Submitted:
29 November 2025
Posted:
01 December 2025
You are already at the latest version
Abstract
Background: Diabetic animals show increased plasma glucose, cholesterol, triglycerides, and low-density lipoprotein levels, along with decreased body weight and glycogen in the liver and muscles. While effective diabetic treatments are still being investigated, plants and herbs have historically contributed to treatment of ailments. The current study explores an alternative, complementary and potential treatment for diabetes. In ethno-medicine, various parts of Mangifera indica are used to treat diabetes mellitus. Methods: The present study aims at investigating antidiabetic, hypolipidemic, and histopathological analysis of Mangifera indica leaf methanolic extract (MIME) in streptozotocin-induced diabetic mice by administering different oral doses (200,400 and 600 mg/kg body weight). After induction of diabetes, the animals are assigned into six groups (one control group and five treatment groups). The treatment groups are treated by different dose of extract, and glibenclamide, and their blood glucose levels were measured. The BGL recording is done using blood glucose test strips OneTouch glucometer on weekly intervals for five weeks. Hematology and other parameters such as liver, renal and total lipid profiles were determined in normal and streptozotocin-induced diabetic mice after oral administration of the extract for 28 days. Post-treatment histopathological changes in the diabetic mice organs, like the pancreas, liver, and kidney, were assessed at the end of treatments. Results: Daily oral administration of MIME (200,400 and 600 mg/kg body weight) and glibenclamide (6 mg/kg) showed significant effects on blood glucose level (P<0.001) as well as in improving kidney, liver functions and hyperlipidemia due to diabetes. The extract treatment also showed to enhanced body weight of diabetic mice as compared to diabetic control group. The NIME showed a restorative effect on pancrease, liver and kidney from the study of histopathological changes. Conclusions: Mageferin indica possess anti-diabetic property in diabetic mice. It improves body weight, liver profile, renal profile and lipid profiles. It has a favorable effect in inhibiting diabetic-induced damage of islets of Langerhans in the pancreas.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Extraction Chemicals and Solvents
2.3. Histopathology and Toxicity Study Reagents
2.4. Methanol Leaf Extraction
2.5. Animal
2.6. Treatment and Control Groups
Experimental Grouping
| GROUP | TITLE | No of mice | TREATMENT |
| 1 | Diabetes control | 6 | Feed + Water |
| 2 | Normal control | 6 | Feed + Water |
| 3 | Diabetic low dose 200mg/kg b.w. | 6 | Feed + extract |
| 4 | Diabetic medium dose 400mg/kg b.w. | 6 | Feed + Extract |
| 5 | Diabetic High dose 600mg/kg b.w. | 6 | Feed +Extract |
| 6 | Diabetic Glibenclamide 6mg/kg b.w. | 6 | Feed standard drug |
2.7. Euthanasia and Sample Collection
2.8. Body Weight Measurements
2.9. Blood Glucose Monitoring
2.10. Hematological Analysis
2.11. Blood Chemistry Analysis
2.12. Histopathological Examination
2.13. Organ Collection and Weight Analysis
2.14. Ethical Issues
3. Results
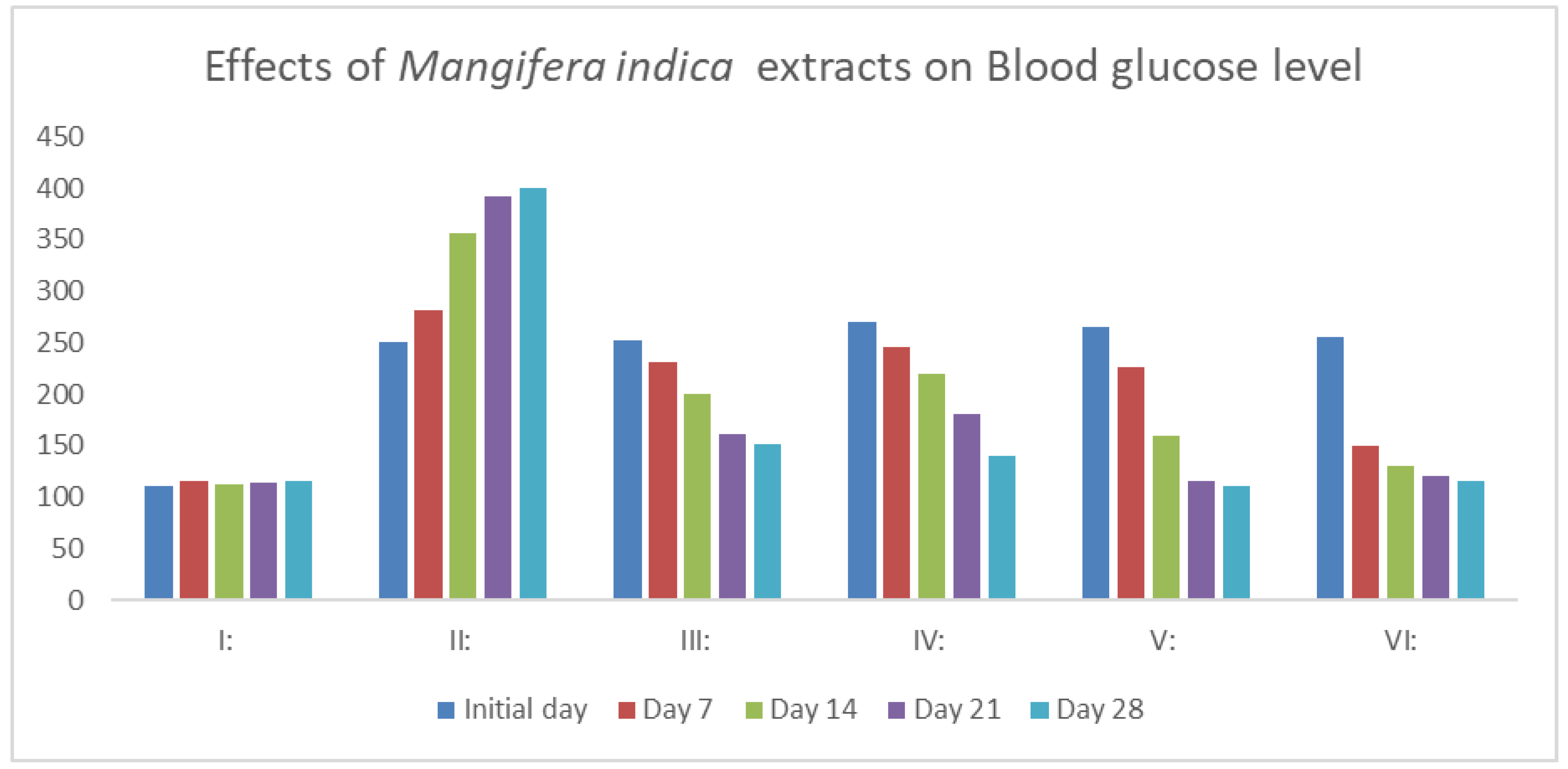
3.1. Serum Glucose Level
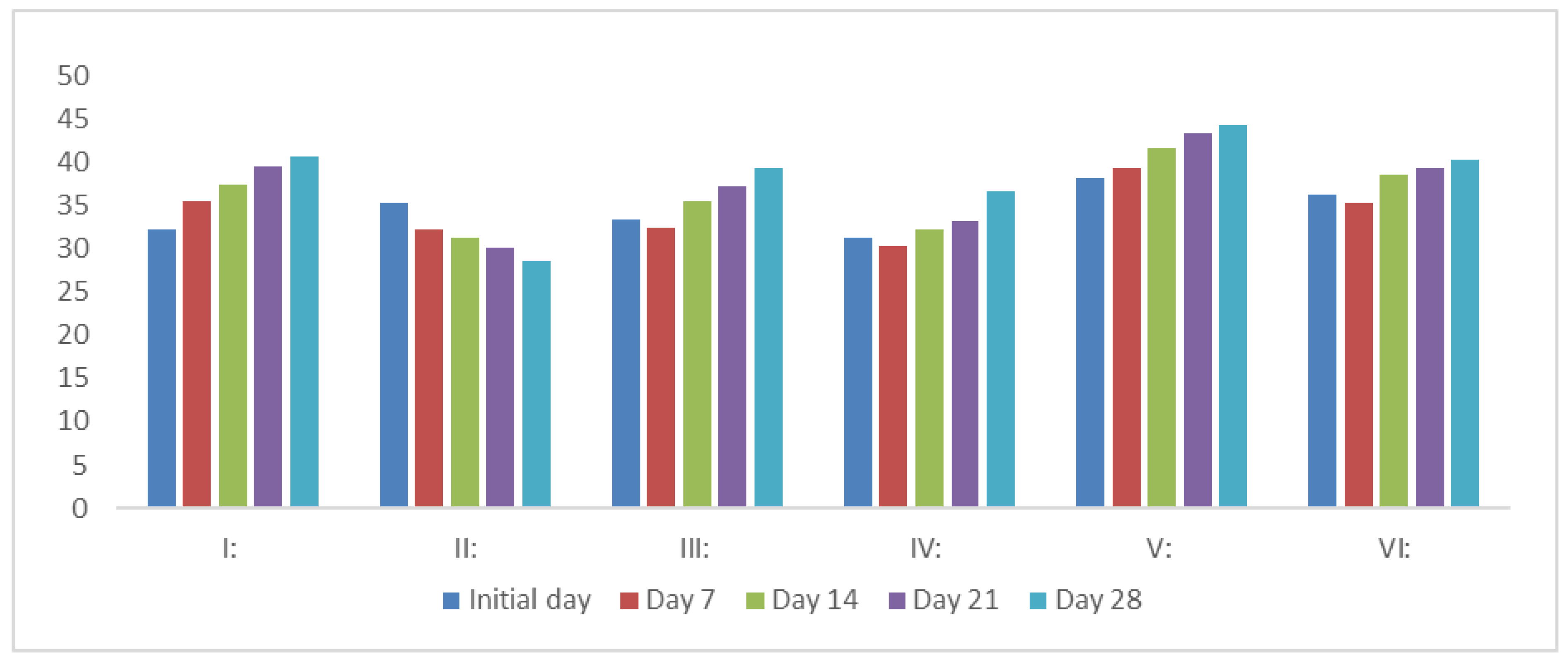
3.2. Effect on the Body Weight of Rats
3.3. Hematology
3.4. Effect on Lipid Profile
3.5. Effect on Liver Functions
3.6. Effect on Kidney Functions
3.7. Histopathological Analysis
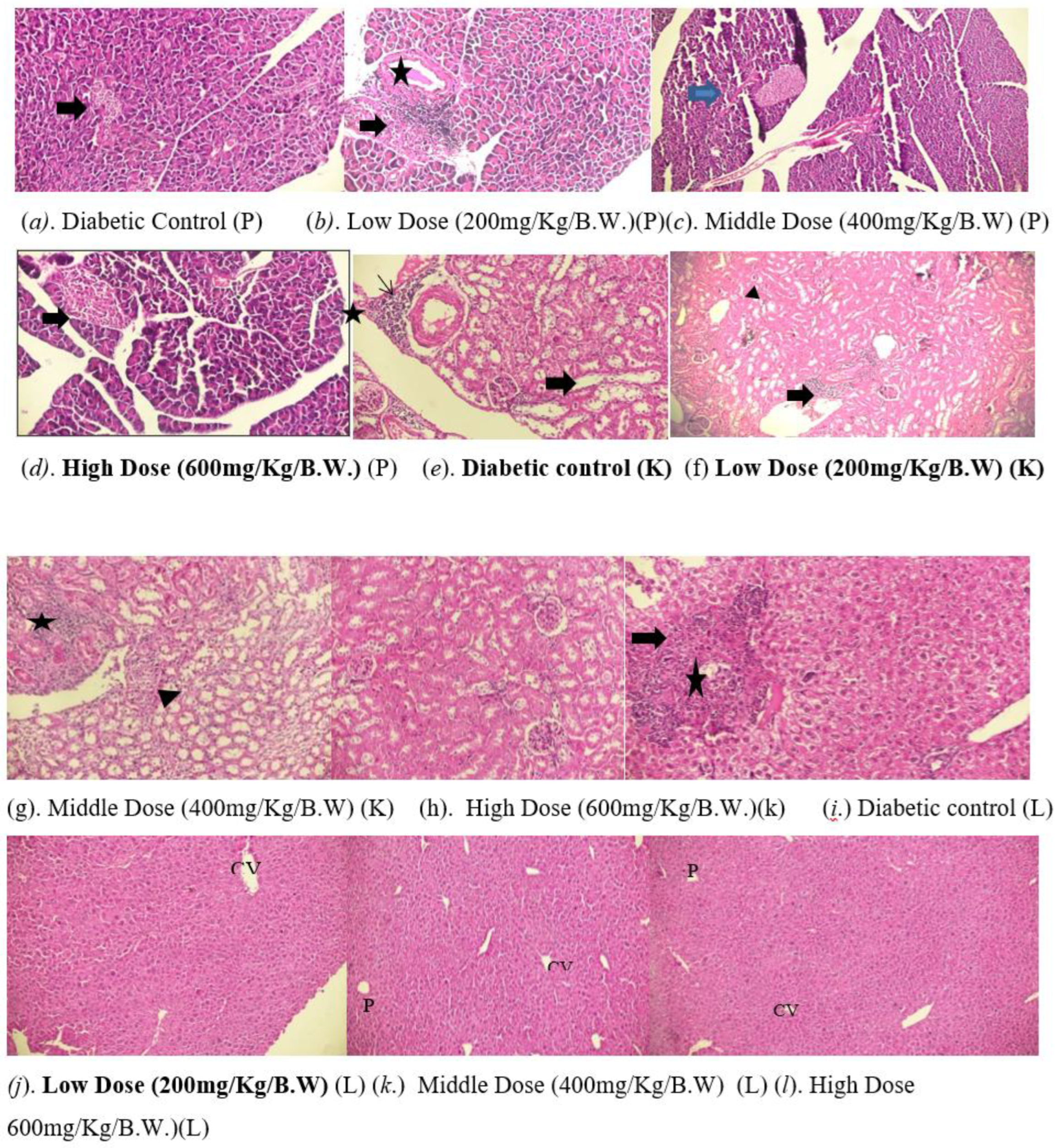
4. Discussion
5. Conclusion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ALP | Alkaline phosphatase |
| ALT | Alanine Aminotransferase |
| AST | Aspartate Transaminase |
| DM | Diabetes Mellitus |
| HDL | High Density lipoprotein |
| HGB | Hemoglobin |
| IRB | institutional review board |
| LDL | low-density lipoprotein |
| MIME | Mangifera indica methanol Extract |
| PLT | Platelets |
| RBC | Red blood cells |
| STZ | streptozotocin |
| WBC | White Blood cells |
References
- Ohiagu, F.; Chikezie, P.; Chikezie, C. Pathophysiology of diabetes mellitus complications: Metabolic events and control. Biomedical Research and Therapy, 2021, 8, 4243–4257. [Google Scholar] [CrossRef]
- Owens, D.R.; et al. IDF diabetes Atlas: A worldwide review of studies utilizing retinal photography to screen for diabetic retinopathy from 2017 to 2024 inclusive. Diabetes Research and Clinical Practice, 2025, 226, 112346. [Google Scholar] [CrossRef]
- Wild, S.; et al. Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes care, 2004, 27, 1047–1053. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.; Singhai, A. Nephroprotective activity of Momordica dioica Roxb. in cisplatin-induced nephrotoxicity. Natural Product Research, 2010, 24, 846–854. [Google Scholar] [CrossRef] [PubMed]
- Stenman, S.; et al. Relationship between sulfonylurea dose and metabolic effect. Diabetes, 1990, 39, 108A. [Google Scholar]
- von Schoen-Angerer, T. , et al., Traditional, complementary and integrative healthcare: global stakeholder perspective on WHO's current and future strategy. BMJ Glob Health, 2023. 8(12).
- Hoenders, R.; et al. A review of the WHO strategy on traditional, complementary, and integrative medicine from the perspective of academic consortia for integrative medicine and health. Front Med (Lausanne), 2024, 11, 1395698. [Google Scholar] [CrossRef]
- Kalita, P. An overview on Mangifera indica: Importance and its various pharmacological action. PharmaTutor, 2014, 2, 72–76. [Google Scholar]
- Jain, C.; et al. Anti-diabetic potential of flavonoids and other crude extracts of stem bark of Mangifera indica Linn: a comparative study. J Sci Innov Res, 2014, 3, 21–27. [Google Scholar] [CrossRef]
- Irondi, E.A.; Oboh, G.; Akindahunsi, A.A. Antidiabetic effects of Mangifera indica Kernel Flour-supplemented diet in streptozotocin-induced type 2 diabetes in rats. Food Sci Nutr, 2016, 4, 828–839. [Google Scholar] [CrossRef]
- Loan, N.T.T.; et al. Purification Process of Mangiferin from Mangifera indica L. Leaves and Evaluation of Its Bioactivities. Processes, 2021. 9(5).
- Kaur, J.; et al. Comprehensive Analysis and Characterization of Mangifera indica L. Leaf Powder. Food Science & Nutrition, 2025. 13(6).
- Bitwell, C.; et al. A review of modern and conventional extraction techniques and their applications for extracting phytochemicals from plants. Scientific African, 2023, 19, e01585. [Google Scholar] [CrossRef]
- Brosius, F. , Low-Dose Streptozotocin Induction Protocol (mouse) v1. 2019.
- Carbon Dioxide for Euthanasia_ Concerns Regarding Pain and Distre.pdf>.
- Parasuraman, S.; Raveendran, R.; Kesavan, R. Blood sample collection in small laboratory animals. J Pharmacol Pharmacother, 2010, 1, 87–93. [Google Scholar] [PubMed]
- Xie, F.; et al. Good Preclinical Bioanalytical Chemistry Requires Proper Sampling from Laboratory Animals: Automation of Blood and Microdialysis Sampling Improves the Productivity of LC/MSMS. Analytical Sciences, 2003, 19, 479–485. [Google Scholar] [CrossRef] [PubMed]
- McLaren, G.; et al. Body weight change as a measure of stress: a practical test. Animal Welfare, 2004, 13, 337–341. [Google Scholar] [CrossRef]
- Schuppli, C.A. Decisions about the use of animals in research: Ethical reflection by Animal Ethics Committee members. Anthrozoös, 2011, 24, 409–425. [Google Scholar] [CrossRef]
- Klonoff, D.C.; et al. Importance of FDA-Integrated Continuous Glucose Monitors to Ensure Accuracy of Continuous Glucose Monitoring. J Diabetes Sci Technol, 2025, 19, 1392–1399. [Google Scholar] [CrossRef]
- Litvinova, E.A.; et al. Modification of Fecal Bacteria Counts and Blood Immune Cells in the Offspring of BALB/c and C57BL/6 Mice Obtained through Interstrain Mouse Embryo Transfer. J Am Assoc Lab Anim Sci, 2020, 59, 401–410. [Google Scholar] [CrossRef]
- Rathkolb, B.; et al. Blood Collection from Mice and Hematological Analyses on Mouse Blood. Curr Protoc Mouse Biol, 2013, 3, 101–19. [Google Scholar] [CrossRef]
- Wolford, S.T.; et al. Reference range data base for serum chemistry and hematology values in laboratory animals. J Toxicol Environ Health, 1986, 18, 161–88. [Google Scholar] [CrossRef]
- Aguila, M.B.; et al. Lipid metabolism in rats fed diets containing different types of lipids. Arq Bras Cardiol, 2002, 78, 25–38. [Google Scholar] [CrossRef] [PubMed]
- Wiedmeyer, C.E.; Ruben, D.; Franklin, C. Complete blood count, clinical chemistry, and serology profile by using a single tube of whole blood from mice. J Am Assoc Lab Anim Sci, 2007, 46, 59–64. [Google Scholar]
- Ahmed, E. , Histological technique quick review. 2022.
- Slaoui, M. and L. Fiette, Histopathology procedures: from tissue sampling to histopathological evaluation, in Drug Safety Evaluation: Methods and Protocols. 2010, Springer. p. 69-82.
- Etuk, E. Animals models for studying diabetes mellitus. Agric Biol JN Am, 2010, 1, 130–134. [Google Scholar]
- Zafar, M. and S.N.-u.-H. Naqvi, Effects of STZ-induced diabetes on the relative weights of kidney, liver and pancreas in albino rats: a comparative study. International Journal of Morphology, 2010. 28(1).
- Yuan, H.; et al. The traditional medicine and modern medicine from natural products. Molecules, 2016, 21, 559. [Google Scholar] [CrossRef] [PubMed]
- Arumugam, G.; Manjula, P.; Paari, N. A review: Anti diabetic medicinal plants used for diabetes mellitus. Journal of Acute Disease, 2013, 2, 196–200. [Google Scholar] [CrossRef]
- Parvez, G.M. Pharmacological activities of mango (Mangifera indica): A review. Journal of Pharmacognosy and phytochemistry, 2016, 5, 1. [Google Scholar]
- Gondi, M.; et al. Anti-diabetic effect of dietary mango (Mangifera indica L.) peel in streptozotocin-induced diabetic rats. Journal of the Science of Food and Agriculture, 2015, 95, 991–999. [Google Scholar] [CrossRef]
- S Mohamed, E.; et al. Evaluation of Hypoglycemic and Hypolipidemic potentials of Aqueous Extracts of Guava and Mango leaves in Streptozotocin Diabetic Rats. مجلة الاقتصاد المنزلي, 2025. 41(4): p. 1-24.
- Fernández-Ochoa, Á.; et al. Evaluation of metabolic changes in liver and serum of streptozotocin-induced diabetic rats after Mango diet supplementation. Journal of Functional Foods, 2020, 64, 103695. [Google Scholar] [CrossRef]
- Moon, D.O. Plant-derived flavonoids as AMPK activators: unveiling their potential in type 2 diabetes management through mechanistic insights, docking studies, and pharmacokinetics. Applied Sciences, 2024, 14, 8607. [Google Scholar] [CrossRef]
- Sathyanarayan, S.; Pillai, K.S. Evaluation of antioxidant and Antidiabetic properties of goa 111, a mixture of Gymnema sylvestre, Ocimum sanctum and Azadirachta indica extract (1: 1: 1): An in vitro approach. Journal of Pharmacognosy and Phytochemistry, 2019, 8, 1889–1895. [Google Scholar]
- Rajesh, M. , Screening of Hypoglycemic Effects of Various Extracts of Costus Speciosus and Aqueous Extract of Mangifera Indica in Normal and Diabetic Rats. 2006, Rajiv Gandhi University of Health Sciences (India).
- Xu, D.; et al. Antioxidant activities of quercetin and its complexes for medicinal application. Molecules, 2019, 24, 1123. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; et al. Therapeutic effects of berberine on liver fibrosis are associated with lipid metabolism and intestinal flora. Frontiers in Pharmacology, 2022, 13, 814871. [Google Scholar] [CrossRef] [PubMed]
- Mada, S.; et al. Hepatoprotective effect of Momordica charantia extract against CCl4 induced liver damage in rats. British Journal of Pharmaceutical Research, 2014, 4, 368. [Google Scholar] [CrossRef]
- Yan, Y.-M.; et al. Anti-diabetic nephropathy compounds from Cinnamomum cassia. Journal of ethnopharmacology, 2015, 165, 141–147. [Google Scholar] [CrossRef]
- Sapin, A.B.; et al. Evaluation of the Antioxidant and Anti-diabetic Bioactivities of Natural Phenolics from Mango (Mangifera indica Linn) Branches. Philippine Journal of Science, 2021. 150(3).
- Varshney, R.; et al. A comparative analysis of various flavonoids in the regulation of obesity and diabetes: An in vitro and in vivo study. Journal of Functional Foods, 2019, 59, 194–205. [Google Scholar] [CrossRef]
- Emejulu, A.; et al. Renal and hepato-protective effects of Irvingia gabonensis juice on sodium fluoride-induced toxicity in Wistar rats. J Clin Toxicol, 2016, 6, 2161–0495. [Google Scholar] [CrossRef]
- Núñez-Sellés, A.J.; et al. The paradox of natural products as pharmaceuticals: Experimental evidences of a mango stem bark extract. Pharmacological Research, 2007, 55, 351–358. [Google Scholar] [CrossRef] [PubMed]
- KHUMYAT, M. and D. Pekthong, Inhibitory and inductive effects of Bacopa monnieri standardized extract and its constituents on human liver cytochrome P450. 2021, Naresuan University.
| Normal control | Diabetic control |
Low dose (200 mg/kg) | Medium dose (400 mg/kg) | High dose (600 mg/kg) | Glebinglemid 6mg/kg/b.w | |
| HGB | 14.12 | 6.5 | 8.25 | 9.0 | 12.67 | 11.55 |
| RBC’s | 6.52 | 3.31 | 4.31 | 4.9 | 6.00 | 5.5 |
| WBC’s | 7.52 | 4.00 | 5.00 | 7.25 | 8.15 | 7.85 |
| Platelet count | 1250 | 865.00 | 950 | 1020.00 | 1130 | 1,000 |
| Groups/Treatments | Total Cholesterol | Triglycerides | HDL Cholesterol | LDL Cholesterol |
| I: Normal | 67.28 ± 2.80 | 85.42 ±5.16 | 38.32 ± 2.90 | 22.25± 2.90 |
| II: Diabetic control | 251.73 ± 7.60 | 165.52 ±4.71 | 26.23 ±2.20 | 159.36± 4.32 |
| III: Diabetic + MIME (200 mg/kg) | 120.32 ±11.20 | 115.24 ±4.51 | 35.24 ±3.50 | 80.55± 3.15 |
| IV: Diabetic + MIME (400 mg/kg) | 110.47 ±4.70 | 92.24 ±6.32 | 41.35 ±2.60 | 60.17± 3.42 |
| V: Diabetic + MIME (600 mg/kg) | 105.47 ±4.7 | 85.24 ±6.32 | 46.35 ±2.60 | 45.23± 3.33 |
| VI: Gilibenglmaid(6 mg/kg. b.w.) | 97.72 ±5.30 | 84.47 ±4.50 | 40.28 ±4.80 | 55.28± 4.3 |
| Groups/Treatments | Liver function tests and total protein | ||||
| T.Protein(g/dL) | Bilirubin(mg/dL) | AST(U/L) | ALT(U/L) | ALP(U/L) | |
| I: Normal | 8.26 ±2.18 | 0.44 ±1.24 | 39.22 ±2.34 | 45.35 ±3.49 | 125.35 ±3.43 |
| II: Diabetic control | 6.26 ±1.29 | 1.00 ±1.29 | 112.26 ±4.87 | 113.23 ±3.45 | 205.26 ±4.37 |
| III: Diabetic + MIME (200 mg/kg) | 6.77 ±1.67 | 0.62 ±0.34 | 74.54 ±3.53 | 66.26 ±3.25 | 154.55 ±3.25 |
| IV: Diabetic + MIME (400 mg/kg) | 7.22 ±2.34 | 0.50 ±1.26 | 49.23 ±3.58 | 48.68 ±3.75 | 124.35 ±3.68 |
| V: Diabetic + MIME (600 mg/kg) | 8.12 ±2.34 | 0.40 ±1.26 | 42.23 ±3.58 | 41.68 ±3.75 | 118.35 ±3.68 |
| VI:Gilibenglmaid(6 mg/kg. b.w.) | 7.21 ±1.25 | 0.38 ±1.83 | 45.56 ±3.54 | 58.86 ±3.58 | 125.25 ±3.25 |
| Groups/Treatments | Serum urea (mg/dL) | Serum creatinine (mg/dL) |
| I: Normal | 30.25 ±1.58 | 0.63 ±1.34 |
| II: Diabetic | 61.24 ±1.57 | 1.97 ±0.54 |
| III: Diabetic + MIME (200 mg/kg) | 45.24 ±1.48 | 0.85 ±1.08 |
| IV: Diabetic + MIME (400 mg/kg) | 47.35 ±0.59 | 0.71 ±0.43 |
| VI: Diabetic + MIME (600 mg/kg) | 36.35 ±0.59 | 0.70 ±0.43 |
| V: Diabetic + gelibenclamide (6 mg/kg.b.w.) | 37.33 ±0.87 | 1.25 ±0.62 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




