Submitted:
16 December 2025
Posted:
18 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Ethical Approval
2.2. Study Population
2.3. Sampling
2.3.1. Blood Sampling
2.3.2. Cerebrospinal Fluid (CSF)
2.4. Sample Analysis
2.4.1. Serum Sample Analysis
2.4.2. CSF Sample Analysis
2.4.3. Methods
2.5. Statistical Analysis
2.5.1. Serum Samples
2.5.2. CSF Samples
3. Results
3.1. Serum Samples
3.1.1. Serum Oxidative Stress Markers
3.1.2. Cholinesterase
3.1.3. C-Reactive Protein (CRP)
3.2. Cerebrospinal Fluid (CSF) Samples
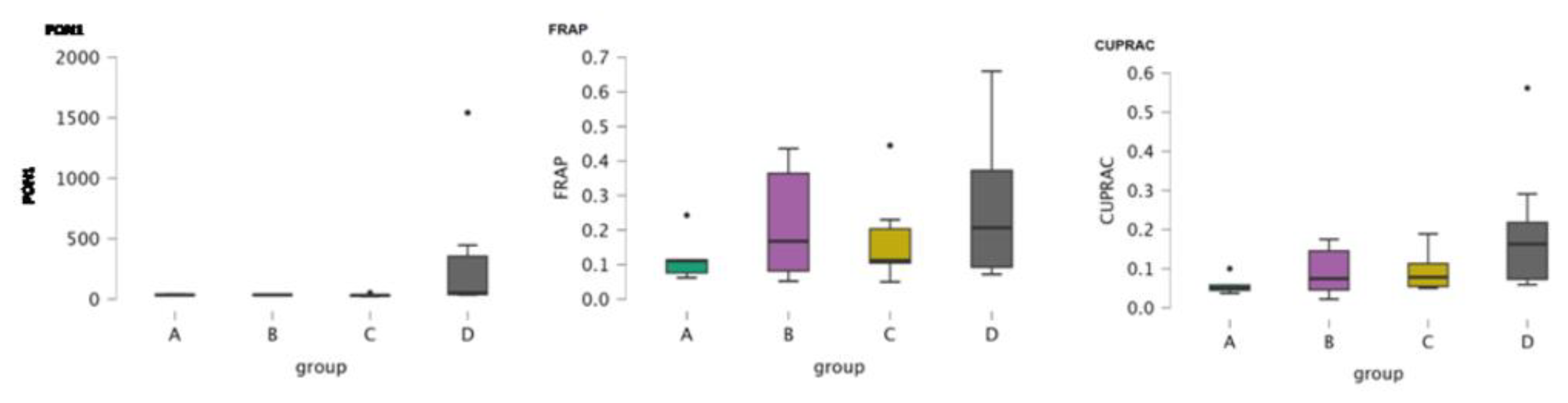
3.2.1. CSF Oxidative Stress Markers
3.2.2. Cholinesterase
3.2.3. Oxytocin
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AChE | Acetylocholine |
| AEM | Antiepileptic Medication |
| ANOVA | Analysis of Variance |
| BBB | Blood Brain Barrier |
| Br | Bromide |
| CBC | Complete Blood Count |
| CNS | Central Nervous System |
| CRP | C-reactive protein |
| CSF | Cerebrospinal Fluid |
| CT | Computed Tomography |
| CVs | Coefficients of variations |
| CUPRAC | Cupric reducing antioxidant capacity |
| EEG | Electroencephalography |
| FRAP | Ferric reducing antioxidant power |
| HDL | High-density lipoprotein |
| ILAE | International League Against Epilepsy |
| LD | Detection limit |
| LEV | Levetiracetam |
| LLQ | Lower limit of quantification |
| MRI | Magnetic Resonance Imaging |
| PB | Phenobarbital |
| PON1 | Paraoxonase 1 |
| RNS | Reactive Nitrogen Species |
| ROS | Reactive Oxygen Species |
References
- Blume, W.T.; Luders, H.O.; Mizrahi, E.; Tassinari, C.; van Emde Boas, W.; Engel, J. Glossary of ictal semiology. Epilepsia 2001, 42, 1212–1218. [Google Scholar] [CrossRef]
- Fisher, R.S.; van Emde Boas, W.; Blume, W.; Elger, C.; Genton, P.; Lee, P.; Engel, J.J. Epileptic seizures and epilepsy: definitions proposed by the International League against Epilepsy (ILAE) and the International Bureau foe Epilepsy (IBE). Epilepsia 2005, 46, 470–472. [Google Scholar] [CrossRef]
- Berendt, M.; Farquhar, R.G.; Mandigers, P.J.J.; Pakozdy, A; Bhatti, S.F.M.; De Risio, L.; Fischer, A.; Long, S.; Matiasek, K.; Munana, K.; Patterson, E.E.; Penderis, J.; Platt, S.; Podell, M.; Potschka, H.; Pumarola, M.B.; Rusbridge, C.; Stein, V.M.; Tipold, A.; Volk, H.A. International Veterinary Task Force Consensus Report on epilepsy definition, classification and terminology in companion animals. Vet. Res. 2015, 182, 1–11. [Google Scholar] [CrossRef]
- Schwartz-Porsche, D. Epidemiological, clinical and pharmacokinetic studies in spontaneously epileptic dogs and cats. Proc. Am. Coll. Vet. Intern. Med. 1986, 1161–1163. [Google Scholar]
- Podell, M.; Hadjiconstantinou, M. Cerebrospinal fluid gamma-aminobutyric acid and glutamate values in dogs with epilepsy. Am. J. Vet. Res. 1997, 58, 451–456. [Google Scholar] [CrossRef]
- Kearsley Fleet, L.O.; Neill, D.G.; Volk, H.A.; Church, D.B.; Broadbelt, D.C. Prevalence and risk factors for canine epilepsy of unknown origin in the UK. Vet. Rec. 2013, 172, 338. [Google Scholar] [CrossRef]
- Asadi-Pooya, A.A.; Malekpour, M.; Taherifard, E.; Mallahzadeh, A.; Farjoud Kouhanjani, M. Coexistence of temporal lobe epilepsy and idiopathic generalized epilepsy. Epilepsy Behav. 2024, 151, 109602. [Google Scholar] [CrossRef] [PubMed]
- Ashjazadeh, N.; Namjoo-Moghadam, A.; Mani, A.; Doostmohammadi, N.; Bayat, M.; Salehi, M.S.; Rafiei, E.; Rostamihosseinkhani, M.; Khani-Robati, A.; Hooshmandi, E. Comparison of executive function in idiopathic generalized epilepsy versus temporal lobe epilepsy. Neurocase. 2024, 30(5), 167–173. [Google Scholar] [CrossRef]
- Steinmetz, S.; Tipold, A.; Loscher, W. Epilepsy after head injury in dogs: a natural model of posttraumatic epilepsy. Epilepsia 2013, 54, 580–588. [Google Scholar] [CrossRef] [PubMed]
- Hall, R.; Labruyere, J.; Volk, H.; Cardy, T.J. Estimation of the prevalence of idiopathic epilepsy and structural epilepsy in a general population of 900 dogs undergoing MRI for epileptic seizures. Vet Rec. 2020, 187(10), e 89. [Google Scholar] [CrossRef]
- De Risio, L.; Bhatti, S.; Munana, K.; Penderis, J.; Stein, V.; Tipold, A.; Brendt, M.; Farqhuar, R.; Fischer, A.; Long, S.; Mandigers, P.J.J.; Matiasek, K.; Packer, R.M.A.; Pakozdy, A.; Patterson, N.; Platt, S.; Podell, M.; Potschka, H.; Batlle, M.P.; Rusbridge, C.; Volk, H.A. International veterinary epilepsy task force consensus proposal: diagnostic approach to epilepsy in dogs. BMC Vet Res 2015, 11, 148. [Google Scholar] [CrossRef] [PubMed]
- Estey, C.M.; Dewey, C.W.; Rishniw, M.; Lin, D.M.; Bouma, J.; Sackman, J. A subset of dogs with presumptive idiopathic epilepsy show hippocampal asymmetry: a volumetric comparison with non-epileptic dogs using MRI. Front. Vet. Sci. 2017, 4, 183. [Google Scholar] [CrossRef] [PubMed]
- Czerwik, A.; Plonek, M.; Podgσrski, P.; Wrzosek, M. Comparison of electroencephalographic findings with hippocampal magnetic resonance imaging volumetry in dogs with idiopathic epilepsy. J.Vet.Intern.Med. 2018, 32, 2037–2044. [Google Scholar] [CrossRef]
- Huaijantug, S.; Yatmark, P.; Chinnabrut, P.; Rueangsawat, N.; Wongkumlue, A.; Teerapan, W. Quantitative brain histogram of canine epilepsy using magnetic resonance imaging. Acta Radiol. 2021, 62, 93–101. [Google Scholar] [CrossRef]
- Nagendran, A.; McConnell, J.F.; De Risio, L.; Jose-Lopez, R.; Quintana, R.G.; Robinson, K.; Platt, S.R.; Masian, D.S.; Maddox, T.; Concalves, R. Peri-ictal magnetic resonance imaging characteristics in dogs with suspected idiopathic epilepsy. J.Vet Intern.Med. 2021, 35(2), 1008–1017. [Google Scholar] [CrossRef] [PubMed]
- Maeso, C.; Sanchez-Masian, D.; Rodenas, S.; Font, C.; Morales, C.; Domνnguez, E. Prevalence, distribution, and clinical associations of suspected postictal changes on brain magnetic resonance imaging in epileptic dogs. J.Am.Vet.Med. Assoc. 2021, 260, 71–81. [Google Scholar] [CrossRef]
- Wirrell, E.C.; Nabbout, R.; Scheffer, I.E.; Alsaadi, T.; Bogacz, A.; French, J.A. Methodology for classification and definition of epilepsy syndromes with list of syndromes: report of the ILAE Task Force on Nosology and Definitions. Epilepsia 2022, 63(6), 1333–1348. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wu, J.; Zeng, Y.; Zheng, W. Neuroinflammation in epileptogenesis: from pathophysiology to therapeutic strategies. Front Immunol. 2023, 14, 1269241. [Google Scholar] [CrossRef]
- Zhao, P.; Ding, X.; Li, L.; Jiang, G. A review of cell-type specific circuit mechanisms underlying epilepsy. Acta Epileptol. 2024, 6(1), 18. [Google Scholar] [CrossRef]
- Birben, E.; Sahiner, U.M.; Sackesen, C.; Erzurum, S.; Kalayci, O. Oxidative stress and antioxidant defense. World Allergy Organ J. 2012, 5(1), 9–19. [Google Scholar] [CrossRef]
- Sies, H. Oxidative Stress: Concept and Some Practical Aspects. Antioxidants (Basel) 2020, 9(9), 852. [Google Scholar] [CrossRef]
- Łukawski, K.; Czuczwar, S.J. Oxidative Stress and Neurodegeneration in Animal Models of Seizures and Epilepsy. Antioxidants (Basel) 2023, 12(5), 1049. [Google Scholar] [CrossRef]
- Rumià, J.; Marmol, F.; Sanchez, J.; Giménez-Crouseilles, J.; Carreño, M.; Bargalló, N.; Boget, T.; Pintor, L.; Setoain, X.; Donaire, A.; Saez, G.T.; Ribalta, T.; Ferrer, E.; Puig-Parellada, P. Oxidative stress markers in the neocortex of drug-resistant epilepsy patients submitted to epilepsy surgery. Epilepsy Res. 2013, 107(1-2), 75–81. [Google Scholar] [CrossRef]
- Işık, M.; Demir, Y.; Kırıcı, M.; Demir, R.; Şimşek, F.; Beydemir, Ş. Changes in the anti-oxidant system in adult epilepsy patients receiving anti-epileptic drugs. Arch Physiol Biochem. 2015, 121(3), 97–102. [Google Scholar] [CrossRef] [PubMed]
- Dönmezdil, N.; Çevik, M.U.; Özdemir, H.H.; Taşin, M. Investigation of PON1 activity and MDA levels in patients with epilepsy not receiving antiepileptic treatment. Neuropsychiatr Dis Treat. 2016, 12, 1013–1017. [Google Scholar] [CrossRef] [PubMed]
- Petrillo, S.; Pietrafusa, N.; Trivisano, M.; Calabrese, C.; Saura, F.; Gallo, M.G.; Bertini, E.S.; Vigevano, F.; Specchio, N.; Piemonte, F. Imbalance of Systemic Redox Biomarkers in Children with Epilepsy: Role of Ferroptosis. Antioxidants (Basel). 2021, 10(8), 1267. [Google Scholar] [CrossRef]
- Michelin, A.P.; Maes, M.H.J.; Supasitthumrong, T.; Limotai, C.; Matsumoto, A.K.; de Oliveira Semeão, L.; de Lima Pedrão, J.V.; Moreira, E.G.; Kanchanatawan, B.; Barbosa, D.S. Reduced paraoxonase 1 activities may explain the comorbidities between temporal lobe epilepsy and depression, anxiety and psychosis. World J Psychiatry. 2022, 12(2), 308–322. [Google Scholar] [CrossRef] [PubMed]
- Korczowska-Łącka, I.; Hurła, M.; Banaszek, N.; Kobylarek, D.; Szymanowicz, O.; Kozubski, W.; Dorszewska, J. Selected Biomarkers of Oxidative Stress and Energy Metabolism Disorders in Neurological Diseases. Mol Neurobiol. 2023, 60(7), 4132–4149. [Google Scholar] [CrossRef]
- Langenbruch, L.; Wiendl, H.; Groß, C.; Kovac, S. Diagnostic utility of cerebrospinal fluid (CSF) findings in seizures and epilepsy with and without autoimmune-associated disease. Seizure. 2021, 91, 233–243. [Google Scholar] [CrossRef]
- Fabisiak, T.; Patel, M. Crosstalk between neuroinflammation and oxidative stress in epilepsy. Front Cell Dev Biol. 2022, 10, 976953. [Google Scholar] [CrossRef]
- Parsons, A.L.M.; Bucknor, E.M.V.; Castroflorio, E.; Soares, T.R.; Oliver, P.L.; Rial, D. The Interconnected Mechanisms of Oxidative Stress and Neuroinflammation in Epilepsy. Antioxidants (Basel). 2022, 11(1), 157. [Google Scholar] [CrossRef]
- Yuen, A.W.; Bell, G.S.; Peacock, J.L.; Koepp, M.M.; Patsalos, P.N.; Sander, J.W. Effects of AEDs on biomarkers in people with epilepsy: CRP, HbA1c and eGFR. Epilepsy Res. 2010, 91(2-3), 187–192. [Google Scholar] [CrossRef]
- Alapirtti, T.; Waris, M.; Fallah, M.; Soilu-Hänninen, M.; Mäkinen, R.; Kharazmi, E.; Peltola, J. C-reactive protein and seizures in focal epilepsy: a video-electroencephalographic study. Epilepsia. 2012, 53(5), 790–796. [Google Scholar] [CrossRef] [PubMed]
- Holtman, L.; van Vliet, E.A.; Aronica, E.; Wouters, D.; Wadman, W.J.; Gorter, J.A. Blood plasma inflammation markers during epileptogenesis in post-status epilepticus rat model for temporal lobe epilepsy. Epilepsia. 2013, 54(4), 589–595. [Google Scholar] [CrossRef]
- Chen, W.; Tan, Y.; Ge, Y.; Chen, Y.; Liu, X. The Effects of Levetiracetam on Cerebrospinal Fluid and Plasma NPY and GAL, and on the Components of Stress Response System, hs-CRP, and S100B Protein in Serum of Patients with Refractory Epilepsy. Cell Biochem Biophys. 2015, 73(2), 489–494. [Google Scholar] [CrossRef]
- Hermann, B.P.; Sager, M.A.; Koscik, R.L.; Young, K.; Nakamura, K. Vascular, inflammatory, and metabolic factors associated with cognition in aging persons with chronic epilepsy. Epilepsia. 2017, 58(11), e152–e156. [Google Scholar] [CrossRef]
- Zhou, T.; Wang, N.; Xu, L.; Huang, H.; Yu, C.; Zhou, H. Effects of carbamazepine combined with vitamin B12 on levels of plasma homocysteine, hs-CRP and TNF-α in patients with epilepsy. Exp Ther Med. 2018, 15(3), 2327–2332. [Google Scholar] [CrossRef] [PubMed]
- Zhong, R.; Chen, Q.; Li, M.; Zhang, X.; Lin, W. Elevated Blood C-Reactive Protein Levels in Patients With Epilepsy: A Systematic Review and Meta-Analysis. Front Neurol. 2019, 10, 974. [Google Scholar] [CrossRef]
- Faria, M.T.; Rego, R.; Rocha, H.; Sá, F.; Farinha, R.; Oliveira, A.; Barata, P.; Alves, D.; Pereira, J.; Rocha-Gonçalves, F.; Gonçalves, H.; Martins, E. cTnI, BNP and CRP profiling after seizures in patients with drug-resistant epilepsy. Seizure. 2020, 80, 100–108. [Google Scholar] [CrossRef]
- Liu, Z.; Li, J.; Yang, F.; Hu, Y.; Liu, J.; Hu, H.; Su, W. Sodium valproate combined with levetiracetam in pediatric epilepsy and its influence on NSE, IL-6, hs-CRP and electroencephalogram improvement. Exp Ther Med. 2020, 20(3), 2043–2048. [Google Scholar] [CrossRef] [PubMed]
- Tao, H.; Gong, Y.; Yu, Q.; Zhou, H.; Liu, Y. Elevated Serum Matrix Metalloproteinase-9, Interleukin-6, Hypersensitive C-Reactive Protein, and Homocysteine Levels in Patients with Epilepsy. J Interferon Cytokine Res. 2020, 40(3), 152–158. [Google Scholar] [CrossRef]
- Wei, H.; Liu, D.; Geng, L.; Liu, Y.; Wang, H.; Yan, F. Application Value of Serum Metabolic Markers for Cognitive Prediction in Elderly Epilepsy. Neuropsychiatr Dis Treat. 2022, 18, 2133–2140. [Google Scholar] [CrossRef]
- Zhou, Y.F.; Huang, Y.; Liu, G.H. Effects of Levetiracetam on the Serum C - reactive protein in Children With Epilepsy: A Meta-Analysis. Front Pharmacol. 2022, 13, 810617. [Google Scholar] [CrossRef]
- Mahon, E.K.; Williams, T.L.; Alves, L. Serum C-reactive protein concentrations in dogs with structural and idiopathic epilepsy. Vet Rec. 2023, 193(12), e3211. [Google Scholar] [CrossRef] [PubMed]
- Despa, A.; Musteata, M.; Solcan, G. Evaluation of Blood C Reactive Protein (CRP) and Neutrophil-to-Lymphocyte Ratio (NLR) Utility in Canine Epilepsy. Vet Sci. 2024, 11(9), 408. [Google Scholar] [CrossRef]
- Kocatürk, M.; Öz, A.D.; Muñoz, A.; Martinez, J.D.; Ceron, J.J.; Yilmaz, Z. Changes in immuno-inflammatory and antioxidant biomarkers in serum and cerebrospinal fluid of dogs with distemper. Microb Pathog. 2025, 198, 107160. [Google Scholar] [CrossRef]
- Sergienko, N.G.; Gonzalez-Quevedo, A.; Gonzalez, N.; Simon y Canon, L.; Marin, G. Role of the acetylocholine—cholinesterase system in the development of epilepsy. ZhNevropatolPsikhiatrIm S S Korsakova 1979, 6, 698–704. [Google Scholar]
- Zimmerman, G.; Njunting, M.; Ivens, S.; Tolner, E.A.; Behrens, C.J.; Gross, M.; Soreq, H.; Heinemann, U.; Friedman, A. Acetylocholin-induced seizure-like activity and modified cholinergic expression in chronically epileptic rats. Eur J Neurosci 2008, 27(4), 965–975. [Google Scholar] [CrossRef] [PubMed]
- Erfanparast, A.; Tamaddonfard, E.; Henareh-Chareh, F. Intra-hippocampal microinjection of oxytocin produced antiepileptic effect on the pentylenetetrazol-induced epilepsy in rats. Pharmacol Rep. 2017, 69(4), 757–763. [Google Scholar] [CrossRef]
- Higashida, H.; Munesue, T.; Kosaka, H.; Yamasue, H.; Yokoyama, S.; Kikuchi, M. Social Interaction Improved by Oxytocin in the Subclass of Autism with Comorbid Intellectual Disabilities. Diseases. 2019, 7(1), 24. [Google Scholar] [CrossRef] [PubMed]
- Wong, J.C.; Shapiro, L.; Thelin, J.T.; Heaton, E.C.; Zaman, R.U.; D'Souza, M.J.; Murnane, K.S.; Escayg, A. Nanoparticle encapsulated oxytocin increases resistance to induced seizures and restores social behavior in Scn1a-derived epilepsy. Neurobiol Dis. 2021, 147, 105147. [Google Scholar] [CrossRef]
- Sahin, H.; Yucel, O.; Emik, S.; Senturk, G.E. Protective Effects of Intranasally Administrated Oxytocin-Loaded Nanoparticles on Pentylenetetrazole-Kindling Epilepsy in Terms of Seizure Severity, Memory, Neurogenesis, and Neuronal Damage. ACS Chem Neurosci. 2022, 13(13), 1923–1937. [Google Scholar] [CrossRef]
- Chen, W.; Man, X.; Zhang, Y.; Yao, G.; Chen, J. Medial prefrontal cortex oxytocin mitigates epilepsy and cognitive impairments induced by traumatic brain injury through reducing neuroinflammation in mice. Sci Rep. 2023, 13(1), 5214. [Google Scholar] [CrossRef]
- De Risio, L.; Platt, S.; De Risio, L.; Platt, S. Idiopathic Epilepsy and Genetics. In BookCanine and Feline Epilepsy: Diagnosis and management; Cabi: Oxfordshire, 2014; pp. 207–218. [Google Scholar]
- Bhatti, F.M.; De Risio, L.; Muñana, K.; Penderis, J.; Stein, V.M.; Tipold, A.; Berendt, M.; Farquhar, R.G.; Fischer, A.; Long, S.; Löscher, W.; Mandigers, P.J.J.; Matiasek, K.; Pakozdy, A.; Patterson, E.E.; Platt, S.; Podell, M.; Potschka, H.; Rusbridge, C.; Volk, H.A. International Veterinary Epilepsy Task Force consensus proposal: medical treatment of canine epilepsy in Europe. Vet Res 2015, 11, 176. [Google Scholar] [CrossRef]
- Charalambous, M.; Shivapour, S.K.; Brodbelt, D.C.; Volk, H.A. Antiepileptic drugs' tolerability and safety--a systematic review and meta-analysis of adverse effects in dogs. BMC Vet Res. 2016, 12, 79. [Google Scholar] [CrossRef]
- Ceron, J.J.; Tecles, F.; Tvarijonaviciute, A. Serum paraoxonase 1 (PON1) measurement: An update. BMC Vet. Res. 2014, 10, 74. [Google Scholar] [CrossRef] [PubMed]
- Rubio, C.P.; Tvarijonaviciute, A.; Martinez-Subiela, S.; Hernández-Ruiz, J.; Cerón, J.J. Validation of an automated assay for the measurement of cupric reducing antioxidant capacity in serum of dogs. BMC Vet. Res. 2016, 12, 137. [Google Scholar] [CrossRef] [PubMed]
- Benzie, I.F.F.; Strain, J.J. The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
- Rubio, C.P.; Martinez-Subiela, S.; Hernández-Ruiz, J.; Tvarijonaviciute, A.; Ceron, J.J. Analytical validation of an automated assay for ferric-reducing ability of plasma in dog serum. J. Vet. Diagnostic Investig. 2017, 29, 574–578. [Google Scholar] [CrossRef] [PubMed]
- Ginoudis, A.; Pardali, D.; Mylonakis, M.E.; Tamvakis, A.; Tvarijonaviciute, A.; Lymperaki, E.; Ceron, J.J.; Polizopoulou, Z. Oxidative Status and Lipid Metabolism Analytes in Dogs with Mast Cell Tumors: A Preliminary Study. Antioxidants 2024, 13, 1473. [Google Scholar] [CrossRef]
- Muñoz-Prieto, A.; Tvarijonaviciute, A.; Escribano, D.; Martínez-Subiela, S.; Cerón, J.J. Use of heterologous immunoassays for quantification of serum proteins: The case of canine C-reactive protein. PLoS One. 2017, 12(2), e0172188. [Google Scholar] [CrossRef]
- López-Arjona, M.; Mateo, S.V.; Manteca, X.; Escribano, D.; Cerón, J.J.; Martínez-Subiela, S. Oxytocin in saliva of pigs: an assay for its measurement and changes after farrowing. Domest Anim Endocrinol 2020, 70, 106384. [Google Scholar] [CrossRef]
- Simoncini, C.; Torri, S.; Montano, V.; Chico, L.; Gruosso, F.; Tuttolomondo, A.; Pinto, A.; Simonetta, I.; Cianci, V.; Salviati, A.; Vicenzi, V.; Marchi, G.; Girelli, D.; Concolino, D.; Sestito, S.; Zedde, M.; Siciliano, G.; Mancuso, M. Oxidative stress biomarkers in Fabry disease: is there a room for them? J Neurol. 2020, 267(12), 3741–3752. [Google Scholar] [CrossRef]
- Radaković, M.; Andrić, J.F.; Spariosu, K.; Vejnović, B.; Filipović, M.K.; Andrić, N. Serum oxidant-antioxidant status and butyrylcholinesterase activity in dogs with idiopathic epilepsy - A pilot study. Res Vet Sci. 2023, 165, 105076. [Google Scholar] [CrossRef]
- Aviram, M.; Rosenblat, M. Paraoxonases and cardiovascular diseases: pharmacological and nutritional influences. Curr Opin Lipidol. 2005, 16(4), 393–399. [Google Scholar] [CrossRef] [PubMed]
- Costa, L.G.; Furlong, C.E. Paraoxonase (PON1): genetics, enzymatic activity, and role in detoxificationa and oxidative stress. Annual Review of Pharmacology and Toxicology 2014, 54, 371–396. [Google Scholar]
- Beggiato, S.; Ferrara, F.; Romani, A.; Cassano, T.; Trentini, A.; Valacchi, G.; Cervellati, C.; Ferraro, L. Signature of paraoxonases in the altered redox homeostasis in Alzheimer's disease. Chem Biol Interact. 2024, 388, 110839. [Google Scholar] [CrossRef]
- Romani, A.; Trentini, A.; Flier, W.M.V.; Bellini, T.; Zuliani, G.; Cervellati, C.; Teunissen, C.E. Arylesterase Activity of Paraoxonase-1 in Serum and Cerebrospinal Fluid of Patients with Alzheimer's Disease and Vascular Dementia. Antioxidants (Basel) 2020, 9(5), 456. [Google Scholar] [CrossRef] [PubMed]
- Marsillach, J.; Cervellati, C. Paraoxonase-1 and Other HDL Accessory Proteins in Neurological Diseases. Antioxidants (Basel). 2021, 10(3), 454. [Google Scholar] [CrossRef]
- Karami, A.; Eriksdotter, M.; Kadir, A.; Almkvist, O.; Nordberg, A.; Darreh-Shori, T. CSF Cholinergic Index, a New Biomeasure of Treatment Effect in Patients With Alzheimer's Disease. Front Mol Neurosci. 2019, 12, 239. [Google Scholar] [CrossRef]
- Sailaja, B.S.; Cohen-Carmon, D.; Zimmerman, G.; Soreq, H.; Meshorer, E. Stress-induced epigenetic transcriptional memory of acetylcholinesterase by HDAC4. ProcNatl Acad Sci U S A 2012, 109(52), E3687–E3695. [Google Scholar] [CrossRef]
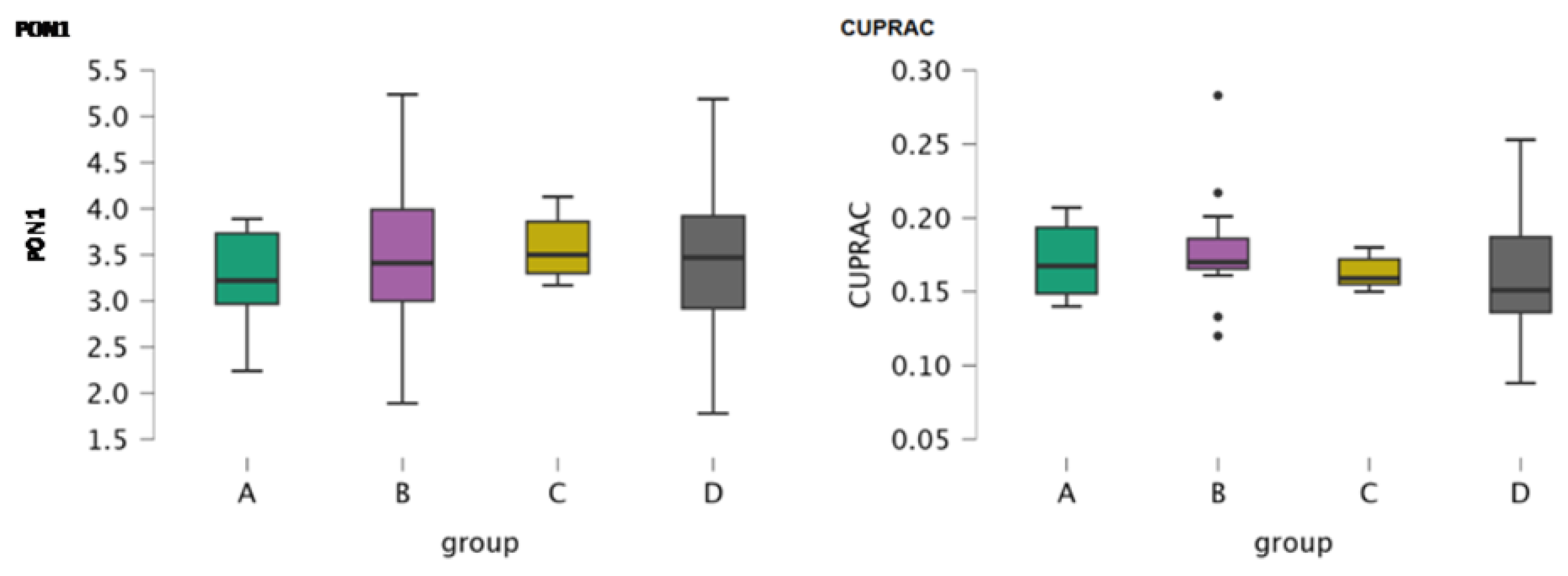
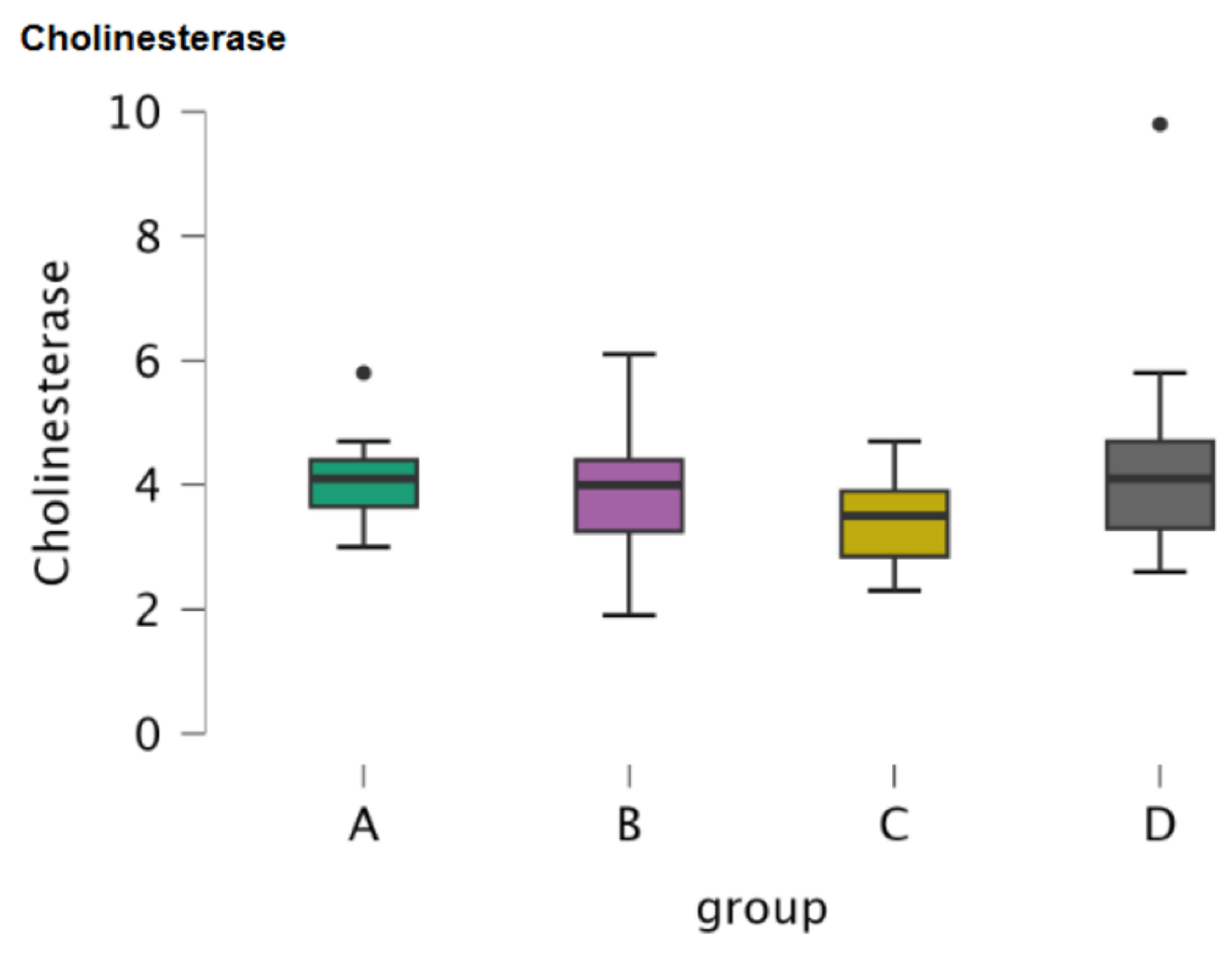
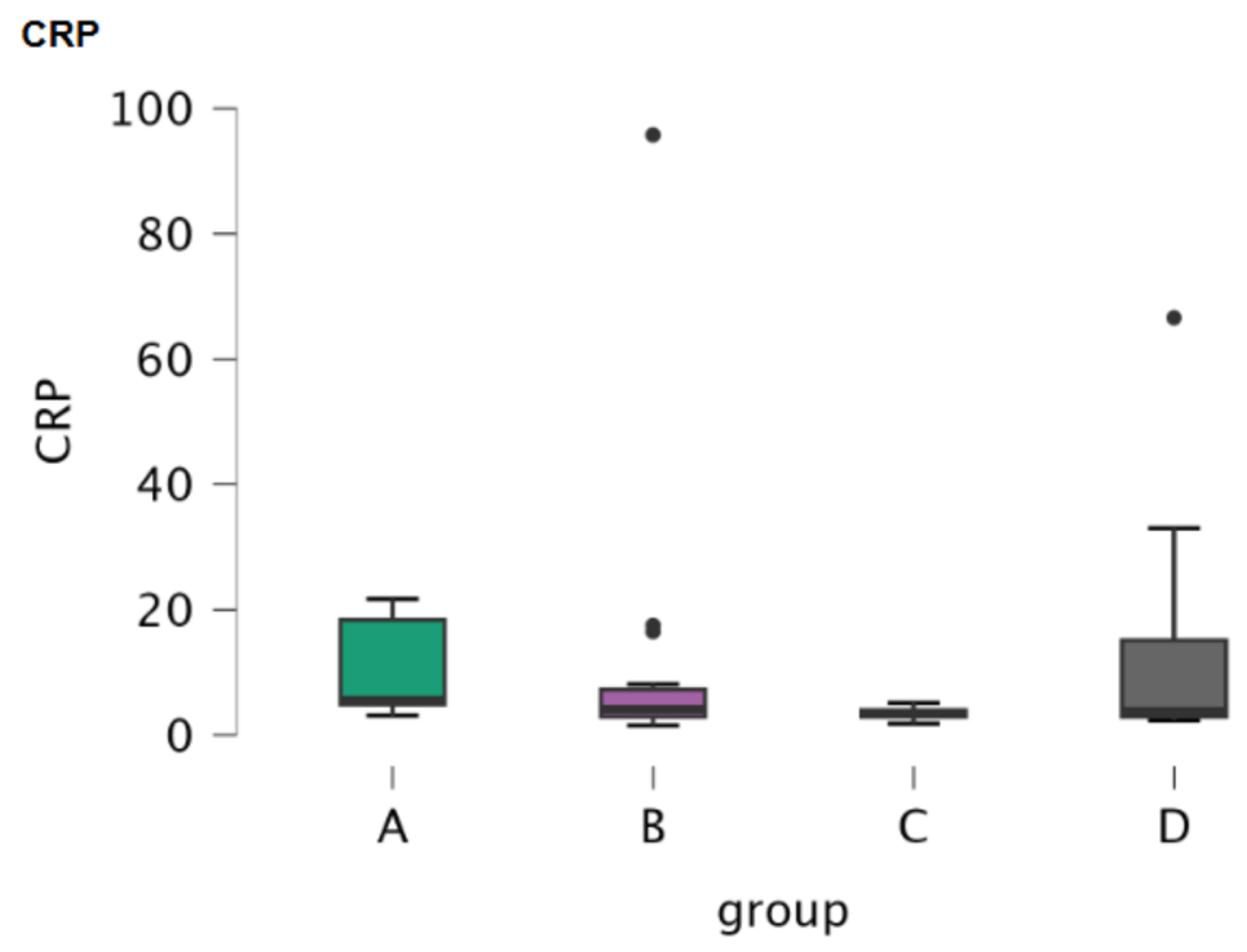

| Parameters | PON1 | CUPRAC | Cholinesterase | CRP | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Groups | A | B | C | D | A | B | C | D | A | B | C | D | A | B | C | D | |
| Valid | 8 | 15 | 11 | 17 | 8 | 15 | 11 | 17 | 8 | 15 | 11 | 17 | 8 | 15 | 11 | 17 | |
| Missing | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Median | 3.220 | 3.410 | 3.500 | 3.470 | 0.167 | 0.170 | 0.159 | 0.151 | 4.100 | 4.000 | 3.500 | 4.100 | 5.550 | 4.100 | 3.400 | 3.800 | |
| Mean | 3.231 | 3.557 | 3.586 | 3.400 | 0.171 | 0.179 | 0.163 | 0.164 | 4.125 | 3.827 | 3.436 | 4.306 | 10.338 | 11.860 | 3.464 | 11.629 | |
| Std. Deviation | 0.583 | 0.937 | 0.337 | 0.867 | 0.027 | 0.037 | 0.010 | 0.047 | 0.880 | 1.065 | 0.757 | 1.682 | 7.773 | 23.722 | 0.904 | 16.521 | |
| 95% CI Std. Dev. Upper | 1.186 | 1.478 | 0.592 | 1.320 | 0.056 | 0.059 | 0.018 | 0.071 | 1.790 | 1.680 | 1.328 | 2.561 | 15.820 | 37.412 | 1.586 | 25.143 | |
| 95% CI Std. Dev. Lower | 0.385 | 0.686 | 0.236 | 0.646 | 0.018 | 0.027 | 0.007 | 0.035 | 0.582 | 0.780 | 0.529 | 1.253 | 5.139 | 17.368 | 0.631 | 12.304 | |
| Skewness | -0.513 | 0.339 | 0.267 | -0.013 | 0.160 | 1.395 | 0.473 | 0.674 | 0.740 | 0.094 | 0.163 | 2.290 | 0.668 | 3.612 | 0.039 | 2.678 | |
| Std. Error of Skewness | 0.752 | 0.580 | 0.661 | 0.550 | 0.752 | 0.580 | 0.661 | 0.550 | 0.752 | 0.580 | 0.661 | 0.550 | 0.752 | 0.580 | 0.661 | 0.550 | |
| Kurtosis | -0.558 | -0.272 | -1.479 | -0.059 | -2.208 | 3.976 | -1.230 | -0.271 | 1.017 | 0.407 | -0.847 | 7.039 | -1.941 | 13.478 | 0.288 | 7.838 | |
| Std. Error of Kurtosis | 1.481 | 1.121 | 1.279 | 1.063 | 1.481 | 1.121 | 1.279 | 1.063 | 1.481 | 1.121 | 1.279 | 1.063 | 1.481 | 1.121 | 1.279 | 1.063 | |
| Shapiro-Wilk | 0.935 | 0.958 | 0.924 | 0.979 | 0.864 | 0.868 | 0.918 | 0.925 | 0.949 | 0.971 | 0.967 | 0.778 | 0.773 | 0.438 | 0.979 | 0.613 | |
| P-value of Shapiro-Wilk | 0.561 | 0.657 | 0.351 | 0.950 | 0.131 | 0.032 | 0.303 | 0.181 | 0.706 | 0.872 | 0.860 | 0.001 | 0.015 | 1.150×10-6 | 0.961 | 1.386×10-5 | |
| Minimum | 2.240 | 1.890 | 3.170 | 1.780 | 0.140 | 0.120 | 0.150 | 0.088 | 3.000 | 1.900 | 2.300 | 2.600 | 3.100 | 1.500 | 1.800 | 2.300 | |
| Maximum | 3.890 | 5.240 | 4.130 | 5.190 | 0.207 | 0.283 | 0.180 | 0.253 | 5.800 | 6.100 | 4.700 | 9.800 | 21.700 | 95.800 | 5.100 | 66.600 | |
| Group comparisons | Mean Difference | SE | df | t | pturkey | |
|---|---|---|---|---|---|---|
| PON1 | ||||||
| A | B | -0.326 | 0.337 | 47 | -0.968 | 0.768 |
| C | -0.355 | 0.358 | 47 | -0.993 | 0.754 | |
| D | -0.169 | 0.330 | 47 | -0.511 | 0.956 | |
| B | C | -0.029 | 0.306 | 47 | -0.095 | 1.000 |
| D | 0.157 | 0.273 | 47 | 0.577 | 0.938 | |
| C | D | 0.186 | 0.298 | 47 | 0.626 | 0.923 |
| CUPRAC | ||||||
| A | B | -0.008 | 0.016 | 47 | -0.496 | 0.960 |
| C | 0.008 | 0.017 | 47 | 0.492 | 0.960 | |
| D | 0.008 | 0.015 | 47 | 0.489 | 0.961 | |
| B | C | 0.016 | 0.014 | 47 | 1.123 | 0.677 |
| D | 0.015 | 0.013 | 47 | 1.205 | 0.627 | |
| C | D | -6.791×10-4 | 0.014 | 47 | -0.049 | 1.000 |
| Cholinesterase | ||||||
| A | B | 0.298 | 0.543 | 47 | 0.549 | 0.946 |
| C | 0.689 | 0.576 | 47 | 1.195 | 0.633 | |
| D | -0.181 | 0.532 | 47 | -0.340 | 0.986 | |
| B | C | 0.390 | 0.492 | 47 | 0.793 | 0.857 |
| D | -0.479 | 0.439 | 47 | -1.091 | 0.697 | |
| C | D | -0.870 | 0.480 | 47 | -1.812 | 0.281 |
| CRP | ||||||
| A | B | -0.326 | 0.337 | 47 | -0.968 | 0.768 |
| C | -0.355 | 0.358 | 47 | -0.993 | 0.754 | |
| D | -0.169 | 0.330 | 47 | -0.511 | 0.956 | |
| B | C | -0.029 | 0.306 | 47 | -0.095 | 1.000 |
| D | 0.157 | 0.273 | 47 | 0.577 | 0.938 | |
| C | D | 0.186 | 0.298 | 47 | 0.626 | 0.923 |
| PON1 | CUPRAC | Cholinesterase | CRP | ||
|---|---|---|---|---|---|
| Factor | group | group | group | group | |
| Statistic | 1.700 | 3.120 | 3.294 | 6.648 | |
| dF | 3 | 3 | 3 | 3 | |
| P | 0.637 | 0.374 | 0.348 | 0.084 | |
| Rank ε2 | 0.034 | 0.062 | 0.066 | 0.133 | |
| 95% CI for Rank ε2 | Lower | 0.009 | 0.010 | 0.017 | 0.059 |
| Upper | 0.272 | 0.358 | 0.299 | 0.305 | |
| Rank η2 | 0.000 | 0.003 | 0.006 | 0.078 | |
| 95% CI for Rank η2 | Lower | 0.000 | 0.000 | 0.000 | 0.016 |
| Upper | 0.174 | 0.295 | 0.229 | 0.296 |
| Comparisons | z | Wi | Wj | rrb | p | pbonf | pholm |
|---|---|---|---|---|---|---|---|
| PON1 | |||||||
| A - B | -0.809 | 20.938 | 26.200 | 0.167 | 0.419 | 1.000 | 1.000 |
| A - C | -1.299 | 20.938 | 29.909 | 0.409 | 0.194 | 1.000 | 1.000 |
| A - D | -0.744 | 20.938 | 25.676 | 0.184 | 0.457 | 1.000 | 1.000 |
| B - C | -0.629 | 26.200 | 29.909 | 0.152 | 0.530 | 1.000 | 1.000 |
| B - D | 0.099 | 26.200 | 25.676 | 0.043 | 0.921 | 1.000 | 1.000 |
| C - D | 0.736 | 29.909 | 25.676 | 0.134 | 0.462 | 1.000 | 1.000 |
| CUPRAC | |||||||
| A - B | -0.595 | 27.063 | 30.933 | 0.083 | 0.552 | 1.000 | 1.000 |
| A - C | 0.292 | 27.063 | 25.045 | 0.034 | 0.770 | 1.000 | 1.000 |
| A - D | 0.831 | 27.063 | 21.765 | 0.176 | 0.406 | 1.000 | 1.000 |
| B - C | 0.998 | 30.933 | 25.045 | 0.406 | 0.318 | 1.000 | 1.000 |
| B - D | 1.742 | 30.933 | 21.765 | 0.278 | 0.082 | 0.490 | 0.490 |
| C - D | 0.571 | 25.045 | 21.765 | 0.262 | 0.568 | 1.000 | 1.000 |
| Cholinesterase | |||||||
| A - B | 0.524 | 29.438 | 26.033 | 0.092 | 0.601 | 1.000 | 1.000 |
| A - C | 1.480 | 29.438 | 19.227 | 0.432 | 0.139 | 0.833 | 0.695 |
| A - D | 0.110 | 29.438 | 28.735 | 0.044 | 0.912 | 1.000 | 1.000 |
| B - C | 1.155 | 26.033 | 19.227 | 0.255 | 0.248 | 1.000 | 0.993 |
| B - D | -0.514 | 26.033 | 28.735 | 0.118 | 0.607 | 1.000 | 1.000 |
| C - D | -1.655 | 19.227 | 28.735 | 0.369 | 0.098 | 0.588 | 0.588 |
| CRP | |||||||
| A - B | -0.809 | 20.938 | 26.200 | 0.167 | 0.419 | 1.000 | 1.000 |
| A - C | -1.299 | 20.938 | 29.909 | 0.409 | 0.194 | 1.000 | 1.000 |
| A - D | -0.744 | 20.938 | 25.676 | 0.184 | 0.457 | 1.000 | 1.000 |
| B - C | -0.629 | 26.200 | 29.909 | 0.152 | 0.530 | 1.000 | 1.000 |
| B - D | 0.099 | 26.200 | 25.676 | 0.043 | 0.921 | 1.000 | 1.000 |
| C - D | 0.736 | 29.909 | 25.676 | 0.134 | 0.462 | 1.000 | 1.000 |
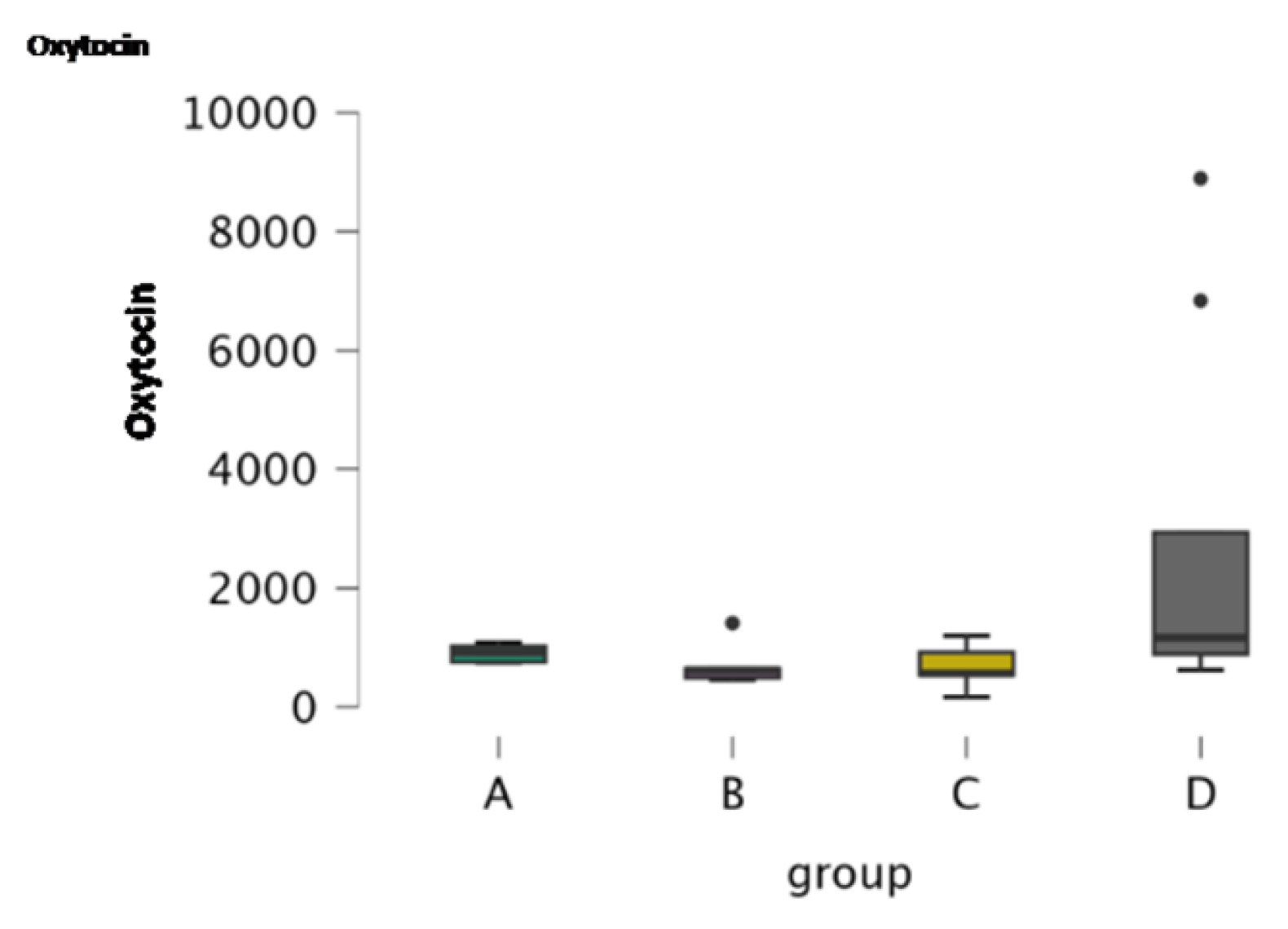
| Parameters | PON1 | FRAP | Cholinestrase | CUPRAC | Oxytocin | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Groups | A | B | C | D | A | B | C | D | A | B | C | D | A | B | C | D | A | B | C | D |
| Valid | 5 | 4 | 5 | 7 | 5 | 6 | 7 | 8 | 5 | 4 | 6 | 7 | 5 | 6 | 7 | 8 | 5 | 6 | 7 | 8 |
| Missing | 0 | 2 | 2 | 1 | 0 | 0 | 0 | 0 | 0 | 2 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Median | 34.100 | 34.800 | 31.000 | 51.000 | 0.111 | 0.167 | 0.112 | 0.206 | 58.000 | 65.600 | 77.550 | 152.700 | 0.050 | 0.074 | 0.078 | 0.163 | 912.920 | 615.285 | 561.190 | 1161.220 |
| Mean | 34.920 | 34.450 | 31.860 | 345.014 | 0.121 | 0.218 | 0.175 | 0.263 | 61.280 | 72.150 | 79.783 | 368.671 | 0.058 | 0.092 | 0.093 | 0.197 | 902.374 | 698.715 | 688.223 | 2746.130 |
| Std. Deviation | 5.186 | 3.580 | 12.054 | 551.093 | 0.072 | 0.171 | 0.133 | 0.208 | 13.030 | 20.756 | 37.847 | 454.231 | 0.025 | 0.064 | 0.052 | 0.167 | 151.306 | 358.166 | 343.063 | 3223.943 |
| Skewness | 0.931 | -0.549 | 1.502 | 2.238 | 1.705 | 0.492 | 1.704 | 1.008 | 0.131 | 1.431 | 0.274 | 2.038 | 1.720 | 0.467 | 1.261 | 1.760 | 0.023 | 2.142 | 0.017 | 1.521 |
| Std. Error of Skewness | 0.913 | 1.014 | 0.913 | 0.794 | 0.913 | 0.845 | 0.794 | 0.752 | 0.913 | 1.014 | 0.845 | 0.794 | 0.913 | 0.845 | 0.794 | 0.752 | 0.913 | 0.845 | 0.794 | 0.752 |
| Kurtosis | 0.139 | 0.952 | 2.565 | 5.186 | 3.215 | -2.187 | 3.133 | 0.387 | -0.393 | 1.739 | -1.070 | 4.165 | 3.235 | -1.954 | 0.740 | 3.379 | -2.550 | 4.890 | -0.364 | 0.726 |
| Std. Error of Kurtosis | 2.000 | 2.619 | 2.000 | 1.587 | 2.000 | 1.741 | 1.587 | 1.481 | 2.000 | 2.619 | 1.741 | 1.587 | 2.000 | 1.741 | 1.587 | 1.481 | 2.000 | 1.741 | 1.587 | 1.481 |
| Shapiro-Wilk | 0.902 | 0.982 | 0.851 | 0.658 | 0.816 | 0.851 | 0.826 | 0.866 | 0.978 | 0.867 | 0.961 | 0.709 | 0.831 | 0.892 | 0.846 | 0.805 | 0.902 | 0.700 | 0.964 | 0.690 |
| P-value of Shapiro-Wilk | 0.424 | 0.911 | 0.198 | 0.001 | 0.109 | 0.162 | 0.073 | 0.139 | 0.924 | 0.286 | 0.826 | 0.005 | 0.141 | 0.331 | 0.112 | 0.032 | 0.422 | 0.006 | 0.851 | 0.002 |
| Minimum | 30.300 | 29.800 | 21.200 | 34.600 | 0.062 | 0.052 | 0.050 | 0.072 | 44.400 | 55.800 | 34.000 | 85.800 | 0.037 | 0.022 | 0.050 | 0.059 | 743.270 | 454.080 | 164.890 | 619.650 |
| Maximum | 42.800 | 38.400 | 51.900 | 1542.700 | 0.243 | 0.436 | 0.445 | 0.660 | 78.600 | 101.600 | 133.800 | 1329.400 | 0.100 | 0.175 | 0.189 | 0.562 | 1081.760 | 1409.120 | 1195.170 | 8894.840 |
| Group comparisons | Mean Difference | SE | df | t | pturkey | |
|---|---|---|---|---|---|---|
| PON1 | ||||||
| A | B | 0.470 | 219.669 | 17 | 0.002 | 1.000 |
| C | 3.060 | 207.106 | 17 | 0.015 | 1.000 | |
| D | -310.094 | 191.743 | 17 | -1.617 | 0.396 | |
| B | C | 2.590 | 219.669 | 17 | 0.012 | 1.000 |
| D | -310.564 | 205.249 | 17 | -1.513 | 0.452 | |
| C | D | -313.154 | 191.743 | 17 | -1.633 | 0.387 |
| FRAP | ||||||
| A | B | -0.096 | 0.098 | 22 | -0.986 | 0.759 |
| C | -0.054 | 0.095 | 22 | -0.566 | 0.941 | |
| D | -0.141 | 0.092 | 22 | -1.534 | 0.435 | |
| B | C | 0.043 | 0.090 | 22 | 0.478 | 0.963 |
| D | -0.045 | 0.087 | 22 | -0.514 | 0.955 | |
| C | D | -0.088 | 0.084 | 22 | -1.050 | 0.723 |
| CUPRAC | ||||||
| A | B | -0.034 | 0.062 | 22 | -0.542 | 0.948 |
| C | -0.035 | 0.060 | 22 | -0.582 | 0.936 | |
| D | -0.139 | 0.059 | 22 | -2.367 | 0.113 | |
| B | C | -0.001 | 0.057 | 22 | -0.023 | 1.000 |
| D | -0.105 | 0.056 | 22 | -1.891 | 0.260 | |
| C | D | -0.104 | 0.053 | 22 | -1.949 | 0.237 |
| Cholinesterase | ||||||
| A | B | -10.870 | 176.571 | 18 | -0.062 | 1.000 |
| C | -18.503 | 159.385 | 18 | -0.116 | 0.999 | |
| D | -307.391 | 154.124 | 18 | -1.994 | 0.227 | |
| B | C | -7.633 | 169.905 | 18 | -0.045 | 1.000 |
| D | -296.521 | 164.980 | 18 | -1.797 | 0.307 | |
| C | D | -288.888 | 146.440 | 18 | -1.973 | 0.235 |
| Oxytocin | ||||||
| A | B | 203.659 | 1112.024 | 22 | 0.183 | 0.998 |
| C | 214.151 | 1075.313 | 22 | 0.199 | 0.997 | |
| D | -1843.756 | 1046.936 | 22 | -1.761 | 0.318 | |
| B | C | 10.492 | 1021.705 | 22 | 0.010 | 1.000 |
| D | -2047.415 | 991.794 | 22 | -2.064 | 0.196 | |
| C | D | -2057.907 | 950.451 | 22 | -2.165 | 0.164 |
| PON1 | FRAP | CUPRAC | Cholinesterase | Oxytocin | ||
|---|---|---|---|---|---|---|
| Factor | group | group | group | group | group | |
| Statistic | 8.489 | 1.224 | 7.202 | 10.763 | 8.013 | |
| dF | 3 | 3 | 3 | 3 | 3 | |
| P | 0.037 | 0.747 | 0.066 | 0.013 | 0.046 | |
| Rank ε2 | 0.424 | 0.049 | 0.288 | 0.513 | 0.321 | |
| 95% CI for Rank ε2 | Lower | 0.179 | 0.011 | 0.110 | 0.379 | 0.113 |
| Upper | 0.824 | 0.403 | 0.687 | 0.797 | 0.645 | |
| Rank η2 | 0.323 | 0.000 | 0.191 | 0.431 | 0.228 | |
| 95% CI for Rank η2 | Lower | 0.108 | 0.000 | 1.106x10-4 | 0.254 | 0.000 |
| Upper | 0.706 | 0.322 | 0.642 | 0.813 | 0.665 |
| Comparisons | z | Wi | Wj | rrb | p | pbonf | pholm |
|---|---|---|---|---|---|---|---|
| PON1 | |||||||
| A - B | -0.006 | 9.100 | 9.125 | 0.050 | 0.995 | 1.000 | 1.000 |
| A - C | 0.586 | 9.100 | 6.800 | 0.280 | 0.558 | 1.000 | 1.000 |
| A - D | -2.018 | 9.100 | 16.429 | 0.771 | 0.044 | 0.262 | 0.218 |
| B - C | 0.559 | 9.125 | 6.800 | 0.400 | 0.576 | 1.000 | 1.000 |
| B - D | -1.879 | 9.125 | 16.429 | 0.786 | 0.060 | 0.362 | 0.241 |
| C - D | -2.651 | 6.800 | 16.429 | 0.771 | 0.008 | 0.048 | 0.048 |
| FRAP | |||||||
| A - B | -0.587 | 10.700 | 13.417 | 0.167 | 0.557 | 1.000 | 1.000 |
| A - C | -0.577 | 10.700 | 13.286 | 0.143 | 0.564 | 1.000 | 1.000 |
| A - D | -1.101 | 10.700 | 15.500 | 0.450 | 0.271 | 1.000 | 1.000 |
| B - C | 0.031 | 13.417 | 13.286 | 0.000 | 0.975 | 1.000 | 1.000 |
| B - D | -0.504 | 13.417 | 15.500 | 0.125 | 0.614 | 1.000 | 1.000 |
| C - D | -0.559 | 13.286 | 15.500 | 0.143 | 0.576 | 1.000 | 1.000 |
| CUPRAC | |||||||
| A - B | -0.879 | 7.600 | 11.667 | 0.200 | 0.380 | 1.000 | 0.759 |
| A - C | -1.255 | 7.600 | 13.214 | 0.543 | 0.210 | 1.000 | 0.629 |
| A - D | -2.574 | 7.600 | 18.813 | 0.850 | 0.010 | 0.060 | 0.060 |
| B - C | -0.364 | 11.667 | 13.214 | 0.095 | 0.716 | 1.000 | 0.759 |
| B - D | -1.731 | 11.667 | 18.813 | 0.500 | 0.083 | 0.500 | 0.417 |
| C - D | -1.415 | 13.214 | 18.813 | 0.482 | 0.157 | 0.942 | 0.628 |
| Cholinesterase | |||||||
| A - B | -0.539 | 6.400 | 8.750 | 0.300 | 0.590 | 1.000 | 1.000 |
| A - C | -0.958 | 6.400 | 10.167 | 0.333 | 0.338 | 1.000 | 1.000 |
| A - D | -3.013 | 6.400 | 17.857 | 1.000 | 0.003 | 0.016 | 0.016 |
| B - C | -0.338 | 8.750 | 10.167 | 0.167 | 0.735 | 1.000 | 1.000 |
| B - D | -2.238 | 8.750 | 17.857 | 0.857 | 0.025 | 0.151 | 0.126 |
| C - D | -2.129 | 10.167 | 17.857 | 0.714 | 0.033 | 0.200 | 0.133 |
| Oxytocin | |||||||
| A - B | 1.468 | 15.800 | 9.000 | 0.667 | 0.142 | 0.852 | 0.568 |
| A - C | 1.359 | 15.800 | 9.714 | 0.429 | 0.174 | 1.000 | 0.568 |
| A - D | -0.677 | 15.800 | 18.750 | 0.300 | 0.499 | 1.000 | 0.997 |
| B - C | -0.168 | 9.000 | 9.714 | 0.000 | 0.867 | 1.000 | 0.997 |
| B - D | -2.360 | 9.000 | 18.750 | 0.708 | 0.018 | 0.110 | 0.110 |
| C - D | -2.283 | 9.714 | 18.750 | 0.679 | 0.022 | 0.135 | 0.112 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




