Submitted:
18 December 2025
Posted:
18 December 2025
You are already at the latest version
Abstract
Mushrooms have long been valued not only as food but also for their medicinal properties, especially in Eastern European traditional medicine. Species such as Inonotus obliquus, Fomitopsis officinalis, Piptoporus betulinus and Fomes fomentarius have been used to treat gastrointestinal problems, cancers, respiratory ailments and more. Modern research confirms their diverse pharmacological effects, including antitumor, immunomodulatory, antioxidant, antiviral, antibacterial and antidiabetic activities. In addition, mushrooms are widely incorporated into functional foods and nutraceuticals that promote health. Their sustainable cultivation, efficient use of agricultural residues, rapid growth cycles and resilience to environmental stressors make them an environmentally friendly source of food and pharmaceuticals. This review focuses on the potential of fungi to inhibit advanced glycation end products (AGEs)—harmful compounds formed through the non-enzymatic binding of sugars to proteins, lipids or nucleic acids. AGEs are strongly associated with the progression of chronic diseases such as diabetes, cardiovascular disorders, neurodegeneration and aging. Natural AGE inhibitors from mushrooms represent a promising therapeutic alternative to synthetic agents, as they may offer broader mechanisms of action with fewer adverse effects.

Keywords:
1. Introduction
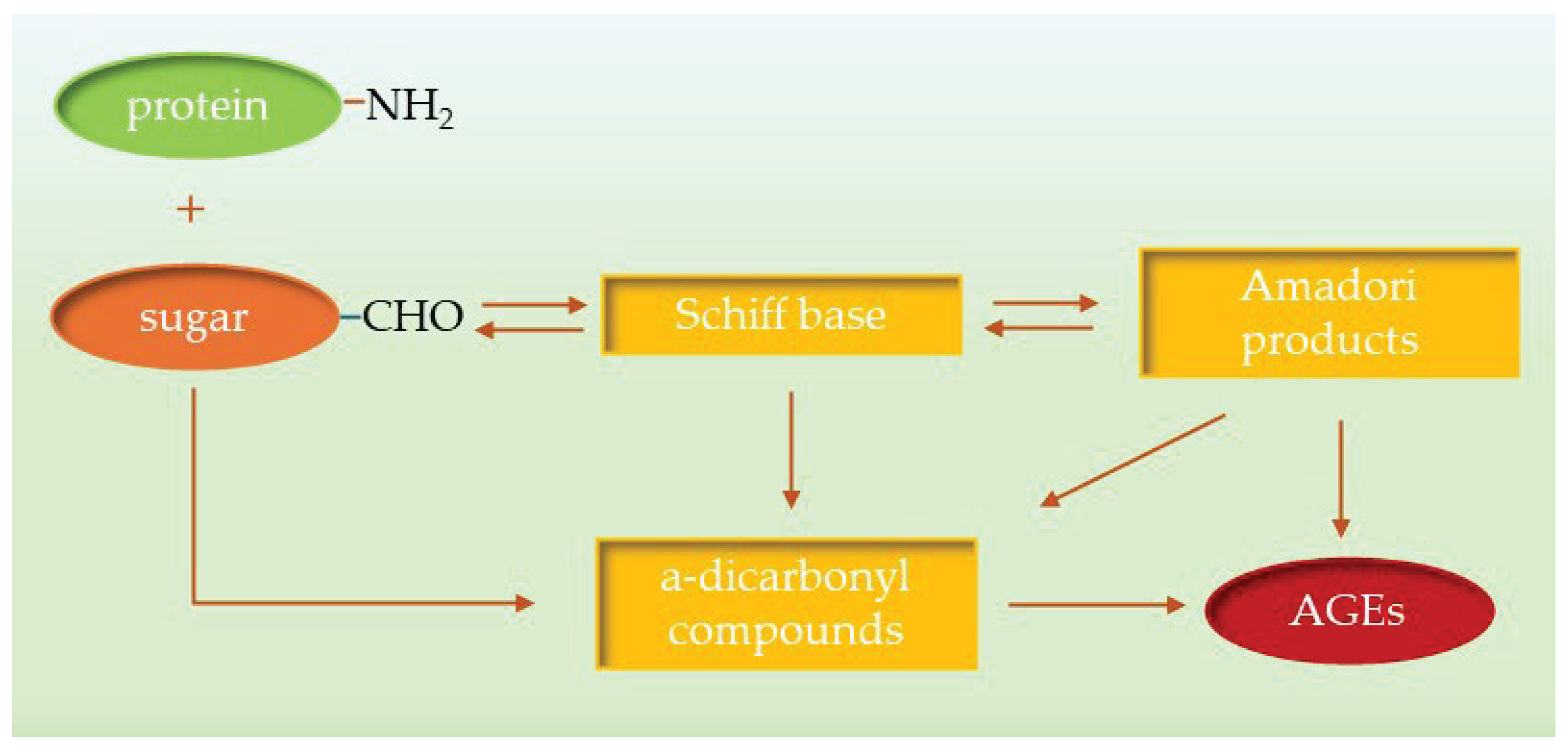
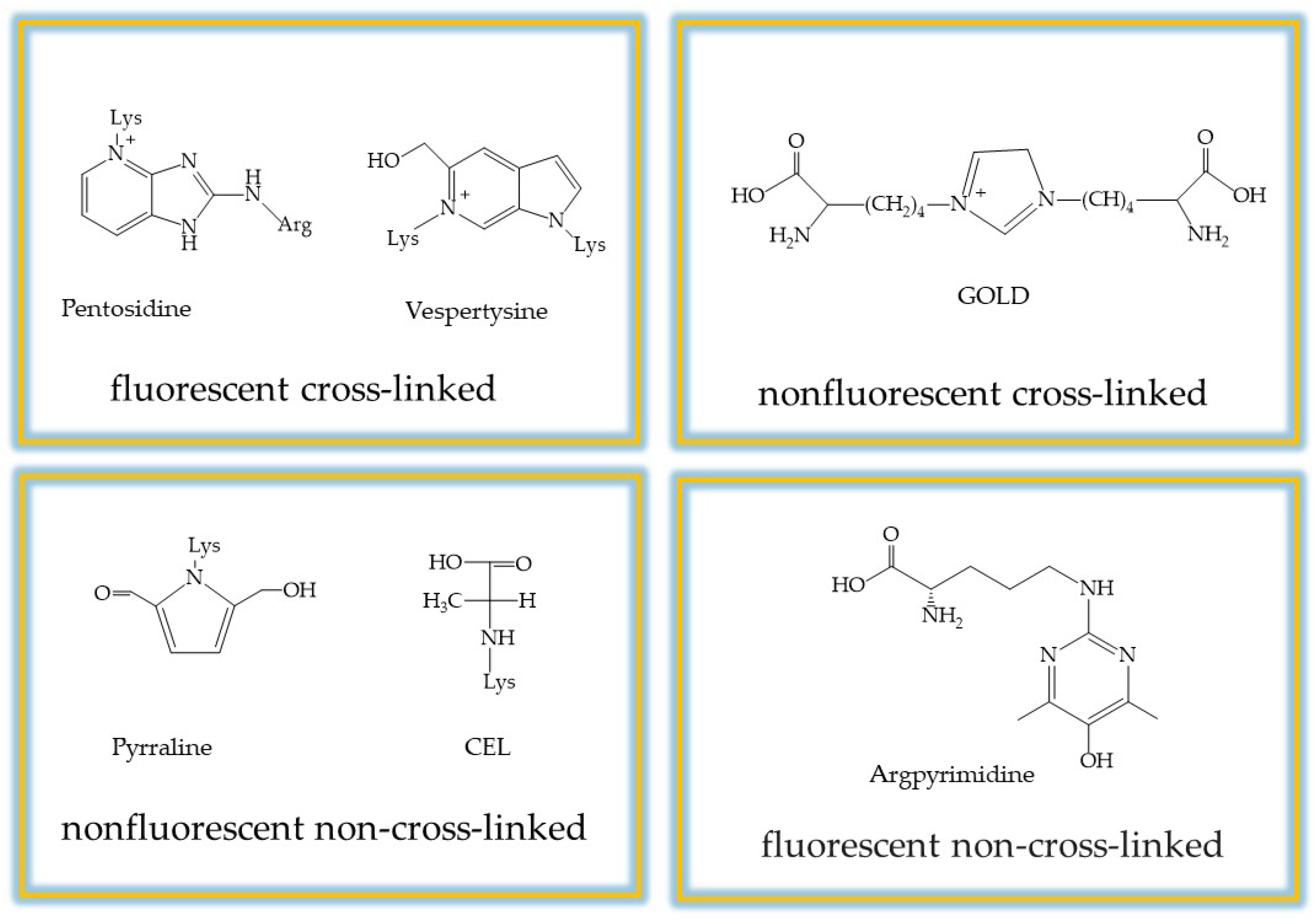
2. Advanced Glycation End-Products (AGEs) and Inhibition Strategies
3. Mushrooms as a Potential Source of Natural AGE Inhibitors
4. Specific Mushroom Species and Their AGE-Inhibiting Properties
| Mushroom Species |
Active Fraction/ Compound(s) |
Proposed Mechanism(s) |
Ref. |
|---|---|---|---|
| Lignosus rhinocerus | Medium-molecular- weight (MMW) fraction |
Inhibition of CML and pentosidine, superoxide anion radical scavenging, glyoxalase I activity | [38] |
| Ganoderma capense | Polysaccharides (GC70 fraction) |
Inhibition of AGE formation, DPPH and hydroxyl radical scavenging |
[39] |
| Ganoderma lucidum | FYGL proteoglycan | Inhibits glycation at every stage; suppresses glycoxidation; forms protective complexes with proteins | [40] |
| Auricularia auricula | Polysaccharides | Inhibition of AGE formation, attenuation of oxidative damage, preservation of sulfhydryl groups, metal chelation, RAGE/TGF-β/NOX4 pathway modulation |
[41,42] |
| Pleurotus ostreatus | Methanolic extract (Fraction F4), phenolic and flavonoid compounds |
Inhibition of glycated hemoglobin formation, fructosamine, protein carbonyls, protein aggregation, fluorescent AGEs, antioxidant activity |
[43] |
| Inonotus obliquus | Polyphenol decoction (CPD) |
Inhibition of albumin and collagen gel glycation, intracellular antioxidant activity |
[44] |
| Pholiota nameko | Polysaccharides | Antioxidant activity and ROS scavenging ability | [46] |
| Lactarius deterrimus | 50% ethanolic extract |
Reduction of the RAGE/NF-κB activation, reduction of enzymatic O-GlcNAcylation, reduction of oxidative stress | [47] |
| Phellinus linteus | Phenolic compounds, polysaccharides, ergothioneine |
Carbonyl trapping, free radical scavenging, metabolic regulation | [48,49] |
| Various mushrooms | Ergothioneine | Antioxidant activity; free radical scavenging | [50] |
| Boletus snicus | Polysaccharides (BSP-2b fraction) |
Activity correlated with high molecular weight and high uronic acid content | [51] |
| Agaricus bisporus, Pleurotus ostreatus | Water-soluble and ethanol-insoluble fractions | Retention of activity post-UV irradiation suggests the involvement of stable, non-phenolic compounds | [52] |
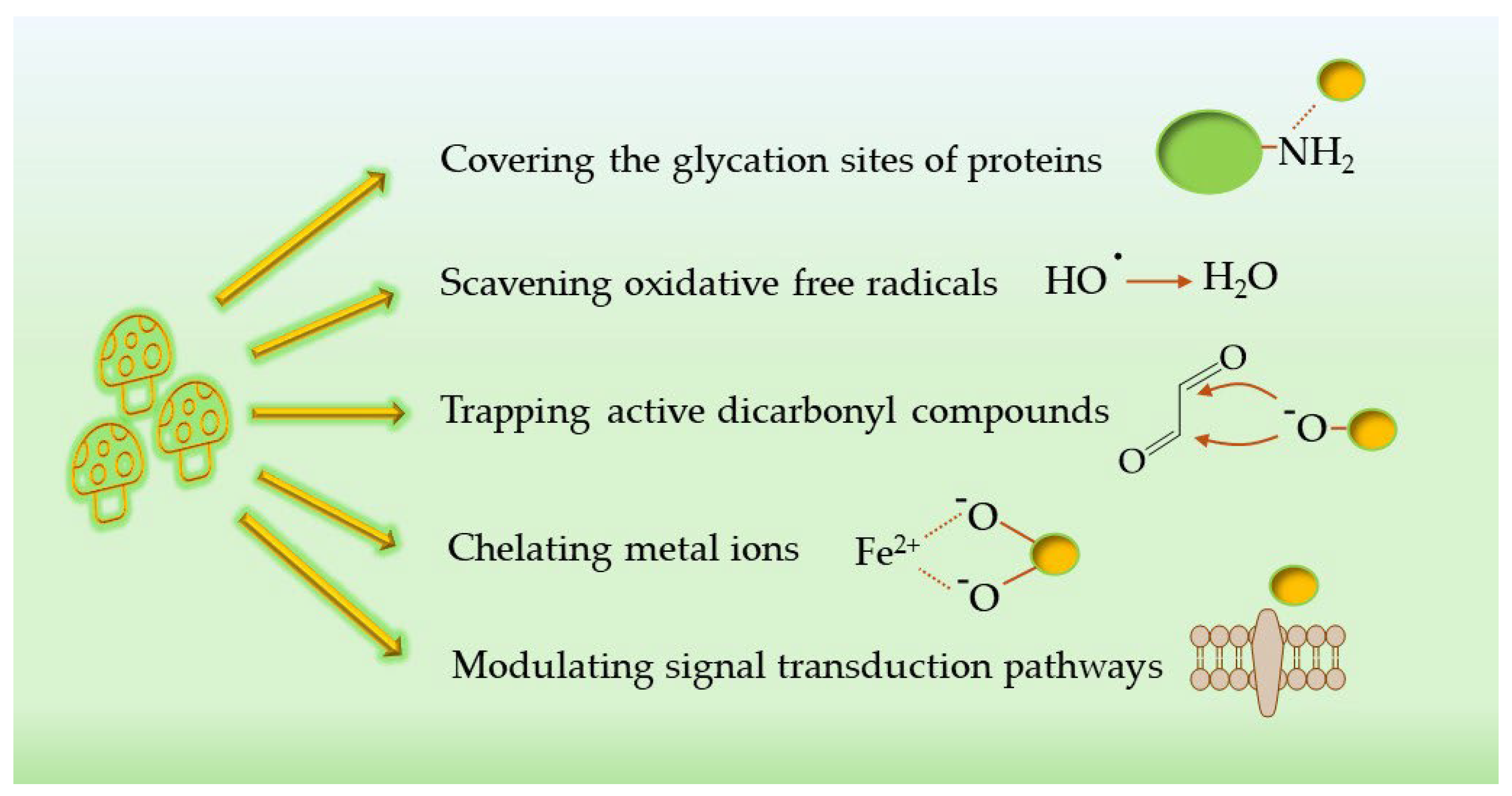
5. Mechanisms of Action of Mushroom-Derived AGE Inhibitors
5.1. Antioxidant Activity
5.2. Scavenging of Carbonyl
5.3. Interfering with the Glycation Process
5.4. Modulation of Signal Transduction Pathways
5.5. Metal-Ion Chelation
5.6. Synergistic Interactions
5.7. The Critical Role of Molecular Weight and Uronic Acid Content
5.8. Role of Stable, High-Molecular-Weight Compounds
6. Evidence from Research Studies
7. Potential Applications and Future Directions
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wani, B.A.; Bodha, R.H.; Wani, A.H. Nutritional and medicinal importance of mushrooms. J. Med. Plant. Res. 2010, 4, 2598–2604. [Google Scholar] [CrossRef]
- Szychowski, K.A.; Skóra, B.; Pomianek, T.; Gmiński. J. Inonotus obliquus – from folk medicine to clinical use. J. Tradit. Complement. Med. 2020, 11, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Elkhateeb, W.; Daba, G.; Elnahas, M.; Thomas, P.W. Fomitopsis officinalis mushroom ancient gold mine of functional components and biological activities for modern medicine. Egypt. Pharm. J. 2019, 18, 285–289. [Google Scholar] [CrossRef]
- Fijałkowska, A.; Muszyńska, B.; Sułkowska-Ziaja, K; Kała, K.; Pawlik, A.; Stefaniuk, D.; Matuszewska, A.; Piska, K.; Pękala, E.; Kaczmarczyk, P.; Piętka, J.; Jaszek, M. Medicinal potential of mycelium and fruiting bodies of an arboreal mushroom Fomitopsis officinalis in therapy of lifestyle diseases. Sci. Rep. 2020, 10, 20081. [Google Scholar] [CrossRef]
- Papp, N.; Rudolf, K.; Bencsik, T.; Czégényi, D. Ethnomycological use of Fomes fomentarius (L.) Fr. and Piptoporus betulinus (Bull.) P. Karst. in Transylvania, Romania. Genet. Resour. Crop. Evol. 2017, 64, 101–111. [Google Scholar] [CrossRef]
- Rašeta, M.; Kebert, M.; Pintać Šarac, D.; Mišković, J.; Berežni, S.; Kulmány, Á.E.; Zupkó, I.; Karaman, M.; Jovanović-Šanta, S. Bioactive Potential of Balkan Fomes fomentarius Strains: Novel Insights into Comparative Mycochemical Composition and Antioxidant, Anti-Acetylcholinesterase, and Antiproliferative Activities. Microorganisms 2025, 13, 1210. [Google Scholar] [CrossRef]
- Ulrike, G.; Zöll, M.; Peintner, U.; Rollinger, J.M. European medicinal polypores – A modern view on traditional uses. J. Ethnopharmaco. 2014, 154, 564–583. [Google Scholar] [CrossRef]
- Gao, Y.; Zhou, S.; Chen, G.; Dai, X; Ye, J. A. Phase I/II study of a Ganoderma lucidum extract (Ganopofy) in patients with advanced cancer. Int. J. Med. Mushrooms. 2002, 4, 207–214. [Google Scholar] [CrossRef]
- Gao, Y.; Zhou, Sh.; Huang, M.; Xu, A. Antibacterial and antiviral value of the genus Ganoderma P. Karst. species (Aphyllophoromycetideae): a review. Int. J. Med. Mushrooms. 2003, 5, 235–246. [Google Scholar] [CrossRef]
- Gao, Y.; Lan, J.; Dai, X.; Ye, J.; Zhou, Sh. A phase I/II study of Ling Zhi mushroom Ganoderma lucidum (W.Curt.:Fr.) Lloyd (Aphyllophoromycetideae) extract in patients with type II diabetes mellitus. Int. J. Med. Mushrooms. 2004, 6, 96–107. [Google Scholar] [CrossRef]
- Didukh, M.Y.; Wasser, S.P.; Nevo, E. Medicinal value of species of the family Agaricaceae Cohn (higher Basidiomycetes) current stage of knowledge and future perspectives. Int. J. Med. Mushrooms. 2003, 5, 133–152. [Google Scholar] [CrossRef]
- Rowan, N.J.; Smith, J.E.; Sullivan, R. Immunomodulatory activities of mushroom glucans and polysaccharide–protein complexes in animals and humans (a review). Int. J. Med. Mushrooms. 2003, 5, 95–110. [Google Scholar] [CrossRef]
- Wasser, S.P. Medicinal mushroom science: history, current status, future trends, and unsolved problems. Int. J. Med. Mushrooms. 2010, 12, 1–16. [Google Scholar] [CrossRef]
- Cheung, P.C.K. Regulatory issues of mushrooms as functional foods and dietary supplements: safety and efficacy. In Mushrooms as functional foods; Cheung, P.C.K. Wiley: New York, 2008; pp. 199–221. [Google Scholar]
- Uceda, A.B.; Mariño, L.; Casasnovas, R.; Adrover, M. An overview on glycation: molecular mechanisms, impact on proteins, pathogenesis, and inhibition. Biophys. Rev. 2024, 16, 189–218. [Google Scholar] [CrossRef]
- Velichkova, S.; Foubert, K.; Pieters, L. Natural Products as a Source of Inspiration for Novel Inhibitors of Advanced Glycation Endproducts (AGEs) Formation. Planta Med. 2021, 87, 780–801. [Google Scholar] [CrossRef]
- Peng, X.; Ma, J.; Chen, F.; Wang, M. Naturally occurring inhibitors against the formation of advanced glycation end-products. Food Funct. 2011, 2, 289–301. [Google Scholar] [CrossRef]
- Reddy, V.P.; Aryal, P.; Darkwah, E.K. Advanced Glycation End Products in Health and Disease. Microorganisms. 2022, 10, 1848. [Google Scholar] [CrossRef]
- Lapolla, A.; Traldi, P.; Fedele, D. Importance of measuring products of non-enzymatic glycation of proteins. Clin. Biochem. 2005, 38, 103–115. [Google Scholar] [CrossRef]
- Twarda-Clapa, A.; Olczak, A.; Białkowska, A.M.; Koziołkiewicz, M. Advanced Glycation End-Products (AGEs): Formation, Chemistry, Classification, Receptors, and Diseases Related to AGEs. Cells. 2022, 11, 1312. [Google Scholar] [CrossRef]
- Perrone, A.; Giovino, A.; Benny, J.; Martinelli, F. Advanced Glycation End Products (AGEs): Biochemistry, Signaling, Analytical Methods, and Epigenetic Effects. Oxid. Med. Cell Longev. 2020, 18, 3818196. [Google Scholar] [CrossRef]
- Chen, N.; Wang, N.; Fang, Q.; Yu, Z.; Hu, Y.; Jin, J.; Yang, S. Inhibition effect of AGEs formation in vitro by the two novel peptides EDYGA and DLLCIC derived from Pelodiscus sinensis. Front. Nutr. 2025, 12, 1537338. [Google Scholar] [CrossRef] [PubMed]
- Chinchansure, A.A.; Korwar, A.M.; Kulkarni, M.J.; Joshi, S.P. Recent development of plant products with anti-glycation activity: a review. RSC Adv. 2015, 5, 31113–31138. [Google Scholar] [CrossRef]
- Chu, J.; Lin, S.; Yuan, Y.; Zhang, S.; Zhang, S. Effects of quercetin and l-ascorbic acid on heterocyclic amines and advanced glycation end products production in roasted eel and lipid-mediated inhibition mechanism analysis. Food Chem. 2024, 441, 138394. [Google Scholar] [CrossRef] [PubMed]
- Song, Q.; Liu, J.; Dong, L.; Wang, X.; Zhang, X. Novel advances in inhibiting advanced glycation end product formation using natural compounds. Biomed. Pharmacother. 2021, 140, 111750. [Google Scholar] [CrossRef]
- Bakris, G.L.; Bank, A.J.; Kass, D.A.; Neutel, J.M.; Preston, R.A.; Oparil, S. Advanced glycation end-product cross-link breakers. A novel approach to cardiovascular pathologies related to the aging process. Am. J. Hypertens. 2004, 17, 23S–30S. [Google Scholar] [CrossRef]
- Jean, D.; Pouligon, M.; Dalle, C. Evaluation in vitro of AGE-crosslinks breaking ability of rosmarinic acid. Glycative Stress Research 2015, 2, 204–207. [Google Scholar] [CrossRef]
- Mizutani, K.; Ikeda, K.; Yamori, Y. Resveratrol Inhibits AGEs-Induced Proliferation and Collagen Synthesis Activity in Vascular Smooth Muscle Cells from Stroke-Prone Spontaneously Hypertensive Rats. Biochem. Biophys. Res. Comm. 2000, 274, 61–67. [Google Scholar] [CrossRef]
- Tang, Y.; Chen, A. Curcumin eliminates the effect of advanced glycation end-products (AGEs) on the divergent regulation of gene expression of receptors of AGEs by interrupting leptin signaling. Lab. Invest. 2014, 94, 503–516. [Google Scholar] [CrossRef]
- Qais, F.A.; Alam, Md.M.; Naseemb, I.; Ahmad, I. Understanding the mechanism of non-enzymatic glycation inhibition by cinnamic acid: an in vitro interaction and molecular modelling study. RSC Adv. 2016, 6, 65322–65337. [Google Scholar] [CrossRef]
- Selvakumar, G.; Venu, D.; Kuttalam, I.; Lonchin, S. Inhibition of Advanced Glycation End Product Formation in Rat Tail Tendons by Polydatin and p-Coumaric acid: an In Vitro Study. Appl. Biochem. Biotechnol. 2022, 194, 339–353. [Google Scholar] [CrossRef]
- Alam, M.M.; Ahmad, I.; Naseem, I. Inhibitory effect of quercetin in the formation of advance glycation end products of human serum albumin: An in vitro and molecular interaction study. Int. J. Biol. Macromol. 2015, 79, 336–343. [Google Scholar] [CrossRef]
- Ou, J.; Huang, J.; Wang, M.; Ou, S. Effect of rosmarinic acid and carnosic acid on AGEs formation in vitro. Food Chem. 2017, 221, 1057–1061. [Google Scholar] [CrossRef] [PubMed]
- Rashedinia, M.; Rasti Arbabi, Z.; Sabet, R.; Emami, L.; Poustforoosh, A.; Sabahi, Z. Comparison of Protective Effects of Phenolic Acids on Protein Glycation of BSA Supported by In Vitro and Docking Studies. Biochem. Res. Int. 2023, 2023, 9984618. [Google Scholar] [CrossRef] [PubMed]
- Chugh, R.M.; Mittal, P.; Mp, N.; Arora, T.; Bhattacharya, T.; Chopra, H.; Cavalu, S.; Gautam, R.K. Fungal Mushrooms: A Natural Compound with Therapeutic Applications. Front. Pharmacol. 2022, 13, 925387. [Google Scholar] [CrossRef] [PubMed]
- Bhambri, A.; Srivastava, M.; Mahale, V.G.; Mahale, S.; Karn, S.K. Mushrooms as Potential Sources of Active Metabolites and Medicines. Front. Microbiol. 2022, 13, 837266. [Google Scholar] [CrossRef]
- Tan, K.W.K.; Yong, P.H.; Ng, Z.X. Synergistic anti-glycation and antioxidant interaction among different mushroom extract combinations. Foods Raw Mater. 2026, 14, 300–320. [Google Scholar] [CrossRef]
- Yap, H.Y.; Tan, N.H.; Ng, S.T.; Tan, C.S.; Fung, S.Y. Inhibition of Protein Glycation by Tiger Milk Mushroom [Lignosus rhinocerus (Cooke) Ryvarden] and Search for Potential Anti-diabetic Activity-Related Metabolic Pathways by Genomic and Transcriptomic Data Mining. Front. Pharmacol. 2018, 9, 103. [Google Scholar] [CrossRef]
- Yan, C.; Kong, F.; Zhang, D.; Cui, J. Anti-glycated and antiradical activities in vitro of polysaccharides from Ganoderma capense. Pharmacogn. Mag. 2013, 33, 23–27. [Google Scholar] [CrossRef]
- Zhang, Y.; Pan, Y.; Li, J.; Zhang, Z.; He, Y.; Yang, H.; Zhou, P. Inhibition on α-Glucosidase Activity and Non-Enzymatic Glycation by an Anti-Oxidative Proteoglycan from Ganoderma lucidum. Molecules 2022, 27, 1457. [Google Scholar] [CrossRef]
- Gong, P.; Pei, S.; Long, H.; Yang, W.; Yao, W.; Li, N.; Wang, J.; Zhao, Y.; Chen, F.; Xie, J.; Guo, Y. Potential inhibitory effect of Auricularia auricula polysaccharide on advanced glycation end-products (AGEs). Int. J. Biol. Macromol. 2024, 262, 129856. [Google Scholar] [CrossRef]
- Shen, M.; Fang, Z.; Chen, Y.; Chen, Y.; Xiao, B.; Guo, L.; Xu, Y.; Wang, G.; Wang, W.; Zhang, Y. Hypoglycemic Effect of the Degraded Polysaccharides from the Wood Ear Medicinal Mushroom Auricularia auricula-judae (Agaricomycetes). Int. J. Med. Mushrooms. 2019, 21, 1033–1042. [Google Scholar] [CrossRef]
- Macwan, D.; Patel, H.V. Inhibitory Effect of Bioassay-Guided Fractionation of Mushroom (Pleurotus ostreatus) Extract on Fructose-induced Glycated Hemoglobin and Aggregation in vitro. Pharmacogn. Res. 2023, 15, 776–784. [Google Scholar] [CrossRef]
- Doi, N.; Araki, K.; Fukuta, Y.; Kuwagaito, Y.; Yamauchi, Y.; Sasai, Y.; Kondo, S-i.; Kuzuya, M. Anti-glycation and antioxidant effects of Chaga mushroom decoction extracted with a fermentation medium. Food Sci. Tech. Res. 2023, 29, 155–161. [Google Scholar] [CrossRef]
- Fordjour, E.; Manful, C.F.; Javed, R.; Galagedara, L.W.; Cuss, C.W.; Cheema, M.; Thomas, R. Chaga mushroom: a super-fungus with countless facets and untapped potential. Front. Pharmacol. 2023, 14, 1273786. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Lin, T.-Y.; Lin, J.-A.; Cheng, K.-C.; Santoso, S.P.; Chou, C.-H.; Hsieh, C.-W. Effect of Pholiota nameko Polysaccharides Inhibiting Methylglyoxal-Induced Glycation Damage In Vitro. Antioxidants. 2021, 10, 1589. [Google Scholar] [CrossRef]
- Jovanović, J.A.; Mihailović, M.; Uskoković, A.S.; Grdović, N.; Dinić, S.; Poznanović, G.; Mujić, I.; Vidaković, M. Evaluation of the Antioxidant and Antiglycation Effects of Lactarius deterrimus and Castanea sativa Extracts on Hepatorenal Injury in Streptozotocin-Induced Diabetic Rats. Front. Pharmacol. 2017, 8, 793. [Google Scholar] [CrossRef]
- Lee, Y.S.; Kang, Y.H.; Jung, J.Y.; Lee, S.; Ohuchi, K.; Shin, K.H.; Kang, I.J.; Park, J.H.; Shin, H.K.; Lim, S.S. Protein glycation inhibitors from the fruiting body of Phellinus linteus. Biol. Pharm. Bull. 2008, 31, 1968–1972. [Google Scholar] [CrossRef]
- Chen, W.; Tan, H.; Liu, Q.; Zheng, X.; Zhang, H.; Liu, Y.; Xu, L. A Review: The Bioactivities and Pharmacological Applications of Phellinus linteus. Molecules. 2019, 24, 1888. [Google Scholar] [CrossRef]
- Use of ergothioneine and/or its derivatives as an anti-glycation agent. Available online: https://patents.google.com/patent/US20020042438A1/en (accessed on 15 June 2025).
- Sun, L.; Su, X.; Zhuang, Y. Preparation, characterization and antiglycation activities of the novel polysaccharides from Boletus snicus. Int. J. Biol. Macromol. 2016, 92, 607–614. [Google Scholar] [CrossRef]
- Gallotti, F.; Lavelli, V. The Effect of UV Irradiation on Vitamin D2 Content and Antioxidant and Antiglycation Activities of Mushrooms. Foods 2020, 9, 1087. [Google Scholar] [CrossRef]
- Liuzzi, G.M.; Petraglia, T.; Latronico, T.; Crescenzi, A.; Rossano, R. Antioxidant Compounds from Edible Mushrooms as Potential Candidates for Treating Age-Related Neurodegenerative Diseases. Nutrients. 2023, 15, 1913. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




