Submitted:
01 December 2025
Posted:
03 December 2025
You are already at the latest version
Abstract
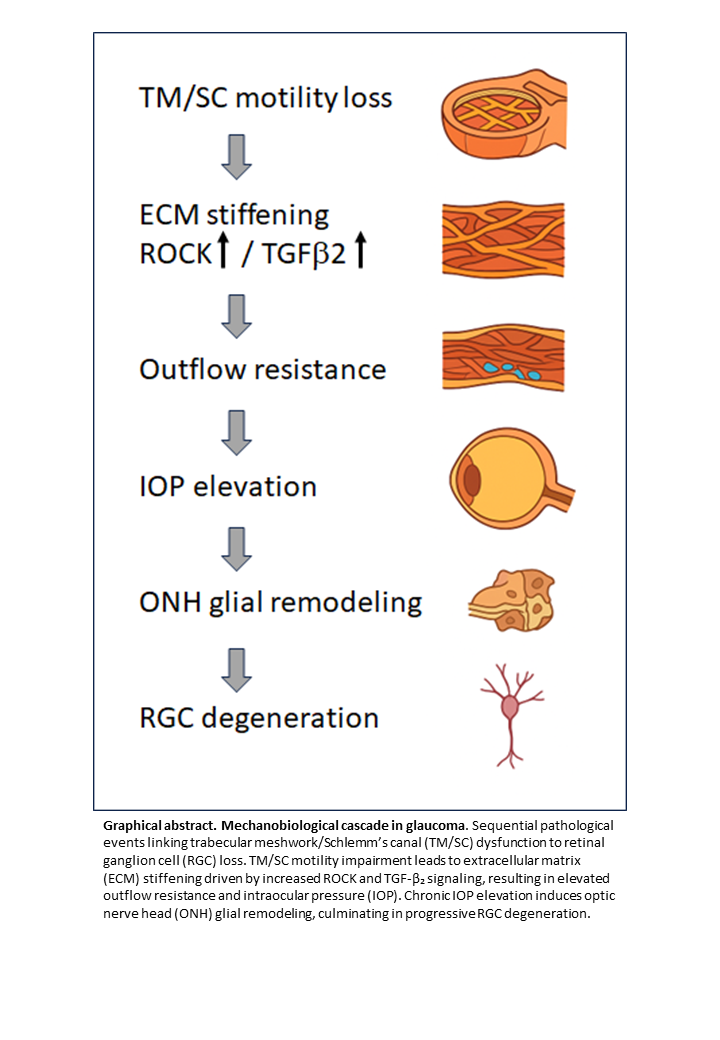
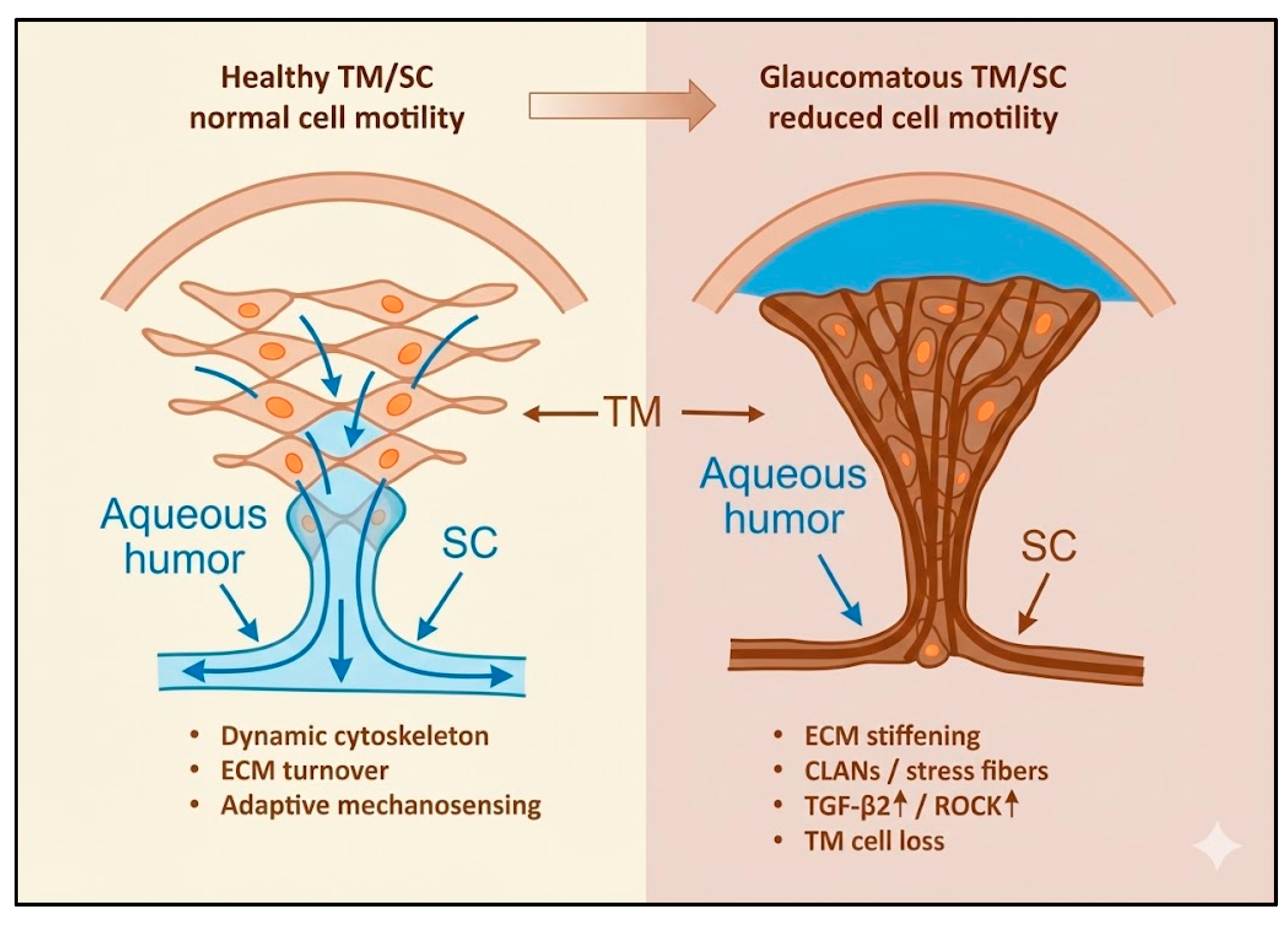
Cell motility—the dynamic process encompassing migration, adhesion modulation, cytoskeletal remodeling, and extracellular matrix (ECM) interactions—is fundamental to ocular homeostasis. In glaucoma, disrupted motility of trabecular meshwork (TM) and Schlemm’s canal (SC) cells contributes to impaired aqueous humor outflow and elevated intraocular pressure (IOP), while reactive motility of optic nerve head (ONH) glial cells promotes fibrosis and neurodegeneration. Mechanistically, TM/SC motility is regulated by Rho GTPase and ROCK signaling, focal adhesion dynamics, and ECM interactions, while glial cells respond to mechanical stress and cytokines such as TGF-β2. Cytoskeletal alterations, ECM stiffening, and endothelial–mesenchymal transition (EndMT) contribute to glaucomatous damage by reducing normal cell motility and tissue remodeling capacity. Aberrant motility at the ONH, including heterogeneous astrocytic reactivity, leads to lamina cribrosa remodeling and retinal ganglion cell degeneration. Therapeutically, ROCK inhibitors improve TM/SC motility and outflow, suppress EndMT, and may confer neuroprotection. Stem cell–based strategies and modulation of TGF-β2 or mechanotransduction pathways represent emerging approaches to restore physiological motility and regenerative potential. Despite promising advances, challenges remain in ensuring targeted, durable, and safe modulation of cellular dynamics. Understanding and therapeutically harnessing cell motility offers a unifying framework to address both pressure-dependent and neurodegenerative mechanisms in glaucoma.
Keywords:
1. Introduction
2. Literature Search Methodology
- Cellular Motility and Cytoskeleton: "cell motility," "cell migration," "cytoskeletal remodeling," "actomyosin contractility," "focal adhesion."
- Glaucoma Pathogenesis: "glaucoma," "trabecular meshwork," "Schlemm's canal," "optic nerve head," "lamina cribrosa," "retinal ganglion cell degeneration."
- Molecular Pathways and Mechanisms: "Rho GTPase," "ROCK signaling," "TGF-beta2," "mechanotransduction," "Piezo1," "TRPV4," "YAP/TAZ," "extracellular matrix stiffening," "endothelial-mesenchymal transition."
- Therapeutic Interventions: "ROCK inhibitor," "netarsudil," "ripasudil," "stem cell therapy," "regenerative medicine," "senolytic," "ferroptosis inhibitor."
3. Mechanisms of Normal Motility in the Aqueous Outflow Pathway
3.1. Cytoskeletal Regulation in TM and SC Cells
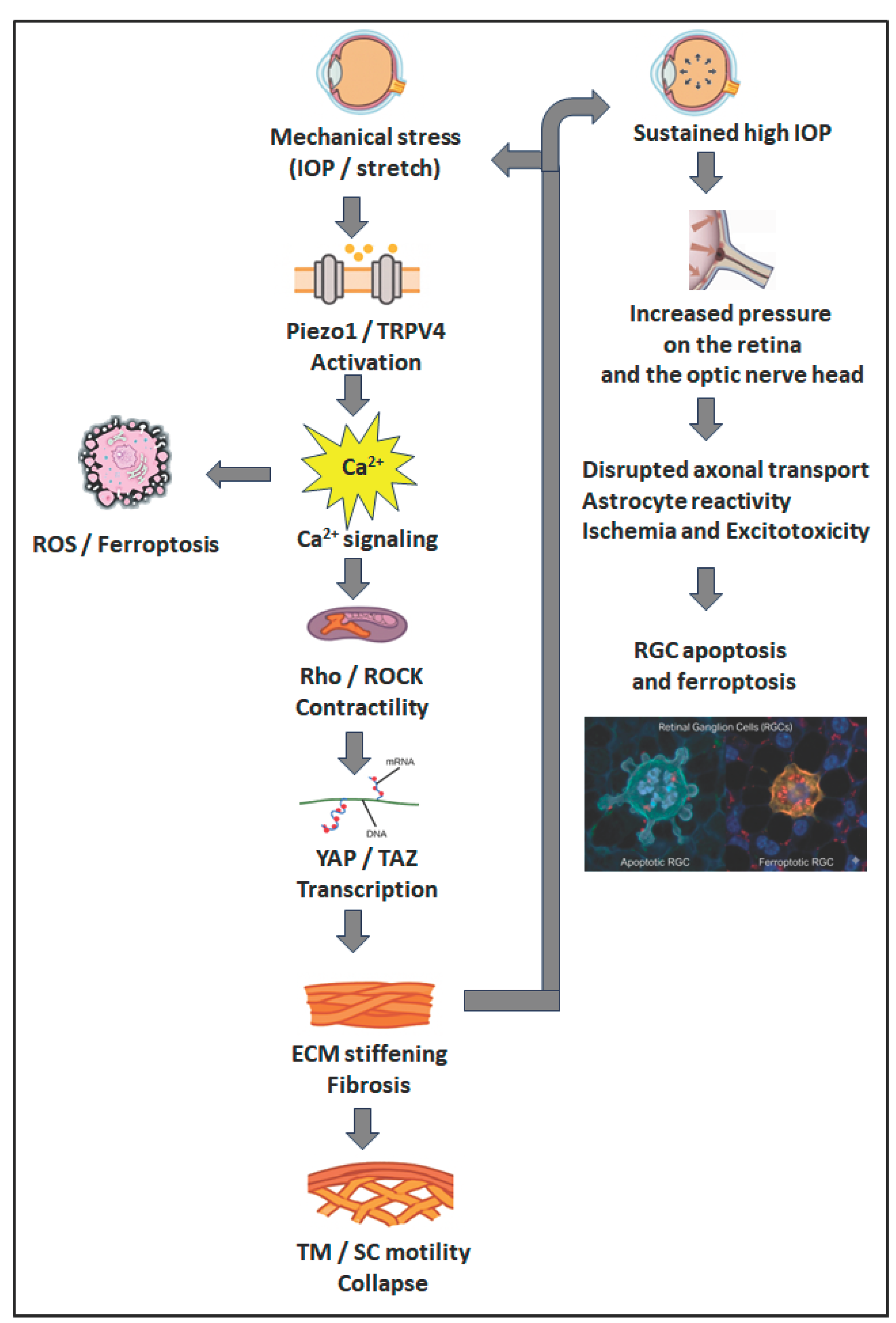
3.2. Mechanosensitive Channels and Ionic Signalling
3.3. Extracellular Cues, Cytokines and ECM Turnover
3.4. Mechanotransducive Transcriptional Effectors
3.5. Integrated View: A Dynamic Equilibrium
4. Altered Motility and the Onset of Glaucomatous Remodeling
4.1. Glial and Optic Nerve Head Remodeling
5. Interplay of Mechanical Forces and Motility
6. Consequences of Impaired Motility
6.1. Impaired Repair and Regeneration
7. Therapeutic Restoration of Motility and Future Directions
7.1. Pharmacologic Modulation: Rho/ROCK Inhibition
7.2. Regenerative and Cell-Based Strategies
7.3. Emerging Targets and Multi-Modal Strategies
7.4. Diagnostic Innovation and Future Paradigms
7.5. Concluding Perspective
8. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Qin M, Yu-Wai-Man C (2023) Glaucoma: Novel antifibrotic therapeutics for the trabecular meshwork. Eur J Pharmacol 954:175882. [CrossRef]
- Zhou Y, Liu Z, Gao W, Yang Y, Peng Q, et al. (2024) Pathological Mechanism and Clinical Therapy Progress of Schlemm's Canal. Journal of ophthalmology 2024:9978312. [CrossRef]
- Tang L TC, Wang Y, Shi X (ed.) (2022) The molecular mechanisms of trabecular meshwork damage in POAG and treatment advances: IntechOpen.
- Wan Y, Guo X, Wang N (2025) Trabecular meshwork: A pivotal target for evolving glaucoma treatments. Survey of ophthalmology 70:1043-1060. [CrossRef]
- Fleenor DL, Shepard AR, Hellberg PE, Jacobson N, Pang IH, et al. (2006) TGFbeta2-induced changes in human trabecular meshwork: implications for intraocular pressure. Investigative ophthalmology & visual science 47:226-234. [CrossRef]
- Inoue-Mochita M, Inoue T, Fujimoto T, Kameda T, Awai-Kasaoka N, et al. (2015) p38 MAP kinase inhibitor suppresses transforming growth factor-beta2-induced type 1 collagen production in trabecular meshwork cells. PloS one 10:e0120774. [CrossRef]
- Li H, Singh A, Perkumas KM, Stamer WD, Ganapathy PS, et al. (2022) YAP/TAZ Mediate TGFbeta2-Induced Schlemm's Canal Cell Dysfunction. Investigative ophthalmology & visual science 63:15.
- Oikawa K, Torne O, Sun D, Moon AKB, Kiland JA, et al. (2023) Aqueous Humor TGF-beta2 and Its Association With Intraocular Pressure in a Naturally Occurring Large Animal Model of Glaucoma. Investigative ophthalmology & visual science 64:18. [CrossRef]
- Yarishkin O, Phuong TTT, Baumann JM, De Ieso ML, Vazquez-Chona F, et al. (2021) Piezo1 channels mediate trabecular meshwork mechanotransduction and promote aqueous fluid outflow. The Journal of physiology 599:571-592. [CrossRef]
- Liu K, Xu J, Yang R, Wang F, Su Y (2024) Ion channel Piezo1 induces ferroptosis of trabecular meshwork cells: a novel observation in the pathogenesis in primary open angle glaucoma. American journal of physiology Cell physiology 327:C1591-C1603. [CrossRef]
- Cullen PF, Sun D (2023) Astrocytes of the eye and optic nerve: heterogeneous populations with unique functions mediate axonal resilience and vulnerability to glaucoma. Frontiers in ophthalmology 3. [CrossRef]
- Zhu Y, Wang R, Pappas AC, Seifert P, Savol A, et al. (2023) Astrocytes in the Optic Nerve Are Heterogeneous in Their Reactivity to Glaucomatous Injury. Cells 12. [CrossRef]
- Fujimoto T, Inoue-Mochita M, Inoue T (2023) A ROCK inhibitor suppresses the transforming growth factor-beta-2-induced endothelial-mesenchymal transition in Schlemm's canal endothelial cells. Scientific reports 13:9655. [CrossRef]
- Balasubramanian R, Kizhatil K, Li T, Tolman N, Bhandari A, et al. (2024) Transcriptomic profiling of Schlemm's canal cells reveals a lymphatic-biased identity and three major cell states. bioRxiv : the preprint server for biology.
- Guan G, Cannon RD, Coates DE, Mei L (2023) Effect of the Rho-Kinase/ROCK Signaling Pathway on Cytoskeleton Components. Genes 14. [CrossRef]
- Singh P, Karkhur S, Verma V, Gupta S, Beri A (2025) Molecular Gatekeepers of Aqueous Outflow: From Mechanotransduction to Gene Therapy in Trabecular Meshwork Health and Disease. Cureus 17:e91633. [CrossRef]
- Inoue T, Tanihara H (2013) Rho-associated kinase inhibitors: a novel glaucoma therapy. Progress in retinal and eye research 37:1-12. [CrossRef]
- Wang K, Read AT, Sulchek T, Ethier CR (2017) Trabecular meshwork stiffness in glaucoma. Experimental eye research 158:3-12. [CrossRef]
- Patel PD, Chen YL, Kasetti RB, Maddineni P, Mayhew W, et al. (2021) Impaired TRPV4-eNOS signaling in trabecular meshwork elevates intraocular pressure in glaucoma. Proceedings of the National Academy of Sciences of the United States of America 118. [CrossRef]
- Di X, Gao X, Peng L, Ai J, Jin X, et al. (2023) Cellular mechanotransduction in health and diseases: from molecular mechanism to therapeutic targets. Signal transduction and targeted therapy 8:282. [CrossRef]
- Li H, Kuhn M, Kelly RA, Singh A, Palanivel KK, et al. (2024) Targeting YAP/TAZ mechanosignaling to ameliorate stiffness-induced Schlemm's canal cell pathobiology. American journal of physiology Cell physiology 326:C513-C528. [CrossRef]
- Rudzitis CN, Lakk M, Singh A, Redmon SN, Kirdajova D, et al. (2025) TRPV4 activation by TGFbeta2 enhances cellular contractility and drives ocular hypertension. eLife 14.
- Lakk M, Krizaj D (2021) TRPV4-Rho signaling drives cytoskeletal and focal adhesion remodeling in trabecular meshwork cells. American journal of physiology Cell physiology 320:C1013-C1030. [CrossRef]
- Uchida T, Shimizu S, Yamagishi R, Tokuoka SM, Kita Y, et al. (2021) Mechanical stretch induces Ca(2+) influx and extracellular release of PGE(2) through Piezo1 activation in trabecular meshwork cells. Scientific reports 11:4044. [CrossRef]
- Soundararajan A, Wang T, Sundararajan R, Wijeratne A, Mosley A, et al. (2022) Multiomics analysis reveals the mechanical stress-dependent changes in trabecular meshwork cytoskeletal-extracellular matrix interactions. Frontiers in cell and developmental biology 10:874828. [CrossRef]
- Wordinger RJ, Fleenor DL, Hellberg PE, Pang IH, Tovar TO, et al. (2007) Effects of TGF-beta2, BMP-4, and gremlin in the trabecular meshwork: implications for glaucoma. Investigative ophthalmology & visual science 48:1191-1200. [CrossRef]
- Ma S, Meng Z, Chen R, Guan KL (2019) The Hippo Pathway: Biology and Pathophysiology. Annual review of biochemistry 88:577-604. [CrossRef]
- Xin C, Sang Q (2025) Trabecular meshwork motion reduces in eyes with normal tension glaucoma using phase-sensitive optical coherence tomography. BMJ open ophthalmology 10. [CrossRef]
- Keller KE, Peters DM (2022) Pathogenesis of glaucoma: Extracellular matrix dysfunction in the trabecular meshwork-A review. Clinical & experimental ophthalmology 50:163-182. [CrossRef]
- Buffault J, Brignole-Baudouin F, Reboussin E, Kessal K, Labbe A, et al. (2022) The Dual Effect of Rho-Kinase Inhibition on Trabecular Meshwork Cells Cytoskeleton and Extracellular Matrix in an In Vitro Model of Glaucoma. Journal of clinical medicine 11. [CrossRef]
- Gong Z, Shi Y, Liu J, Zhang Y, Johnstone MA, et al. (2025) Volumetric imaging of trabecular meshwork dynamic motion using 600 kHz swept source optical coherence tomography. Biomedical optics express 16:267-281. [CrossRef]
- Schneider M, Fuchshofer R (2016) The role of astrocytes in optic nerve head fibrosis in glaucoma. Experimental eye research 142:49-55. [CrossRef]
- Lee EJ, Park DY, Han JC, Kee C (2025) Understanding glaucoma as astrocyte-driven neurodegeneration in the optic nerve head: an integrative clinicopathological perspective. Progress in retinal and eye research 107:101379. [CrossRef]
- Ethier CR, Herberg S (2025) Mechanobiology in the eye. npj biological physics and mechanics 2:18. [CrossRef]
- Singh IP, Fechtner RD, Myers JS, Kim T, Usner DW, et al. (2020) Pooled Efficacy and Safety Profile of Netarsudil Ophthalmic Solution 0.02% in Patients With Open-angle Glaucoma or Ocular Hypertension. Journal of glaucoma 29:878-884. [CrossRef]
- Mazumder AG, Jule AM, Sun D (2023) Astrocytes of the optic nerve exhibit a region-specific and temporally distinct response to elevated intraocular pressure. Molecular neurodegeneration 18:68.
- Karnam S, Maurya S, Ng E, Choudhary A, Thobani A, et al. (2023) Dysregulation of Neuroprotective Lipoxin Pathway in Astrocytes in Response to Cytokines and Ocular Hypertension. bioRxiv : the preprint server for biology. [CrossRef]
- Pattabiraman PP, Rinkoski T, Poeschla E, Proia A, Challa P, et al. (2015) RhoA GTPase-induced ocular hypertension in a rodent model is associated with increased fibrogenic activity in the trabecular meshwork. The American journal of pathology 185:496-512. [CrossRef]
- Wu J, Wei J, Chen H, Dang Y, Lei F (2024) Rho Kinase (ROCK) Inhibitors for the Treatment of Glaucoma. Current drug targets 25:94-107. [CrossRef]
- Bugara K, Pacwa A, Smedowski A (2024) Molecular pathways in experimental glaucoma models. Frontiers in neuroscience 18:1363170. [CrossRef]
- Singh K, Singh A (2023) Rho-kinase Inhibitors in Ocular Diseases: A Translational Research Journey. Journal of current glaucoma practice 17:44-48. [CrossRef]
- Araie M, Sugiyama K, Aso K, Kanemoto K, Iwata R, et al. (2023) Phase 3 Clinical Trial Comparing the Safety and Efficacy of Netarsudil to Ripasudil in Patients with Primary Open-Angle Glaucoma or Ocular Hypertension: Japan Rho Kinase Elevated Intraocular Pressure Treatment Trial (J-ROCKET). Advances in therapy 40:4639-4656. [CrossRef]
- Chatzimichail E, Christodoulaki E, Konstas PAG, Tsiropoulos GN, Amaxilati E, et al. (2025) Rho Kinase Inhibitors in Glaucoma Management: Current Perspectives and Future Directions. Drug design, development and therapy 19:2519-2531. [CrossRef]
- Kelley MJ, Rose AY, Keller KE, Hessle H, Samples JR, et al. (2009) Stem cells in the trabecular meshwork: present and future promises. Experimental eye research 88:747-751. [CrossRef]
- Zhu W, Godwin CR, Cheng L, Scheetz TE, Kuehn MH (2020) Transplantation of iPSC-TM stimulates division of trabecular meshwork cells in human eyes. Scientific reports 10:2905. [CrossRef]
- Coulon SJ, Schuman JS, Du Y, Bahrani Fard MR, Ethier CR, et al. (2022) A novel glaucoma approach: Stem cell regeneration of the trabecular meshwork. Progress in retinal and eye research 90:101063. [CrossRef]
- Xi G, Feng P, Zhang X, Wu S, Zhang J, et al. (2024) iPSC-derived cells stimulate ABCG2(+)/NES(+) endogenous trabecular meshwork cell proliferation and tissue regeneration. Cell proliferation 57:e13611.
- Bahranifard MR, Chan J, Read AT, Li G, Cheng L, et al. (2025) Magnetically steered cell therapy for reduction of intraocular pressure as a treatment strategy for open-angle glaucoma. eLife 13.
- Igarashi N, Honjo M, Aihara M (2021) mTOR inhibitors potentially reduce TGF-beta2-induced fibrogenic changes in trabecular meshwork cells. Scientific reports 11:14111. [CrossRef]
- Doyle C, Callaghan B, Roodnat AW, Armstrong L, Lester K, et al. (2024) The TGFbeta Induced MicroRNAome of the Trabecular Meshwork. Cells 13. [CrossRef]
- Liu LC, Chen YH, Lu DW (2024) The Application of Rho Kinase Inhibitors in the Management of Glaucoma. International journal of molecular sciences 25. [CrossRef]
- Kirby MA, Pelivanov I, Song S, Ambrozinski L, Yoon SJ, et al. (2017) Optical coherence elastography in ophthalmology. Journal of biomedical optics 22:1-28. [CrossRef]
- Ma Y, Moroi SE, Roberts CJ (2021) Non-invasive Clinical Measurement of Ocular Rigidity and Comparison to Biomechanical and Morphological Parameters in Glaucomatous and Healthy Subjects. Frontiers in medicine 8:701997. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



