Submitted:
22 December 2025
Posted:
23 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
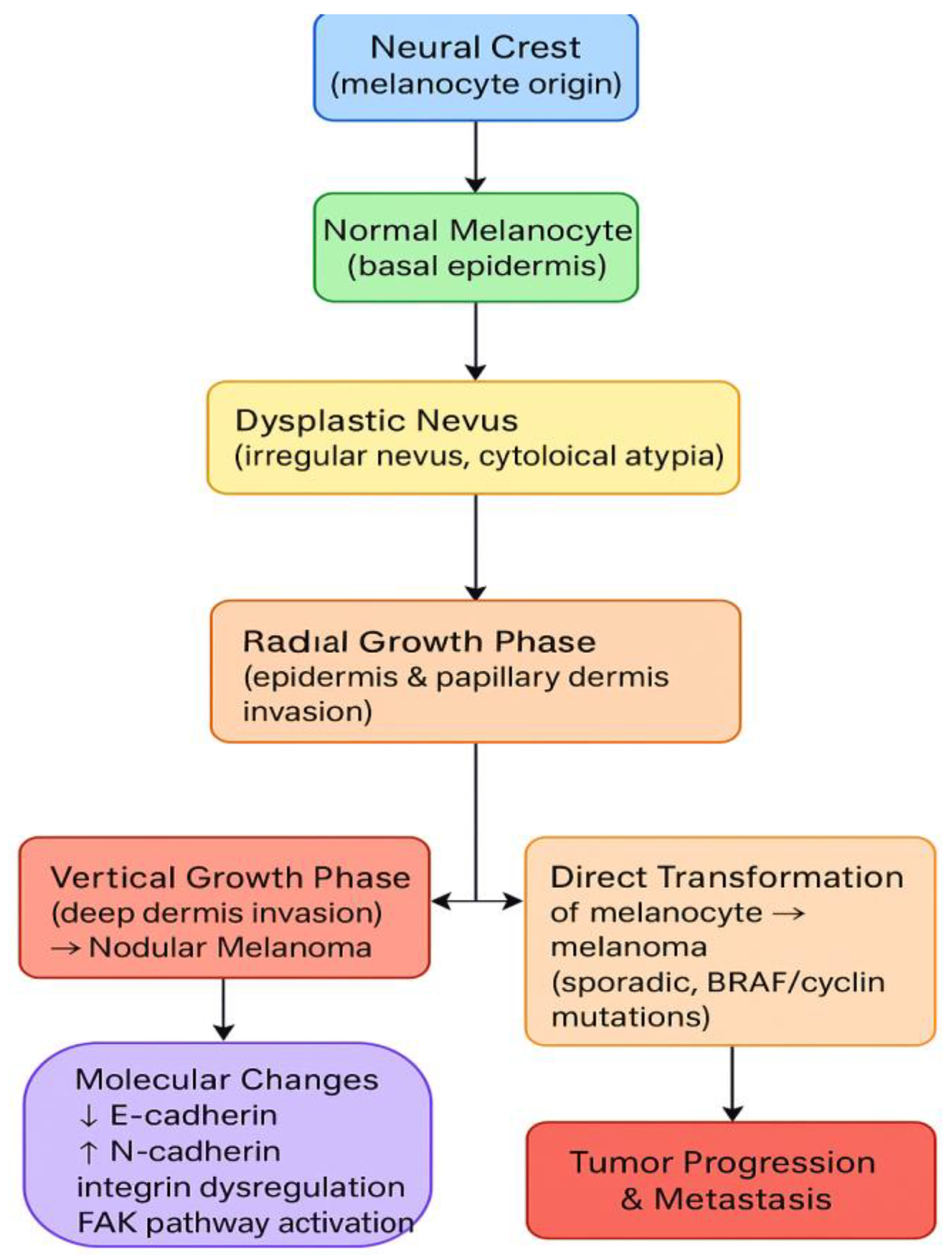
2. Tumor Development
3. Tumor Microenvironment
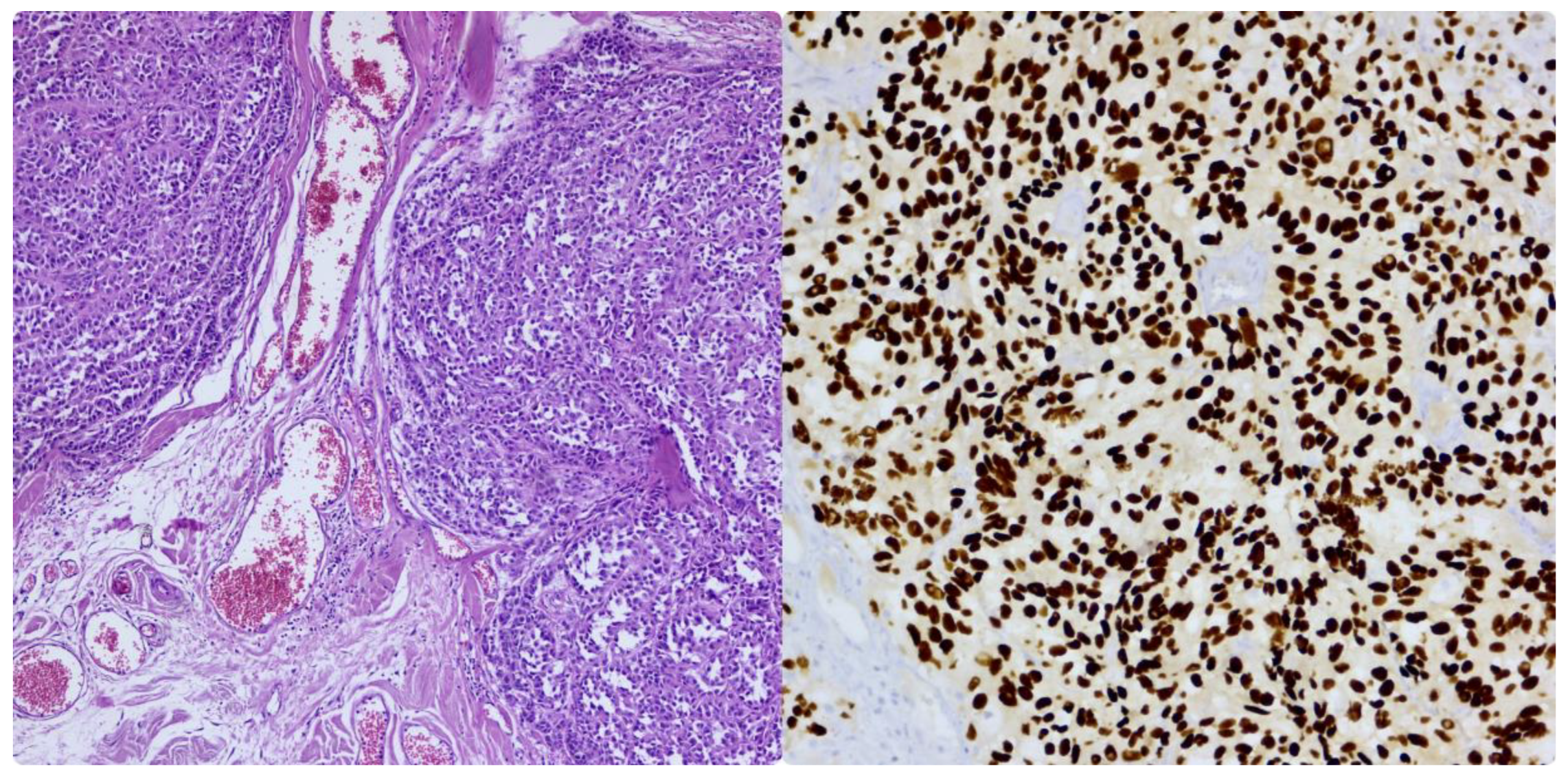
4. Metastatic Melanoma
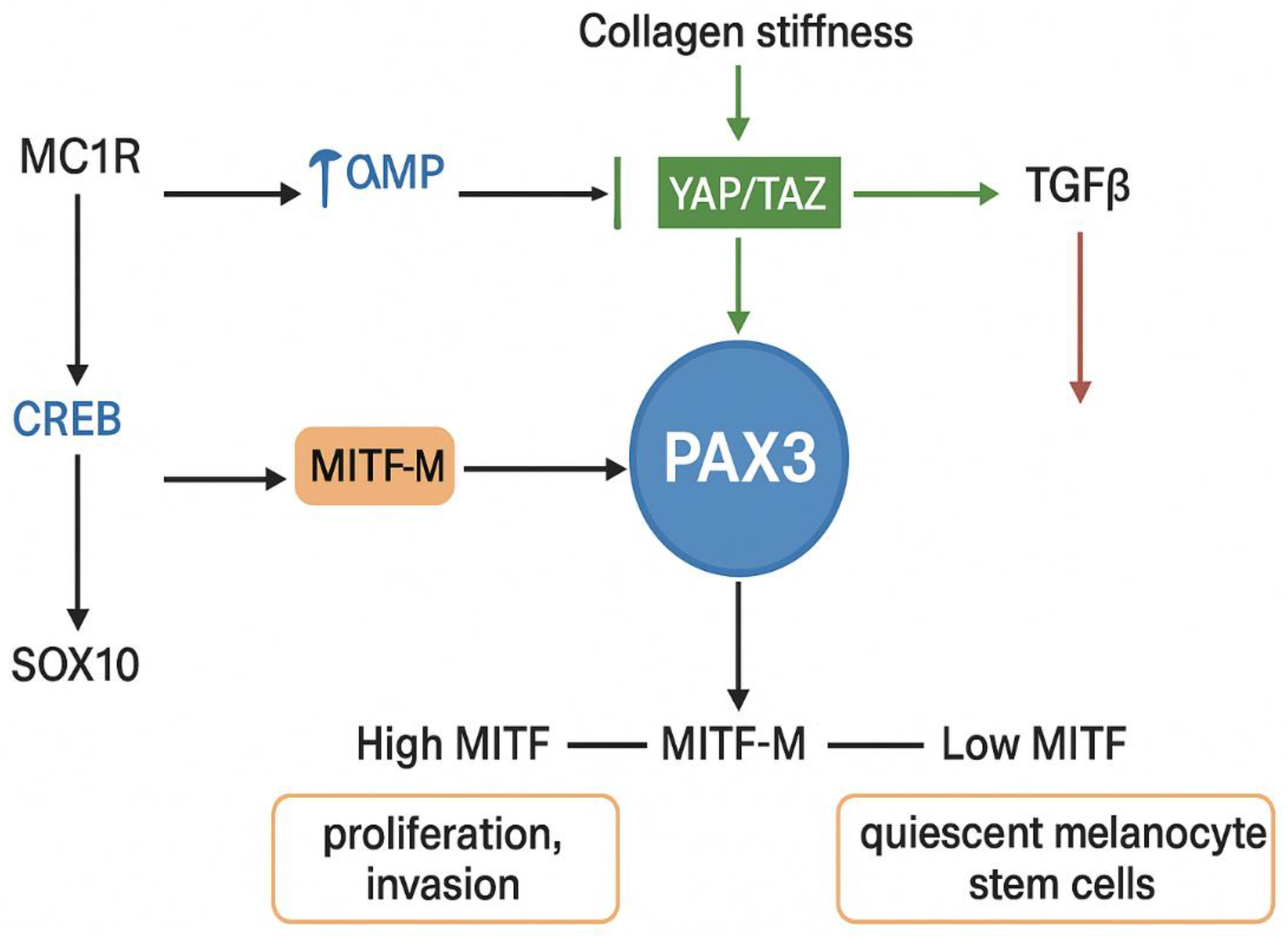
5. Current Therapeutic Options
6. Stem Cells in Melanoma
7. ABCB5
- ABCB5 biology
- Mechanism and roles
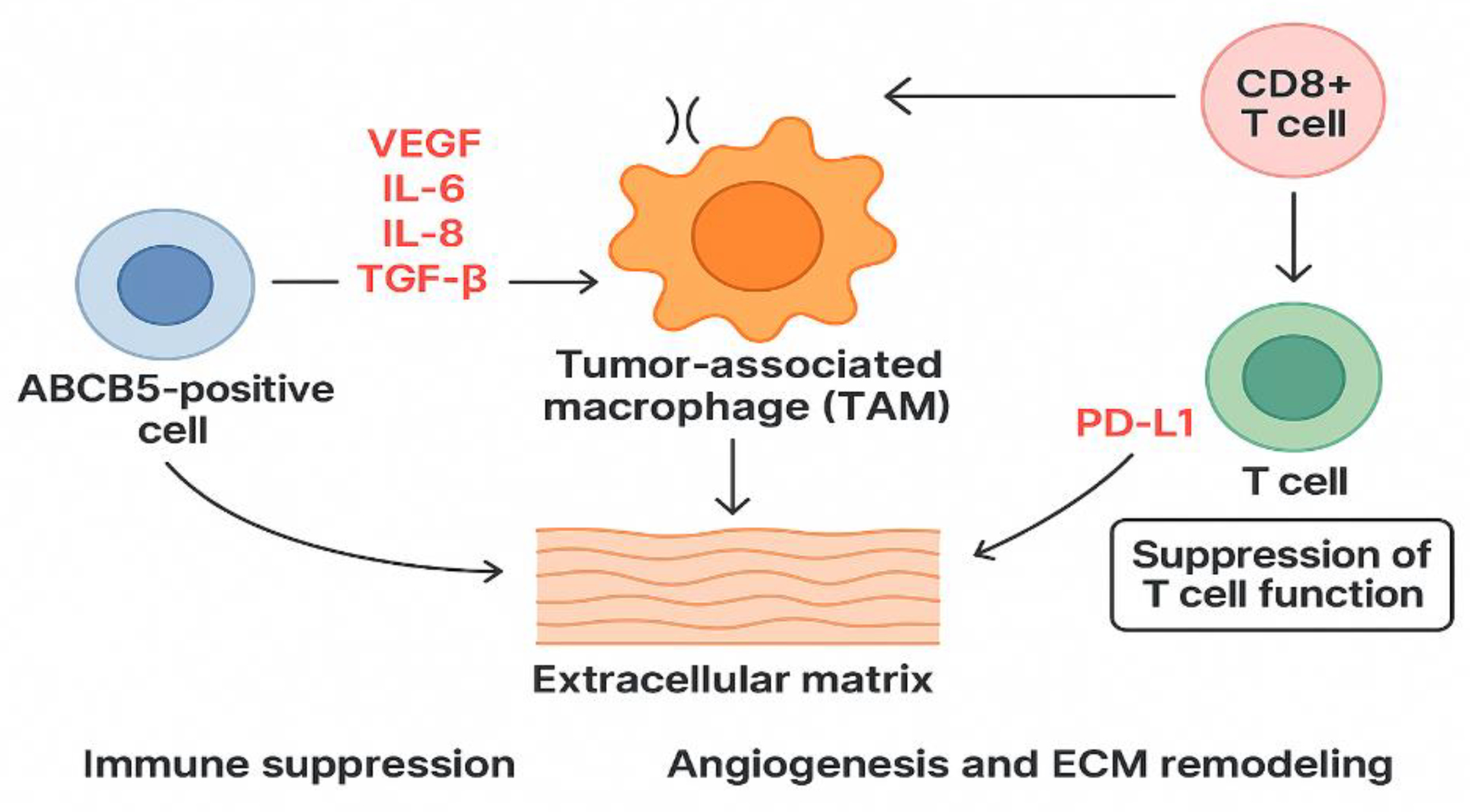
- Clinical significance
| Mechanism | Effect | Clinical Relevance |
| Drug efflux | ↘ intracellular drug concentration | Chemoresistance |
| PI3K/Akt pathway activation | ↗tumor cell survival, ↘ apoptosis | Promotes tumor progression |
| BCL-2 modulation | Anti-apoptotic shift | Promotes tumor progression |
| miR-145 suppression | ↗ ABCB5 expression | Resistance in advanced melanoma |
| Cytokine secretion | Immunosuppressive microenvironment | Poor prognosis, enhanced angiogenesis |
| PD-L1 upregulation | T-cell inhibition | Immune evasion |
| Association with aggressive phenotype | ↗ recurrence, ↘overall survival | Prognostic biomarker |
| Potential therapeutic target | Monoclonal antibodies, inhibitors | Preclinical studies ongoing |
- Therapeutic strategies
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AJCC | American Joint Committee on Cancer |
| WHO | World Health Organization |
| BRAF | B-Raf proto-oncogene, serine/threonine kinase |
| MSCs | Melanoma stem cells |
References
- WHO Classification of Tumours Editorial Board. Skin Tumours. WHO Classification of Tumours, 5th ed. Vol. 12. Lyon: International Agency for Research on Cancer; 2024. Available from: https://publications.iarc.fr.
- Amin, MB; Greene, FL; Edge, SB; Compton, CC; Gershenwald, JE; Brookland, RK; et al. The Eighth Edition AJCC Cancer Staging Manual: Continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J Clin. 2017, 67(2), 93–99. [Google Scholar] [CrossRef]
- Global Cancer Observatory. Cancer Today. Lyon: International Agency for Research on Cancer; 2024. Available from: https://gco.iarc.fr/en.
- Ascierto, P.A.; Kirkwood, J.M.; Grob, J.J.; Simeone, E.; Grimaldi, A.M.; Maio, M.; Palmieri, G.; Testori, A.; Marincola, F.M.; Mozzillo, N. The role of BRAF V600 mutation in melanoma. J. Transl. Med. 2012, 10, 85. [Google Scholar] [CrossRef] [PubMed]
- Wong, P.F.; Wei, W.; Smithy, J.W.; Acs, B.; Toki, M.I.; Blenman, K.R.M.; Zelterman, D.; Kluger, H.M.; Rimm, D.L. Multiplex Quantitative Analysis of Tumor-Infiltrating Lymphocytes and Immunotherapy Outcome in Metastatic Melanoma. Clin. Cancer Res. 2019, 25, 2442–2449. [Google Scholar] [CrossRef] [PubMed]
- Villanueva, J; Herlyn, M. Melanoma and the tumor microenvironment. Curr Oncol Rep. 2008, 10(5), 439–446. [Google Scholar] [CrossRef]
- Falcone, I.; Conciatori, F.; Bazzichetto, C.; Ferretti, G.; Cognetti, F.; Ciuffreda, L.; Milella, M. Tumor Microenvironment: Implications in Melanoma Resistance to Targeted Therapy and Immunotherapy. Cancers 2020, 12, 2870. [Google Scholar] [CrossRef]
- Billottet, C; Jouanneau, J. La relation tumeur-stroma [Tumor-stroma interactions]. Bull Cancer 2008, 95(1), 51–56. [Google Scholar] [CrossRef]
- Labrousse, AL; Ntayi, C; Hornebeck, W; Bernard, P. Stromal reaction in cutaneous melanoma. Crit Rev Oncol Hematol. 2004, 49(3), 269–275. [Google Scholar] [CrossRef]
- Mazurkiewicz, J; Simiczyjew, A; Dratkiewicz, E; Ziętek, M; Matkowski, R; Nowak, D. Stromal Cells Present in the Melanoma Niche Affect Tumor Invasiveness and Its Resistance to Therapy. Int J Mol Sci. 2021, 22(2), 529. [Google Scholar] [CrossRef]
- van der Valk, M.J.M.; Marijnen, C.A.M.; van Etten, B.; Dijkstra, E.A.; Hilling, D.E.; Kranenbarg, E.M.; Putter, H.; Roodvoets, A.G.H.; Bahadoer, R.R.; Fokstuen, T.; et al. Compliance and tolerability of short-course radiotherapy followed by preoperative chemotherapy and surgery for high-risk rectal cancer - Results of the international randomized RAPIDO-trial. Radiother. Oncol. 2020, 147, 75–83. [Google Scholar] [CrossRef]
- Kharouf, N; Flanagan, TW; Hassan, SY; et al. Tumor Microenvironment as a Therapeutic Target in Melanoma Treatment. Cancers (Basel) 2023, 15(12), 3147. [Google Scholar] [CrossRef] [PubMed]
- Sil, H.; Sen, T.; Chatterjee, A. Fibronectin-integrin (alpha5beta1) modulates migration and invasion of murine melanoma cell line B16F10 by involving MMP-9. Oncol. Res. 2011, 19, 335–348. [Google Scholar] [CrossRef] [PubMed]
- Gargalionis, A.N.; Papavassiliou, K.A.; Papavassiliou, A.G. Mechanobiology of solid tumors. Biochim. Biophys. Acta. Mol. Basis Dis. 2022, 1868, 166555. [Google Scholar] [CrossRef] [PubMed]
- Biffi, G.; Tuveson, D.A. Diversity and Biology of Cancer-Associated Fibroblasts. Physiol. Rev. 2021, 101, 147–176. [Google Scholar] [CrossRef]
- Barrett, R.L.; Puré, E. Cancer-associated fibroblasts and their influence on tumor immunity and immunotherapy. Elife 2020, 9. [Google Scholar] [CrossRef]
- Mao, X.; Xu, J.; Wang, W.; Liang, C.; Hua, J.; Liu, J.; Zhang, B.; Meng, Q.; Yu, X.; Shi, S. Crosstalk between cancer-associated fibroblasts and immune cells in the tumor microenvironment: New findings and future perspectives. Mol. Cancer 2021, 20, 131. [Google Scholar] [CrossRef]
- Hinshaw, D.C.; Shevde, L.A. The Tumor Microenvironment Innately Modulates Cancer Progression. Cancer Res. 2019, 79, 4557–4566. [Google Scholar] [CrossRef]
- Hugdahl, E; Aziz, S; Klingen, TA; Akslen, LA. Prognostic value of immune biomarkers in melanoma loco-regional metastases. PLoS One 2025, 20(1), e0315284. [Google Scholar] [CrossRef]
- McDermott, DF; Atkins, MB. PD-1 as a potential target in cancer therapy. Cancer Med. 2013, 2(5), 662–73. [Google Scholar] [CrossRef] [PubMed Central]
- Hartman, ML; Czyz, M. MITF in melanoma: mechanisms behind its expression and activity. Cell Mol Life Sci. 2015, 72(7), 1249–1260. [Google Scholar] [CrossRef]
- Roider, E.; Lakatos, A.I.T.; McConnell, A.M.; et al. MITF regulates IDH1, NNT, and a transcriptional program protecting melanoma from reactive oxygen species. Sci Rep 2024, 14, 21527. [Google Scholar] [CrossRef]
- Rosenbaum, SR; Tiago, M; Caksa, S; et al. SOX10 requirement for melanoma tumor growth is due, in part, to immune-mediated effects. Cell Rep. 2021, 37(10), 110085. [Google Scholar] [CrossRef] [PubMed]
- Lang, D.; Lu, M.; Huang, L.; et al. Pax3 functions at a nodal point in melanocyte stem cell differentiation. Nature 2005, 433, 884–887. [Google Scholar] [CrossRef]
- Medic, S; Ziman, M. PAX3 expression in normal skin melanocytes and melanocytic lesions (naevi and melanomas). PLoS One 2010, 5(4), e9977. [Google Scholar] [CrossRef]
- Moustakas, A. TGF-beta targets PAX3 to control melanocyte differentiation. Dev Cell. 2008, 15(6), 797–799. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y; Cui, S; Li, W; Zhao, Y; Yan, X; Xu, J. PAX3 is a biomarker and prognostic factor in melanoma: Database mining. Oncol Lett. 2019, 17(6), 4985–4993. [Google Scholar] [CrossRef]
- Bonvin, E; Falletta, P; Shaw, H; Delmas, V; Goding, CR. A phosphatidylinositol 3-kinase-Pax3 axis regulates Brn-2 expression in melanoma. Mol Cell Biol. 2012, 32(22), 4674–4683. [Google Scholar] [CrossRef]
- Meng, Z; Moroishi, T; Guan, KL. Mechanisms of Hippo pathway regulation. Genes Dev. 2016, 30(1), 1–17. [Google Scholar] [CrossRef] [PubMed]
- Rastrelli, M; Tropea, S; Rossi, CR; Alaibac, M. Melanoma: epidemiology, risk factors, pathogenesis, diagnosis and classification. In Vivo 2014, 28(6), 1005–1011. [Google Scholar]
- Miskolczi, Z; Smith, MP; Rowling, EJ; Ferguson, J; Barriuso, J; Wellbrock, C. Collagen abundance controls melanoma phenotypes through lineage-specific microenvironment sensing. Oncogene 2018, 37(23), 3166–3182. [Google Scholar] [CrossRef] [PubMed]
- Ryu, HJ; Kim, C; Jang, H; et al. Nuclear Localization of Yes-Associated Protein Is Associated With Tumor Progression in Cutaneous Melanoma. Lab Invest. 2024, 104(5), 102048. [Google Scholar] [CrossRef]
- Lee, JH; Fisher, DE. Melanocyte stem cells as potential therapeutics in skin disorders. Expert Opin Biol Ther. 2014, 14(11), 1569–1579. [Google Scholar] [CrossRef]
- Ostrowski, SM; Fisher, DE. Biology of Melanoma. Hematol Oncol Clin North Am. 2021, 35(1), 29–56. [Google Scholar] [CrossRef]
- Thomas, AJ; Erickson, CA. The making of a melanocyte: the specification of melanoblasts from the neural crest. Pigment Cell Melanoma Res. 2008, 21(6), 598–610. [Google Scholar] [CrossRef] [PubMed]
- Wang, F; Ma, W; Fan, D; Hu, J; An, X; Wang, Z. The biochemistry of melanogenesis: an insight into the function and mechanism of melanogenesis-related proteins. Front Mol Biosci. 2024, 11, 1440187. [Google Scholar] [CrossRef] [PubMed]
- Cornish, D; Holterhues, C; van de Poll-Franse, LV; Coebergh, JW; Nijsten, T. A systematic review of health-related quality of life in cutaneous melanoma. Ann Oncol. 2009, 20 (Suppl 6), vi51–vi58. [Google Scholar] [CrossRef]
- Abel, EV; Basile, KJ; Kugel, CH, 3rd; et al. Melanoma adapts to RAF/MEK inhibitors through FOXD3-mediated upregulation of ERBB3. J Clin Invest. 2013, 123(5), 2155–2168. [Google Scholar] [CrossRef] [PubMed]
- Alver, TN; Lavelle, TJ; Longva, AS; Øy, GF; Hovig, E; Bøe, SL. MITF depletion elevates expression levels of ERBB3 receptor and its cognate ligand NRG1-beta in melanoma. Oncotarget 2016, 7(34), 55128–55140. [Google Scholar] [CrossRef]
- Letsch, A.; Keilholz, U.; Schadendorf, D.; Nagorsen, D.; Schmittel, A.; Thiel, E.; Scheibenbogen, C. High frequencies of circulating melanoma-reactive CD8+ T cells in patients with advanced melanoma. International Journal of Cancer 2000, 87(5), 659–664. [Google Scholar] [CrossRef]
- Westerdahl, J; Ingvar, C; Måsbäck, A; Jonsson, N; Olsson, H. Risk of cutaneous malignant melanoma in relation to use of sunbeds: further evidence for UV-A carcinogenicity. Br J Cancer 2000, 82(9), 1593–1599. [Google Scholar] [CrossRef]
- Cronin, JC; Wunderlich, J; Loftus, SK; et al. Frequent mutations in the MITF pathway in melanoma. Pigment Cell Melanoma Res. 2009, 22(4), 435–444. [Google Scholar] [CrossRef]
- Shakhova, O; Zingg, D; Schaefer, SM; et al. Sox10 promotes the formation and maintenance of giant congenital naevi and melanoma. Nat Cell Biol. 2012, 14(8), 882–890. [Google Scholar] [CrossRef]
- d'Ischia, M; Wakamatsu, K; Napolitano, A; et al. Melanins and melanogenesis: methods, standards, protocols. Pigment Cell Melanoma Res. 2013, 26(5), 616–633. [Google Scholar] [CrossRef]
- Haenssle, HA; Fink, C; Schneiderbauer, R; et al. Man against machine: diagnostic performance of a deep learning convolutional neural network for dermoscopic melanoma recognition in comparison to 58 dermatologists. Ann Oncol. 2018, 29(8), 1836–1842. [Google Scholar] [CrossRef] [PubMed]
- Haq, R; Shoag, J; Andreu-Perez, P; et al. Oncogenic BRAF regulates oxidative metabolism via PGC1α and MITF. Cancer Cell. 2013, 23(3), 302–315. [Google Scholar] [CrossRef] [PubMed]
- Vazquez, F; Lim, JH; Chim, H; et al. PGC1α expression defines a subset of human melanoma tumors with increased mitochondrial capacity and resistance to oxidative stress. Cancer Cell. 2013, 23(3), 287–301. [Google Scholar] [CrossRef] [PubMed]
- Shi, S; Wang, J; Gong, H; et al. PGC-1α-Coordinated Hypothalamic Antioxidant Defense Is Linked to SP1-LanCL1 Axis during High-Fat-Diet-Induced Obesity in Male Mice. Antioxidants (Basel) 2024, 13(2), 252. [Google Scholar] [CrossRef]
- Hossain, SM; Eccles, MR. Phenotype Switching and the Melanoma Microenvironment; Impact on Immunotherapy and Drug Resistance. Int J Mol Sci. 2023, 24(2), 1601. [Google Scholar] [CrossRef]
- Goding, CR; Arnheiter, H. MITF-the first 25 years. Genes Dev. 2019, 33(15-16), 983–1007. [Google Scholar] [CrossRef]
- Clark, WH; Elder, DE; Guerry, D; Braitman, LE; Trock, BJ; Schultz, D; Synnestvedt, M; Halpern, AC. Model predicting survival in stage I melanoma based on tumor progression. J Natl Cancer Inst 1989, 81, 1893–1904. [Google Scholar] [CrossRef]
- Richtig, G; Richtig, E; Massone, C; Hofmann-Wellenhof, R. Analysis of clinical, dermoscopic and histopathological features of primary melanomas of patients with metastatic disease--a retrospective study at the Department of Dermatology, Medical University of Graz, 2000-2010. J Eur Acad Dermatol Venereol 2014, 28(12), 1776–1781. [Google Scholar] [CrossRef]
- Massone, C; Hofman-Wellenhof, R; Chiodi, S; Sola, S. Dermoscopic Criteria, Histopathological Correlates and Genetic Findings of Thin Melanoma on Non-Volar Skin. Genes (Basel) 2021, 12(8), 1288. [Google Scholar] [CrossRef]
- Sundararajan, S; Thida, AM; Yadlapati, S; et al. Metastatic Melanoma. StatPearls [Internet]. 17 Feb 2024. Available online: https://www.ncbi.nlm.nih.gov/books/NBK470358/?utm.
- Lowe, L. Metastatic melanoma and rare melanoma variants: a review. Pathology 2023, 55(2), 236–244. [Google Scholar] [CrossRef] [PubMed]
- Carey, AE; Weeraratna, AT. Entering the TiME machine: How age-related changes in the tumor immune microenvironment impact melanoma progression and therapy response. Pharmacol Ther. 2024, 262, 108698. [Google Scholar] [CrossRef] [PubMed]
- Wick, MR; Gru, AA. Metastatic melanoma: Pathologic characterization, current treatment, and complications of therapy. Semin Diagn Pathol. 2016, 33(4), 204–218. [Google Scholar] [CrossRef]
- Wilson, ML. Histopathologic and Molecular Diagnosis of Melanoma. Clin Plast Surg. 2021, 48(4), 587–598. [Google Scholar] [CrossRef]
- Alexandrov, LB; Nik-Zainal, S; Wedge, DC; et al. Signatures of mutational processes in human cancer. Nature 2013, 500(7463), 415–421. [Google Scholar] [CrossRef]
- Timmons, C; Morris, Q; Harrigan, CF. Regional mutational signature ac-tivities in cancer genomes. PLoS Comput Biol. 2022, 18(12), e1010733. [Google Scholar] [CrossRef]
- Fröhlich, F; Ramelyte, E; Turko, P; et al. Clock-like Mutation Signature May Be Prognostic for Worse Survival Than Signatures of UV Damage in Cutaneous Melanoma. Cancers (Basel) 2023, 15(15), 3818. [Google Scholar] [CrossRef]
- Maul, LV; Ramelyte, E; Dummer, R; Mangana, J. Management of metastatic melanoma with combinations including PD-1 inhibitors. Expert Opin Biol Ther. 2025, 25(5), 1–12. [Google Scholar] [CrossRef]
- Tinca, AC; Raicea, A; Szőke, AR; et al. Morphological aspects and therapeutic options in melanoma: a narrative review of the past decade. Rom J Morphol Embryol 2023, 64(2), 135–141. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D. B. Updates to the management of cutaneous melanoma. JNCCN: Journal of the National Comprehensive Cancer Network 2024, 22, e245015. [Google Scholar] [CrossRef]
- Lodde, G; Albrecht, LJ; Schadendorf, D. Behandlung des metastasierten Melanoms – Update 2025 [Treatment of metastatic melanoma: update 2025]. Dtsch Med Wochenschr 2025, 150(10), 562–569. [Google Scholar] [CrossRef]
- Lu, R; Zhang, X; Li, X; Wan, X. Circ_0016418 promotes melanoma development and glutamine catabolism by regulating the miR-605-5p/GLS axis. Int J Clin Exp Pathol. 2020, 13(7), 1791–1801. [Google Scholar]
- Castellani, G; Buccarelli, M; Arasi, MB; et al. BRAF Mutations in Melanoma: Biological Aspects, Therapeutic Implications, and Circulating Biomarkers. Cancers (Basel) 2023, 15(16), 4026. [Google Scholar] [CrossRef] [PubMed]
- Khaliq, M.; Fallahi-Sichani, M. Epigenetic Mechanisms of Escape from BRAF Oncogene Dependency. Cancers 2019, 11, 1480. [Google Scholar] [CrossRef] [PubMed]
- Solit, D.; Rosen, N. Oncogenic RAF: A brief history of time. Pigment Cell Melanoma Res. 2010, 23, 760–762. [Google Scholar] [CrossRef] [PubMed]
- Menzer, C; Hassel, JC. Targeted Therapy for Melanomas Without BRAF V600 Mutations. Curr Treat Options Oncol. 2022, 23(6), 831–842. [Google Scholar] [CrossRef]
- Knight, A; Karapetyan, L; Kirkwood, JM. Immunotherapy in Melanoma: Recent Advances and Future Directions. Cancers (Basel) 2023, 15(4), 1106. [Google Scholar] [CrossRef]
- Sabău, A.-H.; Tinca, A.-C.; Niculescu, R.; Cocuz, I. G.; Cozac-Szöke, A. R.; Lazar, B. A.; Chiorean, D. M.; Budin, C. E.; Cotoi, O. S. Cancer Stem Cells in Melanoma: Drivers of Tumor Plasticity and Emerging Therapeutic Strategies. International Journal of Molecular Sciences 2025, 26(15), 7419. [Google Scholar] [CrossRef]
- Mu, X; Zhou, Y; Yu, Y; Zhang, M; Liu, J. The roles of cancer stem cells and therapeutic implications in melanoma. Front Immunol 2024, 15, 1486680. [Google Scholar] [CrossRef]
- Gerard, L; Gillet, JP. The uniqueness of ABCB5 as a full transporter ABCB5FL and a half-transporter-like ABCB5β. Cancer Drug Resist 2024, 7, 29. [Google Scholar] [CrossRef]
- He, Z; Starkuviene, V; Keese, M. The Differentiation and Regeneration Potential of ABCB5+ Mesenchymal Stem Cells: A Review and Clinical Perspectives. J Clin Med. 2025, 14(3), 660. [Google Scholar] [CrossRef]
- Chartrain, M; Riond, J; Stennevin, A; et al. Melanoma chemotherapy leads to the selection of ABCB5-expressing cells. PLoS One 2012, 7(5), e36762. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y; Ding, Q; Zhang, M; et al. miR-145 targets ABCB5 to regulate chemosensitivity in melanoma. Oncotarget 2016, 7(35), 57317–57328. [Google Scholar]
- Jilaveanu, LB; Aziz, SA; Kluger, HM. Chemotherapy and biologic therapies for melanoma: do they work? Clin Dermatol 2009, 27, 614–625. [Google Scholar] [CrossRef] [PubMed]
- Frank, NY; Pendse, SS; Lapchak, PH; et al. Regulation of progenitor cell fusion by ABCB5 in human malignant melanoma. Cancer Res 2011, 71(4), 1274–83. [Google Scholar] [PubMed]
- Xiao, J.; Egger, M.E.; McMasters, K.M.; et al. Differential expression of ABCB5 in BRAF inhibitor-resistant melanoma cell lines. BMC Cancer 2018, 18, 675. [Google Scholar] [CrossRef]
- Straussman, R; et al. Tumour micro-environment elicits innate resistance to RAF inhibitors through HGF secretion. Nature 2012, 487(7408), 500–4. [Google Scholar] [CrossRef]
- Perna, D; Karreth, FA; Rust, AG; Perez-Mancera, PA; Rashid, M; Iorio, F; Alifrangis, C; Arends, MJ; Bosenberg, MW; Bollag, G; Tuveson, DA; Adams, DJ. BRAF inhibitor resistance mediated by the AKT pathway in an oncogenic BRAF mouse melanoma model. Proc Natl Acad Sci U S A 2015, 112(6), E536–45. [Google Scholar] [CrossRef]
- Mühlbauer, D; Schatton, T; Schütte, U; et al. Expression of ABCB5 as a molecular marker for poor prognosis in melanoma. Journal of Investigative Dermatology 2016, 136(3), 666–676. [Google Scholar]
- Tu, J; Wang, J; Tang, B; et al. Expression and clinical significance of TYRP1, ABCB5, and MMP17 in sinonasal mucosal melanoma. Cancer Biomark. 2022, 35(3), 331–342. [Google Scholar] [CrossRef]
- Wilson, BJ; Schatton, T; Zhan, Q; et al. ABCB5 identifies a therapy-refractory tumor cell population in colorectal cancer patients. Cancer Res. 2011, 71(15), 5307–5316. [Google Scholar] [CrossRef]
- Kugimiya, N; Nishimoto, A; Hosoyama, T; et al. The c-MYC-ABCB5 axis plays a pivotal role in 5-fluorouracil resistance in human colon cancer cells. J Cell Mol Med. 2015, 19(7), 1569–1581. [Google Scholar] [CrossRef]
- Duvivier, L; Gillet, JP. Deciphering the roles of ABCB5 in normal and cancer cells. Trends Cancer 2022, 8(10), 795–798. [Google Scholar] [CrossRef]
- Rapanotti, MC; Cugini, E; Campione, E; et al. Epithelial-to-Mesenchymal Transition Gene Signature in Circulating Melanoma Cells: Biological and Clinical Relevance. Int J Mol Sci. 2023, 24(14), 11792. [Google Scholar] [CrossRef] [PubMed]
- Kusienicka, A; Bukowska-Strakova, K; Cieśla, M; et al. Heme Oxygenase-1 Has a Greater Effect on Melanoma Stem Cell Properties Than the Expression of Melanoma-Initiating Cell Markers. Int J Mol Sci. 2022, 23(7), 3596. [Google Scholar] [CrossRef] [PubMed]
- Gerard, L; Duvivier, L; Fourrez, M; et al. Identification of two novel heterodimeric ABC transporters in melanoma: ABCB5β/B6 and ABCB5β/B9. J Biol Chem. 2024, 300(2), 105594. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




