1. Introduction
Acute myocardial infarction (AMI) occurring in adults younger than 45 years represents an increasingly recognized and complex cardiovascular entity with profound clinical, epidemiological, and societal implications [
1]. Traditionally perceived as a disease of middle-aged and elderly individuals, AMI is now emerging in younger cohorts at a pace that mirrors shifts in global health determinants. The rising incidence of premature AMI reflects an intricate interplay between biological predispositions, behavioral factors, and environmental influences, together with rapid changes in metabolic health and psychosocial stress exposure. This trend is particularly alarming because it affects individuals in their most productive decades, amplifying the long-term consequences of morbidity, disability-adjusted life years (DALYs) lost, and healthcare expenditure [
2,
3,
4].
From an epidemiological standpoint, early-onset AMI exhibits distinctive characteristics compared with its late-onset counterpart. While classical risk factors remain highly prevalent, younger adults frequently display a different risk profile, characterized by a predominance of smoking, dyslipidemia with pronounced atherogenic profiles, early vascular aging, obesity, insulin resistance, and metabolic syndrome. In addition, non-traditional and emerging risk determinants - including chronic low-grade inflammation, psychosocial distress, sleep disorders, recreational drug use (particularly cocaine and cannabis), and genetic variants affecting lipoprotein metabolism or thrombosis - play a proportionally greater role in precipitating premature coronary events [
5,
6].
These elements collectively contribute to a more heterogeneous pathogenic pathway, ranging from plaque rupture and erosion to coronary vasospasm and microvascular dysfunction, adding further complexity to diagnosis and long-term risk stratification. Although modern reperfusion strategies, improved emergency networks, and adherence to guideline-directed medical therapy have significantly reduced early mortality across age groups, young survivors of AMI face a unique trajectory of long-term vulnerability. Even in the presence of angiographically mild or single-vessel disease, they are at risk of recurrent ischemic episodes, arrhythmic events, adverse ventricular remodeling, and impaired cardiopulmonary fitness. Moreover, persistence of unhealthy behaviors, socioeconomic instability, and psychological distress - including post-traumatic stress, anxiety, depression, and fear of recurrent cardiac events - substantially influence prognosis and quality of life. The intersection between biological recovery and psychosocial adaptation is particularly relevant in this population, given their high expectations for rapid functional reintegration and return to work [
7,
8]. The complexity of this recovery trajectory has highlighted the limitations of traditional follow-up approaches, which rely on episodic, clinic-based assessments that only capture static snapshots of the patient’s physiological status. Advances in digital health technologies have therefore opened a transformative frontier in secondary prevention and long-term cardiovascular monitoring. Wearable devices - equipped with accelerometers, photoplethysmography (PPG), biosensors, and wireless connectivity - enable continuous, real-time acquisition of physiological, behavioral, and environmental data. These technologies support a transition from reactive, physician-driven care to proactive, patient-centered, data-rich models of disease management [
9,
10,
11].
In particular, the integration of wearable systems in the post-AMI setting offers unprecedented opportunities to quantify recovery dynamics with high temporal resolution. Continuous monitoring of heart rate and variability, motor activity, energy expenditure, oxygen saturation, sleep architecture, and circadian rhythms allows for granular assessment of autonomic function, physical conditioning, endothelial health, and behavioral adherence. Patterns of daily activity - such as step count variability, sedentary burden, and intensity of exertion - reflect important correlates of cardiovascular performance and are increasingly recognized as independent predictors of morbidity, mortality, and rehospitalization. Additionally, wearable-derived metrics allow early recognition of maladaptive recovery patterns, such as persistently elevated resting heart rate, reduced physical activity, or dysregulated sleep, which may signal ventricular dysfunction, poor fitness recovery, or psychological distress The potential added value of these technologies becomes even more relevant when combined with patient-reported outcome measures (PROMs). Instruments such as the International Physical Activity Questionnaire (IPAQ), the Depression Anxiety Stress Scale (DASS-21), and the Short Form Health Survey (SF-36) provide validated insight into subjective dimensions of recovery, including functional capacity, mood, emotional burden, fatigue, and perceived quality of life. The combination of continuous biosignal monitoring with PROMs creates a multidimensional framework capable of capturing both objective and experiential aspects of post-AMI health, aligning with emerging principles of precision cardiology and holistic cardiovascular rehabilitation. Nevertheless, despite their extensive availability and rapid adoption in the consumer market, the integration of wearable devices into routine cardiovascular care remains hindered by several knowledge gaps. Concerns persist regarding data accuracy, inter-device variability, sensor drift, and the validity of surrogate parameters for clinical decision-making. Furthermore, challenges related to interoperability, data governance, long-term adherence, digital literacy, and patient privacy raise important questions about feasibility and scalability. These issues are especially relevant in young AMI patients, who represent a heterogeneous and highly dynamic subgroup with diverse expectations, lifestyles, and technology use behaviors. Against this background, the present research provides a comprehensive, multidimensional evaluation of wearable device–enabled monitoring in young adults recovering from AMI. This thesis integrates: 1. objective biosignal analytics focused on motor activity and autonomic markers; 2. validated PROMs capturing psychological, behavioral, and functional recovery; 3. usability and acceptability analyses exploring the patient experience with digital monitoring; 4. methodological evaluation of data quality, adherence, and feasibility within real-world clinical pathways. Through this approach, the study seeks to elucidate the clinical relevance, predictive value, and translational potential of continuous remote monitoring in the early post-AMI phase.
Figure 1 summarizes the study design, outlining the recruitment process, group allocation, and multimodal data acquisition pipeline that forms the methodological backbone of the subsequent chapters. By integrating clinical cardiology, digital health science, behavioral medicine, and data analytics, this work aims to contribute to a more personalized, anticipatory, and technologically enabled model of cardiovascular care for young AMI survivors.
1.1. Risk Factors
Recent evidence confirms that the predominant pathophysiological drivers of acute myocardial infarction (AMI) in young adults remain coronary atherosclerotic plaque rupture and erosion, which collectively account for approximately 90 percent of cases in this population [
12]. Plaque rupture is typically associated with large lipid cores, thin fibrous caps, and heightened inflammatory activity, whereas plaque erosion, more frequently observed in younger patients - especially women - is characterized by endothelial denudation, superficial proteoglycan accumulation, and reduced inflammatory infiltrate. The remaining 10 percent of AMI presentations arise from non-atherosclerotic mechanisms, including spontaneous coronary artery dissection (SCAD), coronary vasospasm, microvascular dysfunction, hypercoagulable states, coronary embolism, myocarditis, and autoimmune-mediated vascular inflammation. These alternative etiologies underscore the heterogeneity of AMI pathogenesis in young adults and highlight the need for tailored diagnostic and therapeutic pathways. Both modifiable and non-modifiable factors intricately modulate susceptibility to these pathogenic processes. Non-modifiable risk factors comprise age, sex, ethnicity, menopausal status, and family history of cardiovascular disease. Young males remain disproportionately affected, a pattern attributed to sex hormone–mediated differences in endothelial function, plaque composition, and thrombogenicity. Genetic predisposition plays a central role: monogenic conditions such as familial hypercholesterolemia, homocystinuria, antiphospholipid antibody syndrome, Marfan syndrome, Ehlers-Danlos syndrome, and fibromuscular dysplasia significantly elevate risk through mechanisms involving dyslipidemia, hypercoagulability, or structural arterial fragility [
13] Beyond monogenic disorders, genome-wide association studies have revealed a continuum of risk determined by the cumulative burden of common genetic variants. Polygenic risk scores (PRS), derived from hundreds to thousands of loci associated with lipid metabolism, inflammation, and vascular biology, have emerged as strong predictors of early-onset AMI, often outperforming traditional risk calculators in young individuals with otherwise unremarkable clinical profiles [
14].
Recent attention has also shifted toward non-traditional risk contributors, which exert profound vascular effects through chronic inflammatory activation, immune dysregulation, oxidative stress, and endothelial dysfunction. Conditions such as HIV infection, systemic lupus erythematosus, rheumatoid arthritis, psoriasis, chronic kidney disease, and obstructive sleep apnea significantly accelerate atherogenesis, promote plaque vulnerability, and impair coronary microvascular function [
15]. Inflammatory cytokines—including IL-6, TNF, and interferons—drive endothelial activation, promote leukocyte adhesion, and mediate prothrombotic states, while intermittent nocturnal hypoxia in obstructive sleep apnea induces sympathetic overactivity, oxidative stress, and cardiometabolic dysregulation. These emerging determinants expand the conventional understanding of cardiovascular risk in younger populations, emphasizing the need for integrated assessment models that incorporate immune, metabolic, and neurohormonal factors. Modifiable risk factors remain highly prevalent among young AMI patients and represent critical intervention targets. Tobacco use stands as the most dominant risk factor, often present in up to 70–90 percent of young AMI cohorts. Smoking induces endothelial dysfunction, increases platelet reactivity, accelerates atherosclerotic plaque formation, and potentiates coronary vasospasm. Excessive alcohol consumption exerts dose-dependent cardiovascular effects, contributing to hypertension, arrhythmias, cardiomyopathy, and pro-inflammatory lipid profiles. Metabolic disorders—including obesity, hypertension, insulin resistance, and overt diabetes—are increasingly common among younger adults, driven by lifestyle changes, nutritional habits, and rising prevalence of sedentary behavior. Diabetes and metabolic syndrome exhibit particularly aggressive coronary phenotypes in young individuals, characterized by diffuse non-calcified plaque, enhanced inflammatory activity, impaired vascular reactivity, and early endothelial senescence. Sedentary behavior, now recognized as an independent cardiovascular risk factor, further contributes to AMI susceptibility by promoting visceral adiposity, dysglycemia, systemic inflammation, and autonomic imbalance. Physical inactivity not only predisposes individuals to a first ischemic event but also markedly amplifies the risk of recurrent events and long-term mortality among AMI survivors [
16].
Modifiable risk factors—tobacco use, excessive alcohol consumption, obesity, hypertension, diabetes, and physical inactivity—further shape AMI incidence and progression. Sedentary behavior is particularly detrimental, not only predisposing to first events but also amplifying cardiovascular risk in post-AMI survivors [
17]. Conversely, robust evidence supports the protective effect of regular physical activity through improvements in endothelial function, nitric oxide bioavailability, lipid metabolism, autonomic regulation, and plaque stability [
18]. Exercise also exerts beneficial effects on psychosocial health, reducing depressive symptoms, stress, and sleep disturbances—factors that themselves modulate cardiovascular risk. Nevertheless, despite growing recognition of its prognostic relevance, long-term outcome data specifically examining the impact of structured physical activity interventions in young AMI survivors remain limited, highlighting a critical need for targeted research within this demographic. Collectively, these risk factors illustrate the multifactorial and often overlapping pathways contributing to premature AMI. Their complex interactions underscore the importance of comprehensive, individualized risk assessment strategies that integrate genetic, metabolic, behavioral, inflammatory, and psychosocial dimensions. Understanding this intricate risk architecture is fundamental not only for elucidating disease mechanisms but also for optimizing prevention, early detection, and long-term management in young adults with AMI.
1.2. Wearable Devices
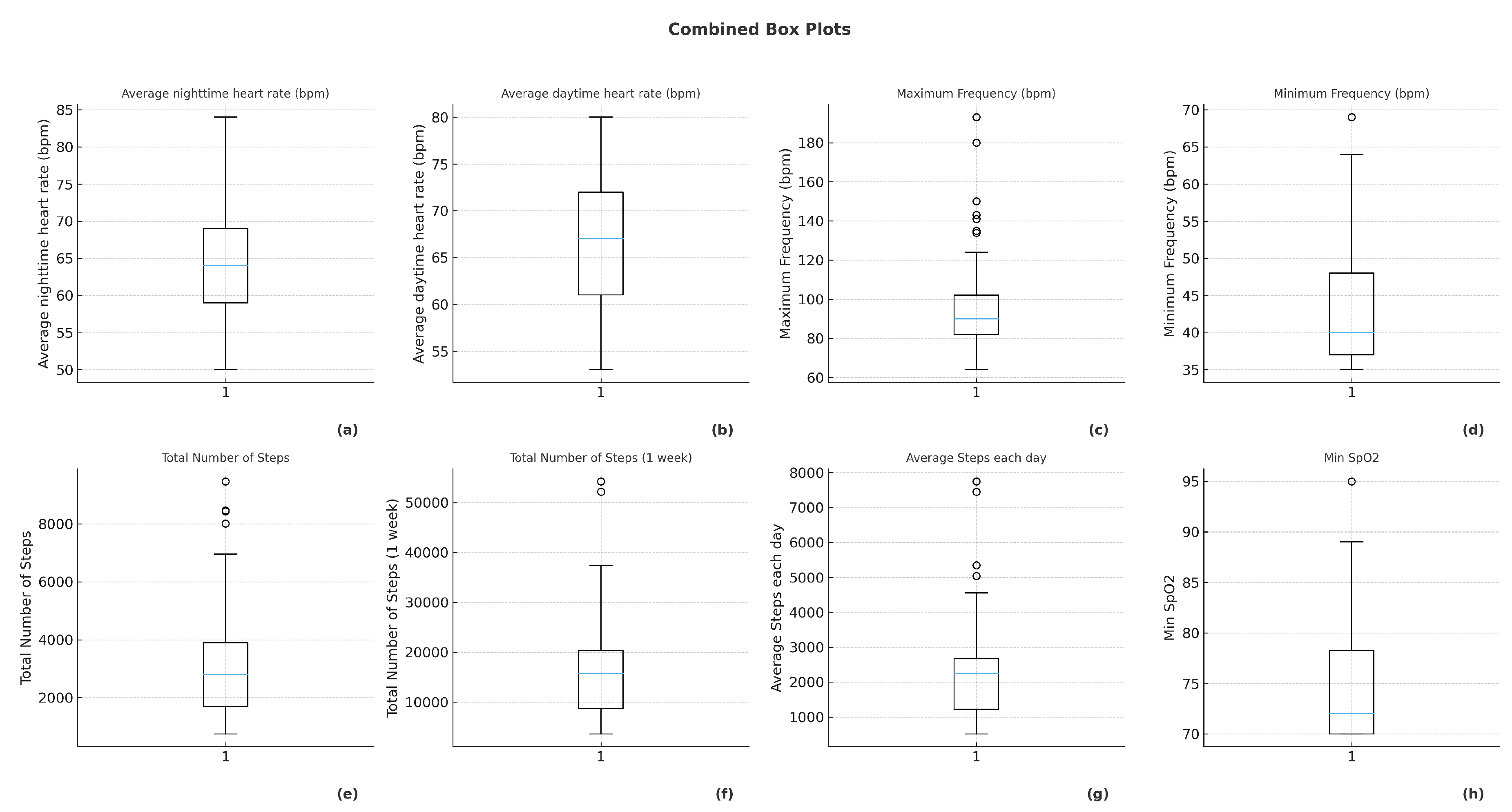
The contemporary landscape of cardiovascular medicine increasingly relies on wearable medical devices equipped with sophisticated multisensor architectures capable of capturing continuous, high-resolution physiological and behavioral data. These systems represent a crucial technological interface between patients and healthcare providers, enabling remote surveillance, early risk detection, and longitudinal monitoring in both clinical and real-world environments. Modern wearables integrate different sensing modalities, including photoplethysmography (PPG), single- and multi-lead electrocardiography (ECG), bioimpedance analysis, accelerometry, gyroscopy, and reflectance oximetry, thereby supporting the acquisition of parameters such as heart rate, cardiac rhythm, heart rate variability (HRV), oxygen saturation (SpO2), respiratory rate, blood pressure (through pulse wave analysis), sleep stages, and step-based physical activity patterns [
19]. These sensor modalities exploit advanced signal processing pipelines. PPG signals, for example, extract cardiac pulsatility via green- or infrared-based optical wavelengths, while embedded computational filters mitigate motion artefacts through adaptive noise cancellation and frequency-domain correction. ECG modules capture depolarization morphology, allowing detection of arrhythmias, premature ventricular complexes, and ischemia-related alterations when configured for multi-lead acquisition. Accelerometers and gyroscopes provide multidimensional kinematic data that enable quantification of gait dynamics, sedentary time, energy expenditure, circadian rhythmicity, and physiologic tremor patterns. Increasingly, wearable devices incorporate environmental sensors (temperature, barometric pressure) to contextualize physiological responses within external conditions. When integrated into secure telemedicine platforms, these wearables support encrypted, bidirectional, and continuous data transmission through Wi-Fi, LTE, and Bluetooth Low Energy channels. Clinicians and algorithmic systems can access dashboard visualizations that display temporal trends, threshold alerts, and multi-sensor summaries, enabling proactive intervention and remote triage.
The SiDLY Care Pro wristband represents a clinically certified Class IIa medical-grade device developed according to the Medical Devices Regulation (EU 2017/745) and ISO 13485 standards. Its design prioritizes accuracy, reliability, and telecare functionality for high-risk cardiovascular populations. The device continuously records: 1. Heart rate and rhythm via dual-channel PPG and optional ECG electrodes 2. Peripheral oxygen saturation (SpO2) using reflectance oximetry validated against hospital-grade co-oximeters 3. Blood pressure (in selected configurations) utilizing pulse wave analysis and individualized calibration algorithms 4. Autonomic surrogates, including HRV-based stress indices 5. Motor activity and posture, assessed using tri-axial accelerometry and gyroscopy
From a safety standpoint, the SiDLY Care Pro integrates real-time fall detection algorithms, which combine free-fall vector recognition with impact kinetics to discriminate between true and false alarms. Immediate SOS alerting is enabled through a dedicated panic button that establishes automatic voice or message contact with caregivers, emergency medical services, or a 24/7 telemonitoring call center. GPS tracking and customizable geo-fencing functions support location monitoring, sending automatic notifications when users exit predefined safe zones—an essential feature for vulnerable or cognitively impaired individuals. Direct two-way voice communication transforms the wristband into an active telecare node, allowing rapid clinician–patient interaction even in areas with fluctuating cellular signal coverage. This functionality ensures real-time assessment during symptomatic episodes and facilitates guided first-aid support when emergency services are required.
All measurements and alerts are processed within a secure, GDPR-compliant cloud ecosystem. Data undergo segmentation into timestamped epochs and pre-processing steps including:
1. artefact suppression via wavelet decomposition and adaptive filtering 2. feature extraction (e.g., RR intervals, PPG pulse amplitude variability, activity bout segmentation) 3. dimensionality reduction using principal component analysis (PCA)
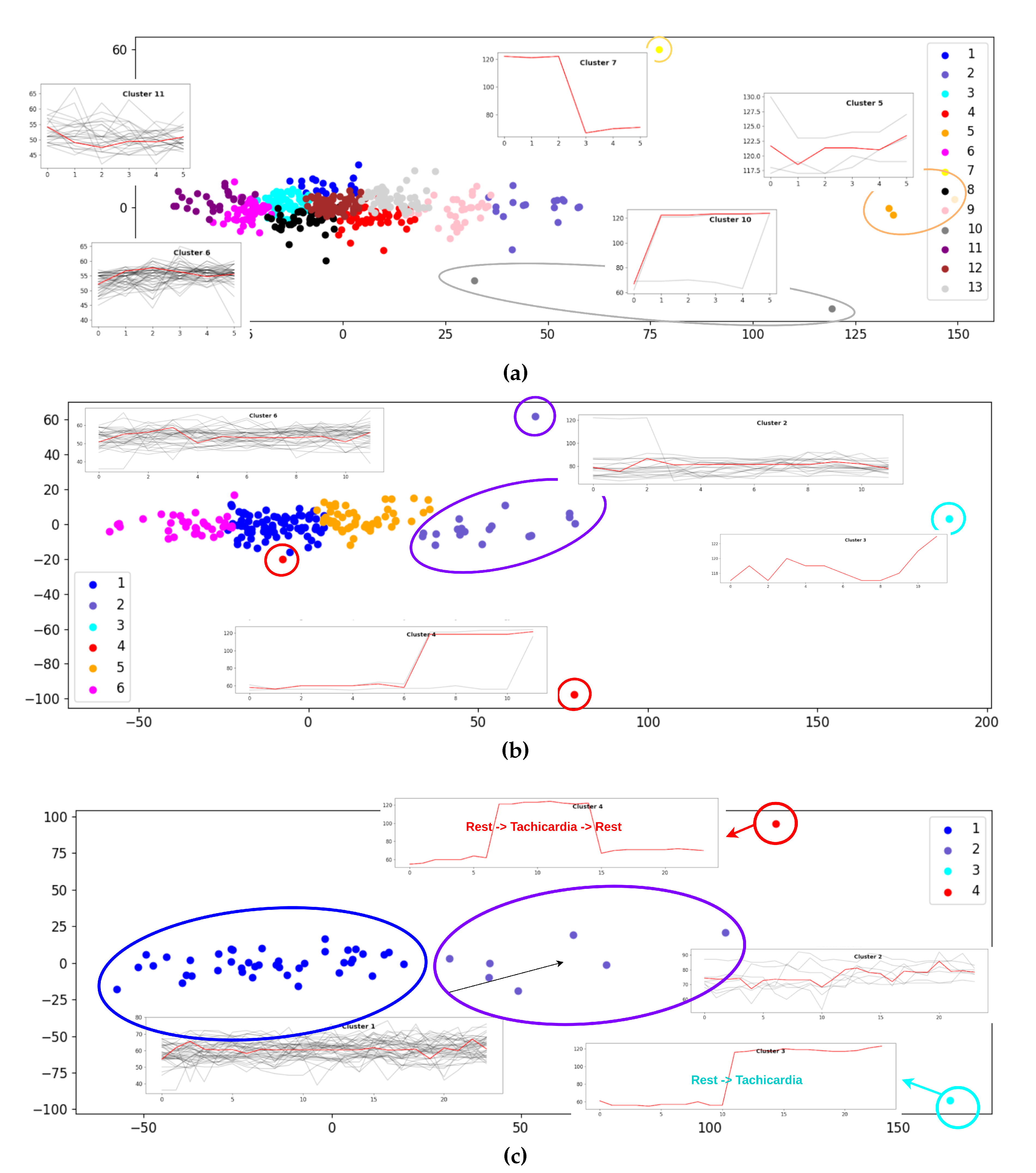
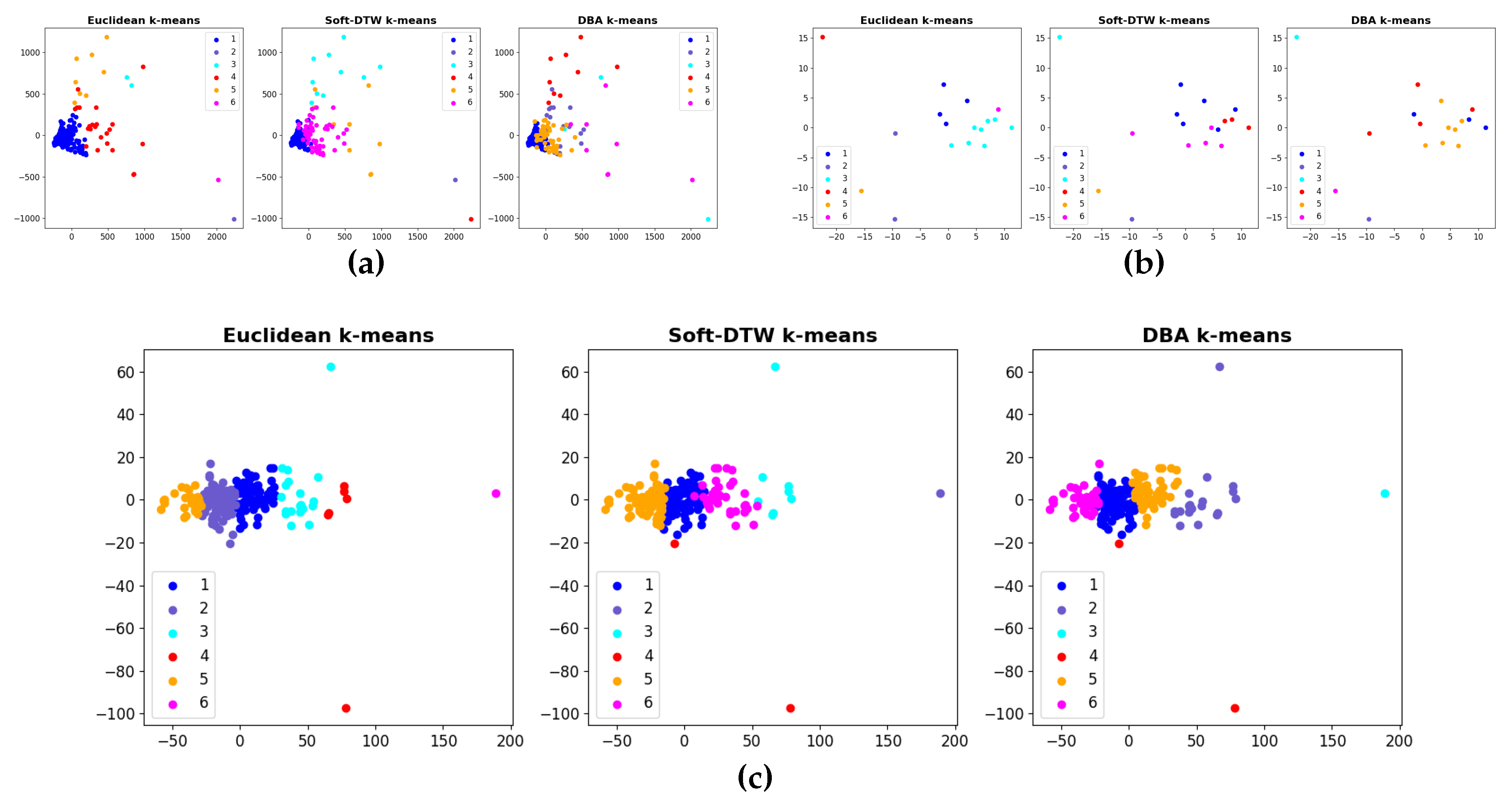
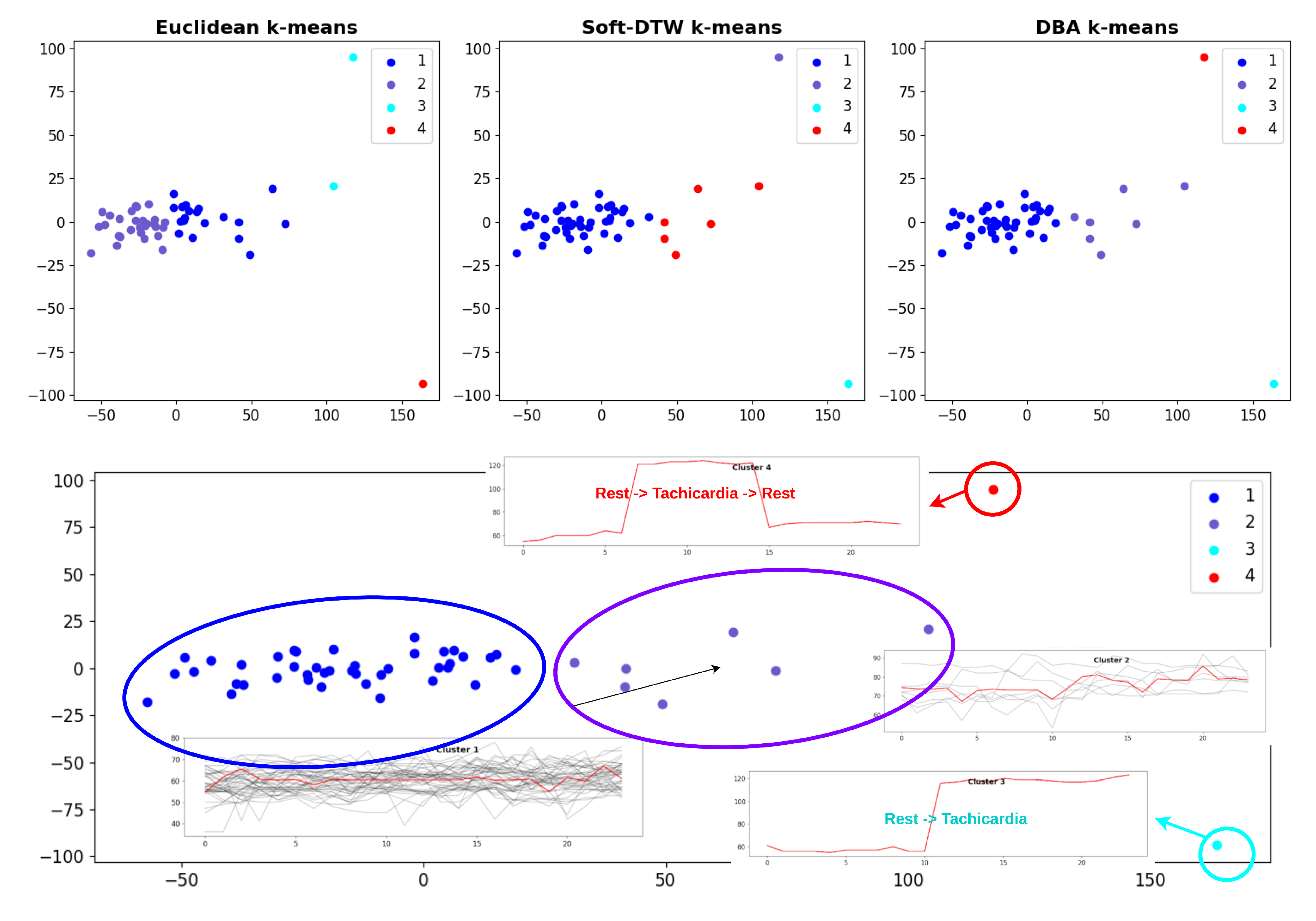
4. pattern recognition through unsupervised clustering (k-means, DBSCAN) to identify phenotypic trajectories
For cardiac time-series, dynamic time warping (DTW) is applied to compare temporally misaligned rhythm patterns and detect deviations from the individual baseline. The analytics layer includes anomaly detection algorithms that signal sudden changes in heart rate, oxygen saturation, or motion consistency, which may indicate arrhythmic events, heart failure decompensation, respiratory compromise, or post-AMI autonomic instability. Clinical dashboards present multi-day trend maps, cumulative activity metrics, nocturnal physiology comparisons, outlier detection, and patient-specific alerts. These visualizations support both retrospective review (e.g., identifying recurrent symptom-associated patterns) and prospective monitoring (e.g., early identification of worsening health status). The platform’s infrastructure enables role-based access control, granting differentiated permissions to clinicians, caregivers, researchers, and patients. Through mobile and web applications, real-time monitoring, historical trends, and emergency alerts become accessible in a user-centered interface optimized for varying levels of digital literacy.
A key innovation lies in the integration of proprietary artificial intelligence algorithms designed to predict imminent health deterioration. These models synthesize multisensor data - including PPG morphology, HRV indices, circadian activity patterns, gait variability, and SpO2 trends - to identify high-risk transitions associated with arrhythmic events, ischemic symptoms, anxiety-related hyperarousal, and post-AMI autonomic dysregulation. Predictive alerts allow for early therapeutic adjustments and may reduce preventable rehospitalizations.
The multilevel data architecture created by wearable devices such as the SiDLY Care Pro provides a scalable environment for:
1. precision cardiovascular phenotyping 2. remote rehabilitation monitoring 3. real-world epidemiological data collection 4. refinement of predictive algorithms through continuous learning 5. rapid clinical decision-making supported by algorithmic triage
Ultimately, wearable-enabled telemonitoring enhances continuity of care, supports post-AMI recovery trajectories, and contributes to a future in which real-time, personalized cardiovascular management becomes a central pillar of secondary prevention strategies [
20].
1.3. Statement of Significance
| Section |
Summary |
| Problem or Issue |
Early myocardial infarction (MI) in adults under 50 is rising, yet post-discharge monitoring remains limited. Traditional follow-up often fails to capture day-to-day physiological fluctuations and physical inactivity, both of which are critical predictors of recovery and recurrent events. |
| What is Already Known |
Wearable devices can monitor heart rate, oxygen saturation, and activity levels, and telemedicine improves follow-up and adherence. However, evidence specific to young MI survivors—regarding real-world usability, clinical relevance, and patient acceptance—is scarce, and existing studies rarely integrate objective biosignals with patient-reported outcomes. |
| What this Paper Adds |
This study provides a multidimensional, real-world evaluation of a certified medical wearable for early MI survivors. It combines time-series analytics, clinical metrics, and usability data, showing that continuous monitoring is feasible, clinically meaningful, and strongly accepted by patients when clinician feedback is integrated. |
| Who Would Benefit |
Clinicians, digital-health researchers, policymakers, and rehabilitation specialists seeking evidence for implementing remote monitoring in young post-MI populations. |