Submitted:
16 December 2025
Posted:
17 December 2025
You are already at the latest version
Abstract
Sulfites are the most extensively used additive in oenology to prevent oxidation and microbiological spoilage. However, their potential adverse health effects have increased the demand for low sulfite wines. Strategies are required to ensure microbiological stability while preserving the quality of the wine. This study evaluated strategies for reducing or replacing added sulfites using chitosan and low doses of lysozyme in Tannat winemaking, measuring their effects on microbial diversity, physicochemical parameters, and sensory attributes. Treatments were vinified by triplicate: reduced sulfites (RS: 30 mg/L), chitosan (C: 100 mg/L), reduced sulfites with chitosan (RS+C: 30 mg/L + 100 mg/L), reduced sulfites with lysozyme (RS+L: 30 mg/L + 5 mg/L), and a traditional winemaking (TW: 125 mg/L sulfites). Sulfur dioxide lowered lactic acid bacteria counts, whereas chitosan and lysozyme treatments maintained higher populations. Metagenomic analyses showed decreased bacterial diversity under sulfur dioxide, while chitosan promoted a more complex microbiota. Lysozyme selectively reduced lactic acid bacteria, mainly affecting Oenococcus spp. Lower sulfite decreased phenolic concentrations possible due to reduced protection against oxidation, leading to color differences among treatments. The results indicate that strategies to reduce or replace sulfites influence microbial dynamics, acidity, phenolic structure, and color, highlighting the importance of careful process management to maintain wine quality.
Keywords:
1. Introduction
2. Materials and Methods
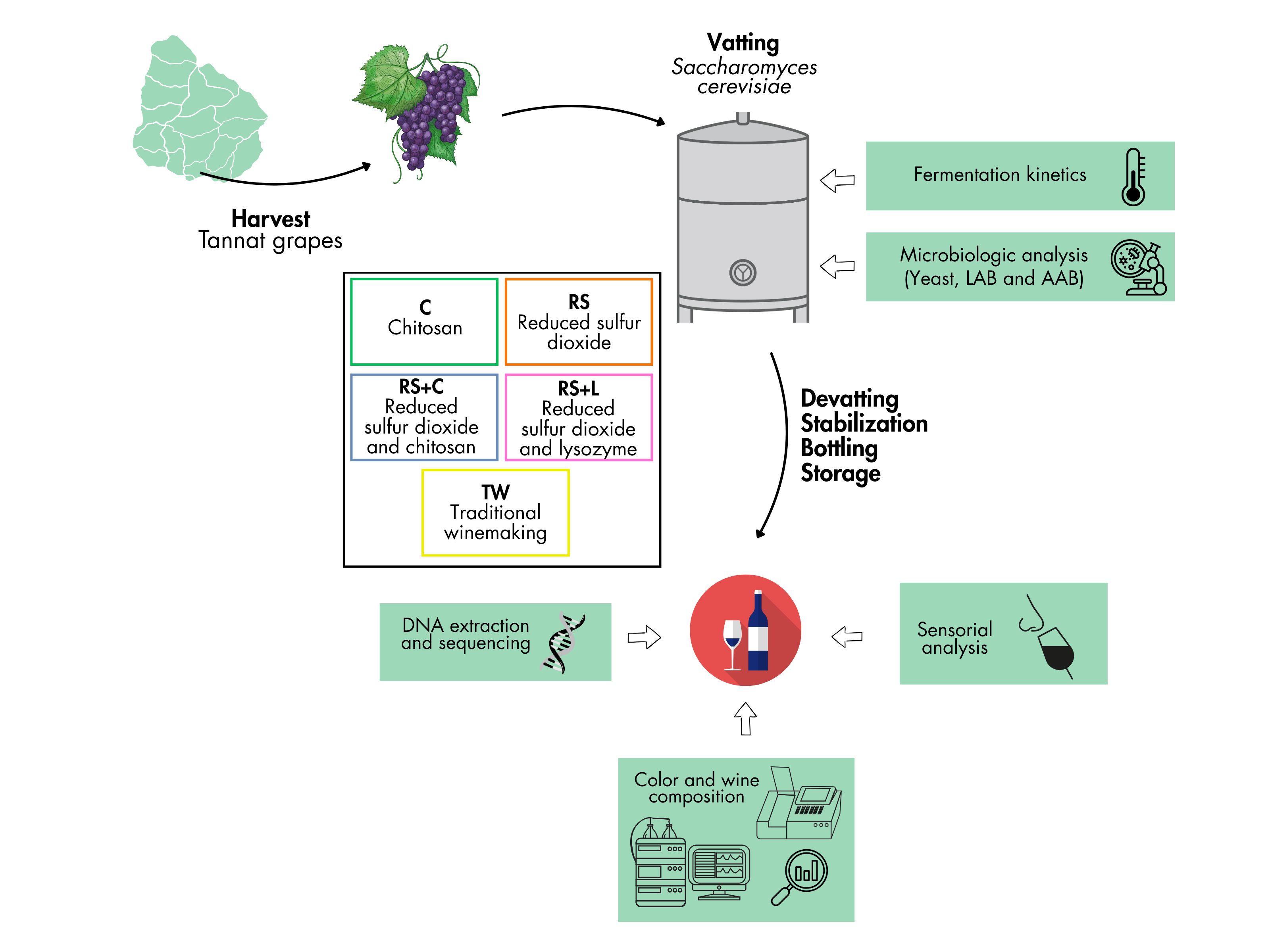
2.1. Grapes and Wines
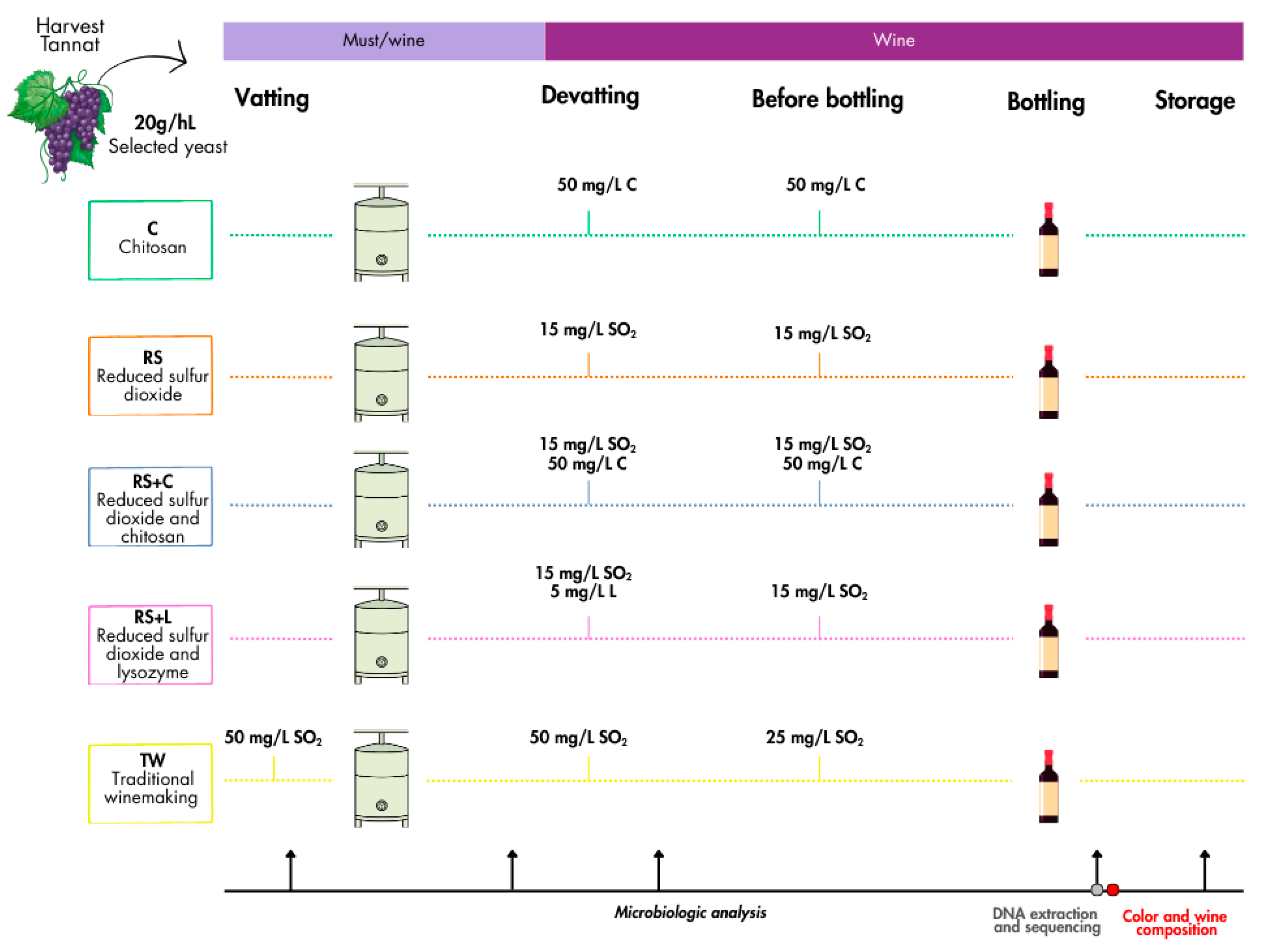
2.2. Harvest
2.3. Experimental Design
2.4. Analytical Determinations
2.4.1. Must Fermentation Kinetics
2.4.2. Microbiological Analysis of the Fermenting Must and the Wine
2.4.3. DNA Extraction and Sequencing
2.4.4. Sequence Analysis
2.4.5. Color and Wine Composition Evaluation
2.4.6. Sensorial Analysis of Wines
2.5. Data Processing and Statistical Analysis
3. Results
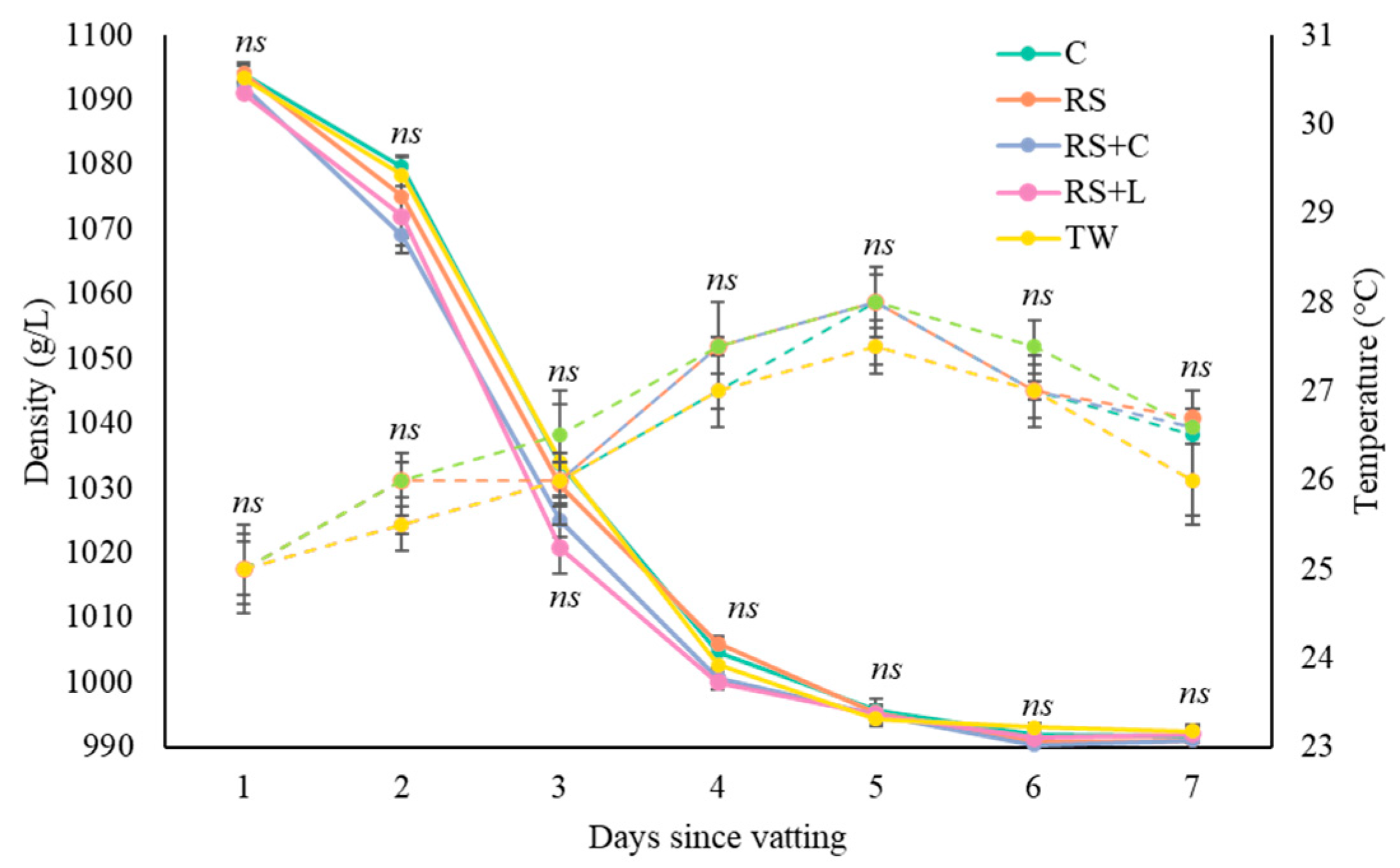
3.1. Grape Juice Fermentation Kinetics
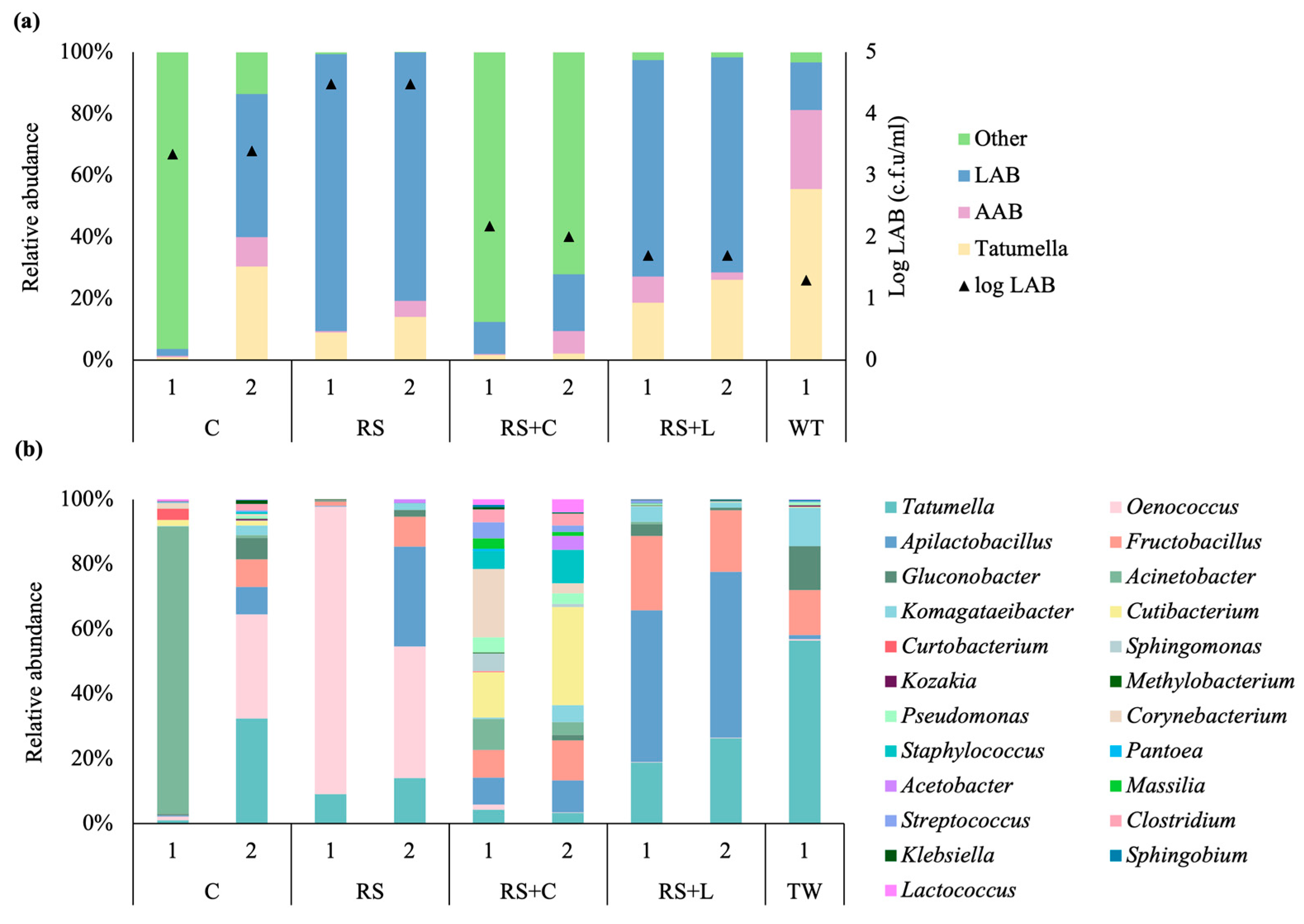
3.2. Microbial Population Dynamics
3.3. Diversity of Microbial Community
3.4. General Composition of Tannat Wines
3.5. Phenolic Composition of Tannat Wines
3.6. Color Parameters of Tannat Wines
3.7. Tannat Wines Sensory Attributes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| SO2 | sulfur dioxide |
| C | Chitosan |
| L | Lysozyme |
| RS | reduce sulfites |
| TW | Traditional Winemaking |
References
- International Organisation of Vine and Wine. Resolution OIV-OENO 631-2020: Review of practices for the reduction of SO2 doses used in winemaking. Available online: https://www.oiv.int/public/medias/7593/oiv-oeno-631-2020-en.pdf (accessed on 29 September 2025).
- International Organisation of Vine and Wine. SO2 and wine: a review. Available online: https://www.oiv.int/public/medias/7840/oiv-collective-expertise-document-so2-and-wine-a-review.pdf (accessed on 12 November 2025).
- Costanigro, M.; Appleby, C.; Menke, SD. The wine headache: consumer perceptions of sulfites and willingness to pay for non-sulfited wines. Food Qual Pref. 2014, 31, 81–89. [Google Scholar] [CrossRef]
- Santos, MC.; Nunes, C.; Saraiva, JÁ.; Coimbra, MA. Chemical and physical methodologies for the replacement/reduction of sulfur dioxide use during winemaking: review of their potentialities and limitations. Eur Food Res Technol. 2012, 234, 1–12. [Google Scholar] [CrossRef]
- Ribéreau-Gayon, P.; Dubourdieu, D.; Donèche, B.; Lonvaud, A. Handbook of enology, Volume 1: The microbiology of wine and vinifications; John Wiley & Sons: New Jersey, United States, 2006; p. 497.
- Limit of sulfur dioxide in wines. Available online: https://www.oiv.int/node/3940 (accessed on 10 October 2025).
- International Oenological Codex. Available online: https://www.oiv.int/sites/default/files/publication/2023-04/CODEX%20complet%202023%20EN.pdf (accessed on 10 October 2025).
- Lárez-Velásquez, C. Chitosan and its applications in oenology. OENO One 2023, 57, 121–132. [Google Scholar] [CrossRef]
- Paulin, M.; Miot-Sertier, C.; Dutilh, L.; Brasselet, C.; Delattre, C.; Pierre, G.; Dubessay, P.; Michaud, P.; Doco, T.; Ballestra, P.; Albertin, W.; Masneuf-Pomarède, I.; Moine, V.; Coulon, J.; Vallet-Courbin, A.; Maupeu, J.; Dols-Lafargue, M. +Brettanomyces bruxellensis Displays Variable Susceptibility to Chitosan Treatment in Wine. Front Microbiol. 2020, 11. [Google Scholar] [CrossRef]
- Valera, MJ.; Sainz, F.; Mas, A.; Torija, MJ. Effect of chitosan and SO2 on viability of Acetobacter strains in wine. Int J Food Microbiol. 2017, 246, 1–4. [Google Scholar] [CrossRef]
- Elmacı, SB.; Gülgör, G.; Tokatlı, M.; Erten, H.; İşci, A.; Özçelik, F. Effectiveness of chitosan against wine-related microorganisms. Antonie Van Leeuwenhoek 2015, 107, 675–686. [Google Scholar] [CrossRef]
- Gómez-Rivas, L.; Escudero-Abarca, BI.; Aguilar-Uscanga, MG.; Hayward-Jones, PM.; Mendoza, P.; Ramírez, M. Selective antimicrobial action of chitosan against spoilage yeasts in mixed culture fermentations. J Ind Microbiol Biotechnol. 2004, 31, 16–22. [Google Scholar] [CrossRef]
- Nunes, C.; Maricato, É.; Cunha, Â.; Rocha, MA.; Santos, S.; Ferreira, P.; Silva, M.; Rodrigues, A.; Amado, O.; Coimbra, J.; Silva, D. Chitosan–genipin film, a sustainable methodology for wine preservation. Green Chem. 2016, 18, 5331–5341. [Google Scholar] [CrossRef]
- Gao, Y.; Tian, Y.; Liu, D.; Li, Z.; Zhang, XX.; Li, JM.; Huang, JH.; Wang, J.; Pan, QH. Evolution of phenolic compounds and sensory in bottled red wines and their co-development. Food Chem. 2015, 172, 565–574. [Google Scholar] [CrossRef]
- Liburdi, K.; Benucci, I.; Esti, M. Lysozyme in wine: An overview of current and future applications. Compr Rev Food Sci Food Saf. 2014, 13, 1062–1073. [Google Scholar] [CrossRef]
- Wüthrich, B. Allergic and intolerance reactions to wine. Allergol. Select 2018, 2, 80–88. [Google Scholar] [CrossRef]
- Programa de vitivinicultura sostenible en Uruguay. Available online: https://www.inavi.com.uy/programa-de-viticultura-sostenible/ (accessed on 10 October 2025).
- Vitis International Variety Catalogue. Available online: https://www.vivc.de/index.php?r=passport%2Fview&id=12257 (accessed on 10 October 2025).
- González-Neves, G.; Ferrer, M.; Gil, G. Differentiation of Tannat, Cabernet Sauvignon and Merlot grapes from Uruguay according to their general composition and polyphenolic potential. Comun. Sci. 2012, 3, 41–49. [Google Scholar] [CrossRef]
- Ferrer, M.; Echeverría, G.; Pereyra, G.; González-Neves, G.; Pan, D.; Mirás-Avalos, JM. Mapping vineyard vigor using airborne remote sensing: relations with yield, berry composition and sanitary status under humid climate conditions. Precision Agric. 2020, 21, 178–197. [Google Scholar] [CrossRef]
- Piccardo, D.; Favre, G.; Pascual, O.; Canals, J. M.; Zamora, F.; González-Neves, G. Impact of must replacement and hot pre-fermentative maceration on the color of Uruguayan Tannat red wines. Fermentation 2019, 5, 80. [Google Scholar] [CrossRef]
- Normas para la producción integrada de uva y vino. Available online: https://www.gub.uy/ministerio-ganaderia-agricultura-pesca/sites/ministerio-ganaderia-agricultura-pesca/files/2023-11/ANEXO%20II%20-%20Norma%20Producciòn%20de%20uva%20para%20vino_0.pdf (accessed on 10 October 2025).
- Syndicat de défense des Vins Nature’l. Available online: https://vinmethodenature.org/wp-content/uploads/Dossier-de-presentation-Syndicat-de-defense-des-vins-naturel_compressed.pdf (accessed on 10 October 2025).
- Rivas, G. A.; Semorile, L.; Delfederico, L. Microbial diversity of the soil, rhizosphere and wine from an emerging wine-producing region of Argentina. Lwt 2022, 153. [Google Scholar] [CrossRef]
- Berbegal, C.; Borruso, L.; Fragasso, M.; Tufariello, M.; Russo, P.; Brusetti, L.; Spano, G.; Capozzi, V. A metagenomic-based approach for the characterization of bacterial diversity associated with spontaneous malolactic fermentations in wine. Int. J. Mol. Sci. 2019, 20. [Google Scholar] [CrossRef] [PubMed]
- R Core Team (2024). R: A Language and Environment for Statistical Computing. Computing (version 4.3.2) [Software] Vienna, Austria: R Foundation for Statistical Computing. https://www.R-project.org/ v4.4.1.
- Callahan, B. J.; McMurdie, P. J.; Rosen, M. J.; Han, A. W.; Johnson, A. J.; Holmes, S. P. DADA2: High-resolution sample inference from Illumina amplicon data (version 1.26.0) [Software]. 2016. [Google Scholar] [CrossRef]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res 2013, 41, 590–596. [Google Scholar] [CrossRef]
- Yilmaz, P; Parfrey, L. W.; Yarza, P.; Gerken, J.; Pruesse, E.; Quast, C.; Schweer, T.; Peplies, J.; Ludwig, W.; Glöckner, F. O. The SILVA and “all-species living tree project (LTP)” taxonomic frameworks. Nucleic Acids Res 2014, 42, 643–648. [Google Scholar] [CrossRef] [PubMed]
- McMurdie, P.J.; Holmes, S. Phyloseq: a bioconductor package for handling and analysis of high-throughput phylogenetic sequence data. Pac. Symp. Biocomput 2012, 235–246. [Google Scholar] [PubMed] [PubMed Central]
- McMurdie, P.J.; Holmes, S. P: phyloseq: An R package for reproducible interactive analysis and graphics of microbiome census data. PLoS One 2013, 8. [Google Scholar] [CrossRef]
- Ayala, F.; Echávarri, J F.; Negueruela, A. A new simplified method for measuring the color of wines. I. Red and Rosé Wines. Am. J. Enol. Vitic. 1997, 48, 357–363. [Google Scholar] [CrossRef]
- Ayala, F.; Echávarri, J F.; Negueruela, A. MSCV [Software]. 2021. [Google Scholar]
- Singleton, V.; Rossi, J. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar] [CrossRef]
- Ribéreau-Gayon, P.; Stonestreet, E. Determination of Anthocyanins in Red Wine. Bull. Soc. Chim. Fr. 1965, 9, 2649–2652. [Google Scholar]
- Di Rienzo, J.A.; Casanoves, F.; Balzarini, M.G.; Gonzalez, L.; Tablada, M.; Robledo, C.W. InfoStat versión 2020. Centro de Transferencia InfoStat, FCA, Universidad Nacional de Córdoba, Argentina. URL http://www.infostat.com.ar.
- Sarneckis, C.; Dambergs, R.; Jones, P.; Mercurio, M.; Herderich, M.; Smith, P. Quantification of condensed tannins by precipitation with methyl cellulose: Development and validation of an optimised tool for grape and wine analysis. Aust. J. Grape Wine Res. 2006, 12, 39–49. [Google Scholar] [CrossRef]
- Martín-García, F.J.; Palacios-Fernández, S.; López de Lerma, N.; García-Martínez, T.; Mauricio, J.C.; Peinado, R. A. The Effect of Yeast, Sugar and Sulfur Dioxide on the Volatile Compounds in Wine. Fermentation 2023, 9. [Google Scholar] [CrossRef]
- Scansani, S.; Rauhut, D.; Brezina, S.; Semmler, H.; Benito, S. The Impact of Chitosan on the Chemical Composition of Wines Fermented with Schizosaccharomyces pombe and Saccharomyces cerevisiae. Foods 2020, 9, 1–11. [Google Scholar] [CrossRef]
- Miot Sertier, C.; Paulin, M.; Dutilh, L.; Ballestra, P.; Albertin, W.; Masneuf Pomarède, I.; Coulon, J.; Moine, V.; Vallet Courbin, A.; Maupeu, J.; Dols La argue, M. Assessment of chitosan antimicrobial effect on wine microbes. Int. J. Food Microbiol. 2022, 381, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Gerbaux, V.; Villa, A.; Monamy, C.; Bertrand, A. Use of lysozyme to inhibit malolactic fermentation and to stabilize wine after malolactic fermentation. Am. J. of Enol. and Vitic. 1997, 48, 49–54. [Google Scholar] [CrossRef]
- Roldán, A; Lasanta, C; Caro, I; Palacios, V. Effect of lysozyme on “flor” velum yeasts in the biological aging of sherry wines. Food Microbiol. 2012, 30, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Ohwofasa, A.; Dhami, M.; Zhang, J.; Tian, B.; Winefield, C.; On, S.L. Influence of climatic variation on microbial communities during organic Pinot noir wine production. PLoS One 2024, 19. [Google Scholar] [CrossRef]
- Windholtz, S.; Miot-Sertier, C.; Maupeu, J.; Vallet-Courbin, A.; Lucas, M.; Pelonnier-Magimel, E.; Duarte, V.; Becquet, S.; Vinsonneau, E.; Lucas, P.; Masneuf-Pomarède, I. Influence of sulphur dioxide management on microbial populations during wine ageing. OENO One 2025, 59, 3. [Google Scholar] [CrossRef]
- Papadopoulou, E.; Bekris, F.; Vasileiadis, S.; Krokida, A.; Rouvali, T.; Veskoukis, A. S.; Liadaki, K.; Kouretas, D.; Karpouzas, D. G. Vineyard-mediated factors are still operative in spontaneous and commercial fermentations shaping the vinification microbial community and affecting the antioxidant and anticancer properties of wines. Food Res. Int. 2023, 173, 113359. [Google Scholar] [CrossRef]
- Liu, Q.; Hao, N; Mi, L.; Peng, S.; Marie-Colette, A.K.; Zhao, X.; Wang, J. From microbial communities to aroma profiles: A comparative study of spontaneous fermentation in merlot and cabernet sauvignon wines. Food Chem: X 2025, 26. [Google Scholar] [CrossRef]
- Bartowsky, E.J.; Henschke, P.A. Acetic acid bacteria spoilage of bottled red wine—a review. Int. J. Food Microbiol 2020, 125, 60–70. [Google Scholar] [CrossRef]
- Prusova, B.; Licek, J.; Kumsta, M.; Baron, M.; Sochor, J. Effect of New Methods for Inhibiting Malolactic Fermentation on the Analytical and Sensory Parameters of Wines. Fermentation 2024, 10. [Google Scholar] [CrossRef]
- Tedesco, F.; Siesto, G.; Pietrafesa, R.; Romano, P.; Salvia, R.; Scieuzo, C.; Falabella, P.; Capece, A. Chemical methods for microbiological control of winemaking: An overview of current and future applications. Beverages 2022, 8. [Google Scholar] [CrossRef]
- Delfini, C.; Cersosimo, M.; Del Prete, V.; Strano, M.; Gaetano, G.; Pagliara, A.; Ambrò, S. Resistance Screening Essay of Wine Lactic Acid Bacteria on Lysozyme: Efficacy of Lysozyme in Unclarified Grape Musts. J. Agric. Food Chem. 2004, 52, 1861–1866. [Google Scholar] [CrossRef]
- Garofalo, C.; El Khoury, M.; Lucas, P.; Bely, M.; Russo, P.; Spano, G.; Capozzi, V. Autochthonous starter cultures and indigenous grape microbiota for sustainable wine production. Front Microbiol 2020, 11, 1395–1408. [Google Scholar] [CrossRef]
- Dicks, L.M.; Endo, A. Taxonomic status of lactic acid bacteria in wine and key characteristics to differentiate species. South African Journal of Enology and Viticulture 2009, 30, 72–90. [Google Scholar] [CrossRef]
- Capozzi, V.; Fragasso, M.; Romaniello, R.; Berbegal, C.; Russo, P.; Spano, G. Spontaneous food fermentations and potential risks for human health. Fermentation 2017, 7. [Google Scholar] [CrossRef]
- Rocha, M.A.M.; Coimbra, M.A.; Nunes, C. Applications of chitosan and their derivatives in beverages: A critical review. Curr. Opin. Food Sci. 2017, 15, 61–69. [Google Scholar] [CrossRef]
- Moulis, P.; Miot-Sertier, C.; Franc, C.; Riquier, L.; Beisert, B.; Marchand, S.; de Revel, G.; Ballestra, P.; Rauhut, D. Impact of Pediococcus parvulus and Saccharomyces cerevisiae on Brettanomyces bruxellensis mousy compound production. OENO One 2024, 58. [Google Scholar] [CrossRef]
- Almeida Santos, C.; Pereira, C.; Martins, N.; Cabrita, M.J.; da Silva, M. Different SO2 Doses and the Impact on Amino Acid and Volatile Profiles of White Wines. Beverage 2023, 9. [Google Scholar] [CrossRef]
- Jackowetz, J.N.; Mira de Orduña, R. Metabolism of SO₂ binding compounds by Oenococcus oeni during and after malolactic fermentation in white wine. Int. J. Food Microbiol. 2012, 155, 153–157. [Google Scholar] [CrossRef]
- He, F.; Liang, N.N.; Mu, L.; Pan, Q.H.; Wang, J.; Reeves, M. J.; Duan, C. Q. Antho-cyanins and their variation in red wines II. Anthocyanin derived pigments and their color evolution. Molecules 2012, 17, 1483–1519. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, C.; Silva Ferreira, A.; De Freitas, V.; Silva, A. Oxidation mechanisms occurring in wines. Food Res Int 2011, 44, 1115–1126. [Google Scholar] [CrossRef]
- Comuzzo, P.; Toniolo, R.; Battistutta, F.; Lizee, M.; Svigelj, R.; Zironi, R. Oxidative behavior of (+)-catechin in the presence of inactive dry yeasts: A comparison with sulfur dioxide, ascorbic acid and glutathione. J. Sci. Food Agric 2017, 97, 5158–5167. [Google Scholar] [CrossRef] [PubMed]
- Baris, F.; Castro Marín, A.; De Aguiar Saldanha Pinheiro, A.C.; Tappi, S.; Chinnici, F. Efficacy of fungoid chitosans from Aspergillus niger and Agaricus bisporus in controlling the oxidative browning of model white wines. Innov. Food Sci. Emerg. Technol. 2023, 86. [Google Scholar] [CrossRef]
- Bartowsky, E.J.; Henschke, P.A. The ‘buttery’ attribute of wine—diacetyl—desirability, spoilage and beyond. Int. J. Food Microbiol. 2004, 96, 235–252. [Google Scholar] [CrossRef]
- Guzzo, F; Cappello, M.S.; Azzolini, M.; Tosi, E.; Zapparoli, G. The inhibitory effects of wine phenolics on lysozyme activity against lactic acid bacteria. Int J Food Microbiol 2011, 148, 184–190. [Google Scholar] [CrossRef]
- Zara, G.; Nardi, T. Yeast Metabolism and Its Exploitation in Emerging Winemaking Trends: From Sulfite Tolerance to Sulfite Reduction. Fermentation 2021, 7. [Google Scholar] [CrossRef]
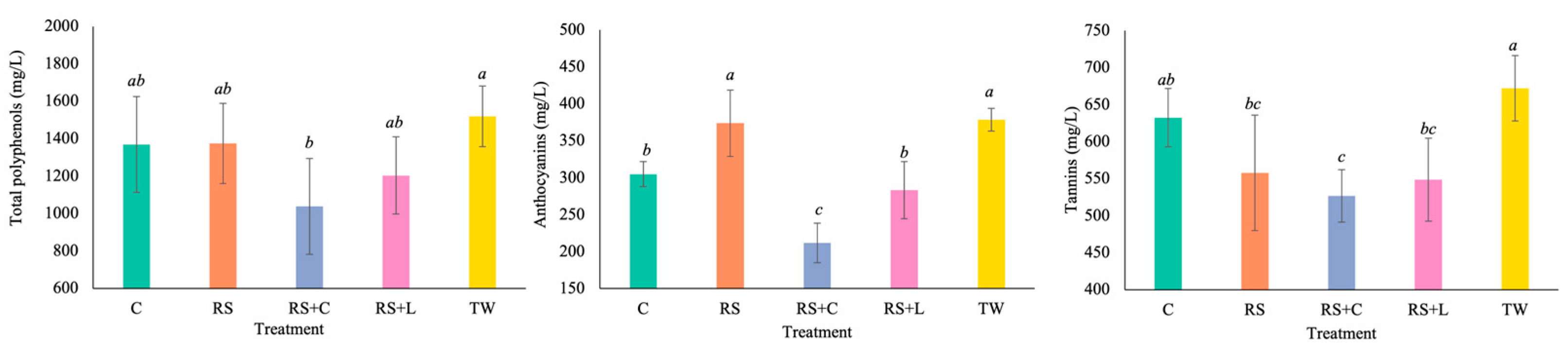
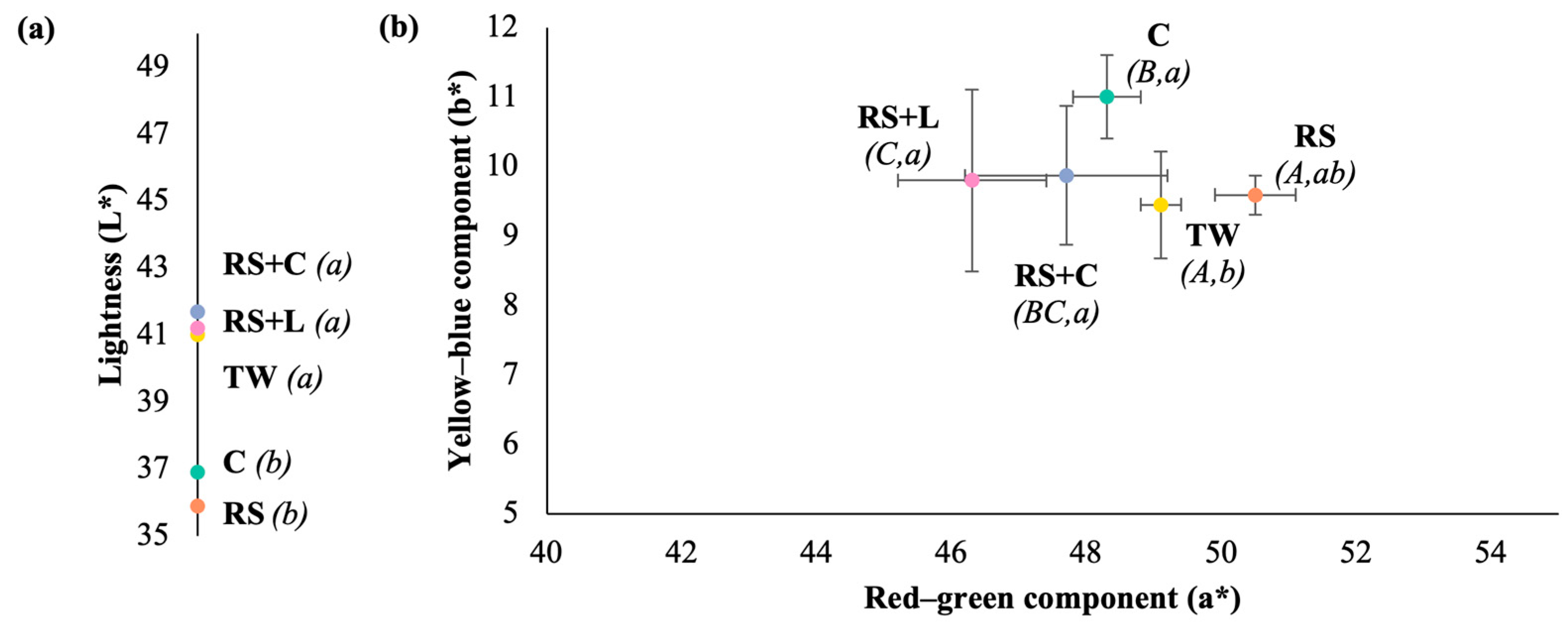
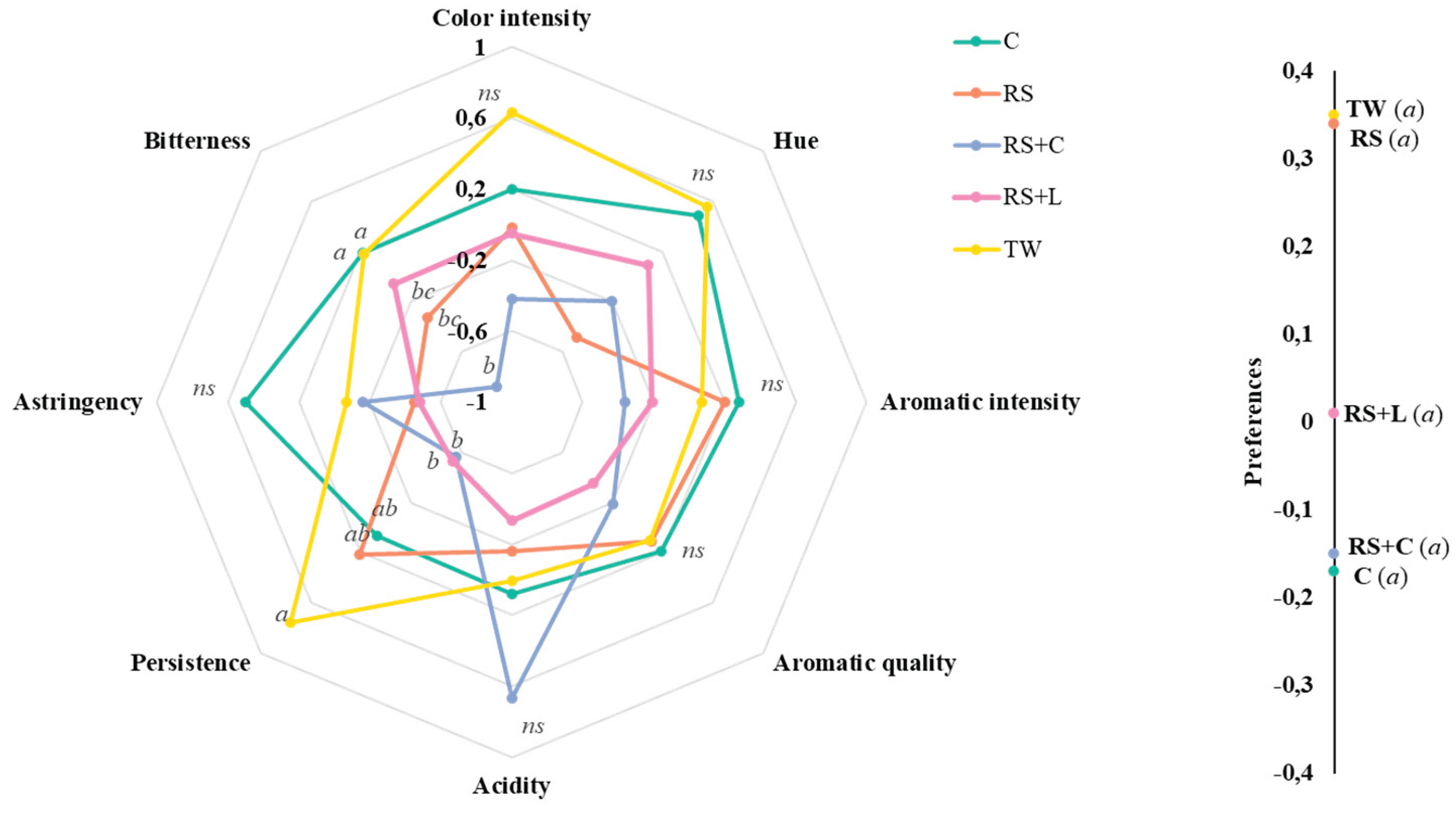
| Microbial count | Analysis stage | Treatments | ||||
| (c.f.u./ml) | C | RS | RS+C | RS+L | TW | |
| Total aerobic count | Vatting | 7,11 ± 0,77 a | 7,56 ± 0,01 a | 7,11 ± 0,77 a | 7,56 ± 0,01 a | 6,28 ± 0,15 b |
| Before devatting | 7,00 ± 0,47 b | 6,67 ± 0,23 b | 7,00 ± 0,47 b | 6,67 ± 0,23 b | 7,67 ± 0,01 a | |
| After devatting | 5,59 ± 0,02 a | - | 2,74 ± 0,37 b | - | - | |
| Afterbottling | - | - | - | - | - | |
| Storage | - | - | - | - | - | |
| Yeast | Vatting | 7,24 ± 0,03 a | 7,24 ± 0,02 a | 7,24 ± 0,03 a | 7,24 ± 0,02 a | 7,32 ± 0,14 a |
| Before devatting | 7,14 ± 0,15 a | 6,92 ± 0,32 a | 7,14 ± 0,15 a | 6,92 ± 0,32 a | 7,63 ± 0,18 a | |
| After devatting | 5,07 ± 0,10 a | 1,00 ± 0,10 b | 1,00 ± 0,10 b | 1,00 ± 0,10 b | 1,00 ± 0,10 b | |
| Afterbottling | 1,60 ± 0,10 a | 1,00 ± 0,10 b | 1,70 ± 0,10 a | 1,66 ± 0,26 a | 1,15 ± 0,21 ab | |
| Storage | 3,34 ± 0,31 a | 2,58 ± 0,25 b | 3,65 ± 0,77 a | 3,09 ± 0,73 a | 3,52 ± 0,06 a | |
| Lactic acid bacteria | Vatting | 6,03 ± 0,17 a | 5,70 ± 0,05 a | 6,03 ± 0,17 a | 5,70 ± 0,06 a | 6,12 ± 0,41 a |
| Before devatting | 3,80 ± 0,28 a | 4,06 ± 0,08 a | 3,80 ± 0,28 a | 4,06 ± 0,08 a | 3,00 ± 0,10 a | |
| After devatting | 2,08 ± 0,34 b | 3,31 ± 0,15 a | 2,48 ± 0,10 b | 2,48 ± 0,10 b | 1,59 ± 0,16 b | |
| Afterbottling | 3,37 ± 0,04 b | 4,48 ± 0,10 a | 1,95 ± 0,10 c | 1,70 ± 0,10 c | 1,30 ± 0,43 c | |
| Storage | 2,58 ± 0,37 b | 2,25 ± 0,26 b | 3,07 ± 0,71 a | 3,01 ± 0,64 a | - | |
| Acetic acid bacteria | Vatting | 3,72 ± 0,36 a | 3,72 ± 0,36 a | 3,72 ± 0,36 a | 3,72 ± 0,36 a | 4,06 ± 0,27 a |
| Before devatting | - | - | - | - | - | |
| After devatting | - | - | - | - | - | |
| Afterbottling | - | - | - | - | - | |
| Storage | - | - | - | - | - | |
| Treatments | |||||||||
| C | RS | RS+C | RS+L | TW | |||||
| 1 | 2 | 1 | 2 | 1 | 2 | 1 | 2 | 1 | |
| Raw reads | 109816 | 179059 | 183538 | 175799 | 180056 | 184566 | 130254 | 185791 | 165553 |
| ASVs | 94 | 294 | 132 | 37 | 608 | 351 | 165 | 167 | 181 |
| % reads assigned at genus level | 0,995 | 0,993 | 0,999 | 1 | 0,893 | 0,938 | 0,999 | 0,997 | 0,997 |
| Number of genus | 54 | 124 | 73 | 15 | 233 | 141 | 107 | 103 | 102 |
| Shannon diversity index | 1,59 | 2,58 | 0,59 | 1,64 | 5,25 | 4,37 | 1,73 | 1,52 | 2,00 |
| Chao1 richness index | 94 | 294 | 133 | 36 | 611 | 352 | 166 | 164 | 183 |
| Analytical parameter | Treatments | ||||
| C | RS | RS+C | RS+L | TW | |
| Ethanol (% V/V) | 13,1 ± 0,2 a | 12,6 ± 0,1 b | 12,7 ± 0,3 b | 12,6 ± 0,1 b | 12,8 ± 0,2 ab |
| pH | 3,89 ± 0,01 a | 3,73 ± 0,03 c | 3,76 ± 0,02 c | 3,81 ± 0,03 b | 3,85 ± 0,02 b |
| Titratable acidity (g/L) | 3,57 ± 0,08 b | 3,97 ± 0,12 ab | 4,32 ± 0,79 a | 3,62 ± 0,16 b | 3,57 ± 0,08 b |
| Malic acid (g/L) | 1,53 ± 0,05 cd | 1,97 ± 0,08 b | 1,73 ± 0,34 bc | 1,42 ± 0,12 d | 2,30 ± 0,09 a |
| Lactic acid (g/L) | 1,78 ± 0,04 b | 1,58 ± 0,10 b | 2,50 ± 0,43 a | 2,23 ± 0,08 a | 1,18 ± 0,17 c |
| Volatile acidity (g/L) | 0,43 ± 0,02 a | 0,44 ± 0,07 a | 0,47 ± 0,10 a | 0,46 ± 0,04 a | 0,33 ± 0,04 a |
| Fructose (g/L) | 0,95 ± 0,14 a | 0,98 ± 0,08 a | 0,82 ± 0,19 a | 0,90 ± 0,14 a | 0,97 ± 0,10 a |
| Glucose (g/L) | 1,80 ± 0,11 d | 2,32 ± 0,12 c | 3,20 ± 0,23 a | 2,77 ± 0,19 b | 1,57 ± 0,08 d |
| Glucose/Fructose ratio | 2,40 ± 0,18 a | 2,17 ± 0,16 a | 1,25 ± 0,10 c | 1,72 ± 0,38 b | 2,27 ± 0,18 a |
| Anthocyanins profile (%) | Treatments | ||||
| C | RS | RS+C | RS+L | TW | |
| Non-acylated anthocyanins | 82,8 ± 0,2 ab | 80,6 ± 1,1 d | 83,8 ± 0,5 a | 82,3 ± 0,9 abc | 80,9 ± 0,1 cd |
| Acetylated anthocyanins | 17,0 ± 0,2 cd | 19,4 ± 0,9 a | 16,2 ± 0,5 d | 17,6 ± 0,9 bcd | 18,7 ± 0,1 ab |
| Comarylated anthocyanins | 0,19 ± 0,01 bc | 0,61 ± 0,25 a | 0,03 ± 0,03 c | 0,11 ± 0,13 bc | 0,37 ± 0,04 ab |
| Delphinidin | 2,20 ± 0,17 ab | 3,00 ± 0,47 a | 0,95 ± 0,24 c | 1,27 ± 0,60 bc | 2,50 ± 0,11 a |
| Cyanidin | - | - | - | - | - |
| Petunidin | 7,00 ± 0,29 a | 7,79 ± 0,63 a | 4,89 ± 0,33 b | 5,35 ± 0,67 b | 6,85 ± 0,20 a |
| Peonidin | 1,06 ± 0,11 bc | 1,90 ± 0,33 b | 0,73 ± 0,50 c | 0,80 ± 0,32 c | 2,76 ± 0,14 a |
| Malvidin | 89,7 ± 0,6 bc | 87,3 ± 1,3 c | 93,4 ± 1,1 a | 92,6 ± 1,6 a | 87,9 ± 0,3 c |
| Color parameter | Treatments | ||||
| C | RS | RS+C | RS+L | TW | |
| C*ab | 49,5 ± 0,5 b | 51,4 ± 0,6 a | 48,7 ± 1,7 bc | 47,3 ± 1,4 c | 50,0 ± 0,4 ab |
| h ab | 12,9 ± 0,6 a | 10,8 ± 0,3 b | 11,7 ± 0,9 ab | 11,9 ± 1,3 ab | 10,9 ± 0,8 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




