Y.K. Dihydrotestosterone-inducible IL-6 inhibits hair shaft elongation. J. Invest. Dermatol. 2012, 132, 43–49. [Google Scholar] [CrossRef]
- Müller-Röver, S.; Bulfone-Paus, S.; Handjiski, B.; Welker, P.; et al. ICAM-1 and hair follicle regression. J. Histochem. Cytochem. 2000, 48, 557–568. [Google Scholar] [CrossRef]
- Borlado, L.R.; Méndez, J. CDC6 from DNA replication to oncogenesis. Carcinogenesis 2008, 29, 237–243. [Google Scholar] [CrossRef]
- Lim, S.; Kaldis, P. Cdks, cyclins and CKIs beyond cell cycle regulation. Development 2013, 140, 3079–3093. [Google Scholar] [CrossRef]
- Marthandan, S.; Baumgart, M.; Priebe, S.; Groth, M.; et al. Conserved senescence-associated genes in fibroblasts. PLoS ONE 2016, 11, e0154531. [Google Scholar] [CrossRef]
- Stein, G.H.; Drullinger, L.F.; Soulard, A.; Dulić, V. Roles of p21 and p16 in senescence. Mol. Cell. Biol. 1999, 19, 2109–2117. [Google Scholar] [CrossRef]
- Búa, S.; Sotiropoulou, P.; Sgarlata, C.; Borlado, L.R.; et al. Deregulated Cdc6 affects hair growth cycle. Cell Cycle 2015, 14, 3897–3907. [Google Scholar] [CrossRef]
- Chen, S.J.; Liu, T.; Liu, Y.J.; Dong, B.; et al. CCNA2 polymorphisms and wool density. Genet. Mol. Res. 2011, 10, 3365–3370. [Google Scholar] [CrossRef]
- Choi, Y.K.; Kang, J.; Hyun, J.W.; Koh, Y.S.; et al. Myristoleic acid promotes anagen signaling. Biomol. Ther. 2021, 29, 211–219. [Google Scholar] [CrossRef]
- Gao, G.; Feng, Y.; Yang, X.; Liu, W.; et al. Genes related to hair follicle cycling in cashmere goats. Front. Vet. Sci. 2022, 9, 894380. [Google Scholar] [CrossRef]
- Gimeno-LLuch, I.; Benito-Jardón, M.; Guerrero-Barberà, G.; Burday, N.; Costell, M. Role of the fibronectin synergy site in wound healing. Cells 2022, 11, 2100. [Google Scholar] [CrossRef]
- Baumann, L.; Bernstein, E.F.; Weiss, A.S.; Bates, D.; et al. Clinical relevance of elastin in skin. Aesthet. Surg. J. Open Forum 2021, 3, ojab019. [Google Scholar] [CrossRef]
- Calleja-Agius, J.; Brincat, M.; Borg, M. Skin connective tissue and ageing. Best Pract. Res. Clin. Obstet. Gynaecol. 2013, 27, 727–740. [Google Scholar] [CrossRef]
- Kothapalli, C.R.; Ramamurthi, A. Lysyl oxidase enhances elastin synthesis. J. Tissue Eng. Regen. Med. 2009, 3, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Cai, L.; Xiong, X.; Kong, X.; Xie, J. Lysyl oxidases in tissue repair. Tissue Eng. Regen. Med. 2017, 14, 15–30. [Google Scholar] [CrossRef]
- Bornstein, P.; Kyriakides, T.R.; Yang, Z.; Armstrong, L.C.; Birk, D.E. Thrombospondin 2 modulates collagen fibrillogenesis. J. Invest. Dermatol. Symp. Proc. 2000, 5, 61–66. [Google Scholar] [CrossRef]
- Freitas-Rodríguez, S.; Folgueras, A.R.; López-Otín, C. Matrix metalloproteinases in aging. Biochim. Biophys. Acta Mol. Cell Res. 2017, 1864, 2015–2025. [Google Scholar] [CrossRef]
- Shin, J.W.; Kwon, S.H.; Choi, J.Y.; Na, J.I.; et al. Molecular mechanisms of dermal aging. Int. J. Mol. Sci. 2019, 20, 2126. [Google Scholar] [CrossRef]
- Huang, D.; Ding, H.; Wang, Y.; Cheng, G.; et al. Hair follicle transcriptome analysis in Angora rabbits. Biology 2023, 12, 445. [Google Scholar] [CrossRef]
- Hayashi, K.; Cao, T.; Passmore, H.; Jourdan-Le Saux, C.; et al. Progressive hair loss in rough coat mice. J. Invest. Dermatol. 2004, 123, 864–871. [Google Scholar] [CrossRef]
- Airola, K.; Ahonen, M.; Johansson, N.; Heikkilä, P.; et al. TIMP-3 expression in hair growth cycle. J. Histochem. Cytochem. 1998, 46, 437–447. [Google Scholar] [CrossRef]
- Jahoda, C.A.; Whitehouse, J.; Reynolds, A.J.; Hole, N. Hair follicle dermal cells differentiate into adipogenic and osteogenic lineages. Exp. Dermatol. 2003, 12, 849–859. [Google Scholar] [CrossRef]
- Wang, M.; Wang, H.; Zheng, H. A mini review of node centrality metrics in biological networks. Int. J. Netw. Inf. Dyn. 2022, 1, 99–110. [Google Scholar] [CrossRef]
- da Silva, P.F.L.; Ogrodnik, M.; Kucheryavenko, O.; Glibert, J.; et al. Bystander effect contributes to senescent cell accumulation. Aging Cell 2019, 18, e12848. [Google Scholar] [CrossRef]
- Najafi, M.; Fardid, R.; Hadadi, G.; Fardid, M. Mechanisms of radiation-induced bystander effect. J. Biomed. Phys. Eng. 2014, 4, 163–172. [Google Scholar]
- Mariotti, L.G.; Bertolotti, A.; Ranza, E.; Babini, G.; Ottolenghi, A. Mechanisms underpinning IL-6 release in bystander responses. Int. J. Radiat. Biol. 2012, 88, 751–762. [Google Scholar] [CrossRef]
- Mahmoudi, S.; Mancini, E.; Xu, L.; Moore, A.; et al. Heterogeneity in old fibroblasts and wound healing variability. Nature 2019, 574, 553–558. [Google Scholar] [CrossRef]
- Higgins, C.A.; Chen, J.C.; Cerise, J.E.; Jahoda, C.A.; Christiano, A.M. Microenvironmental reprogramming enables de novo human hair follicle growth. Proc. Natl. Acad. Sci. USA 2013, 110, 19679–19688. [Google Scholar] [CrossRef] [PubMed]
| Gene symbol | Gene name | UniProt |
|---|---|---|
| LEF1 | Lymphoid enhancer-binding factor 1 | Q9UJU2 |
| GLI2 | Glioma-associated oncogene family zinc finger 2 | P10070 |
| LGR5 | Leucine-rich repeat-containing G protein-coupled receptor 5) | O75473 |
| DKK1 | Dickkopf WNT signalling pathway inhibitor 1 | O94907 |
| SFRP1 | Secreted frizzled-related protein 1 | Q8N474 |
| SOX9 | SRY-Box transcription factor 9 | P48436 |
| NOTCH2 | Notch receptor 2 | Q04721 |
| FZD1 | Frizzled class receptor 1 | Q9UP38 |
| RUNX1 | Runt-related transcription factor 2 | Q13950 |
| MSX2 | Msh homeobox 2, transcription factor) | P35548 |
| MYC | Myelocytomatosis oncogene, transcription factor) | P01106 |
| EGR2 | Early growth response 2, transcription regulatory factor | P11151 |
| KLF4 | Kruppel-like factor 4, transcription factor) | O43474 |
| PAK2 | p21-activated kinase 2 | Q13177 |
| EGR1 | Early growth response 1 | P18146 |
| SMAD3 | Mothers against decapentaplegic homolog 3 | P84022 |
| EBF1 | Early B-cell factor 1 | Q9UH73 |
| KLF5 | Kruppel-like factor 5, transcription factor | Q13887 |
| CCL2 | C-C motif chemokine ligand 2 | P13500 |
| LIF | Leukaemia inhibitory factor, Interleukin 6-class cytokine | P15018 |
| DPP4 | Dipeptyl peptidase 4, T-cell activation antigen CD26 | P27487 |
| Gene symbol | Stem cell markers | |
| LEF1 | Hair follicle development, the anagen phase of the hair follicle cycle [29] | |
| LGR5 | Marker of stem cells, crucial for hair follicle morphogenesis and maintenance[31] | |
| SOX9 | Maintenance of hair follicle stem cells and growth [34] | |
| NOTCH2 | Maintenance of hair follicle structure and morphogenesis [35] | |
| FZD1 | Maintenance of hair follicle structure and morphogenesis [36] | |
| MSX2 | Hair follicle morphogenesis [41] | |
| MYC | Functions in hair follicle development [43] | |
| EGR2 | Dermal cell fate during hair follicle development [45] | |
| LIF | Reduces hair follicle development and growth [54] | |
| EGR1 | Proliferation of dermal papilla cells and control of hair follicle development [55] | |
| SMAD3 | Promotes transition from telogen and anagen and hair follicle regeneration [56] | |
| EBF1 | Supports the anagen phase of the hair follicle cycle [57] | |
| CCL2 | Immune regulation and enhancement of hair follicle regeneration [58] | |
| KLF4 | Inhibition of hair follicle regeneration, arrest in telogen and early anagen [59] | |
| DPP4 | Pro-fibrotic factor linked to hair follicle growth arrest and alopecia [60] | |
| Gene symbol | Inflammation markers | |
| CXCL1 | Inhibition of the growth and maintenance of the hair follicle [67] | |
| CXCL8 | Inhibition of the growth and maintenance of the hair follicle [68] | |
| MMP9 | A possible role in ECM remodelling during the anagen phase [69] | |
| IL6 | Inhibition of hair growth and promotion of hair follicle regression [70] | |
| ICAM1 | A role in late anagen and catagen of hair follicle cycle, hair follicle regression [71] | |
| Gene symbol | Proliferation and senescence markers | |
| CDC6 | Role in prolongation of the telogen phase of hair follicle cycle [76] | |
| CCNA2 | Positive role in the development of hair follicles and hair density [77] | |
| CCNB1 | May be associated with the promotion of anagen signalling [78] | |
| CCNB2 | Increased expression in the anagen phase and downregulated in resting phase[79] | |
| CDK1 | Increased expression in the anagen phase and downregulated in resting phase[79] | |
| Symbol | Extracellular matrix markers | |
| FN1 | Likely co-operation with other genes to control the hair thickness [88] | |
| LOXL | Hair follicle regeneration linked to the expression of elastin and collagen[89] | |
| MMP2 | Expression and activity increased in anagen and decreased in resting phase [69] | |
| TIMP1 | Expression and activity decreased in anagen [69] | |
| TIMP2 | Expression and activity decreased in anagen [69] | |
| TIMP3 | Expression increased at the beginning of anagen and decreased in catagen [90] | |
| Gene symbol | Gene name | UniProt |
|---|---|---|
| CXCL1 | C-X-C chemokine motif ligand 1 | P09341 |
| CXCL8 | C-X-C chemokine motif ligand 8, Interleukin 8) | P10145 |
| MMP9 | Matrix metalloproteinase 9, collagenase | P14780 |
| IL6 | Interleukin 6 | P05231 |
| ICAM1 | Intercellular adhesion molecule 1 | P05362 |
| VCAM1 | Vascular adhesion molecule 1 | P19320 |
| Gene symbol | Gene name | UniProt |
|---|---|---|
| CDC6 | Cell division cycle 6 | Q99741 |
| CCNA2 | Cyclin A2 | P20248 |
| CCNB1 | Cyclin B1 | P14635 |
| CCNB2 | Cyclin B2 | O95067 |
| CDK1 | Cyclin-dependent kinase 1 | P06493 |
| CCNE2 | Cyclin E2 | O96020 |
| FN1 | Fibronectin 1 | P02751 |
| ELN | Elastin | P15502 |
| COL3A1 | Collagen 3A1 | P02461 |
| LOX | Lysyl oxidase | P28300 |
| LOXL1 | Lysyl oxidase-like gene 1 | Q08397 |
| THBS2 | Thrombospondin 2 | P35442 |
| TIMP1 | Tissue inhibitor of metalloproteinases 1 | P01033 |
| TIMP2 | Tissue inhibitors of metalloproteinases 2 | P16035 |
| TIMP3 | Tissue inhibitors of metalloproteinases 3 | P35625 |
| MMP1 | Matrix metalloproteinase, Interstitial collagenase | P03956 |
| MMP3 | Matrix metalloproteinase, Stromelysin-1 | P08254 |
| MMP12 | Matrix metalloproteinase, Macrophage metalloelastase | P39900 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




