Submitted:
18 December 2025
Posted:
19 December 2025
You are already at the latest version
Abstract
Hydrometallurgical pretreatment of pyrite-bearing concentrates and tailings by hydrothermal interaction with Cu(II) solutions is a promising route for chemical beneficiation and mitigation of acid mine drainage but is limited by passivation caused by elemental sulfur and secondary copper sulfides. Here, the effect of sodium lignosulfonate (SLS) on the hydrothermal reaction between natural pyrite and CuSO4 in H2SO4 media at 180–220 °C was studied at [H2SO4]0 = 10–30 g/dm3, [Cu]0 = 6–24 g/dm3 and [SLS]0 = 0–1.0 g/dm3. Process efficiency was evaluated by Fe extraction into solution and Cu precipitation on the solid phase, and products were characterized by XRD and SEM/EDS. SLS markedly intensified pyrite conversion: at 200 °C and 120 min Fe extraction increased from 14 to 26 % and Cu precipitation from 5 to 23 %, while at 220 °C Fe extraction reached 33.4 % and Cu precipitation 26.8 %. XRD confirmed the sequential transformation CuS → Cu1.8S. SEM/EDS showed that SLS converts localized nucleation of CuxS on defect sites into the formation of a fine, loosely packed and well-dispersed copper sulfide phase. The results demonstrate that lignosulfonate surfactants efficiently suppress passivation and enhance mass transfer, providing a basis for intensifying hydrothermal pretreatment of pyrite-bearing industrial materials.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Analysis
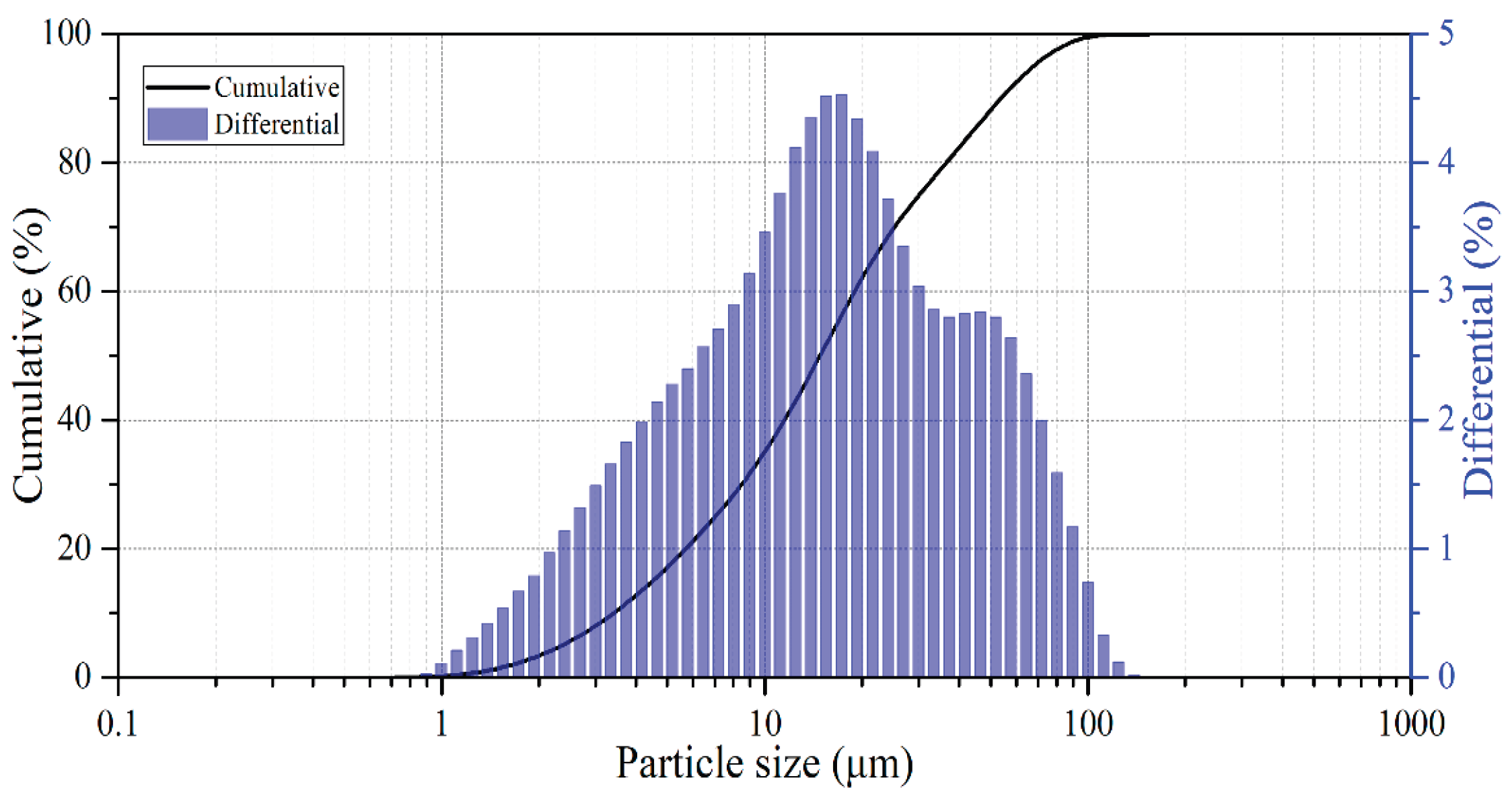
2.2. Materials and Reagents
2.2. Experimental Equipment and Procedures
3. Results and Discussion
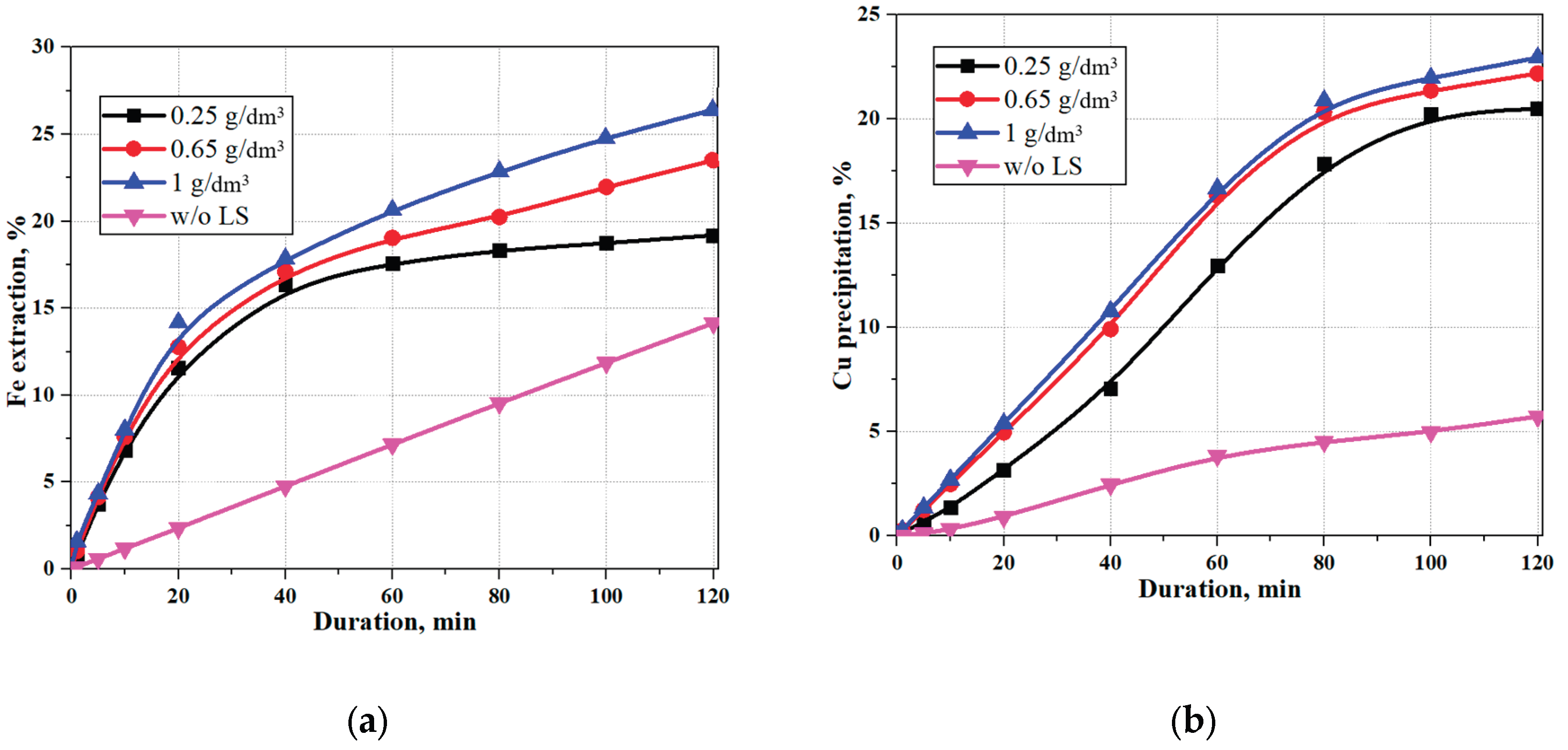
3.1. Effect of Sodium Lignosulfonate
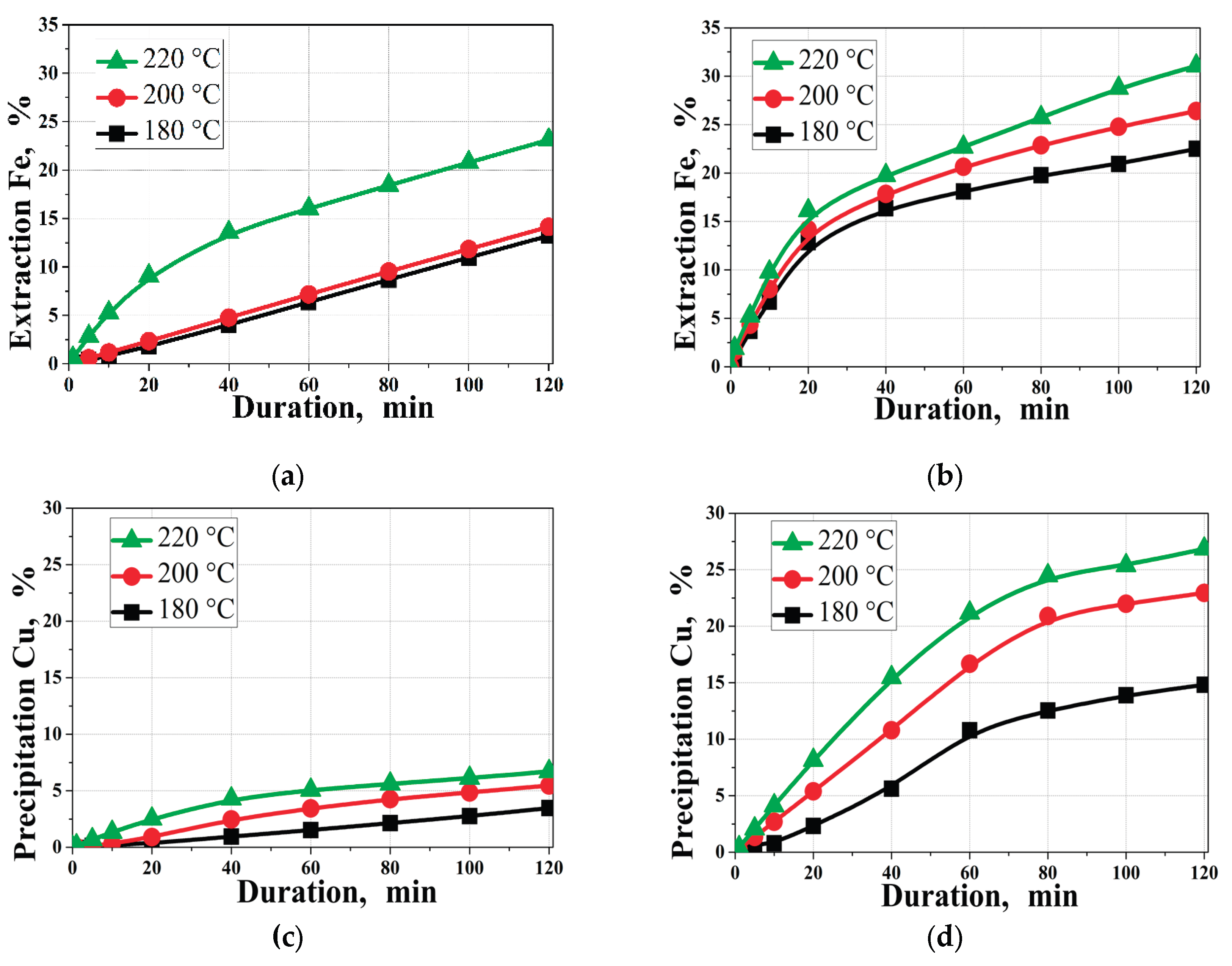
3.2. Effect of Temperature
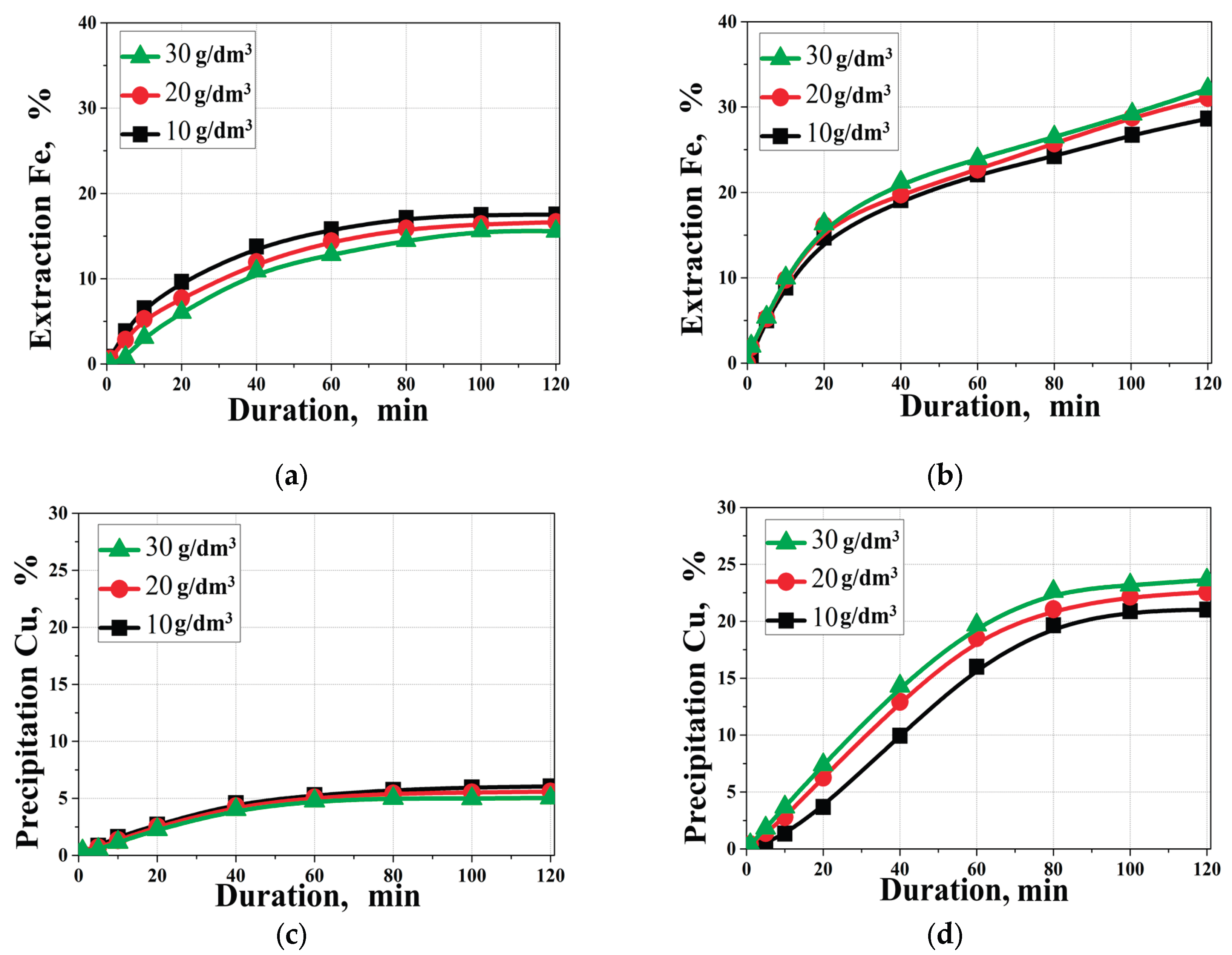
3.3. Effect of Initial Sulfuric Acid Concentration in Solution
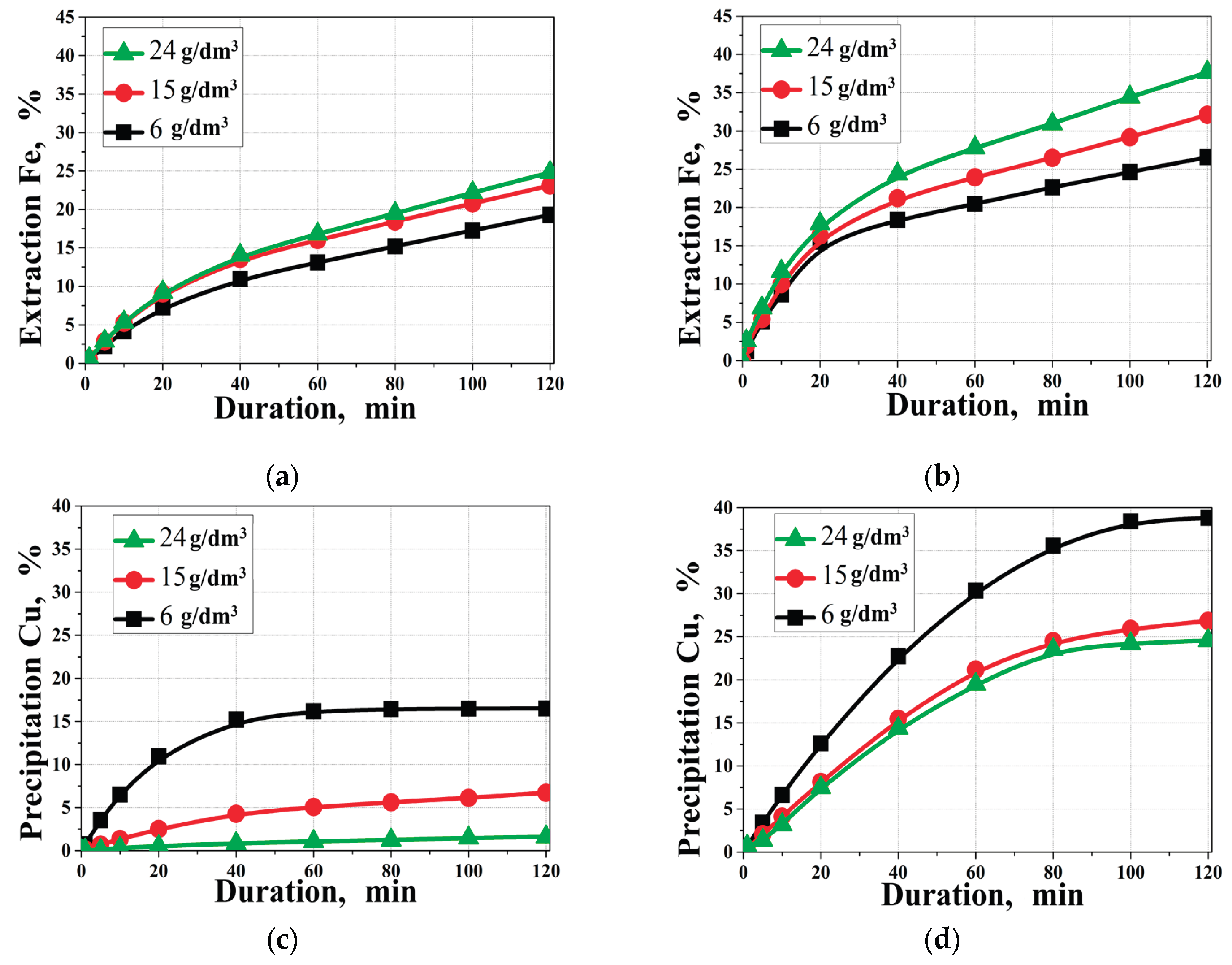
3.4. Effect of Initial Copper Concentration
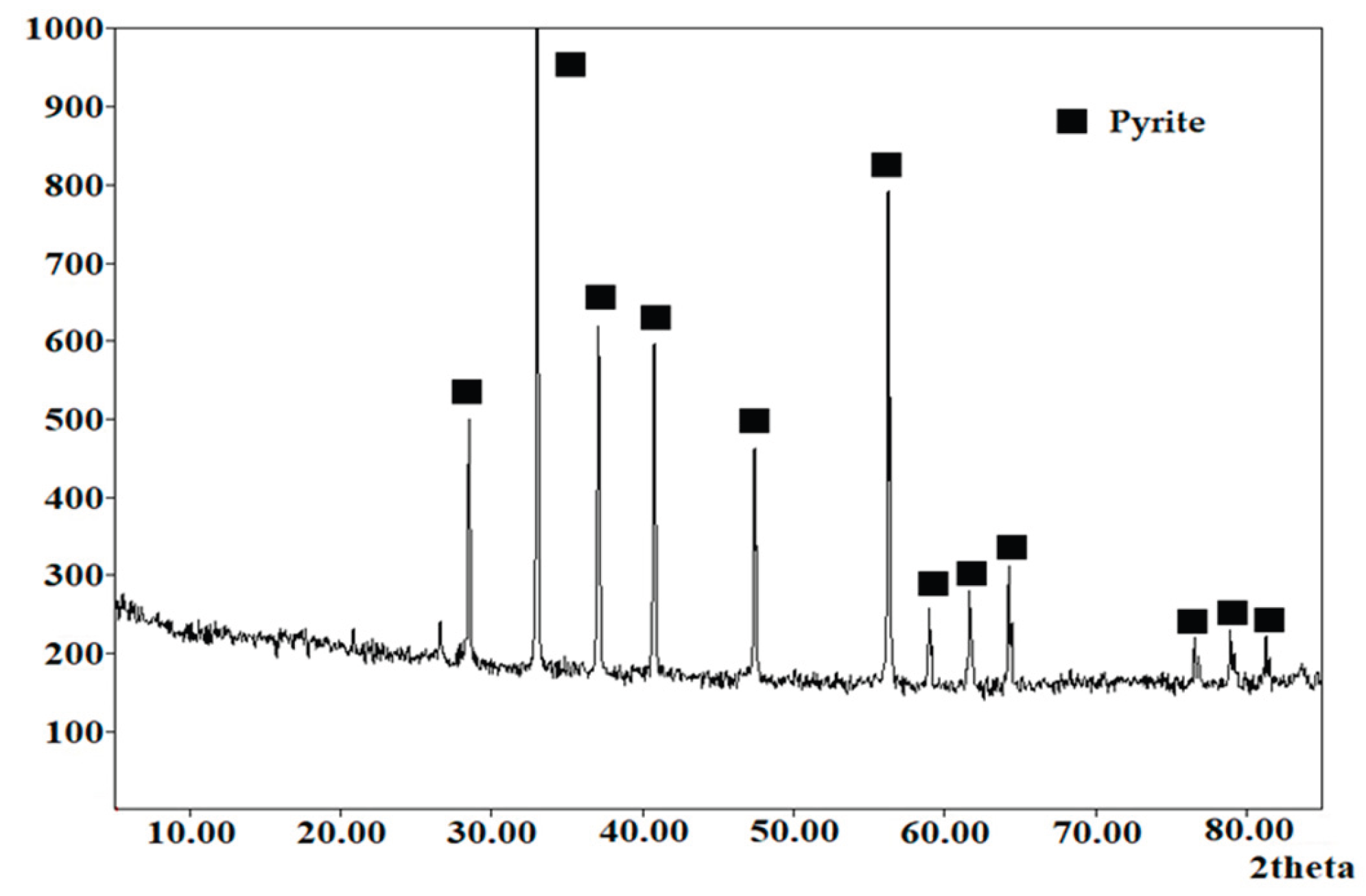
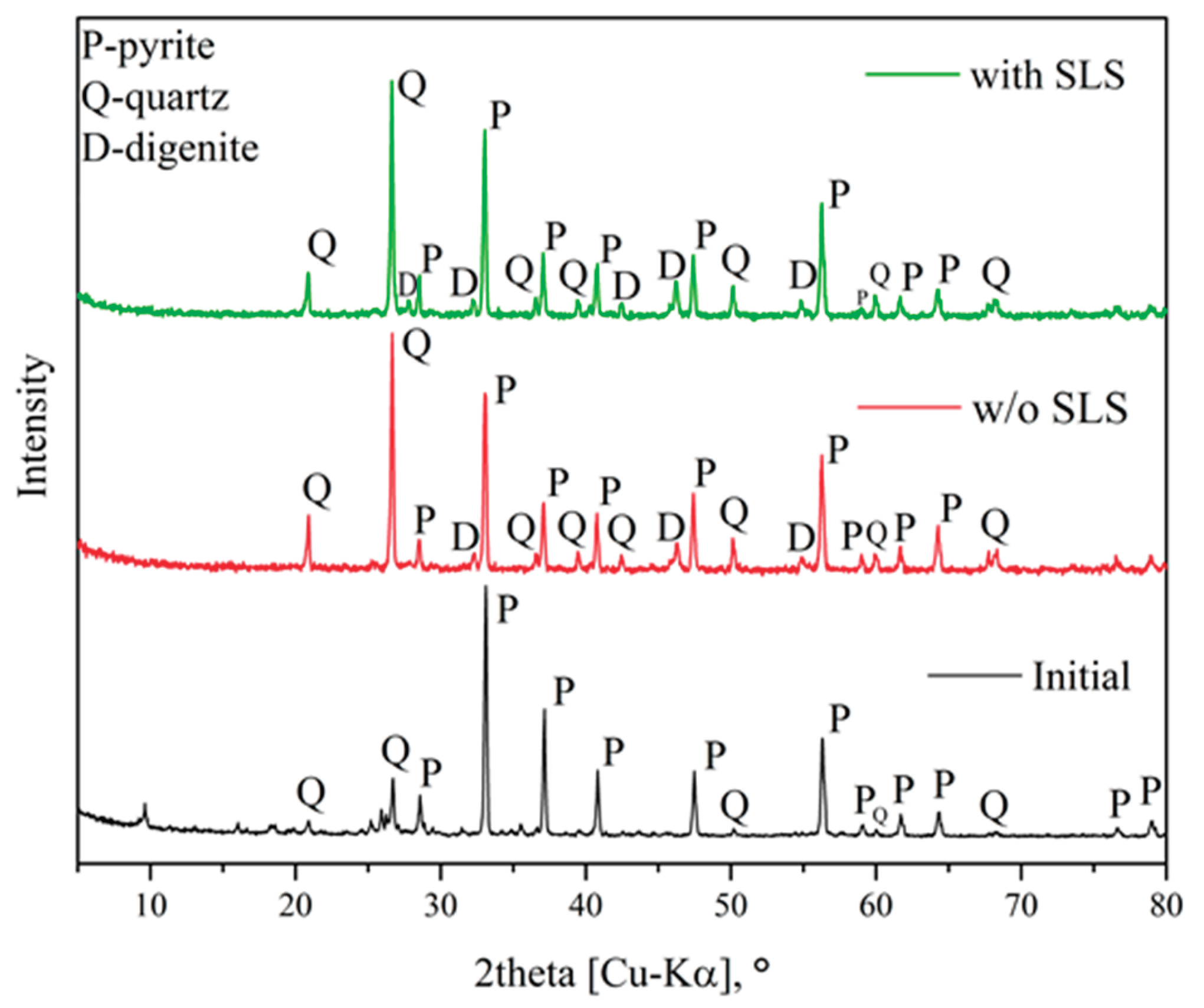
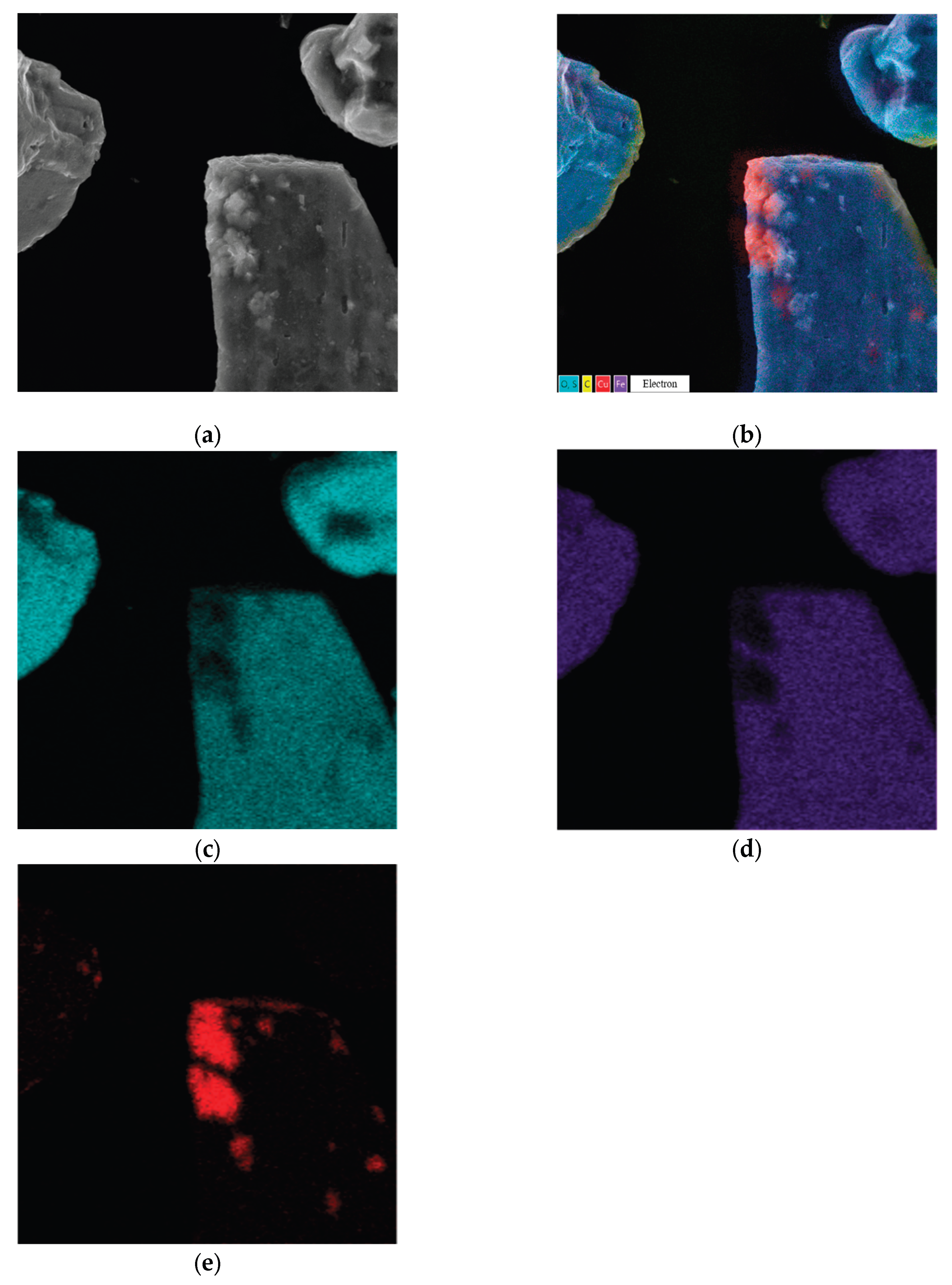
3.5. Characteristics of the Resulting Precipitates
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- US Geological Survey. Copper Data Sheet—Mineral Commodity Summaries 2020; USGS: Reston, VA, USA, 2020. Available online: https://pubs.usgs.gov/periodicals/mcs2020/mcs2020-copper.pdf (accessed on 15 October 2025).
- Dreisinger, D. Copper leaching from primary sulfides: Options for biological and chemical extraction of copper. Hydrometallurgy 2006, 83, 10–20. [Google Scholar] [CrossRef]
- International Copper Study Group. The World Copper Factbook 2019; ICSG: Lisbon, Portugal, 2020. Available online: https://www.icsg.org (accessed on 15 October 2025).
- Kelchevskaya, N.R.; Altushkin, I.A.; Korol, Yu.A.; Bondarenko, N.S. Peculiarities and importance of price formation in non-ferrous metallurgy (the case of copper concentrate production). Tsvetnye Metally 2016, 8, 13–19. [Google Scholar] [CrossRef]
- Altushkin, I.A.; Vorob’ev, A.G.; Kelchevskaya, N.R.; Korol, Yu.A. Pricing in blister copper production: World market features and competitiveness requirements. Tsvetnye Metally 2017, 12, 7–11. [Google Scholar] [CrossRef]
- Schlesinger, M.; Sole, K.; Davenport, W.; Alvear Flores, G. Extractive Metallurgy of Copper, 6th ed.; Elsevier: Amsterdam, The Netherlands, 2021. [Google Scholar] [CrossRef]
- Gorai, B.; Jana, R.K.; Premchand. Characteristics and utilization of copper slag—A review. Resour. Conserv. Recycl. 2003, 39, 299–313. [Google Scholar] [CrossRef]
- Xu, B.; Ma, Y.; Gao, W.; Yang, J.; Yang, Y.; Li, Q.; Jiang, T. A review of the comprehensive recovery of valuable elements from copper smelting open-circuit dust and arsenic treatment. JOM 2020, 72, 3860–3875. [Google Scholar] [CrossRef]
- Nazari, A.M.; Radzinski, R.; Ghahreman, A. Review of arsenic metallurgy: Treatment of arsenical minerals and the immobilization of arsenic. Hydrometallurgy 2016, 174, 258–281. [Google Scholar] [CrossRef]
- Lane, D.J.; Cook, N.J.; Grano, S.R.; Ehrig, K. Selective leaching of penalty elements from copper concentrates: A review. Miner. Eng. 2016, 98, 110–121. [Google Scholar] [CrossRef]
- Wills, B.A.; Finch, J.A. Wills’ Mineral Processing Technology: An Introduction to the Practical Aspects of Ore Treatment and Mineral Recovery, 8th ed.; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar] [CrossRef]
- Voigt, S.; Szargan, R.; Suoninen, E. Interaction of copper(II) ions with pyrite and its influence on ethyl xanthate adsorption. Surf. Interface Anal. 1994, 21, 526–536. [Google Scholar] [CrossRef]
- Yılmaz, T.; Alp, İ.; Deveci, H.; Celep, O. Production of a copper concentrate by flotation from Yomra–Kayabasi massive Cu–Zn sulfide ore. J. Sci. Technol. Dumlupinar Univ. (In Turkish). 2008, 15, 57–64. [Google Scholar]
- Chandra, A.P.; Gerson, A.R. A review of the fundamental studies of copper activation mechanisms for selective flotation of sphalerite and pyrite. Adv. Colloid Interface Sci. 2009, 145, 97–110. [Google Scholar] [CrossRef] [PubMed]
- Bıçak, Ö.; Ekmekçi, Z.; Can, M.; Öztürk, Y. The effect of water chemistry on froth stability and surface chemistry of flotation of a Cu–Zn sulfide ore. Int. J. Miner. Process. 2012, 102–103, 32–37. [Google Scholar] [CrossRef]
- Xu, Z.; Zhang, Q.; Rao, S.R.; Finch, J.A. An in-plant test of sphalerite flotation without copper activation. In Proceedings of the 24th Annual CMP Conference, Ottawa, Canada, 1992. Paper No. 14. [Google Scholar]
- Zhang, Q.; Xu, Z.; Bozkurt, V.; Finch, J.A. Pyrite flotation in the presence of metal ions and sphalerite. Int. J. Miner. Process. 1997, 52, 187–201. [Google Scholar] [CrossRef]
- Lottermoser, B.G. Mine Wastes: Characterization, Treatment and Environmental Impacts, Third Ed. ed; Springer, 2007. [Google Scholar] [CrossRef]
- Lottermoser, B.G. Recycling, reuse and rehabilitation of mine wastes. Elements 2011, 7, 405–410. [Google Scholar] [CrossRef]
- Esposito, M.; Tse, T.; Soufani, K. Is the circular economy a new fast-expanding market? Thunderbird Int. Bus. Rev. 2017, 59, 9–14. [Google Scholar] [CrossRef]
- Kossoff, D.; Dubbin, W.E.; Alfredsson, M.; Edwards, S.J.; Macklin, M.G.; Hudson-Edwards, K.A. Mine tailings dams: Characteristics, failure, environmental impacts, and remediation. Appl. Geochem 2014, 51, 229–245. [Google Scholar] [CrossRef]
- Fernandes, G.W.; Goulart, F.F.; Ranieri, B.D.; Coelho, M.S.; Dales, K.; Boesche, N.; Soares-Filho, B. Deep into the mud: Ecological and socio-economic impacts of the Mariana dam breach (Brazil). Natureza Conserv. 2016, 14, 35–45. [Google Scholar] [CrossRef]
- Sequeira, C.A.C.; Santos, D.M.F.; Chen, Y.; Anastassakis, G. Chemical metathesis of chalcopyrite in acidic solutions. Hydrometallurgy 2008, 92, 135–140. [Google Scholar] [CrossRef]
- Dizer, O.; Rogozhnikov, D.; Karimov, K.; Kuzas, E.; Suntsov, A. Nitric Acid Dissolution of Tennantite, Chalcopyrite and Sphalerite in the Presence of Fe(III) Ions and FeS2. Materials 2022, 15, 1545. [Google Scholar] [CrossRef]
- McDonald, R.G.; Li, J.; Austin, P. High-Temperature Pressure Oxidation of Low-Grade Nickel Sulfide Concentrate with Control of Residue Composition. Minerals 2020, 10, 249. [Google Scholar] [CrossRef]
- Kuzas, E.; Rogozhnikov, D.; Dizer, O.; Karimov, K.; Shoppert, A.; Suntsov, A.; Zhidkov, I. Kinetic Study on Arsenopyrite Dissolution in Nitric Acid Media by the Rotating Disk Method. Miner. Eng. 2022, 187, 107770. [Google Scholar] [CrossRef]
- Karimov, K.A.; Rogozhnikov, D.A.; Naboichenko, S.S.; et al. Autoclave Ammonia Leaching of Silver from Low-Grade Copper Concentrates. Metallurgist 2018, 62, 783–789. [Google Scholar] [CrossRef]
- Rogozhnikov, D.A.; Zakharian, S.V.; Dizer, O.A.; Karimov, K.A. Nitric Acid Leaching of the Copper-Bearing Arsenic Sulphide Concentrate of Akzhal. Tsvetnye Metally 2020, 8, 11–17. [Google Scholar] [CrossRef]
- Rogozhnikov, D.A.; Mamyachenkov, S.V.; Karelov, S.V.; Anisimova, O.S. Nitric Acid Leaching of Polymetallic Middlings of Concentration. Russ. J. Non-Ferr. Met. 2013, 54, 440–442. [Google Scholar] [CrossRef]
- Fuentes, G.; Viñals, J.; Herreros, O. Hydrothermal purification and enrichment of Chilean copper concentrates. Part 2: Behavior of bulk concentrates. Hydrometallurgy 2009, 95, 113–120. [Google Scholar] [CrossRef]
- Kritskii, A.; Naboichenko, S.; Karimov, K.; Agarwal, V.; Lundström, M. Hydrothermal pretreatment of chalcopyrite concentrate with copper sulfate solution. Hydrometallurgy 2020, 195, 105359. [Google Scholar] [CrossRef]
- Kritskii, A.; Fuentes, G.; Deveci, H. A Critical Review of Hydrothermal Treatment of Sulfide Minerals with Cu(II) Solution in H2SO4 Media. Hydrometallurgy 2025, 231, 106413. [Google Scholar] [CrossRef]
- Liu, Q.; Liao, Y.; Wu, M.; Jia, X. Acid-Free Ultrasonic-Enhanced Hydrothermal Leaching of Copper and Zinc from Polymetallic Sulfide Secondary Concentrates. Sep. Purif. Technol. 2025, 378, 134701. [Google Scholar] [CrossRef]
- Peterson, R.D.; Wadsworth, M.E. Solid–solution reactions in the hydrothermal enrichment of chalcopyrite at elevated temperatures. In Proceedings of the EPD Congress; Warren, G.W., Ed.; TMS: Warrendale, PA, USA, 1994; pp. 275–291. [Google Scholar]
- Kritskii, A; Celep, O.; Yazici, E.; Deveci, H.; Naboichenko, S. Hydrothermal treatment of sphalerite and pyrite particles with CuSO4 solution. Minerals Engineering 2022, 180, 107507. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, W.; Cai, Y.; Qu, Y.; Pan, Y.; Zhang, W.; Zhao, K. Experimental investigation of the reactions between pyrite and aqueous Cu(I) chloride solution at 100–250 °C. Geochimica et Cosmochimica Acta 2021, 298, 1–20. [Google Scholar] [CrossRef]
- dos Santos, E.C.; de Souza, C.B.; de Almeida, I.C.; Vieira, M.M.; Dutra, A.J.B. Pyrite Oxidation Mechanism by Oxygen in Aqueous Medium. J. Phys. Chem. C 2016, 120, 17527–17536. [Google Scholar] [CrossRef]
- Rimstidt, J.D.; Vaughan, D.J. Pyrite Oxidation: A State-of-the-Art Assessment of the Reaction Mechanism. Geochim. Cosmochim. Acta 2003, 67, 873–880. [Google Scholar] [CrossRef]
- Jiang, H.; Zhang, H.; Liu, W.; Yang, T. Study on Dissolution and Passivation of Chalcopyrite under Pressurized Leaching in O2–H2O System by Electrochemical and Surface Analysis Methods. Minerals 2023, 13, 996. [Google Scholar] [CrossRef]
- Jiang, Q.; Yang, S.; Zheng, X.; Deng, J.; Zhu, X.; Liu, X.; Li, J. Lignosulphonates in Zinc Pressure Leaching: Improving Sulfur Dispersion and Stabilizing Residue Impurities. J. Clean. Prod. 2024, 438, 140355. [Google Scholar] [CrossRef]
- Tong, L.; Dreisinger, D. Interfacial Properties of Liquid Sulfur in the Pressure Leaching of Nickel Sulfide Concentrates above the Melting Point of Sulfur. Miner. Eng. 2009, 22, 456–461. [Google Scholar] [CrossRef]
- Tong, L.; Dreisinger, D. The Adsorption of Sulfur Dispersing Agents on Sulfur and Nickel Sulfide Concentrate Surfaces. Miner. Eng. 2009, 22, 445–450. [Google Scholar] [CrossRef]
- Khazieva, Z.K.; Mamyachenko, S.V.; Aleksandrova, T.N. Surfactants Influence on Sphalerite Wetting during Zinc Concentrate Pressure Leaching. Solid State Phenom. 2017, 265, 1104–1108. [Google Scholar] [CrossRef]
- Aro, T.; Fatehi, P. Production and Application of Lignosulfonates and Sulfonated Lignin. ChemSusChem 2017, 10, 1861–1877. [Google Scholar] [CrossRef] [PubMed]
- Billich, A.; Kolo, H.; Uhlig, M.; Koch, C.; Niemeyer, J.; Pfeiffer, A. Deciphering Molar Mass Distributions of Lignosulfonates with Asymmetric Flow Field-Flow Fractionation. ACS Sustain. Chem. Eng. 2024, 12, 17385–17397. [Google Scholar] [CrossRef]
- Yang, S.; Jiang, Q.; Zheng, X.; Deng, J.; Zhu, X.; Liu, X.; Li, J. Behavior of Calcium Lignosulphonate under Oxygen Pressure Leaching of a Zinc Sulfide Concentrate. Hydrometallurgy 2024, 230, 106317. [Google Scholar] [CrossRef]
- Wei, D.; Wang, Y.; Li, D.; Chen, M.; Luo, H. Effects of Temperature, Oxygen Partial Pressure and Calcium Lignosulphonate on Chalcopyrite Dissolution in Sulfuric Acid Solution. Trans. Nonferrous Met. Soc. China 2022, 32, 3368–3378. [Google Scholar] [CrossRef]
- Ouyang, X.; Qiu, X.; Chen, P. Adsorption Characteristics of Lignosulfonates in Salt-Free and Electrolyte Solutions. Biomacromolecules 2011, 12, 1652–1659. [Google Scholar] [CrossRef]
- Owusu, C.; Dreisinger, D.B. Interfacial Properties in Liquid Sulfur, Aqueous Zinc Sulfate and Zinc Sulfide Systems. Hydrometallurgy 1996, 43, 207–218. [Google Scholar] [CrossRef]
- Zies, E.G.; Allen, E.T.; Merwin, H.E. Some reactions involved in secondary copper sulphide enrichment. Econ. Geol. 1916, 11, 407–503. [Google Scholar] [CrossRef]
- Fuentes, G.; Viñals, J.; Herreros, O. Hydrothermal purification and enrichment of Chilean copper concentrates. Part 1: Behavior of bornite, covellite, and pyrite. Hydrometallurgy 2009, 95, 104–112. [Google Scholar] [CrossRef]
- Kritskii, A.; Naboichenko, S. Hydrothermal Treatment of Arsenopyrite Particles with CuSO4 Solution. Materials 2021, 14, 7472. [Google Scholar] [CrossRef]
- Viñals, J.; Fuentes, G.; Hernandez, M.C.; Herreros, O. Transformation of sphalerite particles into copper sulfide particles by hydrothermal treatment with Cu(II) ions. Hydrometallurgy 2004, 75, 177–187. [Google Scholar] [CrossRef]
- Chandra, A.P.; Puskar, L.; Simpson, D.J.; Gerson, A.R. Copper and xanthate adsorption onto pyrite surfaces: Implications for mineral separation through flotation. Int. J. Miner. Process. 2012, 114–117, 16–26. [Google Scholar] [CrossRef]
- Guo, B.; Peng, Y. The interaction between copper species and pyrite surfaces in copper cyanide solutions. Int. J. Miner. Process. 2017, 158, 85–92. [Google Scholar] [CrossRef]
- Guo, B. Electrodeposition on pyrite from copper(I) cyanide electrolyte. RSC Adv. 2016, 6, 2183–2190. [Google Scholar] [CrossRef]
- Weisener, C.; Gerson, A.R. An Investigation of the Cu(II) Adsorption Mechanism on Pyrite by ARXPS and SIMS. Miner. Eng. 2000, 13, 1329–1340. [Google Scholar] [CrossRef]
- von Oertzen, G.U.; Skinner, W.; Nesbitt, H.W.; Pratt, A.; Buckley, A.N. Cu Adsorption on Pyrite (100): Ab Initio and Spectroscopic Studies. Surf. Sci. 2007, 601, 5794–5799. [Google Scholar] [CrossRef]
- Yang, B.; Hu, Y.; Sun, W.; Sun, Y.; Gao, Z. Activation of Pyrite by Copper Ions and Its Depression by Thioglycollic Acid in the Flotation of a Complex Sulfide Ore. Minerals 2018, 8, 16. [Google Scholar] [CrossRef]
- You, Y.; Hu, Y.; Sun, W.; Wang, D. Study of Galvanic Interactions between Pyrite and Chalcopyrite in a Flowing System. Environ. Geol. 2007, 52, 1093–1099. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



