Submitted:
04 December 2025
Posted:
09 December 2025
You are already at the latest version
Abstract
Keywords:
Background
The Cheung Glutamatergic Regimen
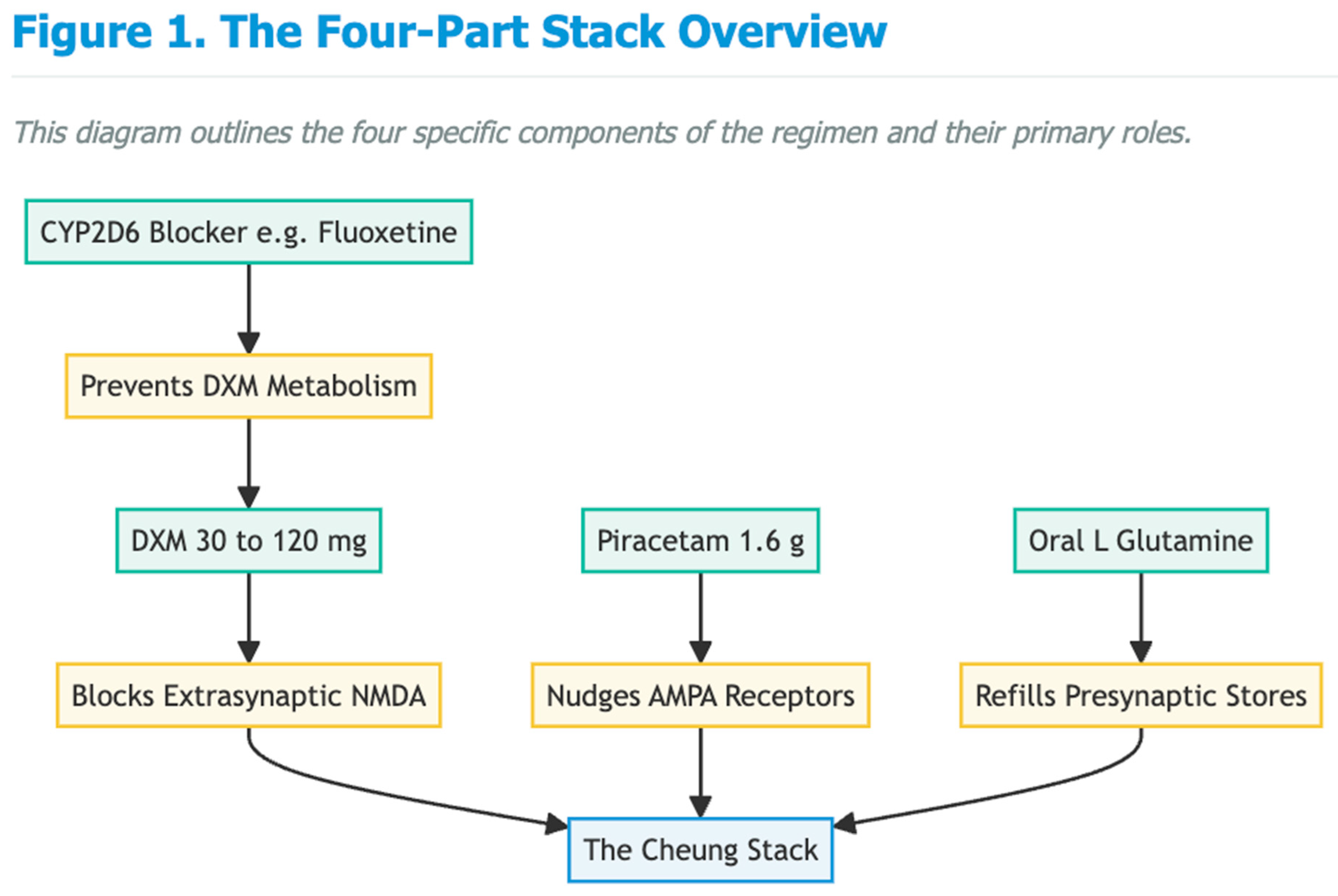
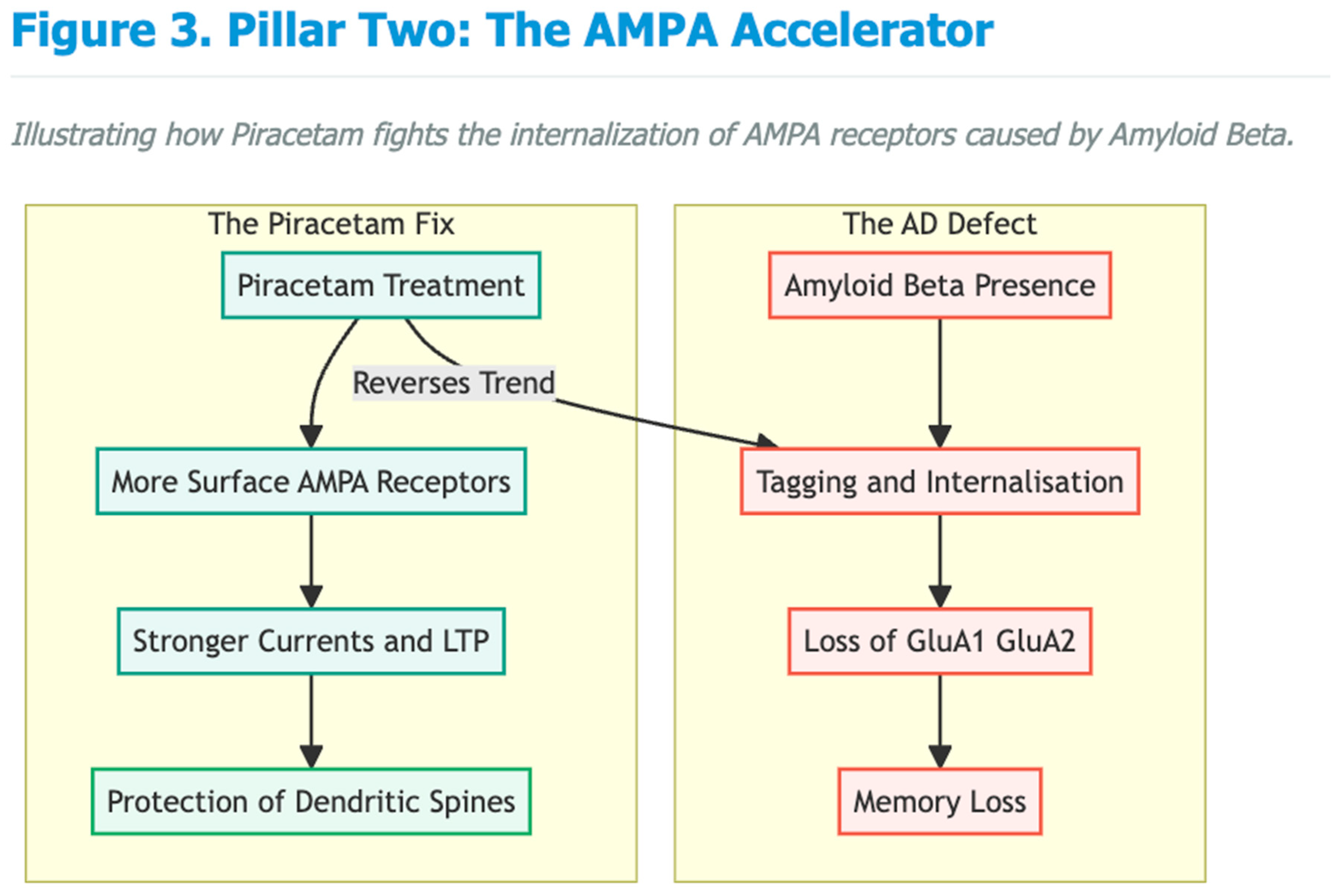
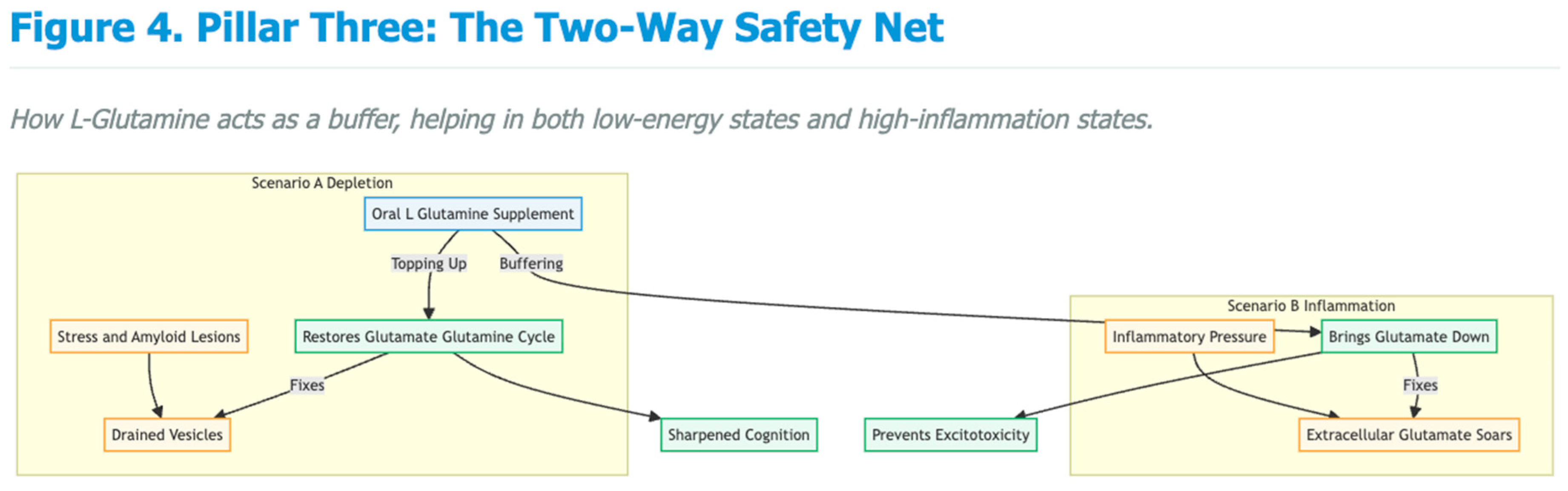
Mechanistic Alignment with Early AD Pathophysiology

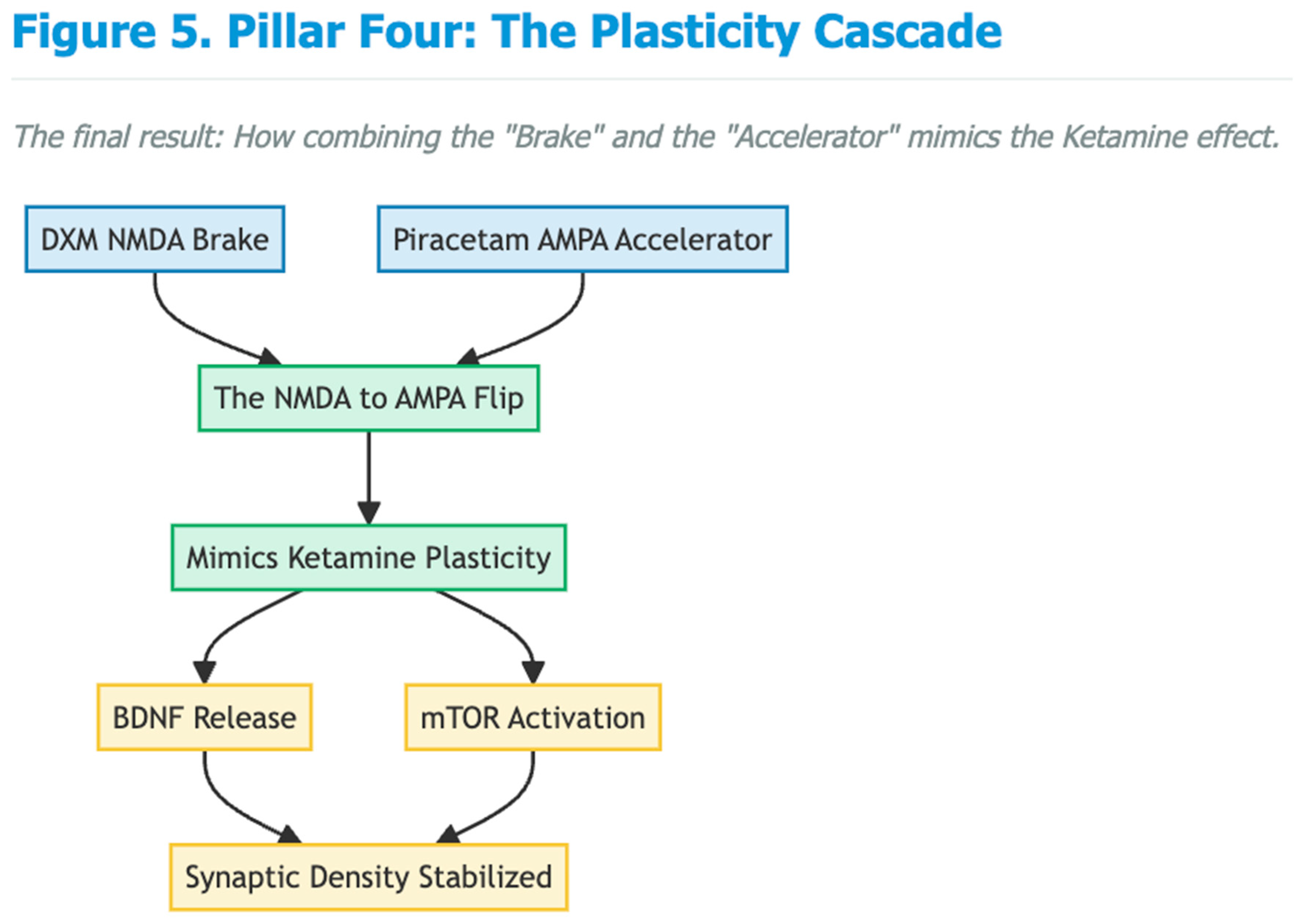
Emerging Clinical Signals Relevant to Cognition and Neuroprotection
Feasibility for Long-Term Prevention
Conclusions
Conflict of Interest and Source of Funding Statement
Funding Declaration
Ethics Declaration
References
- Mota, S.I.; Ferreira, I.L.; Rego, A.C. Dysfunctional synapse in Alzheimer's disease - A focus on NMDA receptors. Neuropharmacology 2014, 76 Pt A, 16–26. [Google Scholar] [CrossRef]
- Wang, R.; Reddy, P.H. Role of Glutamate and NMDA Receptors in Alzheimer's Disease. J Alzheimers Dis. 2017, 57, 1041–1048. [Google Scholar] [CrossRef]
- Chang, E.H.; Savage, M.J.; Flood, D.G.; et al. AMPA receptor downscaling at the onset of Alzheimer's disease pathology in double knockin mice. Proc Natl Acad Sci USA 2006, 103(9), 3410–3415. [Google Scholar] [CrossRef]
- Hsieh, H.; Boehm, J.; Sato, C.; et al. AMPAR removal underlies Abeta-induced synaptic depression and dendritic spine loss. Neuron 2006, 52(5), 831–843. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Guo, O.; Huo, Y.; et al. Amyloid-β Induces AMPA Receptor Ubiquitination and Degradation in Primary Neurons and Human Brains of Alzheimer's Disease. J Alzheimers Dis. 2018, 62(4), 1789–1801. [Google Scholar] [CrossRef] [PubMed]
- Lu, K.; Li, C.; Liu, J.; et al. Impairments in endogenous AMPA receptor dynamics correlates with learning deficits in Alzheimer's disease model mice. Proc Natl Acad Sci USA 2023, 120(40), e2303878120. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Sanz, C.; Balantzategi, U.; Quintela-López, T.; et al. Amyloid β / PKC-dependent alterations in NMDA receptor composition are detected in early stages of Alzheimer´s disease. Cell Death Dis. 2022, 13(3), 253. [Google Scholar] [CrossRef]
- Danysz, W.; Parsons, C.G. Alzheimer's disease, β-amyloid, glutamate, NMDA receptors and memantine--searching for the connections. Br J Pharmacol. 2012, 167, 324–352. [Google Scholar] [CrossRef]
- Cheung, N. Clinical experience and optimisation of the Cheung glutamatergic regimen for refractory psychiatric diseases. Preprints 2025. [Google Scholar] [CrossRef]
- Cheung, N. DXM, CYP2D6-inhibiting antidepressants, piracetam, and glutamine: Proposing a ketamine-class antidepressant regimen with existing drugs. Preprints 2025. [Google Scholar] [CrossRef]
- Cheung, N. Ultra-low-dose paroxetine as CYP2D6 inhibitor enables safe OTC glutamatergic augmentation in a depressive patient on lisdexamfetamine. Preprints 2025. [Google Scholar] [CrossRef]
- Li, N.; Lee, B.; Liu, R.J.; et al. mTOR-dependent synapse formation underlies the rapid antidepressant effects of NMDA antagonists. Science 2010, 329(5994), 959–964. [Google Scholar] [CrossRef]
- Maeng, S.; Zarate, C.A., Jr.; Du, J.; et al. Cellular mechanisms underlying the antidepressant effects of ketamine: role of alpha-amino-3-hydroxy-5-methylisoxazole-4-propionic acid receptors. Biol Psychiatry 2008, 63(4), 349–352. [Google Scholar] [CrossRef]
- Zanos, P.; Moaddel, R.; Morris, P.J.; et al. NMDAR inhibition-independent antidepressant actions of ketamine metabolites. Nature 2016, 533(7604), 481–486. [Google Scholar] [CrossRef]
- Kessels, H.W.; Nabavi, S.; Malinow, R. Metabotropic NMDA receptor function is required for β-amyloid-induced synaptic depression. Proc Natl Acad Sci USA 2013, 110, 4033–4038. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.A.; Müller, W.E. Effects of piracetam on N-methyl-D-aspartate receptor properties in the aged mouse brain. Pharmacology 1993, 47, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Winblad, B. Piracetam: a review of pharmacological properties and clinical uses. CNS Drug Rev. 2005, 11, 169–182. [Google Scholar] [CrossRef]
- Son, H.; Baek, J.H.; Go, B.S.; et al. Glutamine has antidepressive effects through increments of glutamate and glutamine levels and glutamatergic activity in the medial prefrontal cortex. Neuropharmacology 2018, 143, 143–152. [Google Scholar] [CrossRef]
- Baek, J.H.; Park, H.; Kang, H.; et al. The Role of Glutamine Homeostasis in Emotional and Cognitive Functions. Int J Mol Sci. 2024, 25(2), 1302. [Google Scholar] [CrossRef]
- Guerrero-Molina, M.P.; Morales-Conejo, M.; Delmiro, A.; et al. High-dose oral glutamine supplementation reduces elevated glutamate levels in cerebrospinal fluid in patients with mitochondrial encephalomyopathy, lactic acidosis and stroke-like episodes syndrome. Eur J Neurol. 2023, 30(2), 538–547. [Google Scholar] [CrossRef] [PubMed]
- Duman, R.S.; Aghajanian, G.K. Synaptic dysfunction in depression: potential therapeutic targets. Science 2012, 338, 68–72. [Google Scholar] [CrossRef]
- Cheung, N. Clearing the fog with the Cheung's regimen: A case of OTC glutamatergic augmentation in a student with depression, somatic symptoms and cognitive dysfunction. Preprints 2025. [Google Scholar] [CrossRef]
- Cheung, N. An oral "ketamine-like" NMDA/AMPA modulation stack restores cognitive capacity in a young man with schizoaffective disorder—Case report. Preprints 2025. [Google Scholar] [CrossRef]
- Cheung, N. Case series: Marked improvement in treatment-resistant obsessive–compulsive symptoms with over-the-counter glutamatergic augmentation in routine clinical practice. Preprints 2025. [Google Scholar] [CrossRef]
- Cheung, N. Oral glutamatergic augmentation for trauma-related disorders with fluoxetine-/bupropion-potentiated dextromethorphan ± piracetam: A four-patient case series. Preprints 2025. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




