Submitted:
16 December 2025
Posted:
17 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
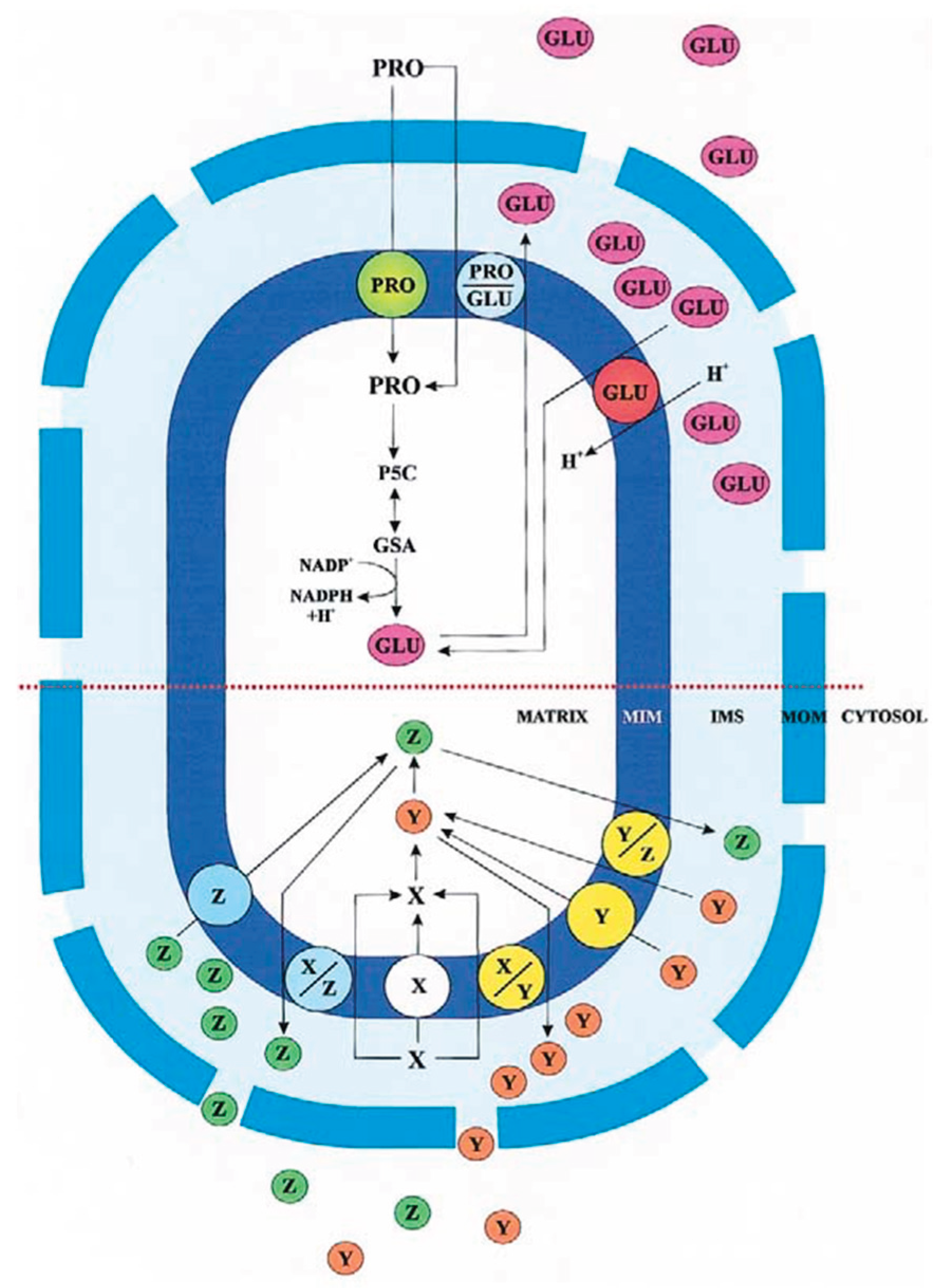
2. The Role of the Intermembrane Space in Metabolite Trafficking
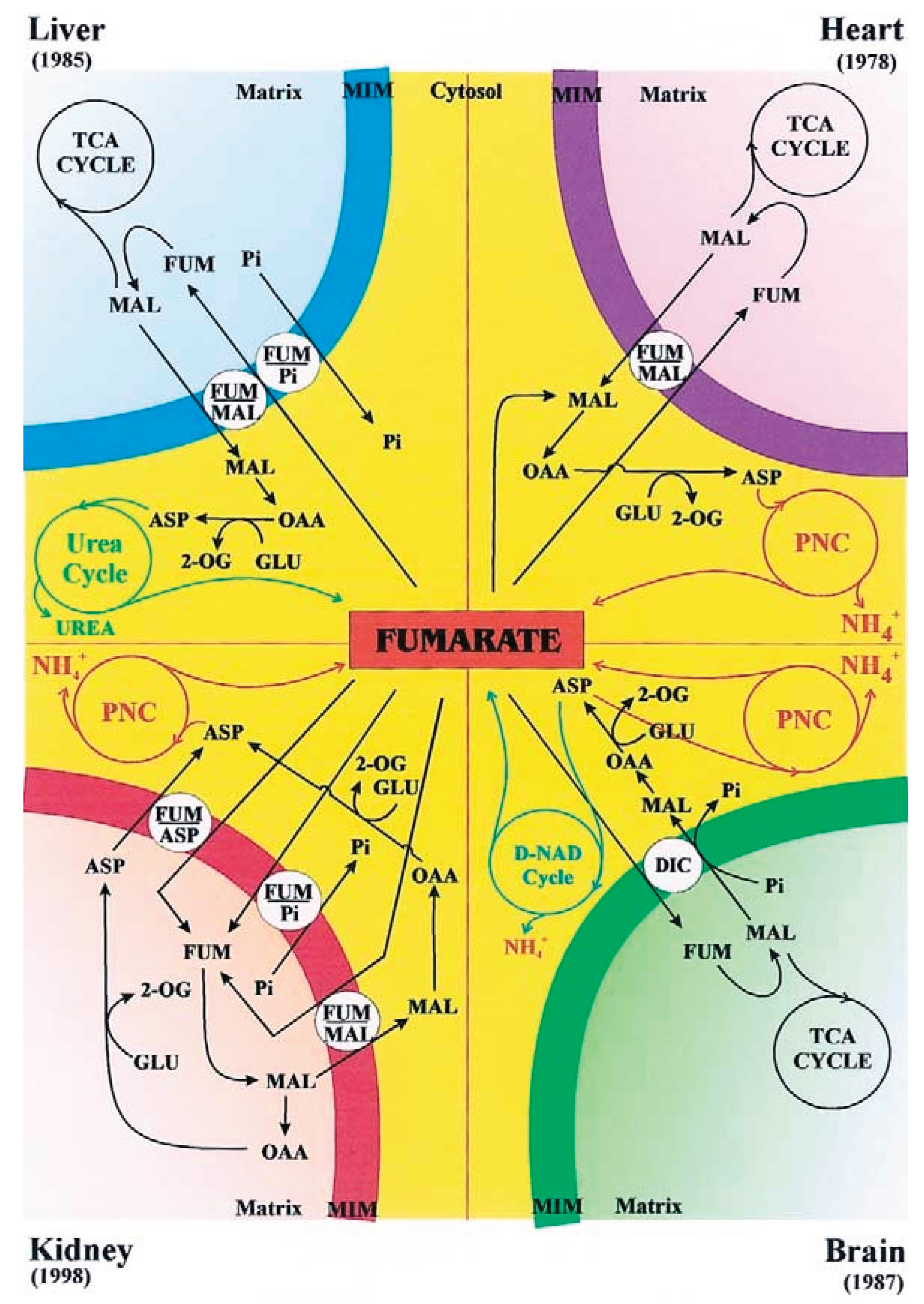
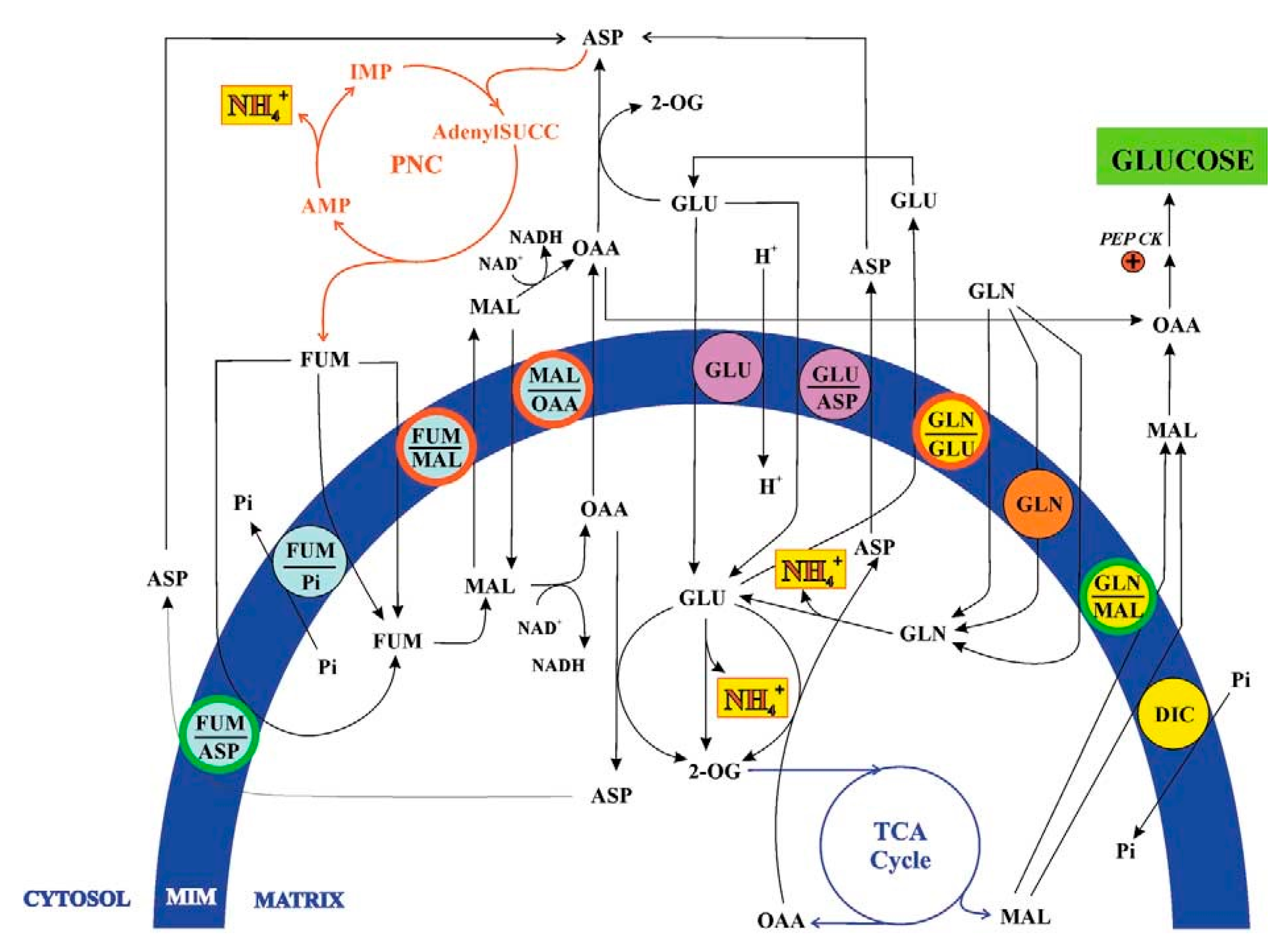
3. Fumarate Transport in Mitochondria
3. Glutamine Transport in Mitochondria
4. Phosphoenolpyruvate Transport and Mitochondrial Pyruvate Kinase
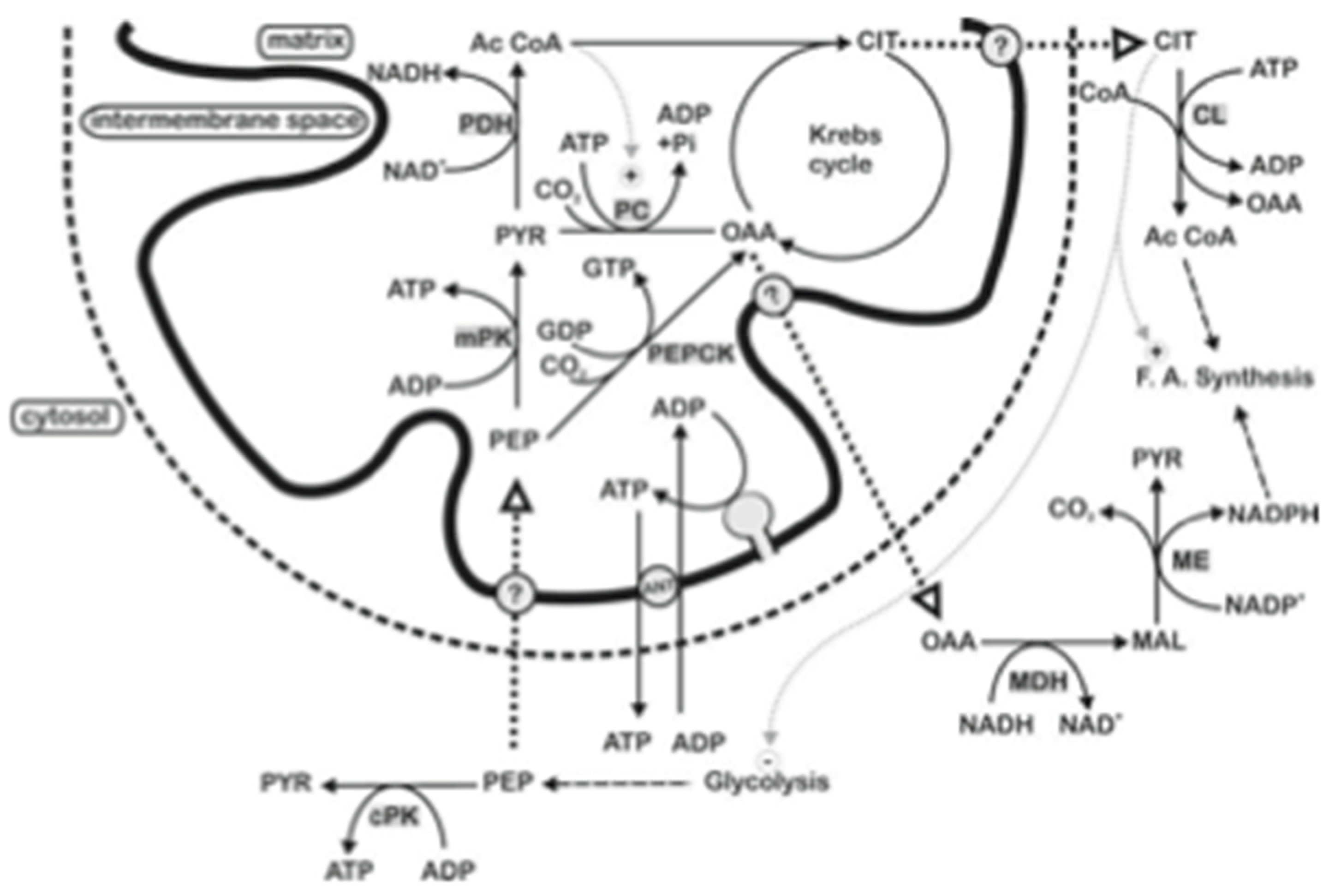
5. Conclusions
References
- Passarella, S.; Atlante, A.; Valenti, D.; de Bari, L. The role of mitochondrial transport in energy metabolism. Mitochondrion 2003, 2, 319–343. [Google Scholar] [CrossRef]
- Atlante, A.; Passarella, S.; Pierro, P.; Di Martino, C.; Quagliariello, E. The mechanism of proline/glutamate antiport in rat kidney mitochondria. Energy dependence and glutamate-carrier involvement. Eur J Biochem 1996, 241, 171–177. [Google Scholar] [CrossRef]
- Passarella, S.; Schurr, A.; Portincasa, P. Mitochondrial Transport in Glycolysis and Gluconeogenesis: Achievements and Perspectives. Int. J. Mol. Sci. 2021, 22, 12620. [Google Scholar] [CrossRef]
- Passarella, S. Revisiting concepts of mitochondrial transport and energy metabolism in health and cancer. Academia Biology in press. [Google Scholar]
- Akiba, T.; Hiraga, K.; Tuboi, S. Intracellular distribution of fumarase in various animals. J Biochem 1984, 96, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Chappell, J.B.; Haarhoff, K.N. The Penetration of the Mitochondrial Membrane by Anions and Cations; Slater, E.C., Kaniuga, Z., Wojtczak, L., Eds.; Academic Press: London, UK, 1966; pp. 75–91. [Google Scholar]
- Passarella, S.; Fasano, E.; Carrieri, S.; Quagliariello, E. Prime evidenze sperimentali del trasporto del fumarato in mitocondri di cuore di ratto [Initial experimental evidence of fumarate transport in rat heart mitochondria]. Boll Soc Ital Biol Sper. 1978, 54, 40–45. [Google Scholar] [PubMed]
- Passarella, S.; Fasano, E.; Carrieri, S.; Quagliariello, E. Fumarate permeation in rat heart mitochondria. Biochem Biophys Res Commun 1979, 90, 498–505. [Google Scholar] [CrossRef]
- Atlante, A.; Passarella, S.; Giannattasio, S.; Quagliariello, E. Fumarate permeation in rat liver mitochondria: fumarate/malate and fumarate/phosphate translocators. Biochem Biophys Res Commun. 1985, 132, 8–18. [Google Scholar] [CrossRef]
- Passarella, S.; Atlante, A.; Barile, M.; Quagliariello, E. Anion transport in rat brain mitochondria: fumarate uptake via the dicarboxylate carrier. Neurochem Res 1987, 12, 255–264. [Google Scholar] [CrossRef]
- Pallotta, ML.; Fratianni, A.; Passarella, S. Metabolite transport in isolated yeast mitochondria: fumarate/malate and succinate/malate antiports. FEBS Lett 1999, 462, 313–316. [Google Scholar] [CrossRef]
- Atlante, A.; Gagliardi, S.; Passarella, S. Fumarate permeation in normal and acidotic rat kidney mitochondria: fumarate/malate and fumarate/aspartate translocators. Biochem Biophys Res Commun 1998, 24, 711–718. [Google Scholar] [CrossRef] [PubMed]
- Passarella, S.; Atlante, A. Teaching the role of mitochondrial transport in energy metabolism. Biochem Mol Biol Educ 2007, 35, 125–132. [Google Scholar] [CrossRef]
- Arinze, I.J. Facilitating understanding of the purine nucleotide cycle and the one-carbon pool: Part I: The purine nucleotide cycle. Biochem Mol Biol Educ. 2005, 33, 165–168. [Google Scholar]
- Araújo, W.L.; Nunes-Nesi, A.; Fernie, A.R. Fumarate: Multiple functions of a simple metabolite. Phytochemistry 2011, 72, 838–843. [Google Scholar] [CrossRef] [PubMed]
- King, A.; Selak, M.A.; Gottlieb, E. Succinate dehydrogenase and fumarate hydratase: linking mitochondrial dysfunction and cancer. Oncogene 2006, 25, 4675–4682. [Google Scholar] [CrossRef] [PubMed]
- Hajaj, E.; Sciacovelli, M.; Frezza, C.; Erez, A. The context-specific roles of urea cycle enzymes in tumorigenesis. Mol Cell 2021, 81, 3749–3759. [Google Scholar] [CrossRef]
- Keshet, R.; Szlosarek, P.; Carracedo, A.; Erez, A. Rewiring urea cycle metabolism in cancer to support anabolism. Nat Rev Cancer 2018, 18, 634–645. [Google Scholar]
- Ghosh, N.; Mahalanobish, S.; Sil, P.C. Reprogramming of urea cycle in cancer: Mechanism, regulation and prospective therapeutic scopes. Biochem Pharmacol 2024, 228, 116326. [Google Scholar] [CrossRef]
- Schatton, D.; Frezza, C. Fumarate. Trends Endocrinol Metab 2025, S1043-2760(24)00332-1. [Google Scholar] [CrossRef]
- Atlante, A.; Passarella, S.; Minervini, G.M.; Quagliariello, E. Glutamine transport in normal and acidotic rat kidney mitochondria. Arch Biochem Biophys 1994, 15, 369–381. [Google Scholar] [CrossRef]
- Curthoys, N.P.; Watford, M. Regulation of glutaminase activity and glutamine metabolism. Annu Rev Nutr 1995, 15, 133–159. [Google Scholar] [CrossRef]
- Stumvoll, M.; Perriello, G.; Meyer, C.; Gerich, J. Role of glutamine in human carbohydrate metabolism in kidney and other tissues. Kidney Int 1999, 55, 778–792. [Google Scholar] [CrossRef]
- Yoo, H.C.; Park, S.J.; Nam, M.; Kang, J.; Kim, K.; Yeo, J.H.; Kim, J.K.; Heo, Y.; Lee, H.S.; Lee, M.Y.; Lee, C.W.; Kang, J.S.; Kim, Y.H.; Lee, J.; Choi, J.; Hwang, G.S.; Bang, S.; Han, J.M. A Variant of SLC1A5 Is a Mitochondrial Glutamine Transporter for Metabolic Reprogramming in Cancer Cells. Cell Metab 2020, 31, 267–283. [Google Scholar] [CrossRef]
- Stine, Z.E.; Dang, C.V. Glutamine Skipping the Q into Mitochondria. Trends Mol Med 2020, 26, 6–7. [Google Scholar] [CrossRef]
- Newsholme, P.; Lima, M.M.; Procopio, J.; Pithon-Curi, T.C.; Doi, S.Q.; Bazotte, R.B.; Curi, R. Glutamine and glutamate as vital metabolites. Braz J Med Biol Res 2003, 36, 153–163. [Google Scholar] [CrossRef]
- Matés, J.M.; Segura, J.A.; Campos-Sandoval, J.A.; Lobo, C.; Alonso, L.; Alonso, F.J.; Márquez, J. Glutamine homeostasis and mitochondrial dynamics. Int J Biochem Cell Biol 2009, 41, 2051–2061. [Google Scholar] [CrossRef]
- Scalise, M.; Pochini, L.; Galluccio, M.; Indiveri, C. Glutamine transport. From energy supply to sensing and beyond. Biochim Biophys Acta 2016, 1857, 1147–1157. [Google Scholar] [CrossRef]
- Scalise, M.; Pochini, L.; Galluccio, M.; Console, L.; Indiveri, C. Glutamine transport and Mitochondrial Metabolism in Cancer Cell Growth. Front Oncol 2017, 7, 306. [Google Scholar] [CrossRef]
- Kahya, U.; Lukiyanchuk, V.; Gorodetska, I.; Weigel, MM.; Köseer, AS.; Alkan, B.; Savic, D.; Linge, A.; Löck, S.; Peitzsch, M.; Skvortsova, II; Krause, M.; Dubrovska, A. Disruption of glutamine transport uncouples the NUPR1 stress-adaptation program and induces prostate cancer radiosensitivity. Cell Commun Signal 2025, 23, 351–375. [Google Scholar] [CrossRef]
- de Bari, L.; Valenti, D.; Pizzuto, R.; Atlante, A.; Passarella, S. Phosphoenolpyruvate metabolism in Jerusalem artichoke mitochondria. Biochim Biophys Acta 2007, 1767, 281–294. [Google Scholar] [CrossRef]
- Pizzuto, R.; Paventi, G.; Chieppa, G.; Atlante, A.; Passarella, S. Is there a pyruvate kinase in pig liver mitochondria? Ital J Biochem 2007, 56, 270–274. [Google Scholar] [CrossRef] [PubMed]
- Pizzuto, R.; Paventi, G.; Atlante, A.; Passarella, S. Pyruvate kinase in pig liver mitochondria. Arch Biochem Biophys 2010, 495, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Schormann, N.; Hayden, K.L.; Lee, P.; Banerjee, S.; Chattopadhyay, D. An overview of structure, function, and regulation of pyruvate kinases. Protein Sci 2019, 28, 1771–1784. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



