Submitted:
04 December 2025
Posted:
05 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
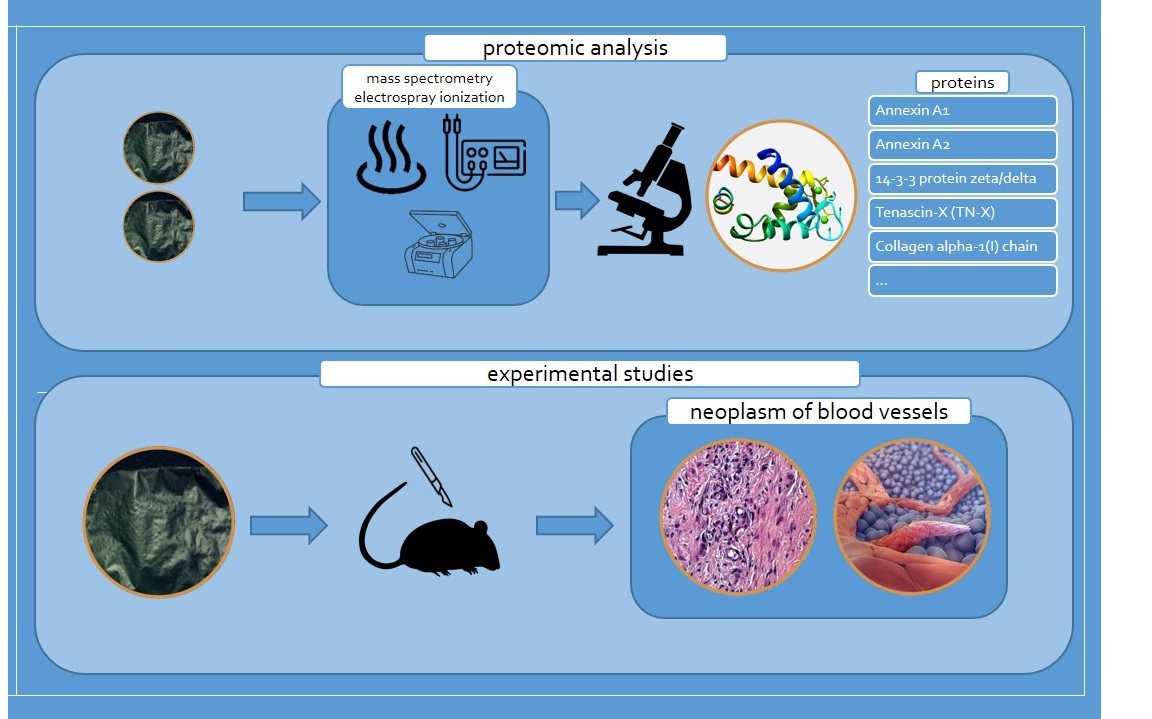
2. Materials and Methods
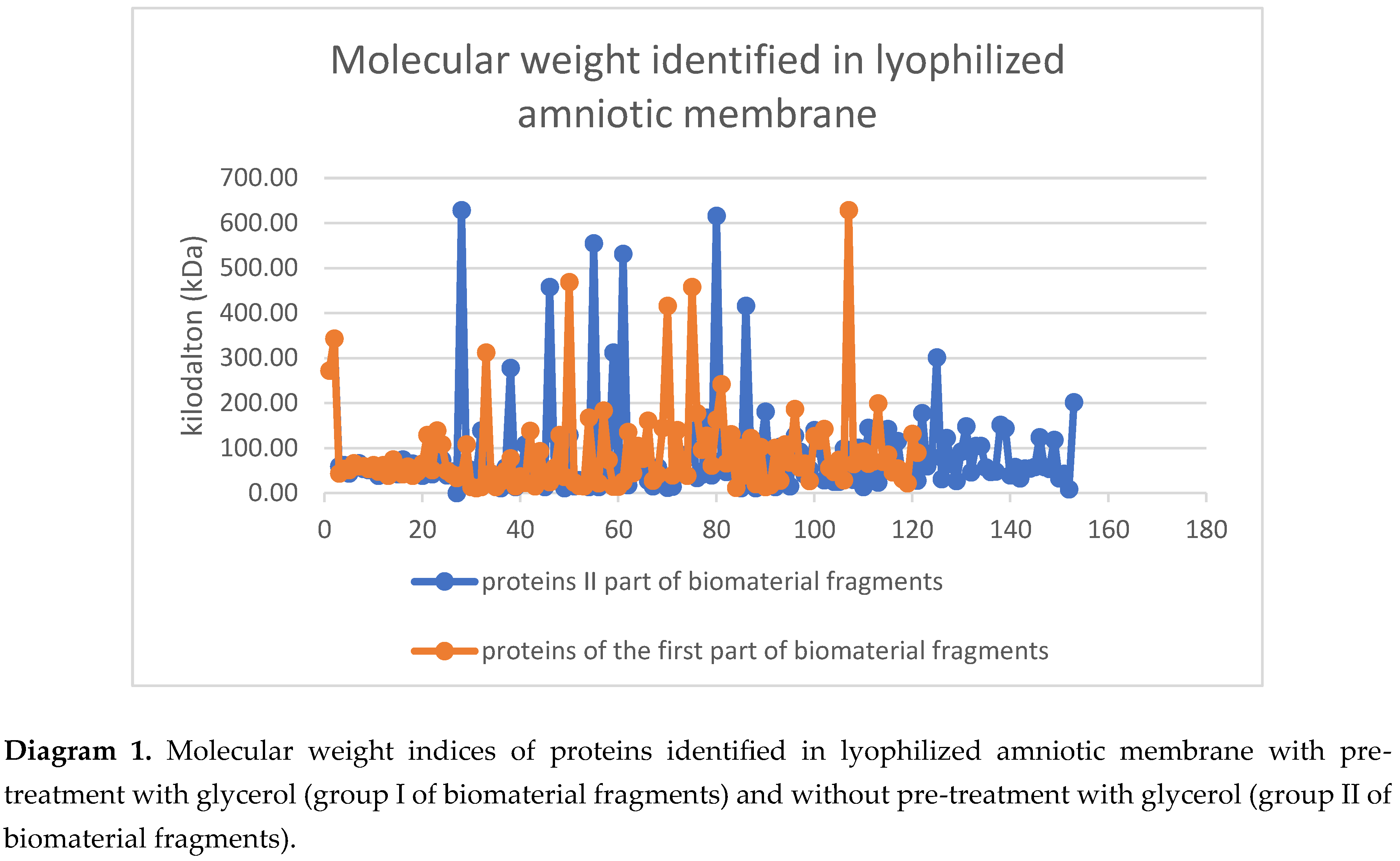
3. Results
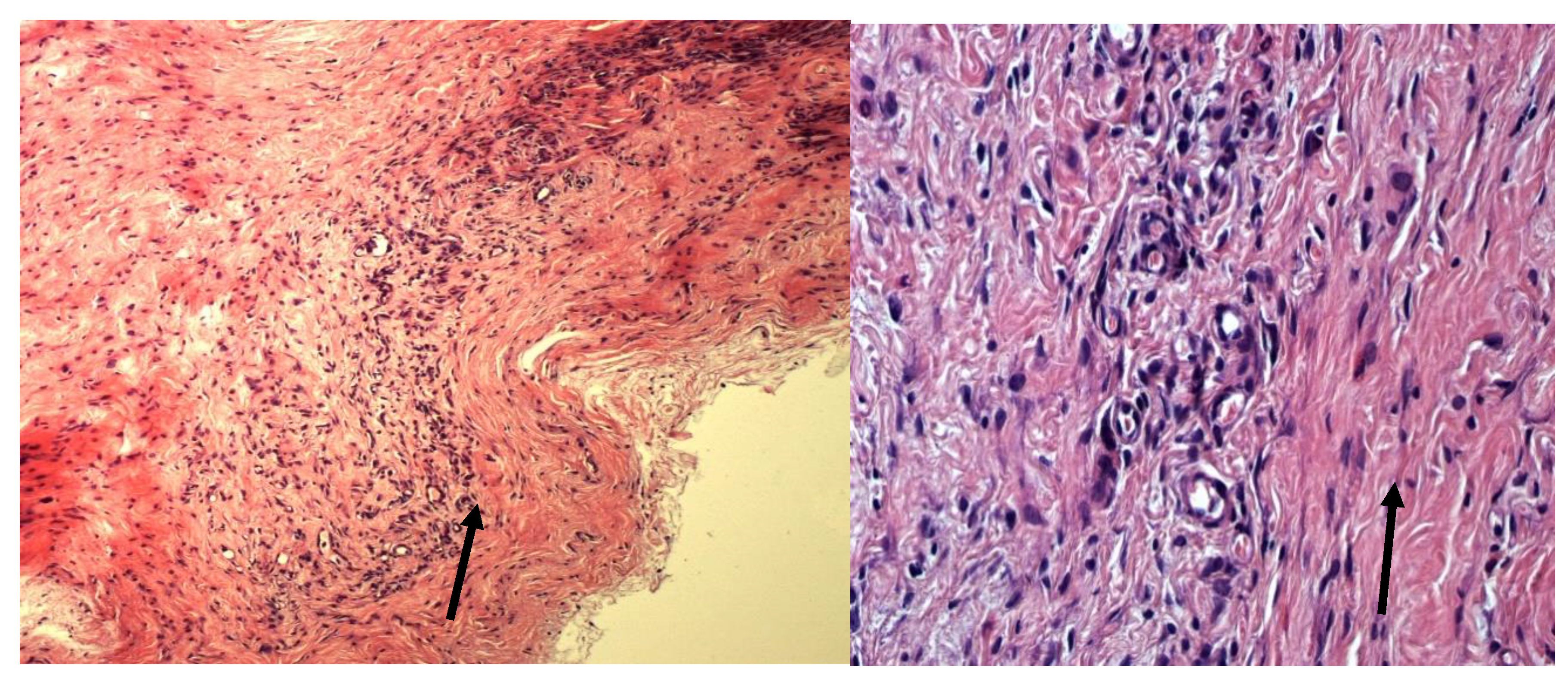
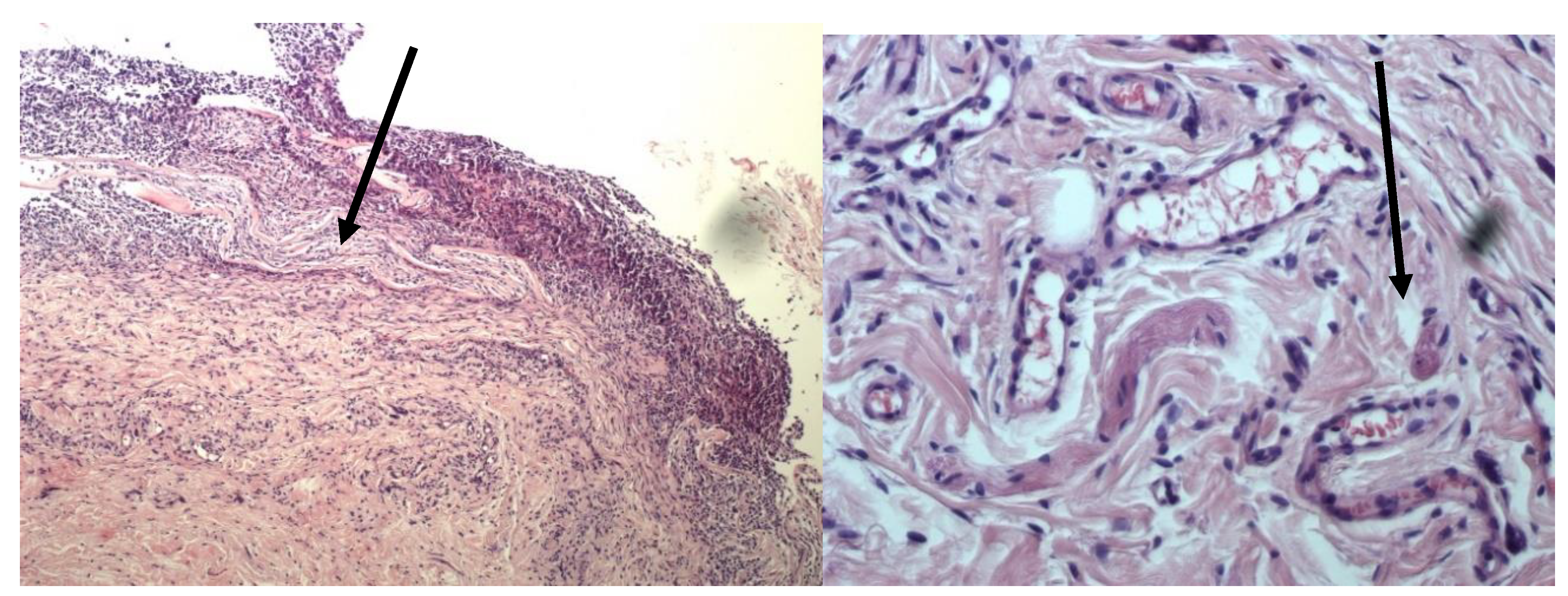
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Meller D, Pires RT, Mack RJ, Figueiredo F, Heiligenhaus A, Park WC, Prabhasawat P, John T, McLeod SD, Steuhl KP, Tseng SC. Amniotic membrane transplantation for acute chemical or thermal burns. Ophthalmology. 2000 May;107(5):980-9; discussion 990. PMID: 10811094. [CrossRef]
- Niknejad H, Peirovi H, Jorjani M, Ahmadiani A, Ghanavi J, Seifalian AM. Properties of the amniotic membrane for potential use in tissue engineering. Eur Cells Mater. 2008;15:88–99.
- Tsai R., Li L., Chan J. Reconstruction of damaged corneas by transplantation of autologous limbal epithelial cells. N. Engl. J. Med; 2000.
- Nakamura T, Inatomi T, Sotozono C, et al. Ocular surface reconstruction using stem cell and tissue engineering. Progress in retinal and eye research 2016; 51: 187–207.
- Koizumi N., Fullwood N., Bairaktaris G., et al. Cultivasion of corneal epithelial cells on intact and denuded human amniotic membrane. Invest. Ophthalmol. Vis. Sci. 2000; 41.
- Adds P. J., Hunt C. J., Dart J. K. G. Amniotic membrane grafts, “fresh” or frozen? A clinical and in vitro comparison//Br J Ophthalmol 2001;85:905-907.
- Milyudin E.S. Technology of preservation of amniotic membrane by drying over silica gel//Technologies of living systems 2006. Vol.—3. No. -3. -P.44-50.
- Kim JC, Tseng SC. The effects on inhibition of corneal neovascularization after human amniotic membrane transplantation in severely damaged rabbit corneas. Korean J Ophthalmol. 1995 Jun;9(1):32-46. PMID: 7674551. [CrossRef]
- Shao C, Sima J, Zhang SX, Jin J, Reinach P, Wang Z, Ma JX. Suppression of corneal neovascularization by PEDF release from human amniotic membranes. Invest Ophthalmol Vis Sci. 2004 Jun;45(6):1758-62. PMID: 15161837. [CrossRef]
- Liu J, Yang L, Liu K and Gao F (2023) Hydrogel scaffolds in bone regeneration: Their promising roles in angiogenesis. Front. Pharmacol. 14:1050954. [CrossRef]
- Hodde, J. P. Vascular endothelial growth factor in porcine-derived extracellular matrix/J. P. Hodde, R. D. Record, H. A. Liang, S. F. Badylak//Endothelium.—2001.—№ 8 (1).—P. 10–15.
- Gilbert, T. W. Decellularization of tissues and organs/T. W. Gilbert, T. L. Sellaro, S. F. Badylak//Biomaterials.—2006.—№ 27 (19). – P. 3532–3537.
- Niknejad H, Peirovi H, Jorjani M, Ahmadiani A, Ghanavi J, Seifalian AM. Properties of the amniotic membrane for potential use in tissue engineering. Eur Cell Mater. 2008 Apr 29;15:88-99. PMID: 18446690. [CrossRef]
- Portmann-Lanz CB, Ochsenbein-Kölble N, Marquardt K, Lüthi U, Zisch A, Zimmermann R. Manufacture of a cell-free amnion matrix scaffold that supports amnion cell outgrowth in vitro. Placenta. 2007 Jan;28(1):6-13. Epub 2006 Mar 3. PMID: 16516964. [CrossRef]
- Munoz-Torres JR, Martínez-González SB, Lozano-Luján AD, Martínez-Vázquez MC, Velasco-Elizondo P, Garza-Veloz I, et al. Biological properties and surgical applications of the human amniotic membrane. Front Bioeng Biotechnol. (2023) 10:1–20. [CrossRef]
- Koizumi NJ, Inatomi TJ, Sotozono CJ, Fullwood NJ, Quantock AJ, Kinoshita S. Growth factor mRNA and protein in preserved human amniotic membrane. Curr Eye Res. 2000 Mar;20(3):173-7. PMID: 10694891.
- Roy I, Gupta MN. Freeze-drying of proteins: some emerging concerns. Biotechnol Appl Biochem. 2004 Apr;39(Pt 2):165-77. PMID: 15032737. [CrossRef]
- López-Valladares MJ, Touriño R, Vieites B, Gude F, Silva MT, Couceiro J. Effects of lyophilization on human amniotic membrane. Acta Ophthalmol. 2009 Jun;87(4):396-403. Epub 2008 Oct 15. PMID: 18937812. [CrossRef]
- Hopkinson A, McIntosh RS, Shanmuganathan V, Tighe PJ, Dua HS. Proteomic analysis of amniotic membrane prepared for human transplantation: characterization of proteins and clinical implications. J Proteome Res. 2006 Sep;5(9):2226-35. PMID: 16944934. [CrossRef]
- Milyudin, E.; Volova, L.T.; Kuchuk, K.E.; Timchenko, E.V.; Timchenko, P.E. Amniotic Membrane Biopolymer for Regenerative Medicine. Polymers 2023, 15, 1213. [CrossRef]
- Volova L.T., Milyudin E.S., Kuchuk K.E. Method for preparing allogeneic amniotic membrane for reconstructive medicine. Russian Federation Patent No. 2835347.
- Andri K. Riau, Roger W. Beuerman, Laurence S. Lim, Jodhbir S. Mehta Preservation, sterilization and de-epithelialization of human amniotic membrane for use in ocular surface reconstruction Biomaterials, Volume 31, Issue 2,2010,Pages 216-225,ISSN 0142-9612. (https://www.sciencedirect.com/science/article/pii/S0142961209009648). [CrossRef]
- Ingraldi, A.L.; Audet, R.G.; Tabor, A.J. The Preparation and Clinical Efficacy of Amnion-Derived Membranes: A Review. J. Funct. Biomater. 2023, 14, 531. [CrossRef]
- Redl, H., Wolbank, S., & Hennerbichler, S. (2009). Impact of human amniotic membrane preparation on release of angiogenic factors. J Tissue Eng Regen Med 2009; 3: 651–654. [CrossRef]
- Bukhdruker S, Varaksa T, Orekhov P, Grabovec I, Marin E, Kapranov I, Kovalev K, Astashkin R, Kaluzhskiy L, Ivanov A, Mishin A, Rogachev A, Gordeliy V, Gilep A, Strushkevich N, Borshchevskiy V. Structural insights into the effects of glycerol on ligand binding to cytochrome P450. Acta Crystallogr D Struct Biol. 2023 Jan 1;79(Pt 1):66-77. Epub 2023 Jan 1. PMID: 36601808. [CrossRef]
- Zahn-Zabal M, Michel PA, Gateau A, Nikitin F, Schaeffer M, Audot E, Gaudet P, Duek Roggli P, Teixeira D, Rech de Laval V, Samarasinghe K, Bairoch A, Lane L. The neXtProt knowledgebase in 2020: data, tools and usability improvements. Nucleic Acids Res. 2020 Jan 8;48 (D1):D328-D334.
- 27. The UniProt Consortium UniProt: the Universal Protein Knowledgebase in 2023 Nucleic Acids Res. 51:D523–D531 (2023).
- The Gene Ontology Consortium. The Gene Ontology knowledgebase in 2023. Genetics. 2023 May 4;224 (1):iyad031. [CrossRef]
- Zhang F, Zhang L, Zhang B, Wei X, Yang Y, Qi RZ, Ying G, Zhang N, Niu R. Anxa2 plays a critical role in enhanced invasiveness of the multidrug resistant human breast cancer cells. J Proteome Res. 2009 Nov;8(11):5041-7. PMID: 19764771. [CrossRef]
- Sharma MR, Koltowski L, Ownbey RT, Tuszynski GP, Sharma MC. Angiogenesis-associated protein annexin II in breast cancer: selective expression in invasive breast cancer and contribution to tumor invasion and progression. Exp Mol Pathol. 2006 Oct;81(2):146-56. Epub 2006 Apr 27. PMID: 16643892. [CrossRef]
- Sharma M, Ownbey RT, Sharma MC. Breast cancer cell surface annexin II induces cell migration and neoangiogenesis via tPA dependent plasmin generation. Exp Mol Pathol. 2010 Apr;88(2):278-86. Epub 2010 Jan 15. PMID: 20079732. [CrossRef]
- Zhang J, Guo B, Zhang Y, Cao J, Chen T. Silencing of the annexin II gene down-regulates the levels of S100A10, c-Myc, and plasmin and inhibits breast cancer cell proliferation and invasion. Saudi Med J. 2010 Apr;31(4):374-81. PMID: 20383413.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



