3. Results and Discussions
3.1. Cultural and Physiological-Biochemical Properties of Isolated Microorganisms
All isolated microorganisms and used in the study were Gram-positive bacteria belonging to five different genus (
Table 1).
CBT are spore-forming, solitary, round bacteria that are urease-, phosphatase-, and amylase-positive, catalase-negative, no proteolytic active, and utilize nitrogen from nitrate salts and are capable of degrading cellulose. Their optimal growth temperature is 45–55 °C, with growth occurring at temperatures up to 65 °C and no growth at 30 °C.
NFBT2 are spore-forming, rod-shaped bacteria that are catalase-, phosphatase-, urease-, and amylase-negative, possessing proteolytic activity, atmospheric nitrogen-fixer and able to utilize ammonium salts. Their optimal growth temperature is 47–50 °C, with no growth at 30 °C.
NFBT9 are spore-forming, solitary, elongated bacteria that are urease-negative, catalase-, phosphatase-, and amylase-positive, lacking proteolytic activity, and assimilate nitrogen from atmospheric air. Optimum growth temperature is 47–50 °C; no growth occurs at 30 °C.
NFBT10 are spore-forming, solitary, round-ended bacteria that are urease-negative, catalase-, phosphatase-, and amylase-positive, lacking proteolytic activity, and assimilate nitrogen from atmospheric air. Optimum growth temperature is 47–50 °C; no growth occurs at 30 °C.
NBT1 are thermotolerant, non-spore-forming, round, solitary bacteria that are urease- and catalase-positive, phosphatase- and amylase-negative, and possess proteolytic activity. They assimilate nitrogen from atmospheric air, nitrates, and ammonium salts, and possess the ability to mobilize phosphates. The optimal growth temperature is 30–40 °C, with a maximum temperature of up to 50 °C.
NBT2 are thermotolerant, non-spore-forming, solitary, elongated bacteria that are urease-, catalase-, and phosphatase-positive, and amylase-positive, possessing proteolytic activity. They assimilate nitrogen from nitrates and ammonium salts, and possess the ability to mobilize phosphates. The optimal growth temperature is 30–40 °C, with a maximum temperature of up to 50 °C.
3.2. Degree of Organic Matter Destruction
Accelerating the decomposition of organic matter is a fundamental element of composting technologies. However, the authors did not find any use the proportion of individual fractions (fragmentation) in assessing the composting process of poultry manure in the scientific literature. It is logical to assume that an increase in the proportion of the smallest fractions reflects the intensity of biochemical reactions in the compost. At the same time, the authors propose separating the concepts of “decomposition,” which reflects deeper processes of biochemical transformation of organic matter, such as humification, from “destruction,” which reflects the quantitative expression of physical parameters such as particle size of the composted material. Thus, the fractions presented in the table can be roughly divided into small (≤0.25 mm and 0.5 mm), reflecting the intensity of decomposition, and large (1.0 mm and ≥2.0 mm), the proportion of which reflects the advisability of continuing the composting process. The analysis results demonstrate the influence of the studied microorganisms on the destruction of organic matter with varying degrees of intensity (
Table 2).
Considering the effect of the isolated strains on the destruction of organic matter in poultry manure, it should be noted that, based on the sum of fractions ≤0.25–0.5 mm, the strains of nitrifying thermotolerant microorganisms were the most effective, increasing the breakdown rate of organic matter by 9.7%. A cellulose-degrading strain and a consortium of thermophilic nitrogen-fixing microorganisms increased this rate by 7.9% and 8.8%, respectively. It was expected that cellulolytic microorganisms would be more aggressive in increasing the proportion of degraded matter. The greater effectiveness of nitrifying microorganisms may be due to their ability to release mineral nitrogen, which acts as an activator of the native cellulolytic microflora of poultry manure. The proportion of undestroyed or slightly destroyed organic material (fraction ≥2.0 mm) was observed in the control variant (20.8%), which turned out to be 18.8–30.8% higher than under the influence of the studied microorganisms.
3.3. Organic Matter Mineralization by Fraction
During composting, active decomposition of organic matter occurs, and the degree of mineralization is estimated by the proportion of total carbon in the biomass. The analysis data (
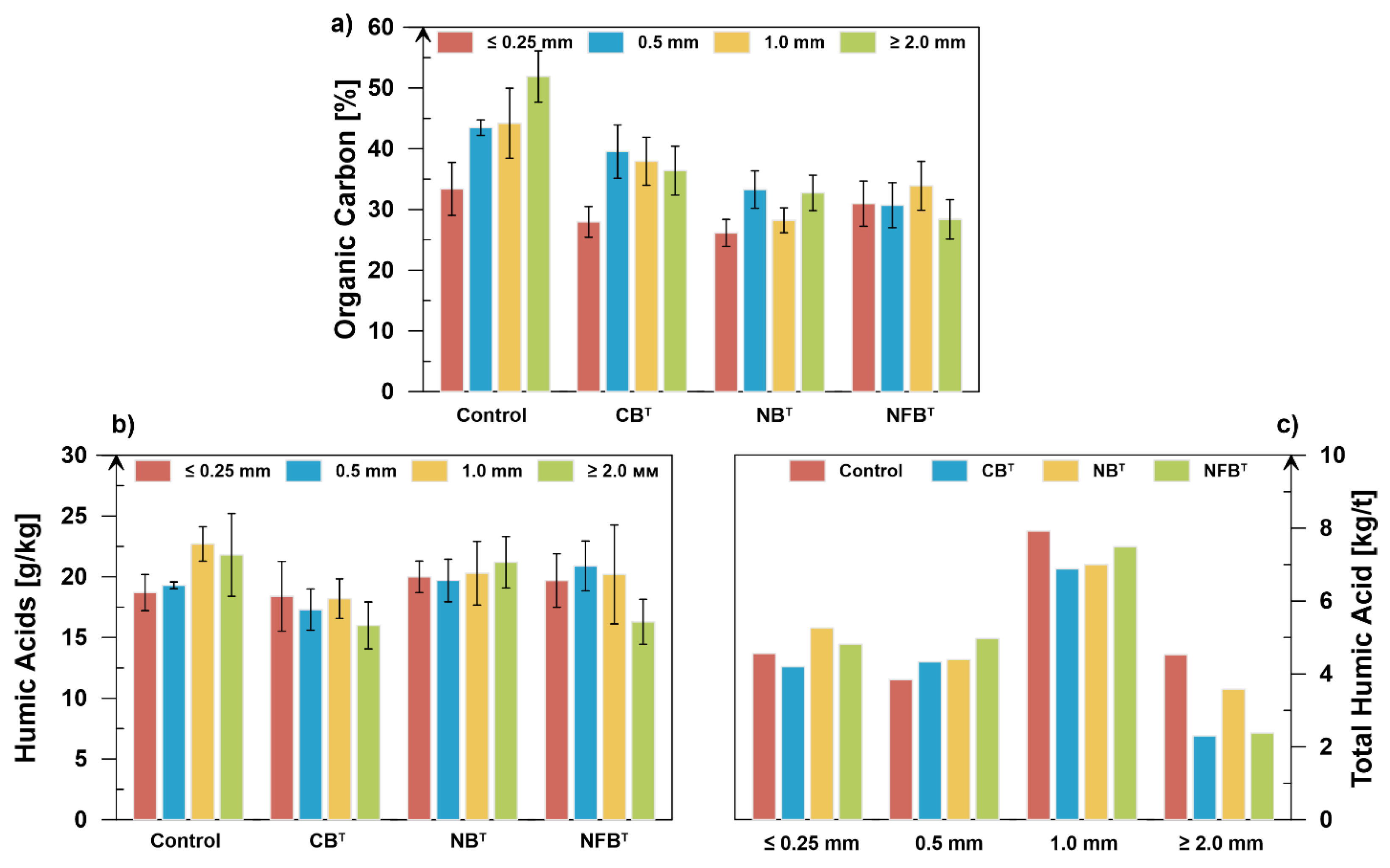
Figure 2a) demonstrate that the introduction of the studied strains into the composted material can accelerate the mineralization of organic matter.
The minimum degree of mineralization (43.2% on average) was observed in the control variant, in which the proportion of organic carbon increased by 33.4%–51.9% as the fraction size increased.
Of the compared strains, nitrifying (30.1%) and nitrogen-fixing microorganisms (31.0%) contributed the most to mineralization. No clear pattern of mineralization by fraction was observed - carbon content by fraction ranged from 26.2% to 33.3% for nitrifying microorganisms and from 28.4% to 33.9% for nitrogen-fixing microorganisms. The cellulose-decomposing microorganisms mineralizes organic matter on average up to 35.5%, but the spread across fractions is more significant – 28.0–39.5%.
3.4. Organic Matter Humification by Fraction
One of the ultimate goals of composting livestock byproducts is the accumulation of HA, transformed from primary organic matter by microorganisms [
25].
The influence of microorganisms on humic substance accumulation varies by fraction (
Figure 2b). The maximum amount of HA in the smallest fraction (≤0.25 mm) is formed under the influence of nitrifiers (20.0 g/kg) and nitrogen fixers (19.7 g/kg), which is 7.0% and 5.3% higher than in the control variant. In the 0.5 mm fraction, nitrogen fixers increase the HA content by 8.3%.
As fractions increase (1.0 and ≥2.0 mm), the maximum HA content is observed in the control variant – 22.7 and 21.8 g/kg, respectively. The impact of cellulose-degrading microorganisms on the humification process is minimal. At the same HA value in the ≤0.25 mm fraction as in the control, a decrease of 10.4%, 19.8%, and 26.6% was observed in the 0.5 mm, 1.0 mm, and ≥2.0 mm fractions, respectively. These data may serve as the basis for reconsidering the use of cellulolytics in the processing of animal byproducts. Thus, they can be added to the composted mass in combination with microbial strains of other physiological groups, or used at later stages of fermentation. A more objective assessment of the humus status of fractions can be made based on their total HA content, calculated based on the HA concentration (g/kg) and the fraction’s proportion (%) in the compost (
Figure 2c).
The figure shows that the smallest fraction (≤0.25 mm) in 1 ton of finished compost accumulates between 4.2 and 5.26 kg of HA, depending on the microorganisms used. The maximum effect is achieved with the addition of nitrifying (5.26 kg/t) and nitrogen-fixing (4.81 kg/t) microorganisms. This is 15.4% and 5.5% higher than in the control, respectively. In the next-largest fraction, 0.5 mm, the maximum accumulation of HA occurs under the influence of nitrogen-fixing microorganisms (4.97 kg/t), which is 29.4% higher than in the control (4.56 kg/t). The effects of cellulose-degrading organisms and nitrifiers were relatively equal: 4.33 and 4.39 kg/t, respectively, which is 12.8 and 14.3% higher than the control value (3.84 kg/t).
As the fraction size increases, the total HA content in the experimental variants decreases relative to the control value. Thus, the 1.0 mm fraction contains 7.92 kg/t of HA compost, which is 15.1% higher than in the CBT variant, 13.1% higher than in the nitrifier variant, and 5.7% higher than in the nitrogen-fixing variant. This trend persists in the ≥2.0 mm fraction. The maximum content (4.53 kg/t) was found in the control variant, which is 26.5–96.7% higher than in the variants using microorganisms.
3.5. Total Nitrogen Content in Compost Fractions
The objective of preserving nitrogen in compost during the processing of waste by-products is a key quality indicator.
The analysis results (
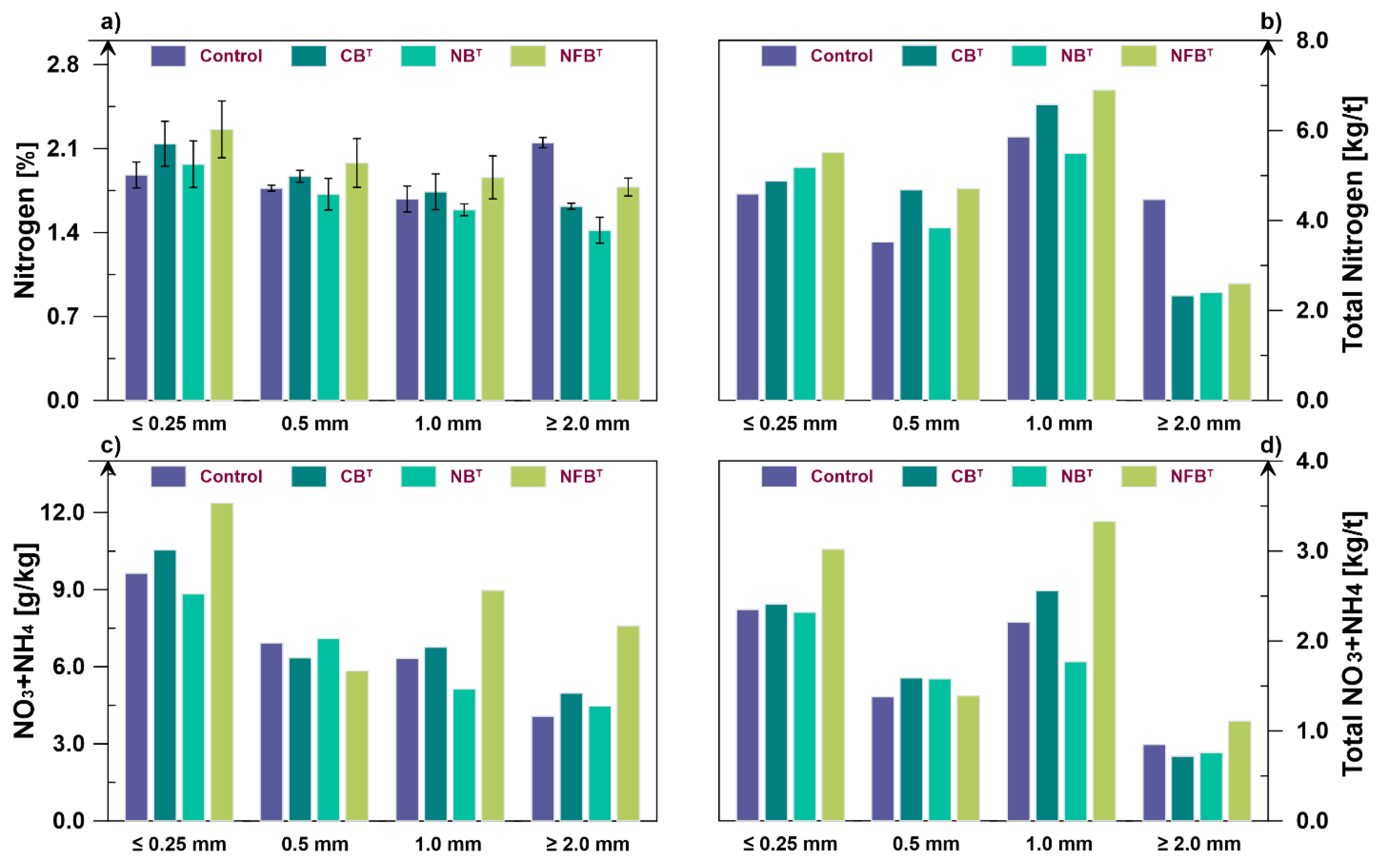
Figure 3a) indicate variations in compost nitrogen content by fraction depending on the physiological characteristics of the microbial strains used in biofermentation. In the ≤0.25 mm fraction, the nitrogen content was higher than in the control by 13.8% (CB
T), 4.8% (NB
T), and 20.2% (NFB
T). In the 0.5 mm and 1.0 mm fractions, the difference between treatments narrowed. The maximum total nitrogen content (1.98%) relative to the control (1.77%) was observed with the use of nitrogen fixers in the 0.5 mm fraction, with a relative difference of 11.9%. In the 1.0 mm fraction, the difference was 10.7%.
The effect of cellulolytics in the 0.5 mm and 1.0 mm fractions was noted by a slight increase in total nitrogen – by 5.7% and 3.6%. Under the influence of nitrifiers, there was a tendency for the total nitrogen content to decrease as the fraction size increased. Thus, compared to the control variant, in the 0.5 mm, 1.0 mm, and ≥2.0 mm fractions, nitrogen decreased by 2.8%, 5.4%, and 34.0%, respectively.
The control variant, relative to the experimental variants, shows an excess of total nitrogen content in the ≥2.0 mm fraction. Compared to cellulolytics, nitrifiers, and nitrogen fixers, the difference is 32.7%, 51.4%, and 20.8%, respectively. The fertilizer value for total nitrogen in individual fractions, as in the case of HA, should be assessed taking into account their share in the finished compost (
Figure 3b).
The total nitrogen content in the compost under the influence of the studied microorganisms varies within the range of 9.1–7.0%, where the minimum content was noted in the variant with nitrifying microorganisms (16.9 kg/t). The maximum (19.73 kg/t) was found in the variant with nitrogen-fixing thermophiles. The total nitrogen content of cellulolytics does not differ from that of the control variant. At the same time, differentiation of nitrogen content by fractions demonstrates more significant differences. The effect of microorganisms on the accumulation of total nitrogen is more pronounced in the smallest fractions (≤0.25 mm and 0.5 mm). Thus, the nitrogen content under the influence of cellulolytics, nitrifiers and nitrogen fixers increases by 6.3%, 12.9% and 20.0%, respectively, for the fraction ≤0.25 mm and by 33.0%, 9.1% and 33.8% for the fraction 0.5 mm.
A large amount of nitrogen accumulates in the 1.0 mm fraction. For this fraction, the maximum effect was observed in the treatments with cellulolytics (6.58 kg/t) and nitrogen fixers (6.90 kg/t), which is 12.3% and 17.78% higher than in the control. Nitrifiers were less effective for this fraction (5.49 kg/t), representing a 6.3% reduction.
In the largest fraction (≥2.0 mm), the maximum total nitrogen content (4.47 kg/t) was observed in the control, that can be attributed to its lower degree of mineralization. Under the influence of microorganisms, 2.33–2.60 kg of nitrogen is accumulated in this fraction per 1 ton of compost. The importance of the method for differentiated compost quality assessment by fractions is associated not only with the ability to monitor the intensity and direction of biofermentation processes but also with the ability to more reliable interpretation the obtained data. As an example, the correlations between total nitrogen and HA values based on the initial analysis results and after their conversion to gross values taking into account the proportion of each individual fraction (
Table 3) can be seen.
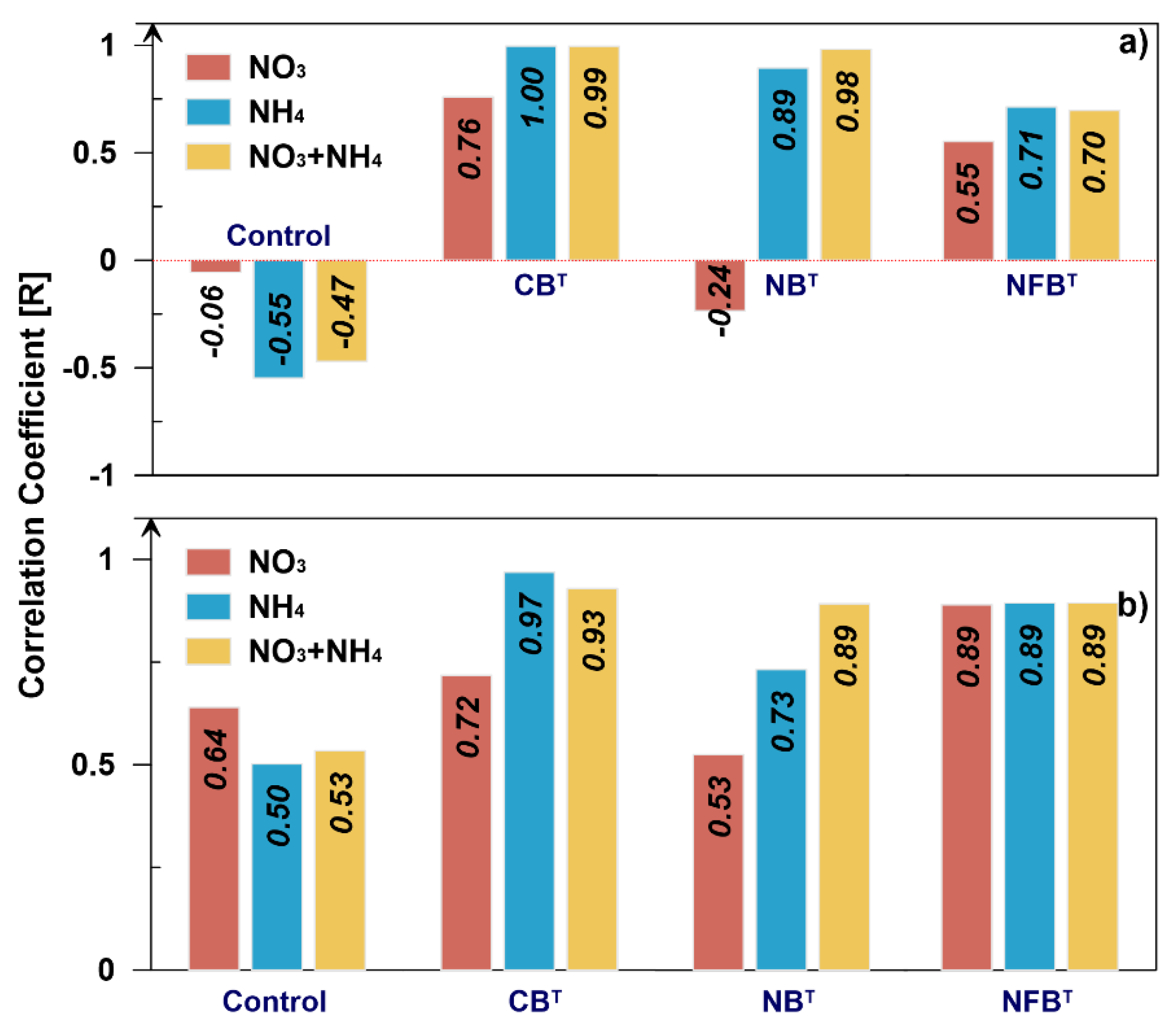
As can be seen from the table, the correlations between total nitrogen and HA values based on relative values do not show a consistent pattern in their content across fractions. Correlation coefficients vary and range from r = -0.765 to r = 0.716.
When converting relative values to absolute values and then taking into account the proportion of each fraction, we obtain highly positive. The correlation coefficients range from r = 0.902 to 0.976.
3.6. The Impact of Microorganisms on the Content of Mineral Nitrogen
One of the main goals of biological fermentation of livestock byproducts is to increase nitrogen availability to plants through the intensive mineralization of nitrogen-containing organic compounds. At the same time, the risk of significant nitrogen loss as ammonia must be considered. The study of various microbial strains is aimed at finding ways to limit non-productive nitrogen losses while improving the overall nitrogen status of compost.
The expediency of introducing selected strains into compostable material is confirmed by test results (
Figure 3c).
Thermophilic nitrogen-fixing microorganisms had the greatest impact on the accumulation of mineral nitrogen. Thus, with the exception of the 0.5 mm fraction, which contained 15.7% less nitrogen, in the fractions ≤0.25 mm, 1.0 mm, and ≥2.0 mm, the increase was 28.3%, 41.9%, and 83.6%, respectively. The cellulolytic thermophilic strain also proved effective, but to a lesser extent. For fractions ≤0.25 mm, 1.0 mm, and ≥2.0 mm, increases of 9.4%, 7.0%, and 22.1% were observed. In the 0.5 mm fraction, a decrease of 8.4% was observed.
The effectiveness of nitrifying microorganisms in mineral nitrogen accumulation was minimal compared to the other strains. For fractions 0.5 mm and ≥2.0 mm, increases of 2.5% and 9.6% were observed. Decreases of 8.3% and 18.8% were observed in the ≤0.25 mm and 1.0 mm fractions.
The proportions of compost fractions when calculating the mineral nitrogen content by physical weight (kg per ton of compost) introduce certain adjustments that more accurately reflect the nitrogen status of each fraction (
Figure 3d). In terms of the overall accumulation of mineral nitrogen forms in compost, nitrogen fixers are the most effective, with an accumulation rate of 8.85 kg per ton of compost. This is more than 30% higher than the control (6.79 kg/t). The increase in nitrogen due to cellulolytics is 7.2% (7.28 kg/t). Nitrifiers contribute to a decrease in mineral nitrogen content by 5.3% (6.43 kg/t).
Given the narrow functionality of nitrifying microorganisms, it is more accurate to evaluate their effect not through their influence on the total mineral nitrogen content, but on its nitrate form in the compost. In this case, for individual fractions, we see results consistent with their function, specifically the accumulation of nitrate nitrogen and a significant reduction in the ratio with ammonium nitrogen (
Figure 4).
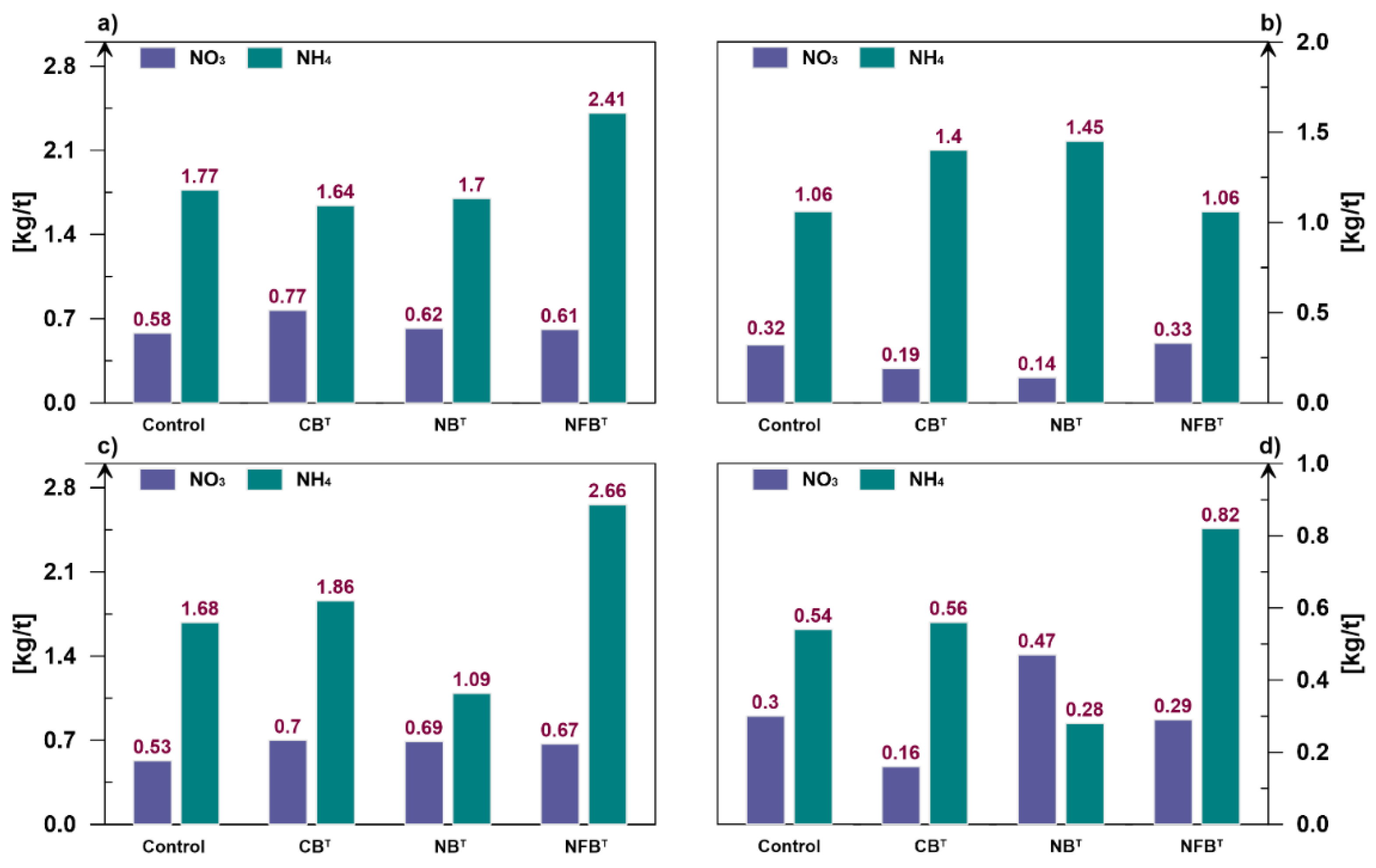
Looking at the figure, it should be noted that the ammonium nitrogen content is higher than the nitrate nitrogen content in most variants. On average, nitrogen-fixing microorganisms have the greatest influence on the total ammonium content across fractions. Under their influence, 6.95 kg/t accumulates in the compost, which is 37.6% higher than in the control variant (5.05 kg/ha). Ammonium nitrogen, 5.46 kg/t, accumulates in the compost under the influence of cellulolytic bacteria (+8.1%). The minimum accumulation of ammonium nitrogen (4.52 kg/t) was observed in the variant with nitrifying bacteria, which is 10.5% lower than in the control variant. In terms of nitrate nitrogen, nitrifying microorganisms proved to be the most effective, producing 1.92 kg/t of NO3 in the compost, which is 9.7% higher than the control (1.75 kg/t) and 5.5% higher than the cellulolytics-based treatment (1.82 kg/t). Nitrogen fixers were slightly inferior to nitrifiers in their ability to accumulate nitrate nitrogen in the compost (1.90 kg/t).
A distinctive feature of the nitrifying strain was their activity in larger compost fractions. For example, in the 1.0 mm and ≥2.0 mm fractions, NO3 accumulation was higher than in the control by 30.2% and 51.6%. In the largest fraction, nitrifiers were significantly more effective than cellulolytics and nitrogen fixers by 193.8% and 62.1%, respectively. It can be assumed that large fractions of compost mass provide a favorable ecological niche for nitrifying activity. This thesis is supported by the narrow ratio of nitrate to ammonium nitrogen. This ratio in the fraction ≥2.0 mm is 1.68, while for other variants it is 0.57 (Control), 0.29 (CBT), and 0.35 (NFBT).
The influence of nitrifying microorganisms in this case can be assessed as climatically favorable, as it is accompanied by a decrease in the proportion of ammonium, a key predictor of nitrous oxide flux into the atmosphere [
26].
Fractional differentiation of the nitrogen status of the compost demonstrates closer correlations when comparing nitrate, ammonium forms, and their sums with the total nitrogen content (
Figure 5). The figure shows that recalculating the mineral nitrogen content of compost by fractions more accurately reflects their relationship with the total nitrogen content.
3.7. Bioreclamation Properties of Individual Compost Fractions
Biological indicators are increasingly being used as indicators of soil fertility. Given the subject of the study—the influence of microorganisms on the transformation of organic matter from livestock byproducts—microbial biomass carbon and invertase activity were used as bioindicators. Microbial biomass carbon represents an important part of the labile fraction of soil organic matter, responsible for effective fertility [
27]. Invertase, in turn, indicates the intensity of organic matter decomposition [
28].
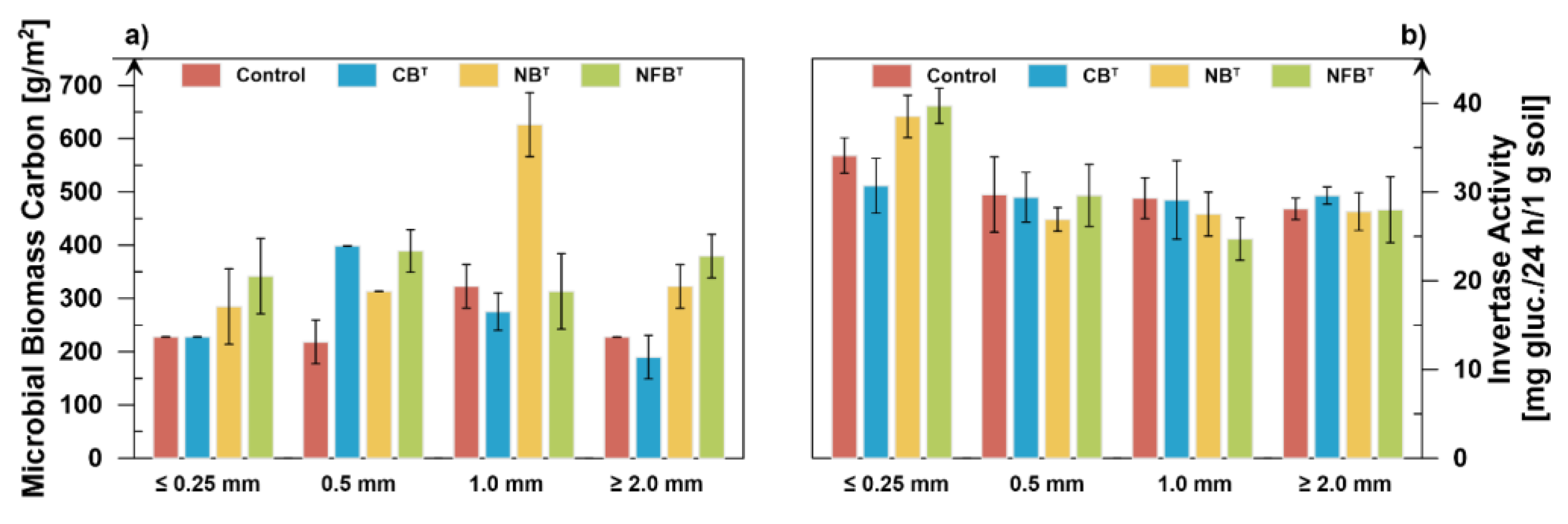
The application of compost treated with different microbial strains to soil has different effects on the accumulation of microbial biomass carbon (
Figure 6a).
Thus, under the influence of the smallest compost fraction (≤0.25 mm), the maximum accumulation of microbial biomass carbon is observed in the soil variant using thermophilic nitrogen fixers (342 g/m2), which is 50.0% higher than in the control variant (228 g/m2). Nitrifiers increase this indicator by 25.0% (285 g/m2). The Microbial Biomass Carbon (MBC) content under the influence of cellulolytics is equivalent to that in the control variant.
Under the influence of the 0.5 mm compost fraction, all studied strains (NFBT, NBT, CBT) increase the MBC content by 78.4%, 43.6%, and 83.0%, respectively.
Under the influence of the 1.0 mm compost fraction, an increase in MBC content is observed only in the variant using nitrifiers, and it is maximum – 93.8%. Cellulolytics reduce the content by 14.9%. The reduction under the influence of nitrogen fixers does not exceed the 5% threshold.
The coarse compost fraction (≥2.0 mm) obtained with nitrogen fixers and nitrifiers increased MBC by 66.7% and 41.7%, respectively. Cellulosolytics, on the other hand, reduced microbial carbon content by 16.7%.
Summarizing the assessment of compost bioreclamation properties based on microbial biomass carbon, it can be concluded that nitrogen fixers and nitrifiers in compost in most cases act as activators of the accumulation of labile organic matter in the soil.
Changes in soil invertase activity under the influence of biofermented compost are observed mainly with the addition of the smallest fraction (Figure 6b). The highest invertase activity is observed in soil samples fertilized with the smallest compost fraction (≤0.25 mm). This trend applies to all experimental variants, but with different degrees. When examining the effects of the treatments within a single fraction of ≤0.25 mm, it can be noted that the use of nitrogen-fixing and nitrifying strains in biofermentation increases the bioremediation properties of compost by 16.4% and 12.9%, respectively. Conversely, the treatment with cellulolytics reduces soil invertase activity by 10.0%.
Differences in the effects of microorganisms on invertase activity in larger fractions of 0.5 mm, 1.0 mm, and ≥2.0 mm narrow and range from 26.9–29.7, 24.7–29.3, and 27.8–29.6 mg glucose/24h/1 g soil, respectively.