Submitted:
15 December 2025
Posted:
17 December 2025
You are already at the latest version
Abstract
Keywords:
Introduction
Results
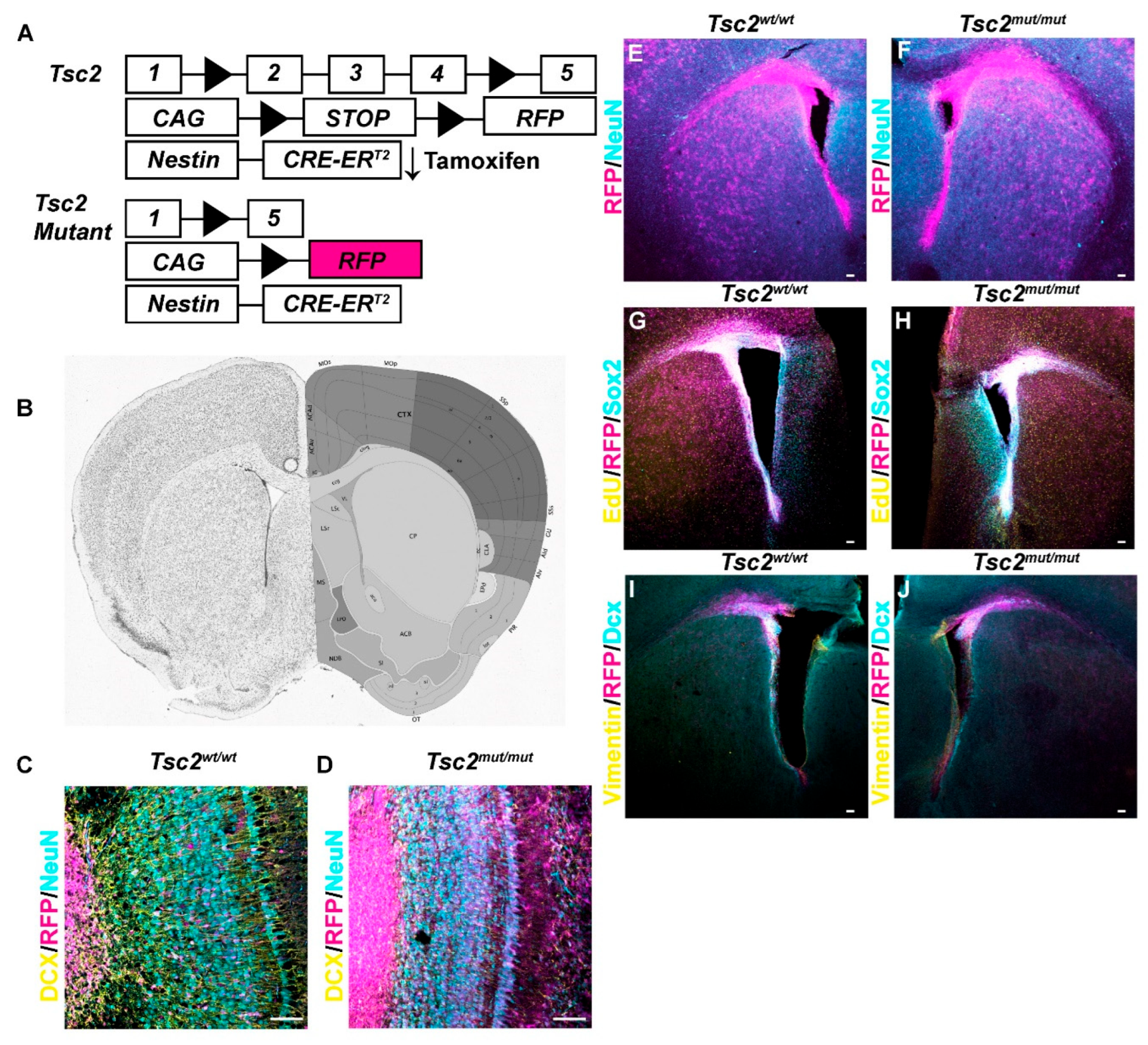
Targeted Recombination in Gliogenic and Neurogenic Stem Cells
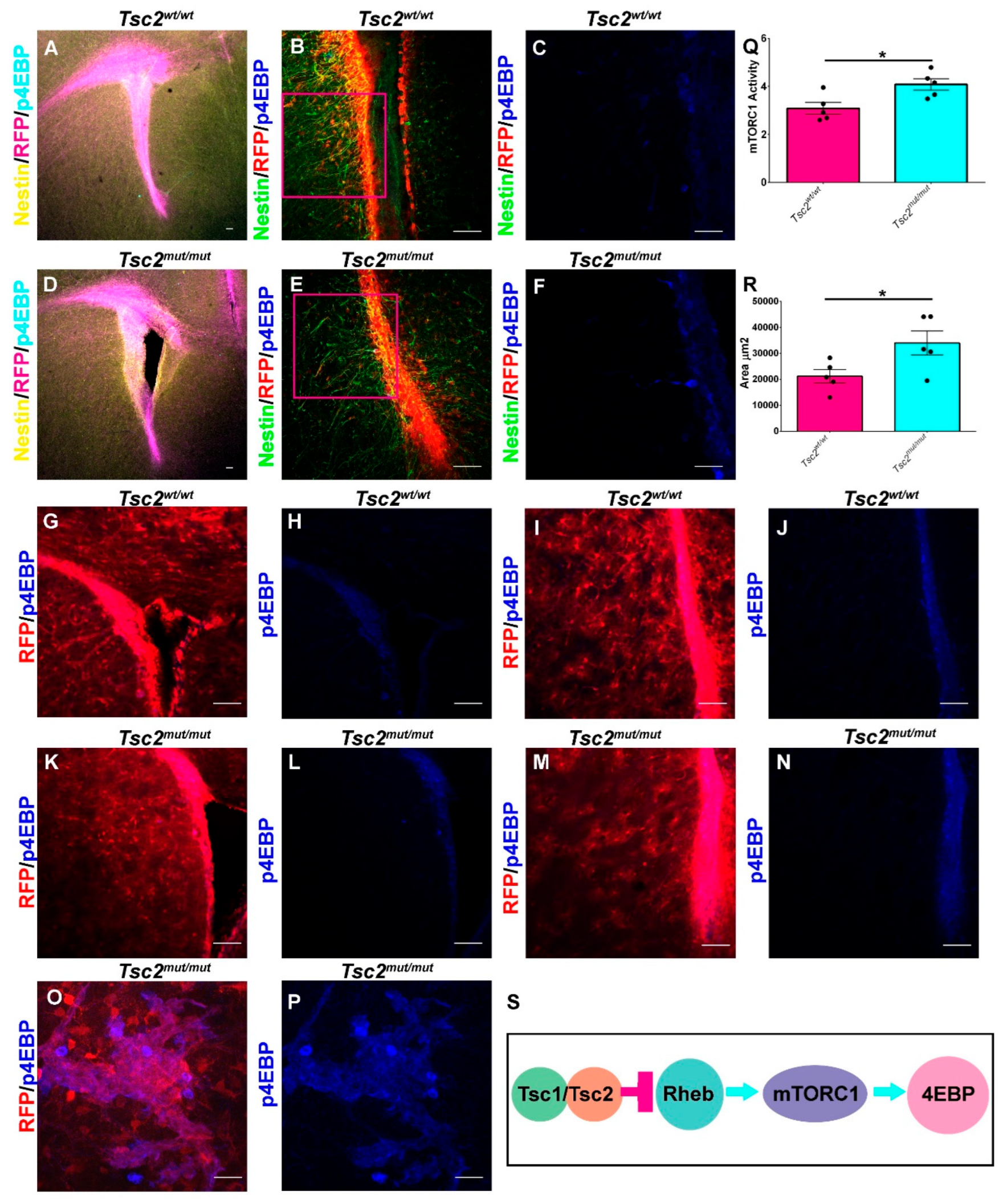
Loss of Tsc2 in NSCs Increases mTORC1 Activity
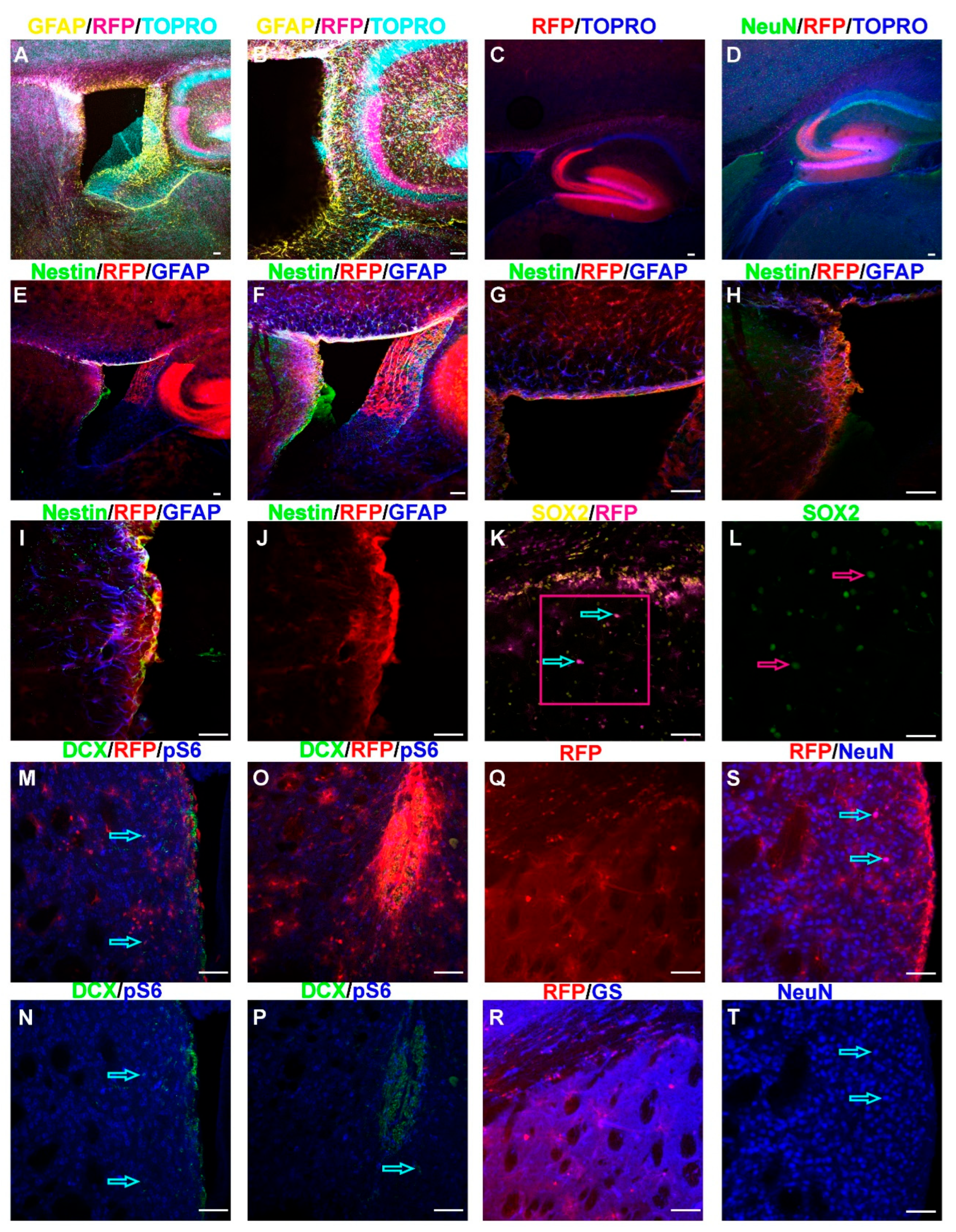
Cellular Phenotypes Associated with Tsc2 Mutation
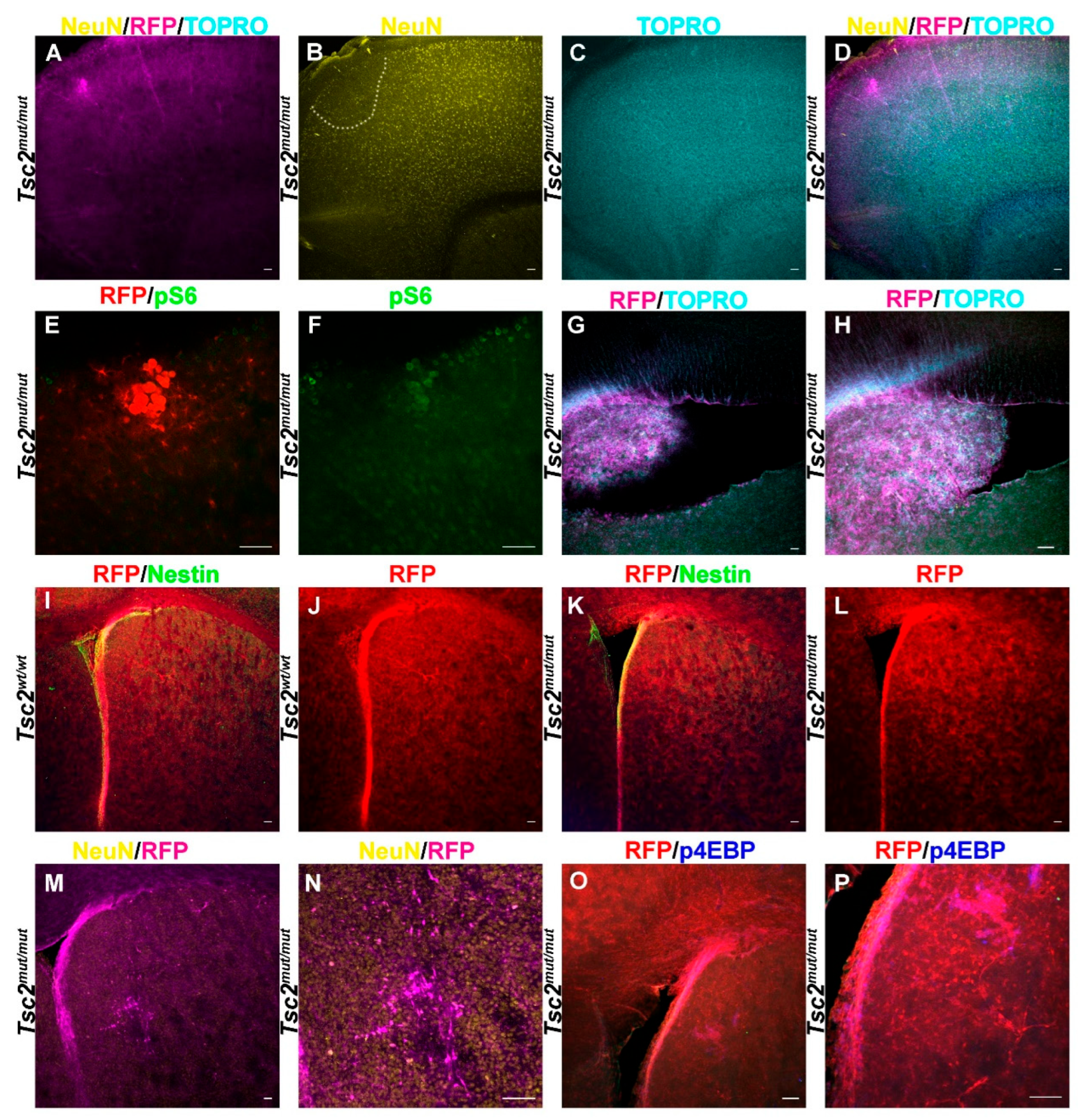
Loss of Tsc2 Models TSC Brain Pathological Features
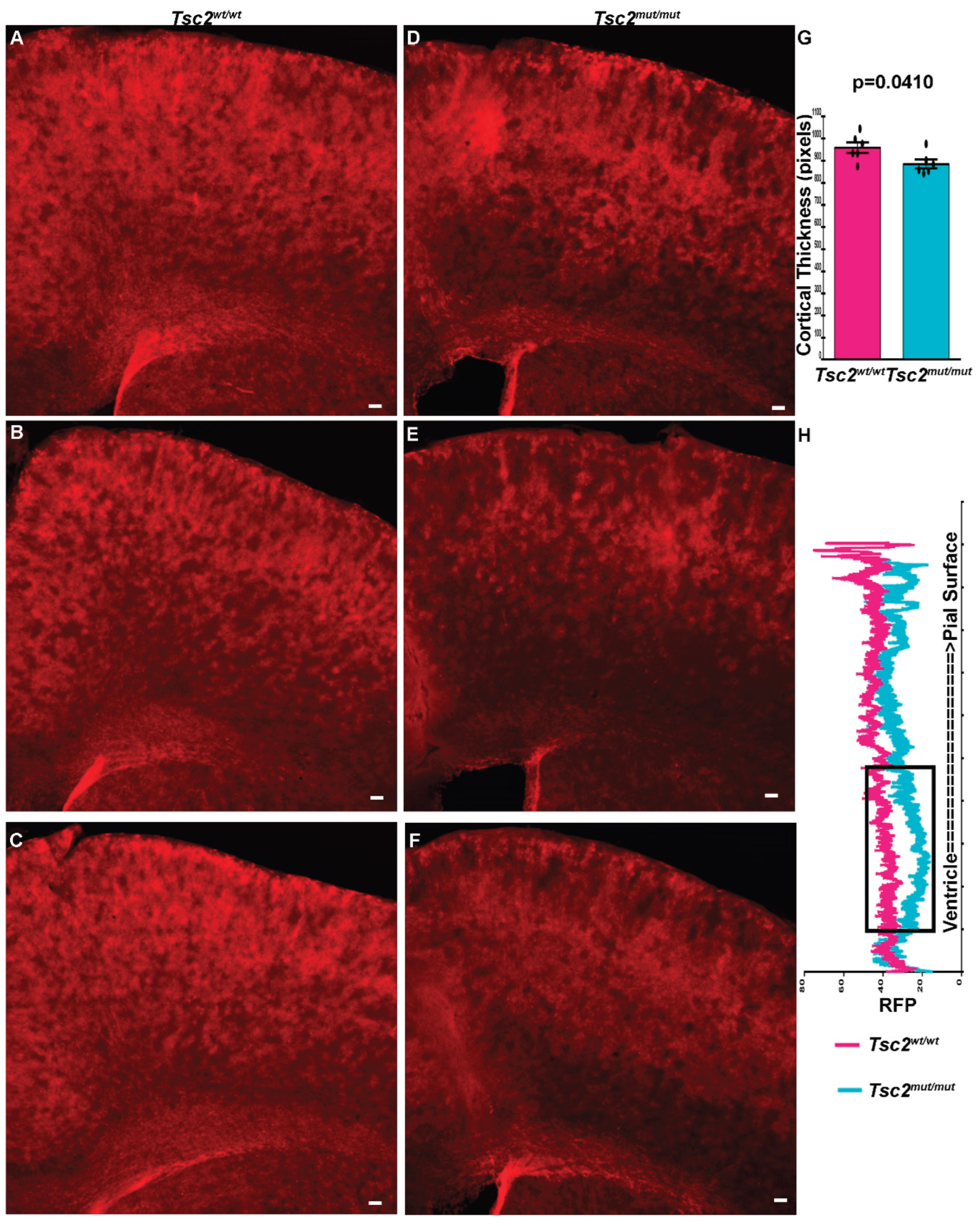
Neonatal Tsc2 Deletion Alters Cortical Development
Discussion
Materials and Methods
References
- Hasbani, D.M.; Crino, P.B. Tuberous sclerosis complex. Handbook of Clinical Neurology 2018, Vol. 148, 813–822. [Google Scholar]
- Feliciano, D.M.; Lin, T. V.; Hartman, N.W.; Bartley, C.M.; Kubera, C.; Hsieh, L.; Lafourcade, C.; O’Keefe, R.A.; Bordey, A. A circuitry and biochemical basis for tuberous sclerosis symptoms: From epilepsy to neurocognitive deficits. A circuitry and biochemical basis for tuberous sclerosis symptoms: From epilepsy to neurocognitive deficits. International Journal of Developmental Neuroscience 2013, 31, 667–678. [Google Scholar] [CrossRef]
- Feliciano, D.M. The Neurodevelopmental Pathogenesis of Tuberous Sclerosis Complex (TSC). The Neurodevelopmental Pathogenesis of Tuberous Sclerosis Complex (TSC). Front Neuroanat 2020, 14. [Google Scholar]
- Van Slegtenhorst, M.; De Hoogt, R.; Hermans, C.; Nellist, M.; Janssen, B.; Verhoef, S.; Lindhout, D.; Van Den Ouweland, A.; Halley, D.; Young, J.; et al. Identification of the tuberous sclerosis gene TSC1 on chromosome 9q34. Science (1979) 1997, 277, 805–808. [Google Scholar] [CrossRef]
- The European Chromosome 16 Tuberous Sclerosis Consortium Identification and characterization of the tuberous sclerosis gene on chromosome 16. Cell 1993, 75, 1305–1315. [CrossRef]
- Henske, E.P.; Jóźwiak, S.; Kingswood, J.C.; Sampson, J.R.; Thiele, E.A. Tuberous sclerosis complex. Nat Rev Dis Primers 2016, 2, 16035. [Google Scholar] [CrossRef]
- Henske, E.P.; Jóźwiak, S.; Kingswood, J.C.; Sampson, J.R.; Thiele, E.A. Tuberous sclerosis complex. Nat Rev Dis Primers 2016, 2, 16035. [Google Scholar] [CrossRef]
- Chan, D.L.; Calder, T.; Lawson, J.A.; Mowat, D.; Kennedy, S.E. The natural history of subependymal giant cell astrocytomas in tuberous sclerosis complex: A review. Rev Neurosci 2018, 29, 295–301. [Google Scholar] [CrossRef]
- Hasbani, D.M.; Crino, P.B. Tuberous sclerosis complex. Handbook of Clinical Neurology 2018, Vol. 148, 813–822. [Google Scholar]
- Knudson, A.G. Mutation and cancer: statistical study of retinoblastoma. Proc Natl Acad Sci U S A 1971, 68, 820–823. [Google Scholar] [CrossRef]
- Hino, O.; Klein-Szanto, A.J.P.; Freed, J.J.; Testa, J.R.; Brown, D.Q.; Vilensky, M.; Yeung, R.S.; Tartof, K.D.; Knudson, A.G. Spontaneous and radiation-induced renal tumors in the Eker rat model of dominantly inherited cancer. Proc Natl Acad Sci U S A 1993, 90, 327–331. [Google Scholar] [CrossRef]
- Yeung, R.S.; Xiao, G.H.; Jin, F.; Lee, W.C.; Testa, J.R.; Knudson, A.G. Predisposition to renal carcinoma in the Eker rat is determined by germ- line mutation of the tuberous sclerosis 2 (TSC2) gene. Proc Natl Acad Sci U S A 1994, 91, 11413–11416. [Google Scholar] [CrossRef]
- Yeung, R.S.; Xiao, G.H.; Jin, F.; Lee, W.C.; Testa, J.R.; Knudson, A.G. Predisposition to renal carcinoma in the Eker rat is determined by germ- line mutation of the tuberous sclerosis 2 (TSC2) gene. Proc Natl Acad Sci U S A 1994, 91, 11413–11416. [Google Scholar] [CrossRef]
- Xiao, G.H.; Jin, F.; Yeung, R.S. Germ-line Tsc2 mutation in a dominantly inherited cancer model defines a novel family of rat intracisternal-A particle elements. Oncogene 1995, 11. [Google Scholar]
- Yeung, R.S.; Katsetos, C.D.; Klein-Szanto, A. Subependymal astrocytic hamartomas in the Eker rat model of tuberous sclerosis. American Journal of Pathology 1997, 151, 1477–1486. [Google Scholar] [PubMed]
- Wenzel, H.J.; Patel, L.S.; Robbins, C.A.; Emmi, A.; Yeung, R.S.; Schwartzkroin, P.A. Morphology of cerebral lesions in the Eker rat model of tuberous sclerosis. Acta Neuropathol 2004, 108. [Google Scholar] [CrossRef]
- Kubo, Y.; Mitani, H.; Hino, O. Allelic Loss at the Predisposing Gene Locus in Spontaneous and Chemically Induced Renal Cell Carcinomas in the Eker Rat. Cancer Res 1994, 54, 2633–2635. [Google Scholar]
- Kubo, Y.; Klimek, F.; Kikuchi, Y.; Bannasch, P.; Hino, O. Early Detection of Knudson’s Two-Hits in Preneoplastic Renal Cells of the Eker Rat Model by the Laser Microdissection Procedure. Cancer Res 1995, 55, 989–990. [Google Scholar] [PubMed]
- Kobayashi, T.; Urakami, S.; Hirayama, Y.; Yamamoto, T.; Nishizawa, M.; Takahara, T.; Kubo, Y.; Hino, O. Intragenic Tsc2 somatic mutations as Knudson’s second hit in spontaneous and chemically induced renal carcinomas in the Eker rat model. Japanese Journal of Cancer Research 1997, 88, 254–261. [Google Scholar] [CrossRef]
- Kútna, V.; Uttl, L.; Waltereit, R.; Krištofiková, Z.; Kaping, D.; Petrásek, T.; Hoschl, C.; Ovsepian, S. V. Tuberous Sclerosis (tsc2+/-) Model Eker Rats Reveals Extensive Neuronal Loss with Microglial Invasion and Vascular Remodeling Related to Brain Neoplasia. Neurotherapeutics 2020, 17. [Google Scholar] [CrossRef] [PubMed]
- Zordan, P.; Cominelli, M.; Cascino, F.; Tratta, E.; Poliani, P.L.; Galli, R. Tuberous sclerosis complex-associated CNS abnormalities depend on hyperactivation of mTORC1 and Akt. Journal of Clinical Investigation 2018, 128, 1688–1706. [Google Scholar] [CrossRef]
- Riley, V.A.; Shankar, V.; Holmberg, J.C.; Sokolov, A.M.; Neckles, V.N.; Williams, K.; Lyman, R.; Mackay, T.F.C.; Feliciano, D.M. Tsc2 coordinates neuroprogenitor differentiation. iScience 2023, 26, 108442. [Google Scholar] [CrossRef]
- Zhou, J.; Shrikhande, G.; Xu, J.; Mckay, R.M.; Burns, D.K.; Johnson, J.E.; Parada, L.F. Tsc1 mutant neural stem/progenitor cells exhibit migration deficits and give rise to subependymal lesions in the lateral ventricle. Genes Dev 2011, 25, 1595–1600. [Google Scholar] [CrossRef] [PubMed]
- Feliciano, D.M.; Quon, J.L.; Su, T.; Taylor, M.M.; Bordey, A. Postnatal neurogenesis generates heterotopias, olfactory micronodules and cortical infiltration following single-cell TSC1 deletion. Hum Mol Genet 2012, 21, 799–810. [Google Scholar] [CrossRef] [PubMed]
- Feliciano, D.M.; Bordey, A. TSC-mTORC1 Pathway in Postnatal V-SVZ Neurodevelopment. Biomolecules 2025, 15, 573. [Google Scholar] [CrossRef]
- Holmberg, J.C.; Riley, V.A.; Sokolov, A.M.; Mukherjee, S.; Feliciano, D.M. Protocol for electroporating and isolating murine (sub)ventricular zone cells for single-nuclei omics. STAR Protoc 2024, 5, 103095. [Google Scholar] [CrossRef]
- Mukherjee, S.; Wolan, M.J.; Scott, M.K.; Riley, V.A.; Sokolov, A.M.; Feliciano, D.M. A bitopic mTORC inhibitor reverses phenotypes in a tuberous sclerosis complex model. Sci Rep 2025, 15, 20367. [Google Scholar] [CrossRef]
- Riley, V.A.; Holmberg, J.C.; Sokolov, A.M.; Feliciano, D.M. Tsc2 shapes olfactory bulb granule cell molecular and morphological characteristics. Front Mol Neurosci 2022, 15, 19. [Google Scholar] [CrossRef]
- Lagace, D.C.; Whitman, M.C.; Noonan, M.A.; Ables, J.L.; DeCarolis, N.A.; Arguello, A.A.; Donovan, M.H.; Fischer, S.J.; Farnbauch, L.A.; Beech, R.D.; et al. Dynamic contribution of nestin-expressing stem cells to adult neurogenesis. Journal of Neuroscience 2007, 27, 12623–12629. [Google Scholar] [CrossRef]
- Pollen, A.A.; Bhaduri, A.; Andrews, M.G.; Nowakowski, T.J.; Meyerson, O.S.; Mostajo-Radji, M.A.; Di Lullo, E.; Alvarado, B.; Bedolli, M.; Dougherty, M.L.; et al. Establishing Cerebral Organoids as Models of Human-Specific Brain Evolution. Cell 2019, 176, 743–756.e17. [Google Scholar] [CrossRef]
- Northrup, H.; Krueger, D.A.; Roberds, S.; Smith, K.; Sampson, J.; Korf, B.; Kwiatkowski, D.J.; Mowat, D.; Nellist, M.; Povey, S.; et al. Tuberous sclerosis complex diagnostic criteria update: Recommendations of the 2012 international tuberous sclerosis complex consensus conference. Pediatr Neurol 2013, 49, 243–254. [Google Scholar] [CrossRef]
- Lind, W.A.T. EPILOIA. Med J Aust 1924, 293, 1–5. [Google Scholar] [CrossRef]
- Crithchley, M.; Earl, C.J.C. Tuberose sclerosis and allied conditions. Brain 1932, 55. [Google Scholar] [CrossRef]
- Staley, B.A.; Vail, E.A.; Thiele, E.A. Tuberous sclerosis complex: Diagnostic challenges, presenting symptoms, and commonly missed signs. Pediatrics 2011, 127. [Google Scholar] [CrossRef]
- Krueger, D.A.; Northrup, H.; Krueger, D.A.; Roberds, S.; Smith, K.; Sampson, J.; Korf, B.; Kwiatkowski, D.J.; Mowat, D.; Nellist, M.; et al. Tuberous sclerosis complex surveillance and management: Recommendations of the 2012 international tuberous sclerosis complex consensus conference. Pediatr Neurol 2013, 49, 255–265. [Google Scholar] [CrossRef] [PubMed]
- Adriaensen, M.E.A.P.M.; Schaefer-Prokop, C.M.; Stijnen, T.; Duyndam, D.A.C.; Zonnenberg, B.A.; Prokop, M. Prevalence of subependymal giant cell tumors in patients with tuberous sclerosis and a review of the literature. Eur J Neurol 2009, 16, 691–696. [Google Scholar] [CrossRef]
- Fujiwara, S.; Takaki, T.; Hikita, T.; Nishio, S. Subependymal giant-cell astrocytoma associated with tuberous sclerosis - Do subependymal nodules grow? Child’s Nervous System 1989, 5. [Google Scholar]
- Morimoto, K.; Mogami, H. Sequential CT study of subependymal giant-cell astrocytoma associated with tuberous sclerosis. Case report. J Neurosurg 1986, 65. [Google Scholar] [CrossRef]
- Cuccia, V.; Zuccaro, G.; Sosa, F.; Monges, J.; Lubienieky, F.; Taratuto, A.L. Subependymal giant cell astrocytoma in children with tuberous sclerosis. Child’s Nervous System 2003, 19, 232–243. [Google Scholar] [CrossRef] [PubMed]
- Zordan, P.; Cominelli, M.; Cascino, F.; Tratta, E.; Poliani, P.L.; Galli, R. Tuberous sclerosis complex-associated CNS abnormalities depend on hyperactivation of mTORC1 and Akt. Journal of Clinical Investigation 2018, 128, 1688–1706. [Google Scholar] [CrossRef]
- Jóźwiak, S.; Nabbout, R.; Curatolo, P. Management of subependymal giant cell astrocytoma (SEGA) associated with tuberous sclerosis complex (TSC): Clinical recommendations. European Journal of Paediatric Neurology 2013, 17. [Google Scholar] [CrossRef]
- Roth, J.; Roach, E.S.; Bartels, U.; Jóźwiak, S.; Koenig, M.K.; Weiner, H.L.; Franz, D.N.; Wang, H.Z. Subependymal giant cell astrocytoma: Diagnosis, screening, and treatment. Recommendations from the international tuberous sclerosis complex consensus conference 2012. Pediatr Neurol 2013, 49, 439–444. [Google Scholar] [CrossRef]
- Northrup, H.; Aronow, M.E.; Bebin, E.M.; Bissler, J.; Darling, T.N.; de Vries, P.J.; Frost, M.D.; Fuchs, Z.; Gosnell, E.S.; Gupta, N.; et al. Updated International Tuberous Sclerosis Complex Diagnostic Criteria and Surveillance and Management Recommendations. Pediatr Neurol 2021, 123. [Google Scholar] [CrossRef]
- Jansen, A.C.; Belousova, E.; Benedik, M.P.; Carter, T.; Cottin, V.; Curatolo, P.; Dahlin, M.; D’Amato, L.; D’Augères, G.B.; De Vries, P.J.; et al. Clinical characteristics of subependymal giant cell astrocytoma in tuberous sclerosis complex. Front Neurol. 2019. [Google Scholar] [CrossRef]
- Goto, J.; Talos, D.M.; Klein, P.; Qin, W.; Chekaluk, Y.I.; Anderl, S.; Malinowska, I.A.; Di Nardo, A.; Bronson, R.T.; Chan, J.A.; et al. Regulable neural progenitor-specific Tsc1 loss yields giant cells with organellar dysfunction in a model of tuberous sclerosis complex. Proc Natl Acad Sci U S A 2011, 108. [Google Scholar] [CrossRef]
- Jansen, A.C.; Belousova, E.; Benedik, M.P.; Carter, T.; Cottin, V.; Curatolo, P.; D’Amato, L.; D’Augères, G.B.; De Vries, P.J.; Ferreira, J.C.; et al. Newly diagnosed and growing subependymal giant cell astrocytoma in adults with tuberous sclerosis complex: Results from the International TOSCA Study. Front Neurol 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Kotulska, K.; Borkowska, J.; Mandera, M.; Roszkowski, M.; Jurkiewicz, E.; Grajkowska, W.; Bilska, M.; Jóźwiak, S. Congenital subependymal giant cell astrocytomas in patients with tuberous sclerosis complex. Child’s Nervous System 2014, 30, 2037–2042. [Google Scholar] [CrossRef] [PubMed]
- Knudson, A.G. Mutation and cancer: statistical study of retinoblastoma. Proc Natl Acad Sci U S A 1971, 68, 820–823. [Google Scholar] [CrossRef]
- Henske, E.P.; Wessner, L.L.; Golden, J.; Scheithauer, B.W.; Vortmeyer, A.O.; Zhuang, Z.; Klein-Szanto, A.J.P.; Kwiatkowski, D.J.; Yeung, R.S. Loss of tuberin in both subependymal giant cell astrocytomas and angiomyolipomas supports a two-hit model for the pathogenesis of tuberous sclerosis tumors. American Journal of Pathology 1997, 151, 1639–1647. [Google Scholar]
- Giannikou, K.; Zhu, Z.; Kim, J.; Winden, K.D.; Tyburczy, M.E.; Marron, D.; Parker, J.S.; Hebert, Z.; Bongaarts, A.; Taing, L.; et al. Subependymal giant cell astrocytomas are characterized by mTORC1 hyperactivation, a very low somatic mutation rate, and a unique gene expression profile. Modern Pathology 2021, 34. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.A.; Zhang, H.; Roberts, P.S.; Jozwiak, S.; Wieslawa, G.; Lewin-Kowalik, J.; Kotulska, K.; Kwiatkowski, D.J. Pathogenesis of tuberous sclerosis subependymal giant cell astrocytomas: Biallelic inactivation of TSC1 or TSC2 leads to mTOR activation. J Neuropathol Exp Neurol 2004, 63, 1236–1242. [Google Scholar] [CrossRef] [PubMed]
- Bongaarts, A.; Giannikou, K.; Reinten, R.J.; Anink, J.J.; Mills, J.D.; Jansen, F.E.; Spliet, W.G.M.; den Dunnen, W.F.A.; Coras, R.; Blümcke, I.; et al. Subependymal giant cell astrocytomas in Tuberous Sclerosis Complex have consistent TSC1/TSC2 biallelic inactivation, and no BRAF mutations. Oncotarget 2017, 8, 95516–95529. [Google Scholar] [CrossRef]
- Feliciano, D.M.; Su, T.; Lopez, J.; Platel, J.C.; Bordey, A. Single-cell Tsc1 knockout during corticogenesis generates tuber-like lesions and reduces seizure threshold in mice. Journal of Clinical Investigation 2011, 121, 1596–1607. [Google Scholar] [CrossRef]
- Feliciano, D.M.; Zhang, S.; Quon, J.L.; Bordey, A. Hypoxia-inducible factor 1a is a Tsc1-regulated survival factor in newborn neurons in tuberous sclerosis complex. Hum Mol Genet 2013, 22, 1725–1734. [Google Scholar] [CrossRef]
- Rushing, G. V.; Brockman, A.A.; Bollig, M.K.; Leelatian, N.; Mobley, B.C.; Irish, J.M.; Ess, K.C.; Fu, C.; Ihrie, R.A. Location-dependent maintenance of intrinsic susceptibility to mTORC1-driven tumorigenesis. Life Sci Alliance 2019, 2. [Google Scholar] [CrossRef] [PubMed]
- Holmberg, J.C.; Shankar, V.; Lyman, R.A.; Mackay, T.F.C.; Feliciano, D.M. Single Nuclei Transcriptomics reveals cellular diversity in TSC Subependymal Giant Cell Astrocytomas. iScience 2025, 113389. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



