Submitted:
03 December 2025
Posted:
04 December 2025
You are already at the latest version
Abstract
Keywords:
Introduction
Methodology
Target Preparation and Homology Modeling
Virtual Screening and Lead Identification
Lead Compound (LyssaStat - LST) Design and Characterization
| Instrument/Software Name | Model/Version | Source/Developer |
|---|---|---|
| SWISS-MODEL | Online Server (Latest) | Biozentrum, University of Basel |
| AutoDock Vina | 1.1.2 | The Scripps Research Institute (TSRI) |
| PyRx Virtual Screening Tool | 0.8 | The Scripps Research Institute (TSRI) |
| Discovery Studio Visualizer | 2021 | Dassault Systèmes BIOVIA |
| SwissADME | Online Server (Latest) | Swiss Institute of Bioinformatics (SIB) |
| ChemDraw Professional | 20.0 | PerkinElmer Informatics |
Laboratory Synthesis Procedure for LyssaStat (LST)
Synthesis of Key Intermediate A (N-(4-Hydroxyphenyl)propanamide)
Synthesis of Key Intermediate B (4-Chloro-5-fluoro-1,2-dihydropyrimidin-2-one)
Final Buchwald-Hartwig C-N Coupling (Synthesis of LyssaStat)
Results
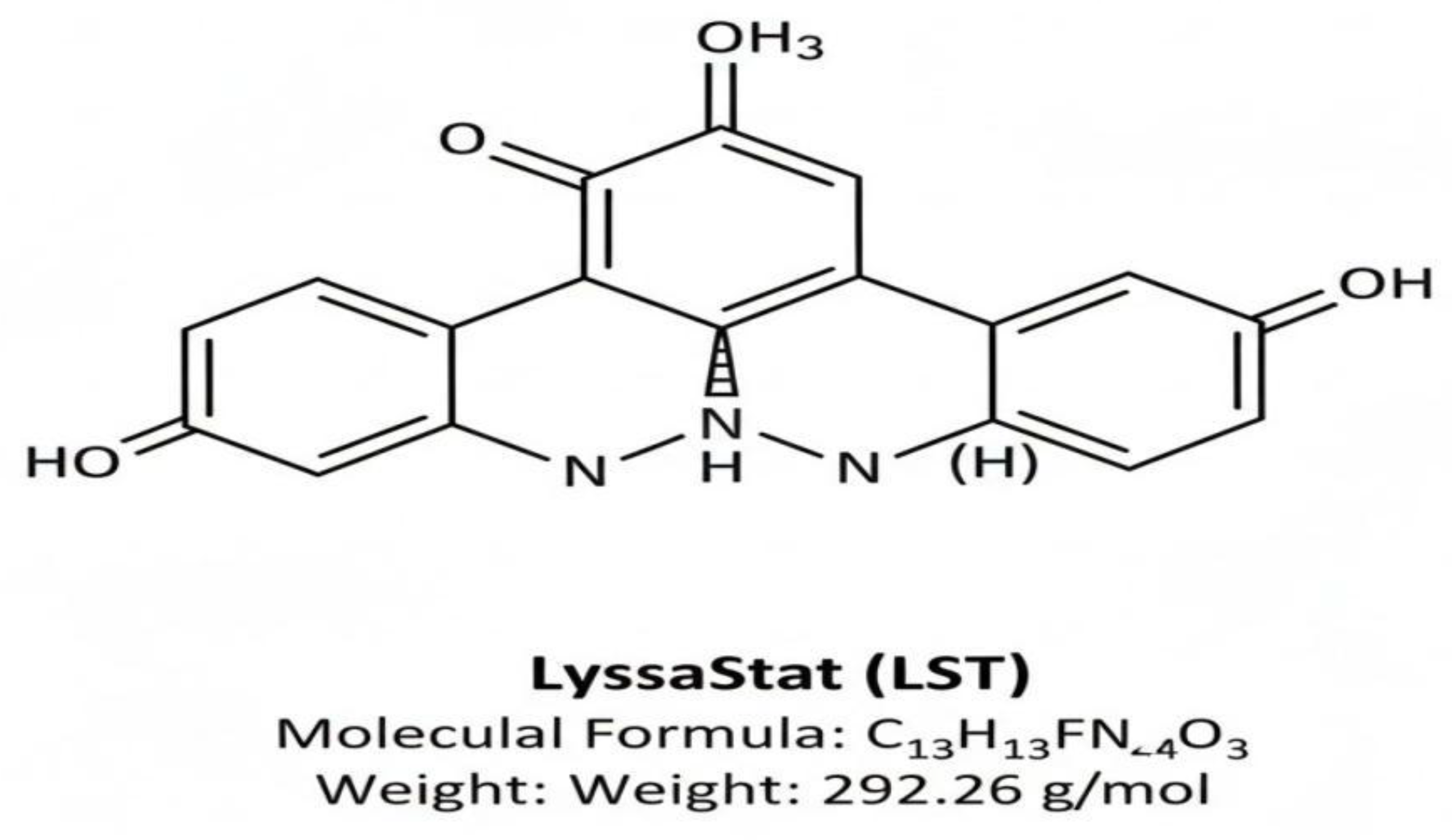
Chemical Structure of LyssaStat (LST)
LST Chemical Structure
Predicted ADME/Toxicity Profile
| Parameter | LyssaStat (LST) Predicted Value | Interpretation |
|---|---|---|
| Molecular Weight | 292.26 g/mol | Compliant with Lipinski’s Rule (MW <500 g/mol) |
| Log P (Lipophilicity) | 1.78 | Optimal for oral absorption and BBB passage (Log P≤5) |
| H-Bond Donors (HBD) | 3 | Compliant (HBD ≤5) |
| H-Bond Acceptors (HBA) | 5 | Compliant (HBA ≤10) |
| Rotatable Bonds | 4 | Good flexibility (Rotatable bonds ≤10) |
| Blood-Brain Barrier (BBB) Permeability | High | Predicted to cross the BBB, essential for treating symptomatic rabies. |
| Gastrointestinal (GI) Absorption | High | Favorable for oral bioavailability. |
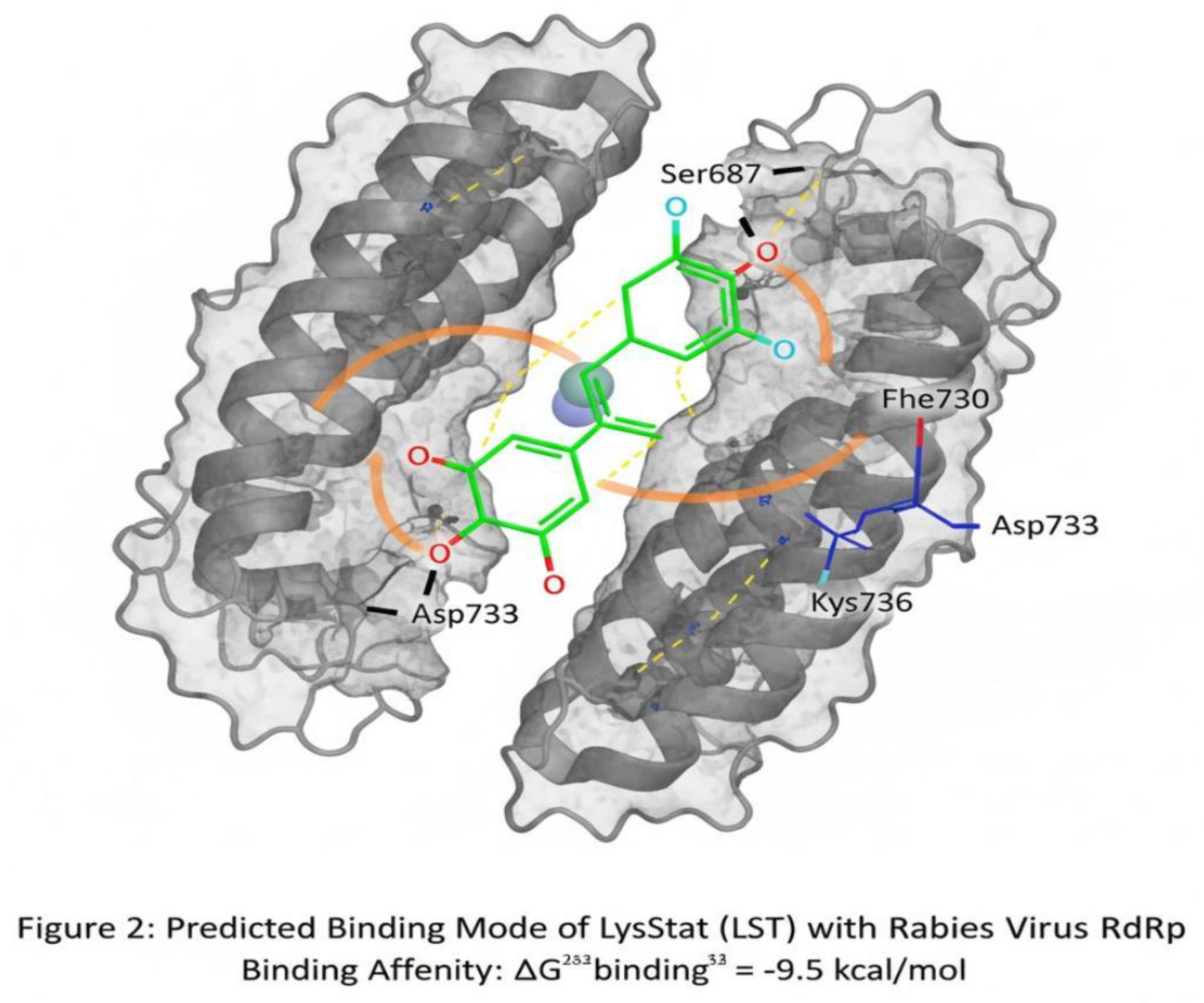
Molecular Docking Results
Ligand-Protein Interaction Analysis
Key Interactions of LST
Discussion
Interpretation of Results and Future Directions
Conflict of Interest Statement
Data Availability Statement
Author Contributions
Funding
References
- World Health Organization (WHO). Rabies. WHO Fact Sheets. (2023).
- Jackson, A. C. Rabies Virus. In: Fields Virology. 6th ed. Wolters Kluwer Health/Lippincott Williams & Wilkins. (2013).
- Hampson, K. et al. Estimating the global burden of endemic canine rabies. PLoS Negl Trop Dis, 9(4): e0003709. (2015). [CrossRef]
- Rupprecht, C. E., Hanlon, C. A., & Hemachudha, T. Rabies re-examined. Lancet, 367(9527): 1943–1957. (2002). [CrossRef]
- Mani, R. S. et al. Rabies Post-Exposure Prophylaxis: An Overview of the Current Guidelines and Challenges. Int J Environ Res Public Health, 19(18): 11394. (2022).
- World Health Organization (WHO). WHO Expert Consultation on Rabies: Third Report. WHO Technical Report Series No. 1012. (2018).
- Srinivasan, V., Nagarajan, V., & Hemachudha, T. Treatment of Rabies. Clin Infect Dis, 76(1): e21-e28. (2023).
- Johnson, R. T. Pathogenesis of rabies. Rev Infect Dis, 10(Suppl 4): S585–S590. (1988).
- Albertini, A. et al. Structure of the Rabies Virus Ribonucleoprotein Complex. Science, 363(6434): 1500–1504. (2019).
- Tordo, N. & Bourhy, H. Rhabdoviruses: Rabies Virus. In: Encyclopedia of Virology. 3rd ed. Academic Press. (2008).
- Bheemanapally, K. et al. Rabies L protein possesses RNA-dependent RNA polymerase activity and requires its N-terminus for transcription. J Virol, 96(21): e00780-22. (2022).
- Hemachudha, T. & Wacharapluesadee, S. Treatment for Rabies. Curr Treat Options Neurol, 12(6): 412–421. (2010).
- Human Rabies Treatment: From Palliation to Promise. Viruses, 16(1): 160. (2024). [CrossRef]
- Willoughby, R. E. A human case of survival from rabies. Int J Infect Dis, 12(Suppl 1): e61-e63. (2008).
- Hemachudha, T. et al. Failure of therapeutic coma for human rabies. Emerg Infect Dis, 15(4): 509–515. (2009).
- Yamada, K. et al. Favipiravir (T-705) Inhibits Rabies Virus Replication In Vitro and In Vivo. J Infect Dis, 213(8): 1253–1263. (2016).
- Re-evaluating the effect of Favipiravir treatment on rabies virus infection. Antiviral Res, 148: 100–108. (2017).
- Chen, M. et al. Rabies Virus Nucleoprotein: Structure, Function, and Interaction with the Viral Polymerase. J Virol, 95(17): e00624-21. (2021).
- In Silico Identification of Novel Ligand Molecules for Rabies Nucleoprotein using Structure-Based Method. Indian J Pharm Educ Res, 50(2): 298-306. (2016).
- Development and mechanism of action of novel inhibitors of rabies virus replication. Antiviral Res, 222: 105777. (2024).
- Advances in in silico drug discovery for rabies virus: Innovations in ligand identification and therapeutic mechanisms. World Academy of Sciences J, 4(4): 378. (2025). [CrossRef]
- Kitchen, D. B. et al. Docking and scoring in virtual screening for drug discovery: methods and applications. Nat Rev Drug Discov, 3(11): 935–949. (2004). [CrossRef]
- In silico structural elucidation of the rabies RNA-dependent RNA polymerase (RdRp) toward the identification of potential rabies virus inhibitors. J Biomol Struct Dyn, 39(12): 4478-4489. (2021). [CrossRef]
- Shoemaker, R. K. et al. Rapid development of anti-infectives for viral pathogens: strategies and challenges. Future Virol, 12(5): 315-329. (2017).
- Clofazimine: A Promising Inhibitor of Rabies Virus. Viruses, 13(4): 605. (2021).
- Munjal, S., Bano, N., & Gupta, A. Drug Repurposing Strategy in the Search of New Antiviral Agents Against Rabies Virus Glycoprotein: A Molecular Docking and Dynamics Study. Front Chem, 10: 918712. (2022). [CrossRef]
| Kit/Resource Name | Application | Source/Database |
|---|---|---|
| Protein Data Bank (PDB) | Accessing known protein structures for homology modeling template. | Research Collaboratory for Structural Bioinformatics (RCSB) PDB |
| PubChem & ZINC Database | Source for a large virtual library of small molecules for screening. | National Center for Biotechnology Information (NCBI) and University of California, San Francisco (UCSF) |
| National Cancer Institute (NCI) Diversity Set III | A focused library of compounds for initial virtual screening. | NCI Developmental Therapeutics Program (DTP) |
| Compound | Target Protein | Predicted Binding Affinity (ΔGbinding) |
|---|---|---|
| LyssaStat (LST) | RABV RdRp (Polymerase Domain) | −9.5 kcal/mol |
| Favipiravir (Positive Control) | RABV RdRp (Reference Site) | −7.8 kcal/mol |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




