Submitted:
12 December 2025
Posted:
15 December 2025
You are already at the latest version
Abstract
Keywords:
Introduction
2.1. Natural Bone Structure and Composition
| Component | Site or Specific Molecule | Volume (%) |
|---|---|---|
| Water | Free/pore; bound at collagen-mineral interfaces; structural | 15–25 vol % |
| Organic matrix | Type I collagen (~90 wt %), non-collagenous proteins (~10 wt %) | ~32–40 vol % |
| Mineral (apatite) | Hydroxyapatite with carbonate substitutions | 35–45 vol % |
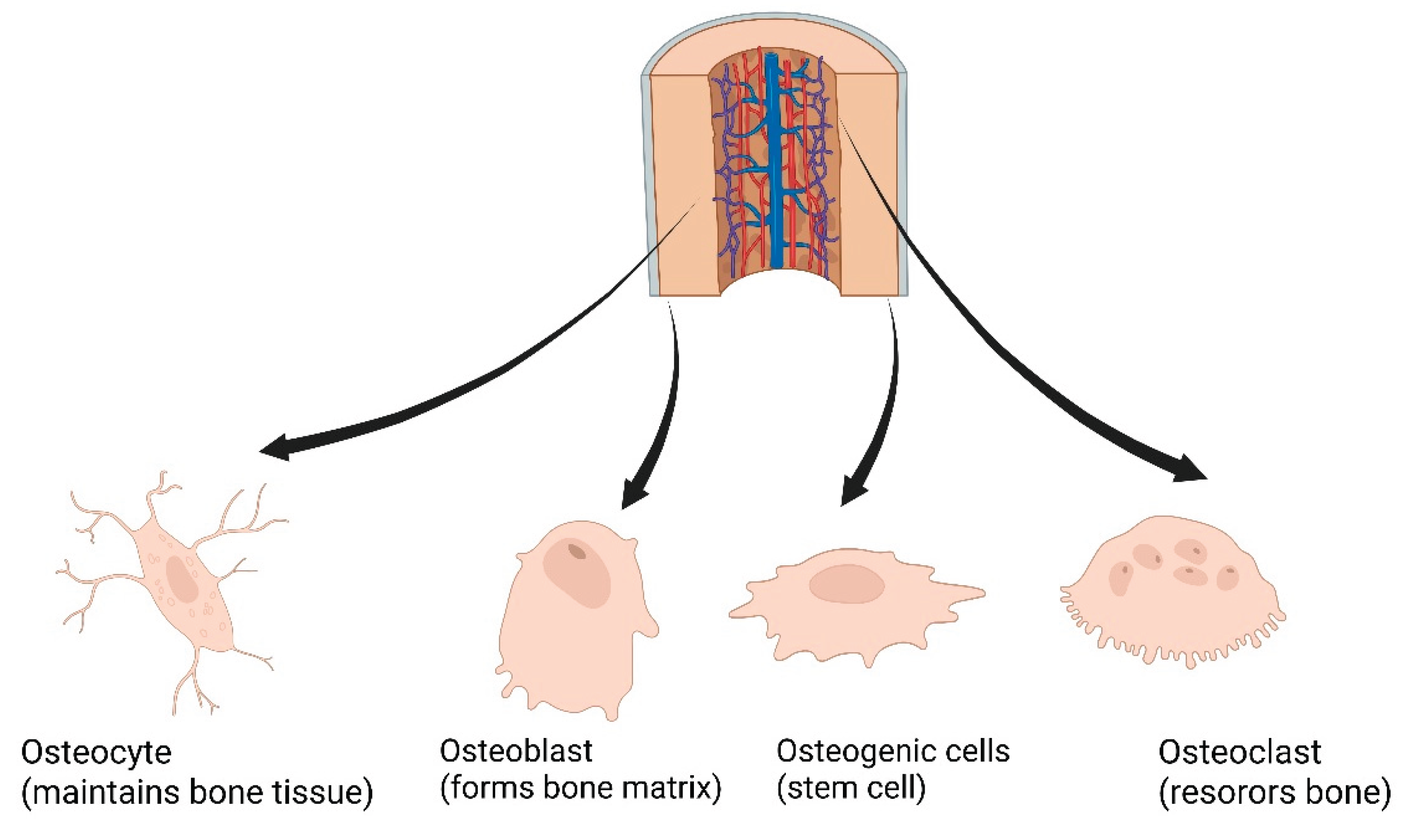
2.1.1. Bone Cells and Their Functions
2.1.2. Bone Healing Process
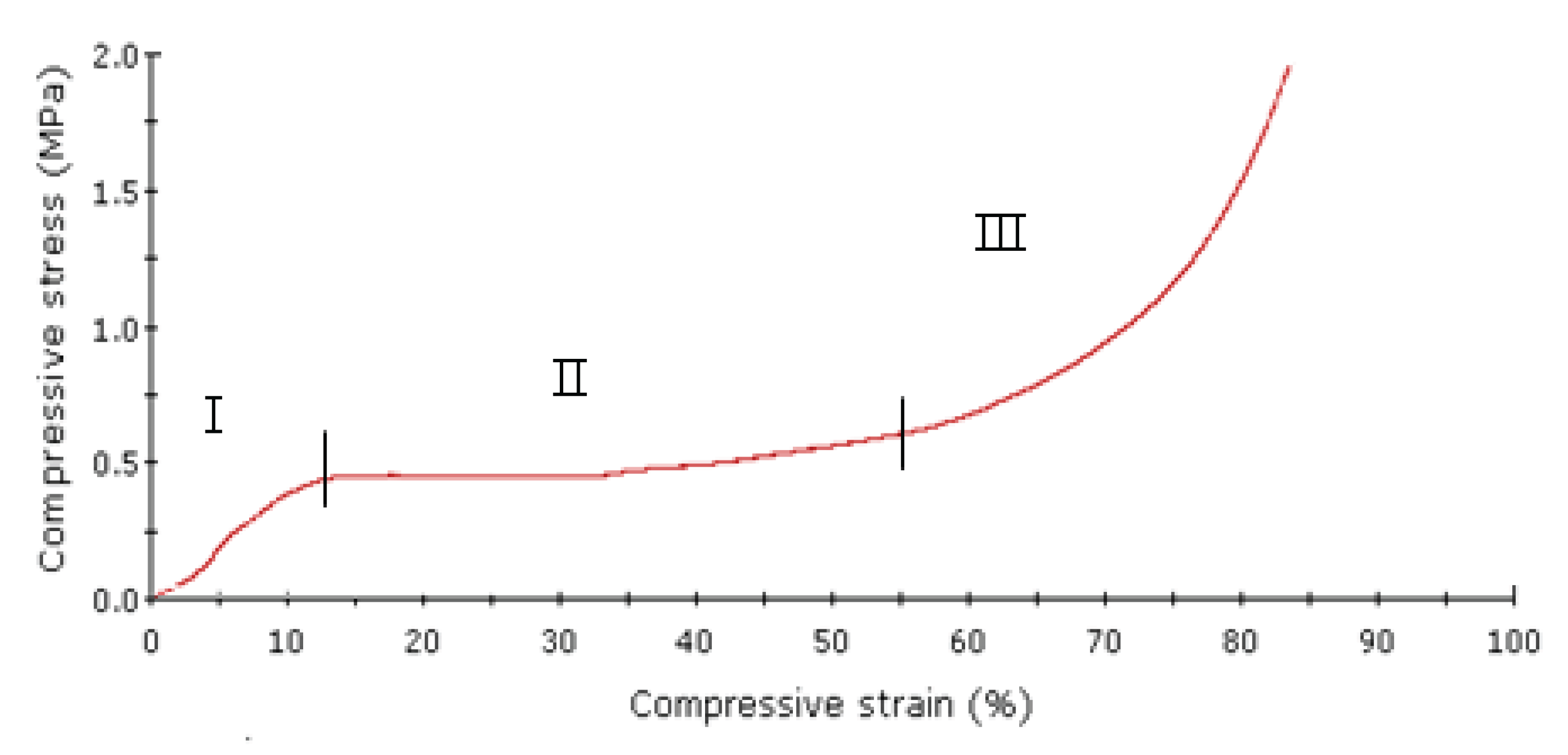
2.1.3. Mechanical Properties of Natural Bone
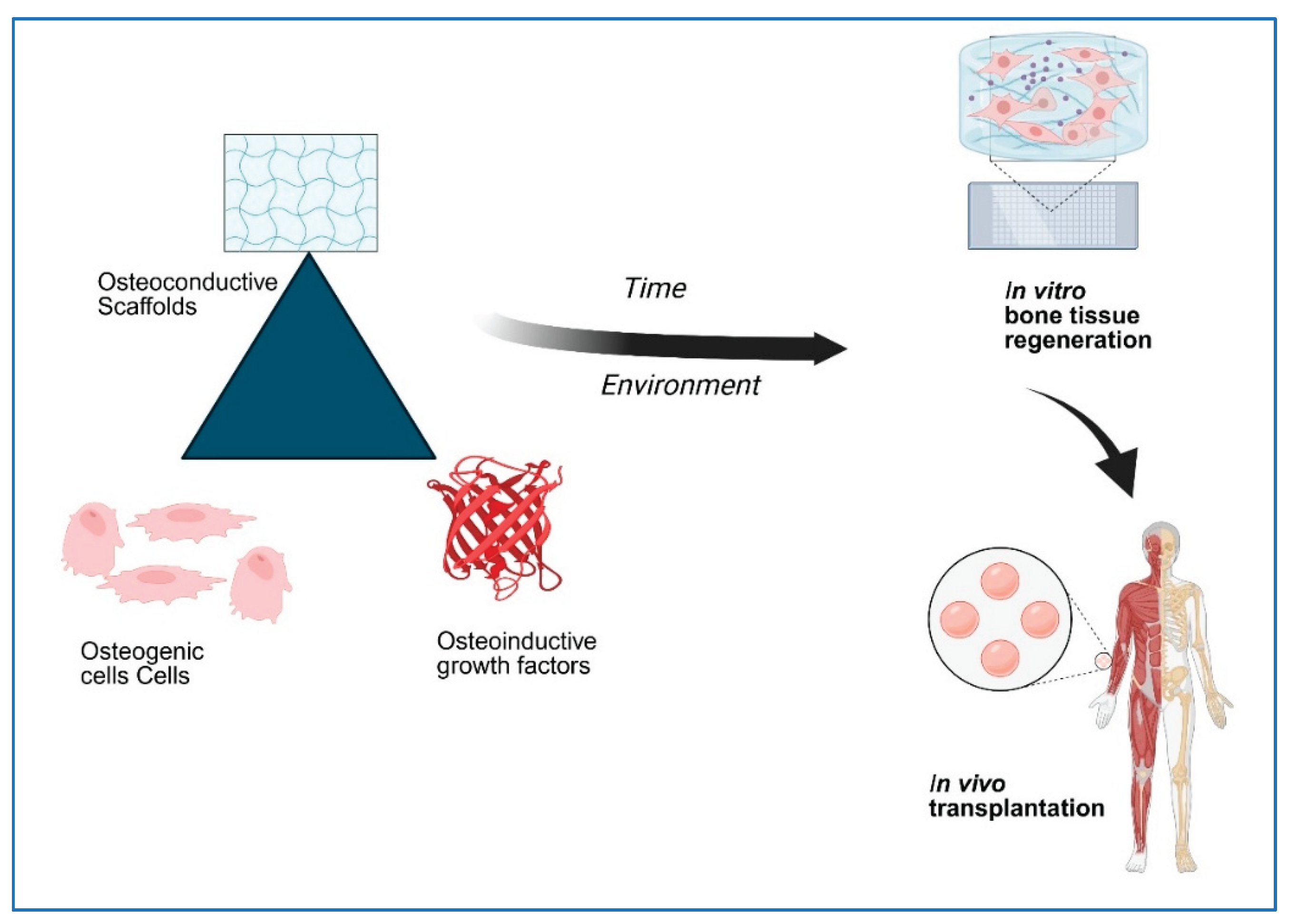
2.2. Properties of Bone TE Scaffold
2.2.1. Chemical and Mechanical Properties of Bone TE Scaffold
2.2.2. Physical Properties of Bone TE Scaffold
2.2.3. Surface Properties of Bone TE Scaffold
2.2.4. Bioactivity Properties
2.2.5. Degradation Properties
2.3. Bone-Substituted Biomaterials
2.3.1. Metals
| Category | Examples | Key Properties | Biomedical Applications | References |
| Permanent Metals | Stainless steel, Titanium alloys (Ti-6Al-4V), Cobalt-chromium alloys | High mechanical strength, corrosion resistance, long-term stability, non-degradable | Joint replacements, fracture fixation devices, spinal implants | [56] |
| Biodegradable Metals | Magnesium alloys, Zinc alloys, Iron-based alloys | Biodegradable in physiological environment, good biocompatibility, promotes bone growth | Temporary fracture fixation, bone scaffolds, pediatric implants | [57] |
2.3.2. Bioceramics
| Form of HA | Key Properties | Biomedical Performance | References |
| Micro-hydroxyapatite (Micro-HA) | Lower surface area, larger particle size (1–100 µm), lower reactivity, limited mechanical strength | Moderate osteoconductivity, slower resorption, less effective in promoting cell adhesion and proliferation | [60] |
| Nano-hydroxyapatite (Nano-HA) | High surface area, nanoscale particle size (<100 nm), enhanced reactivity, better mechanical integration | Superior osteoconductivity, faster resorption, improved cell adhesion, proliferation, and differentiation | [61] |
2.3.3. Polymers
2.5. Fabrication Techniques of Tissue Engineering Scaffolds
| Technique | Description | Advantages | Limitations |
| Gas Foaming | Uses blowing agents (CO₂, N₂) under supercritical conditions to create porous polymer structures. | Solvent-free process; low temperature reduces polymer degradation. | Produces small pore sizes; requires ultrasound for better interconnectivity. |
| Sintering | Compacts ceramic powders using heat or pressure without melting. | Allows control over porosity; suitable for ceramics. | High fragility; poor pore interconnectivity. |
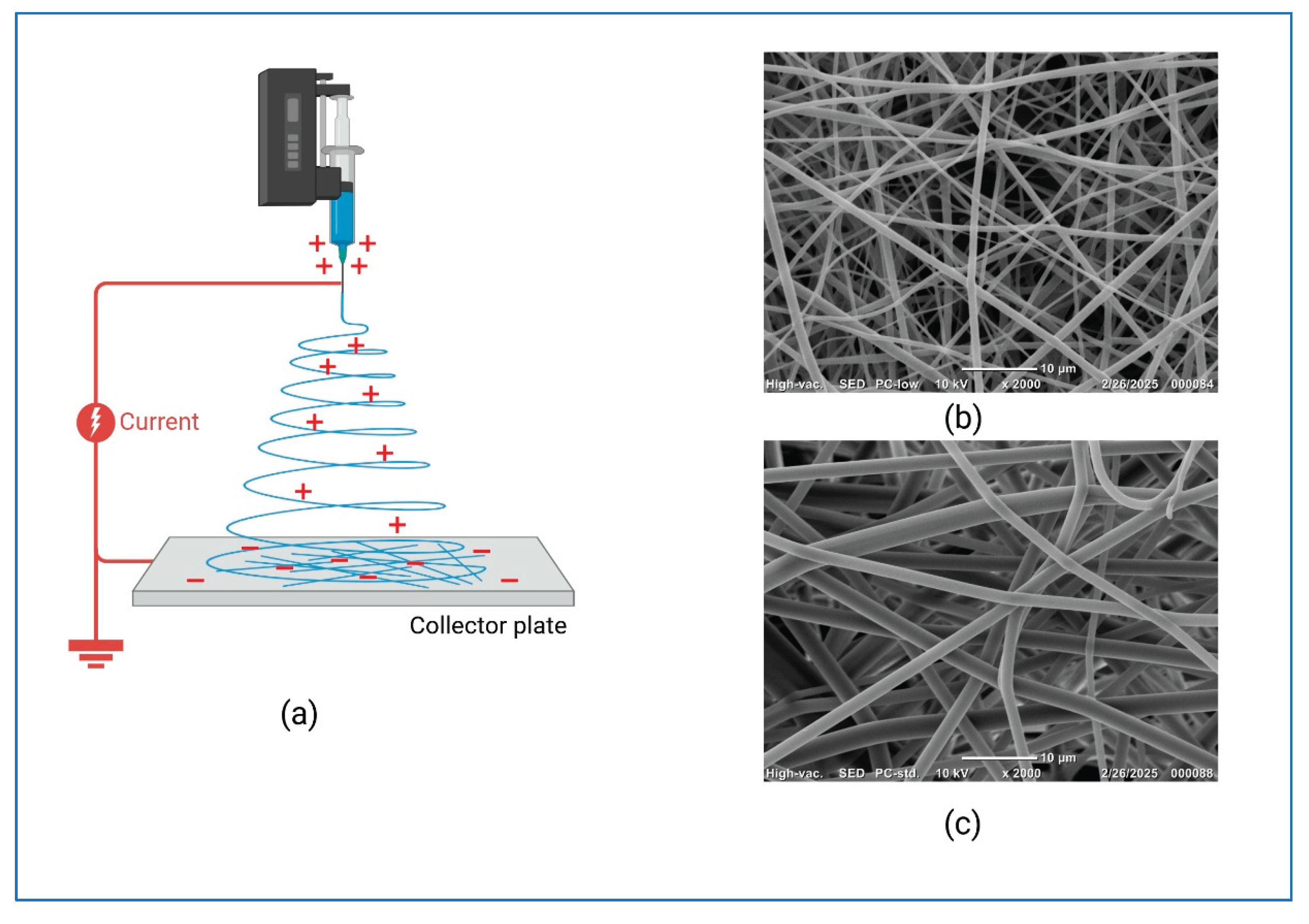
| Electrospinning | Uses electric field to produce nanofibers from polymer solutions. | Mimics ECM structure; versatile for different polymers. | Limited control over 3D architecture; requires post-processing. |
| Casting & Particle Leaching | Introduces porogens (salt, sugar) into polymer solution; leached to form pores. | Produces highly porous scaffolds; simple method. | Time-consuming; residual particles may remain. |
| Polymer Phase Separation (TIPS) | Thermally induced phase separation to control pore morphology. | Effective for micro/nano-scale pores; adaptable for drug delivery. | Requires precise control of parameters; solvent handling needed. |
| Rapid Prototyping (3D Printing) | CAD-based layer-by-layer fabrication for complex architectures. | High precision; customizable for patient-specific defects. | Expensive equipment; slower than conventional methods. |
| Freeze-Drying | Creates porous scaffolds by freezing polymer solution and sublimating solvent. | Highly porous structure; good for drug/growth factor incorporation. | Requires careful parameter control; limited scalability. |
3. In Vitro Biological Evaluation of Bone Tissue Engineering Scaffolds
4. In Vivo Biological Evaluation of Bone Tissue Engineering Scaffolds
5. Clinical Translation Challenges
6. Limitations and Future Directions
References
- Zhu, T.; Zhou, H.; Chen, X.; Zhu, Y. Recent advances of responsive scaffolds in bone tissue engineering. Front. Bioeng. Biotechnol. 2023, 11, 1296881. [Google Scholar] [CrossRef] [PubMed]
- Puppi, D.; Chiellini, F.; Piras, A.M.; Chiellini, E. Polymeric materials for bone and cartilage repair. Prog. Polym. Sci. 2010, 35, 403–440. [Google Scholar] [CrossRef]
- Rose, F.R.A.J.; Oreffo, R.O.C. Bone tissue engineering: Hope vs hype. Biochem. Biophys. Res. Commun. 2002, 292, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Gharibshahian, M.; Salehi, M.; Beheshtizadeh, N.; Nourbakhsh, M.S.; Alizadeh, M. Recent advances on 3D-printed PCL-based composite scaffolds for bone tissue engineering. Front. Bioeng. Biotechnol. 2023, 11, 1168504. [Google Scholar] [CrossRef]
- Khobragade, S.S.; Deshmukh, M.; Vyas, U.; Ingle, R.G. Innovative approaches in bone tissue engineering: Strategies for cancer treatment and recovery. Int. J. Mol. Sci. 2025, 26, 3937. [Google Scholar] [CrossRef]
- Mikos, A.G.; et al. Principles of tissue engineering. J. Biomed. Mater. Res. 1993, 27, 123–138. [Google Scholar]
- Hansmann, J.; et al. Bioreactor systems for bone tissue engineering. Adv. Drug Deliv. Rev. 2013, 65, 62–79. [Google Scholar]
- Chen, Q.; et al. Bone tissue engineering: Advances and challenges. Tissue Eng. 2006, 12, 1137–1151. [Google Scholar]
- Schieker, M.; et al. Tissue engineering of bone: Biomaterials, cell sources and growth factors. J. Cell. Mol. Med. 2006, 10, 6–17. [Google Scholar]
- Rastogi, S.; Verma, R.; Gouru, S.A.; Venkatesan, K.; Pandian, P.M.; Khan, M.I.; Deka, T.; Kumar, P. Emerging technologies in bone tissue engineering: A review. J. Bionic Eng. 2025, 22, 2261–2285. [Google Scholar] [CrossRef]
- Rosa, N.; Moura, M.F.S.F.; Olhero, S.; et al. Bone: An Outstanding Composite Material. Appl. Sci. 2022, 12, 3381. [Google Scholar] [CrossRef]
- Hamandi, F.; Goswami, T. Hierarchical Structure and Properties of the Bone at Nano Level. Bioengineering 2022, 9, 677. [Google Scholar] [CrossRef] [PubMed]
- Wittig, N.K.; Birkedal, H. Bone Hierarchical Structure: Spatial Variation across Length Scales. IUCrJ 2022, 9, 3. [Google Scholar] [CrossRef] [PubMed]
- Alcântara, A.C.S.; Felix, L.C.; Galvão, D.S.; et al. Devising Bone Molecular Models at the Nanoscale. Materials 2022, 15, 2274. [Google Scholar] [CrossRef] [PubMed]
- Schwarcz, A.V.; et al. The Ultrastructure of Bone and Its Relevance to Mechanical Properties. Front. Phys. 2017, 5, 45. [Google Scholar] [CrossRef]
- Luo, Y.; Amromanoh, O. Bone Organic-Inorganic Phase Ratio Is a Fundamental Determinant of Bone Material Quality. Appl. Bionics Biomech. 2021, 2021, 1–10. [Google Scholar] [CrossRef]
- Allen, M.R.; Wallace, J. Mechanical Properties of Bone. In Skeletal Tissue Mechanics; Springer: Cham, Switzerland, 2025; pp. 151–192. [Google Scholar]
- Wu, B.; Wu, Y.; Liu, M.; et al. Mechanical Behavior of Human Cancellous Bone in Alveolar Bone under Uniaxial Compression and Creep Tests. Materials 2022, 15, 5912. [Google Scholar] [CrossRef]
- Donsante, S.; Palmisano, B.; Serafini, M.; et al. From Stem Cells to Bone-Forming Cells. Int. J. Mol. Sci. 2021, 22, 3989. [Google Scholar] [CrossRef]
- Kitaura, H.; Marahleh, A.; Ohori, F.; et al. Osteocyte-Related Cytokines Regulate Osteoclast Formation and Bone Resorption. Int. J. Mol. Sci. 2020, 21, 5169. [Google Scholar] [CrossRef]
- Maruyama, M.; et al. Modulation of the Inflammatory Response and Bone Healing. Front. Endocrinol. 2020, 11, 386. [Google Scholar] [CrossRef]
- ElHawary, H.; et al. Bone Healing and Inflammation: Principles of Fracture and Repair. Semin Plast Surg 2021, 35, 141–147. [Google Scholar] [CrossRef]
- Sanders, E.J.; Parker, T.; Smith, A. Scaffold Design for Bone Tissue Engineering: Biological Considerations. J. Biomed. Mater. Res. 2000, 52, 231–240. [Google Scholar] [CrossRef]
- Wan, C.; Li, J.; Zhou, Y. Influence of Scaffold Architecture on Cell Migration and ECM Deposition. Biomaterials 1997, 18, 953–960. [Google Scholar]
- Flemming, R.; Green, D.; Brown, K. Mechanical Loading and Scaffold Performance in Bone Regeneration. Tissue Eng. 1999, 5, 291–302. [Google Scholar]
- Green, D.; Patel, M.; Jones, R. Spatial Organization of Bone Cells in Engineered Scaffolds. J. Orthop. Res. 1994, 12, 465–472. [Google Scholar]
- Li, J.; Yang, F.; Xu, H. Scaffold Architecture and Osteogenic Cell Behavior. Biomaterials 2002, 23, 123–135. [Google Scholar]
- Longer, R.; Vacant, A. Advances in Scaffold Fabrication for Regenerative Medicine. Adv. Drug Deliv. Rev. 1999, 39, 211–220. [Google Scholar]
- Hutmacher, D.W. Scaffolds in Tissue Engineering Bone and Cartilage. Biomaterials 2001, 22, 2529–2543. [Google Scholar]
- Khan, Y.; Yaszemski, M.J.; Mikos, A.G. Tissue Engineering Scaffolds for Bone Repair: Chemical Properties. Tissue Eng. 2008, 14, 163–174. [Google Scholar]
- Liu, X.; Ma, P.X. Polymeric Scaffolds for Bone Tissue Engineering. Biomaterials 2006, 27, 2414–2431. [Google Scholar] [CrossRef]
- Hassenkam, T.; Jørgensen, L.; Pedersen, M. AFM Imaging of Bone Surface Nanostructure. Bone 2004, 35, 4–10. [Google Scholar] [CrossRef]
- Williams, D.F. Surface Modification Strategies for Biomaterials. Biomaterials 1999, 20, 1321–1335. [Google Scholar]
- Park, J.B. Biomaterials Science: Introduction to Materials in Medicine; Academic Press, 1979. [Google Scholar]
- Ong, J.L.; Lucas, L.C. Interaction Zone Between Tissue and Implant Surfaces. J. Biomed. Mater. Res. 1998, 39, 152–160. [Google Scholar]
- Chen, Q.; Roether, J.A.; Boccaccini, A.R. Surface Engineering for Bone Tissue Scaffolds. Biomaterials 2006, 27, 2414–2431. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Xu, H.; Li, J. Collagen Coating Enhances Scaffold Hydrophilicity and Cell Adhesion. Biomaterials 2002, 23, 123–135. [Google Scholar]
- Chang, H. C.; Sun, T.; Sultana, N.; Lim, M. M.; Khan, T. H.; Ismail, A. F. Conductive PEDOT:PSS coated polylactide (PLA) and poly(3-hydroxybutyrate-co-3-hydroxyvalerate) (PHBV) electrospun membranes: Fabrication and characterization. Materials Science and Engineering: C 2016, 61, 396–410. [Google Scholar] [CrossRef]
- Shendage, S.; Kachare, K.; Gaikwad, K.; Kashte, S.; Mai, Fu.; Ghule, A. V. Cellulose-based bioactive material and turmeric-impregnated flexible and biocompatible scaffold for bone tissue engineering applications. J. Mater. Chem. B 2025, 15, 4570–4581. Available online: https://pubs.rsc.org/en/content/articlelanding/2025/tb/d4tb02028f. [CrossRef]
- Shin, K.; Acri, T.; Geary, S.; Salem, A. K. Biomimetic Mineralization of Biomaterials Using Simulated Body Fluids for Bone Tissue Engineering and Regenerative Medicine. Tissue Engineering. Part A 2017, 23(19-20), 1169. [Google Scholar] [CrossRef]
- Oktay, B.; Akkad, H.; Özerol, E.A. Recent advances in conducting tissue engineering based on conducting polymers. J. Biomater. Sci. Polym. Ed. 2025, 36, 2527910. [Google Scholar] [CrossRef]
- Aboagye, J.; Edwards, M.; Ge, J.; Hong, Y.; Yang, H. Evaluating and improving biocompatibility of conductive polymers for cardiac tissue engineering. J. Mater. Chem. B 2026, 14, D5TB01523E. [Google Scholar] [CrossRef]
- Castana, R.; et al. Title of study. Journal Name 2004, Volume, Pages. (Insert full MDPI citation).
- Bendrea, A.-D.; et al. Title of study. Journal Name 2011, Volume, Pages. (Insert full MDPI citation).
- Campione, P.; Rizzo, M.G.; Bauso, L.V.; Ielo, I.; Messina, G.M.L.; Calabrese, G. Osteoblastic differentiation of human adipose-derived mesenchymal stem cells on P3HT thin polymer film. J. Funct. Biomater. 2024, 16, 10. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Liao, R.; Li, X.; Zhang, C.; Huo, S.; Qin, L.; Xiong, Y.; He, T.; Xiao, G.; Zhang, T. Mesenchymal stem cells in treating human diseases: molecular mechanisms and clinical studies. Signal Transduct. Target. Ther. 2025, 10, 2313. [Google Scholar] [CrossRef] [PubMed]
- Hartung, N.; Abrams, S.A.; Huisinga, W.; Weisser, K. Consolidated calcium kinetic rates in a Caucasian population as a function of age and sex. bioRxiv 2024. [Google Scholar] [CrossRef]
- Choi, I.A.; Umemoto, A.; Mizuno, M.; Park-Min, K.H. Bone metabolism: an underappreciated player in metabolic health and disease. npj Metab. Health Dis. 2024, 1, 10. [Google Scholar]
- Sultana, N.; Khan, T. H. In Vitro Degradation of PHBV Scaffolds and nHA/PHBV Composite Scaffolds Containing Hydroxyapatite Nanoparticles for Bone Tissue Engineering. Journal of Nanomaterials 2012, 2012(1), 190950. [Google Scholar] [CrossRef]
- Feng, P.; Jia, J.; Liu, M.; Peng, S.; Zhao, Z.; Shuai, C. Degradation mechanisms and acceleration strategies of poly (lactic acid) scaffold for bone regeneration. Materials & Design 2021, 210, 110066. [Google Scholar] [CrossRef]
- Mohseni, M; Cometta, S; Klein, L; Wille, ML; Vaquette, C; Hutmacher, DW; Medeiros Savi, F. In vitro and in vivo degradation studies of a dual medical-grade scaffold design for guided soft tissue regeneration. Biomater Sci. 2025, 13(8), 2115–2133. [Google Scholar] [CrossRef] [PubMed]
- Oldani, C; Dominguez, A. Titanium as a Biomaterial for Implants [Internet]. Recent Advances in Arthroplasty. InTech. Available from. [CrossRef]
- Thomas, K.K.; Zafar, M.N.; Pitt, W.G.; Husseini, G.A. Biodegradable Magnesium Alloys for Biomedical Implants: Properties, Challenges, and Surface Modifications . Appl. Sci. 2024, 14(1). [Google Scholar] [CrossRef]
- Khan, A.R.; Shabbir, M.T.; Grewal, N.S.; Zhang, H.J.; Jun, Z. Recent Progress in the Application of Biodegradable Metal Implants . Biomed. Lett. 2024, 10(1), 1–13. [Google Scholar] [CrossRef]
- Abbasi, I. Biodegradable Metallic Materials: Revolutionizing Medical Implants and Devices. AZoM Feature Article, June 2024.
- Niinomi, M. Recent research and development in titanium alloys for biomedical applications and healthcare goods. Science and Technology of Advanced Materials 2003, 4(5), 445–454. [Google Scholar] [CrossRef]
- Witte, F. The history of biodegradable magnesium implants: a review. Acta Biomater 2010, 6(5), 1680–92. [Google Scholar] [CrossRef] [PubMed]
- Sultana, N.; Wang, M. Fabrication of HA/PHBV composite scaffolds through the emulsion freezing/freeze-drying process and characterisation of the scaffolds. J Mater Sci: Mater Med 2008, 19, 2555–2561. [Google Scholar] [CrossRef]
- Hassan, M I; Sultana, N.; Hamdan, S. Bioactivity Assessment of Poly(ɛ-caprolactone)/Hydroxyapatite Electrospun Fibers for Bone Tissue Engineering Application. Journal of Nanomaterials 2014, 2014, 573238, 6 pages. [Google Scholar] [CrossRef]
- Dorozhkin, S. V. Calcium Orthophosphates in Nature, Biology and Medicine. Materials 2009, 2(2), 399. [Google Scholar] [CrossRef]
- Webster, TJ; Ergun, C; Doremus, RH; Siegel, RW; Bizios, R. Enhanced functions of osteoblasts on nanophase ceramics. Biomaterials 2000, 21(17), 1803–10. [Google Scholar] [CrossRef] [PubMed]
- Vaiani, L.; Boccaccio, A.; Uva, A. E.; Palumbo, G.; Piccininni, A.; Guglielmi, P.; Cantore, S.; Santacroce, L.; Charitos, I. A.; Ballini, A. Ceramic Materials for Biomedical Applications: An Overview on Properties and Fabrication Processes. Journal of Functional Biomaterials 2023, 14(3), 146. [Google Scholar] [CrossRef]
- Budharaju, H.; Suresh, S.; Sekar, M. P.; De Vega, B.; Sethuraman, S.; Sundaramurthi, D.; Kalaskar, D. M. Ceramic materials for 3D printing of biomimetic bone scaffolds – Current state-of-the-art & future perspectives. Materials & Design 2023, 231, 112064. [Google Scholar] [CrossRef]
- Sultana, N.; Cole, A.; Strachan, F. Biocomposite Scaffolds for Tissue Engineering: Materials, Fabrication Techniques and Future Directions. Materials 2024, 17, 5577. [Google Scholar] [CrossRef] [PubMed]
- Dhandayuthapani, B.; Yoshida, Y.; Maekawa, T.; Kumar, D. S. Polymeric Scaffolds in Tissue Engineering Application: A Review. International Journal of Polymer Science 2011, 2011(1), 290602. [Google Scholar] [CrossRef]
- Habiburrohman, M.R.; Jamilludin, M.A.; et al. Fabrication and in vitro cytocompatibility evaluation of porous bone scaffold based on cuttlefish bone-derived nano-carbonated hydroxyapatite reinforced with polyethylene oxide/chitosan fibrous structure. RSC Adv. 2025, 15, 8457. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, A.; Tirnoveanu, A.; et al. In Vitro Characterization of 3D-Printed PLA/CPO Oxygen Releasing Scaffolds: Mechanical and Biological Properties for Bone Tissue Engineering. J. Manuf. Mater. Process. 2025, 9, 149. [Google Scholar] [CrossRef]
- Li, C.; Chen, G.; et al. Indirect co-culture of osteoblasts and endothelial cells in vitro based on a biomimetic 3D composite hydrogel scaffold. PLoS ONE 2024, 19, e0298689. [Google Scholar] [CrossRef]
- Zohoor, A.; et al. Accelerated degradation testing of porous PLA scaffolds under alkaline conditions. Polymers 2023, 16, 876. [Google Scholar]
- Dixon, D.T.; Landree, E.N.; Gomillion, C.T. Biomimetic Dual-Sensing Bone Scaffolds: Characterization and In Vitro Evaluation Under Dynamic Culturing Conditions. Biomimetics 2025, 10, 598. [Google Scholar] [CrossRef]
- ASTM F2739-19; Standard Guide for Quantifying Cell Viability and Related Attributes within Biomaterial Scaffolds. ASTM International, 2023.
- Buckley, C.; Madhavarapu, S.; Kamara, Z.; Pellegrini, M.; Perry, B.; Gatt, C.J., Jr.; Freeman, J.W. In Vivo Evaluation of the Regenerative Capacity of a Nanofibrous, Prevascularized, Load-Bearing Scaffold for Bone Tissue Engineering. Regen. Eng. Transl. Med. 2023, 10, 56–67. [Google Scholar] [CrossRef]
- Wang, L.; Shen, M.; Tang, Z.; Tan, J.; Li, K.; Ma, H.; et al. 3D-Printed Magnesium Silicate/β-Tricalcium Phosphate Scaffolds Promote Coupled Osteogenesis and Angiogenesis by Activating PI3K/Akt Signaling Pathway. Front. Bioeng. Biotechnol. 2025, 12, 1518145. [Google Scholar] [CrossRef]
- Slavin, B.V.; Wu, S.; Sturm, S.R.; Hwang, K.K.; Almada, R.; Mirsky, N.A.; Nayak, V.V.; Witek, Ł.; Coelho, P.G. An Evaluation of Novel AMP2-Coated Electrospun Composite Scaffolds for Intraoral Bone Regeneration: A Proof-of-Concept In Vivo Study. Front. Bioeng. Biotechnol. 2025, 13, 1443280. [Google Scholar] [CrossRef]
- Ximenes-Carballo, C.; Rey-Viñolas, S.; Blanco-Fernandez, B.; Pérez-Amodio, S.; Engel, E.; Castano, O. Combining Three-Dimensionality and CaP Glass-PLA Composites: Towards an Efficient Vascularization in Bone Tissue Healing. Biomater. Adv. 2024, 164, 213985. [Google Scholar] [CrossRef]
- Vila, O.F.; Bagó, J.R.; Navarro, M.; Alieva, M.; Aguilar, E.; Engel, E.; Planell, J.; Rubio, N.; Blanco, J. Calcium Phosphate Glass Improves Angiogenesis Capacity of Poly(Lactic Acid) Scaffolds and Stimulates Differentiation of Adipose Tissue-Derived Mesenchymal Stromal Cells to the Endothelial Lineage. J. Biomed. Mater. Res. Part A 2013, 101A, 932–941. [Google Scholar] [CrossRef]
- Xu, C.; Ivanovski, S. Clinical translation of personalized bioengineered implant scaffolds. Nat Rev Bioeng 2025, 3, 390–407. [Google Scholar] [CrossRef]
- Romero, D.J.; Hussey, G.; Capella-Monsonís, H. Immune Response to Extracellular Matrix Bioscaffolds: A Comprehensive Review. Biologics 2025, 5, 28. [Google Scholar] [CrossRef]
- Zhao, X.; Li, N.; Zhang, Z.; et al. Beyond hype: unveiling the Real challenges in clinical translation of 3D printed bone scaffolds and the fresh prospects of bioprinted organoids. J Nanobiotechnol 2024, 22, 500. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




