Submitted:
16 December 2025
Posted:
17 December 2025
You are already at the latest version
Abstract
Keywords:
Author Summary
Overview
- Part I: The Human Sex Recognition (HSR) model
1. Introduction
Origins and Evolution of the Model
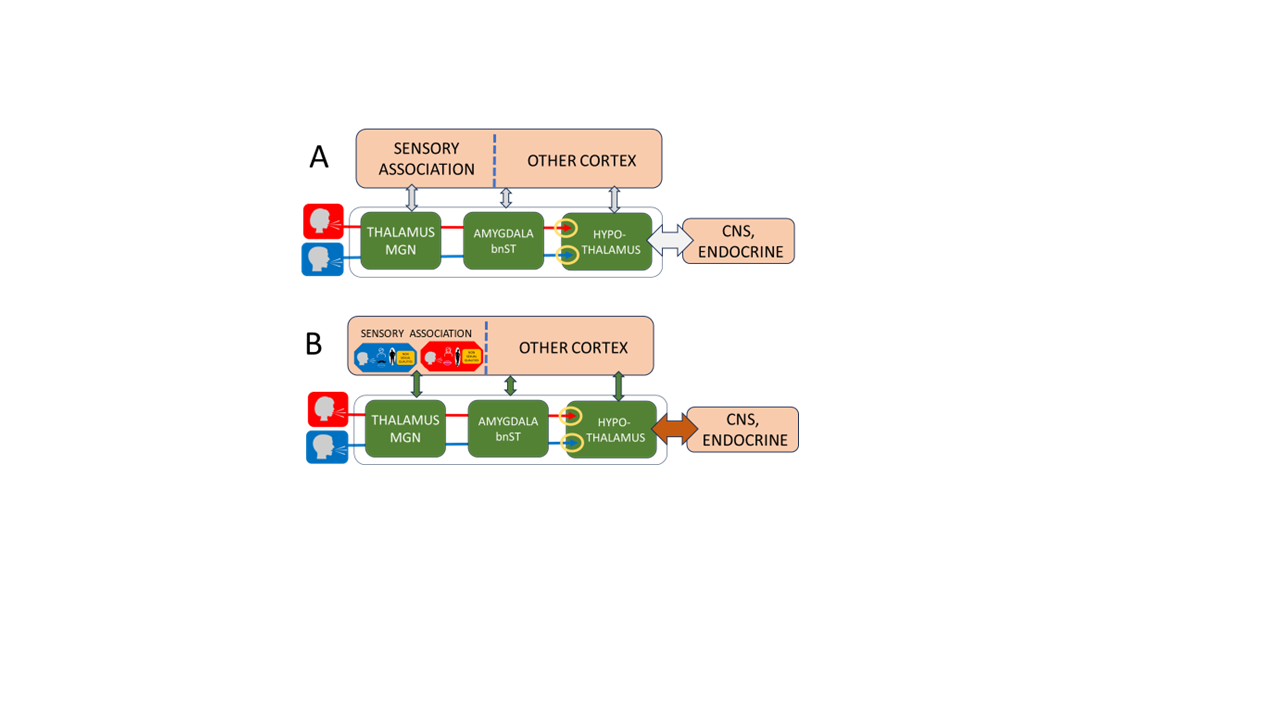
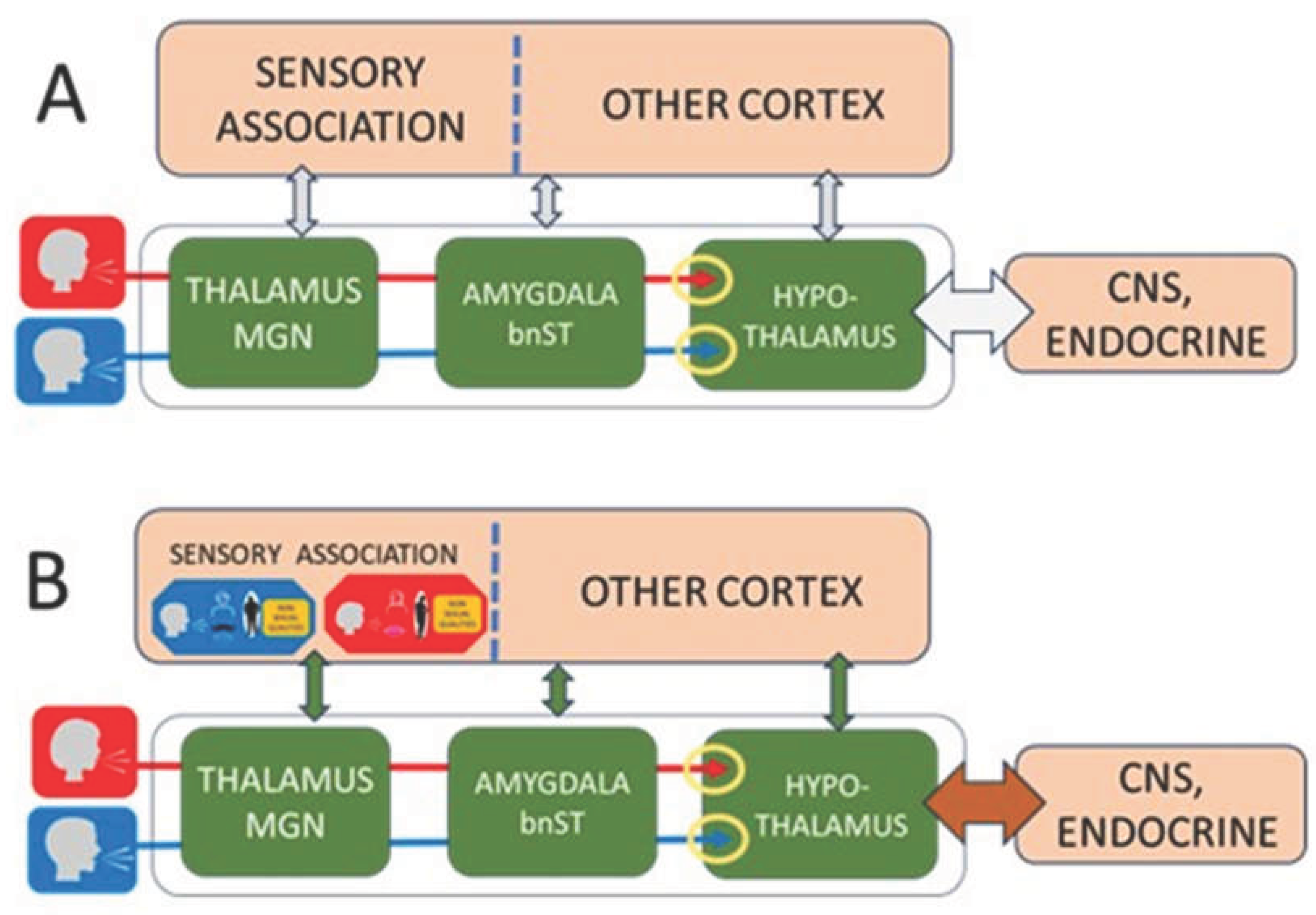
2. Developmental Architecture of Human Sex Recognition
Prenatal foundations
Auditory scaffold in infancy
Childhood cross-modal learning
Pubertal expansion and motivational reorganization
- Part II: Evaluation, Complementarity, and Testable Implications
3. Complementary Evaluation of Human Sex Recognition Models
4. Scientific Criteria and Model Evaluation
5. Predictions and Testable Hypotheses
-
Developmental timingChildren should reliably classify sex by voice earlier than by face, reflecting earlier auditory access and learning.
-
Sensory deprivationCongenital or early auditory deprivation should impair the development of sex recognition more strongly than olfactory impairment.
-
Pubertal reorganizationAdolescents should exhibit increased hypothalamic and limbic responsiveness to visual sex cues compared to children, reflecting the integration of earlier learned associations into motivational circuits.
-
Chemosensory predictionsIf pheromone-based models are correct, olfactory deprivation should directly impair sex recognition from auditory or visual cues. Although anosmia and smell loss are associated with changes in sexual behavior, intimacy, or mood (Croy et al., 2013; Oleszkiewicz et al., 2020), no studies to date demonstrate a direct impairment in voice- or face-based sex recognition in individuals with olfactory deficits.
-
Neural pathway differentiationThe HSR model predicts that male and female voices may engage partially distinct neural pathways from the medial geniculate nucleus (MGN) to hypothalamic and limbic structures, with possible overlap. Existing neuroimaging studies already demonstrate sex-differentiated cortical processing of voices, including tonotopic organization (Talavage et al., 2004), hemispheric asymmetries (Lattner et al., 2004), sex-specific voice activations (Sokhi et al., 2005), gender-sensitive processing (Charest et al., 2012), and distinct representations of pitch and timbre (Allen et al., 2016). These approaches could be extended to test subcortical pathway differentiation predicted by the HSR framework.
6. Chemosensory Influences in Humans: A Modulatory Role
7. Biological Feasibility and Comparative Perspective
8. Interpreting Effect Size, Sample Scale, and Error Bounds
9. Conclusion
References
- Allen, E.J.; Burton, P.C.; Olman, C.A.; Oxenham, A.J. Representations of pitch and timbre variation in human auditory cortex. The Journal of Neuroscience 2016, 37, 1284–1293. [Google Scholar] [CrossRef]
- Belin, P.; Zatorre, R.; Ahad, P. Human temporal-lobe regions sensitive to voice. Nature Neuroscience 2002, 5, 965–970. [Google Scholar]
- Blasi, C.H.; et al. Young children’s attributes are better conveyed by voices than by faces. Journal of Experimental Child Psychology 2023. [Google Scholar] [CrossRef]
- Calce, R.P.; et al. Voice categorization in the four-month-old human brain. Current Biology 2024, 34, 292–299. [Google Scholar] [CrossRef]
- Campanella, S.; Belin, P. Integrating face and voice in person perception. Trends in Cognitive Sciences 2007, 11, 535–543. [Google Scholar] [CrossRef] [PubMed]
- Charest, I.; Pernet, C.; Latinus, M.; Crabbe, F.; Belin, P. Cerebral processing of voice gender studied using a continuous carryover fMRI design. Cerebral Cortex 2012, 23, 958–966. [Google Scholar] [CrossRef]
- Chelli, D.; Chanoufi, B. Fetal audition: Myth or reality? Journal de Gynécologie Obstétrique et Biologie de la Reproduction 2008, 37, 554–558. [Google Scholar] [CrossRef]
- Chen, L.; Togneri, R.; Maybery, M.; Tan, T. Automated sex classification of children’s voices and changes in differentiating factors with age. arXiv. 2022. Available online: https://arxiv.org/abs/2209.13112.
- Croy, I.; Bojanowski, V.; Hummel, T. Men without a sense of smell exhibit a strongly reduced number of sexual relationships, women exhibit reduced partnership security – a reanalysis of previously published data. Biological Psychology 2013, 92, 292–294. [Google Scholar] [CrossRef] [PubMed]
- DeCasper, A.J.; Fifer, W. Of human bonding: Newborns prefer their mother’s voice. Science 1980, 208, 1174–1176. [Google Scholar] [CrossRef]
- DeCasper, A.J.; Spence, M.J. Prenatal maternal speech influences newborn listening preferences. Infant Behavior and Development 1986, 9, 133–150. [Google Scholar] [CrossRef]
- Dulac, C.; Torello, A.T. Molecular detection of pheromone signals in mammals. Nature Reviews Neuroscience 2003, 4, 551–562. [Google Scholar] [CrossRef]
- Gluckman, M.; Johnson, S. Early developmental differences in voice and face recognition. Developmental Science 2013, 16, 739–748. [Google Scholar]
- Grossmann, T.; Oberecker, R.; Koch, S.P.; Friederici, A.D. The developmental origins of voice processing in the human brain. Neuron 2010, 65, 852–858. [Google Scholar] [CrossRef]
- Herz, R.S.; Inzlicht, M. Sex differences in response to physical and social cues: A review. Hormones and Behavior 2014, 66, 545–560. [Google Scholar] [CrossRef]
- Hillenbrand, J.; Getty, L.A.; Clark, M.J.; Wheeler, K. Acoustic characteristics of American English vowels. Journal of the Acoustical Society of America 97 1995, 3099–3111. [Google Scholar] [CrossRef]
- Hummer, T.A.; McClintock, M.K. Putative human pheromones and their effects on attention. Psychoneuroendocrinology 34 2009, 142–153. [Google Scholar]
- Jacob, S.; et al. Androstadienone modulates women’s mood and cortisol. Journal of Neuroscience 2002, 22, 239–245. [Google Scholar]
- Johnson, S.P.; Dong, M.; Ogren, M.; Senturk, D. Infants’ identification of gender in biological motion displays. Infancy 2021, 26, 687–706. [Google Scholar] [CrossRef]
- Kisilevsky, B.S.; et al. Effects of experience on fetal voice recognition. Psychological Science 2003, 14, 220–224. [Google Scholar] [CrossRef]
- Kisilevsky, B.; et al. Fetal sensitivity to properties of maternal speech and language. Infant Behavior and Development 2009, 32, 59–71. [Google Scholar] [CrossRef]
- Lattner, S.; Meyer, M.E.; Friederici, A.D. Voice perception: Sex, pitch, and the right h Lecanuet, J.P.; Schaal, B. (1996). Fetal sensory competencies. European Journal of Obstetrics & Gynecology and Reproductive Biology;emisphere. Human Brain Mapping 2004, 68 24, 11–20. [Google Scholar] [CrossRef]
- Meredith, M. Human vomeronasal organ: Vestigial or functional? Chemical Senses 2001, 26, 433–445. [Google Scholar] [CrossRef]
- Nowling, D.; et al. Sex differences in development of functional connections in the face processing network. NeuroImage 2024, 283, 120384. [Google Scholar] [CrossRef]
- Oleszkiewicz, A.; et al. Consequences of undetected olfactory loss for human romantic and sexual activity. Archives of Sexual Behavior 2020, 49, 1295–1302. [Google Scholar] [CrossRef]
- Pernet, C.R.; McAleer, P.; Latinus, M.; et al. The human voice areas: Anatomical and functional organization. Cerebral Cortex 25 2015, 4656–4669. [Google Scholar]
- Richoz, A.R.; et al. Audio-visual perception of gender by infants emerges earlier for female than male faces. Frontiers in Psychology 2017, 8, 149. [Google Scholar]
- Salu, Y. The roots of sexual arousal and sexual orientation. Medical Hypotheses 2011, 76, 384–387. [Google Scholar] [CrossRef]
- Salu, Y. Nature controls nurture in the development of sexual orientation, and voice is nature’s agent. Preprints 2020. [Google Scholar] [CrossRef]
- Savic, I.; Berglund, H.; Gulyas, B.; Roland, P. Smelling of odorous sex hormone-like compounds causes sex-differentiated hypothalamic activations. Neuron 2001, 31, 661–668. [Google Scholar] [CrossRef]
- Savic, I.; Lindström, P. PET and MRI show differences in cerebral asymmetry and functional connectivity between homosexual and heterosexual subjects. Proceedings of the National Academy of Sciences of the USA 2008, 105, 9403–9408. [Google Scholar] [CrossRef]
- Saxton, T.K.; Lyndon, A.; Little, A.C.; Roberts, S.C. Evidence for sex pheromones in humans? Proceedings of the Royal Society B 275 2008, 2723–2729. [Google Scholar]
- Sisk, C.L.; Zehr, J.L. Pubertal hormones organize the adolescent brain and behavior. Frontiers in Neuroendocrinology 2005, 26, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Sokhi, D.S.; Hunter, M.D.; Wilkinson, I.D.; Woodruff, P.W.R. Male and female voices activate distinct regions in the male brain. NeuroImage 2005, 27, 572–578. [Google Scholar] [CrossRef] [PubMed]
- Talavage, T.M.; Sereno, M.I.; Melcher, J.R.; Ledden, P.J.; Rosen, B.R.; Dale, A.M. Tonotopic organization in human auditory cortex revealed by progressions of frequency sensitivity. Journal of Neurophysiology 2004, 91, 1282–1296. [Google Scholar] [CrossRef] [PubMed]
- Trotier, D. Vomeronasal organ and human pheromones. European Annals of Otorhinolaryngology, Head and Neck Diseases 2011, 128, 184–190. [Google Scholar] [CrossRef]
- Yu, M.E.; Johnson, T.J.; Fecher, N. Learning to identify talkers: Do 4.5-month-old infants recognize unfamiliar female talkers? JASA Express Letters 2024, 4, 015203. [Google Scholar] [CrossRef]
| Dimension | HSR (Human Voice-Based Sex Recognition) | HPSR (Human Pheromone-Based Sex Recognition) |
| Typical experimental samples | 50–500 participants per study; supplemented by continuous naturalistic exposure across development | 20–60 participants per laboratory study |
| Effective observational scale | Extremely large (lifelong exposure across millions of real-world interactions) | Limited to episodic laboratory exposure |
| Typical classification accuracy | ~90–98% for voice-based sex classification | ~55–60%, often near chance |
| Cohen’s h (effect size) | Large to very large (h ≈ 1.1–1.3), reflecting robust sex classification | Very small (h ≈ 0.05–0.25), reflecting subtle or context-dependent effects |
| Biological mechanisms engaged | Auditory, cortical, subcortical, and developmental learning systems | Chemosensory pathways with unclear developmental primacy in humans |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




