Submitted:
13 December 2025
Posted:
15 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Synthesis of Epitaxial Thin Films
2.2. Synthesis of Freestanding Membranes
2.3. Thin Film and Freestanding Membrane Characteristics
2.4. Oxygen Surface Exchange Kinetics
2.5. Surface Analysis of Thin Films
3. Results
3.1. Crystallinity and Strain of the LSC214 Thin Films and Freestanding Membranes
3.2. Electrochemical Properties of the Epitaxial LSC214 Thin Films and Freestanding Membranes
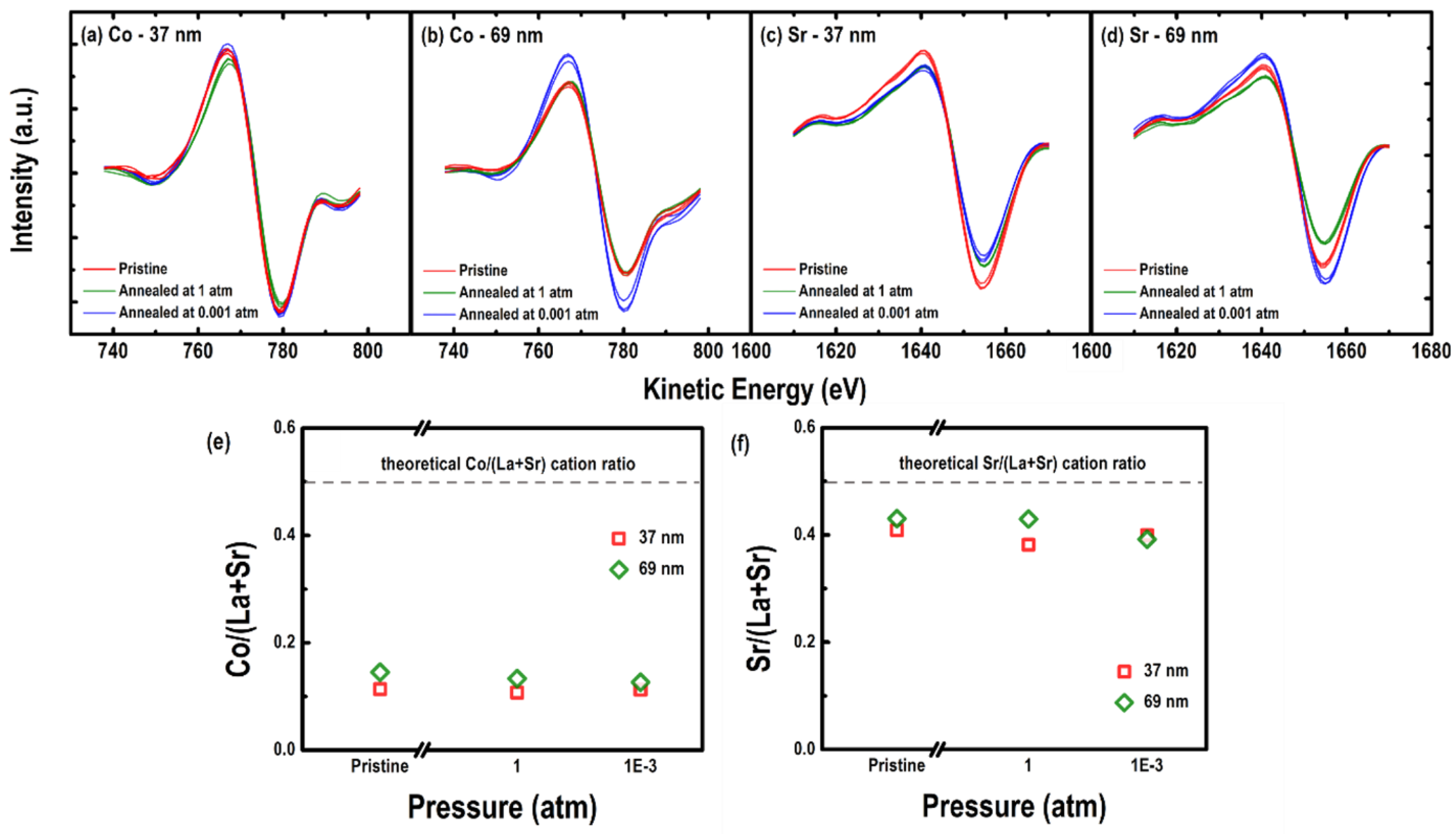
3.3. Surface Chemistry of the Epitaxial LSC214 Thin Films
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sahini, M.G.; Mwankemwa, B.S.; Kanas, N. BaxSr1-xCoyFe1-yO3-δ (BSCF) mixed ionic-electronic conducting (MIEC) materials for oxygen separation membrane and SOFC applications: insights into processing, stability, and functional properties. Ceramics International 2022, 48, 2948–2964. [Google Scholar] [CrossRef]
- Zhang, K.; Ge, L.; Ran, R.; Shao, Z.; Liu, S. Synthesis, characterization and evaluation of cation-ordered LnBaCo2O5+ δ as materials of oxygen permeation membranes and cathodes of SOFCs. Acta Materialia 2008, 56, 4876–4889. [Google Scholar] [CrossRef]
- Adler, S.B. Chemical expansivity of electrochemical ceramics. Journal of the American Ceramic Society 2001, 84, 2117–2119. [Google Scholar] [CrossRef]
- Mutoro, E.; Crumlin, E.J.; Biegalski, M.D.; Christen, H.M.; Shao-Horn, Y. Enhanced oxygen reduction activity on surface-decorated perovskite thin films for solid oxide fuel cells. Energy & Environmental Science 2011, 4, 3689–3696. [Google Scholar] [CrossRef]
- Yang, Q.; Wang, G.; Wu, H.; Beshiwork, B.A.; Tian, D.; Zhu, S.; Yang, Y.; Lu, X.; Ding, Y.; Ling, Y. A high-entropy perovskite cathode for solid oxide fuel cells. Journal of Alloys and Compounds 2021, 872, 159633. [Google Scholar] [CrossRef]
- Kim, J.H.; Kim, D.; Ahn, S.; Kim, K.J.; Jeon, S.; Lim, D.-K.; Kim, J.K.; Kim, U.; Im, H.-N.; Koo, B. An universal oxygen electrode for reversible solid oxide electrochemical cells at reduced temperatures. Energy & Environmental Science 2023, 16, 3803–3814. [Google Scholar] [CrossRef]
- Bai, J.; Zhou, D.; Zhu, X.; Wang, N.; Liang, Q.; Chen, R.; Lu, H.; Li, J.; Yan, W. Bi0. 5Sr0. 5FeO3-δ perovskite B-site doped Ln (Nd, Sm) as cathode for high performance Co-free intermediate temperature solid oxide fuel cell. Ceramics International 2023, 49, 28682–28692. [Google Scholar] [CrossRef]
- Yang, G.; Jung, W.; Ahn, S.-J.; Lee, D. Controlling the oxygen electrocatalysis on perovskite and layered oxide thin films for solid oxide fuel cell cathodes. Applied Sciences 2019, 9, 1030. [Google Scholar] [CrossRef]
- Crumlin, E.J.; Ahn, S.-J.; Lee, D.; Mutoro, E.; Biegalski, M.D.; Christen, H.M.; Shao-Horn, Y. Oxygen electrocatalysis on epitaxial La0. 6Sr0. 4CoO3-δ perovskite thin films for solid oxide fuel cells. Journal of the Electrochemical Society 2012, 159, F219. [Google Scholar] [CrossRef]
- Crumlin, E.J.; Mutoro, E.; Liu, Z.; Grass, M.E.; Biegalski, M.D.; Lee, Y.-L.; Morgan, D.; Christen, H.M.; Bluhm, H.; Shao-Horn, Y. Surface strontium enrichment on highly active perovskites for oxygen electrocatalysis in solid oxide fuel cells. Energy & Environmental Science 2012, 5, 6081–6088. [Google Scholar] [CrossRef]
- Safian, S.D.; Abd Malek, N.I.; Jamil, Z.; Lee, S.W.; Tseng, C.J.; Osman, N. Study on the surface segregation of mixed ionic-electronic conductor lanthanum-based perovskite oxide La1− xSrxCo1− yFeyO3− δ materials. International Journal of Energy Research 2022, 46, 7101–7117. [Google Scholar]
- Wei, M.; Li, H.; Chen, X.; Guo, G.; Liu, Y.; Zhang, D. First-principles study of interfacial effects toward oxygen reduction reaction of palladium/La1-xSrxCo1-yFeyO3-δ cathodes in solid oxide fuel cells. Applied Surface Science 2021, 562, 150218. [Google Scholar] [CrossRef]
- Adler, S.B. Mechanism and kinetics of oxygen reduction on porous La1− xSrxCoO3− δ electrodes. Solid State Ionics 1998, 111, 125–134. [Google Scholar] [CrossRef]
- Tyunina, M.; Pacherova, O.; Kocourek, T.; Dejneka, A. Anisotropic chemical expansion due to oxygen vacancies in perovskite films. Scientific Reports 2021, 11, 15247. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Peng, M.; Zhao, Y.; Li, J.; Sun, Y. Minimized thermal expansion mismatch of cobalt-based perovskite air electrodes for solid oxide cells. Nanoscale 2021, 13, 20299–20308. [Google Scholar] [CrossRef]
- Rupp, G.M.; Limbeck, A.; Kubicek, M.; Penn, A.; Stöger-Pollach, M.; Friedbacher, G.; Fleig, J. Correlating surface cation composition and thin film microstructure with the electrochemical performance of lanthanum strontium cobaltite (LSC) electrodes. Journal of Materials Chemistry A 2014, 2, 7099–7108. [Google Scholar] [CrossRef]
- Wen, Y.; Yang, T.; Lee, D.; Lee, H.N.; Crumlin, E.J.; Huang, K. Temporal and thermal evolutions of surface Sr-segregation in pristine and atomic layer deposition modified La 0.6 Sr 0.4 CoO 3− δ epitaxial films. Journal of Materials Chemistry A 2018, 6, 24378–24388. [Google Scholar] [CrossRef]
- Koo, B.; Kim, K.; Kim, J.K.; Kwon, H.; Han, J.W.; Jung, W. Sr segregation in perovskite oxides: why it happens and how it exists. Joule 2018, 2, 1476–1499. [Google Scholar] [CrossRef]
- Lee, D.; Grimaud, A.; Crumlin, E.J.; Mezghani, K.; Habib, M.A.; Feng, Z.; Hong, W.T.; Biegalski, M.D.; Christen, H.M.; Shao-Horn, Y. Strain influence on the oxygen electrocatalysis of the (100)-oriented epitaxial La2NiO4+ δ thin films at elevated temperatures. The Journal of Physical Chemistry C 2013, 117, 18789–18795. [Google Scholar] [CrossRef]
- Lee, D.; Lee, Y.-L.; Grimaud, A.; Hong, W.T.; Biegalski, M.D.; Morgan, D.; Shao-Horn, Y. Strontium influence on the oxygen electrocatalysis of La 2− x Sr x NiO 4±δ (0.0≤ x Sr≤ 1.0) thin films. Journal of Materials Chemistry A 2014, 2, 6480–6487. [Google Scholar]
- Burriel, M.; Garcia, G.; Santiso, J.; Kilner, J.A.; Chater, R.J.; Skinner, S.J. Anisotropic oxygen diffusion properties in epitaxial thin films of La 2 NiO 4+ δ. Journal of materials chemistry 2008, 18, 416–422. [Google Scholar] [CrossRef]
- Yang, G.; El Loubani, M.; Hill, D.; Keum, J.K.; Lee, D. Control of crystallographic orientation in Ruddlesden-Popper for fast oxygen reduction. Catalysis Today 2023, 409, 87–93. [Google Scholar] [CrossRef]
- Ma, T.; Xia, T.; Li, Q.; Sun, L.; Huo, L.; Zhao, H. Highly electrocatalytic activity Ruddlesden− Popper type electrode materials for solid oxide fuel cells. Journal of the European Ceramic Society 2022, 42, 490–498. [Google Scholar] [CrossRef]
- Lee, D.; Lee, H.N. Controlling oxygen mobility in Ruddlesden–Popper oxides. Materials 2017, 10, 368. [Google Scholar] [CrossRef]
- Chen, Y.; Téllez, H.; Burriel, M.n.; Yang, F.; Tsvetkov, N.; Cai, Z.; McComb, D.W.; Kilner, J.A.; Yildiz, B. Segregated chemistry and structure on (001) and (100) surfaces of (La1–x Sr x) 2CoO4 override the crystal anisotropy in oxygen exchange kinetics. Chemistry of Materials 2015, 27, 5436–5450. [Google Scholar] [CrossRef]
- Boehm, E.; Bassat, J.-M.; Steil, M.; Dordor, P.; Mauvy, F.; Grenier, J.-C. Oxygen transport properties of La2Ni1− xCuxO4+ δ mixed conducting oxides. Solid State Sciences 2003, 5, 973–981. [Google Scholar] [CrossRef]
- Munnings, C.; Skinner, S.; Amow, G.; Whitfield, P.; Davidson, I. Oxygen transport in the La2Ni1− xCoxO4+ δ system. Solid State Ionics 2005, 176, 1895–1901. [Google Scholar] [CrossRef]
- Brown, I.; Altermatt, D. Bond-valence parameters obtained from a systematic analysis of the inorganic crystal structure database. Acta Crystallographica Section B: Structural Science 1985, 41, 244–247. [Google Scholar] [CrossRef]
- Nitadori, T.; Muramatsu, M.; Misono, M. Valence control, reactivity of oxygen, and catalytic activity of lanthanum strontium cobalt oxide (La2-xSrxCoO4). Chemistry of Materials 1989, 1, 215–220. [Google Scholar] [CrossRef]
- Zhao, C.; Liu, X.; Zhang, W.; Zheng, Y.; Li, Y.; Yu, B.; Wang, J.; Chen, J. Measurement of oxygen reduction/evolution kinetics enhanced (La, Sr) CoO3/(La, Sr) 2CoO4 hetero-structure oxygen electrode in operating temperature for SOCs. International Journal of Hydrogen Energy 2019, 44, 19102–19112. [Google Scholar] [CrossRef]
- Lee, D.; Lee, Y.-L.; Hong, W.T.; Biegalski, M.D.; Morgan, D.; Shao-Horn, Y. Oxygen surface exchange kinetics and stability of (La, Sr) 2 CoO 4±δ/La 1− x Sr x MO 3− δ (M= Co and Fe) hetero-interfaces at intermediate temperatures. Journal of Materials Chemistry A 2015, 3, 2144–2157. [Google Scholar] [CrossRef]
- Fleig, J.; Baumann, F.; Brichzin, V.; Kim, H.R.; Jamnik, J.; Cristiani, G.; Habermeier, H.U.; Maier, J. Thin film microelectrodes in SOFC electrode research. Fuel Cells 2006, 6, 284–292. [Google Scholar] [CrossRef]
- Lee, D.; Jacobs, R.; Jee, Y.; Seo, A.; Sohn, C.; Ievlev, A.V.; Ovchinnikova, O.S.; Huang, K.; Morgan, D.; Lee, H.N. Stretching epitaxial La0. 6Sr0. 4CoO3− δ for fast oxygen reduction. The Journal of Physical Chemistry C 2017, 121, 25651–25658. [Google Scholar]
- Yang, G.; El Loubani, M.; Handrick, D.; Stevenson, C.; Lee, D. Understanding the influence of strain-modified oxygen vacancies and surface chemistry on the oxygen reduction reaction of epitaxial La0. 8Sr0. 2CoO3-δ thin films. Solid State Ionics 2023, 393, 116171. [Google Scholar] [CrossRef]
- Yang, G.; El Loubani, M.; Chalaki, H.R.; Kim, J.; Keum, J.K.; Rouleau, C.M.; Lee, D. Tuning ionic conductivity in fluorite Gd-doped CeO2-Bixbyite RE2O3 (RE= Y and Sm) multilayer thin films by controlling interfacial strain. ACS Applied Electronic Materials 2023, 5, 4556–4563. [Google Scholar] [CrossRef]
- Wen, K.; Lv, W.; He, W. Interfacial lattice-strain effects on improving the overall performance of micro-solid oxide fuel cells. Journal of Materials Chemistry A 2015, 3, 20031–20050. [Google Scholar] [CrossRef]
- Santiso, J.; Burriel, M. Deposition and characterisation of epitaxial oxide thin films for SOFCs. Journal of Solid State Electrochemistry 2011, 15, 985–1006. [Google Scholar] [CrossRef]
- Bassat, J.-M.; Burriel, M.; Wahyudi, O.; Castaing, R.m.; Ceretti, M.; Veber, P.; Weill, I.; Villesuzanne, A.; Grenier, J.-C.; Paulus, W. Anisotropic oxygen diffusion properties in Pr2NiO4+ δ and Nd2NiO4+ δ single crystals. The Journal of Physical Chemistry C 2013, 117, 26466–26472. [Google Scholar] [CrossRef]
- Burriel, M.; Téllez, H.; Chater, R.J.; Castaing, R.; Veber, P.; Zaghrioui, M.; Ishihara, T.; Kilner, J.A.; Bassat, J.-M. Influence of crystal orientation and annealing on the oxygen diffusion and surface exchange of La2NiO4+ δ. The Journal of Physical Chemistry C 2016, 120, 17927–17938. [Google Scholar] [CrossRef]
- Tsvetkov, N.; Lu, Q.; Chen, Y.; Yildiz, B. Accelerated oxygen exchange kinetics on Nd2NiO4+ δ thin films with tensile strain along c-axis. ACS nano 2015, 9, 1613–1621. [Google Scholar] [CrossRef] [PubMed]
- Yun, S.; le Cozannet, T.E.; Christoffersen, C.H.; Brand, E.; Jespersen, T.S.; Pryds, N. Strain engineering: perfecting freestanding perovskite oxide fabrication. Small 2024, 20, 2310782. [Google Scholar] [CrossRef]
- Lu, Q.; Liu, Z.; Yang, Q.; Cao, H.; Balakrishnan, P.; Wang, Q.; Cheng, L.; Lu, Y.; Zuo, J.-M.; Zhou, H. Engineering magnetic anisotropy and emergent multidirectional soft ferromagnetism in ultrathin freestanding LaMnO3 films. ACS nano 2022, 16, 7580–7588. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Xiang, C.; Chiabrera, F.M.; Yun, S.; Zhang, H.; Kelly, D.J.; Dahm, R.T.; Kirchert, C.K.; Cozannet, T.E.L.; Trier, F. Stacking and twisting of freestanding complex oxide thin films. Advanced materials 2022, 34, 2203187. [Google Scholar] [CrossRef]
- Ji, D.; Cai, S.; Paudel, T.R.; Sun, H.; Zhang, C.; Han, L.; Wei, Y.; Zang, Y.; Gu, M.; Zhang, Y. Freestanding crystalline oxide perovskites down to the monolayer limit. Nature 2019, 570, 87–90. [Google Scholar] [CrossRef]
- Varshney, S.; Ramis, M.; Choo, S.; Coll, M.; Jalan, B. Epitaxially grown single-crystalline SrTiO 3 membranes using a solution-processed, amorphous SrCa 2 Al 2 O 6 sacrificial layer. Journal of Materials Chemistry C 2024, 12, 13809–13815. [Google Scholar] [CrossRef]
- Dong, G.; Li, S.; Li, T.; Wu, H.; Nan, T.; Wang, X.; Liu, H.; Cheng, Y.; Zhou, Y.; Qu, W. Periodic wrinkle-patterned single-crystalline ferroelectric oxide membranes with enhanced piezoelectricity. Advanced materials 2020, 32, 2004477. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Yuan, H.; Dong, G.; Wu, K.; Liu, G.; Sun, J.; Zhou, Z.; Liu, M. Tunable friction properties of periodic wrinkled BaTiO3 membranes. Advanced Materials Interfaces 2022, 9, 2102316. [Google Scholar] [CrossRef]
- Liu, W.; Wang, H. Flexible oxide epitaxial thin films for wearable electronics: Fabrication, physical properties, and applications. Journal of Materiomics 2020, 6, 385–396. [Google Scholar] [CrossRef]
- la O, G.J.; Ahn, S.J.; Crumlin, E.; Orikasa, Y.; Biegalski, M.D.; Christen, H.M.; Shao-Horn, Y. Catalytic activity enhancement for oxygen reduction on epitaxial perovskite thin films for solid-oxide fuel cells. Angewandte Chemie (International ed. in English) 2010, 49. [Google Scholar] [CrossRef]
- Mitterdorfer, A.; Gauckler, L. La2Zr2O7 formation and oxygen reduction kinetics of the La0. 85Sr0. 15MnyO3, O2 (g)| YSZ system. Solid State Ionics 1998, 111, 185–218. [Google Scholar]
- Yashima, M.; Sasaki, S.; Kakihana, M.; Yamaguchi, Y.; Arashi, H.; Yoshimura, M. Oxygen-induced structural change of the tetragonal phase around the tetragonal–cubic phase boundary in ZrO2–YO1. 5 solid solutions. Structural Science 1994, 50, 663–672. [Google Scholar]
- James, M.; Tedesco, A.; Cassidy, D.; Colella, M.; Smythe, P. The phase diagram and crystal chemistry of strontium-doped rare earth cobaltates: Ln2− xSrxCoO4+ δ (Ln= La–Dy). Journal of alloys and compounds 2006, 419, 201–207. [Google Scholar]
- Christen, H.M.; Specht, E.D.; Silliman, S.S.; Harshavardhan, K. Ferroelectric and antiferroelectric coupling in superlattices of paraelectric perovskites at room temperature. Physical Review B 2003, 68, 020101. [Google Scholar] [CrossRef]
- Noh, D.; Hwu, Y.; Je, J.; Hong, M.; Mannaerts, J. Strain relaxation in Fe3 (Al, Si)/GaAs: An x-ray scattering study. Applied physics letters 1996, 68, 1528–1530. [Google Scholar] [CrossRef]
- Maier, J. On the correlation of macroscopic and microscopic rate constants in solid state chemistry. Solid State Ionics 1998, 112, 197–228. [Google Scholar] [CrossRef]
- Fleig, J.; Maier, J. The polarization of mixed conducting SOFC cathodes: Effects of surface reaction coefficient, ionic conductivity and geometry. Journal of the European Ceramic Society 2004, 24, 1343–1347. [Google Scholar] [CrossRef]
- Maier, J. Physical chemistry of ionic materials: ions and electrons in solids; John Wiley & Sons, 2023. [Google Scholar]
- Vashook, V.; Ullmann, H.; Olshevskaya, O.; Kulik, V.; Lukashevich, V.; Kokhanovskij, L. Composition and electrical conductivity of some cobaltates of the type La2− xSrxCoO4. 5− x/2±δ. Solid State Ionics 2000, 138, 99–104. [Google Scholar]
- Lee, Y.-L.; Lee, D.; Wang, X.R.; Lee, H.N.; Morgan, D.; Shao-Horn, Y. Kinetics of oxygen surface exchange on epitaxial Ruddlesden–Popper phases and correlations to first-principles descriptors. The Journal of Physical Chemistry Letters 2016, 7, 244–249. [Google Scholar] [CrossRef] [PubMed]
- Adler, S.B. Factors governing oxygen reduction in solid oxide fuel cell cathodes. Chemical reviews 2004, 104, 4791–4844. [Google Scholar]
- Adler, S.B.; Lane, J.A.; Steele, B.C. Electrode kinetics of porous mixed-conducting oxygen electrodes. Journal of the electrochemical society 1996, 143, 3554. [Google Scholar]
- Kawada, T.; Suzuki, J.; Sase, M.; Kaimai, A.; Yashiro, K.; Nigara, Y.; Mizusaki, J.; Kawamura, K.; Yugami, H. Determination of oxygen vacancy concentration in a thin film of La0. 6Sr0. 4CoO3− δ by an electrochemical method. Journal of the Electrochemical Society 2002, 149, E252. [Google Scholar] [CrossRef]
- Yang, Y.; Chen, C.; Chen, S.; Chu, C.; Jacobson, A. Impedance studies of oxygen exchange on dense thin film electrodes of La0. 5Sr0. 5CoO3− δ. Journal of the Electrochemical Society 2000, 147, 4001. [Google Scholar]
- Takeda, Y.; Kanno, R.; Noda, M.; Tomida, Y.; Yamamoto, O. Cathodic polarization phenomena of perovskite oxide electrodes with stabilized zirconia. Journal of the Electrochemical Society 1987, 134, 2656. [Google Scholar] [CrossRef]
- Escudero, M.; Aguadero, A.; Alonso, J.A.; Daza, L. A kinetic study of oxygen reduction reaction on La2NiO4 cathodes by means of impedance spectroscopy. Journal of Electroanalytical Chemistry 2007, 611, 107–116. [Google Scholar]
- Tealdi, C.; Ferrara, C.; Mustarelli, P.; Islam, M.S. Vacancy and interstitial oxide ion migration in heavily doped La 2− x Sr x CoO 4±δ. Journal of Materials Chemistry 2012, 22, 8969–8975. [Google Scholar] [CrossRef]
- Xie, W.; Lee, Y.-L.; Shao-Horn, Y.; Morgan, D. Oxygen Point Defect Chemistry in Ruddlesden–Popper Oxides (La1–x Sr x) 2MO4±δ (M= Co, Ni, Cu). The journal of physical chemistry letters 2016, 7, 1939–1944. [Google Scholar]
- Miyoshi, S.; Takeshita, A.; Okada, S.; Yamaguchi, S. Rate-determining elementary step of oxygen reduction reaction at (La, Sr) CoO3-based cathode surface. Solid State Ionics 2016, 285, 202–208. [Google Scholar]
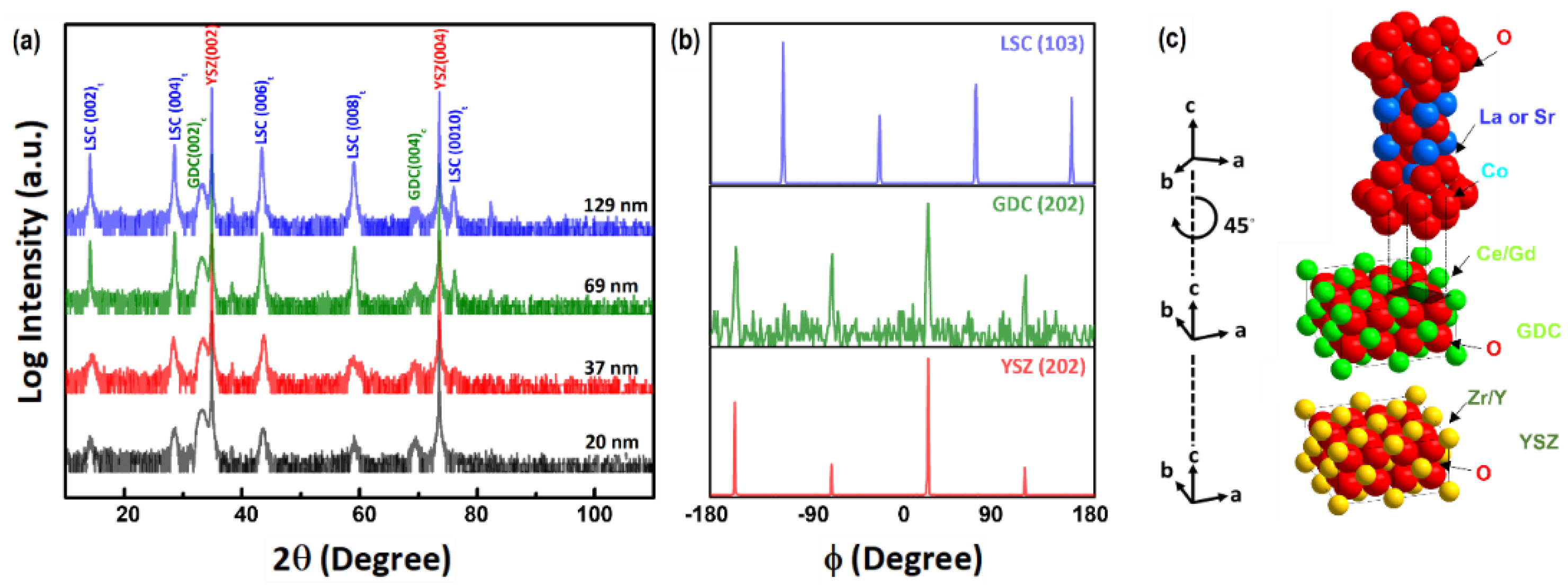
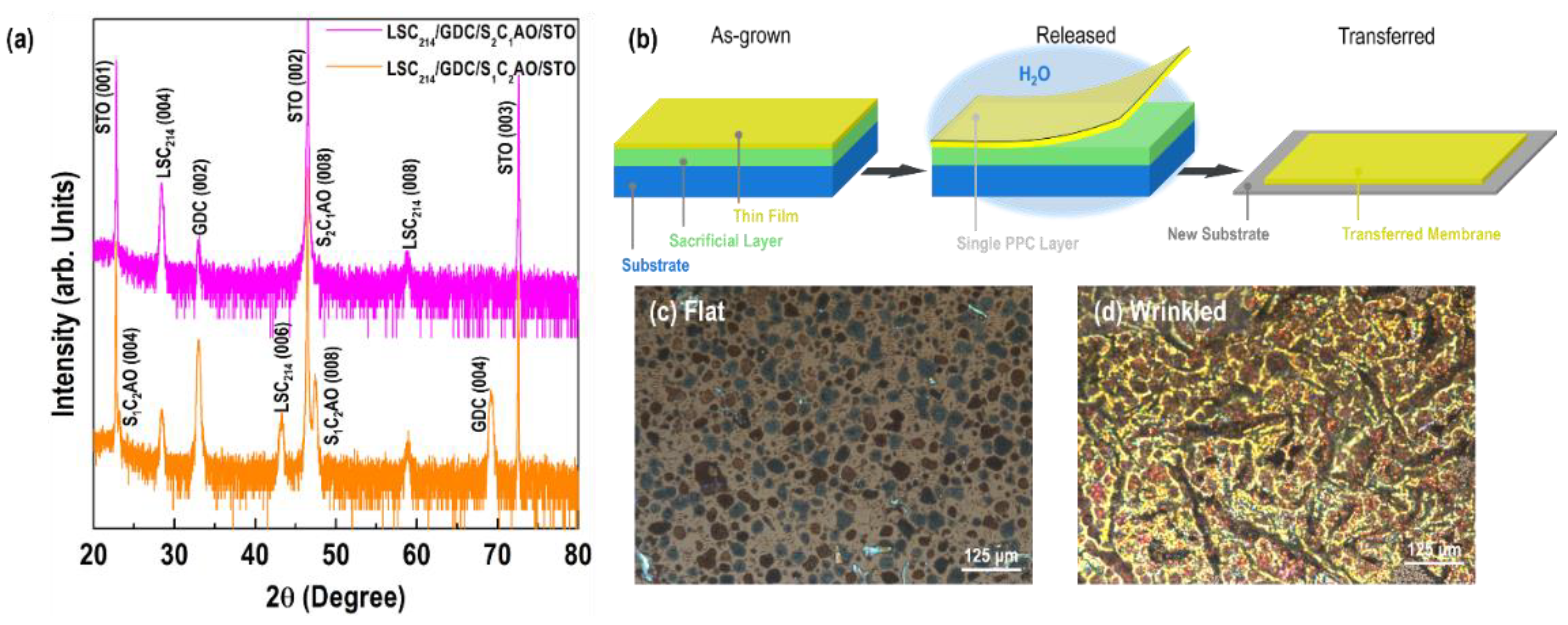
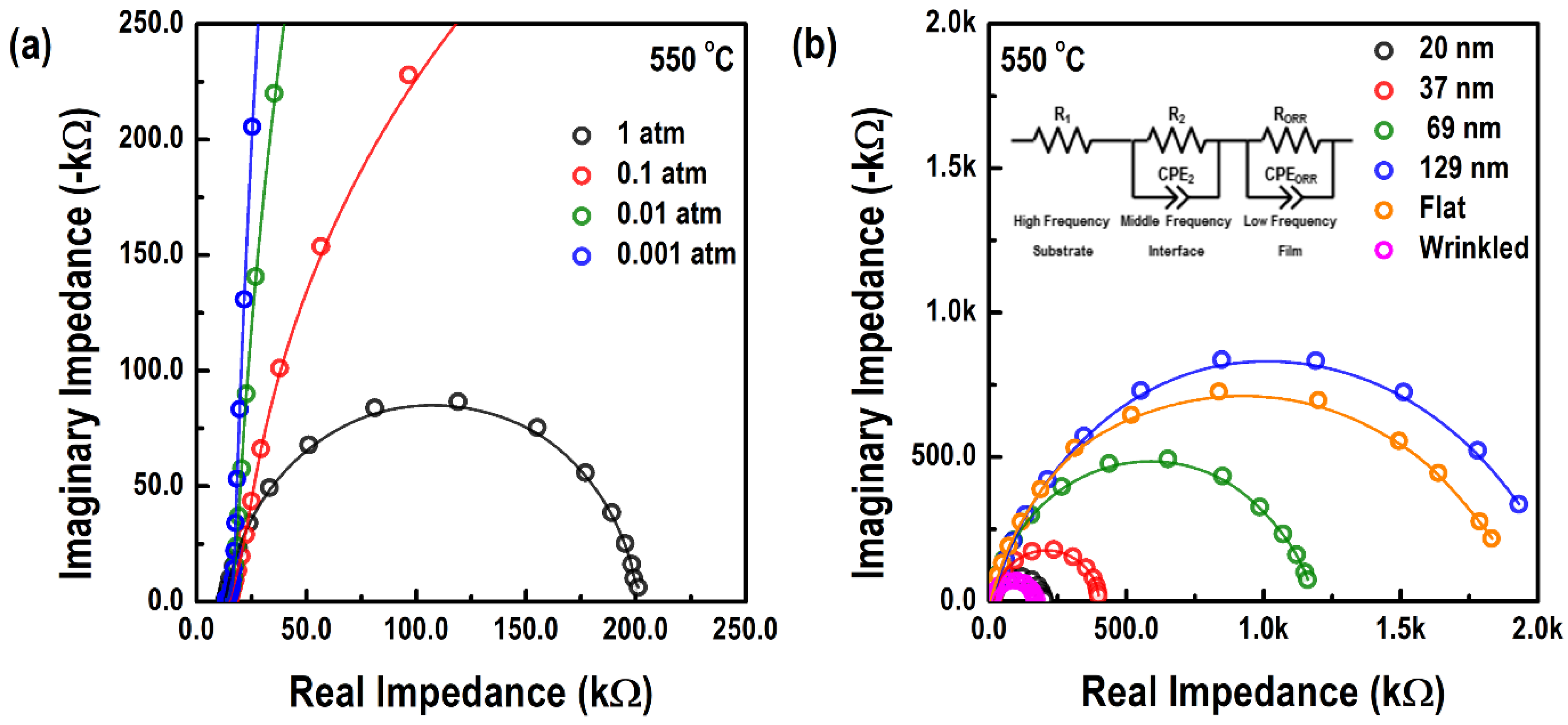
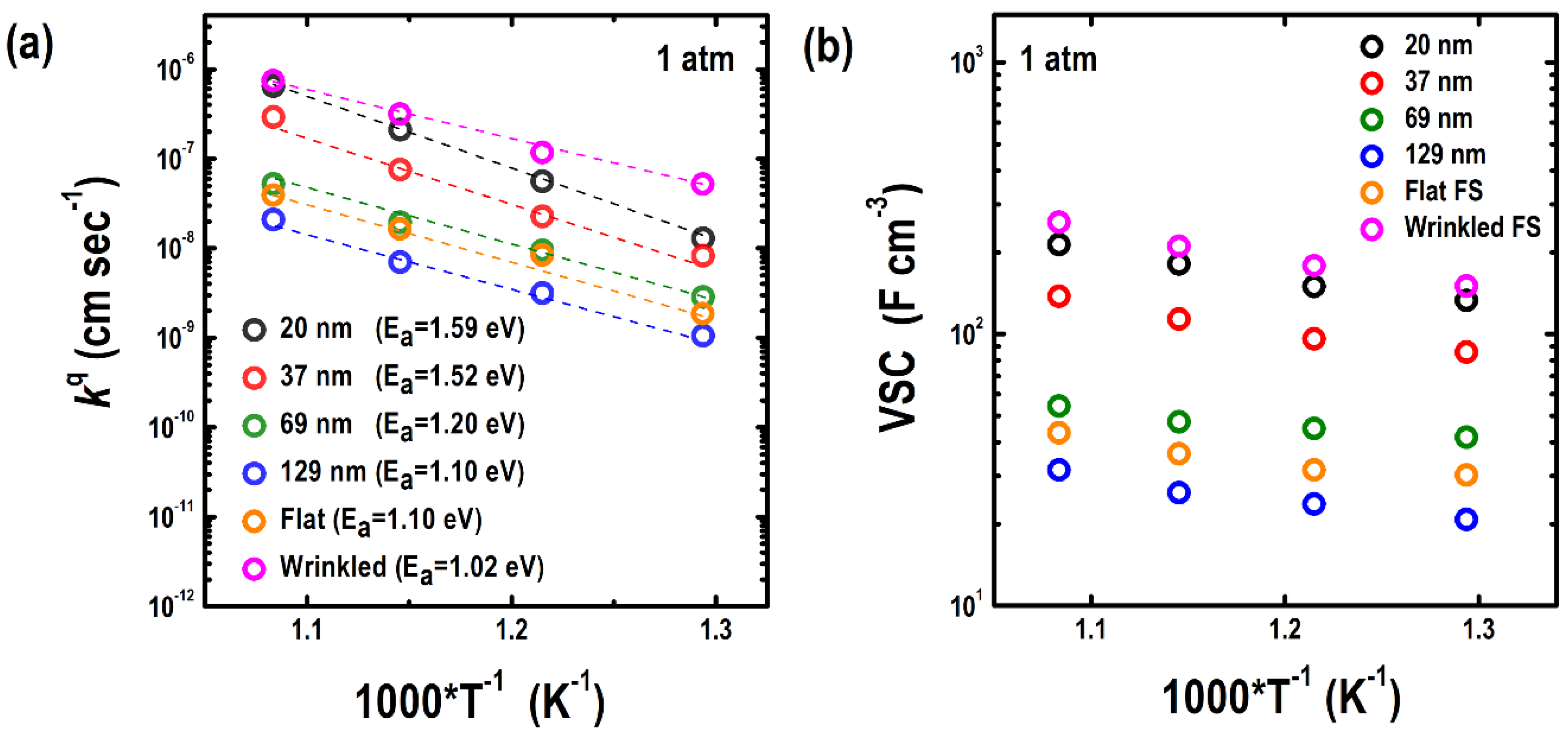
| Film Thickness (nm) | a (Å) | c (Å) | â (Å) | ĉ (Å) | a strain (%) | c strain (%) |
|---|---|---|---|---|---|---|
| 20 | 3.832 | 12.454 | 3.811 | 12.515 | 0.574 | -0.492 |
| 37 | 3.828 | 12.467 | 3.811 | 12.516 | 0.457 | -0.392 |
| 69 | 3.813 | 12.491 | 3.808 | 12.505 | 0.134 | -0.115 |
| 129 | 3.806 | 12.506 | 3.807 | 12.503 | -0.020 | 0.017 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




