Submitted:
13 December 2025
Posted:
15 December 2025
You are already at the latest version
Abstract
Keywords:
Key Points
Introduction
Physiology of Respiratory Mechanics
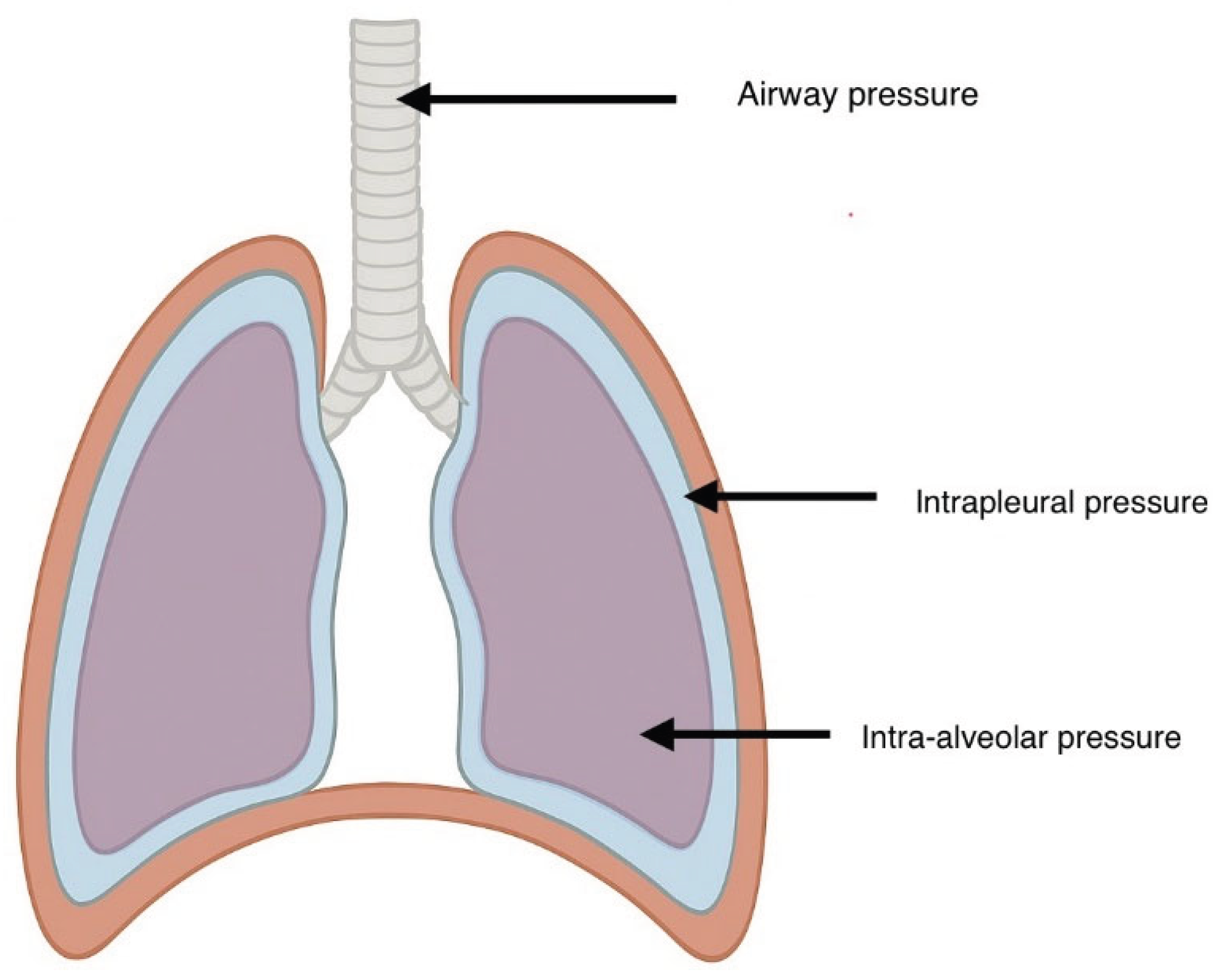
- Airway pressure (Paw): Airway pressure refers to the pressure measured at the airway opening, typically by the ventilator. It reflects the sum of pressures required to overcome both the resistive load of the conducting airways and the elastic recoil of the lung and chest wall. During positive pressure ventilation, Paw rises during inspiration as gas is delivered, and it falls back toward baseline during expiration. Clinically, Paw is easily monitored and forms the basis for measurements such as peak inspiratory pressure (PIP) and plateau pressure (Pplat). In zero flow static states such as during end-inspiratory or end- expiratory holds, airway pressure approximates the alveolar pressure (Palv) [5].
- Pleural Pressure (Ppl): Pleural pressure represents the pressure within the pleural space, which surrounds the lungs. It is the external pressure acting on the lung parenchyma and is a critical determinant of the mechanics of breathing. Because direct measurement of Ppl is impractical at the bedside, it is commonly estimated using esophageal manometry, under the assumption that esophageal pressure approximates pleural pressure. Ppl varies with the respiratory cycle: becoming more negative during spontaneous inspiration, and more positive during positive pressure ventilation [5].
- Trans-pulmonary Pressure (PL):
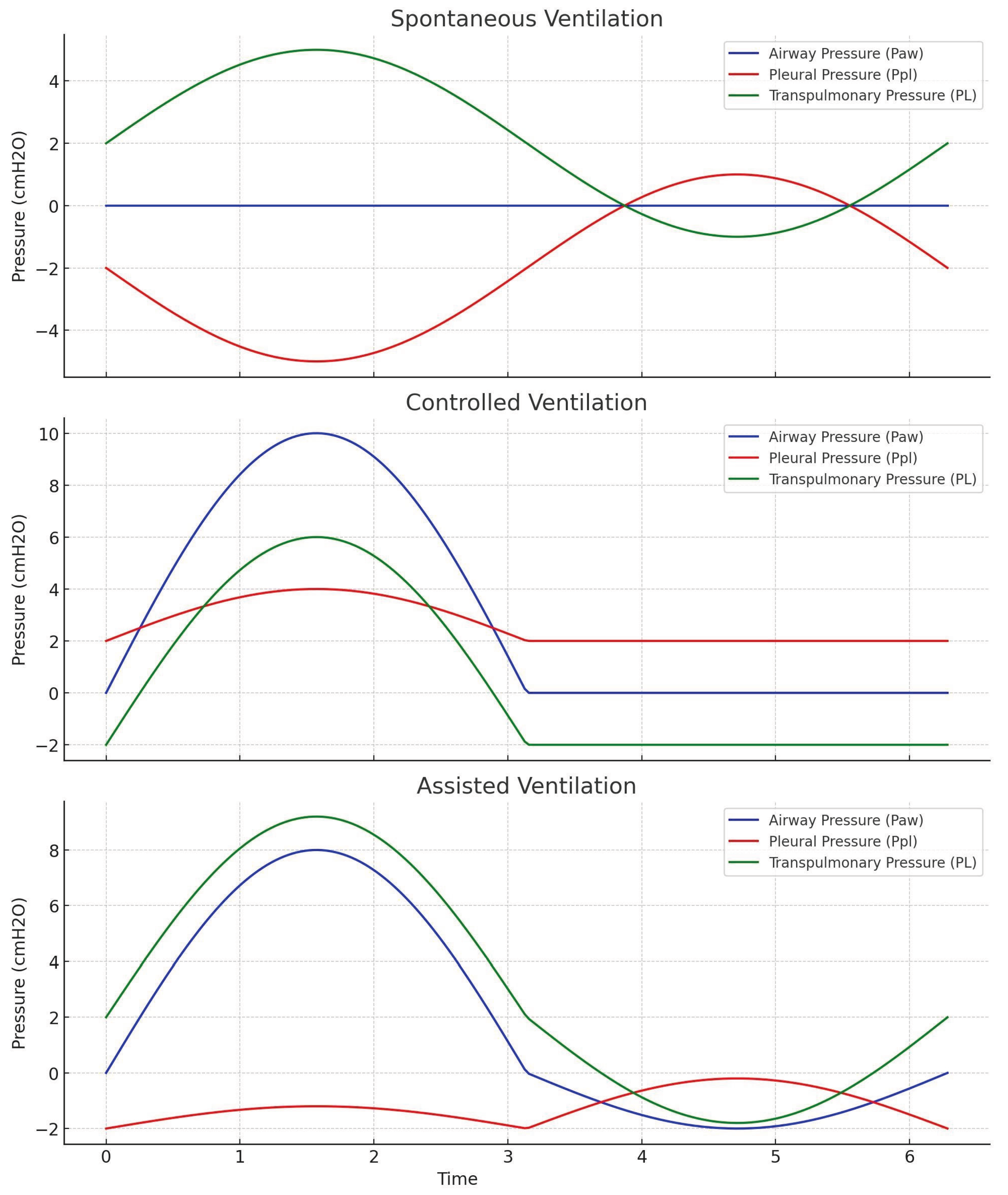
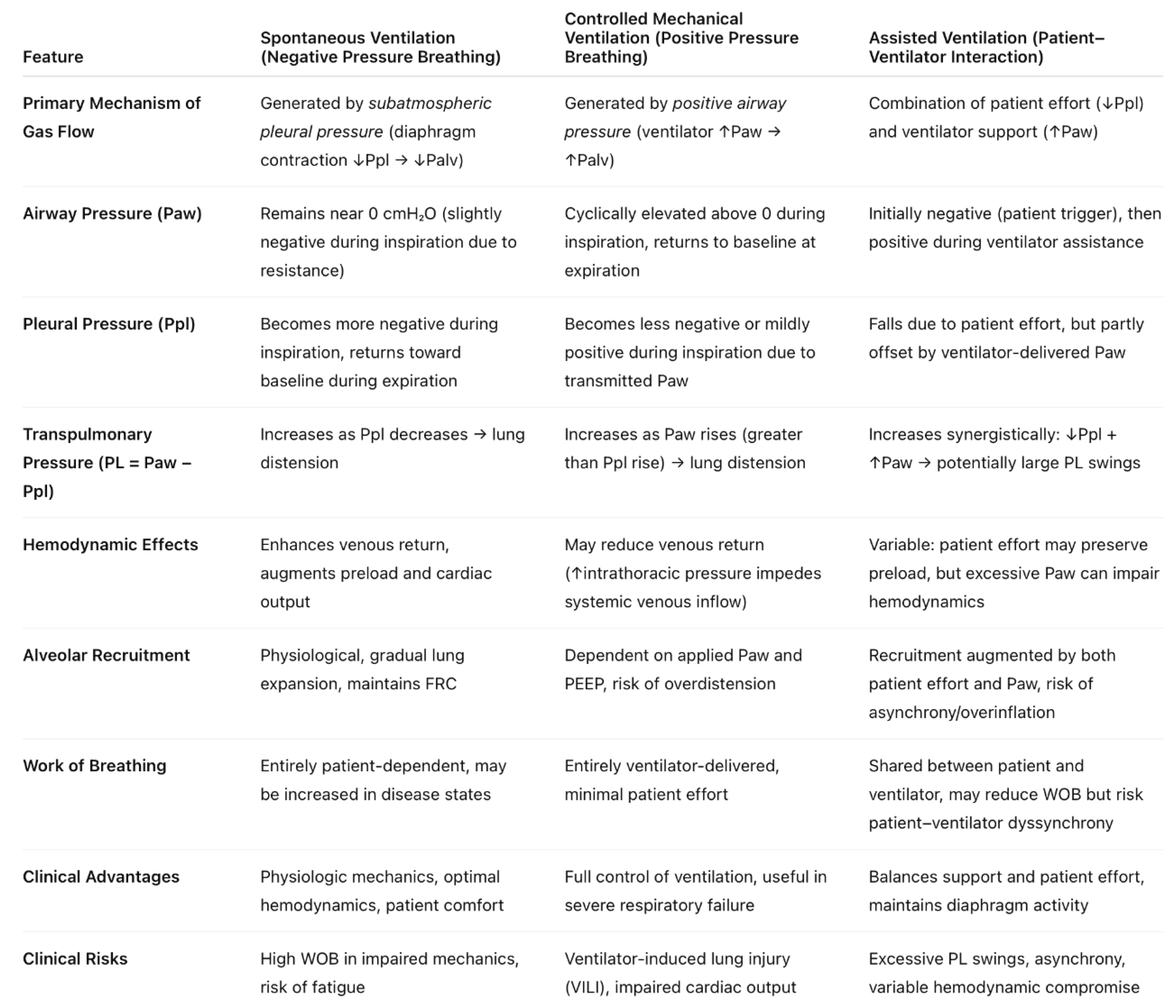
- Airway Pressure (Paw): At the airway opening, Paw remains close to atmospheric pressure (0 cmH₂O) throughout most of the cycle, with only small variations due to resistance in the conducting airways.
- Pleural Pressure (Ppl): Becomes more negative during inspiration due to diaphragmatic contraction and expansion of the thoracic cavity.
- Trans-pulmonary Pressure (PL): Increases during inspiration because Ppl decreases while Paw remains nearly constant. This rising PL generates alveolar expansion and airflow into the lungs.
- Airway Pressure (Paw): Actively raised above baseline during inspiration by the ventilator. This positive pressure overcomes airway resistance and elastic recoil.
- Pleural Pressure (Ppl): Rises slightly (becomes more positive) during inspiration, since part of Paw is transmitted to the pleural space.
- Trans-pulmonary Pressure (PL): Increases because Paw rises more than Ppl, leading to alveolar inflation.
- Airway Pressure (Paw): Initially falls slightly as the patient generates a negative inspiratory effort, which triggers the ventilator. Once triggered, Paw rises as positive pressure is delivered.
- Pleural Pressure (Ppl): Drops due to patient effort, often more negative than in spontaneous breathing alone, then partially countered by the delivered Paw.
- Trans-pulmonary Pressure (PL): May rise synergistically: both falling Ppl (patient effort) and rising Paw (ventilator assistance) increase PL together, leading to enhanced alveolar distension.
2. Static and Dynamic Trans-pulmonary Pressure
- Static PL provides insight into alveolar distending pressure and the risk of over distension under set ventilatory conditions.
- Dynamic PL reflects the combined mechanical forces during tidal breathing and is useful for understanding the interaction of airflow, resistance, and patient effort with ventilator-delivered pressure.
3. Stress, Strain, and Ventilator-Associated Lung Injury (VALI)
- Stress refers to the distending force per unit area applied to lung tissue. Clinically, this is represented by the trans-pulmonary pressure (PL), which reflects the difference between alveolar and pleural pressures. It is the direct mechanical load experienced by the alveolar walls and interstitium during ventilation [13].
- Strain denotes the resultant deformation of the lung parenchyma, expressed as the ratio of the change in lung volume (ΔV) to the reference resting volume, usually the functional residual capacity (FRC). Thus, strain quantifies how much the lung is inflated relative to its baseline size, independent of absolute volume [13].
- Esophageal varices, due to the risk of rupture and life-threatening hemorrhage.
- Coagulopathy, which increases the risk of bleeding from mucosal trauma.
- Severe facial or basilar skull fractures, where transnasal passage of the catheter may be hazardous [18].
4. Equipment and Catheters for Esophageal Pressure Monitoring
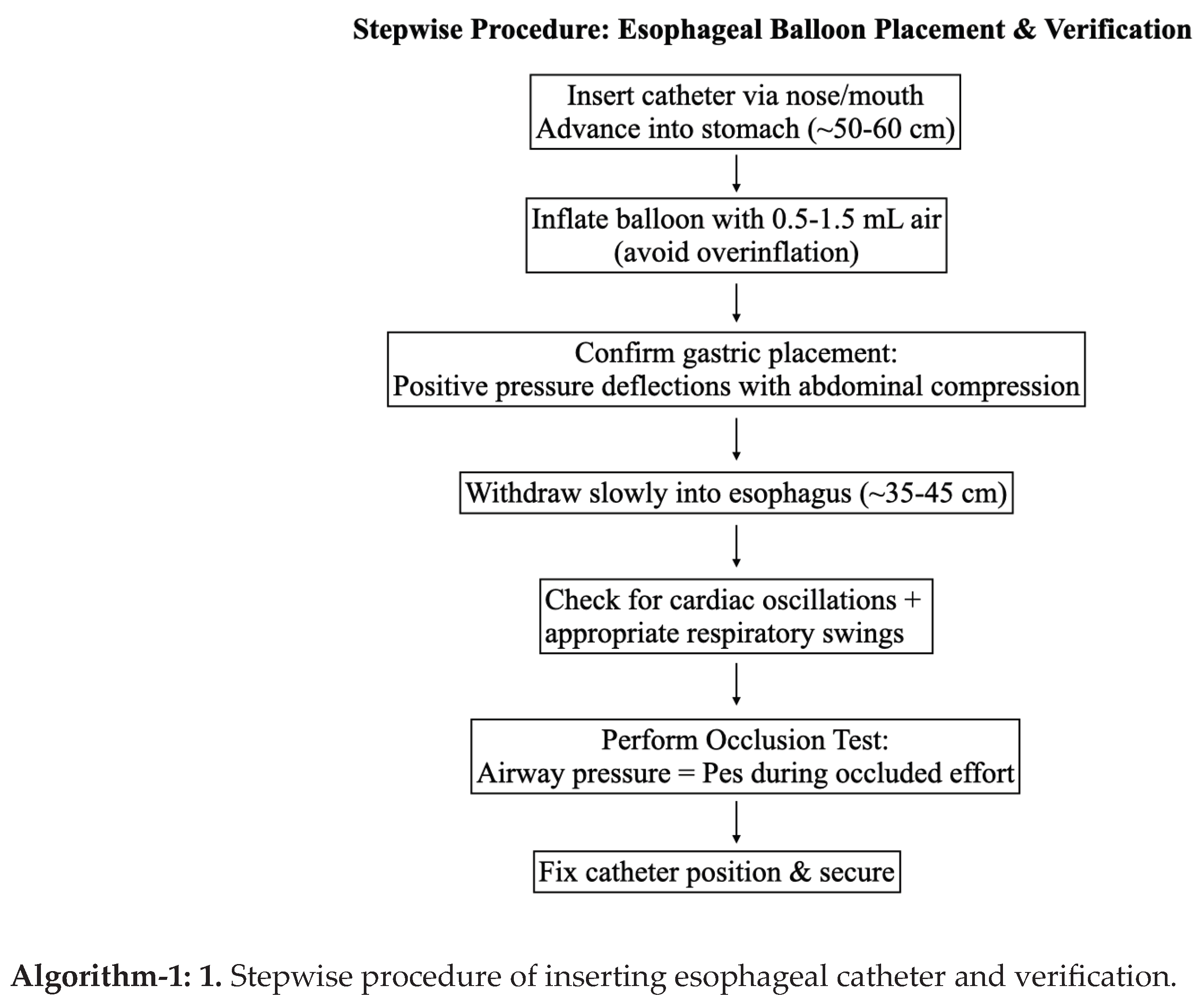
5. Technique: Insertion and Positioning
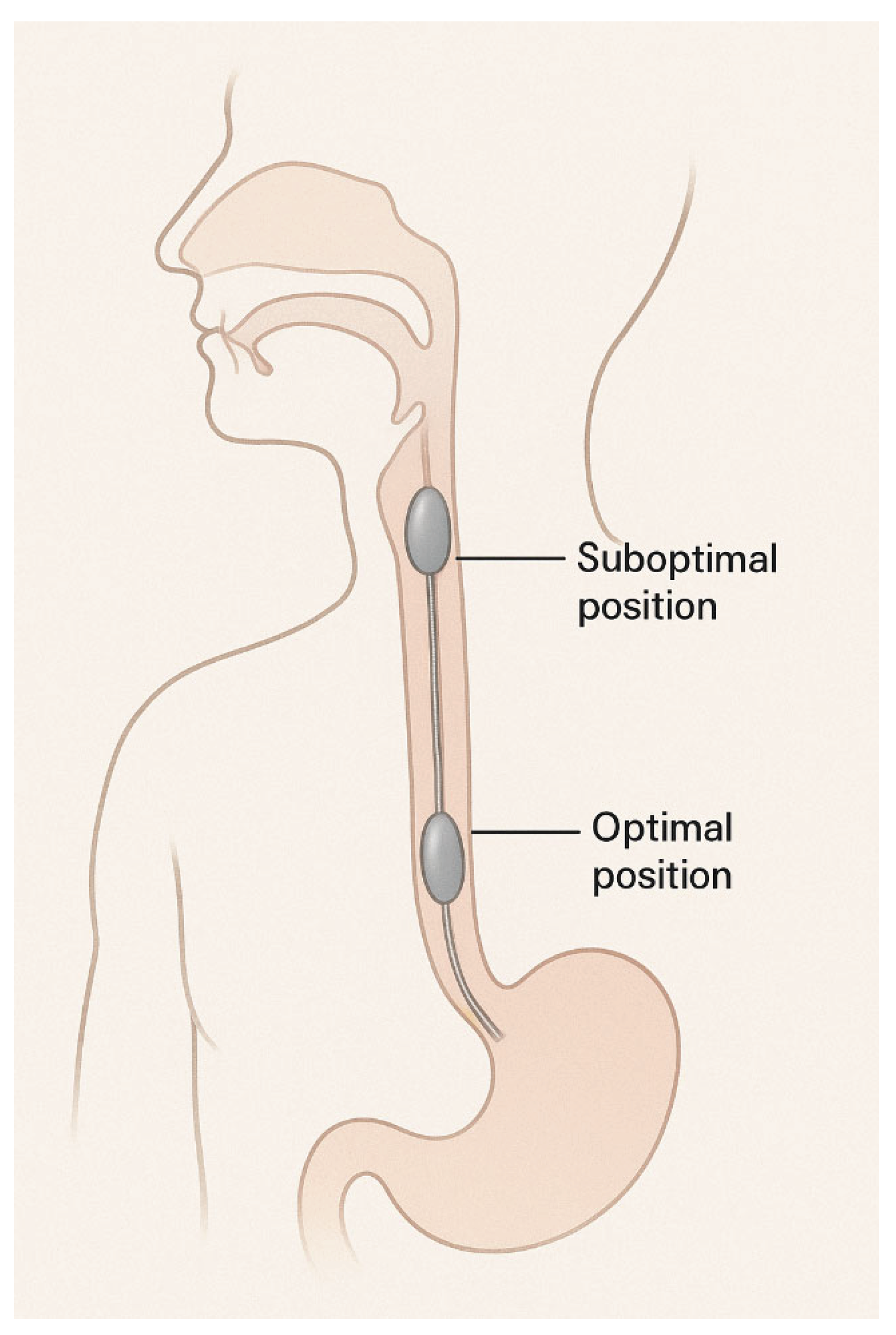
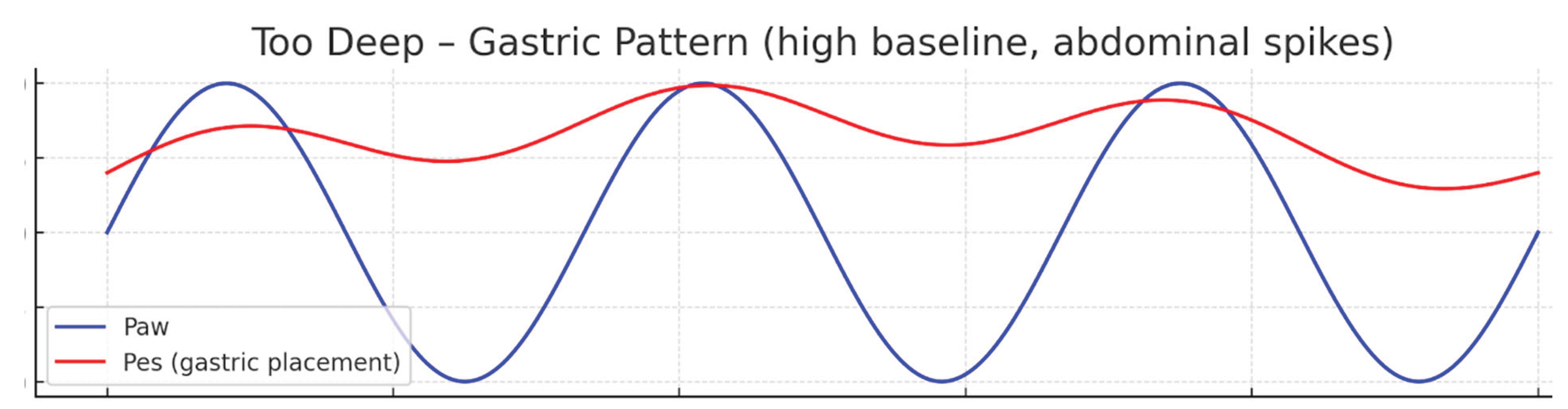
- Cooper catheter: Typically inflated with 2 mL of air and then deflated to approximately 1.2 mL [20].
- Nutrivent catheter: Inflated with 4 mL of air and deflated to approximately 1.5 mL [21].
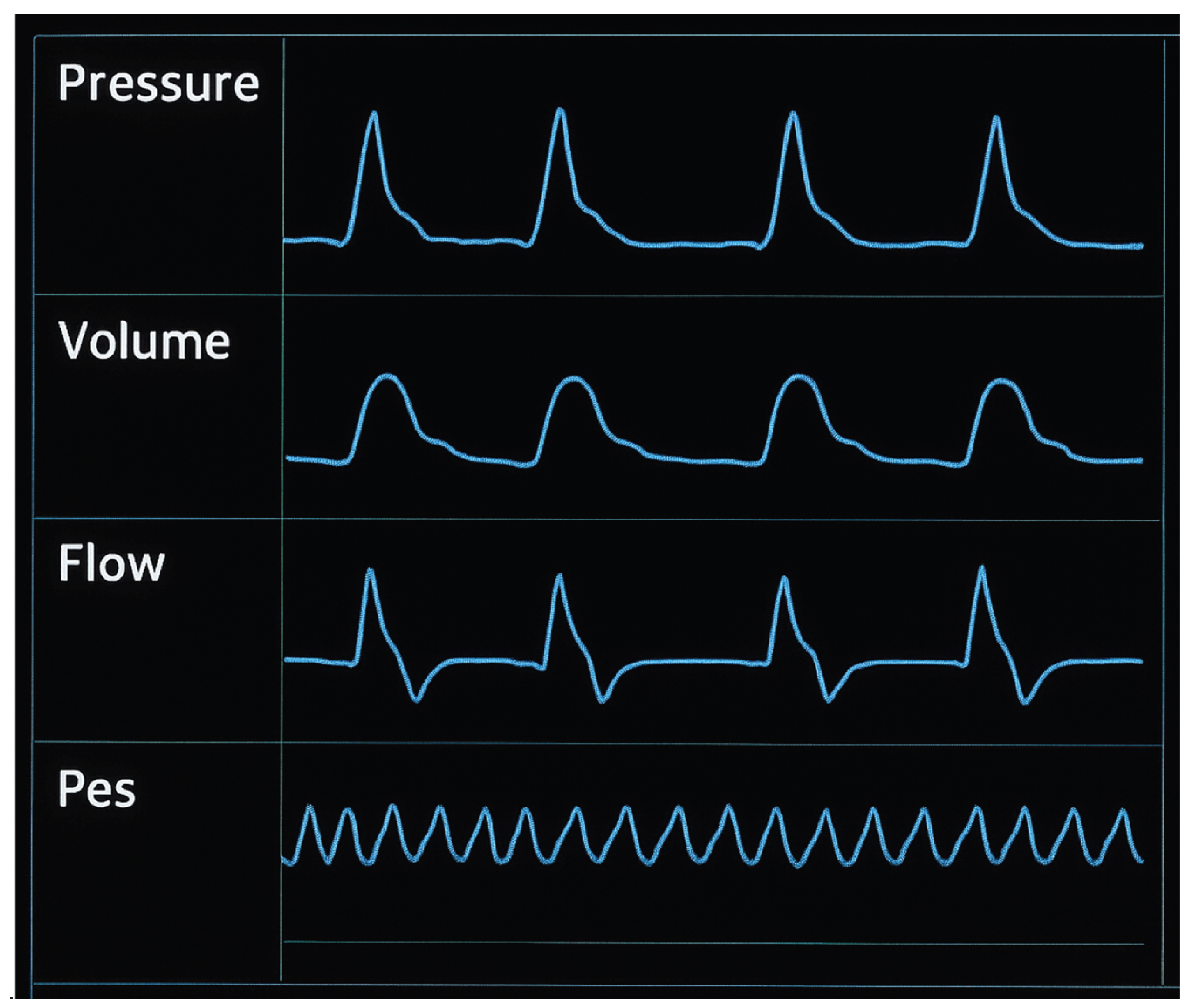
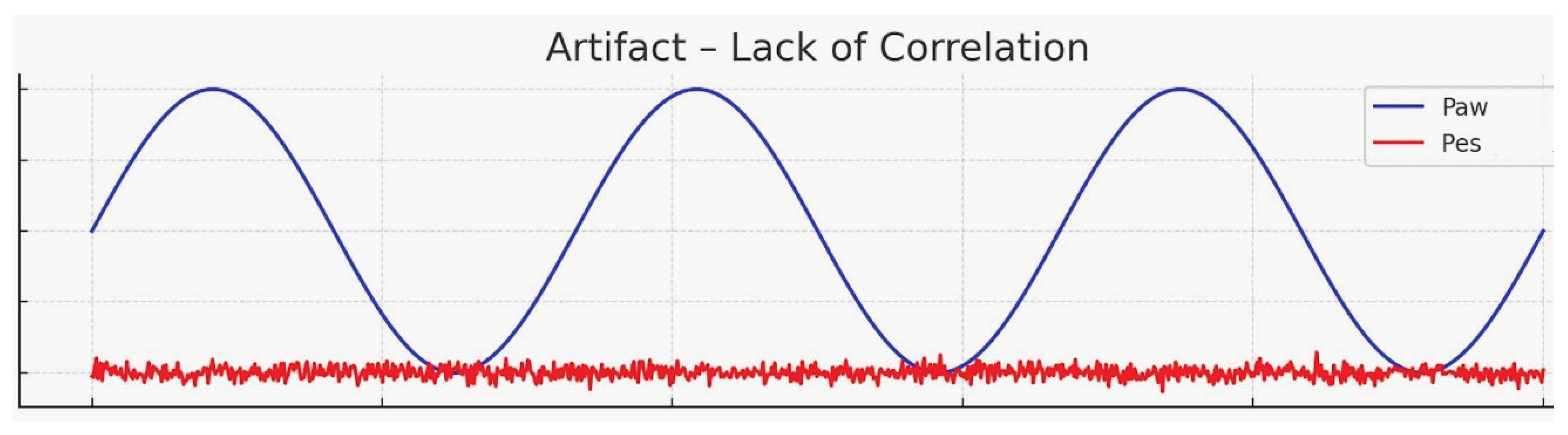
- Cardiac oscillations
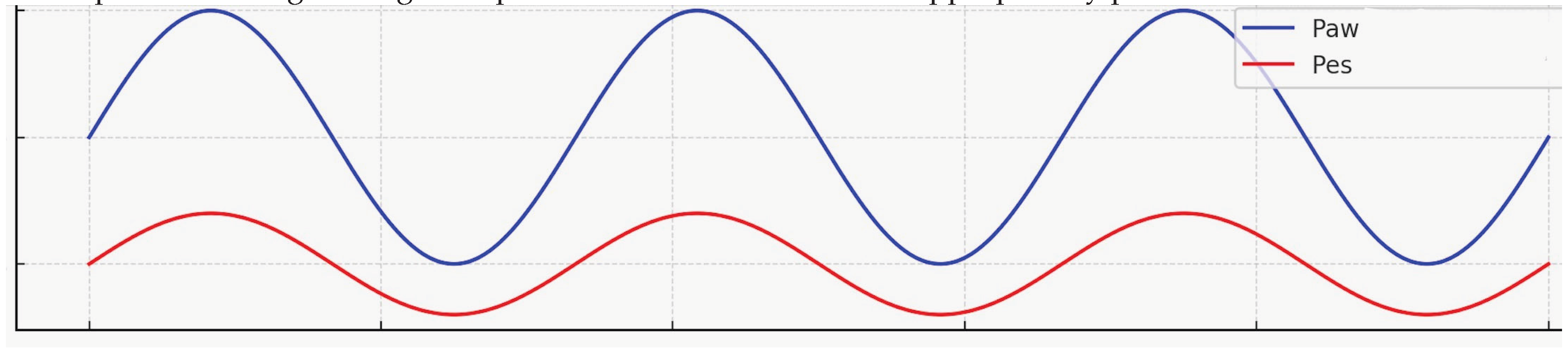
- Comparison of airway and esophageal pressure waveforms
- Occlusion test: ΔPaw/ΔPes ratio 0.8–1.2 (thoracic compression or inspiratory effort during occlusion).
- Zero system to atmosphere.
- Fully inflate balloon, then gradually deflate to manufacturer-recommended volume (Vbest).
- Examples:
- Cooper: inflate 2 ml → deflate to 1.2 ml
- Nutrivent: inflate 4 ml → deflate to 1.5 ml
- Vbest defined as balloon volume yielding maximal ΔPes with minimal artifact.
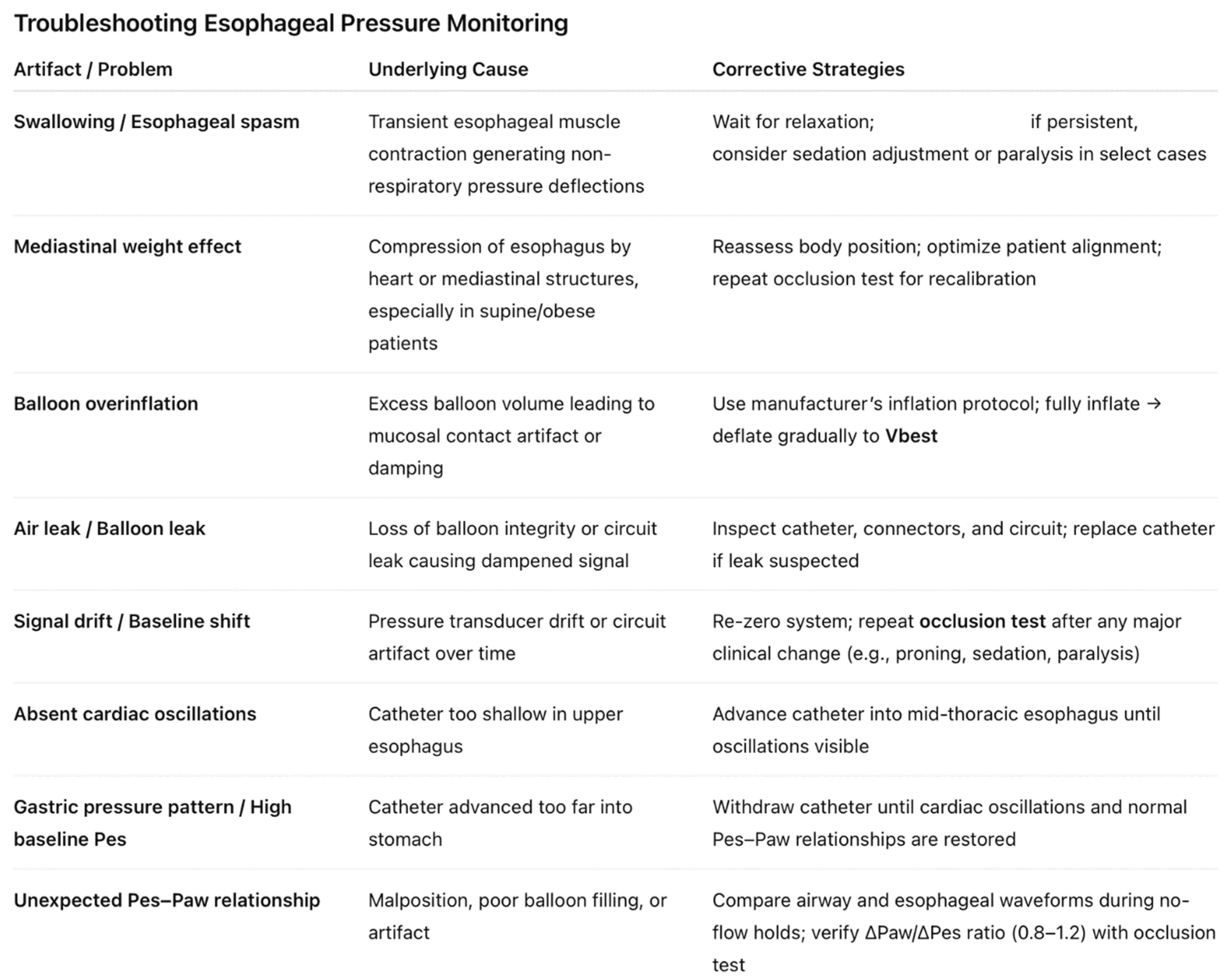
- Large cardiac oscillations → reposition or slightly ramp patient.
- Esophageal spasm → transient artifact, sometimes refractory to paralysis.
- Loss of signal → balloon leak or migration.
- Prone position or Paralysis: may need recalibration.
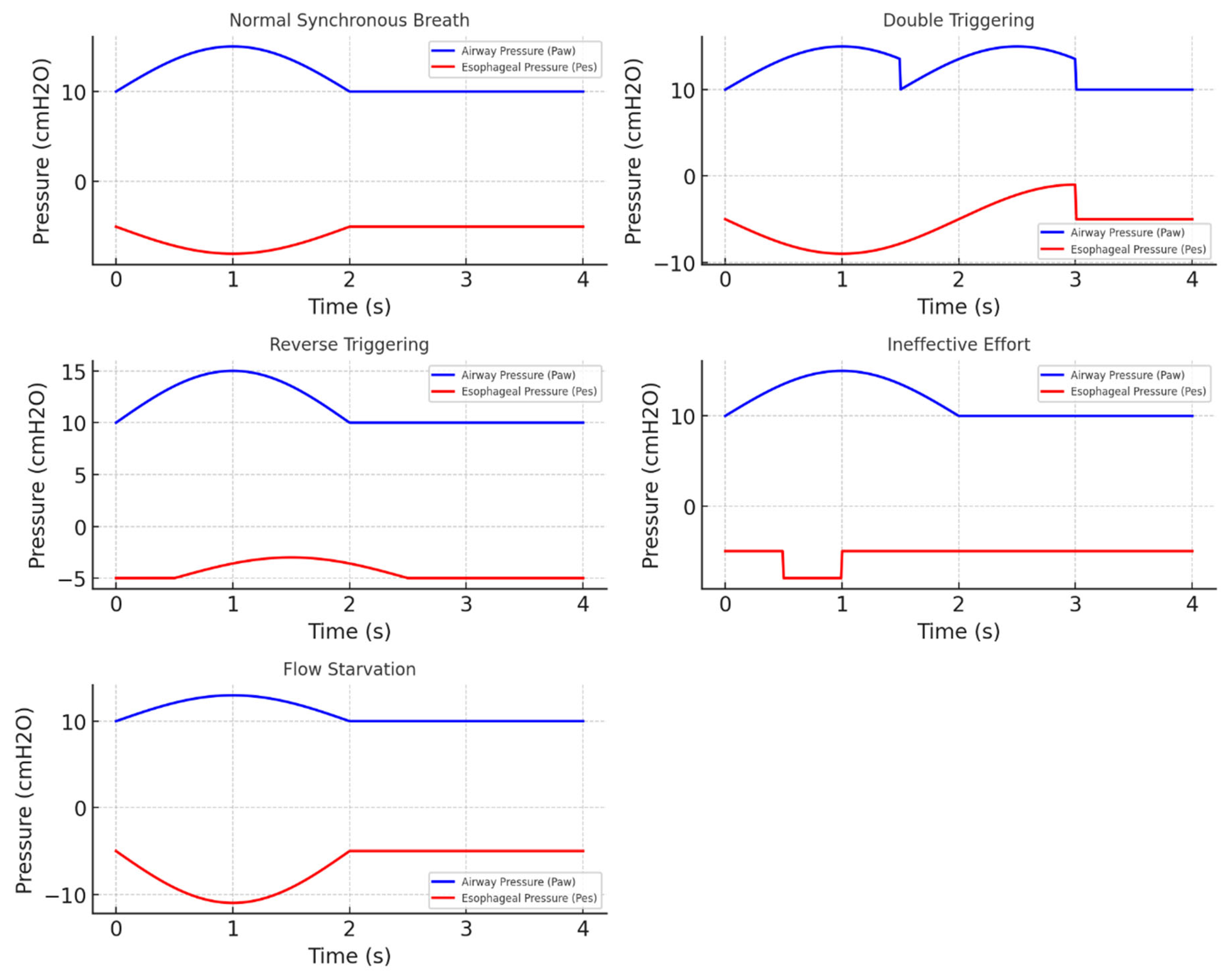
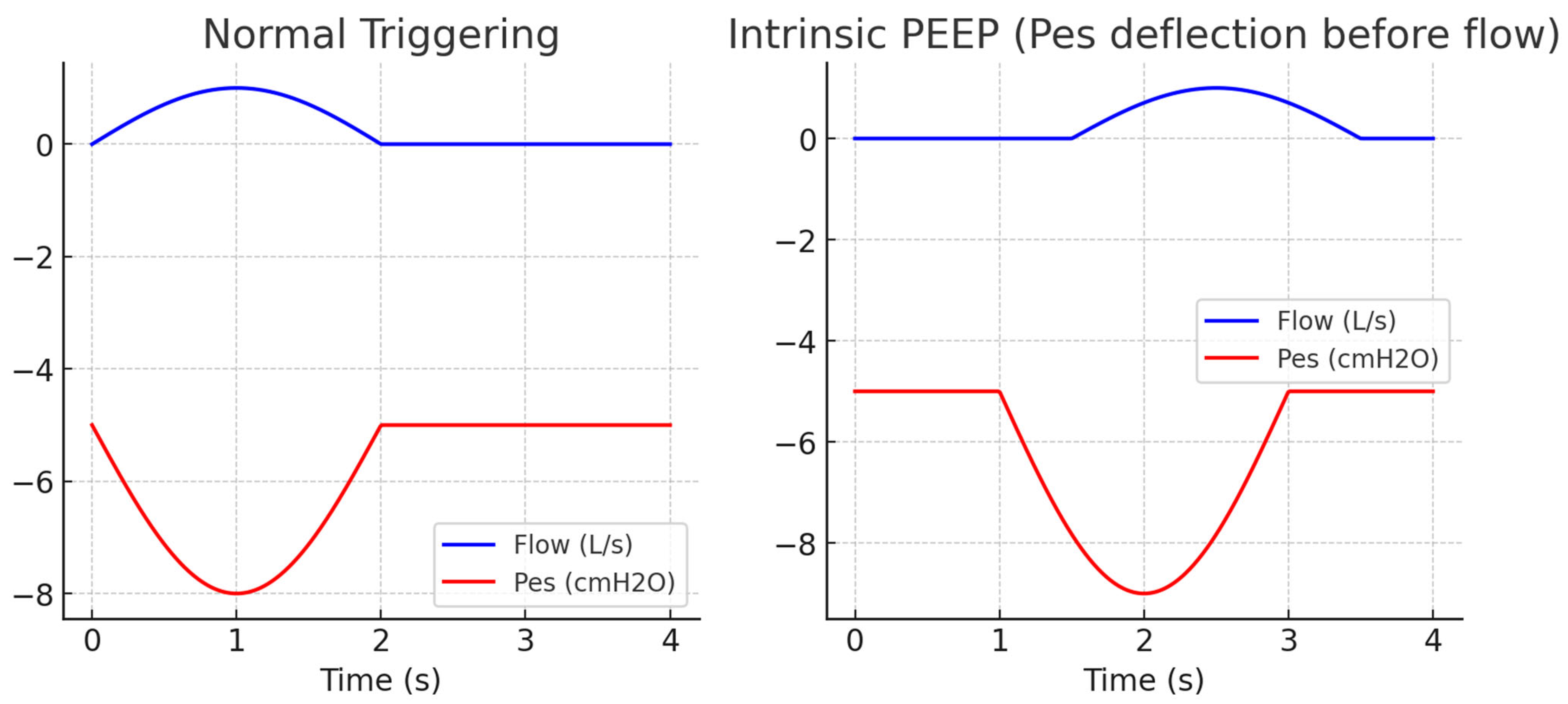
- High levels of positive end-expiratory pressure (PEEP) or intrinsic PEEP (“auto-PEEP”), which impose an inspiratory threshold load that the patient cannot consistently overcome.
- Over-assistance or deep sedation, leading to reduced patient drive and delayed or insufficient inspiratory effort relative to the ventilator’s trigger window.
- Short inspiratory times or high cycling thresholds, resulting in early termination of mechanical inspiration and difficulty for the patient to reinitiate a breath.
- Fixed inspiratory flow delivery in volume-controlled modes that is inadequate relative to patient demand.
- Excessively low flow settings or slow rise time in pressure-controlled or pressure-support modes, delaying the attainment of sufficient inspiratory pressure.
- Increased respiratory drive, such as in hypoxemia, hypercapnia, metabolic acidosis, or discomfort, amplifying the disparity between demand and supply [31].
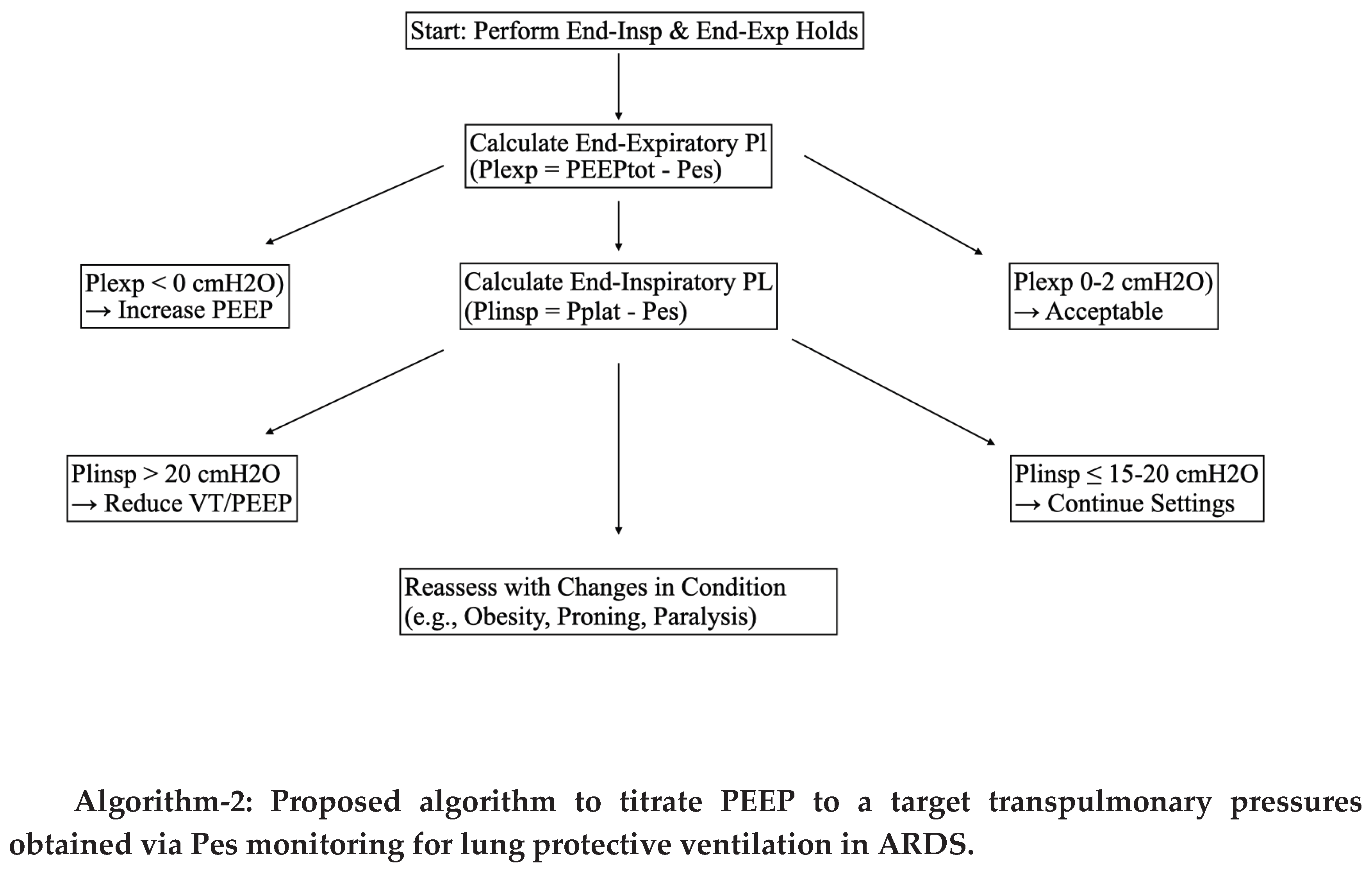
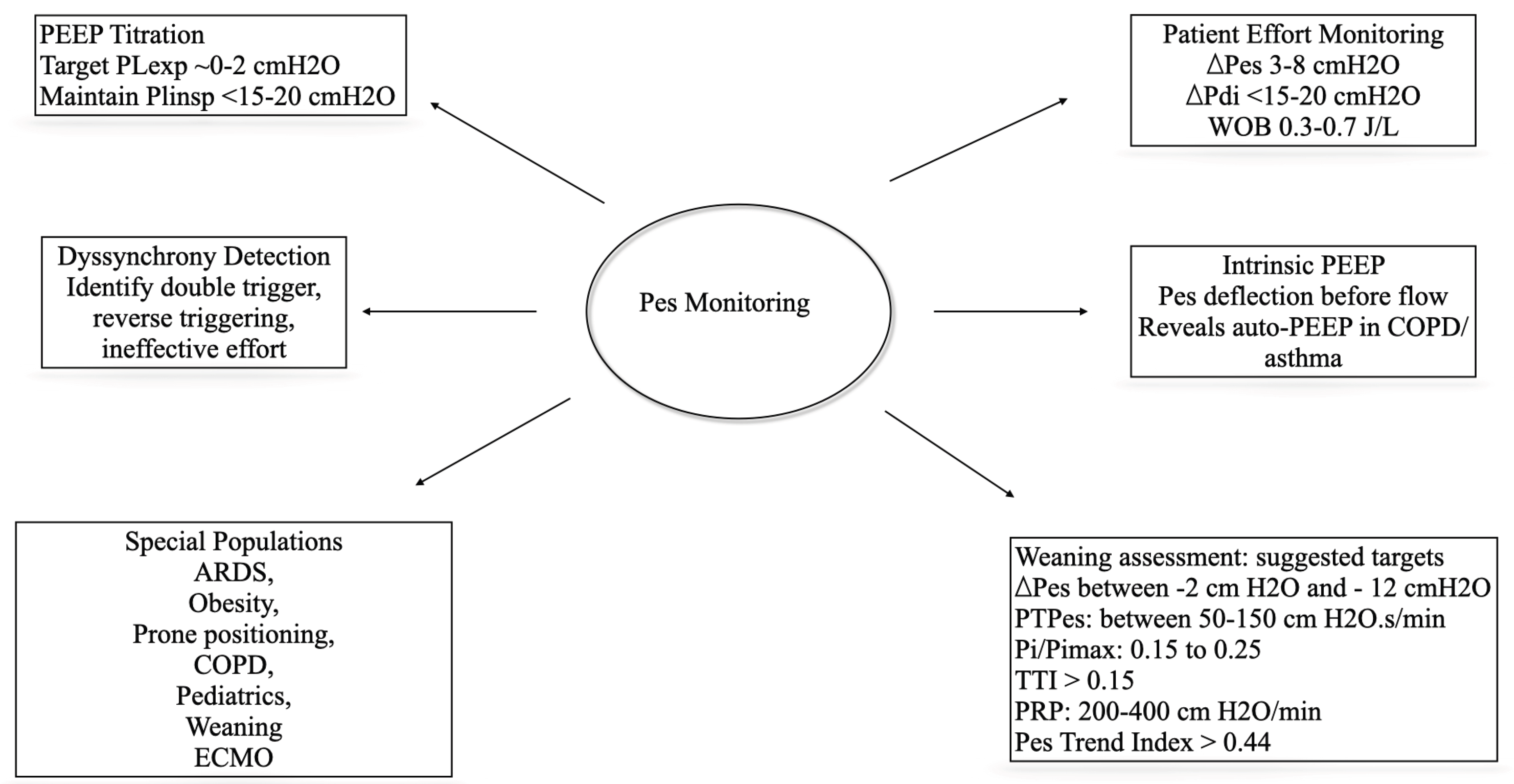
- Methodology: In the intervention arm, PEEP was adjusted to achieve a transpulmonary pressure at end-expiration (PLexp) of 0 to +2 cmH₂O, thereby preventing alveolar collapse.
- Findings: The Pes-guided group demonstrated:
- Improved oxygenation (higher PaO₂/FiO₂ ratios).
- Better respiratory system compliance.
- A trend toward reduced mortality, though not powered for survival as a primary endpoint.
- Significance: This trial provided proof-of-concept that Pes-guided PEEP titration could physiologically optimize ventilation and potentially improve clinical outcomes.
- Methodology: Patients with moderate-to-severe ARDS were randomized to Pes-guided PEEP titration (targeting PLexp 0–6 cmH₂O) versus a high-PEEP ARDSNet strategy.
- Findings:
- No significant difference in the primary outcome of 28-day mortality or ventilator-free days between groups.
- Both groups had similar safety profiles, with no excess of barotrauma or hemodynamic compromise.
- Oxygenation and compliance improvements observed in EPVent were not consistently replicated, possibly because the high-PEEP control arm already approximated physiologically appropriate PEEP levels.
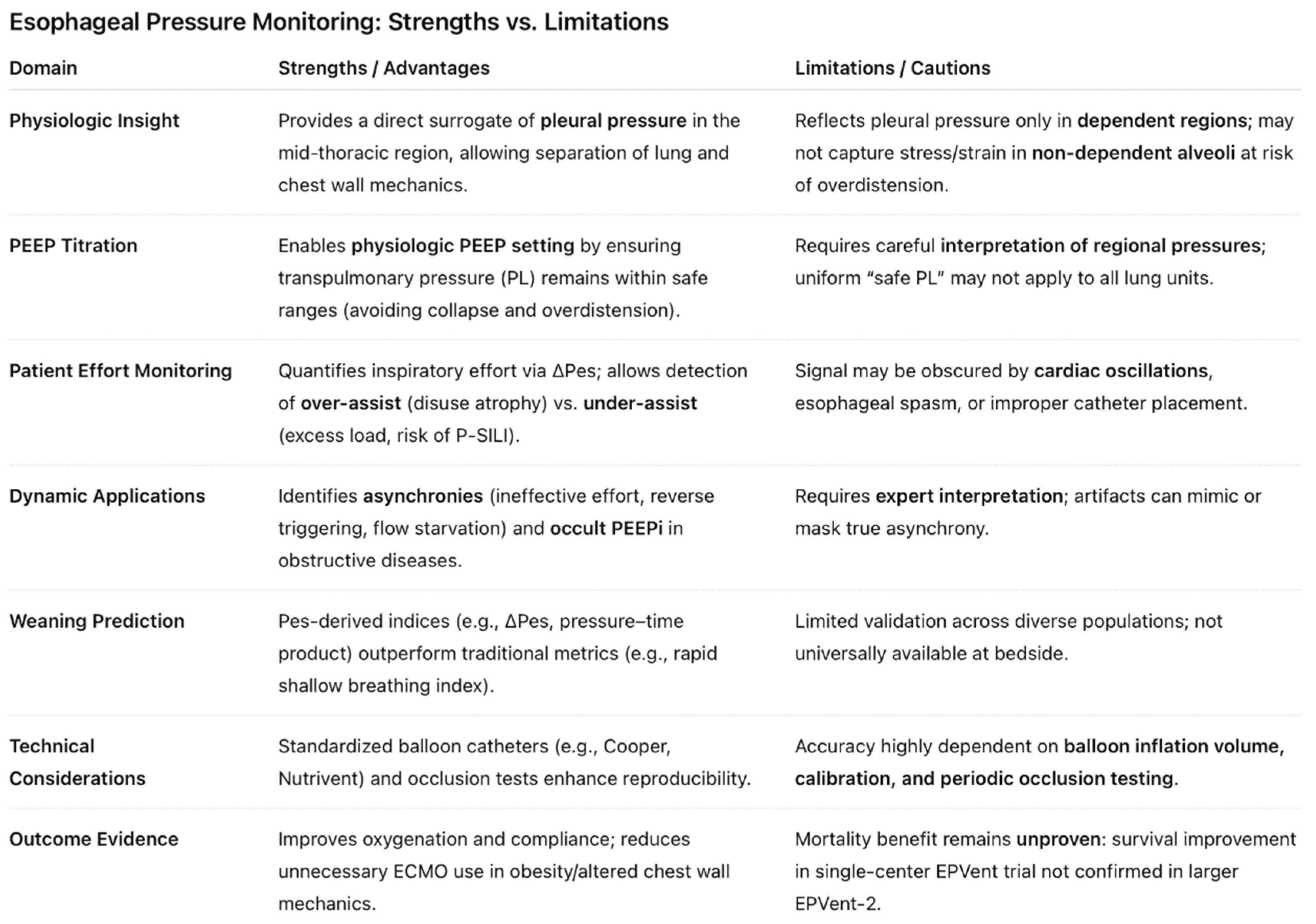
- Significance: EPVent-2 suggested that while Pes-guided titration is safe, its benefit may be most pronounced compared with low-PEEP strategies, whereas its advantage over high-PEEP strategies is less clear. Patients in Pes guided PEEP required less rescue therapies and prone position was considered as a rescue therapy rather than a standard of care.
4. Optimization of Tidal Volume
- Pplat = 32 cmH₂O
- Pes = 25 cmH₂O
- PLinsp = Pplat – Pes = 7 cmH₂O
- Pplat = 25 cmH₂O
- Pes = 5 cmH₂O
- PLinsp = 20 cmH₂O
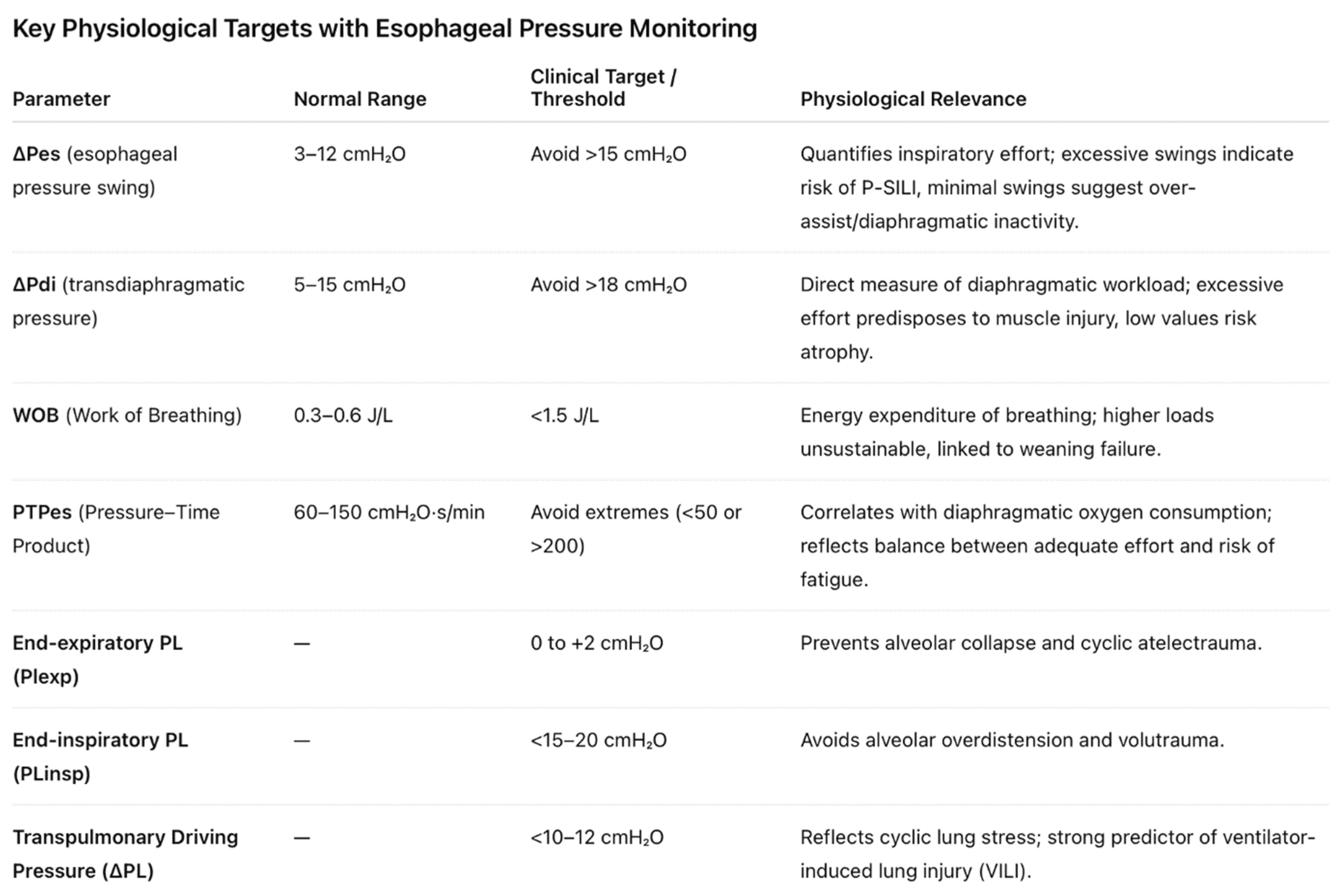
- End-inspiratory transpulmonary pressure (PLinsp): Preferably maintained <15 cmH₂O, though values up to <20 cmH₂O may be acceptable in selected cases.
- Transpulmonary driving pressure (ΔPL): Ideally limited to <10–12 cmH₂O, paralleling evidence linking driving pressure to ventilator-induced lung injury (VILI).
5. Quantifying Patient Effort in Assisted Ventilation
- Suggested Optimal range: A ΔPes of 3–12 cmH₂O is generally considered to represent physiologically appropriate effort. Within this range, the patient contributes to ventilation without incurring undue respiratory muscle strain or alveolar stress.
- Excessive effort: A ΔPes >15 cmH₂O indicates markedly elevated inspiratory drive. Such vigorous efforts can amplify transpulmonary pressures, precipitating regional overdistension, increased shear stress, and ultimately P-SILI.
- Suggested Optimal range: 5–15 cmH₂O.
- Excessive effort: >18 cmH₂O, signaling risk of diaphragmatic injury and increased metabolic load.
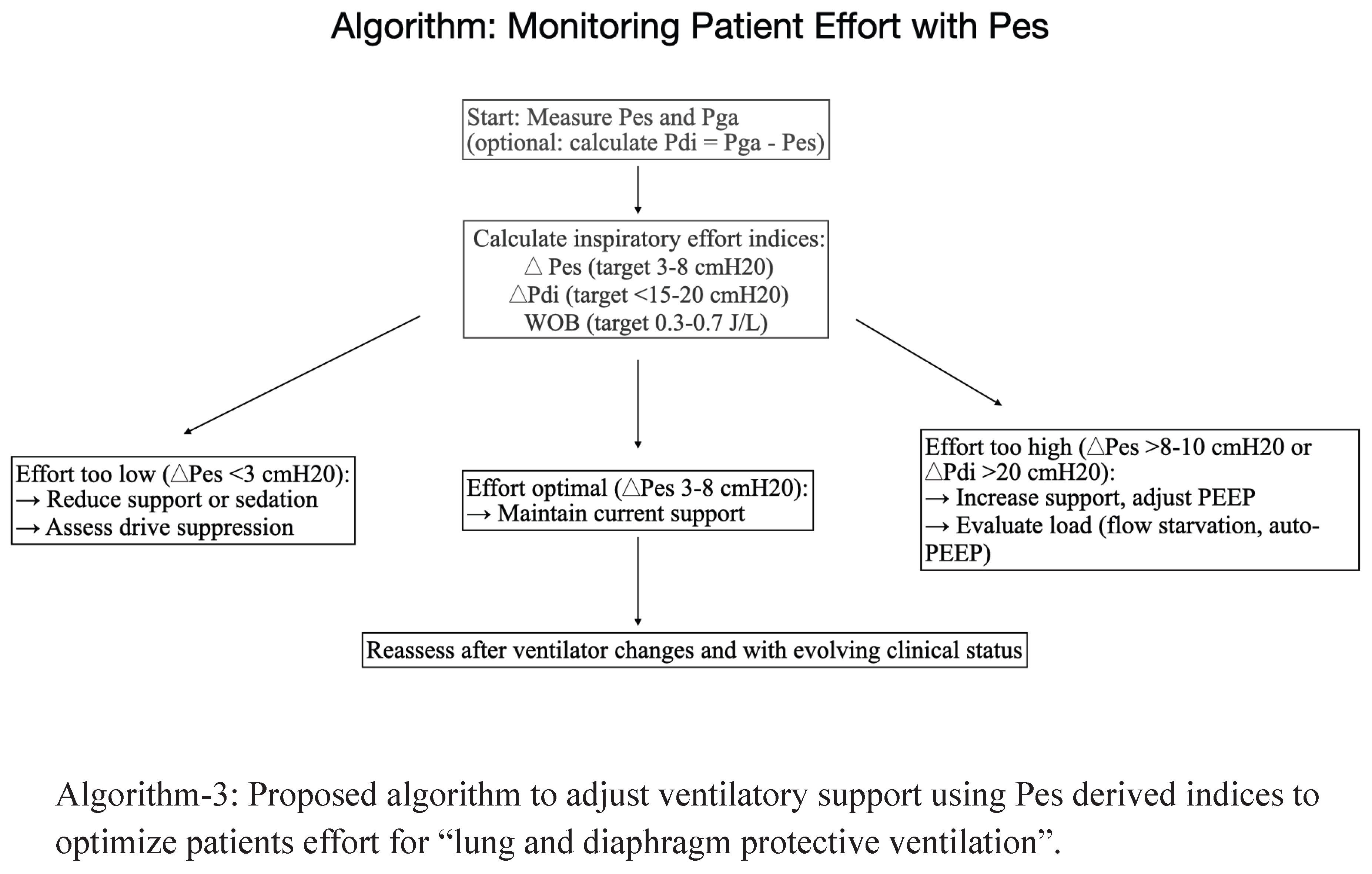
6. Work of Breathing
- Suggested target range: 60–150 cmH₂O·s/min.
- Low values indicate over-assist and underuse of respiratory muscles.
- High values (>200 cmH₂O·s/min) denote unsustainable loads, predictive of weaning failure and impending fatigue.
7. Weaning from mechanical ventilation
- ΔPes (Swing in Esophageal Pressure): Directly measures patient inspiratory effort during spontaneous breathing trials (SBTs). This is calculated as the difference between lowest Pes value just before the beginning and during inspiration. This is considered as a surrogate of muscle power, as this does not incorporate recoil of the chest wall.
- 2.
- PTPes (Esophageal Pressure–Time Product): This is a time based integral of Pmus (pressure generated by the inspiratory muscles) which captures both the magnitude and duration of inspiratory effort, making it a robust indicator of the energetic cost of breathing. This is calculated as an area bounded by the negative esophageal pressure during inspiration and the chest wall elastance slope multiplied by the respiratory rate. This seems to co-relate with the oxygen consumption of respiratory muscles better than the work of breathing.
- 3.
- Pi/Pimax: This is the ratio of change in esophageal pressure during inspiration to the maximum change during occlusion.
- 4.
- Tension-time index (TTI): calculated as a ratio of (Pdi/Pdimax) to (TI/Ttot). This is the ratio of fractional diaphragmatic pressure to the inspiratory duty cycle.
- 5.
- 6.
- Trend Index: Unlike RSBI (RR/VT), which ignores effort, TI incorporates ΔPes, providing a more direct measure of patient work. This is a composite Pes-derived parameter that integrates ΔPes swings in the first 9-10 minutes, reflecting the trajectory of effort rather than a single time-point measurement. Progressive increases in ΔPes or PTPes during an SBT (even if within acceptable ranges at initiation) may signal fatigue and impending failure, allowing earlier intervention [64,65,66].
- X1 = max (0, 7.411 –∆Pes 9);
- X2 = max (0, ∆Pes 9- 5.967);
- X3 = max (0, ∆Pes 1-0.094) * max(0, ∆Pes 9-7.411);
- X4 = max (0, ∆Pes 1 + 1.679) * max(0, ∆Pes 9-10.729)
- ∆Pes 9 represents the estimated value of Pes at the ninth-to-tenth minute transition, and ∆Pes 1 represents the slope of the swings in Pes throughout the first minute
8. Hemodynamic Assessment
9. Utility in special populations
- Phasic Pga rises during expiration and a positive end-expiratory Pdi (Pga–Pes).
- Abdominal paradox or visible abdominal muscle recruitment.
- End-inspiratory stress (PLinsp). Use paralysis or deep sedation (at least during measurement) to eliminate patient effort, then titrate VT/pressure support to keep PLinsp low (commonly ≤10–12 cmH₂O, and even 5–10 cmH₂O in the early “ultraprotective” phase).
- End-expiratory stability (Plexp). Adjust PEEP to maintain Plexp ≈ 0 to +2 cmH₂O, preventing cyclic collapse without overdistending non-dependent lung.
- Driving pressure. Minimize ΔPL (PLinsp − Plexp)—ideally <10–12 cmH₂O. With sweep gas handling CO₂, accept very low VT (3–4 mL/kg PBW or less) and lower respiratory rates as needed to achieve these PL targets.
- Obesity/high IAP. Expect higher measured Paw at any given PL because Pes is elevated. With Pes guidance, accept higher Pplat/Paw (and often higher PEEP) so long as PL targets are respected—a strategy that sustains recruitment in dependent lung without exceeding safe distending pressures.
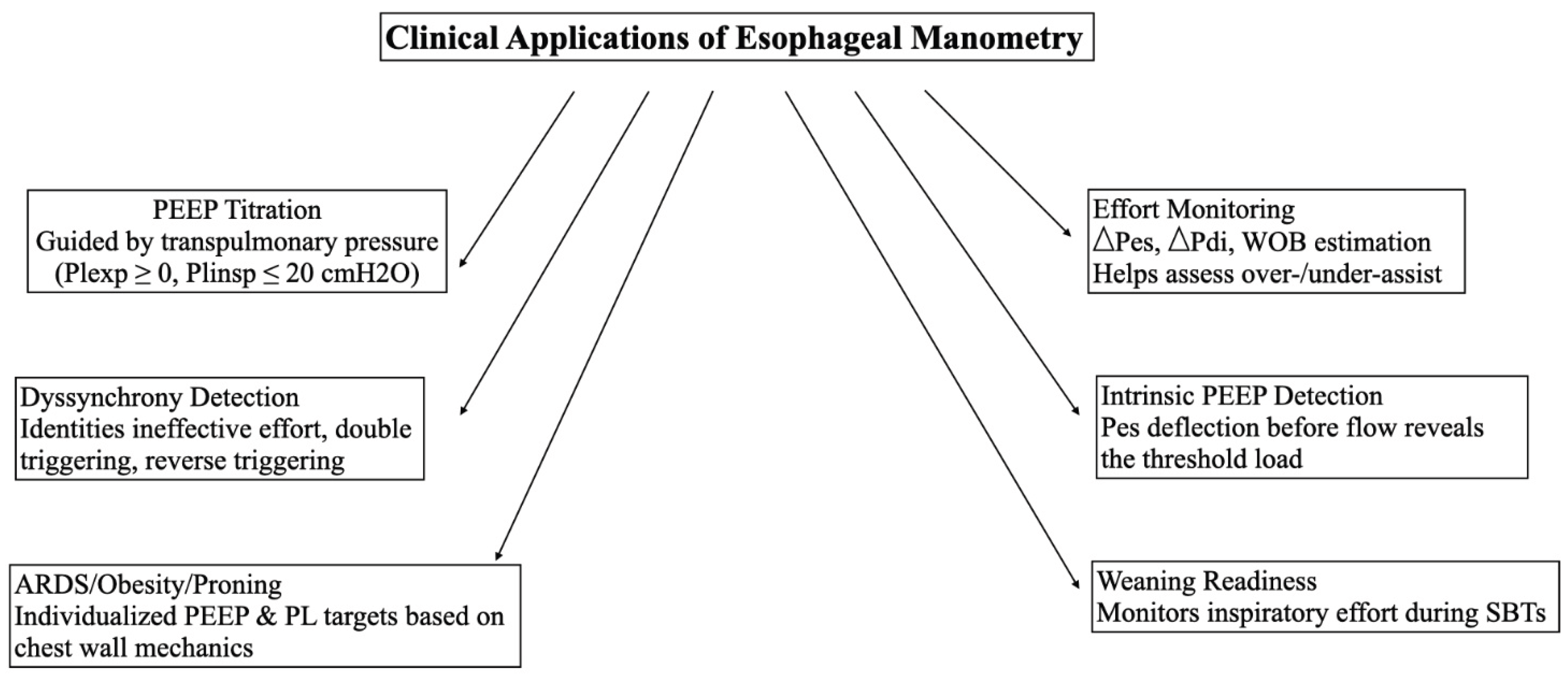
10. Integration with other monitoring for comprehensive assessment
11. Conclusions
Author Contributions
References
- Gattinoni, L.; Tonetti, T.; Quintel, M. Regional physiology of ARDS. Crit Care 2017, 21, 312. [Google Scholar] [CrossRef]
- Otto, C.M.; Markstaller, K.; Kajikawa, O.; et al. Spatial and temporal heterogeneity of ventilator-associated lung injury after surfactant depletion. J Appl Physiol Bethesda Md 1985 2008, 104, 1485–1494. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, S.F.; Shaikh, A.; Khan, D.A.; Surani, S.; Ratnani, I. Driving pressure in mechanical ventilation: A review. World J Crit Care Med. 2024, 13, 88385. [Google Scholar] [CrossRef]
- Shimatani, T.; Kyogoku, M.; Ito, Y.; Takeuchi, M.; KhemaniRG. Fundamental concepts and the latest evidence for esophageal pressure monitoring. J Intensive Care 2023, 11, 22. [Google Scholar] [CrossRef]
- Silva, P.L.; Rocco, P.RM. The basics of respiratory mechanics: ventilator-derived parameters. Ann Transl Med. 2018, 6, 376–376. [Google Scholar] [CrossRef]
- FernandesCR. Importance of pleural pressure for the evaluation of respiratory mechanics. Rev Bras Anestesiol. 2006, 56, 287–303. [Google Scholar] [CrossRef]
- Neupane, K.; Jamil, R.T. Physiology, Transpulmonary Pressure. In StatPearls; StatPearls Publishing, 2025; Available online: http://www.ncbi.nlm.nih.gov/books/NBK559004/ (accessed on 9 September 2025).
- Hickey, S.M.; Sankari, A.; GiwaAO. Mechanical Ventilation. In StatPearls; StatPearls Publishing, 2025; Available online: http://www.ncbi.nlm.nih.gov/books/NBK539742/ (accessed on 9 September 2025).
- Corp, A.; Thomas, C.; Adlam, M. The cardiovascular effects of positive pressure ventilation. BJA Educ. 2021, 21, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Mora Carpio, A.L.; Mora, JI. Assist-Control Ventilation. In StatPearls; StatPearls Publishing, 2025; Available online: http://www.ncbi.nlm.nih.gov/books/NBK441856/ (accessed on 9 September 2025).
- Mahmood, S.S.; Pinsky, MR. Heart-lung interactions during mechanical ventilation: the basics. Ann Transl Med. 2018, 6, 349. [Google Scholar] [CrossRef] [PubMed]
- Pinto, E.F.; Santos, R.S.; Antunes, M.A.; et al. Static and Dynamic Transpulmonary Driving Pressures Affect Lung and Diaphragm Injury during Pressure-controlled versus Pressure-support Ventilation in Experimental Mild Lung Injury in Rats. Anesthesiology 2020, 132, 307–320. [Google Scholar] [CrossRef]
- Modesto i Alapont, V.; Aguar Carrascosa, M.; Medina, V. Stress, strain and mechanical power: Is material science the answer to prevent ventilator induced lung injury? Med Intensiva Engl Ed. 2019, 43, 165–175. [Google Scholar] [CrossRef]
- Florio, G.; Carlesso, E.; Mojoli, F.; et al. Esophageal pressure as estimation of pleural pressure: a study in a model of eviscerated chest. BMC Anesthesiol. 2024, 24, 415. [Google Scholar] [CrossRef] [PubMed]
- Bastia, L.; Engelberts, D.; Osada, K.; et al. Role of Positive End-Expiratory Pressure and Regional Transpulmonary Pressure in Asymmetrical Lung Injury. Am J Respir Crit Care Med. 2021, 203, 969–976. [Google Scholar] [CrossRef] [PubMed]
- Katira, B.H.; Osada, K.; Engelberts, D.; et al. Positive End-Expiratory Pressure, Pleural Pressure, and Regional Compliance during Pronation. An Experimental Study. Am J Respir Crit Care Med. 2021, 203, 1266–1274. [Google Scholar] [CrossRef]
- Esophageal catheter placement | Hamilton Medical. Available online: http://www.hamilton-medical.com/en_US/Article-page~knowledge-base~7e2f2536-5057-4307-ae54-4eacfd81071b~Esophageal-catheter-placement~.html (accessed on 9 September 2025).
- Sigmon, D.F.; An, J. Nasogastric Tube. In StatPearls; StatPearls Publishing, 2025; Available online: http://www.ncbi.nlm.nih.gov/books/NBK556063/ (accessed on 9 September 2025).
- Mireles-Cabodevila, E.; Fischer, M.; Wiles, S.; Chatburn, RL. Esophageal Pressure Measurement: A Primer. Respir Care 2023, 68, 1281–1294. [Google Scholar] [CrossRef]
- https://www.coopersurgical.com/product/esophageal-balloon-catheter-set/. Available online: https://www.coopersurgical.com/product/esophageal-balloon-catheter-set/ (accessed on 9 September 2025).
- e-Catalog | Hamilton Medical. Available online: http://www.hamilton-medical.com/en_US/E-catalog.html?category=aed4b027-8713-4747-8d16-24b69f76fa3a&tab=7b4882e0-13b1-406a-a224-7ba7480a8b71&product=c7529755-4ea7-4fac-994f-07c54f1fee46 (accessed on 9 September 2025).
- Rudolph, M.W.; Koopman, A.A.; Blokpoel, R.G.T.; KneyberMCJ. Evaluation of Optimal Esophageal Catheter Balloon Inflation Volume in Mechanically Ventilated Children. Respir Care 2024, 22 69, 325–332. [Google Scholar] [CrossRef]
- Pham, T.; Telias, I.; Beitler, JR. Esophageal Manometry. Respir Care 2020, 65, 772–792. [Google Scholar] [CrossRef]
- Cheng, W.; Jiang, J.; Long, Y.; et al. Phenotypes of esophageal pressure response to the change of positive end-expiratory pressure in patients with moderate acute respiratory distress syndrome. J Thorac Dis. 2024, 16, 979–988. [Google Scholar] [CrossRef]
- The Application of Esophageal Pressure Measurement in Patients with Respiratory Failure | American Journal of Respiratory and Critical Care Medicine. Available online: https://www.atsjournals.org/doi/10.1164/rccm.201312-2193CI (accessed on 9 September 2025).
- Sun, X.M.; Chen, G.Q.; Huang, H.W.; et al. Use of esophageal balloon pressure-volume curve analysis to determine esophageal wall elastance and calibrate raw esophageal pressure: a bench experiment and clinical study. BMC Anesthesiol. 2018, 18, 21. [Google Scholar] [CrossRef] [PubMed]
- Enrico, B.; Cristian, F.; Stefano, B.; Luigi, P. Patient-ventilator asynchronies: types, outcomes and nursing detection skills. Acta Bio Medica Atenei Parm. 2018, 89 Suppl 7, 6–18. [Google Scholar] [CrossRef]
- Jackson, R.; Kim, A.; Moroz, N.; et al. Reverse triggering ? a novel or previously missed phenomenon? Ann Intensive Care 2024, 14, 78. [Google Scholar] [CrossRef]
- Ciorba, C.; Gonzalez-Bermejo, J.; Salva, M.A.Q.; et al. Flow and airway pressure analysis for detecting ineffective effort during mechanical ventilation in neuromuscular patients. Chron Respir Dis. 2018, 16, 1479972318790267. [Google Scholar] [CrossRef]
- Saavedra, S.N.; Barisich, P.V.S.; Maldonado, J.B.P.; Lumini, R.B.; Gómez-González, A.; Gallardo, A. Asynchronies during invasive mechanical ventilation: narrative review and update. Acute Crit Care 2022, 37, 491–501. [Google Scholar] [CrossRef]
- de Haro, C.; Santos-Pulpón, V.; Telías, I.; et al. Flow starvation during square-flow assisted ventilation detected by supervised deep learning techniques. Crit Care 2024, 28, 75. [Google Scholar] [CrossRef] [PubMed]
- Parthasarathy, S.; Jubran, A.; Tobin, MJ. Cycling of Inspiratory and Expiratory Muscle Groups with the Ventilator in Airflow Limitation. Am J Respir Crit Care Med. 1998, 158, 1471–1478. [Google Scholar] [CrossRef]
- Coppola, S.; Caccioppola, A.; Froio, S.; et al. Dynamic hyperinflation and intrinsic positive end-expiratory pressure in ARDS patients. Crit Care 2019, 23, 375. [Google Scholar] [CrossRef] [PubMed]
- Lessard, M.R.; Lofaso, F.; Brochard, L. Expiratory muscle activity increases intrinsic positive end-expiratory pressure independently of dynamic hyperinflation in mechanically ventilated patients. Am J Respir Crit Care Med. 1995, 151 2 Pt 1, 562–569. [Google Scholar] [CrossRef]
- van Oosten, J.P.; Akoumianaki, E.; Jonkman, AH. Monitoring respiratory muscles effort during mechanical ventilation. Curr Opin Crit Care 2025, 31, 12. [Google Scholar] [CrossRef]
- Somhorst, P.; Mousa, A.; Jonkman, AH. Setting positive end-expiratory pressure: the use of esophageal pressure measurements. Curr Opin Crit Care 2024, 30, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Jonkman, A.H.; Telias, I.; Spinelli, E.; Akoumianaki, E.; Piquilloud, L. The oesophageal balloon for respiratory monitoring in ventilated patients: updated clinical review and practical aspects. Eur Respir Rev. 2023, 32, 220186. [Google Scholar] [CrossRef]
- Alvey, N.J.; Hlaing, M.; Piccoli, J.; Kukreja, N.; Tran, TT. Positive end-expiratory pressure titration via esophageal balloon monitoring in a morbidly obese patient undergoing laparoscopic nephrectomy. Can J Anaesth. 2020, 67, 1086–1087. [Google Scholar] [CrossRef]
- Talmor, D.; Sarge, T.; Malhotra, A.; et al. Mechanical ventilation guided by esophageal pressure in acute lung injury. N Engl J Med. 2008, 359, 2095–2104. [Google Scholar] [CrossRef]
- Effect of Titrating Positive End-Expiratory Pressure (PEEP) With an Esophageal Pressure–Guided Strategy vs an Empirical High PEEP-Fio2 Strategy on Death and Days Free From Mechanical Ventilation Among Patients With Acute Respiratory Distress Syndrome: A Randomized Clinical Trial | Critical Care Medicine | JAMA | JAMA Network. Available online: https://jamanetwork.com/journals/jama/fullarticle/2725206 (accessed on 9 September 2025).
- Lung protective ventilation strategy for the acute respiratory distress syndrome - Petrucci, N - 2013 | Cochrane Library. Available online: https://www.cochranelibrary.com/cdsr/doi/10.1002/14651858.CD003844.pub4/full (accessed on 9 September 2025).
- De Santis Santiago, R.; Teggia Droghi, M.; Fumagalli, J.; et al. High Pleural Pressure Prevents Alveolar Overdistension and Hemodynamic Collapse in Acute Respiratory Distress Syndrome with Class III Obesity. A Clinical Trial. Am J Respir Crit Care Med. 2021, 203, 575–584. [Google Scholar] [CrossRef] [PubMed]
- Carteaux, G.; Parfait, M.; Combet, M.; Haudebourg, A.F.; Tuffet, S.; Mekontso, D.e.s.s.a.p A. Patient-Self Inflicted Lung Injury: A Practical Review. J Clin Med. 2021, 10, 2738. [Google Scholar] [CrossRef] [PubMed]
- Cornejo, R.; Telias, I.; Brochard, L. Measuring patient’s effort on the ventilator. Intensive Care Med. 2024, 50, 573–576. [Google Scholar] [CrossRef] [PubMed]
- Benditt, JO. Esophageal and gastric pressure measurements. Respir Care discussion 75-77. 2005, 50, 68–75. [Google Scholar]
- Gell, L.K.; Reynolds, K.J.; McEvoy, R.D.; Nguyen, D.P.; Catcheside, PG. A novel method to quantify breathing effort from respiratory mechanics and esophageal pressure. J Appl Physiol Bethesda Md 1985 2024, 136, 1418–1428. [Google Scholar] [CrossRef]
- Mojoli, F.; Pozzi, M.; Orlando, A.; et al. Timing of inspiratory muscle activity detected from airway pressure and flow during pressure support ventilation: the waveform method. Crit Care 2022, 26, 32. [Google Scholar] [CrossRef]
- Goligher, E.C.; Fan, E.; Herridge, M.S.; et al. Evolution of Diaphragm Thickness during Mechanical Ventilation. Impact of Inspiratory Effort. Am J Respir Crit Care Med. 2015, 192, 1080–1088. [Google Scholar] [CrossRef]
- Vries Hde Jonkman, A.; Shi, Z.H.; Man ASde, H.e.u.n.k.s L. Assessing breathing effort in mechanical ventilation: physiology and clinical implications. Ann Transl Med. 2018, 6, 387–387. [Google Scholar] [CrossRef]
- Mojoli, F.; Iotti, G.A.; Torriglia, F.; et al. In vivo calibration of esophageal pressure in the mechanically ventilated patient makes measurements reliable. Crit Care Lond Engl. 2016, 20, 98. [Google Scholar] [CrossRef]
- Mauri, T.; Yoshida, T.; Bellani, G.; et al. Esophageal and transpulmonary pressure in the clinical setting: meaning, usefulness and perspectives. Intensive Care Med. 2016, 42, 1360–1373. [Google Scholar] [CrossRef]
- Wu, W.; Zhang, X.; Lin, L.; et al. Transdiaphragmatic pressure and neural respiratory drive measured during inspiratory muscle training in stable patients with chronic obstructive pulmonary disease. Int J Chron Obstruct Pulmon Dis. 2017, 12, 773–781. [Google Scholar] [CrossRef]
- Calzia, E.; Lindner, K.H.; Witt, S.; et al. Pressure-time product and work of breathing during biphasic continuous positive airway pressure and assisted spontaneous breathing. Am J Respir Crit Care Med. 1994, 150, 904–910. [Google Scholar] [CrossRef]
- Bertoni, M.; Spadaro, S.; Goligher, EC. Monitoring Patient Respiratory Effort During Mechanical Ventilation: Lung and Diaphragm-Protective Ventilation. Crit Care 2020, 24, 106. [Google Scholar] [CrossRef]
- Banner, M.J.; Kirby, R.R.; Gabrielli, A.; Blanch, P.B.; Layon, AJ. Partially and totally unloading respiratory muscles based on real-time measurements of work of breathing. A clinical approach. Chest 1994, 106, 1835–1842. [Google Scholar] [CrossRef]
- Jubran, A.; Grant, B.J.B.; Laghi, F.; Parthasarathy, S.; Tobin, MJ. Weaning prediction: esophageal pressure monitoring complements readiness testing. Am J Respir Crit Care Med. 2005, 171, 1252–1259. [Google Scholar] [CrossRef]
- Albashir, S.M.; Robert, R.C.; Jairath, N.N.; Raub, C.B.; Alzumai, O.A.; Salem, SS. The rapid shallow breathing index (RSBI) as a predictor for extubation success in medical and surgical ICU patients: A retrospective cohort study. Heart Lung J Crit Care 2025, 70, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Vaporidi, K.; Soundoulounaki, S.; Papadakis, E.; Akoumianaki, E.; Kondili, E.; Georgopoulos, D. Esophageal and transdiaphragmatic pressure swings as indices of inspiratory effort. Respir Physiol Neurobiol. 2021, 284, 103561. [Google Scholar] [CrossRef] [PubMed]
- Sassoon, C.S.; Light, R.W.; Lodia, R.; Sieck, G.C.; Mahutte, CK. Pressure-time product during continuous positive airway pressure, pressure support ventilation, and T-piece during weaning from mechanical ventilation. Am Rev Respir Dis. 1991, 143, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Khemani, R.G.; Sekayan, T.; Hotz, J.; et al. Risk Factors for Pediatric Extubation Failure: The Importance of Respiratory Muscle Strength. Crit Care Med. 2017, 45, e798–e805. [Google Scholar] [CrossRef]
- Harikumar, G.; Egberongbe, Y.; Nadel, S.; et al. Tension-time index as a predictor of extubation outcome in ventilated children. Am J Respir Crit Care Med. 2009, 180, 982–988. [Google Scholar] [CrossRef] [PubMed]
- Bellemare, F.; Grassino, A. Effect of pressure and timing of contraction on human diaphragm fatigue. J Appl Physiol. 1982, 53, 1190–1195. [Google Scholar] [CrossRef]
- McGregor, M.; Becklake, MR. THE RELATIONSHIP OF OXYGEN COST OF BREATHING TO RESPIRATORY MECHANICAL WORK AND RESPIRATORY FORCE. J Clin Invest. 1961, 40, 971–980. [Google Scholar] [CrossRef]
- Bellani, G.; Pesenti, A. Assessing effort and work of breathing. Curr Opin Crit Care 2014, 20, 352. [Google Scholar] [CrossRef]
- Carteaux, G.; Mancebo, J.; Mercat, A.; et al. Bedside Adjustment of Proportional Assist Ventilation to Target a Predefined Range of Respiratory Effort. Crit Care Med. 2013, 41, 2125. [Google Scholar] [CrossRef]
- Khemani, R.G.; Hotz, J.; Morzov, R.; et al. Pediatric extubation readiness tests should not use pressure support. Intensive Care Med. 2016, 42, 1214–1222. [Google Scholar] [CrossRef]
- Spinelli, E.; Scaramuzzo, G.; Slobod, D.; Mauri, T. Understanding cardiopulmonary interactions through esophageal pressure monitoring. Front Physiol. 2023, 14, 1221829. [Google Scholar] [CrossRef]
- Jardin, F.; Genevray, B.; Brun-Ney, D.; Bourdarias, JP. Influence of lung and chest wall compliances on transmission of airway pressure to the pleural space in critically ill patients. Chest 1985, 88, 653–658. [Google Scholar] [CrossRef] [PubMed]
- Slobod, D.; Assanangkornchai, N.; Alhazza, M.; Mettasittigorn, P.; Magder, S. Right Ventricular Loading by Lung Inflation during Controlled Mechanical Ventilation. Am J Respir Crit Care Med. 2022, 205, 1311–1319. [Google Scholar] [CrossRef] [PubMed]
- Repessé, X.; Vieillard-Baron, A.; Geri, G. Value of measuring esophageal pressure to evaluate heart-lung interactions—applications for invasive hemodynamic monitoring. Ann Transl Med. 2018, 6, 351. [Google Scholar] [CrossRef]
- Rankin, J.S.; Olsen, C.O.; Arentzen, C.E.; et al. The effects of airway pressure on cardiac function in intact dogs and man. Circulation 1982, 66, 108–120. [Google Scholar] [CrossRef] [PubMed]
- Khirfan, G.; Melillo, C.A.; Al Abdi, S.; et al. Impact of Esophageal Pressure Measurement on Pulmonary Hypertension Diagnosis in Patients With Obesity. Chest 2022, 162, 684–692. [Google Scholar] [CrossRef]
- Thind, G.S.; Mireles-Cabodevila, E.; Chatburn, R.L.; Duggal, A. Evaluation of Esophageal Pressures in Mechanically Ventilated Obese Patients. Respir Care 2022, 67, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Reddy, VG. Auto-PEEP: how to detect and how to prevent--a review. Middle East J Anaesthesiol. 2005, 18, 293–312. [Google Scholar]
- Chen, L.; Grieco, D.L.; Beloncle, F.; et al. Partition of respiratory mechanics in patients with acute respiratory distress syndrome and association with outcome: a multicentre clinical study. Intensive Care Med. 2022, 48, 888–898. [Google Scholar] [CrossRef]
- Tonelli, R.; Fantini, R.; Tabbì, L.; et al. Early Inspiratory Effort Assessment by Esophageal Manometry Predicts Noninvasive Ventilation Outcome in De Novo Respiratory Failure. A Pilot Study. Am J Respir Crit Care Med. 2020, 202, 558–567. [Google Scholar] [CrossRef]
- Ahmed, S.M.; Athar, M. Mechanical ventilation in patients with chronic obstructive pulmonary disease and bronchial asthma. Indian J Anaesth. 2015, 59, 589. [Google Scholar] [CrossRef]
- Mein, S.A.; Ferrera, MC. Management of Asthma and COPD Exacerbations in Adults in the ICU. CHEST Crit Care 2025, 3. [Google Scholar] [CrossRef] [PubMed]
- Rehder, K.J.; Alibrahim, OS. Mechanical Ventilation during ECMO: Best Practices. Respir Care 2023, 68, 838–845. [Google Scholar] [CrossRef]
- Bishop, M.A.; Moore, A. Extracorporeal Membrane Oxygenation Weaning. In StatPearls; StatPearls Publishing, 2025; Available online: http://www.ncbi.nlm.nih.gov/books/NBK570564/ (accessed on 10 September 2025).
- Beck, J.; Reilly, M.; Grasselli, G.; et al. Patient-ventilator interaction during neurally adjusted ventilatory assist in low birth weight infants. Pediatr Res. 2009, 65, 663–668. [Google Scholar] [CrossRef]
- Liu, L.; Liu, S.; Xie, J.; et al. Assessment of patient-ventilator breath contribution during neurally adjusted ventilatory assist in patients with acute respiratory failure. Crit Care 2015, 19, 43. [Google Scholar] [CrossRef]
- Vassilakopoulos, T. Ultrasonographic Monitoring of the Diaphragm during Mechanical Ventilation: The Vital Pump Is Vivid, Plastic, and Vulnerable. Am J Respir Crit Care Med. 2015, 192, 1030–1032. [Google Scholar] [CrossRef]
- Laguado-Nieto, M.A.; Roberto-Avilán, S.L.; Naranjo-Junoy, F.; et al. Diaphragmatic Dynamics and Thickness Parameters Assessed by Ultrasonography Predict Extubation Success in Critically Ill Patients. Clin Med Insights Circ Respir Pulm Med. 2023, 17, 11795484231165940. [Google Scholar] [CrossRef]
- Umbrello, M.; Formenti, P. Ultrasonographic Assessment of Diaphragm Function in Critically Ill Subjects. Respir Care 2016, 61, 542–555. [Google Scholar] [CrossRef]
- Tomicic, V.; Cornejo, R. Lung monitoring with electrical impedance tomography: technical considerations and clinical applications. J Thorac Dis. 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- Kobylianskii, J.; Murray, A.; Brace, D.; Goligher, E.; Fan, E. Electrical impedance tomography in adult patients undergoing mechanical ventilation: A systematic review. J Crit Care 2016, 35, 33–50. [Google Scholar] [CrossRef]
- Maciejewski, D.; Putowski, Z.; Czok, M.; Krzych, ŁJ. Electrical impedance tomography as a tool for monitoring mechanical ventilation. An introduction to the technique. Adv Med Sci. 2021, 66, 388–395. [Google Scholar] [CrossRef] [PubMed]
- Reinartz, S.D.; Imhoff, M.; Tolba, R.; et al. EIT monitors valid and robust regional ventilation distribution in pathologic ventilation states in porcine study using differential DualEnergy-CT (ΔDECT). Sci Rep. 2019, 9, 9796. [Google Scholar] [CrossRef] [PubMed]
- PEEP titration guided by ventilation homogeneity: a feasibility study using electrical impedance tomography | Critical Care | Full Text. Available online: https://ccforum.biomedcentral.com/articles/10.1186/cc8860 (accessed on 10 September 2025).
- Costa, E.L.V.; Borges, J.B.; Melo, A.; et al. Bedside estimation of recruitable alveolar collapse and hyperdistension by electrical impedance tomography. Intensive Care Med. 2009, 35, 1132–1137. [Google Scholar] [CrossRef]
- Beloncle, F.; Piquilloud, L.; Olivier, P.Y.; et al. Accuracy of P0.1 measurements performed by ICU ventilators: a bench study. Ann Intensive Care 2019, 9, 104. [Google Scholar] [CrossRef]
- Kuhlen, R.; Mohnhaupt, R.; Slama, K.; et al. Validation and clinical application of a continuous P0.1 measurement using standard respiratory equipment. Technol Health Care Off J Eur Soc Eng Med. 1996, 4, 415–424. [Google Scholar] [CrossRef]
- Le Marec, J.; Hajage, D.; Decavèle, M.; et al. High Airway Occlusion Pressure Is Associated with Dyspnea and Increased Mortality in Critically Ill Mechanically Ventilated Patients. Am J Respir Crit Care Med. 2024, 210, 201–210. [Google Scholar] [CrossRef]
- Pérez, J.; Telias, I. Airway Occlusion Pressure and Dyspnea during Mechanical Ventilation: Giving Words to the Pleas of the Respiratory Centers. Am J Respir Crit Care Med. 2024, 210, 139–141. [Google Scholar] [CrossRef]
- Tonelli, R.; Protti, A.; Spinelli, E.; et al. Assessing inspiratory drive and effort in critically ill patients at the bedside. Crit Care 2025, 29, 339. [Google Scholar] [CrossRef] [PubMed]
- Whitelaw, W.A.; Derenne, J.P.; Milic-Emili, J. Occlusion pressure as a measure of respiratory center output in conscious man. Respir Physiol. 1975, 23, 181–199. [Google Scholar] [CrossRef] [PubMed]
- Pata, R.; Kristeva, J.; Stocks, J.; Mian, I.S.; Guadarrama, D.; Datar, P. Occlusion Pressure at 100msec for Predicting Outcomes in Patients Ventilated Via Pressure Regulated Volume Control Mode in Moderate Adult Respiratory Distress Syndrome (Oculus Study). In B38: OPTIMIZING MECHANICAL VENTILATION AND VENTILATOR LIBERATION; American Thoracic Society, 2024; pp. A3428–A3428. [Google Scholar] [CrossRef]
- Terzi, N.; Bayat, S.; Noury, N.; et al. Comparison of pleural and esophageal pressure in supine and prone positions in a porcine model of acute respiratory distress syndrome. J Appl Physiol. 2020, 128, 1617–1625. [Google Scholar] [CrossRef]
- Dostal, P.; Dostalova, V. Practical Aspects of Esophageal Pressure Monitoring in Patients with Acute Respiratory Distress Syndrome. J Pers Med. 2023, 13, 136. [Google Scholar] [CrossRef] [PubMed]
- Beitler, J.R.; Sarge, T.; Banner-Goodspeed, V.M.; et al. Effect of Titrating Positive End-Expiratory Pressure (PEEP) With an Esophageal Pressure–Guided Strategy vs an Empirical High PEEP-Fio2 Strategy on Death and Days Free From Mechanical Ventilation Among Patients With Acute Respiratory Distress Syndrome: A Randomized Clinical Trial. JAMA 2019, 321, 846–857. [Google Scholar] [CrossRef]
 |
 |
 |
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




