Submitted:
14 December 2025
Posted:
15 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
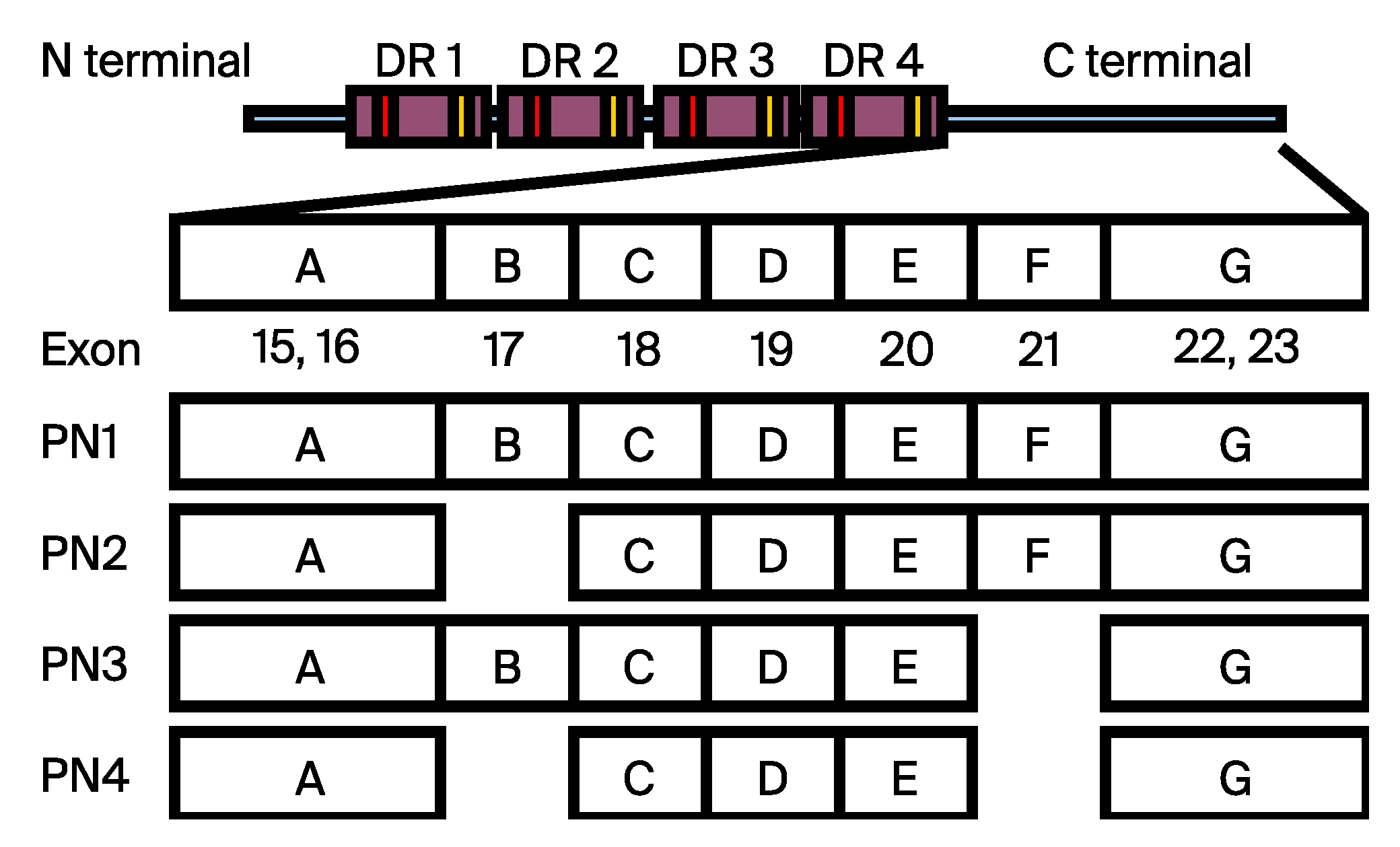
2. Materials and Methods
2.1. Cell Culture
2.2. Quantitative Real-Time PCR
- GAPDH (5′-GAAGCAGGCATCTGAGGGC-3′, 5′-TTGAAGTCGCAGGAGACAACC-3′)
- hPN1(5′-GTGATTGAAGGCAGTCTTCAGCC-3′,5′-CTCCCTGAAGCAGTCTTTTA-3′), hPN2 (5′-AATCCCCGTGACTGTCTATAGACC-3′, 5′-CTCCCTGAAGCAGTCTTTTA-3′),
- hPN3 (5′-GTGATTGAAGGCAGTCTTCAGCC-3′, 5′-TCCTCACGGGTGTGTCTTCT-3′),
- hPN4(5′-AATCCCCGTGACTGTCTATAGACC-3′,5′-TCCTCACGGGTGTGTCTTCT-3′), CDH1 (5′-TGCCCAGAAAATGAAAAAGG-3′, 5′-GTGTATGTGGCAATGCGTTC-3′), SNAI1 (5′-AAGATGCACATCCGAAGCC-3′,5′-CGCAGGTTGGAGCGGTCAGC-3′), SNAI2 (5′-AAGCATTTCAACGCCTCCAAA-3′, 5′-GGATCTCTGGTTGTGGTATGACA-3′), TWIST1 (5′-AAGAGGTCGTGCCAATCAG-3′,5′-GGCCAGTTTGATCCCAGTAT-3′), ZEB1 (5′-GATGATGAATGCGAGTCAGATGC-3′,5′-ACAGCAGTGTCTTGTTGTTGTAG-3′),
2.3. In Vivo Mouse Experiment
2.4. Anti-Human POSTN Exon 21 Antibody
2.5. Statistical Analysis
2.6. Ethical Statement
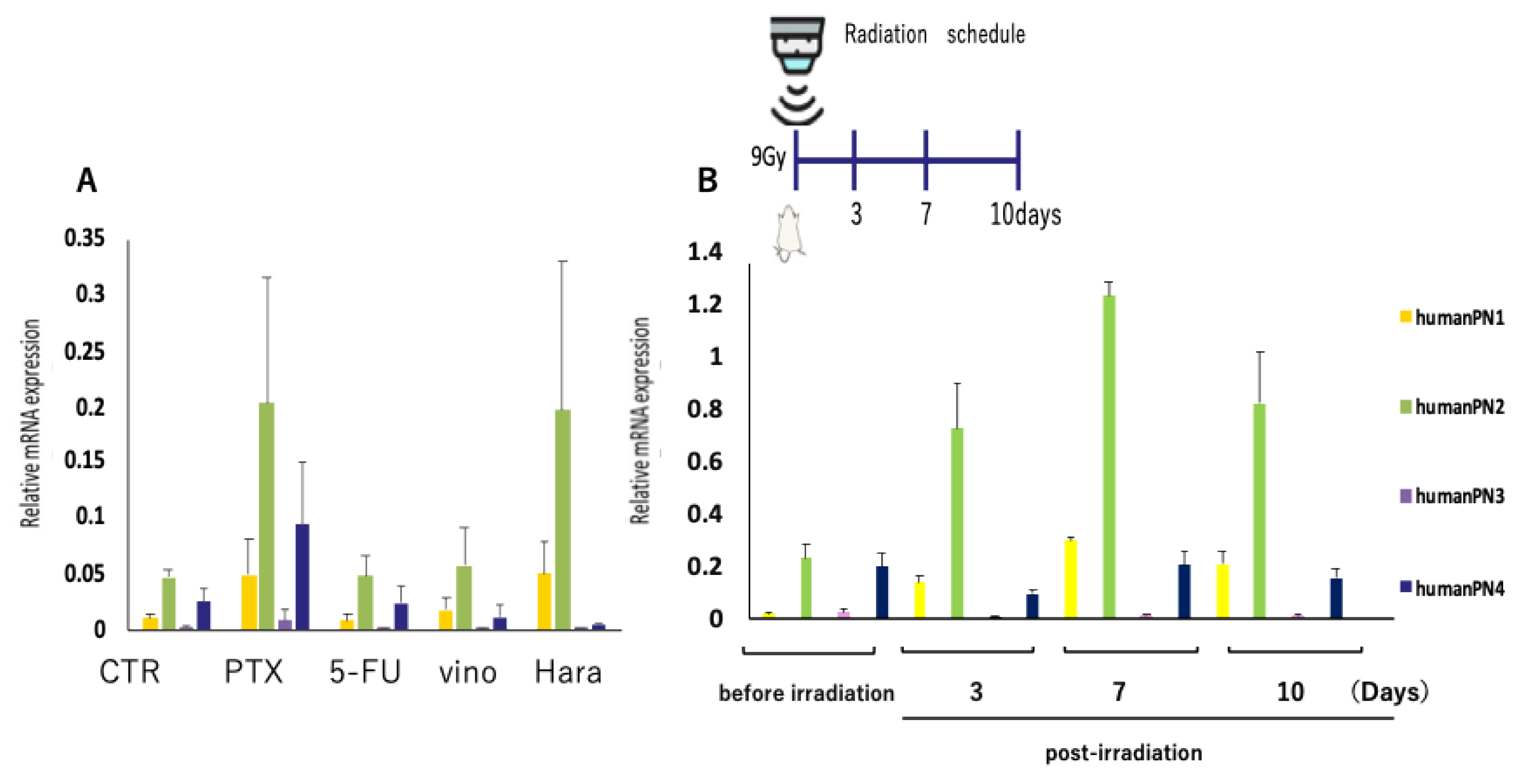
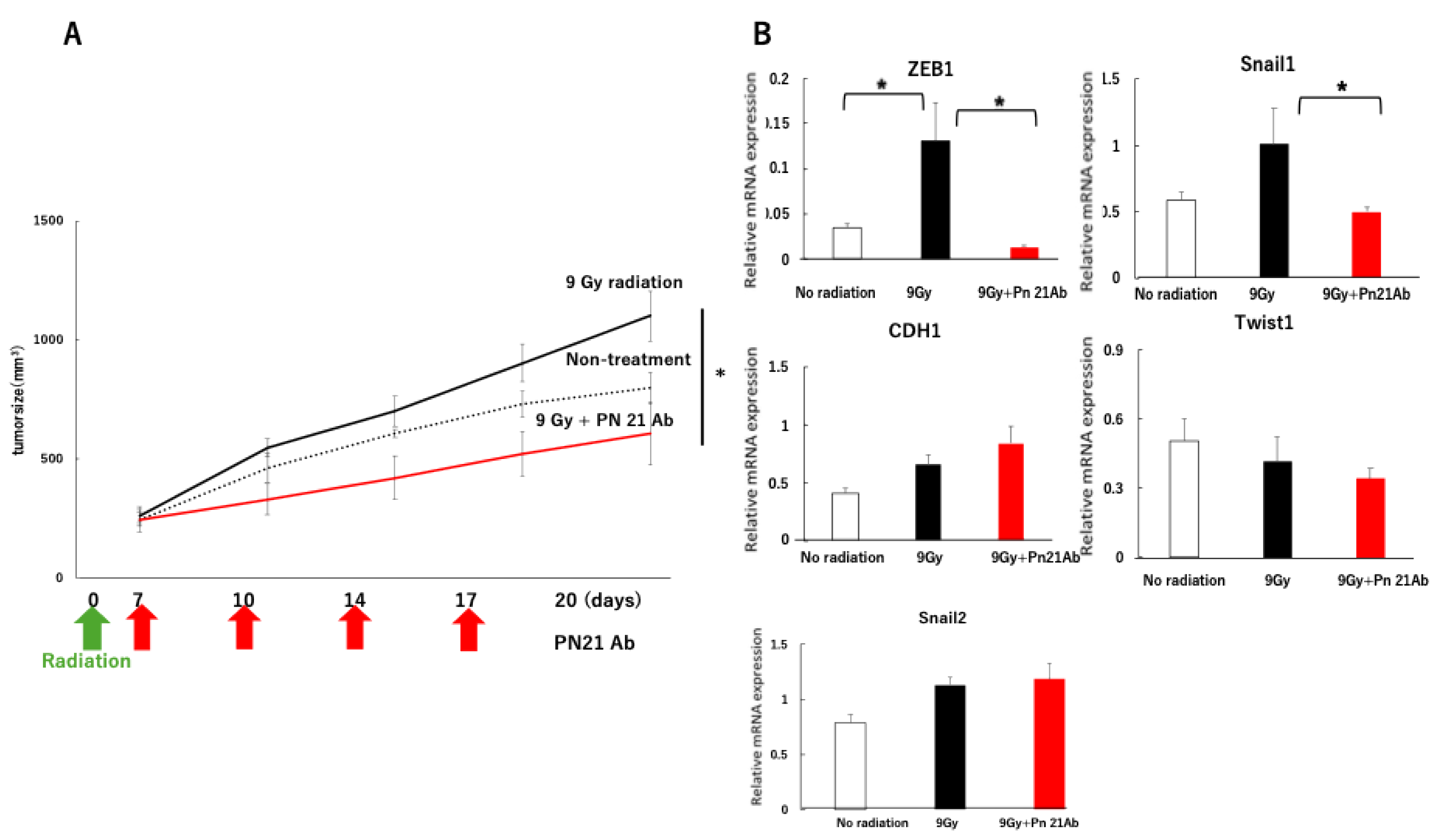
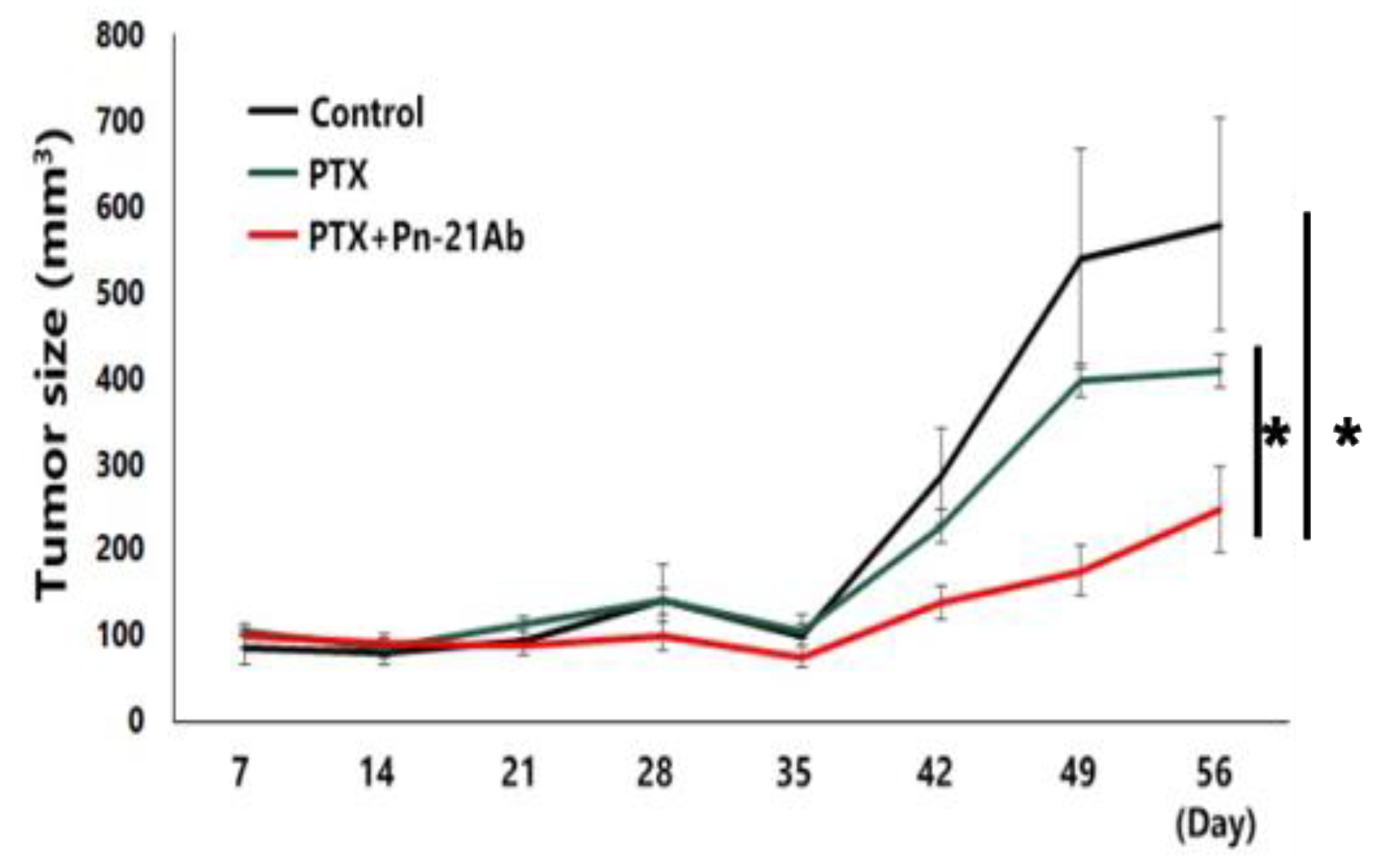
3. Results
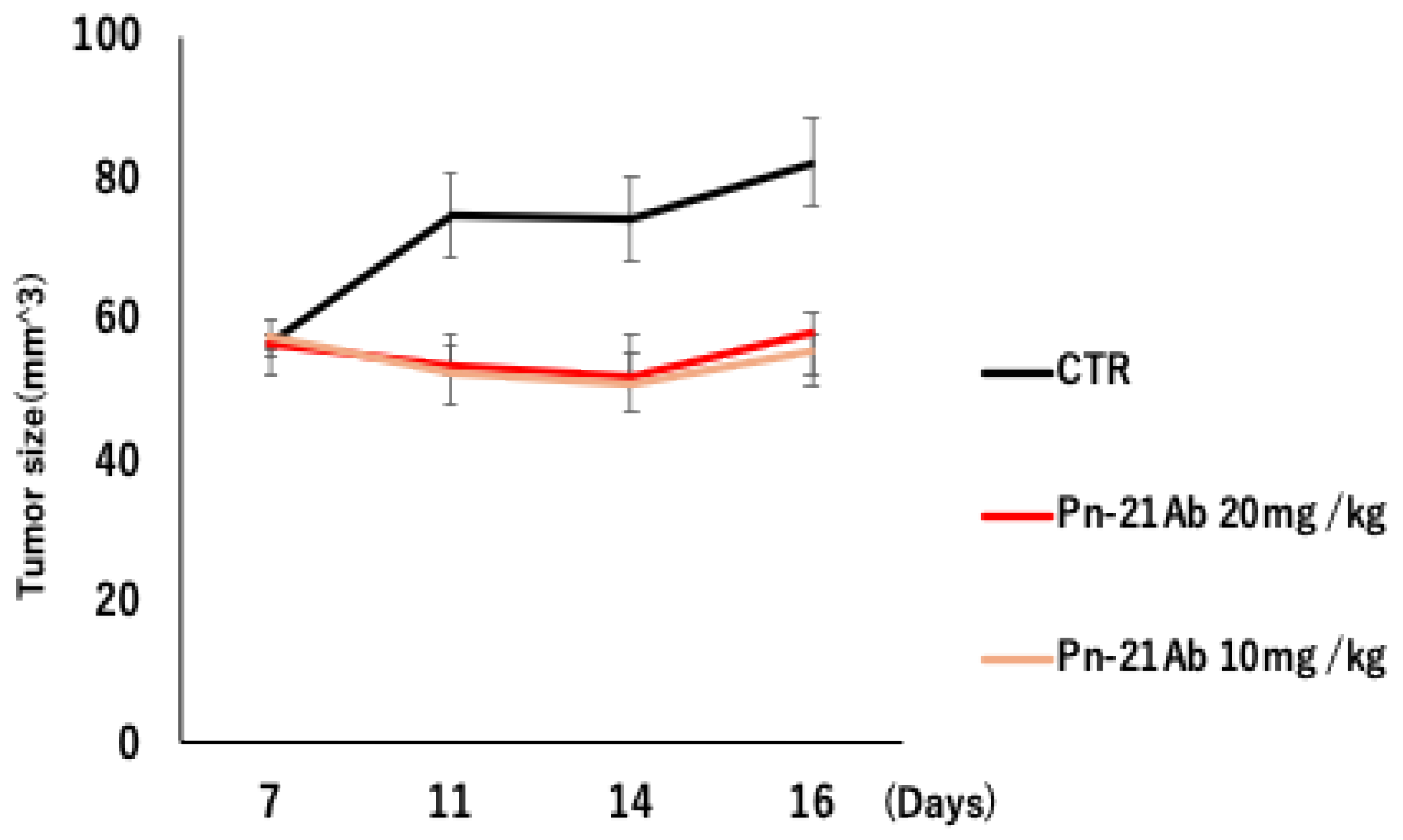
3.1. Antitumor Activity of PN-21Ab in Luminal Breast Cancer Model (ZR75-1)
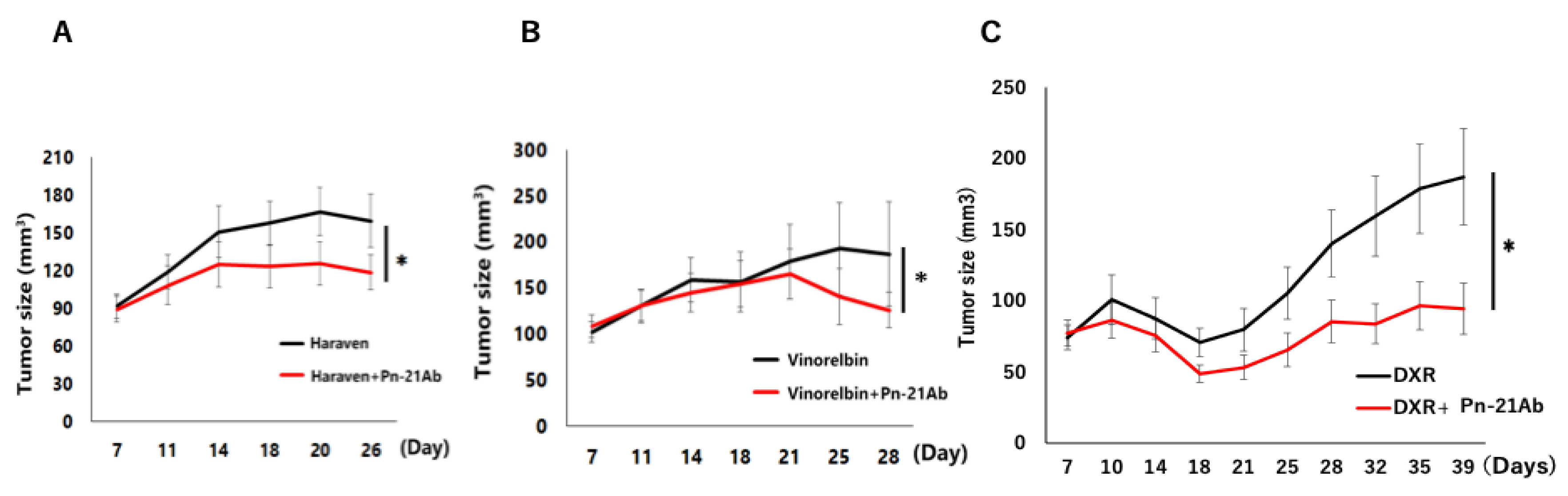
3.2. PN-21Ab Synergistically Enhances the Antitumor Effects of Microtubule-Targeting and Cytotoxic Agents
3.3. PN-21Ab Enhances the Antitumor Efficacy of Radiation and Suppresses EMT Activation
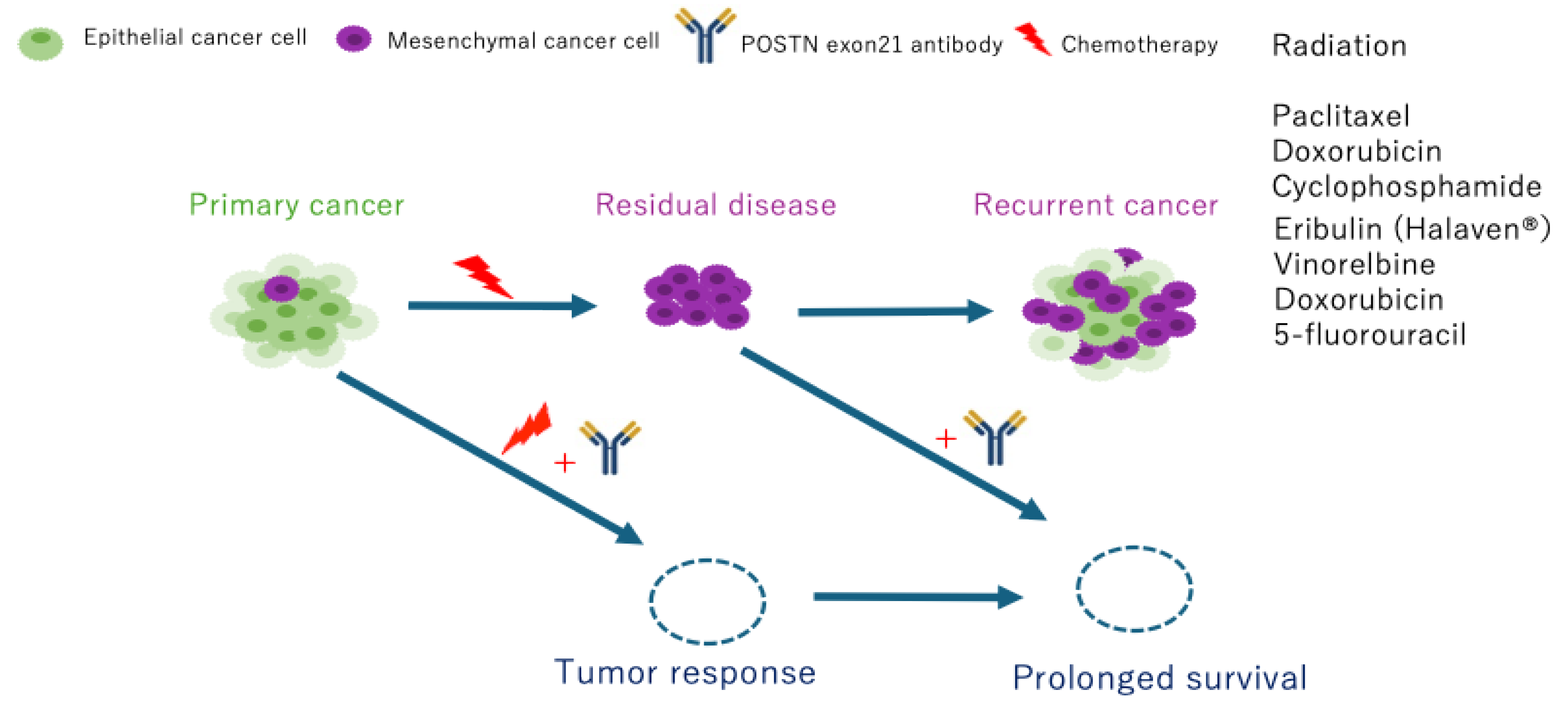
4. Discussion
5. Patents
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Abbreviation | Full term |
| ADC | Antibody–drug conjugate |
| Akt | Protein kinase B |
| CAF | Cancer-associated fibroblast |
| CDH1 | Cadherin 1 (E-cadherin) |
| cDNA | Complementary DNA |
| DMEM | Dulbecco’s Modified Eagle’s Medium |
| ECM | Extracellular matrix |
| EMT | Epithelial–mesenchymal transition |
| ER | Estrogen receptor |
| ERK | Extracellular signal-regulated kinase |
| Ex | Exon |
| FAS1 | Fasciclin-like domain 1 |
| FBS | Fetal bovine serum |
| 5-FU | 5-Fluorouracil |
| GAPDH | Glyceraldehyde-3-phosphate dehydrogenase |
| Gy | Gray (unit of absorbed radiation dose) |
| HER2 | Human epidermal growth factor receptor 2 |
| IL | Interleukin |
| i.p. | Intraperitoneal injection |
| i.v. | Intravenous injection |
| MCF10DCIS | MCF10DCIS.com human breast cancer cell line |
| MDA-MB-231 | Human triple-negative breast cancer cell line |
| MEXT | Ministry of Education, Culture, Sports, Science and Technology (Japan) |
| mRNA | Messenger RNA |
| PBS | Phosphate-buffered saline |
| PCR | Polymerase chain reaction |
| PN | Periostin isoform (PN1–PN4) |
| PN-21Ab | Anti-periostin exon 21 monoclonal antibody |
| POSTN | Periostin |
| PR | Progesterone receptor |
| PTX | Paclitaxel |
| qRT-PCR | Quantitative reverse transcription polymerase chain reaction |
| SD | Standard deviation |
| SPF | Specific pathogen-free |
| TGF-β | Transforming growth factor beta |
| TME | Tumor microenvironment |
| TNBC | Triple-negative breast cancer |
| VC | Vinorelbine combination group |
| ZEB1 | Zinc finger E-box-binding homeobox 1 |
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin 2021, 71, 209–249. [CrossRef]
- Bianchini, G.; Balko, J.M.; Mayer, I.A.; Sanders, M.E.; Gianni, L. Triple-Negative Breast Cancer: Challenges and Opportunities of a Heterogeneous Disease. Nat Rev Clin Oncol 2016, 13, 674–690.
- Foulkes, W.D.; Smith, I.E.; Reis-Filho, J.S. Triple-Negative Breast Cancer; 2010; Vol. 363;.
- Articles Comparisons between Diff Erent Polychemotherapy Regimens for Early Breast Cancer: Meta-Analyses of Long-Term Outcome among 100 000 Women in 123 Randomised Trials. 2011. [CrossRef]
- Conway, S.J.; Izuhara, K.; Kudo, Y.; Litvin, J.; Markwald, R.; Ouyang, G.; Arron, J.R.; Holweg, C.T.J.; Kudo, A. The Role of Periostin in Tissue Remodeling across Health and Disease. Cellular and Molecular Life Sciences 2014, 71, 1279–1288.
- Kudo, A. Periostin in Fibrillogenesis for Tissue Regeneration: Periostin Actions inside and Outside the Cell. Cellular and Molecular Life Sciences 2011, 68, 3201–3207.
- Takayama, G.; Arima, K.; Kanaji, T.; Toda, S.; Tanaka, H.; Shoji, S.; McKenzie, A.N.J.; Nagai, H.; Hotokebuchi, T.; Izuhara, K. Periostin: A Novel Component of Subepithelial Fibrosis of Bronchial Asthma Downstream of IL-4 and IL-13 Signals. Journal of Allergy and Clinical Immunology 2006, 118, 98–104. [CrossRef]
- Katsuragi, N.; Morishita, R.; Nakamura, N.; Ochiai, T.; Taniyama, Y.; Hasegawa, Y.; Kawashima, K.; Kaneda, Y.; Ogihara, T.; Sugimura, K. Periostin as a Novel Factor Responsible for Ventricular Dilation. Circulation 2004, 110, 1806–1813. [CrossRef]
- Yoshida, S.; Nakao, S.; Nakama, T.; Kita, T.; Asato, R.; Sassa, Y.; Arita, R.; Miyazaki, M.; Enaida, H.; Oshima, Y.; et al. Periostin Promotes the Generation of Fibrous Membranes in Proliferative Vitreoretinopathy Keijiro Ishikawa1. FASEB Journal 2014, 28, 131–142. [CrossRef]
- Sidhu, S.S.; Yuan, S.; Innes, A.L.; Kerr, S.; Woodruff, P.G.; Hou, L.; Muller, S.J.; Fahy, J. V. Roles of Epithelial Cell-Derived Periostin in TGF-β Activation, Collagen Production, and Collagen Gel Elasticity in Asthma. Proc Natl Acad Sci U S A 2010, 107, 14170–14175. [CrossRef]
- Katsuragi, N.; Morishita, R.; Nakamura, N.; Ochiai, T.; Taniyama, Y.; Hasegawa, Y.; Kawashima, K.; Kaneda, Y.; Ogihara, T.; Sugimura, K. Periostin as a Novel Factor Responsible for Ventricular Dilation. Circulation 2004, 110, 1806–1813. [CrossRef]
- Malanchi, I.; Santamaria-Martínez, A.; Susanto, E.; Peng, H.; Lehr, H.A.; Delaloye, J.F.; Huelsken, J. Interactions between Cancer Stem Cells and Their Niche Govern Metastatic Colonization. Nature 2012, 481, 85–91. [CrossRef]
- González-González, L.; Alonso, J. Periostin: A Matricellular Protein with Multiple Functions in Cancer Development and Progression. Front Oncol 2018, 8.
- Shao, R.; Bao, S.; Bai, X.; Blanchette, C.; Anderson, R.M.; Dang, T.; Gishizky, M.L.; Marks, J.R.; Wang, X.-F. Acquired Expression of Periostin by Human Breast Cancers Promotes Tumor Angiogenesis through Up-Regulation of Vascular Endothelial Growth Factor Receptor 2 Expression. Mol Cell Biol 2004, 24, 3992–4003. [CrossRef]
- Nakazawa, Y.; Taniyama, Y.; Sanada, F.; Morishita, R.; Nakamori, S.; Morimoto, K.; Yeung, K.T.; Yang, J. Periostin Blockade Overcomes Chemoresistance via Restricting the Expansion of Mesenchymal Tumor Subpopulations in Breast Cancer. Sci Rep 2018, 8. [CrossRef]
- Litvin, J.; Selim, A.H.; Montgomery, M.O.; Lehmann, K.; Rico, M.C.; Devlin, H.; Bednarik, D.P.; Safadi, P.F. Expression and Function of Periostin-Isoforms in Bone. J Cell Biochem 2004, 92, 1044–1061. [CrossRef]
- Morra, L.; Moch, H. Periostin Expression and Epithelial-Mesenchymal Transition in Cancer: A Review and an Update. Virchows Archiv 2011, 459, 465–475.
- Cui, D.; Huang, Z.; Liu, Y.; Ouyang, G. The Multifaceted Role of Periostin in Priming the Tumor Microenvironments for Tumor Progression. Cellular and Molecular Life Sciences 2017, 74, 4287–4291.
- Shibata, K.; Koibuchi, N.; Sanada, F.; Katsuragi, N.; Kanemoto, Y.; Tsunetoshi, Y.; Ikebe, S.; Yamamoto, K.; Morishita, R.; Shimazu, K.; et al. The Importance of Suppressing Pathological Periostin Splicing Variants with Exon 17 in Both Stroma and Cancer. Cells 2024, 13. [CrossRef]
- Ikeda-Iwabu, Y.; Taniyama, Y.; Katsuragi, N.; Sanada, F.; Koibuchi, N.; Shibata, K.; Shimazu, K.; Rakugi, H.; Morishita, R. Periostin Short Fragment with Exon 17 via Aberrant Alternative Splicing Is Required for Breast Cancer Growth and Metastasis. Cells 2021, 10. [CrossRef]
- Kanemoto, Y.; Sanada, F.; Shibata, K.; Tsunetoshi, Y.; Katsuragi, N.; Koibuchi, N.; Yoshinami, T.; Yamamoto, K.; Morishita, R.; Taniyama, Y.; et al. Expression of Periostin Alternative Splicing Variants in Normal Tissue and Breast Cancer. Biomolecules 2024, 14. [CrossRef]
- Balbi, C.; Milano, G.; Fertig, T.E.; Lazzarini, E.; Bolis, S.; Taniyama, Y.; Sanada, F.; Silvestre, D. Di; Mauri, P.; Gherghiceanu, M.; et al. An Exosomal-Carried Short Periostin Isoform Induces Cardiomyocyte Proliferation. Theranostics 2021, 11, 5634–5649. [CrossRef]
- Fujikawa, T.; Sanada, F.; Taniyama, Y.; Shibata, K.; Katsuragi, N.; Koibuchi, N.; Akazawa, K.; Kanemoto, Y.; Kuroyanagi, H.; Shimazu, K.; et al. Periostin Exon-21 Antibody Neutralization of Triple-Negative Breast Cancer Cell-Derived Periostin Regulates Tumor-Associated Macrophage Polarization and Angiogenesis. Cancers (Basel) 2021, 13. [CrossRef]
- Kalluri, R. The Biology and Function of Fibroblasts in Cancer. Nat Rev Cancer 2016, 16, 582–598.
- Dongre, A.; Weinberg, R.A. New Insights into the Mechanisms of Epithelial–Mesenchymal Transition and Implications for Cancer. Nat Rev Mol Cell Biol 2019, 20, 69–84.
- Pastushenko, I.; Blanpain, C. EMT Transition States during Tumor Progression and Metastasis. Trends Cell Biol 2019, 29, 212–226. [CrossRef]
- Lamouille, S.; Xu, J.; Derynck, R. Molecular Mechanisms of Epithelial-Mesenchymal Transition. Nat Rev Mol Cell Biol 2014, 15, 178–196.
- Nieto, M.A.; Huang, R.Y.Y.J.; Jackson, R.A.A.; Thiery, J.P.P. EMT: 2016. Cell 2016, 166, 21–45. [CrossRef]
- Balbi, C.; Milano, G.; Fertig, T.E.; Lazzarini, E.; Bolis, S.; Taniyama, Y.; Sanada, F.; Silvestre, D. Di; Mauri, P.; Gherghiceanu, M.; et al. An Exosomal-Carried Short Periostin Isoform Induces Cardiomyocyte Proliferation. Theranostics 2021, 11, 5634–5649. [CrossRef]
- Kyutoku, M.; Taniyama, Y.; Katsuragi, N.; Shimizu, H.; Kunugiza, Y.; Iekushi, K.; Koibuchi, N.; Sanada, F.; Oshita, Y.; Morishita, R. Role of Periostin in Cancer Progression and Metastasis: Inhibition of Breast Cancer Progression and Metastasis by Anti-Periostin Antibody in a Murine Model. Int J Mol Med 2011, 28, 181–186. [CrossRef]
- Tsunetoshi, Y.; Sanada, F.; Kanemoto, Y.; Shibata, K.; Masamune, A.; Taniyama, Y.; Yamamoto, K.; Morishita, R. A Role for Periostin Pathological Variants and Their Interaction with HSP70-1a in Promoting Pancreatic Cancer Progression and Chemoresistance. Int J Mol Sci 2024, 25. [CrossRef]
- Fischer, K.R.; Durrans, A.; Lee, S.; Sheng, J.; Li, F.; Wong, S.T.C.; Choi, H.; El Rayes, T.; Ryu, S.; Troeger, J.; et al. Epithelial-to-Mesenchymal Transition Is Not Required for Lung Metastasis but Contributes to Chemoresistance. Nature 2015, 527, 472–476. [CrossRef]
- Yoshida, T.; Ozawa, Y.; Kimura, T.; Sato, Y.; Kuznetsov, G.; Xu, S.; Uesugi, M.; Agoulnik, S.; Taylor, N.; Funahashi, Y.; et al. Eribulin Mesilate Suppresses Experimental Metastasis of Breast Cancer Cells by Reversing Phenotype from Epithelial-Mesenchymal Transition (EMT) to Mesenchymal-Epithelial Transition (MET) States. Br J Cancer 2014, 110, 1497–1505. [CrossRef]
- Taguchi, E.; Horiuchi, K.; Senoo, A.; Susa, M.; Inoue, M.; Ishizaka, T.; Rikitake, H.; Matsuhashi, Y.; Chiba, K. Eribulin Induces Tumor Vascular Remodeling through Intussusceptive Angiogenesis in a Sarcoma Xenograft Model. Biochem Biophys Res Commun 2021, 570, 89–95. [CrossRef]
- Hong, J.; Zhou, J.; Fu, J.; He, T.; Qin, J.; Wang, L.; Liao, L.; Xu, J. Phosphorylation of Serine 68 of Twist1 by MAPKs Stabilizes Twist1 Protein and Promotes Breast Cancer Cell Invasiveness. Cancer Res 2011, 71, 3980–3990. [CrossRef]
- Chhabra, Y.; Weeraratna, A.T. Fibroblasts in Cancer: Unity in Heterogeneity. Cell 2023, 186, 1580–1609. [CrossRef]
- Ouanouki, A.; Lamy, S.; Annabi, B. Oncotarget 22023 Www.Oncotarget.Com Periostin, a Signal Transduction Intermediate in TGF-β-Induced EMT in U-87MG Human Glioblastoma Cells, and Its Inhibition by Anthocyanidins; 2018; Vol. 9;.
- Chen, Y.; McAndrews, K.M.; Kalluri, R. Clinical and Therapeutic Relevance of Cancer-Associated Fibroblasts. Nat Rev Clin Oncol 2021, 18, 792–804.
- Kim, R.-K.; Kaushik, N.; Suh, Y.; Yoo, K.-C.; Cui, Y.-H.; Kim, M.-J.; Lee, H.-J.; Kim, I.-G.; Lee, S.-J. Radiation Driven Epithelial-Mesenchymal Transition Is Mediated by Notch Signaling in Breast Cancer; 2016; Vol. 7;.
- He, E.; Pan, F.; Li, G.; Li, J. Fractionated Ionizing Radiation Promotes Epithelial-Mesenchymal Transition in Human Esophageal Cancer Cells through PTEN Deficiency-Mediated Akt Activation. PLoS One 2015, 10. [CrossRef]
- Lin, Y.; Bai, X.; Zhou, W.; He, Y.; Wu, Y.; Wang, X. Radiation Exposure Triggers the Progression of Triple Negative Breast Cancer via Stabilizing ZEB1. Biomedicine & Pharmacotherapy 2018, 107, 1624–1630. [CrossRef]
- Mani, S.A.; Guo, W.; Liao, M.J.; Eaton, E.N.; Ayyanan, A.; Zhou, A.Y.; Brooks, M.; Reinhard, F.; Zhang, C.C.; Shipitsin, M.; et al. The Epithelial-Mesenchymal Transition Generates Cells with Properties of Stem Cells. Cell 2008, 133, 704–715. [CrossRef]
- Brabletz, S.; Schuhwerk, H.; Brabletz, T.; Stemmler, M.P. Dynamic EMT: A Multi-tool for Tumor Progression. EMBO J 2021, 40. [CrossRef]
- Thiery, J.P.; Acloque, H.; Huang, R.Y.J.; Nieto, M.A. Epithelial-Mesenchymal Transitions in Development and Disease. Cell 2009, 139, 871–890. [CrossRef]
- Kalluri, R. The Biology and Function of Fibroblasts in Cancer. Nat Rev Cancer 2016, 16, 582–598.
- Lamouille, S.; Xu, J.; Derynck, R. Molecular Mechanisms of Epithelial-Mesenchymal Transition. Nat Rev Mol Cell Biol 2014, 15, 178–196.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



