1. Introduction
All aerobic organisms, including the human body, are harmed when exposed to higher than normal concentrations of oxygen. The peroxyl radical (ROO•) is a reactive oxygen species formed because of oxidative damage to lipids, DNA, and carbohydrates. Oxidative damage is continuous and, over time, may be linked to cellular disorders and the development of diseases [
1]. Erythrocytes are particularly exposed to oxidative stress as they transport oxygen [
2]. Therefore, antioxidants are required to prevent this oxidative stress, in this case could be C-phycocyanin (C-PC).
C-PC is a phycobiliprotein with a photosynthetic pigment function, present in various species of algae, cyanobacteria, and some cryptomonads. Based on their maximum absorbance, these light harvesting pigments are classified as allophycocyanin, phycocyanin, and phycoerythrin. This protein is a hexamer of monomers with two subunits containing chromophores: alpha (1 chromophore) and beta (2 chromophores), totaling 18 phycocyanobilin chromophores. It possesses functional properties in the body such as antiinflammatory and anticancer effects and has a high antioxidant capacity. It also acts directly on oxidative stress, apoptosis, and inflammatory processes. Currently, C-PC is obtained from
Spirulina sp. It is recognized as a relevant natural blue colorant applied in the food industry, which can also be incorporated as an ingredient due to its multiple health benefits [
3,
4].
Damage to the erythrocyte membrane caused by the formation of peroxyl radicals can denationalize the cell, causing long-term disorders. For this reason, antioxidants such as phycocyanin are proposed to reduce or inhibit damage to membranes caused by free radicals. Damage to the erythrocyte membrane caused by the formation of peroxyl radicals can denature the cell, leading to long-term disorders. For this reason, antioxidants such as C-PC are proposed to reduce or inhibit membrane damage caused by free radicals. The antioxidant capacity and antihemolytic compatibility of phycocyanin could vary according to blood group. The ABO blood group antigens play a relevant role in susceptibility to infections and the severity of various diseases, being related to a wide spectrum of pathologies, including metabolic diseases, autoimmune diseases, different types of cancer (such as ovarian, gastric and prostate cancer), as well as neuropsychiatric and rheumatological disorders, in addition to infectious conditions (
Plasmodium falciparum, Escherichia coli, Helicobacter pylori, parvovirus B19, hepatitis B virus, chikungunya virus and others) [
5,
6,
7,
8,
9].
Therefore, the objective of this research was to quantify the antioxidant activity of C-PC in erythrocytes of different blood groups and Rh (+/-) factors and in an in vitro digestion model. Currently, there are no studies comparing the erythroprotective capacity of C-PC from Spirulina sp. before and after in vitro digestion to observe its bioavailability.
2. Results
2.1. Quantification of the Antioxidant Capacity of Phycocyanin Before and After Digestion In Vitro
Antioxidant Capacity
The antioxidant activity provided by C-PC before and after
in vitro digestion is summarized in
Table 1. The ABTS+•, DPPH•, and FRAP assays allow the antioxidant capacity to be estimated through different oxidation-reduction reactions. The antioxidant capacity of the bioaccessible fraction increased compared to baseline in all assays. However, a slight decrease in DPPH and FRAP was observed in the bioavailable fraction, while ABTS increased. These techniques are among the most frequently used methods for analyzing the antioxidant activity of foods, nutrition, and supplements. It is advisable to perform at least two antioxidant activity tests, as their physicochemical characteristics and the antioxidant mechanisms involved (hydrogen atom transfer--HAT or single electron transfer--SET) make it possible to identify variations that would otherwise go unnoticed [
10,
11,
12,
13]. In this case, FRAP only uses the SET mechanism, while DPPH and ABTS can use both depending on their reaction system. Based on the results obtained, the samples appear to have low electron transfer capacity, meaning they likely rely more on a HAT mechanism than a SET mechanism.
2.2. Cytotoxic and Erythroprotective Effects Before and After In Vitro Digestion
2.2.1. Cytotoxicity of C-PC from the Cyanobacterium Spirulina sp. at Different Concentrations Before In Vitro Digestion
In the cytotoxicity or blood biocompatibility test of C-PC from the cyanobacterium
Spirulina sp., different concentrations were applied to different blood groups to quantify its cytotoxicity (
Table 2). According to the results, C-PC is not cytotoxic in most blood groups. However, no cytotoxicity was observed in blood groups O- and B+ at the maximum concentration (300 µg/mL). In the remaining blood groups, a slight increase was observed starting at 125 µg/mL, with AB+, AB-, A+, and O+ being the most susceptible. Generally, Rh-negative blood groups are less susceptible to cytotoxicity. However, compared with AAPH, that was used as positive control, the cytotoxicity of P-CP was almost nil.
2.2.2. Erythroprotection of C-Phycocyanin from the Cyanobacterium Spirulina sp. at Different Concentrations Prior to In Vitro Digestion
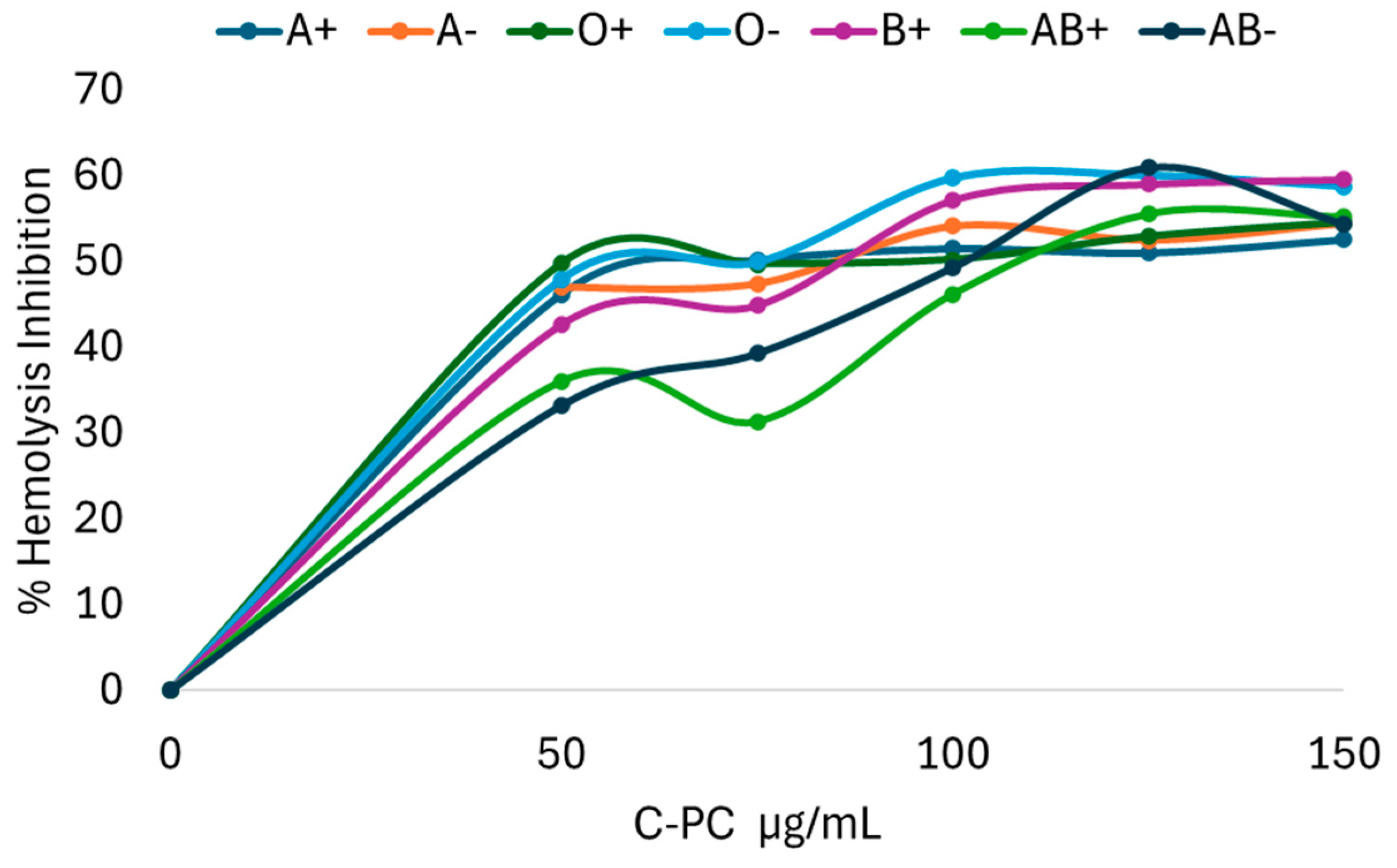
This was performed by inhibiting hemolysis of different concentrations of C-PC against peroxyl radicals caused by the azo compound AAPH. Different responses were obtained in different blood groups and Rh factors. C-PC is capable of inhibiting the formation of peroxyl radicals in a differentiated manner in blood groups. Blood type O- requires a lower concentration than the other groups for radical inhibition. In contrast, type AB- requires a higher concentration of C-PC to achieve the same result, as can be seen in
Figure 1.
Since C-PC acts differently in each blood group and Rh factor, the inhibitory concentration 50 (IC50) was estimated using equations obtained from the graphs, yielding the results in
Table 3, where it was found that O- requires the lowest concentration of C-PC to inhibit 50% of hemolysis against the azo compound AAPH, followed by type AB+, which has a different relationship with the azo compound compared to AB-, which requires the highest concentration of C-PC to achieve the same result.
2.2.3. Cytotoxic Effect of IC50 of C-PC
The analysis provides information on the biocompatibility of C-phycocyanin from the cyanobacterium
Spirulina sp. solubilized in physiological solution and applied to erythrocytes of different blood groups and Rh factors (
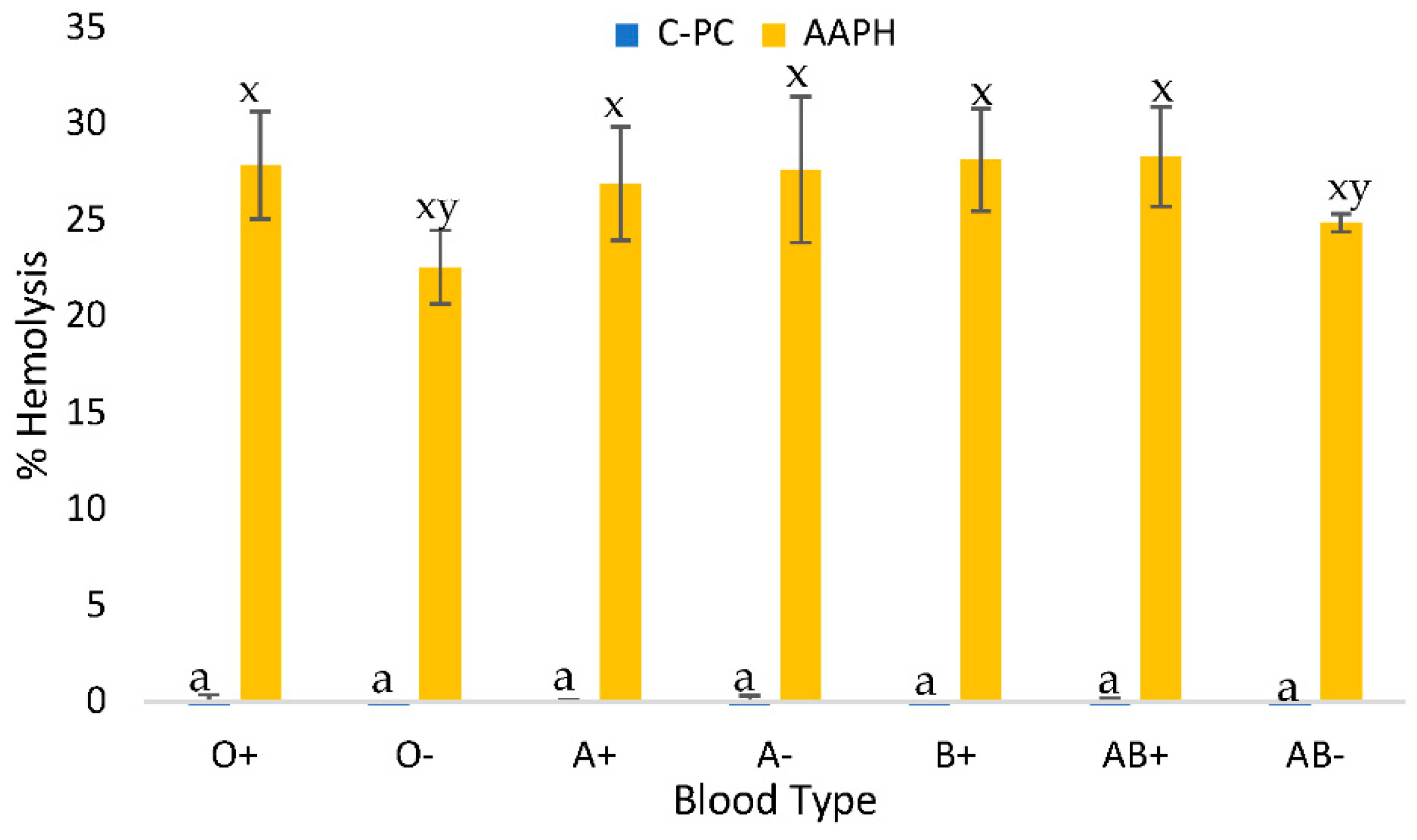
Figure 2). This study was conducted to verify that C-PC at specific IC50 levels did not cause damage to erythrocytes. The results showed that it did not cause damage at these concentrations, compared to AAPH administered at the same IC50 levels.
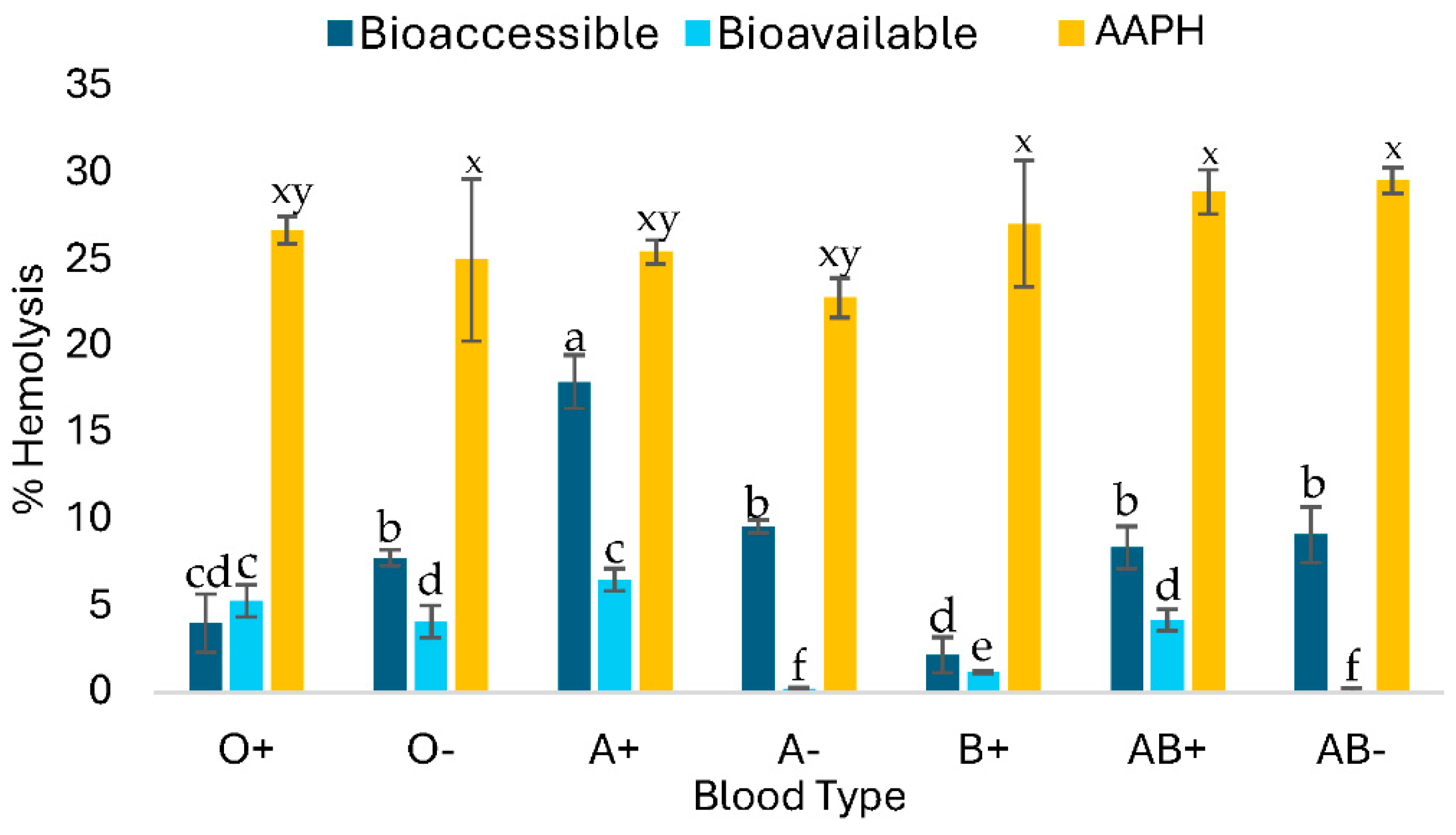
2.2.4. Cytotoxic Effect of C-PC After In Vitro Digestion
The blood biocompatibility test shows cytotoxicity results of the compound on human erythrocytes. In this case, an
in vitro digestion of 1 mg/mL of C-PC was performed on the different ABO blood groups (
Figure 3). Group O- showed more hemolysis in the bioaccessible fraction, followed by AB+ and AB-. Approximately 30% of C-PC is bioavailable and, instead of causing toxicity, it will help protect erythrocytes from damage, principally A- and AB- groups.
2.3. Erythroprotection Assays Before and After In Vitro Digestion
2.3.1. Erythroprotection of C-PC Using its IC50 Before in Vitro Digestion
The IC50 is the concentration of a compound necessary to inhibit 50% of biological activity. In this case, the determination of IC50 in erythrocytes of different blood groups are observed in
Table 4. Blood groups A- and O+ require the least amount of C-PC to inhibit 50% of hemolysis, while groups AB+ and AB- require larger quantities.
2.3.2. Erythroprotective Effect of C-Phycocyanin After In Vitro Digestion
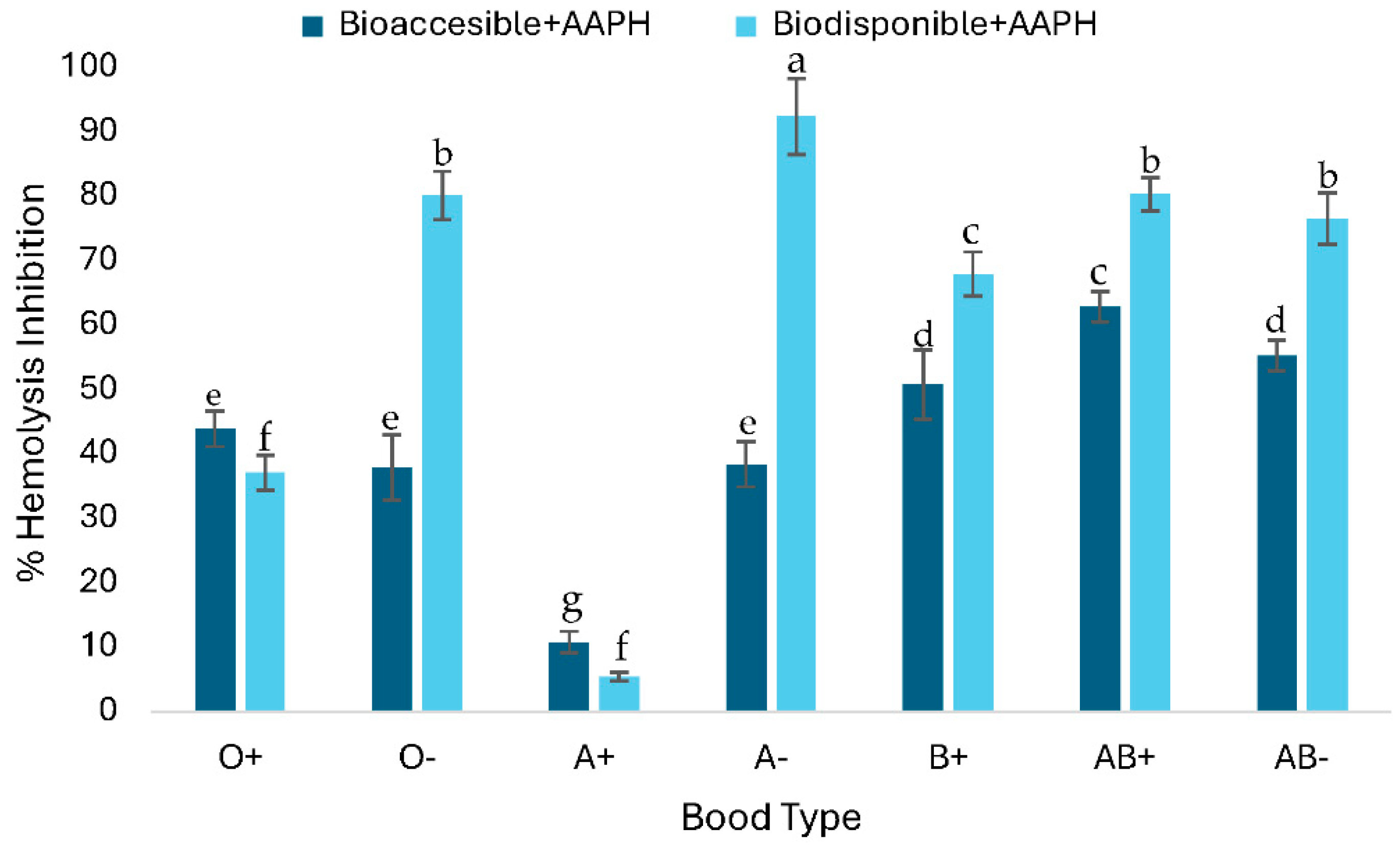
C-PC at a concentration of 1 mg/mL, evaluated using the
in vitro post digestion erythroprotective assay, was able to inhibit peroxyl radicals generated by the azocompound AAPH (40 mM) differentially among the different ABO blood groups of human erythrocytes. As shown in
Figure 4, significant differences were observed between the bioavailable and bioaccessible fractions in all blood groups. In Rh positive groups, the differences in the percentage of hemolysis inhibition between the two fractions were smaller compared to Rh negative groups. In general, the bioavailable fraction showed a higher percentage of hemolysis inhibition in most blood groups, except for types O and A+, in which the bioaccessible fraction showed greater inhibitory capacity. The values obtained were variable, with inhibition percentages ranging from 5% to 90% depending on the blood group and the type of fraction. These variations could be explained by the differential affinity of the compounds towards the components of the erythrocyte membrane, particularly by the variability in the terminal sugars characteristic of each blood type.
2.4. Damage to Erythrocytes Observed Using Immersion Optical Microscopy
Images were taken using an optical microscope of erythrocytes added with samples of Triton x 1%, AAPH, and C-PC before and after in vitro digestion to observe the morphological damage caused to the erythrocytes.
2.4.1. Microscope Images of Blood Type O+
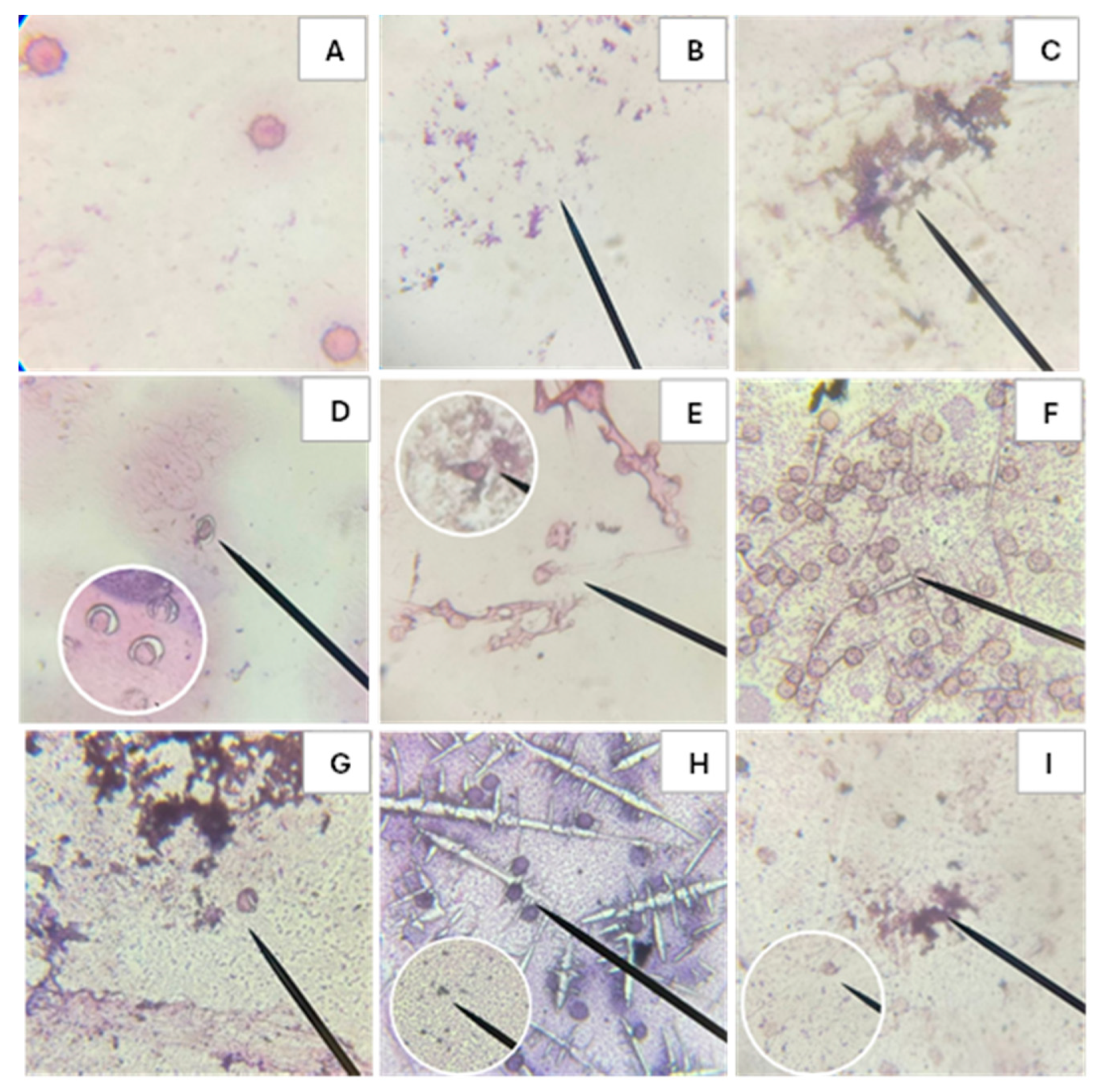
Microscopic observation provides visual evidence of the morphological changes occurring in erythrocytes of blood type O+ after exposure to the different treatments shown in
Figure 5. Image A depicts healthy erythrocytes with intact cell membranes, whereas Image B illustrates complete hemolysis produced by Triton, used as the positive control of hemolysis. AAPH induces both hemolysis and oxidative damage to red blood cells, as observed in Image C. In contrast, C-PC does not exhibit cytotoxic effects (Image D). A comparison between Images C and E shows that C-PC confers protective activity against AAPH-induced damage, as indicated by the presence of erythrocytes retaining their normal morphology. Furthermore, the
in vitro digestion products demonstrate that both the bioavailable and bioaccessible fractions do not induce morphological alterations in erythrocytes (Images F and H) and are capable of mitigating AAPH-induced damage, as evidenced by the presence of healthy cells (Images G and I).
2.4.2. Microscope Images of Blood Type O-
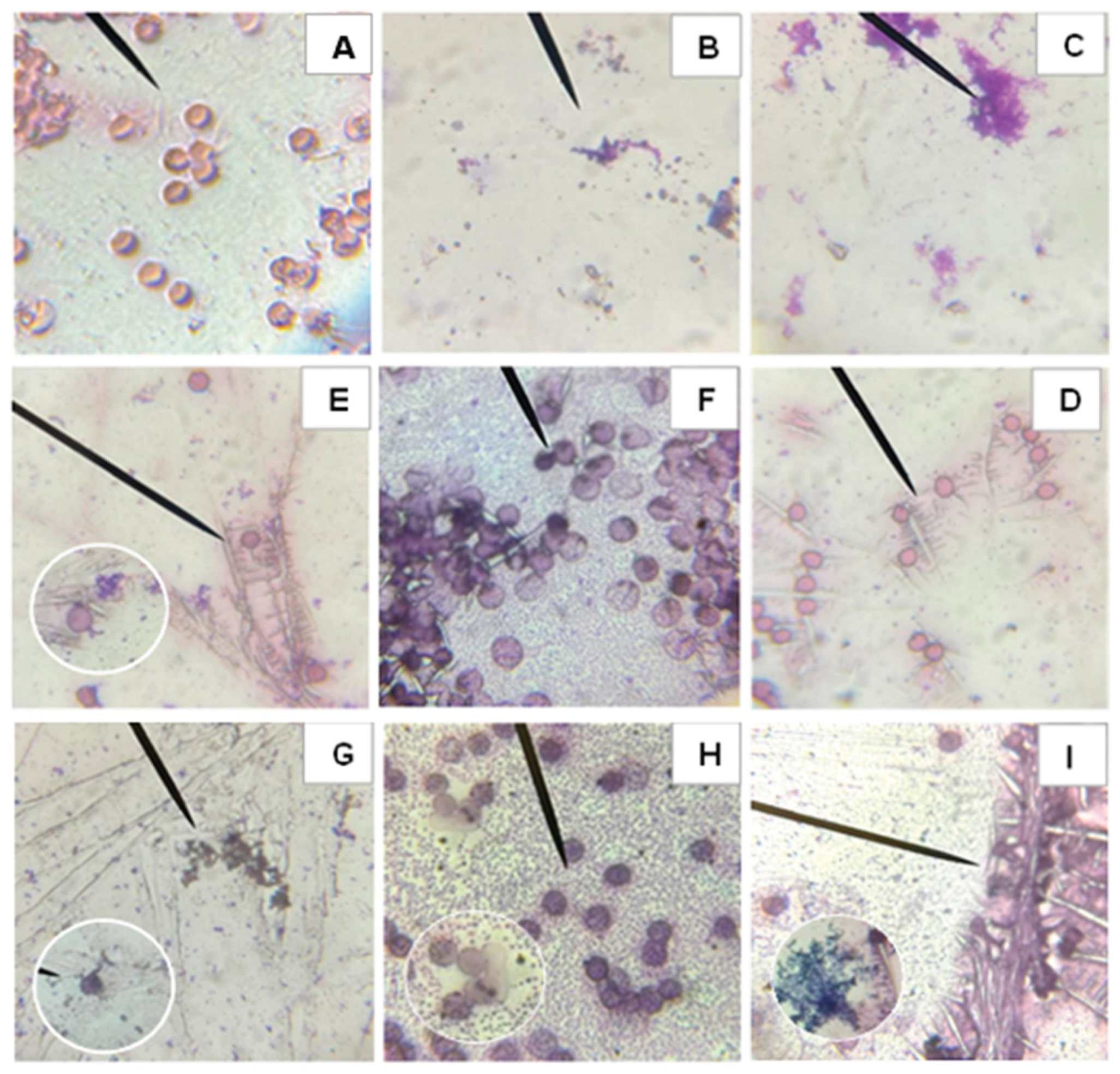
Similar to
Figure 5, microscopic evaluation of O- was performed, allowing visualization of the morphological alterations in erythrocytes after the application of the different samples shown in
Figure 6. Image A shows intact erythrocytes with undamaged membranes, while Image B shows completely hemolyzed cells due to the Triton treatment, used as a positive control. AAPH causes hemolysis and oxidative stress in red blood cells, as seen in Image C. In contrast, C-PC does not show cytotoxic effects (Image D). Comparing Images C and E, it can be observed that C-PC exerts a protective effect against AAPH-induced damage, since erythrocytes with healthy morphology can still be distinguished. Furthermore,
in vitro digestions indicate that both the bioavailable and bioaccessible fractions do not generate morphological alterations in erythrocytes (Images F and H) and contribute to reducing the damage caused by AAPH, with healthy cells evident in images G and I.
3. Discussion
3.1. Quantifying the Antioxidant Capacity of C- Phycocyanin Before and After Digestion In Vitro Digestion
Antioxidant Capacity
The results in
Table 1 show that C-PC exhibits antioxidant activity in the ABTS+• assay. The differences in the antioxidant capacity of C-PC depending on the methods used can be explained by the HAT and SET antioxidant mechanisms specific to each test performed. Compared to DPPH•, the ABTS radical is more sensitive to hydrophilic compounds, such as soluble polyphenols and some water-soluble pigments (phycobiliproteins, oxygenated carotenoids). In the ABTS+• assay, the bioavailable fractions of C-PC showed slightly higher activity than the bioaccessible fractions in two of the three digestions. On the other side, in the DPPH• assay, the opposite behavior was observed, with the bioavailable fractions showing lower antioxidant activity than the bioaccessible fractions. This is probably because not all antioxidant compounds released during digestion are able to cross the simulated intestinal membrane (mainly larger compounds or those associated with complex matrices). The FRAP assay showed the same trend as DPPH•, with bioavailable fractions exhibiting lower antioxidant capacity than bioaccessible fractions. The fact that both DPPH• and FRAP (assays mainly associated with the SET mechanism, Single Electron Transfer) show similar results could indicate a low contribution of this mechanism in bioavailable fractions. This suggests that compounds capable of transferring electrons (such as certain phenolic acids and flavonoids) may not be absorbed efficiently or may be degraded during gastrointestinal transit.
3.2. Cytotoxic and Erythroprotective Effects Before and After In Vitro Digestion
3.2.1. Cytotoxic Effect of IC50 of C-Phycocyanin
When using the IC50 concentrations obtained from the equations corresponding to each blood group, cytotoxicity below 5% hemolysis was observed in all blood groups studied. On the other hand, the azo compound AAPH presented an average cytotoxicity of 27.48±3.8% (
Table 2). Evaluating the cytotoxicity results, it was found that C-phycocyanin extract is harmless in the seven blood groups studied when applying the IC50, since the critical hemolysis limit for good blood biocompatibility must be less than 5% according to ISO/TR7406. In the case of AB+, the hemolytic damage presented is considered minimal.
3.2.2. Cytotoxic Effect of C-Phycocyanin After In Vitro Digestion
In the case of the bioaccessible fraction, cytotoxic results exceeding the permitted 5% hemolysis were obtained, with the exception of groups O and B+, which were within the biocompatibility values. In the case of the bioavailable fraction, C-PC is only cytotoxic in blood group A+, with a value above that permitted by ISO/TR7406. As a result, the C-PC compound is harmless after
in vitro digestion, with the exception of blood type A+, inferring that digestion helps the absorbed compound to be biocompatible with red blood cells and the unabsorbed fraction is cytotoxic. The azo compound AAPH showed cytotoxicity with significant differences between blood groups, with an average hemolysis of 27.18±2.9% (
Figure 3). Control of hemolysis caused by peroxyl radicals produced by the azo compound AAPH showed cytotoxicity with an average hemolysis of 27.18±2.9%.
3.2.3. Erythroprotection of C-PC Using its IC50 Prior to In Vitro Digestion
The IC50 of C-PC obtained from the equations was confirmed, as it inhibits half of the damage caused by the azo compound AAPH. Concentrations ranging from 82.66 to 104.37 µg/mL of C-PC were applied. This variation could be caused by the different conformation of the blood group membrane, which contain different terminal sugars or antigens that can interfere with the action of compounds in terms of both antioxidant capacity and sensitivity to hemolysis, in this case of C-PC or AAPH, respectively (
Table 4). C-PC stood out in the erythroprotective effect determinations to obtain IC50 because values close to this were reached with relatively low concentrations. C-PC has a potent antioxidant effect thanks to its phycocyanobilin chromophores. The main mechanism of cell damage associated with AAPH is the generation of free radicals. Phycocyanin is highly efficient at inhibiting radicals through tetrapyrrole chromophore groups (phycocyanobilin) that react quickly with ROO• radicals, neutralizing them before they initiate chain reactions and prevent lipoperoxidation. Although the potential of phycocyanin is evident, there is some variation in the concentrations required to reach the IC50, due to the different interactions between C-PC and the different compositions of erythrocyte membranes and blood types [
14,
15,
16].
3.2.4. Erythroprotective Effect of C-Phycocyanin After In Vitro Digestion
According to
Figure 4, the bioaccessible fraction shows inhibitions of up to 62.87% with significant differences depending on blood type. In contrast, the bioavailable fraction shows hemolysis inhibition of up to 92.38% with significant differences depending on blood type, with both fractions inhibiting AAPH-induced radicals. The bioaccessible fraction has a significantly lower hemolysis inhibitory capacity than the bioavailable fraction. The erythroprotective assay of C-PC against AAPH-induced hemolysis after
in vitro digestion, specifically the bioaccessible fraction samples, showed that there are no significant differences in each blood group except for O-, where hemolysis inhibition does not differ between samples. However, there are significant differences between the bioavailable fraction and its % hemolysis inhibition between blood groups when applying the same fraction. The erythroprotective assay against hemolysis caused by AAPH of C-PC after
in vitro digestion, specifically of the bioavailable fraction, shows that the cytotoxicity of AAPH resulted in an average of 27.18±2.9% hemolysis. The bioavailable fractions of C-PC inhibiting between 40-90% of hemolysis depending on the blood group.
The ABO and Rh blood groups are differentiated by the presence of specific antigens on the surface of red blood cells. As demonstrated in the results, C-phycocyanin interacts differently with these antigens, affecting its erythroprotective efficacy. It is hypothesized that C-phycocyanin may have specific affinities for blood groups due to its structural properties and electrostatic charge. This explains why the effectiveness of C-phycocyanin varies with different blood groups (A, B, AB, O), with a more pronounced effect observed in one group compared to another. In the case of Rh groups, the difference between positive and negative lies in the presence of the D antigen on the surface of red blood cells. C-phycocyanin interacts differently with Rh-positive erythrocytes due to the presence of this antigen, which modulates its antioxidant and protective effect. However, further experimental studies directly evaluating these interactions and including diverse population samples are essential to confirm these findings.
4. Materials and Methods
4.1. Reagents
C-phycocyanin from Spirulina sp., ABTS [2,2-azino-bis-(3-ethylbenzothiazoline-6-sulfonic acid], AAPH [2,2-Azobis(2-methylpropionamidine) dihydrochloride], DPPH [2,2-diphenyl-1-picrylhydrazyl], Trolox [6-hydroxy -2,5,7,8-tetramethylchroman-2-carboxylic acid], TPTZ [2,4,6-Tripridil-s-triazine], and Tris-HCl biological buffer, were purchased from Sigma-Aldrich (St. Louis, MO, USA). All other chemicals and solvents were of the highest commercial grade.
4.2. Quantification of the Antioxidant Capacity of C-Phycocyanin Before and After In Vitro Digestion
The antioxidant capacity of C-PC from the cyanobacterium
Spirulina sp. was determined before and after
in vitro digestion by evaluating its antiradical potential (ABTS+• and DPPH• inhibition assay) and reducing potential (FRAP). A Trolox curve was constructed to express the results of the different antioxidant activity assays as µmol ET (Trolox equivalents)/g PS. Trolox is a synthetic hydrophilic antioxidant standard analogous to vitamin E. The Trolox equivalent unit implies the antiradical and reduced capacity of the Trolox antioxidant. In addition, the ability to inhibit peroxyl radicals generated by AAPH on human erythrocytes was analyzed [
17].
4.2.1. Inhibition Assay of 2,2-azinobis-(3-ethylbenzothiazolin)-6-sulfonic acid Free Radical (ABTS+•)
The antiradical activity against the ABTS+• free radical was analyzed based on the proposed technique with some modifications [
18]. ABTS salt (19.3 mg) was dissolved in distilled water (5 mL), then 88 μL of potassium persulfate (K2S2O8) solution (88 μL) (0.0378 g/L) was added and left to stand in the dark for 12 h in order to oxidize the ABTS to ABTS+•. The ABTS+• solution was diluted with 99% ethanol until an absorbance of 0.7±0.05 at 734 nm was obtained. The following treatments were performed in triplicate in a 96-well microplate: negative control (20 µL ethanol + 270 µL ABTS+•), standard control (20 μL standard antioxidant + 270 μL ABTS+•), and samples (20 μL extract + 270 μL ABTS+•). The samples were left to stand for 30 min in the dark at room temperature, after which readings were taken at 734 nm in a microplate spectrophotometer (Multiskan Go, Thermo Scientific, Waltham, MA, USA). The results were reported as µmol ET (Trolox equivalents)/g DW.
4.2.2. Free Radical Inhibition Assay 1,1-diphenyl-2-picrylhydrazyl (DPPH•)
The inhibitory capacity of the DPPH• free radical was evaluated following the published protocol [
19]. The methanolic solution of DPPH• (6×10-5 mol·L
-1) was prepared and 99% methanol was gradually added until an adjusted absorbance of 0.7 ± 0.05 at 515 nm was obtained. The following treatments were performed in triplicate in a 96-well microplate: negative control (20 μL methanol + 200 μL DPPH•), standard control (20 μL standard antioxidant + 200 μL DPPH•), and sample (20 μL extract + 200 μL DPPH•). The samples were left to stand for 30 min at room temperature in the dark. The absorbances were read at 515 nm in a spectrophotometer (Multiskan Go, Thermo Scientific, Waltham, MA, USA). The results were reported as µmol ET (Trolox equivalents)/g DW.
4.2.3. Ferric Ion Antioxidant Reducing Power (FRAP)
The reducing potential was determined following the methodology proposed by with some modifications [
20]. The stock solutions were sodium acetate buffer (300 mM, pH 3.6), ferric chloride (FeCl₃) solution (20 mM), and TPTZ (2,4,6-triphenyl-s-triazine) solution (10 mM) in HCl (40 mM). The FRAP working solution was prepared in a ratio of 10:1:1 (buffer: FeCl₃: TPTZ). The following treatments were carried out in triplicate in a 96-well microplate: negative control (20 µL methanol + 280 µL FRAP), standard control (20 µL standard antioxidant + 280 µL FRAP) and sample (20 µL extract + 280 µL FRAP). The samples were left to stand for 30 min in the dark at room temperature. The optical densities at 638 nm were obtained using a spectrophotometer (Multiskan Go, Thermo Scientific, Waltham, MA, USA). The results were reported as µmol ET (Trolox equivalents)/g DW.
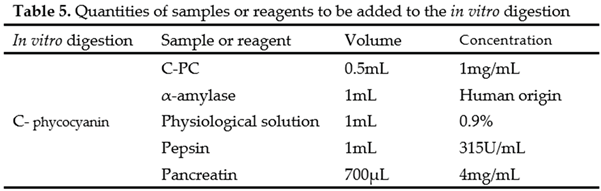
4.3. In Vitro Digestion
This was carried out using an established protocol, with modifications [
21]. Human amylase (saliva) was added to the sample and left to stand for 2 minutes. After this time, physiological solution was added, followed by homogenization. It was then acidified to pH 2 with 6M HCl. Once adjusted, pepsin at 315 U/mL was added, and it was placed in a water bath for 2 hours at 37°C and 80 rpm. After this time, the pH was neutralized to 7.4 with 1.25M NaHCO3, 4mg/mL pancreatin was added, homogenized, and previously hydrated cellulose membranes were filled with physiological solution for 1 day, placed in a water bath for 4 hours at 37°C and 80rpm. After the incubation period, the antioxidant capacity was quantified (Table 5).
4.4. Cytotoxic and Erythroprotective Effect
4.4.1. Erythrocyte Cytotoxicity Assay
All methods that utilized human red blood cells (RBCs) were conducted in accordance with Mexican regulations (NOM-253-SSA1-2012) and international standards (FDA: CFR—Code of Federal Regulations Title 21, part 640, Additional Standards for human blood and blood products, Support. B Red blood cells, Sec. 640.14, Blood testing (21CFR640.14)). The protocol was approved by the ethics committee of the General Hospital of the state of Sonora (CI 2023-47).
Red blood cell (RBC) samples were collected by venipuncture into EDTA tubes using the Vacutainer system. Blood was obtained from all possible groups (ABO with Rh + and –) from apparently healthy individuals over the age of 18 with their respective informed consent. Washing was performed with physiological solution (PS) by adding 3000 µL to 1000 µL of blood, which was then centrifuged at 1500 rpm for 10 min. After this time, the supernatant was removed, and the process was repeated three times to remove all plasma. Subsequently, a 2% erythrocyte suspension was made from all blood groups obtained (using 0.9% physiological solution). Negative control (100 µL erythrocyte suspension + 200 µL physiological solution), positive control (100 µL erythrocyte suspension + 100 µL physiological solution + 100 µL Triton 1%), sample (100 µL erythrocyte suspension + 100 µL physiological solution + 100 µL sample). Subsequently, the controls and tubes with the samples were incubated for 3 hours at 37 °C, the reaction sample was diluted with 1 mL of physiological solution and centrifuged at 7,000 rpm for 2 minutes. Three hundred µL of the supernatant was taken and read in triplicate in a spectrophotometer with a microplate reader at 540 nm. The cytotoxic effect was measured by quantifying the percentage of hemolysis according to equation 1:
4.4.2. Antihemolytic Activity in Erythrocytes by the 2,2'-azobis-(2-methylpropionic-(2-methylpropionamidine) (AAPH)
Oxidative hemolysis was induced with the compound AAPH, which is listed as a free radical initiator, based on the proposed methods [
22,
23]. Negative control (100 μL of 2% erythrocyte suspension + 200 μL of physiological solution), positive control (100 μL of erythrocyte suspension, 100 μL of physiological solution, and 100 μL of AAPH), and sample (100 μL erythrocyte suspension + 100 μL AAPH + 100 μL). The tubes were incubated at 37°C with agitation at 40 rpm in the dark for 3 h. After incubation, the reaction sample was diluted with 1 mL of physiological solution and centrifuged at 7000 rpm for 2 minutes. The absorbance of the supernatant was measured at 540 nm in a spectrophotometer using 96-well microplates. All measurements were performed in triplicate. The percentage of hemolysis inhibition was calculated using equation 2:
4.5. Erythrocyte Cell Membrane
To examine alterations in the erythrocyte cell membrane, an optical microscope (Eclipse FN1 model with 100x magnification) was used. In this analysis, the supernatant samples were evaluated immediately after reading. Approximately 50 µL of fresh plasma were added to the packed red blood cells obtained from the hemolysis inhibition test, mixing carefully to avoid any mechanical damage to the membranes. A drop of the blood suspension was placed on a slide, forming a thin layer of erythrocytes. Wright staining was used to observe the structure of these membranes. The micrographs were examined at 100x magnification and are presented with a 5 µm scale bar to facilitate comparison of erythrocyte size. The images were acquired using NIS-Elements F v4.11.0 software (Nikon Instruments Inc, Americas, New York, NY, USA;
https://www.microscope.healthcare.nikon.com/es_AMS/products/software/nis-elements/viewer, accessed on 13 May 2025).
5. Statistical Analysis
The experimental results were analyzed using inferential statistics with a 95% confidence level. All assays were performed with a minimum of three independent replicates (n ≥ 3), and the data were expressed as mean ± standard deviation (SD). To determine significant differences among treatments, blood groups, and fractions (bioaccessible/bioavailable), a one-way analysis of variance (one-way ANOVA) was applied. Data processing and statistical analysis were conducted using specialized software, and the interpretation was based on comparisons among ABO/Rh blood groups, fractions before and after in vitro digestion, as well as on the interaction between C-phycocyanin, the peroxyl radical generated by AAPH, and the erythrocyte membrane.
6. Conclusions
The results of the analyses of the erythroprotective effect of C-phycocyanin from the cyanobacterium Spirulina sp. revealed a probable relationship between the different antigens (ABO and RhD) and the antihemolytic effect. The results of erythroprotection after in vitro digestion show that the bioavailable fractions have a higher percentage of hemolysis inhibition against peroxyl radicals; the digested fraction of C-phycocyanin has better inhibition than the undigested samples. The optical differences between treatments are noticeable under the microscope, where the cytotoxic activity of AAPH and erythroprotection of the samples can be observed. These findings highlight potential biotechnological and biomedical applications of the extract as a safe ingredient in the development of nutraceuticals and functional foods, as well as in formulations with preventive potential against oxidative stress.
Author Contributions
Conceptualization, Y.I.C.-R., R.I.G.-V. and M.A.R.-G.; writing—original draft preparation, C.J.G.-C.; writing—review and editing, A.T.B.-M. and S.R.-C.; visualization J.J.O.-P. and J.B.-F.; resources J.R.R.-E.; and supervision C.L.D.-T.-S and D.F.-O. All authors have read and agreed to the published version of the manuscript.
Funding
The work was supported by the project CBF2023-2024-3196 from Secretaría de Ciencia, Humanidades, Tecnología e Innovación (SECIHTI).
Institutional Review Board Statement
The study was carried out in accordance with the Declaration of Helsinki of 1975. The protocol was approved by the ethics committee of the General Hospital of the state of Sonora (CI 2023-47).
Informed Consent Statement
All subjects gave their informed consent for inclusion in the study prior to their participation.
Data Availability Statement
The original contributions of data presented in this research are included in the article; further inquiries can be directed to the corresponding authors.
Acknowledgments
The authors are pleased to acknowledge of Secretaría de Ciencia, Humani-dades, Tecnología e Innovación (SECIHTI) for awarding Jesús Cinthia Jael Gaxiola Calvo for his master’s scholarship.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| AAPH |
2,2-Azobis(2-methylpropionamidine) dihydrochloride |
| ABTS |
2,2-azino-bis-(3-ethylbenzothiazoline-6-sulfonic acid |
| APC |
Allophycocyanin |
| B-PE |
B- Phycoerythrin |
| C-PC |
C- Phycocyanin |
| DPPH |
2,2-diphenyl-1-picrylhydrazyl |
| DW |
Dry Weight |
| FRAP |
Ferric Reducing Antioxidant Power |
| PBP |
Phycobiliprotein |
| PSU |
Practical Salinity Units |
| PS |
Physiological Solution |
| R-PC |
R- Phycocyanin |
| TPTZ |
2,4,6-Tripridil-s-triazine |
| Trolox |
6-hydroxy -2,5,7,8-tetramethylchroman-2-carboxylic acid |
References
- Alcides González Gil, L., Yisel González Madariaga, I., Dra Grecia Martínez Leyva, I., Felipe Hernández Ugalde, I., & Adalberto Suárez González, I. V. (2012). Daño oxidativo en un modelo experimental de hiperglicemia e hiperlipidemia inducida por sacarosa en ratas wistar. Revista Médica Electrónica, 34(4), 406–416. http://scielo.sld.cu/scielo.php?script=sci_arttext&pid=S1684-18242012000400001&lng=es&nrm=iso&tlng=es.
- Alfaro-Alfaro, Á. E., Alpízar-Cambronero, V., Duarte-Rodríguez, A. I., Feng-Feng, J., Rosales-Leiva, C., & Mora-Román, J. J. (2020). C-ficocianinas: Modulación del sistema inmune y su posible aplicación como terapia contra el cáncer. Revista Tecnología En Marcha, 33, ág. 125-139. [CrossRef]
- Carlos, S.-B., Celestino, G.-G., Claudio, G.-B., Julia, M.-R., Juan, N.-A., Julio, B.-R., Villa, F. S., Escobedo, G., & León, N. (2023). Optimización para la extracción de ficocianina de la cianobacteria Spirulina maxima. Investigación y Desarrollo En Ciencia y Tecnología de Alimentos, 8(1), 834–840. [CrossRef]
- Contreras, N. A. V., & Caicedo, C. R. (2022). Análisis bibliométrico del efecto de la luz en la producción de ficobiliproteínas. TecnoLógicas, 25(54), e2386–e2386. [CrossRef]
- Alchazal, R., Zaitoun, K. J., Al-Qudah, M., Zaitoun, G., Taha, A. M., Saleh, O., Alqudah, M., Abuawwad, M., Taha, M., & Aldalati, A. Y. (2025). Relationship between ABO blood group antigens and Rh factor with breast cancer: A systematic review and meta-analysis. Oncotarget, 16, 311–326. [CrossRef]
- Asadi-Pooya, A. A., Namjoo-Moghadam, A., & Cheraghipour, M. (2022). ABO blood group and Rh factor distributions in patients with epilepsy. Epilepsy and Behavior, 128. [CrossRef]
- Dogan, O. (2023). Are ABO/Rh blood groups A risk factor for polycystic ovary syndrome? Medicine, 102(36), e34944. [CrossRef]
- Torres-Alarcón, C. G., García-Ruíz, A., Cañete-Ibáñez, C. R., Morales-Pogoda, I. I., Muñoz-Arce, C. M., Cid-Domínguez, B. E., Montalvo-Bárcenas, M., Maza-de la Torre, G., Sandoval-López, C., Gaytán-Guzmán, E., Correo- Zamora, J. D., Torres-Alarcón, C. G., García-Ruíz, A., Cañete-Ibáñez, C. R., Morales-Pogoda, I. I., Muñoz-Arce, C. M., Cid-Domínguez, B. E., Montalvo-Bárcenas, M., Maza-de la Torre, G., … Correo- Zamora, J. D. (2021). Antígenos del sistema sanguíneo ABO como factor de riesgo para la gravedad de la infección por SARS-CoV-2. Gaceta Médica de México, 157(2), 181–187. [CrossRef]
- Zhao, J., Yang, Y., Huang, H., Li, D., Gu, D., Lu, X., Zhang, Z., Liu, L., Liu, T., Liu, Y., He, Y., Sun, B., Wei, M., Yang, G., Wang, X., Zhang, L., Zhou, X., Xing, M., & Wang, P. G. (2020). Relationship between the ABO Blood Group and the COVID-19 Susceptibility. MedRxiv, 2020.03.11.20031096. [CrossRef]
- Benzie, I. F. F., & Strain, J. J. (1996). The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay. Analytical Biochemistry, 239(1), 70–76. [CrossRef]
- Brand-Williams, W., Cuvelier, M. E., & Berset, C. (1995). Use of a free radical method to evaluate antioxidant activity. LWT - Food Science and Technology, 28(1), 25–30. [CrossRef]
- González-Vega, R. I., Robles-García, M. Á., Mendoza-Urizabel, L. Y., Cárdenas-Enríquez, K. N., Ruiz-Cruz, S., Gutiérrez-Lomelí, M., Iturralde-García, R. D., Avila-Novoa, M. G., Villalpando-Vargas, F. V., & Del-Toro-Sánchez, C. L. (2023). Impact of the ABO and RhD Blood Groups on the Evaluation of the Erythroprotective Potential of Fucoxanthin, β-Carotene, Gallic Acid, Quercetin and Ascorbic Acid as Therapeutic Agents against Oxidative Stress. Antioxidants 2023, Vol. 12, Page 2092, 12(12), 2092. [CrossRef]
- Halliwell, B. (2006). Reactive species and antioxidants. Redox biology is a fundamental theme of aerobic life. Plant Physiology, 141(2), 312–322. [CrossRef]
- Hernández-Ruiz, K. L., Ruiz-Cruz, S., Cira-Chávez, L. A., Gassos-Ortega, L. E., de Jesús Ornelas-Paz, J., Del-Toro-Sánchez, C. L., Márquez-Ríos, E., López-Mata, M. A., & Rodríguez-Félix, F. (2018). Evaluation of antioxidant capacity, protective effect on human erythrocytes and phenolic compound identification in two varieties of plum fruit (Spondias spp.) by UPLC-MS. Molecules, 23(12). [CrossRef]
- Li, W., Su, H. N., Pu, Y., Chen, J., Liu, L. N., Liu, Q., & Qin, S. (2019). Phycobiliproteins: Molecular structure, production, applications, and prospects. Biotechnology Advances, 37(2), 340–353. [CrossRef]
- McCarty, M. F. (2007). Clinical potential of Spirulina as a source of phycocyanobilin. Journal of Medicinal Food, 10(4), 566–570. [CrossRef]
- Ozgen, M., Reese, R. N., Tulio, A. Z., Scheerens, J. C., & Miller, A. R. (2006). Modified 2,2-azino-bis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS) method to measure antioxidant capacity of selected small fruits and comparison to ferric reducing antioxidant power (FRAP) and 2,2′-diphenyl-1- picrylhydrazyl (DPPH) methods. Journal of Agricultural and Food Chemistry, 54(4), 1151–1157. [CrossRef]
- Parcheta, M., Świsłocka, R., Orzechowska, S., Akimowicz, M., Choińska, R., & Lewandowski, W. (2021). Recent developments in effective antioxidants: The structure and antioxidant properties. Materials, 14(8). [CrossRef]
- Re, R., Pellegrini, N., Proteggente, A., Pannala, A., Yang, M., & Rice-Evans, C. (1999). Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Biology and Medicine, 26(9–10), 1231–1237. [CrossRef]
- Rodríguez-Roque, M. J., Del-Toro-sánchez, C. L., Chávez-Ayala, J. M., González-Vega, R. I., Pérez-Pérez, L. M., Sánchez-Chávez, E., Salas-Salazar, N. A., Soto-Parra, J. M., Iturralde-García, R. D., & Flores-Córdova, M. A. (2022). Digestibility, Antioxidant and Anti-Inflammatory Activities of Pecan Nutshell (Carya illioinensis) Extracts. Journal of Renewable Materials, 10(10), 2569–2580. [CrossRef]
- Ruiz-Hernández, Y. A., Garza-Valverde, E., Márquez-Reyes, J. R., & García-Gómez, C. (2023). Extracción de ficocianina para uso como colorante natural: optimización por metodología de superficie de respuesta. Investigación y Desarrollo En Ciencia y Tecnología de Alimentos, 8(1), 84–91. [CrossRef]
- Sadeer, N. B., Montesano, D., Albrizio, S., Zengin, G., & Mahomoodally, M. F. (2020). The versatility of antioxidant assays in food science and safety—chemistry, applications, strengths, and limitations. Antioxidants, 9(8), 1–39. [CrossRef]
- Siddeeg, A., AlKehayez, N. M., Abu-Hiamed, H. A., Al-Sanea, E. A., & AL-Farga, A. M. (2021). Mode of action and determination of antioxidant activity in the dietary sources: An overview. Saudi Journal of Biological Sciences, 28(3), 1633–1644. [CrossRef]
Figure 1.
Percentage of hemolysis inhibition using C-phycocyanin at different concentrations on erythrocytes supplemented with AAPH.
Figure 1.
Percentage of hemolysis inhibition using C-phycocyanin at different concentrations on erythrocytes supplemented with AAPH.
Figure 2.
Blood cytotoxicity assay of the inhibitory concentration 50 of C-phycocyanin obtained from the cyanobacterium Spirulina sp. on human erythrocytes with different ABO blood groups. Data are presented as the mean ± SD (standard deviation) of at least three replicates (n ≥ 3). Different lowercase letters (x, y, z) represent significant differences in the effect of AAPH (40mM) between blood groups (p < 0.001). Different lowercase letters (a, b, c) represent significant differences with IC50 of C-PC between blood groups (p < 0.001).
Figure 2.
Blood cytotoxicity assay of the inhibitory concentration 50 of C-phycocyanin obtained from the cyanobacterium Spirulina sp. on human erythrocytes with different ABO blood groups. Data are presented as the mean ± SD (standard deviation) of at least three replicates (n ≥ 3). Different lowercase letters (x, y, z) represent significant differences in the effect of AAPH (40mM) between blood groups (p < 0.001). Different lowercase letters (a, b, c) represent significant differences with IC50 of C-PC between blood groups (p < 0.001).
Figure 3.
Blood cytotoxicity assay of 1 mg/mL C-phycocyanin obtained from the cyanobacterium Spirulina sp. after in vitro digestion on human erythrocytes with different ABO blood groups. Where Bioaccessible: sample inside the cellulose membrane and Bioavailable: sample outside the cellulose membrane. Different lowercase letters (x, y, z) represent significant differences in the AAPH (40mM) positive control between blood groups, and lowercase letters (a, b, c) indicate significant differences between in vitro digestion fractions and blood groups (p<0.005).
Figure 3.
Blood cytotoxicity assay of 1 mg/mL C-phycocyanin obtained from the cyanobacterium Spirulina sp. after in vitro digestion on human erythrocytes with different ABO blood groups. Where Bioaccessible: sample inside the cellulose membrane and Bioavailable: sample outside the cellulose membrane. Different lowercase letters (x, y, z) represent significant differences in the AAPH (40mM) positive control between blood groups, and lowercase letters (a, b, c) indicate significant differences between in vitro digestion fractions and blood groups (p<0.005).
Figure 4.
Erythroprotection assay against hemolysis induced by the azocompound AAPH (40mM) using 1mg/mL of C-phycocyanin after in vitro digestion on human erythrocytes with different ABO blood groups. Where Bioaccessible+AAPH: sample inside the cellulose membrane against peroxyl radicals and Bioavailable+AAPH: sample outside the cellulose membrane against peroxyl radicals. Different capital letters indicate significant differences in the AAPH positive control be-tween blood groups, and lowercase letters indicate significant differences between in vitro digestion fractions and blood groups (p<0.005).
Figure 4.
Erythroprotection assay against hemolysis induced by the azocompound AAPH (40mM) using 1mg/mL of C-phycocyanin after in vitro digestion on human erythrocytes with different ABO blood groups. Where Bioaccessible+AAPH: sample inside the cellulose membrane against peroxyl radicals and Bioavailable+AAPH: sample outside the cellulose membrane against peroxyl radicals. Different capital letters indicate significant differences in the AAPH positive control be-tween blood groups, and lowercase letters indicate significant differences between in vitro digestion fractions and blood groups (p<0.005).
Figure 5.
Smears from biocompatibility and hemolysis inhibition tests before and after in vitro digestion of C-phycocyanin (C-PC) from the cyanobacterium Spirulina sp. in blood group O+. Where A: Negative control, B: Positive control Triton 1%, C: Positive control AAPH, D: C-PC, E: C-PC + AAPH, F: Fraction inside the cellulose membrane after in vitro digestion of the C-PC sample, G: Fraction within the cellulose membrane after in vitro digestion of the C-PC + AAPH sample, H: Fraction outside the cellulose membrane after in vitro digestion of the C-PC sample, I: Fraction outside the cellulose membrane after in vitro digestion of the C-PC + AAPH sample.
Figure 5.
Smears from biocompatibility and hemolysis inhibition tests before and after in vitro digestion of C-phycocyanin (C-PC) from the cyanobacterium Spirulina sp. in blood group O+. Where A: Negative control, B: Positive control Triton 1%, C: Positive control AAPH, D: C-PC, E: C-PC + AAPH, F: Fraction inside the cellulose membrane after in vitro digestion of the C-PC sample, G: Fraction within the cellulose membrane after in vitro digestion of the C-PC + AAPH sample, H: Fraction outside the cellulose membrane after in vitro digestion of the C-PC sample, I: Fraction outside the cellulose membrane after in vitro digestion of the C-PC + AAPH sample.
Figure 6.
Smears from biocompatibility and hemolysis inhibition tests before and after in vitro digestion of C-phycocyanin (C-PC) from the cyanobacterium Spirulina sp. in blood group O-. Where A: Negative control, B: Positive control Triton 1%, C: Positive control AAPH, D: C-PC, E: C-PC + AAPH, F: Fraction inside the cellulose membrane after in vitro digestion of the C-PC sample, G: Fraction within the cellulose membrane after in vitro digestion of the C-PC + AAPH sample, H: Fraction outside the cellulose membrane after in vitro digestion of the C-PC sample, I: Fraction outside the cellulose membrane after in vitro digestion of the C-PC + AAPH sample.
Figure 6.
Smears from biocompatibility and hemolysis inhibition tests before and after in vitro digestion of C-phycocyanin (C-PC) from the cyanobacterium Spirulina sp. in blood group O-. Where A: Negative control, B: Positive control Triton 1%, C: Positive control AAPH, D: C-PC, E: C-PC + AAPH, F: Fraction inside the cellulose membrane after in vitro digestion of the C-PC sample, G: Fraction within the cellulose membrane after in vitro digestion of the C-PC + AAPH sample, H: Fraction outside the cellulose membrane after in vitro digestion of the C-PC sample, I: Fraction outside the cellulose membrane after in vitro digestion of the C-PC + AAPH sample.
Table 1.
Determination of antioxidant activity of C-PC before and after in vitro digestion (µmol ET/g DW).
Table 1.
Determination of antioxidant activity of C-PC before and after in vitro digestion (µmol ET/g DW).
| Sample |
ABTS+• |
DPPH• |
FRAP |
| C-PC before digestion in vitro
|
1.58Ba±0.42 |
1.32ABa±0.08 |
0.03Cb±0.02 |
| Bioaccessible Fraction |
6.97Aa±0.42 |
1.21bA±0.48 |
0.13Ac±0.01 |
| Bioavailable Fraction |
7.34Aa±0.08 |
0.89Ab±0.42 |
0.07Bc±0.01 |
Table 2.
Percentage of hemolysis caused by C-phycocyanin from the cyanobacterium Spirulina sp. at different concentrations on human erythrocytes in different blood types and Rh factors.
Table 2.
Percentage of hemolysis caused by C-phycocyanin from the cyanobacterium Spirulina sp. at different concentrations on human erythrocytes in different blood types and Rh factors.
| |
µg/mL |
ABO
Rh +/-* |
50 |
75 |
100 |
125 |
150 |
300 |
AAPH |
| A+ |
0A±1.35 |
0A±1.10 |
0.58A±0.59 |
0.29A±0.54 |
2.26A±1.58 |
1.47B± 0.47 |
25.13ABC±2.53 |
| A- |
0B±0.21 |
0.12A±0.77 |
0A±0.78 |
0A±1.68 |
0B±1.66 |
0.65B±1.44 |
20.95E±0.96 |
| O+ |
0A±0.97 |
0.03A±1.26 |
0A±0.83 |
0.34A±0.85 |
0.04B±0.81 |
1.13A±0.94 |
27.01AB±0.49 |
| O- |
0A±0.89 |
0A±0.39 |
0A±0.37 |
0A±0.93 |
0B±0.91 |
0B±1.31 |
26.23BC±0.97 |
| B+ |
0.97A±0.75 |
0.43A±0.95 |
0A±0.44 |
0A±0.35 |
0B±1.41 |
0.98A±2.03 |
25.29C±0.55 |
| AB+ |
0A±0.73 |
0.25A±0.90 |
0A±0.51 |
0A±2 |
1.01B±0,61 |
2A±0.5 |
23.35D±1.24 |
| AB- |
0A±0.77 |
0A±0.50 |
0A±0.91 |
0.70A±0.39 |
0.28B±0.95 |
1.75AB±0.30 |
28.10A±1.00 |
Table 3.
Inhibitory concentration 50 in µg/mL of C-phycocyanin against hemolysis caused by peroxyl radicals obtained by equations.
Table 3.
Inhibitory concentration 50 in µg/mL of C-phycocyanin against hemolysis caused by peroxyl radicals obtained by equations.
ABO
Rh +/-*
|
C-PC
µg/mL
|
Equation |
R2
|
Model |
| A+ |
82.66 |
y = 0.5216x + 6.8853 |
0.9593 |
Linear |
| A- |
95.25 |
y = -0.0055x2 + 1.0478x + 1.0108 |
0.9352 |
Polynomial |
| O+ |
92.53 |
y = 0.5512x – 1.0054 |
0.9896 |
Linear |
| O- |
82.05 |
y = -0.0072x2 + 1.1813x + 1.5912 |
0.9316 |
Polynomial |
| B+ |
88.26 |
y = 0.4973x + 6.1085 |
0.9677 |
Linear |
| AB+ |
104.37 |
y = -0.0057x2 + 1.1897x + 1.3272 |
0.9704 |
Polynomial |
| AB- |
101.94 |
y = -0.0051x2 + 1.0397x + 0.0859 |
0.9986 |
Polynomial |
Table 4.
Percentage inhibitor of AAPH 40Mm induced hemolysis using the IC50 of C-PC.
Table 4.
Percentage inhibitor of AAPH 40Mm induced hemolysis using the IC50 of C-PC.
| Bood Type |
µg/mL |
% Hemolysis Inhibition |
| A+ |
82.66 |
50.08 ± 1.7 |
| A- |
95.25 |
48.58 ± 1.9 |
| O+ |
92.53 |
50.07 ± 1.3 |
| O- |
82.05 |
50.70 ± 0.7 |
| B+ |
88.26 |
50.96 ± 1.1 |
| AB+ |
104.37 |
50.27 ± 1.9 |
| AB- |
101.94 |
49.95 ± 1.7 |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).