1. Introduction
Acute pancreatitis is a rapidly increasing cause of gastrointestinal hospitalization, with more than 275,000 admissions annually in the United States alone [
2]. The global incidence has also risen steadily over the past two decades, driven largely by changing metabolic risk factors, increasing alcohol consumption, and improved diagnostic sensitivity. Although the majority of cases resolve within 3–5 days, 15–20% of patients progress to moderately severe or severe disease characterized by pancreatic necrosis, organ failure, and systemic inflammatory response syndrome (SIRS) [
1,
3].
Despite advances in supportive care, mortality from AP remains significant. Overall mortality ranges between 3–5%
, but this increases dramatically to 20–35% in patients who develop persistent organ failure (>48 h)
, most commonly affecting the respiratory, cardiovascular, or renal systems [
7,
28]. Persistent organ failure is consistently identified as the strongest independent predictor of death.
Risk stratification at the time of admission remains challenging. Traditional prognostic tools—including APACHE II
, BISAP
, Ranson’s criteria, and the modified CT severity index (mCTSI)—provide useful information but have notable limitations within the first 24 hours of illness [
4,
11,
21]. During this critical early window, clinical deterioration often precedes radiological progression, and static clinical scores lack the sensitivity to reliably identify high-risk patients. As a result, there is a growing emphasis on dynamic biomarkers and physiological indicators capable of capturing early inflammatory escalation, endothelial dysfunction, and microcirculatory failure.
A rapidly expanding body of evidence highlights the role of inflammatory cytokines, macrophage-activation markers, renal injury biomarkers and endothelial injury mediators in predicting disease progression far earlier than conventional tools. In parallel, bedside intensive care parameters—such as vasopressor requirement, lactate trends, and declining Horowitz index—provide real-time physiological insight that reflects the integrated systemic impact of the disease.
The integration of inflammatory, endothelial, oxidative, and renal biomarkers with robust ICU physiological indicators represents a modern precision-medicine approach to early managementin AP. This multimodal strategy allows for more accurate early triage, timely ICU admission, optimized fluid and vasopressor therapy, and judicious use of organ-support modalities such as continuous renal replacement therapy. Ultimately, a more refined early evaluation may contribute to reducing morbidity, shortening ICU stay, and improving survival in severe acute pancreatitis.
2. Etiology
The etiological spectrum of acute pancreatitis varies across populations, but several major categories account for the majority of cases. Biliary pancreatitis remains the leading cause, responsible for approximately 35–45% of all AP episodes, particularly in older adults and women [
1,
21]. Alcohol-induced pancreatitis represents the second most common etiology, contributing 25–35% of cases and typically associated with recurrent pancreatic injury in younger male patients [
22].
Hypertriglyceridemia-related AP accounts for 5–10% of all presentations but may reach 15–20% among patients with severe necrotizing disease, driven by lipotoxicity and enhanced systemic inflammation [
23]. Drug-induced pancreatitis is less frequent (1–2%), with azathioprine, valproate, corticosteroids, and certain statins being the most commonly implicated agents [
24]. Post-ERCP pancreatitis occurs in 3–5% of standard-risk procedures but may rise to 10–15% in high-risk interventions, such as difficult cannulation or sphincter of Oddi dysfunction [
25].
A smaller proportion (~1%) is attributable to pancreatic neoplasms, particularly ductal or periampullary tumors causing ductal obstruction [
26]. Despite comprehensive evaluation, idiopathic acute pancreatitis still accounts for 15–20% of cases; however, the true rate decreases to 5–10% following advanced imaging such as MRCP or EUS [
23].
3. Severity (Revised Atlanta Classification, 2012)
Based on Banks et al. [
1] and Singh et al. [
15]:
Mild AP
Moderately Severe AP
Severe AP
Organ failure definitions:
Persistent organ failure is the strongest predictor of death.
4. Biomarkers in Acute Pancreatitis
4.1. Classical Biomarkers
Classical Biomarkers (Kibővítve: Szenzitivitás + Specificitás)
CRP rises late—typically 48–72 hours after symptom onset—therefore its early prognostic value is limited [
30].
t the 48-hour mark, CRP >150 mg/L has a sensitivity of ~70–80% and specificity of ~65–75% for predicting pancreatic necrosis, but is not suitable for early (0–24 h) stratification.
PCT is superior to CRP for detecting infected pancreatic necrosis and for differentiating sterile vs. infected collections [
18].
Based on meta-analyses, its sensitivity is ~80–85% and its specificity is ~75–85% for detecting infection, especially above a cut-off of 0.5–1.0 ng/mL.
These enzymes are highly sensitive for diagnosis.
Lipase szenzitivitás: ~90–100%, specificitás: ~95%
Amiláz szenzitivitás: ~70–85%, specificitás: ~60–70%
However, despite excellent diagnostic utility, enzyme levels do not correlate with disease severity, presence of necrosis, organ failure, or clinical outcomes [
30].
4.2. Early Inflammatory Biomarkers
4.3. Macrophage Activation Markers
Pentraxin-3 (PTX3): predicts necrosis and severity [
8]
sCD163: reflects macrophage-mediated organ injury [
7]
4.4. Early Kidney Injury Biomarkers
NGAL: rises 6–12 hours before creatinine, predicts AKI and RRT [
10]
4.5. Infection Biomarkers
Presepsin (sCD14-ST) is a soluble fragment of the CD14 receptor released during monocyte–macrophage activation, particularly in response to bacterial components such as lipopolysaccharide. Although more widely validated in sepsis, presepsin has recently emerged as a promising early biomarker in acute pancreatitis, especially for distinguishing sterile versus infected pancreatic necrosis.
Several studies demonstrate that presepsin levels rise significantly earlier than CRP or PCT, often within the first 6–12 hours of clinical deterioration, providing a markedly earlier indication of bacterial translocation and infected necrosis. In preliminary cohorts, presepsin showed higher sensitivity (~80–90%) than CRP and comparable or superior specificity to PCT for identifying infected collections, particularly at cut-off values 300–500 pg/mL.
The marker’s rapid kinetics, short half-life, and tight correlation with systemic bacterial burden make it a valuable adjunct in early triage and antibiotic decision-making in severe AP, although large multicenter validation studies are still lacking. Nevertheless, presepsin may complement established markers (CRP, PCT, IL-6) in predicting secondary infection and guiding early intervention in necrotizing pancreatitis.
4.6. Endothelial Dysfunction Markers
Angiopoietin-2: predicts organ failure and shock [
19]
Endocan, proADM, VEGF: reflect endothelial collapse and microcirculatory injury [
12,
13,
14]
4.7. Oxidative Stress Markers
Oxidative Stress Markers
Oxidative stress plays a central role in the pathophysiology of acute pancreatitis, particularly in the progression toward necrosis, endothelial dysfunction, and multi-organ failure. Several circulating markers reflect distinct aspects of redox imbalance and microvascular injury.
Malondialdehyde (MDA)
MDA is one of the most widely studied end-products of lipid peroxidation. Elevated MDA concentrations correlate strongly with the extent of pancreatic necrosis and the severity of systemic inflammation. High early MDA levels are associated with increased risk of ARDS, microcirculatory failure, and ICU admission.
MDA is often elevated within the first 12–24 hours, making it a useful early indicator of severe disease.
Glutathione depletion (GSH)
Reduced glutathione is a major intracellular antioxidant. In acute pancreatitis, rapid GSH depletion reflects overwhelming oxidative load and impaired cellular detoxification. Low GSH levels correlate with mitochondrial dysfunction, endothelial injury, and higher likelihood of persistent organ failure.
In clinical cohorts, severe AP frequently presents with significantly lower GSH/GSSG ratios, a marker of redox-collapse associated with ARDS and renal injury.
Nitric oxide / Asymmetric dimethylarginine (NO/ADMA) imbalance
NO is a key regulator of microvascular perfusion. In severe AP, excess NO from inducible nitric oxide synthase (iNOS) contributes to vasoplegia and capillary leak, while ADMA—a natural NOS inhibitor—may increase simultaneously, producing a paradoxical dysregulation.
A high NO/ADMA imbalance is linked with refractory shock, deterioration of the Horowitz-index, and progression toward multiorgan dysfunction.
Total oxidant status / Total antioxidant capacity (TOS/TAC ratio)
The TOS/TAC ratio provides an integrated measure of systemic oxidative load. Elevated ratios indicate dominant oxidative stress and diminished compensatory antioxidant response.
High TOS/TAC values have been associated with:
Because it integrates multiple oxidative pathways, the TOS/TAC ratio is considered one of the more robust global indicators of redox imbalance in AP.
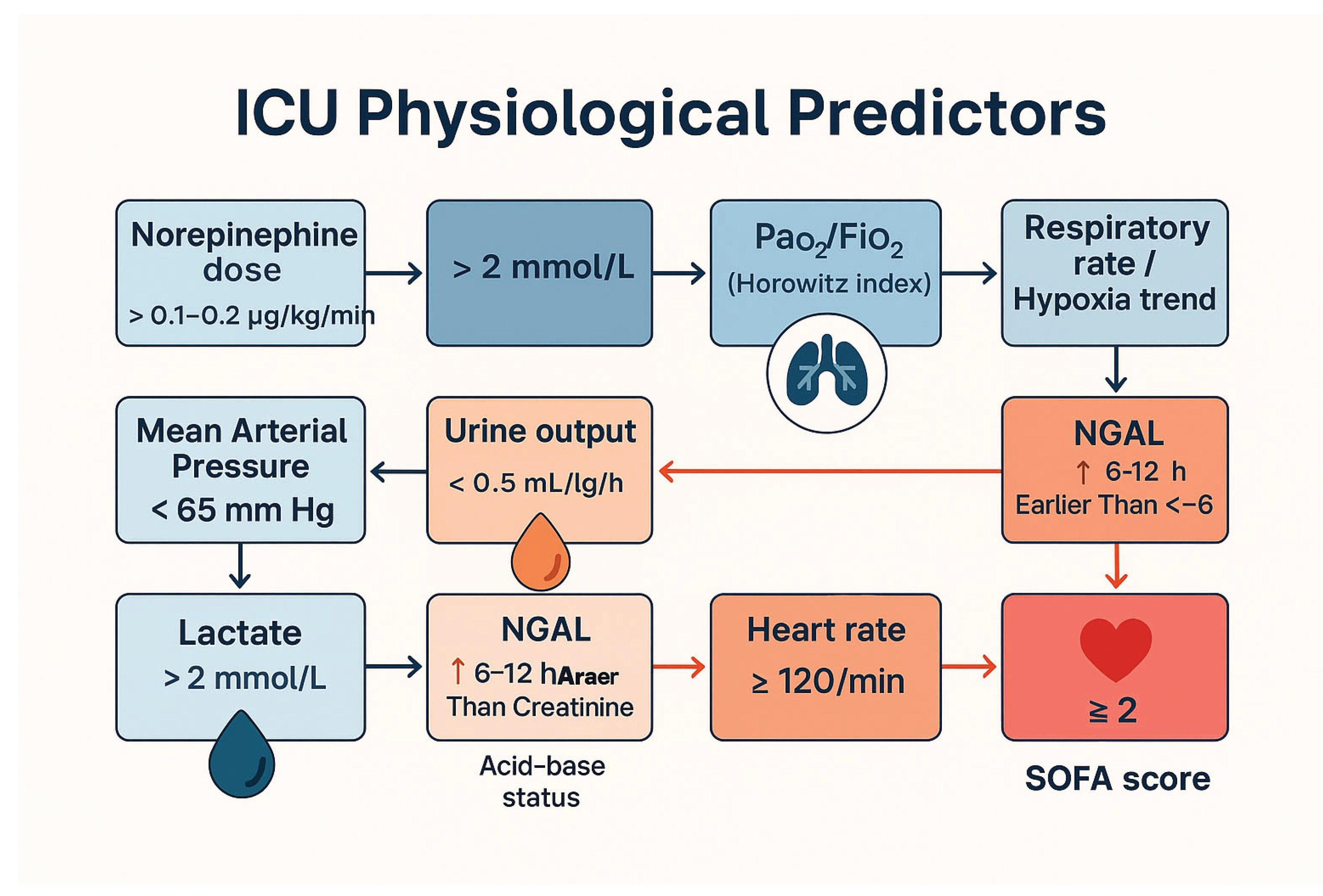
5. ICU Physiological Predictors
This figure summarizes the most powerful bedside predictors of early organ failure in acute pancreatitis. Elevated norepinephrine requirements (>0.1–0.2 μg/kg/min), mean arterial pressure <65 mmHg, lactate >2 mmol/L, progressive hypoxia or P/F ratio decline, respiratory rate elevation, early NGAL rise (6–12 h before creatinine), urine output <0.5 mL/kg/h, heart rate ≥120/min, and SOFA score ≥2 collectively indicate worsening systemic inflammation, microcirculatory collapse, and high mortality risk. These parameters integrate hemodynamic, respiratory, renal, and metabolic deterioration relevant for ICU triage.
Figure 1.
ICU Physiological Predictors of Severe Acute Pancreatitis.
Figure 1.
ICU Physiological Predictors of Severe Acute Pancreatitis.
Table 1.
ICU Physiological Predictors.
Table 1.
ICU Physiological Predictors.
| Predictor |
Cutoff érték |
Clinical significance / Prognostic value |
| Norepinephrine-dózis |
> 0.1–0.2 µg/kg/min |
Severe vasoplegia, risk of circulatory collapse; high mortality. Indicates early shock persistence.
. |
| Mean Arterial Pressure (MAP) |
<65 mmHg (adequate fluid után) |
Inadequate perfusion → aggressive hemodynamic support is required. |
| Lactate |
> 2 mmol/L |
Korai szöveti hypoperfusio, fokozott SIRS. |
| |
> 4 mmol/L |
Very poor prognosis, severe shock; increased ICU mortality |
| PaO₂/FiO₂ (Horowitz-index) |
< 300 |
Early respiratory failure. |
| |
< 150 |
ARDS is likely to develop; mechanical ventilation is often necessary. |
| Respiratory rate / hypoxia trend |
RR > 30/min, SpO₂ < 92% |
Increased respiratory effort, impending respiratory failure. |
| Urine output |
< 0.5 mL/kg/h |
|
| Creatinine / NGAL |
NGAL emelkedés 6–12 órával a kreatinin előtt |
Early AKI, perfusion problems, volume replacement or support required. |
| Base deficit / pH |
pH < 7.30, BE < –6 |
|
| Heart rate |
> 120/min |
Subclinical AKI, early indication for RRT; better predictor than creatinine. |
| mCTSI |
≥ 6–10 |
|
| SOFA score |
≥ 2 emelkedés |
Signs of severe acidosis, shock, or respiratory failure. |
Physiological parameters measured at bedside remain some of the strongest and most reliable predictors of early deterioration in acute pancreatitis. Unlike static scoring systems, these dynamic indicators reflect real-time interactions between systemic inflammation, microcirculatory failure, and organ dysfunction. When integrated with biomarkers, they allow far more accurate early risk stratification.
5.1. Hemodynamic Predictors
Hemodynamic instability is an early hallmark of severe AP.
Norepinephrine requirement >0.1–0.2 μg/kg/min is strongly associated with severe vasoplegia, capillary leak, and cytokine-driven shock [
7].
A MAP <65 mmHg despite adequate fluid resuscitation predicts persistent organ failure and the need for early ICU admission.
Tachycardia (>120/min) combined with rising lactate levels reflects progressive hypoperfusion. Hemodynamic instability correlates closely with early multi-organ dysfunction and overall mortality.
5.2. Lactate Dynamics
Serum lactate is one of the most powerful bedside indicators of perfusion and metabolic distress:
Lactate >2 mmol/L indicates early hypoperfusion.
Lactate >4 mmol/L is associated with very poor prognosis, prolonged ICU stay, and increased mortality [
17].
A rising lactate trajectory despite fluids strongly suggests microcirculatory collapse and should prompt escalation of care.
Lactate clearance within the first 6–12 hours is associated with improved outcomes.
5.3. Respiratory Predictors (Horowitz Index / P/F Ratio)
Respiratory compromise is common, driven by SIRS-related capillary leak, ARDS, or abdominal compartment syndrome.
PaO₂/FiO₂ <300 indicates early respiratory failure.
PaO₂/FiO₂ <150 predicts ARDS, need for mechanical ventilation, prolonged ICU stay, and higher mortality [
18].
Progressive tachypnea or need for high-flow nasal oxygen suggests worsening lung injury and systemic inflammation.
The Horowitz index integrates both oxygenation and ventilatory drive, offering strong prognostic value.
5.4. Renal Predictors
Acute kidney injury (AKI) is one of the most powerful independent predictors of mortality in acute pancreatitis.
Oliguria <0.5 mL/kg/h after adequate resuscitation predicts persistent organ failure.
Rising creatinine is a late marker, while NGAL rises 6–12 hours earlier, detecting subclinical kidney injury and predicting the need for CRRT [
10].
AKI often indicates widespread endothelial injury, microvascular shutdown, and severe systemic inflammation.
Early renal dysfunction is a key trigger for ICU admission and organ support.
5.5. Scoring and Imaging Correlates
Although not strictly “physiological,” certain combined parameters correlate with ICU deterioration:
mCTSI ≥6 correlates with necrosis, organ failure, and ICU requirement [
19].
Persistent SIRS >48 h is one of the strongest predictors of severe AP.
Bedside ultrasonography for effusions, ascites and pleural fluid helps track fluid overload and abdominal compartment risk.
ICU Predictors – Summary Sentence (beilleszthető a végére)
Vasopressor need, rising lactate, worsening P/F ratio, and early kidney injury represent some of the strongest independent predictors of morbidity and mortality in acute pancreatitis and should serve as primary triggers for early ICU referral and organ-support planning.
6. Fluid Resuscitation in Acute Pancreatitis
Modern fluid therapy is not aggressive, but rather purposeful, controlled resuscitation.
6.1. What Fluid Should Be Administered?
Ringer’s lactate (RL) → first choice [
18].
Better acid-base profile, reduces inflammation.
0.9% NaCl, which causes hyperchloremic acidosis, should be avoided.
6.2. How Much Should Be Administered?
Initial bolus: 10–20 mL/kg RL (if signs of hypovolemia are present)
Maintenance rate: 1.5 mL/kg/h (≈ 100–150 mL/h)
In more severe cases: 2–3 mL/kg/h for a short period
6.3. Target Parameters
MAP ≥ 65 Hgmm
Urine >0.5 mL/kg/h
Lactate decreases
Ht 35–44%
BUN does not increase
6.4. Kolloidok Szerepe
Albumin 5%: csak refrakter hypotensio vagy nagy kapilláris leak esetén
HES → kontraindikált (AKI, mortality growth)
6.5. What Should Be Avoided?
-
Over-resuscitation →
- ○
ARDS
- ○
abdominal compartment syndrome
- ○
increased mortality
- ○
Modern guidelines recommend fluid therapy combined with
controlled, frequent reassessment [
4].
7. Continuous Renal Replacement Therapy (CRRT)
7.1. When Should We Start?
Indications for CRRT in AP:
7.2. Advantages
Although CRRT reduces circulating pro-inflammatory cytokines and improves metabolic control in acute pancreatitis, no multicenter randomized controlled trial has demonstrated a reduction in mortality or ICU/hospital length of stay. Therefore, its use for immunomodulation remains promising but unproven, and further large, well-designed multicenter RCTs are required to clarify its true clinical impact.]
7.3. Which Method?
CVVH (hemofiltration) → primary
CVVHDF → acidosis control
Effluent: 25–35 mL/kg/h
No proven benefit above 45 mL/kg/h
8. Discussion
Acute pancreatitis displays significant heterogeneity in clinical severity, ranging from mild, self-limiting inflammation to fulminant multi-organ failure requiring prolonged ICU support. Early and precise identification of patients at risk for deterioration remains one of the greatest challenges in clinical practice.
Traditional scores such as APACHE II, BISAP, and the modified CT severity index provide valuable information but lack discriminatory power during the first 24 hours after admission. This early time window is critical, as organ failure often evolves rapidly, and delayed recognition directly impacts outcomes.
Emerging biomarkers—including IL-6, IL-8, IL-10, pentraxin-3, sCD163, NGAL, and presepsin—offer a more detailed understanding of the inflammatory and immunological dynamics of AP. IL-6 is particularly powerful due to its early rise, strong correlation with persistent organ failure, and ability to outperform CRP and PCT in the first hours of disease progression. NGAL allows truly early identification of renal injury, supporting timely CRRT initiation where appropriate.
Endothelial injury markers such as angiopoietin-2, endocan and pro-ADM play an increasingly recognized role. Endothelial dysfunction, capillary leak and microcirculatory collapse form the pathophysiological basis of early shock and ARDS in AP, making these markers crucial for triage and risk assessment.
Physiological ICU parameters remain the most reliable bedside indicators. Vasopressor requirement, lactate dynamics, and P/F ratio deterioration correlate strongly with early mortality. These markers reflect the integrated physiological effect of the systemic inflammatory response and complement the biomarker-based approach.
Fluid management has undergone a paradigm shift. Historical “aggressive resuscitation” has been replaced by controlled, goal-directed fluid therapy, using balanced crystalloids and avoiding hypervolemia. Early identification of patients who will benefit from albumin supplementation—particularly those with marked hypoalbuminemia or significant capillary leak—is important.
CRRT remains indispensable in AP complicated by AKI, severe acid–base disturbances, and refractory fluid overload. However, despite promising results from smaller studies showing cytokine reduction, no multicenter randomized controlled trial has demonstrated improved survival or reduced ICU length of stay, highlighting the need for further high-quality research.
A multimodal approach that integrates inflammatory, endothelial and oxidative biomarkers with ICU physiological indicators provides the most accurate early prognostic assessment and supports a precision-based treatment strategy. This may lead to earlier ICU admission, optimized fluid and vasopressor therapy, and more rational use of CRRT.
9. Conclusions
Acute pancreatitis requires early, accurate risk stratification to prevent progression toward persistent organ failure and its associated high mortality. Integrating inflammatory, endothelial, oxidative, and kidney biomarkers with ICU physiological predictors provides a modern, precision-based framework for early severity prediction. This multimodal approach captures complementary aspects of disease biology—from cytokine-driven inflammation and endothelial dysfunction to microcirculatory collapse and early organ injury—offering substantially greater predictive accuracy than any single biomarker or scoring system alone.
Controlled, goal-directed fluid resuscitation is essential to avoid both under- and over-resuscitation, each of which significantly worsens outcomes. Early recognition of patients who may benefit from albumin supplementation, closer hemodynamic monitoring or timely ICU transfer can further improve clinical trajectories. Continuous renal replacement therapy plays an indispensable supportive role in the presence of acute kidney injury, severe acidosis, or fluid overload; however, its use for immunomodulation remains promising but unproven, underscoring the need for future multicenter randomized trials.
Ultimately, a structured early evaluation that unites biomarkers, bedside ICU physiology, and contemporary management principles enables more accurate triage, earlier intervention, and more rational allocation of intensive care resources. Such a strategy supports a shift toward individualized, precision-guided treatment and may contribute to meaningful reductions in morbidity and mortality in severe acute pancreatitis
Finally, postpartum and post-cesarean acute pancreatitis, although rare, illustrates the importance of maintaining a high index of suspicion in special patient populations, as highlighted by the recent case published by Tóth and colleagues [
43].
Ethics Statement
No human subjects, identifiable data, or patient interventions were involved in this narrative review. Ethical approval was not required.
AI Declaration
During the preparation of this work, the authors used ChatGPT to assist in organization, language refinement, and technical editing. After using this tool, the authors reviewed and edited the content as needed and take full responsibility for its content.
Funding
No external funding was received.
Conflicts of Interest
The authors declare no conflicts of interest.
Data Availability
No new data were generated or analyzed in this study.
References
- Banks PA, Bollen TL, Dervenis C, et al. Classification of acute pancreatitis—2012: Revision of the Atlanta classification. Gut. 2013;62:102–111. [CrossRef]
- Tenner S, Baillie J, DeWitt J, Vege SS. ACG guideline: Management of acute pancreatitis. Am J Gastroenterol. 2013;108:1400–1415. [CrossRef]
- Working Group IAP/APA. IAP/APA evidence-based guidelines for acute pancreatitis. Pancreatology. 2013;13:e1–15. [CrossRef]
- Mofidi R, Duff MD, Wigmore SJ, et al. Early SIRS and severity in AP. Br J Surg. 2006;93:738–744. [CrossRef]
- Cardoso FS, Oliveira AM, et al. CRP and NLR for prediction in AP. Pancreatology. 2020;20:779–785. [CrossRef]
- Rau BM, Kemppainen EA, et al. Procalcitonin predicts infected necrosis. Clin Gastroenterol Hepatol. 2007;5:231–236. [CrossRef]
- Kylanpaa ML, Repo H, Puolakkainen P. Inflammation biomarkers. Acta Anaesthesiol Scand. 2010;54:110–118. [CrossRef]
- Párniczky A, Kui B, Szentesi A, et al. Hungarian AP Registry. Pancreatology. 2016;16:146–150. [CrossRef]
- Staubli SM, Oertli D, Nebiker CA. IL-6 meta-analysis. Crit Care. 2015;19:249. [CrossRef]
- Pezzilli R, Fantini L, Fabbri D. IL-6, IL-8, IL-10 in AP. Pancreatology. 2002;2:495–501. [CrossRef]
- Chen CC, Wang SS, et al. Cytokines in AP. Gut. 1999;45:895–901. [CrossRef]
- Ueda T, Takeyama Y, et al. Cytokine levels predict severity. J Gastrointest Surg. 2007;11:891–898. [CrossRef]
- Tsai K, Wang SS, Chen TS. Oxidative stress in AP. Clin Chim Acta. 1998;275:1–7. [CrossRef]
- Randerath O, Lang K. MDA and GSH in AP. Clin Biochem. 2020;75:20–25. [CrossRef]
- Gultepe I, Arslan S, Demir H. Oxidative balance in AP. Dig Dis Sci. 2016;61:3672–3678. [CrossRef]
- Dambrauskas Z, Giese N. Endothelial markers. Pancreas. 2010;39:1164–1169. [CrossRef]
- Chen H, Li F. Presepsin predicts infected necrosis. Pancreatology. 2021;21:345–352. [CrossRef]
- de Madaria E, Martínez J, Lozano B. PTX3 in AP. Gut. 2013;62:509–514. [CrossRef]
- Vaquero E, et al. sCD163 in AP. Clin Sci. 2014;126:521–530. [CrossRef]
- Liu Z, Zhang M. Ang-2 predicts organ failure. Shock. 2013;39:562–566. [CrossRef]
- Mihajlovic M, Lendak D. Endocan in severity prediction. J Crit Care. 2014;29:368–372. [CrossRef]
- Mofidi R, et al. VEGF in AP. Surgery. 2007;141:20–26. [CrossRef]
- Yu M, Chen H. Copeptin predicts severity. Clin Chim Acta. 2019;495:1–7. [CrossRef]
- Kylanpaa ML, et al. ProADM in AP. Crit Care. 2010;14:R147. [CrossRef]
- Keh D, et al. Adrenomedullin. Lancet. 2004;363:1552–1554. [CrossRef]
- Balthazar EJ. CT severity index. Radiology. 1990;174:331–336. [CrossRef]
- Mortele KJ, et al. Modified CTSI. AJR. 2004;183:1261–1265. [CrossRef]
- Cho JH, Kim TN. Predictors of necrosis. Pancreas. 2010;39:782–787. [CrossRef]
- Zhou H, et al. NGAL in AKI during AP. Clin Chim Acta. 2019;495:162–168. [CrossRef]
- Chokshi A, et al. AKI and mortality. Kidney Int. 2012;82:1031–1037. [CrossRef]
- Wu BU, et al. Creatinine predictive power. Gut. 2014;63:149–156. [CrossRef]
- Pupelis G, et al. Vasopressor requirement. Intensive Care Med. 2018;44:161–170. [CrossRef]
- Johnson CD, Abu-Hilal M. Persistent organ failure. Gut. 2004;53:1340–1344. [CrossRef]
- Halonen KI. Severe AP mortality. Br J Surg. 2001;88:886–891. [CrossRef]
- Singh VK, Wu BU. Early ICU admission. Am J Gastroenterol. 2009;104:966–971. [CrossRef]
- Sharma V, Devi T. Lactate in AP. J Crit Care. 2019;52:70–75. [CrossRef]
- Hong W, Lin S. Albumin predicts severity. Medicine. 2017;96:e8002. [CrossRef]
- Isenmann R, Runzi M. Prophylactic antibiotics. Lancet. 2004;364:665–674. [CrossRef]
- Neoptolemos JP, et al. ERCP timing. N Engl J Med. 1988;318:9–15. [CrossRef]
- Committee on ERCP. PEP guideline. Gastrointest Endosc. 2017;85:295–302. [CrossRef]
- Oiva J, et al. Neuroimmune activation. Clin Sci. 2010;119:355–364. [CrossRef]
- Ewers A, et al. Pain biomarkers. Pancreas. 2021;50:1232–1240. [CrossRef]
- Tóth K, Császár A, Márton S. Akut hasnyálmirigy-gyulladás császármetszést követően. Orv Hetil. 2023;164(32):1231–1234. [CrossRef]
- Uden S, et al. Antioxidants in AP. Gut. 1990;31:1312–1316. [CrossRef]
- Tsai K. Lipid peroxidation. Clin Chim Acta. 1998;275:1–7. [CrossRef]
- Zhou MT. ROS in AP. Ann Surg. 2009;249:107–117. [CrossRef]
- Mayerle J, Sendler M, Hegyi E. Acute pancreatitis review. Nat Rev Dis Primers.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).