Submitted:
28 November 2025
Posted:
28 November 2025
You are already at the latest version
Abstract

Keywords:
1. Introduction
| 2 Solvent | cavoxin/cavoxone | photopiperazines |
| 2.1 DMSO | pyrrolizin-3-ones | aspochracin/sclerotiolides |
| migrastatins/dorrigocins | 2.7 dichloromethane | clavosines/calyculins |
| discorhabdins | bromotyramines | 5.2 Photooxidation |
| dendrillic acids | 2.8 benzene | cadinanes |
| methylsulfonated polyketides | theonellastrols | 5.3 Photoreactive |
| cerulenin | 2.9 ethyl acetate | chetomins |
| bisanthraquinones | sorbicillinol/sorbivetone | talaromycins/purpactins |
| glyclauxins | 2.10 aprotic vs protic | 6 Air oxidation |
| aculeaxanthones/chrysoxanthones | oxandrastins | ketidocillinones |
| versixanthones | alaeolide | pseudopyronines |
| secalonic acid/parnafungins | pratensilins | norpectinatone |
| 2.2 pyridine | 3 Heat | linfuranones |
| acremoxanthone/acremonidins | psammaplins/bastadins | hyafurones/aurafurones |
| 2.3 methanol | creolophins | avermectins |
| brevianamides | neobulgarones | penilumamides |
| talaronins | 4 pH | 7 Acetal/ketal equilibration |
| pyrasplorins | 4.1 basic | okichromanone |
| penicipyridones | pestalone/pestalachloride | sphydrofuran |
| varacins | neoenterocins | 8 Trans-esterification |
| epithiodiketopiperazines | asperazepanones | kipukasins |
| eleutherobins/caribaeoranes | hydroxybrevianamides | glenthmycins |
| 2.4 acetone | salinosporamides | amaurones |
| kutzneridines | 4.2 acidic | 9 Epimerization |
| enamidonins/K97-0239A and B | enterocins | aspergillazines |
| autucedines | serratiochelin | quinolactacins |
| madurastatins | franklinolides | 10 Cryptic natural products |
| drimanes | oxanthromicins/eurotones | N-amino-l-proline methyl ester |
| duclauxin/verruculosins | 4.3 silica gel | prolinimines |
| 2.5 acetonitrile | sphydrofurans | N-amino-anthranilic acid |
| talcarpones | duclauxin/bacillisporins | penipacids |
| 2.6 chloroform | xenoclauxin/talaromycesone B | elansolids |
| greensporones | xanthepinone | 11 Bioassay biotransformation |
| alkyl resorcinols | daldinones | abyssomicins |
| azodyrecins | 5 Light | roseopurpurins |
| schipenindolenes | 5.1 Photoisomerization | kendomycin/goondomycins |
| shearinines | pyranpolyenolides |
2. Solvents
2.1. DMSO
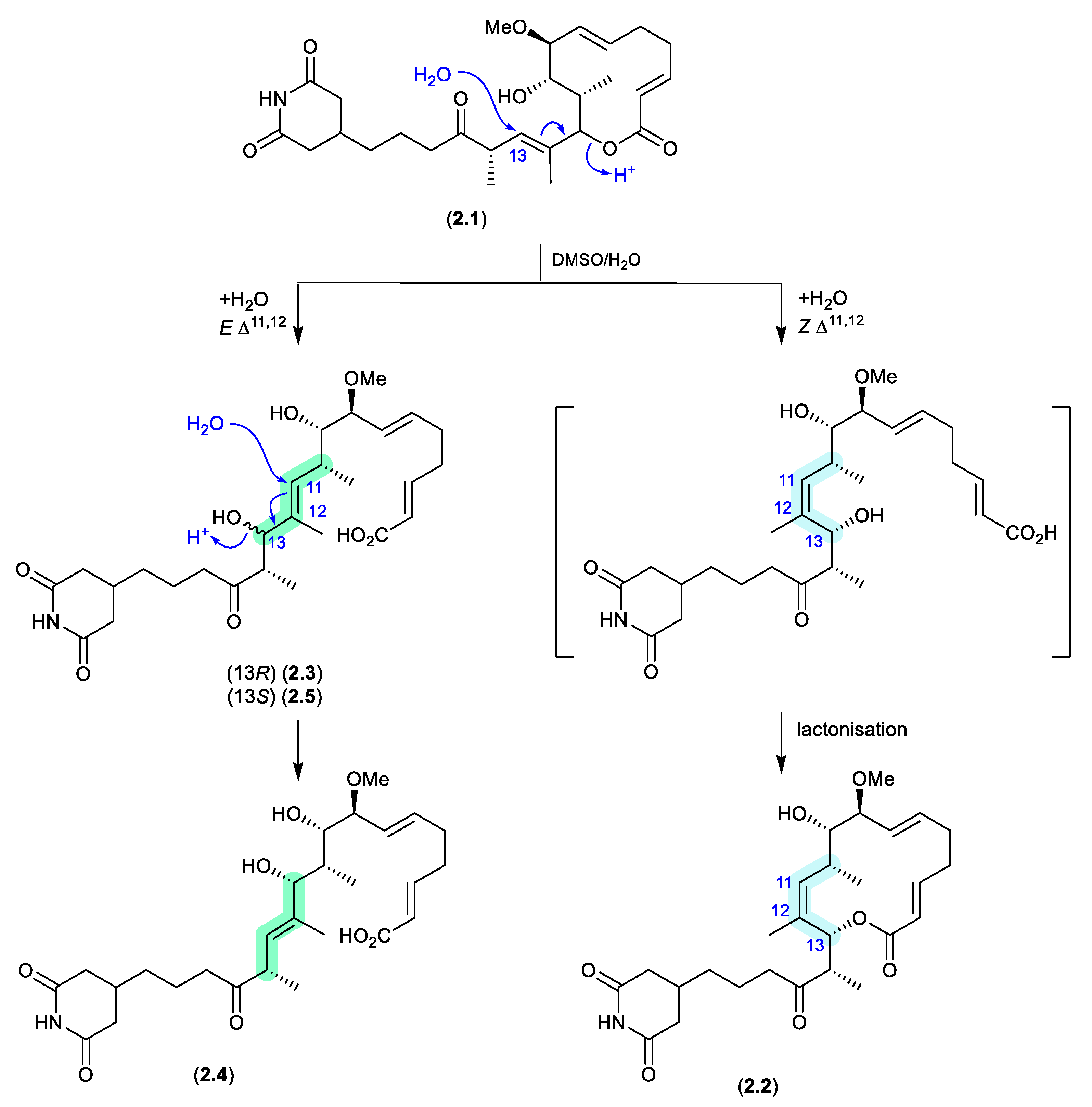
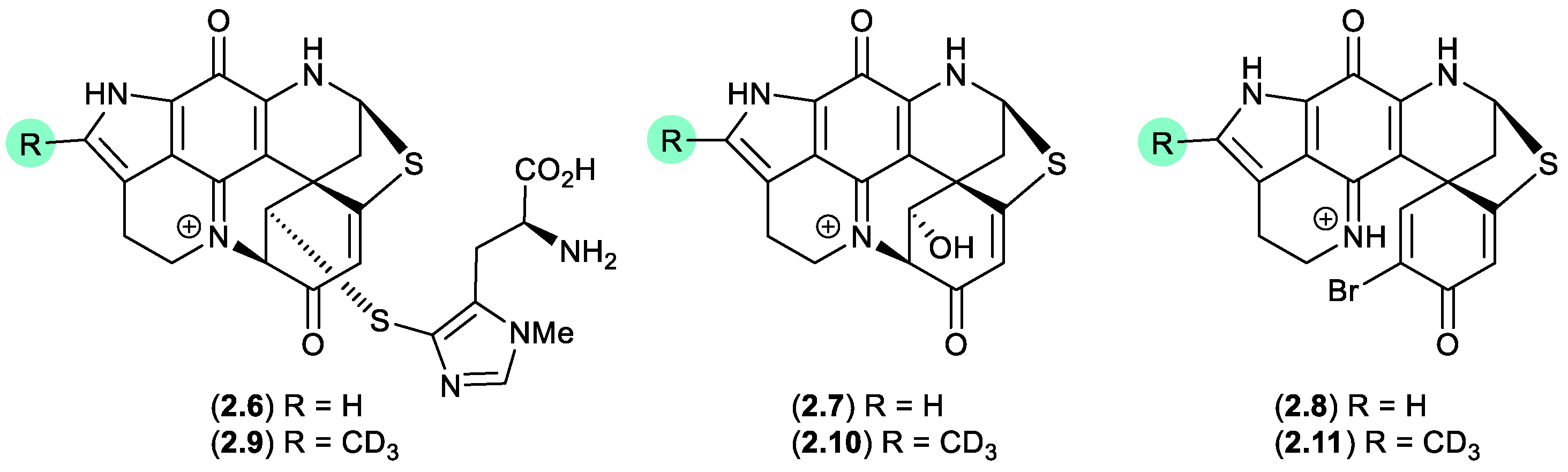
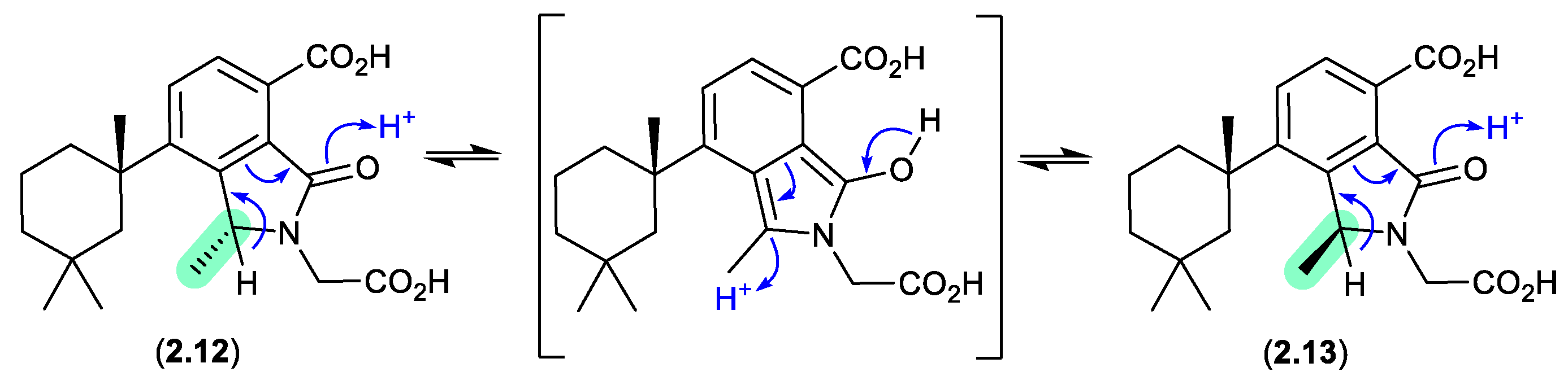
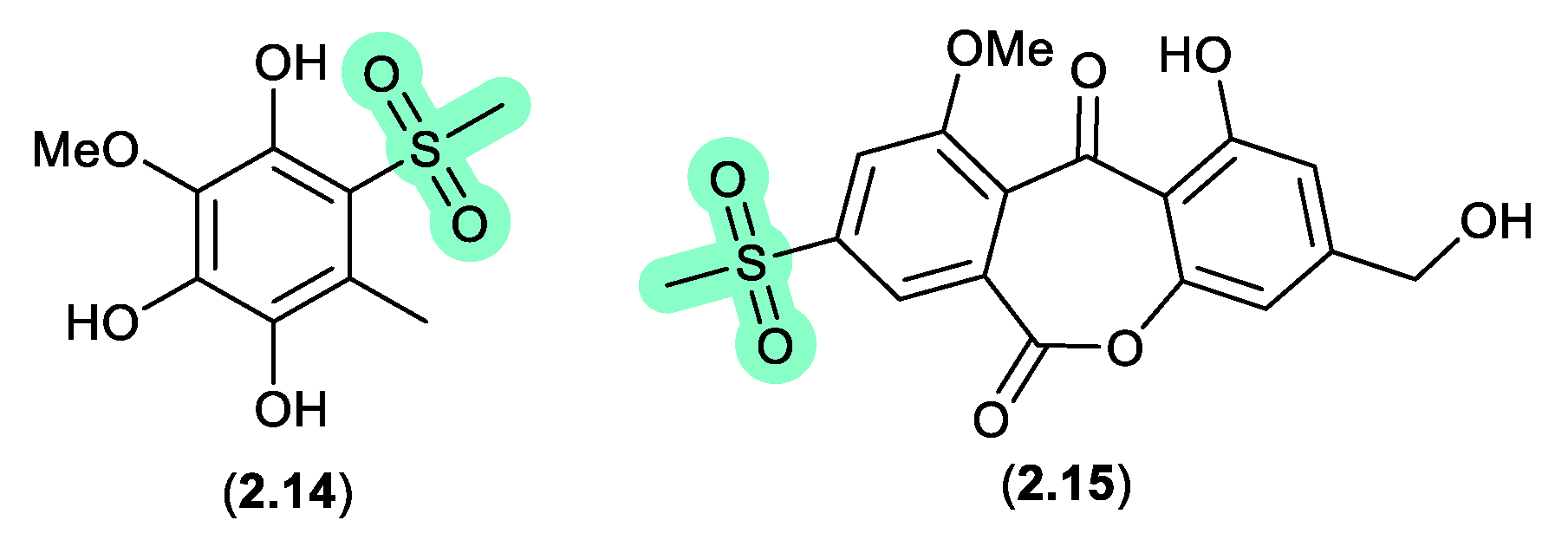
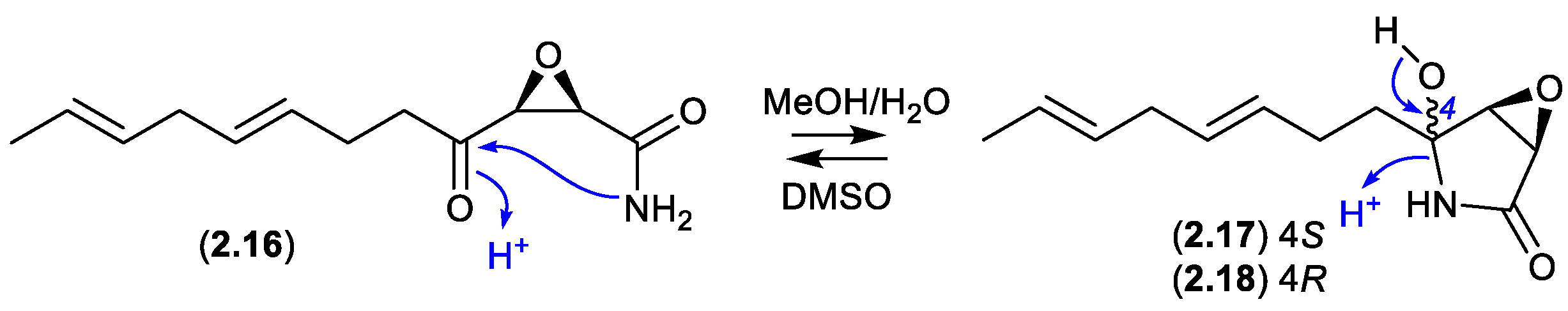
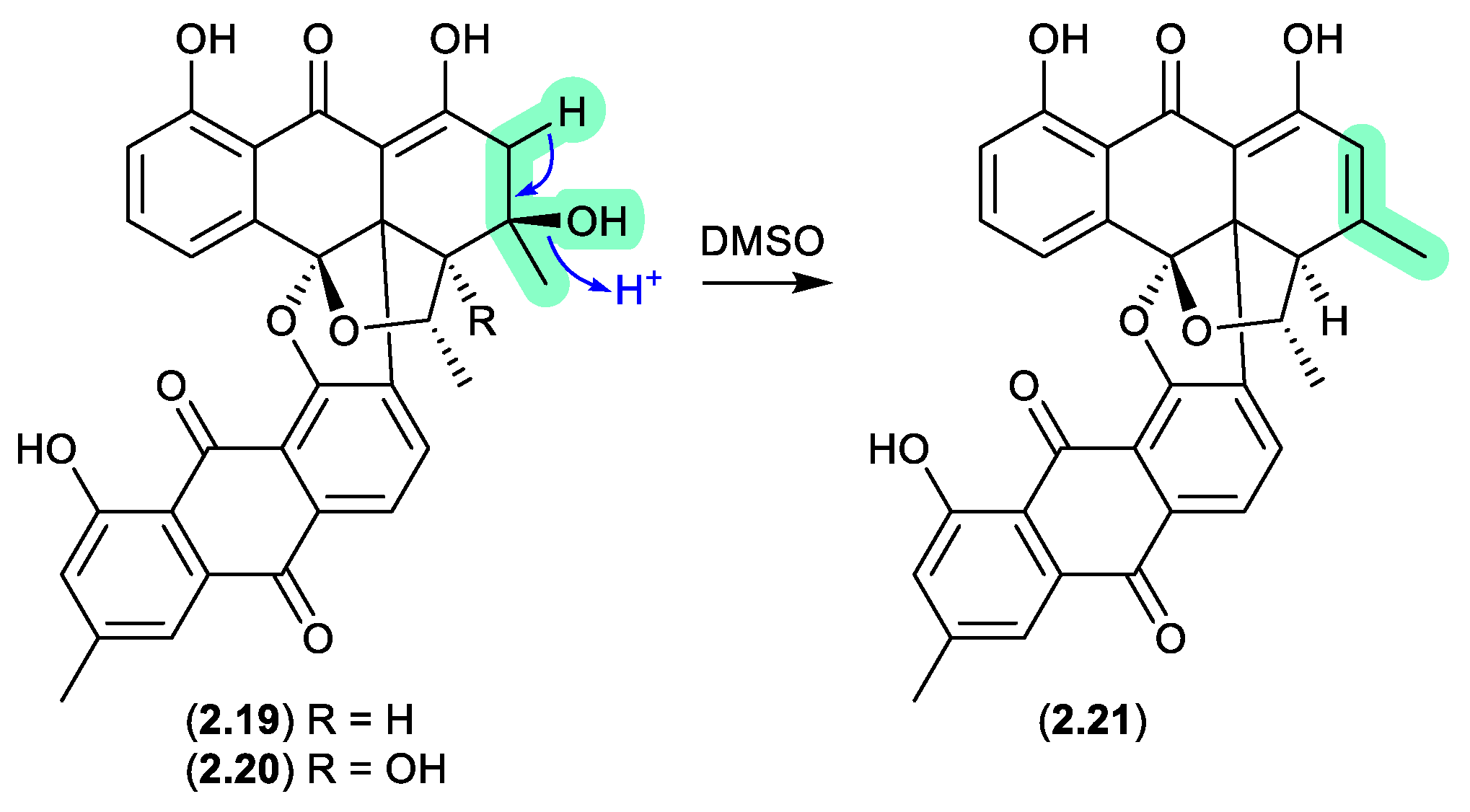
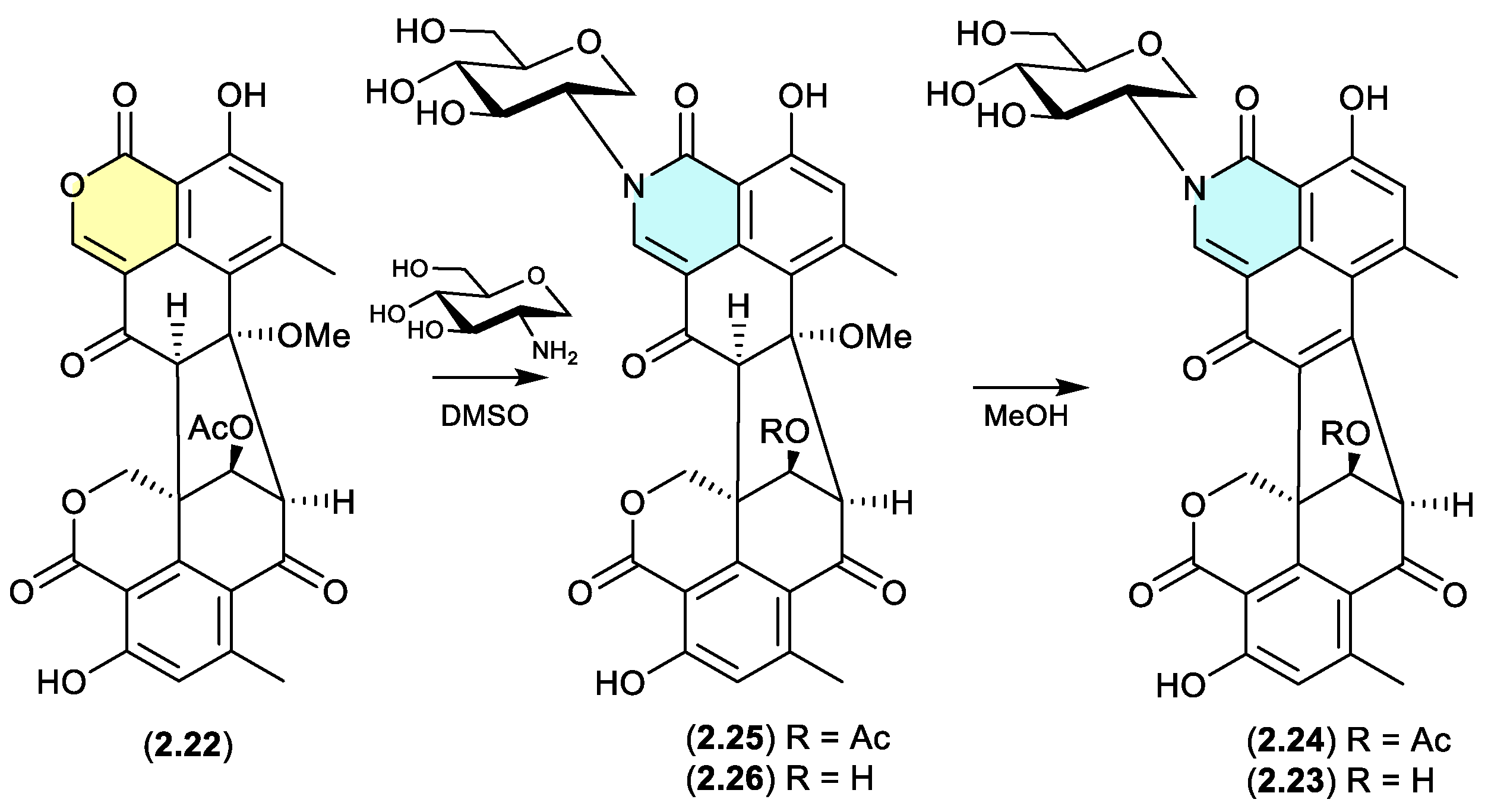
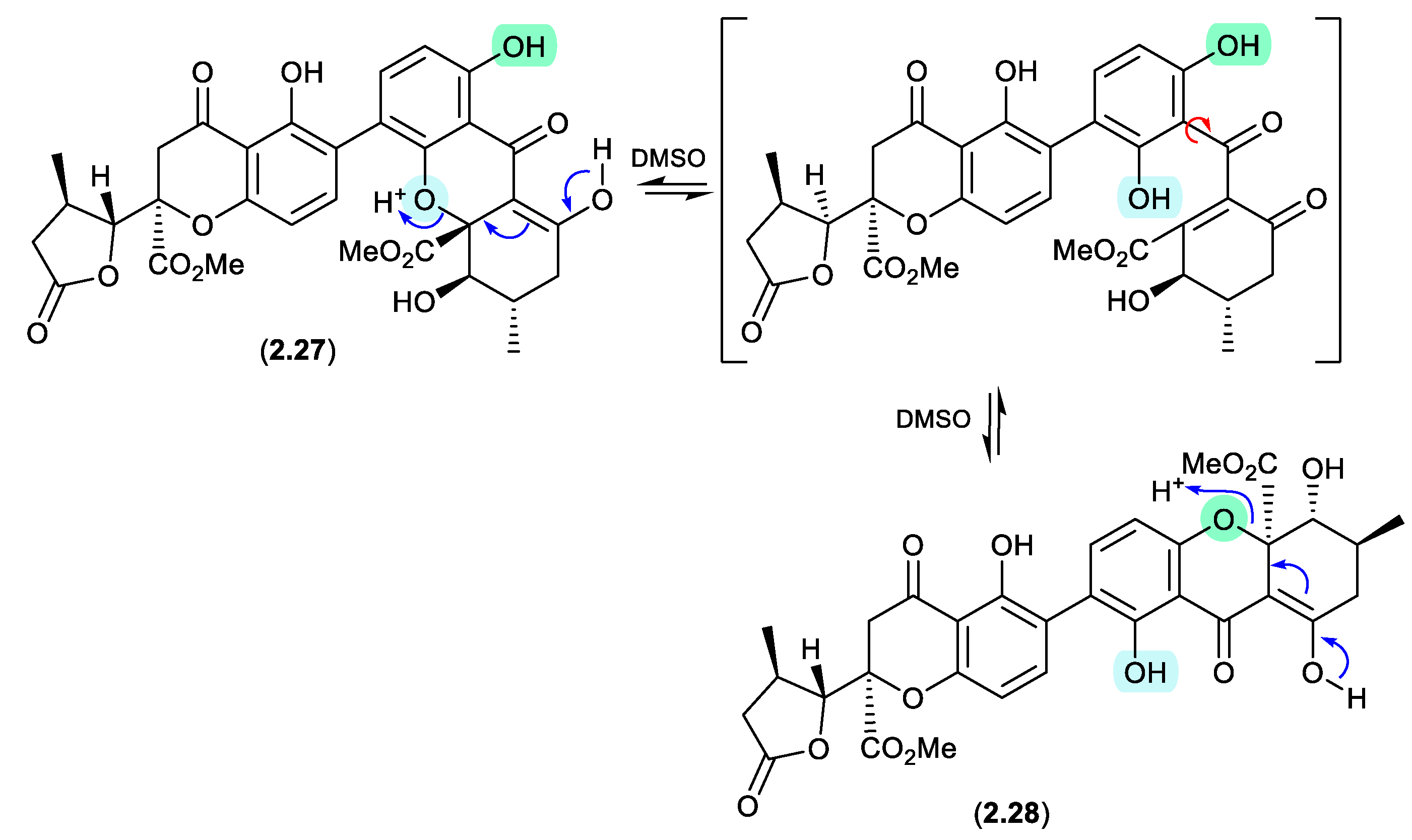
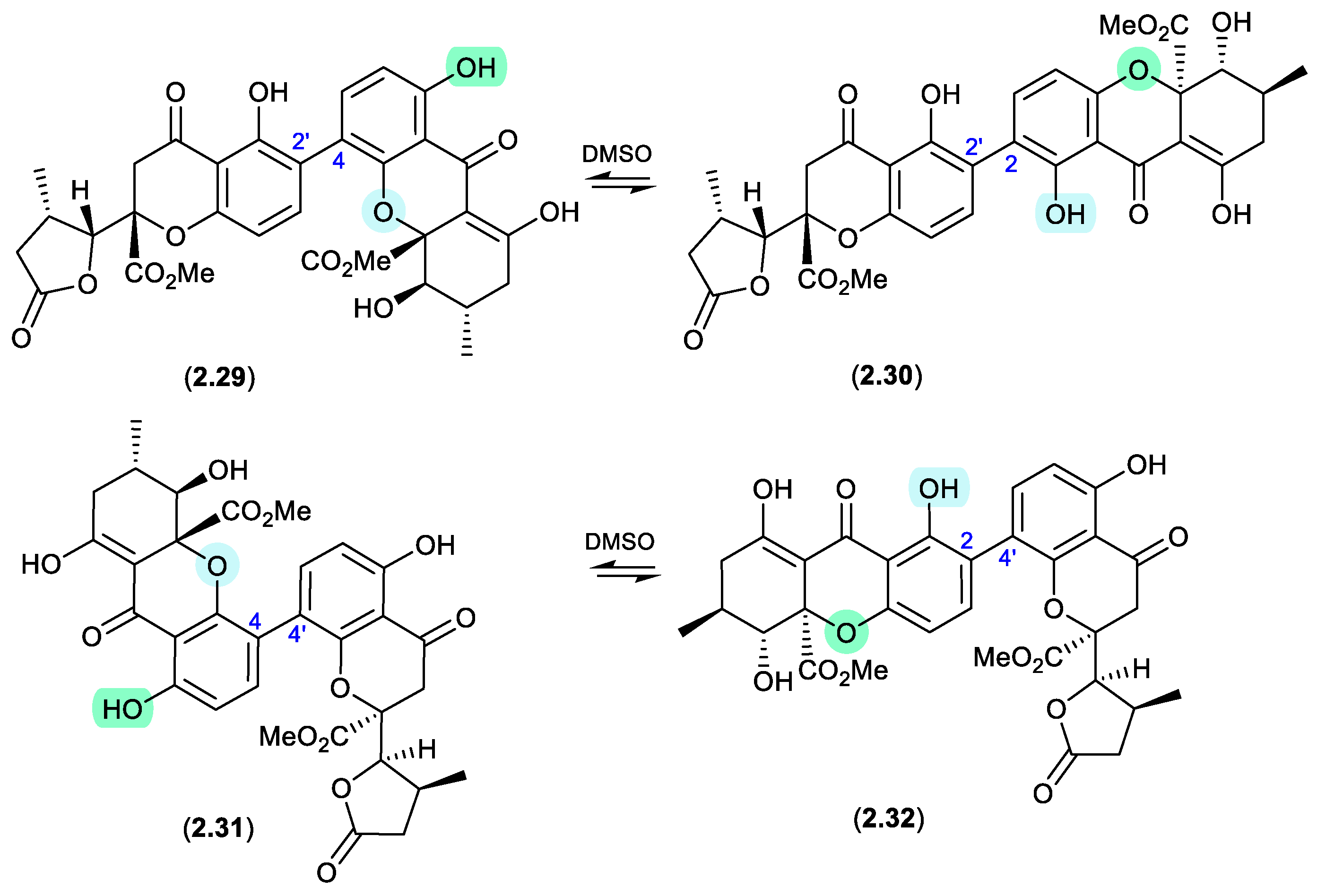
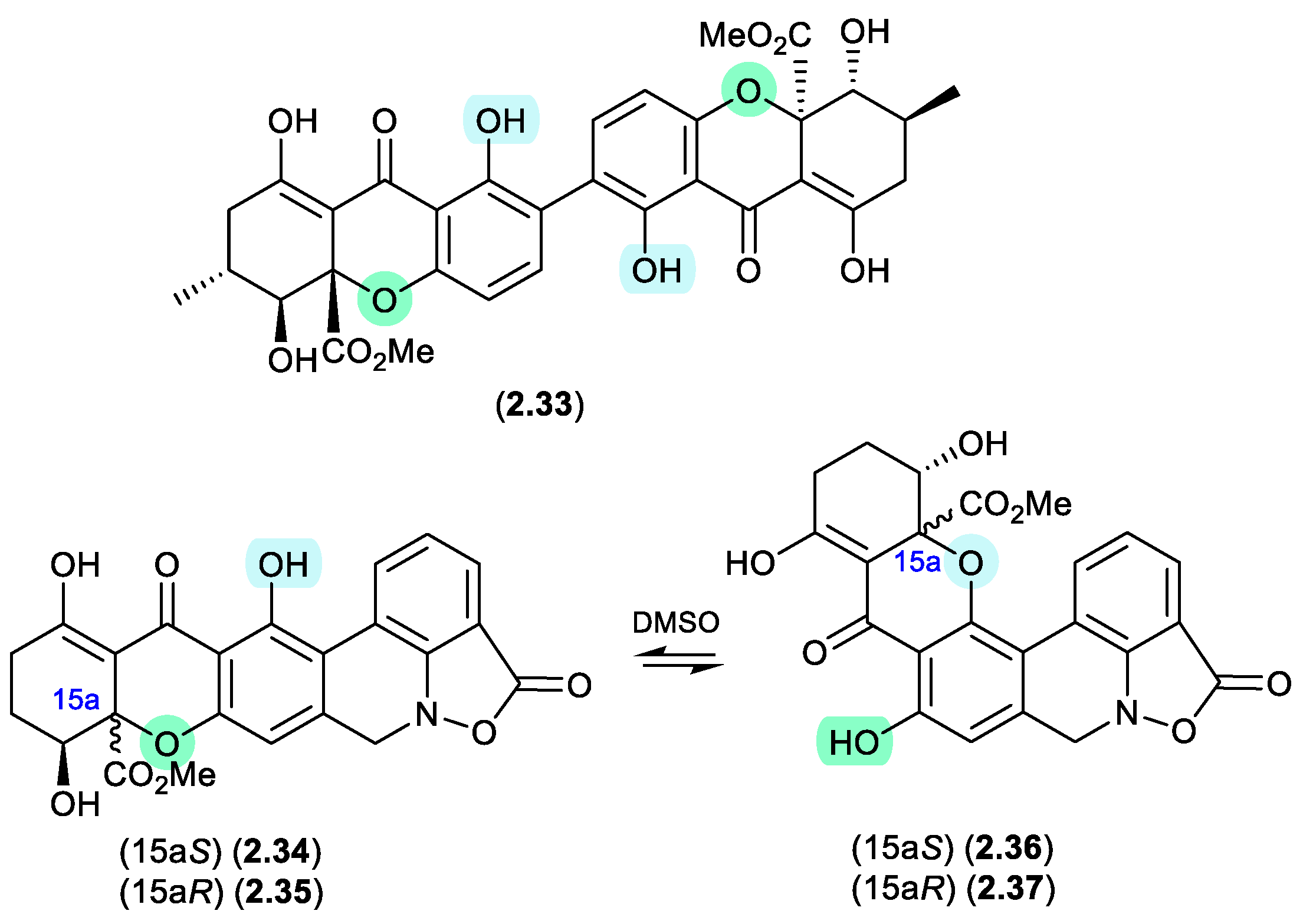
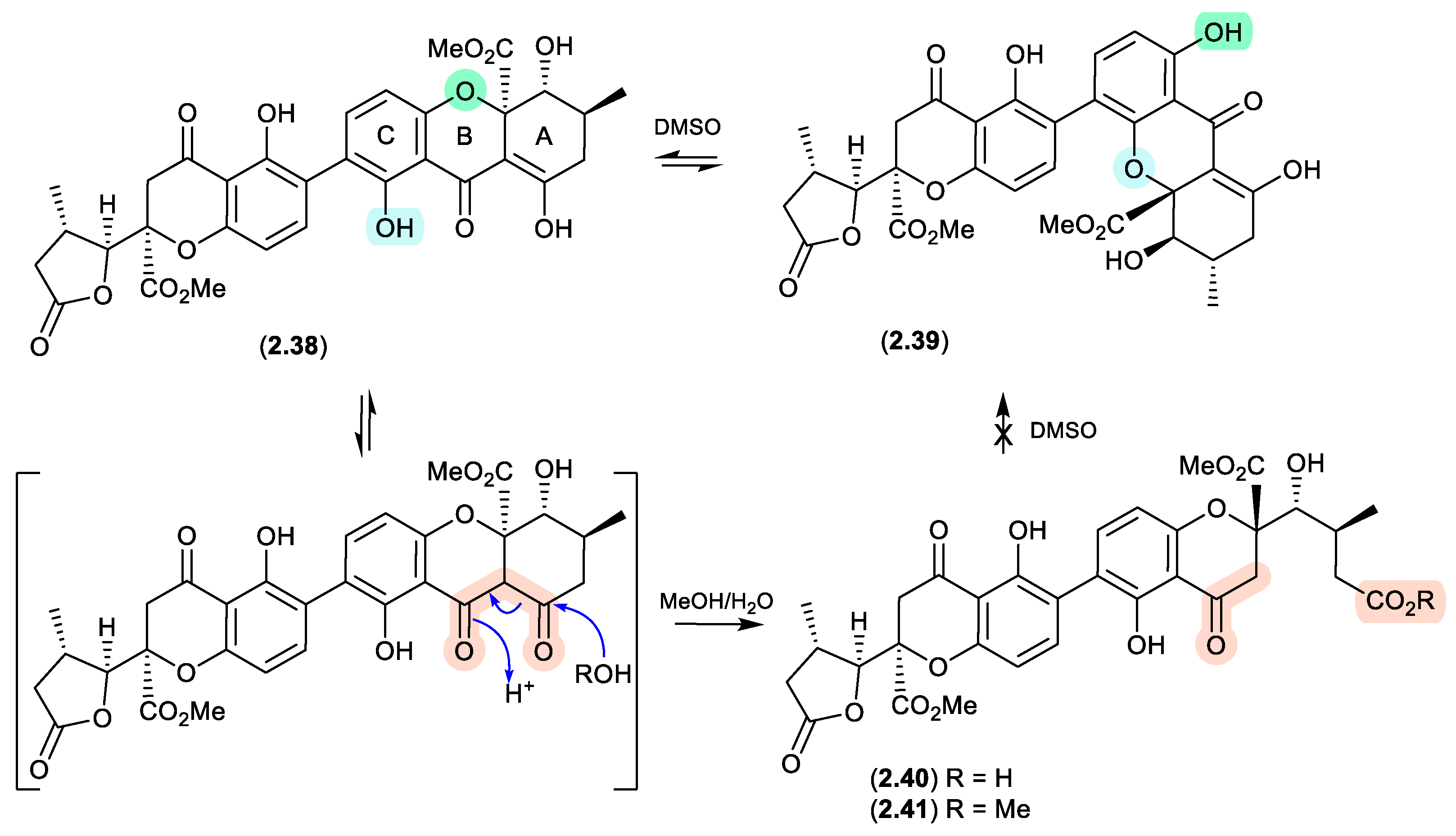
2.2. Pyridine
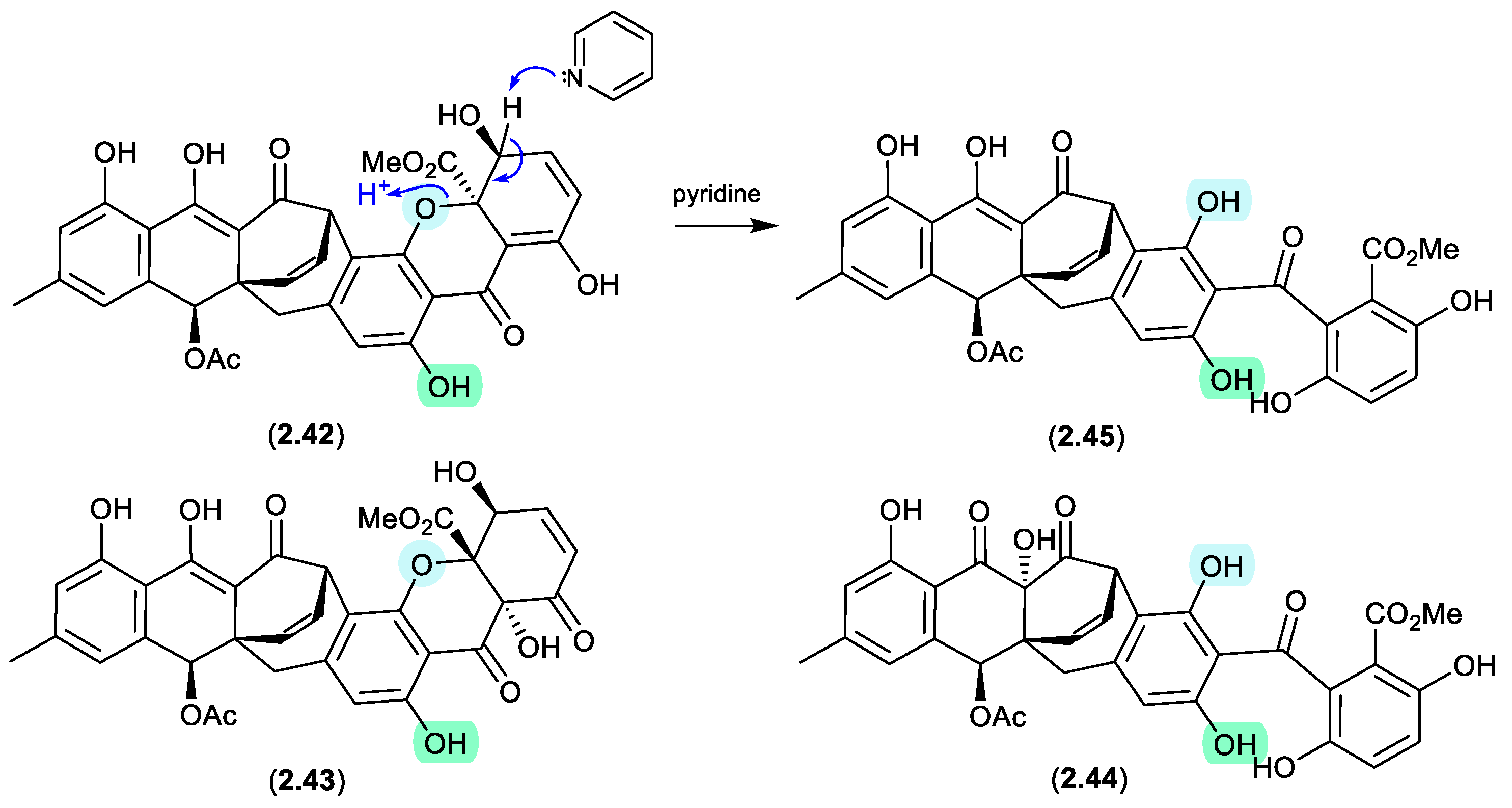
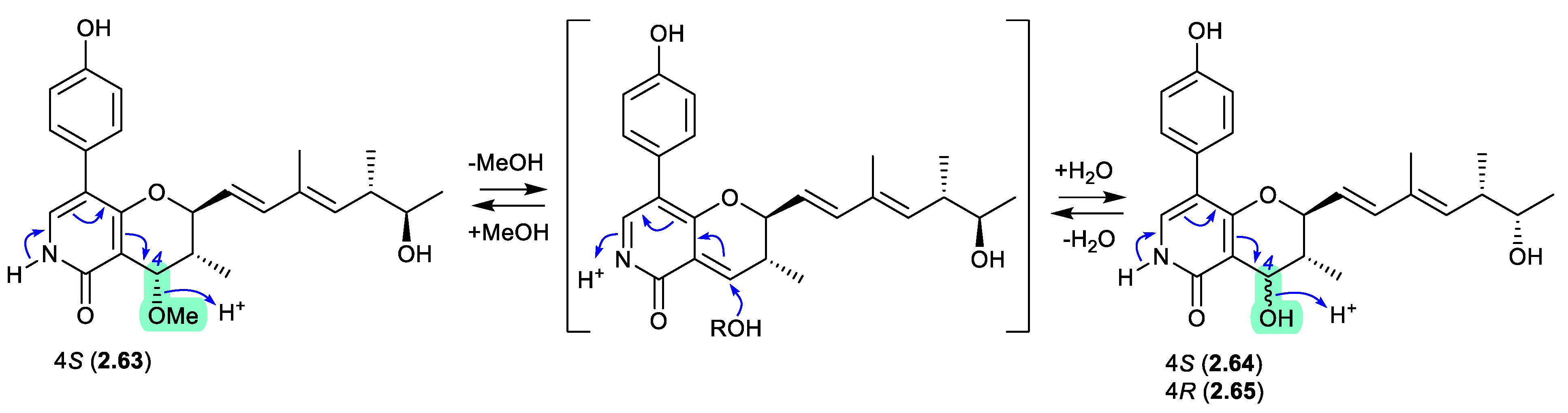
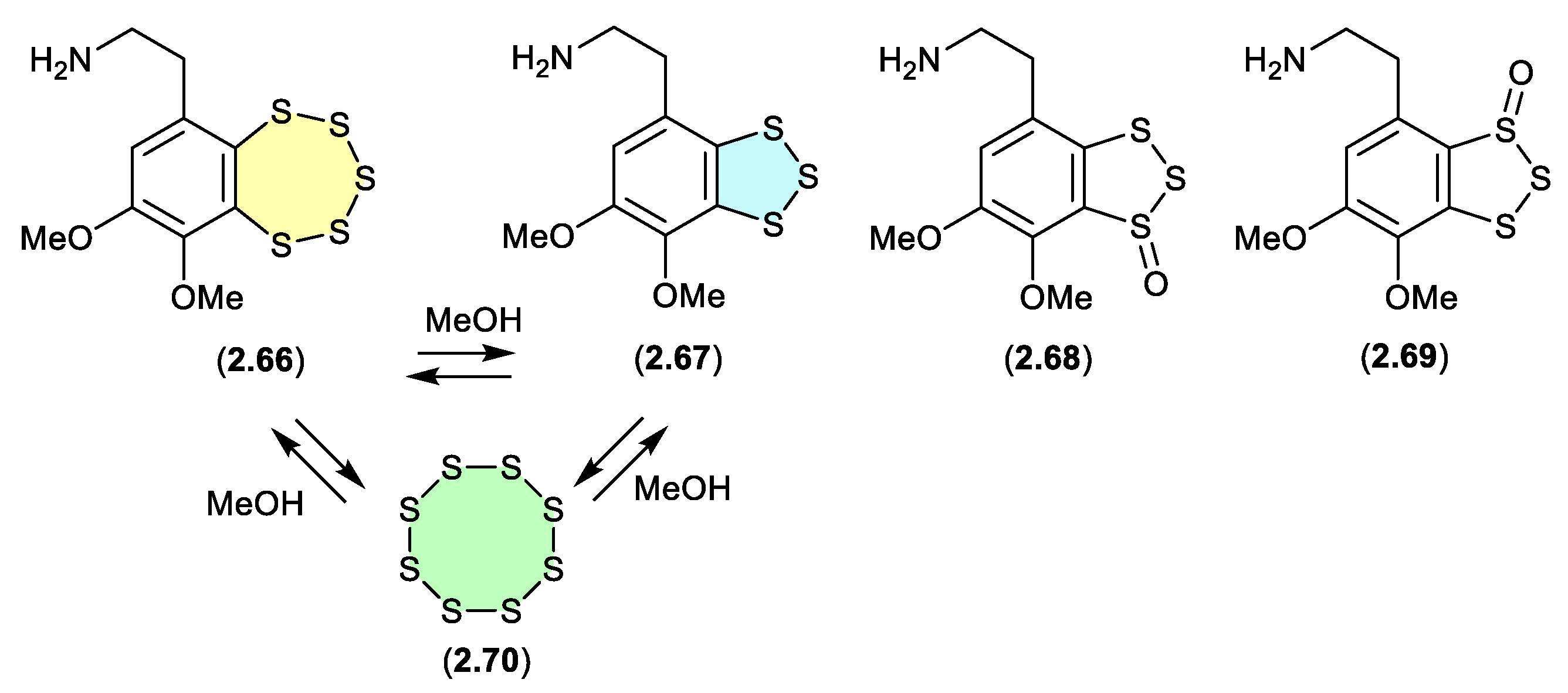
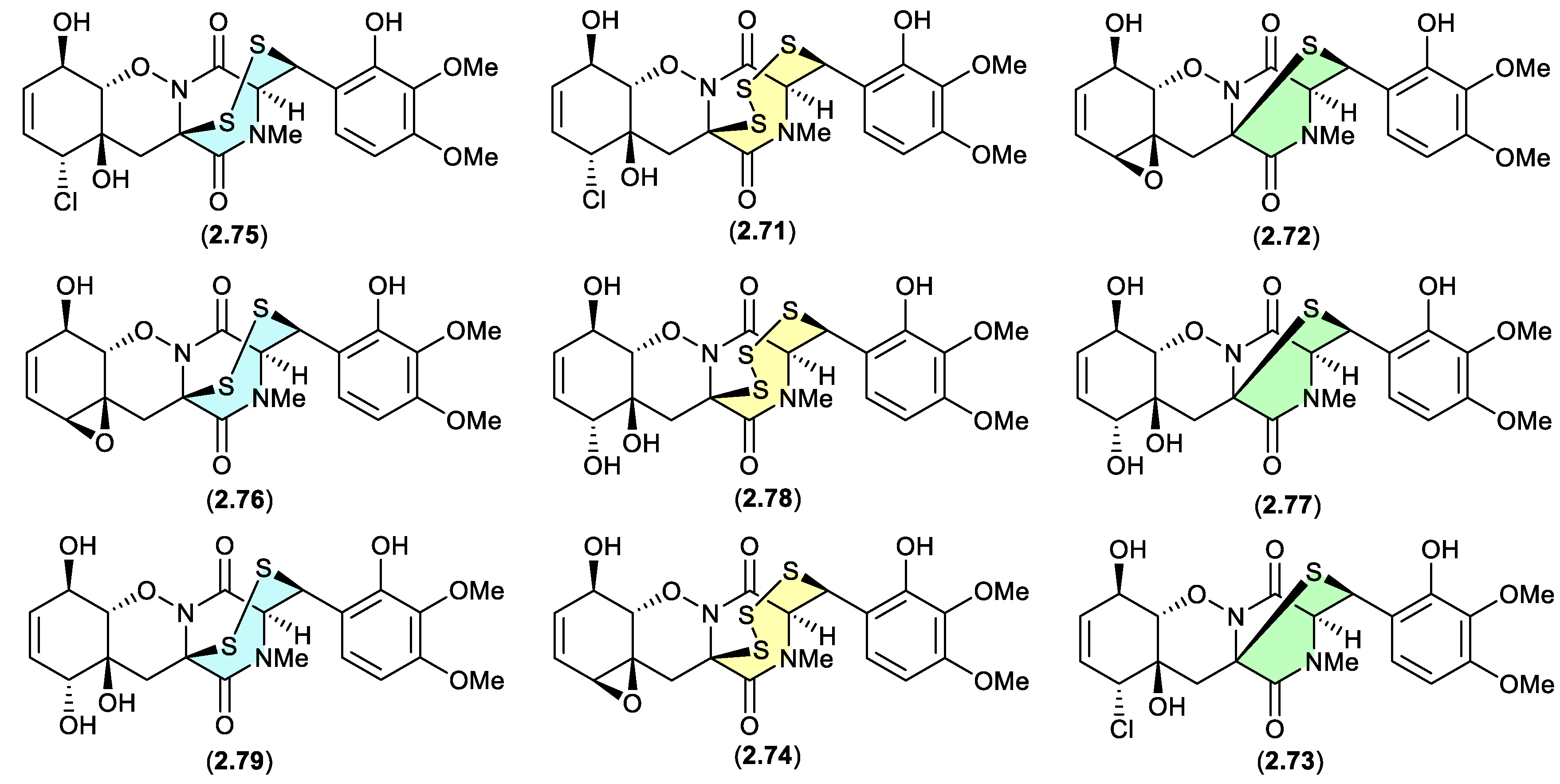
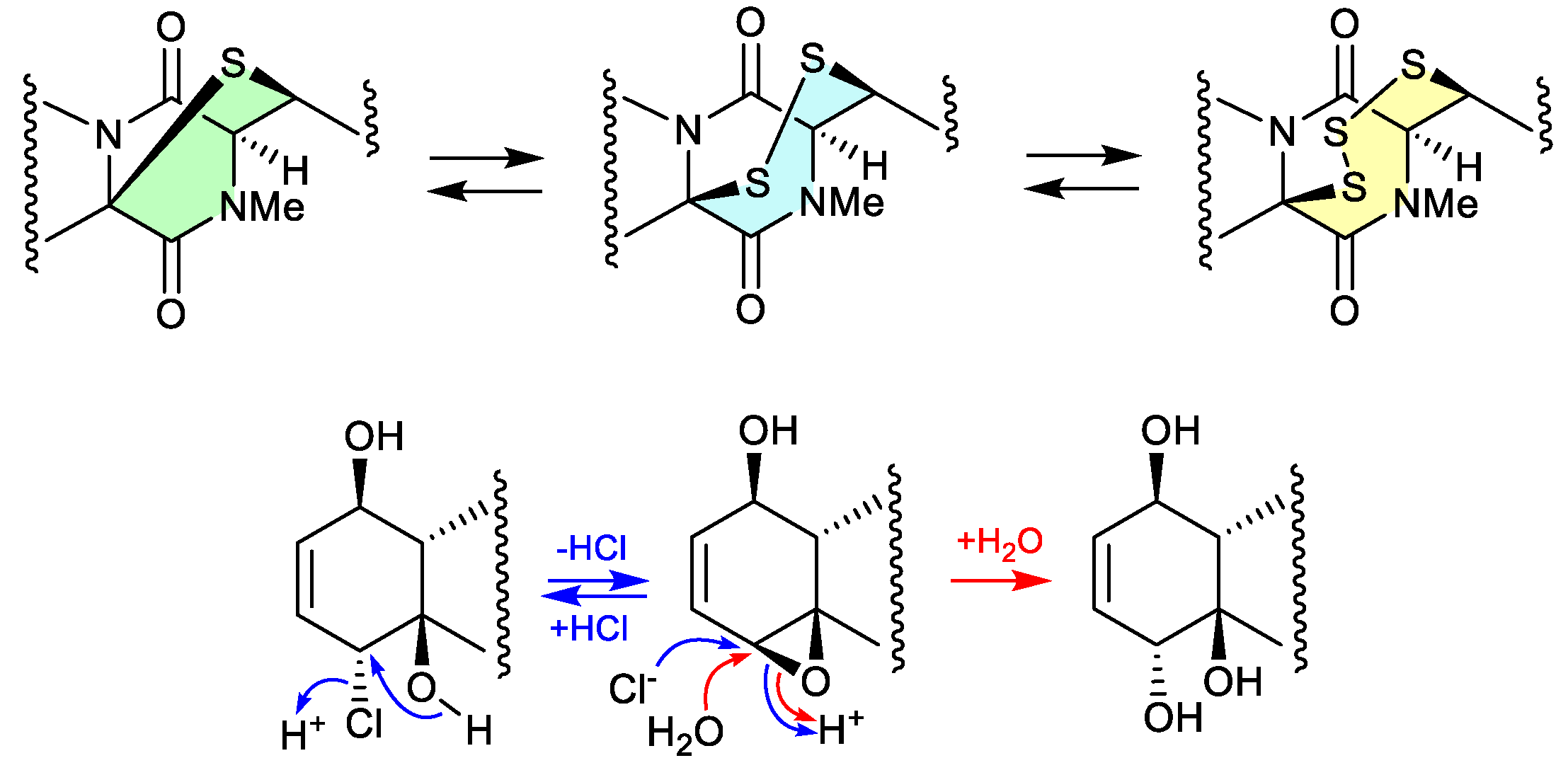
2.3. Methanol
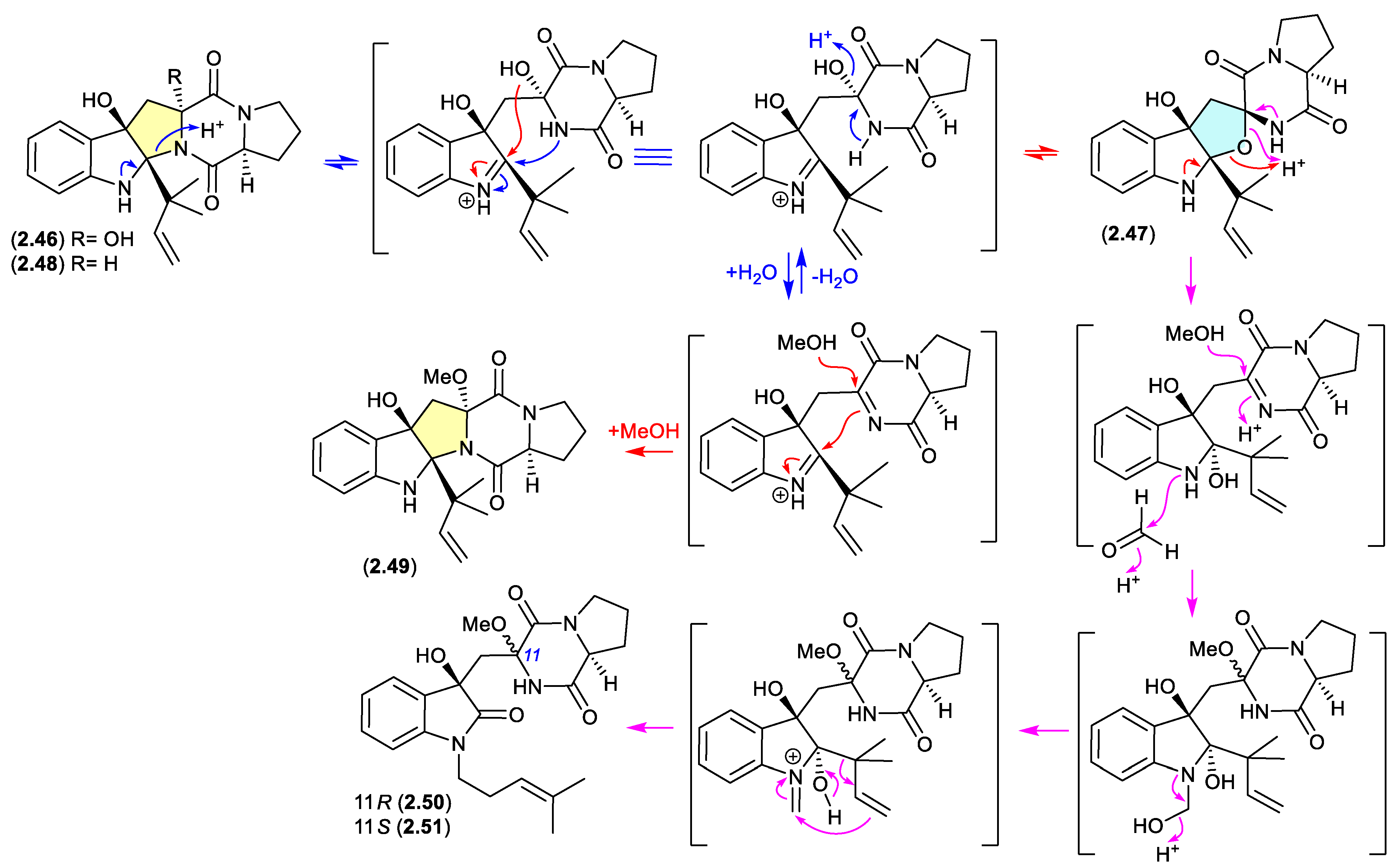
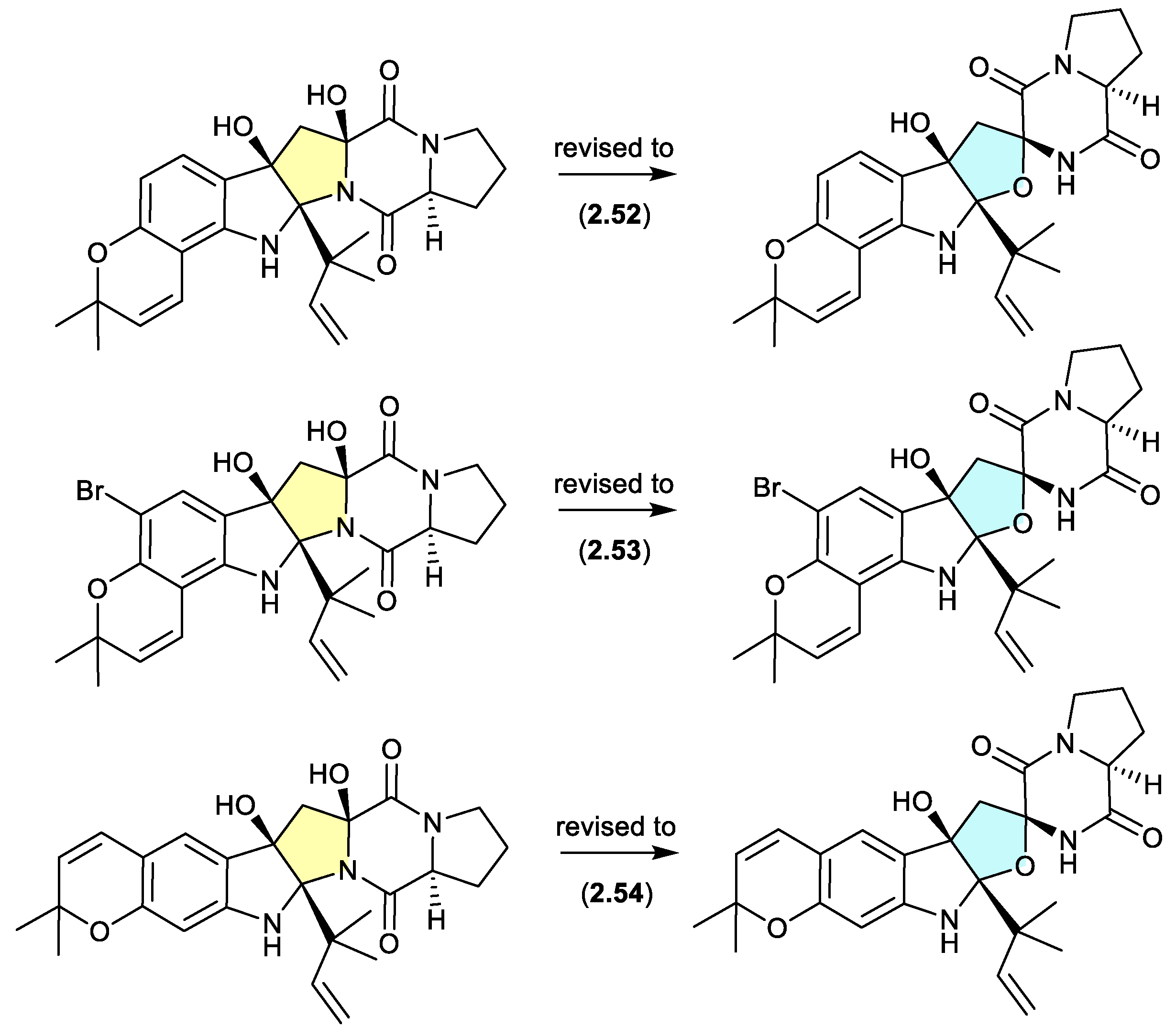
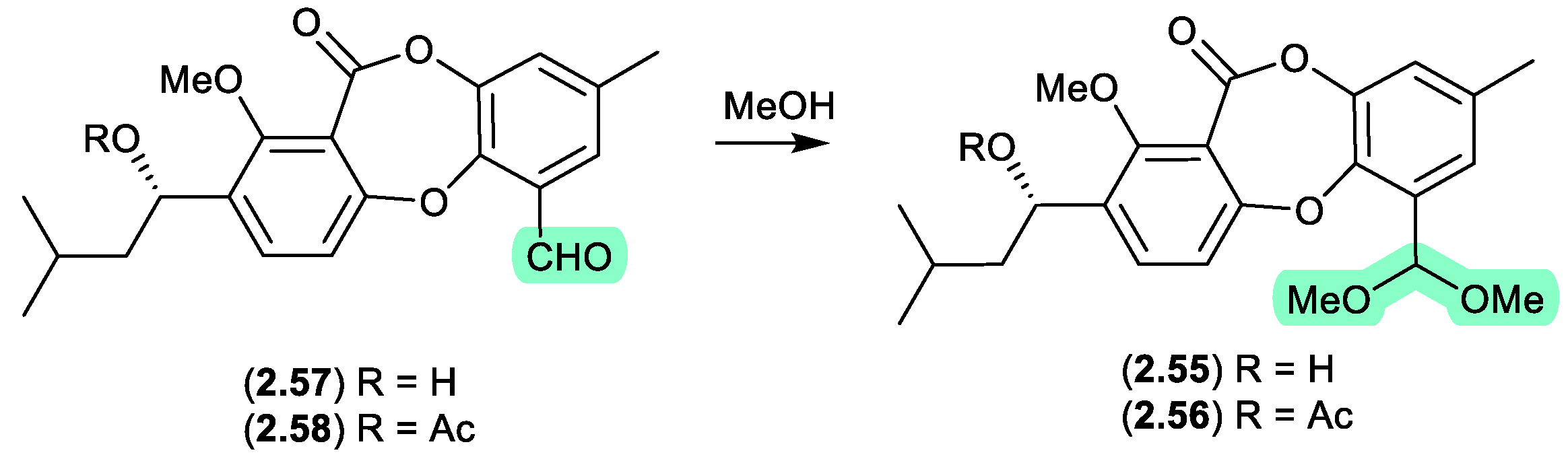
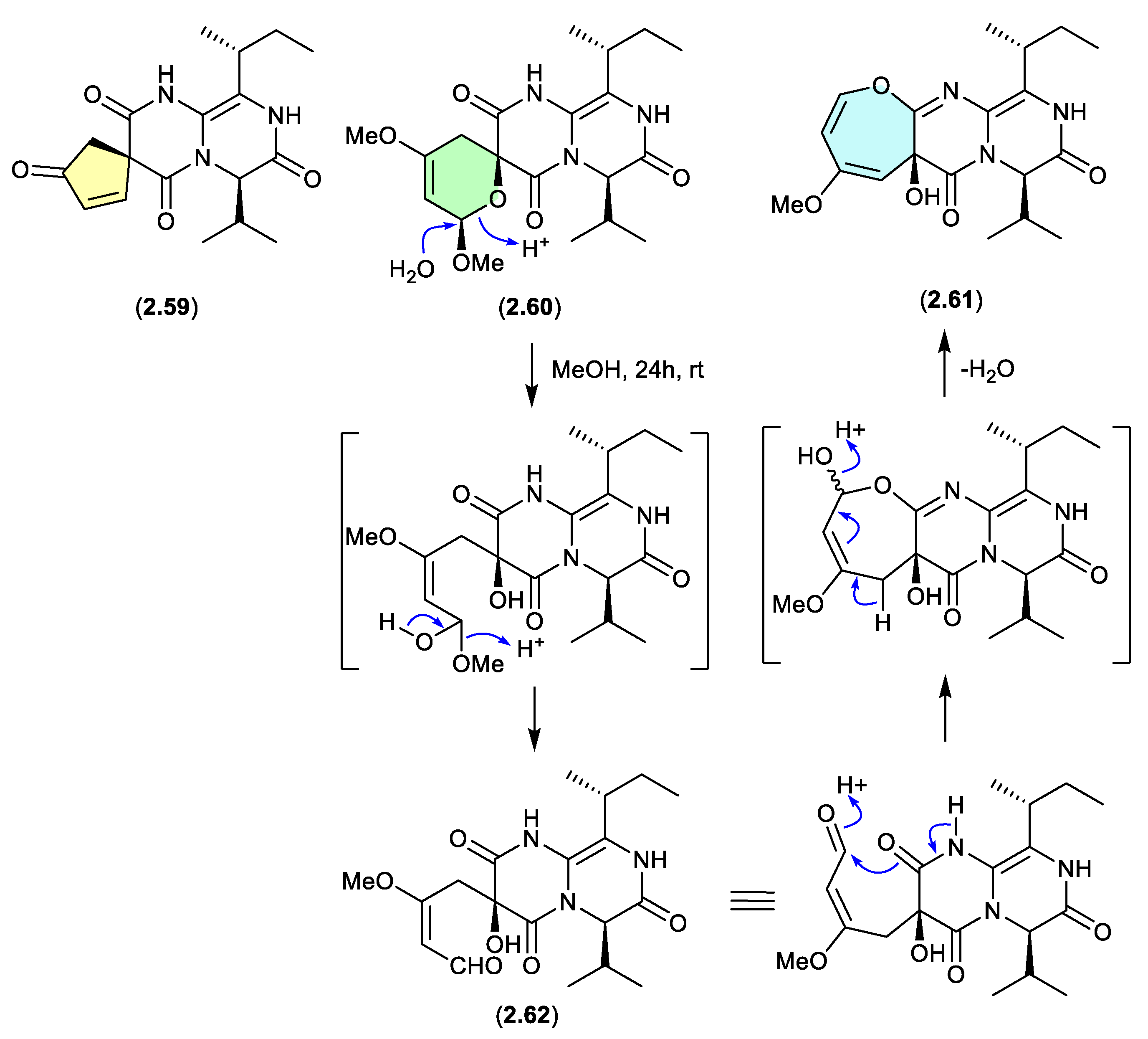
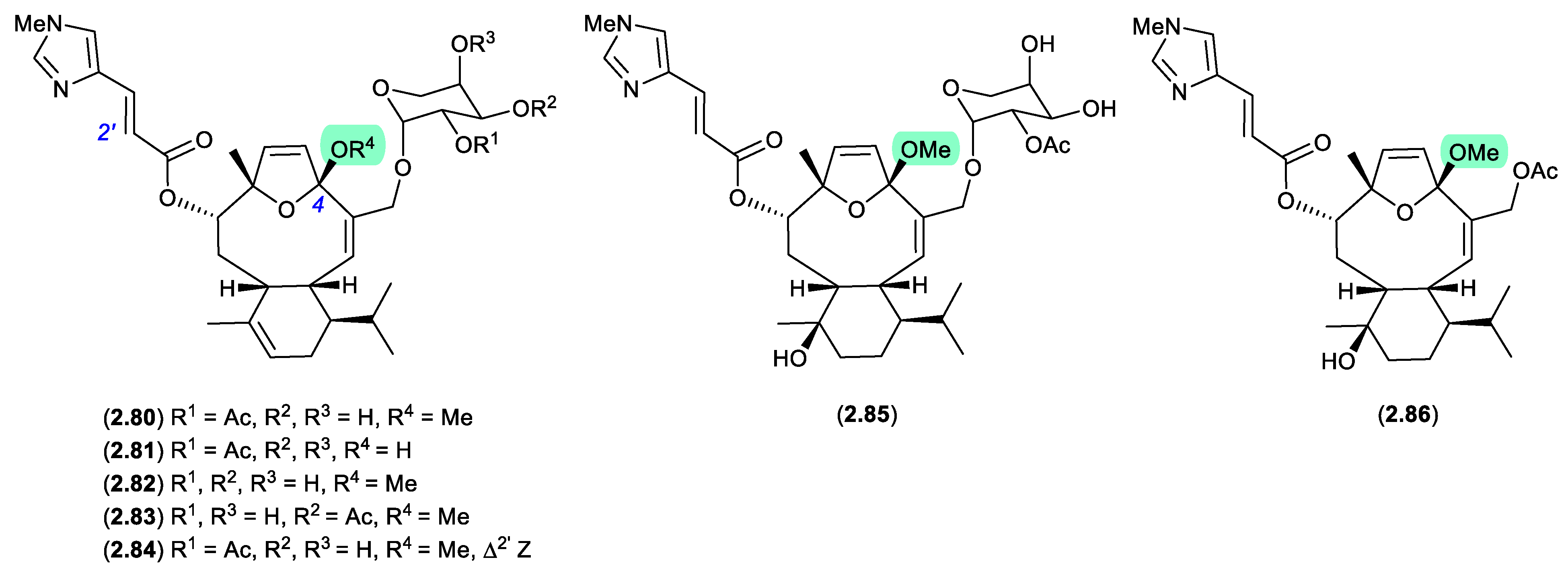
2.4. Acetone
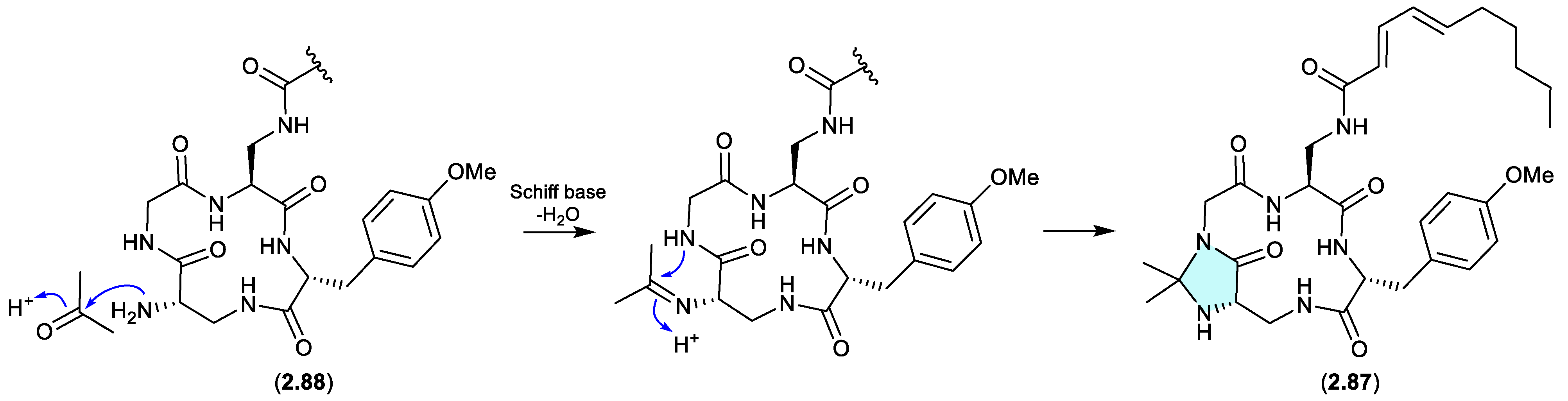
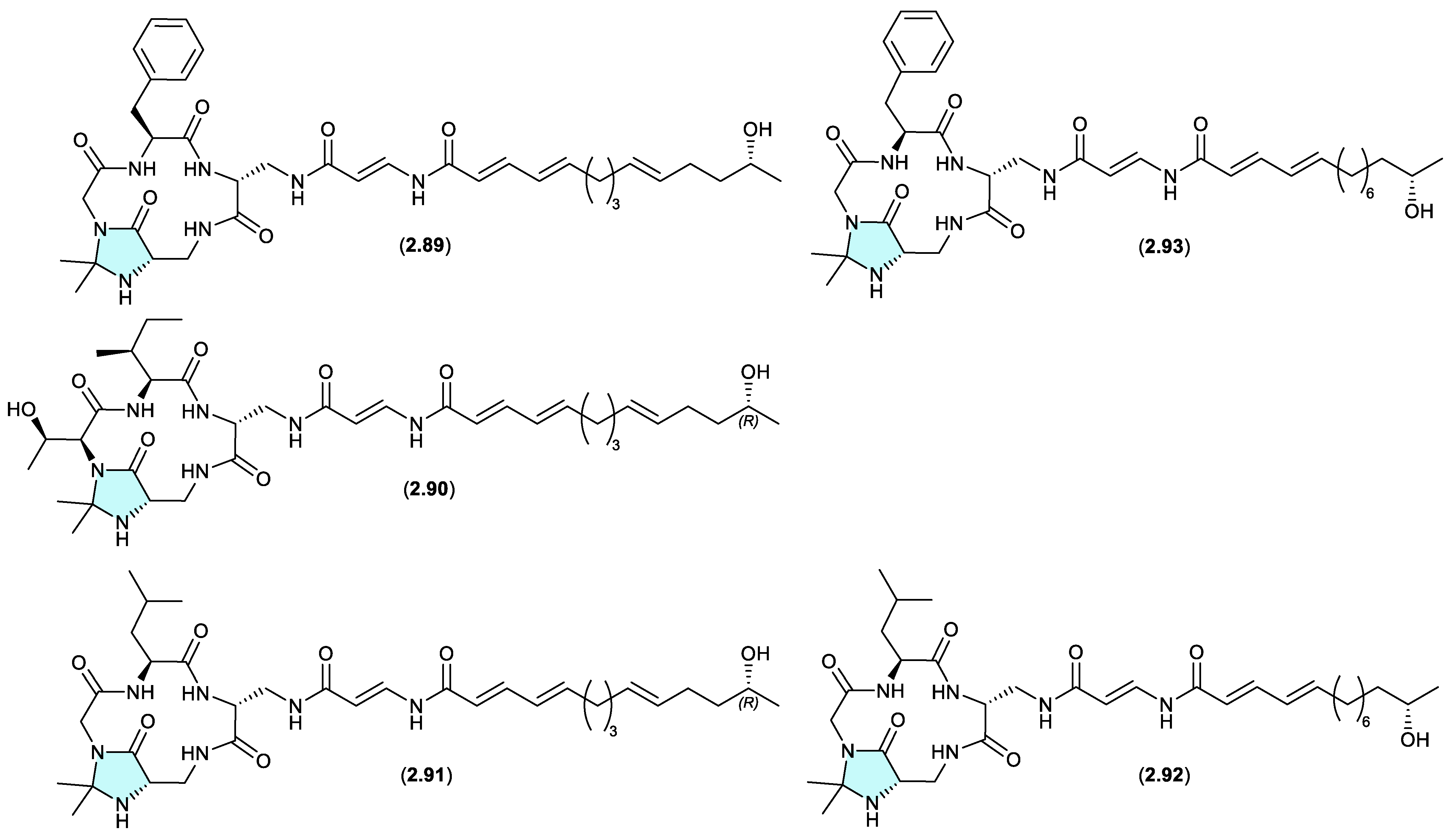
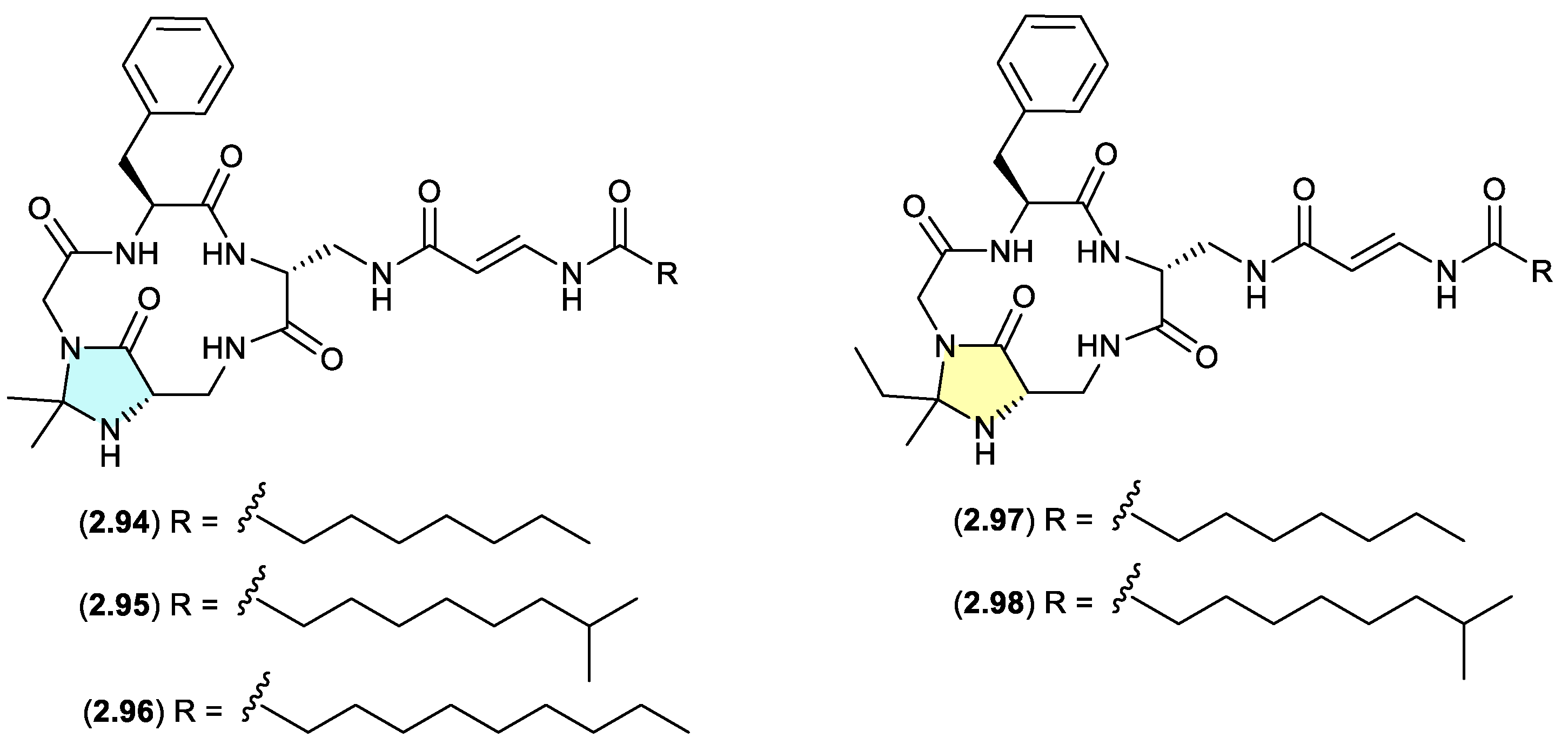
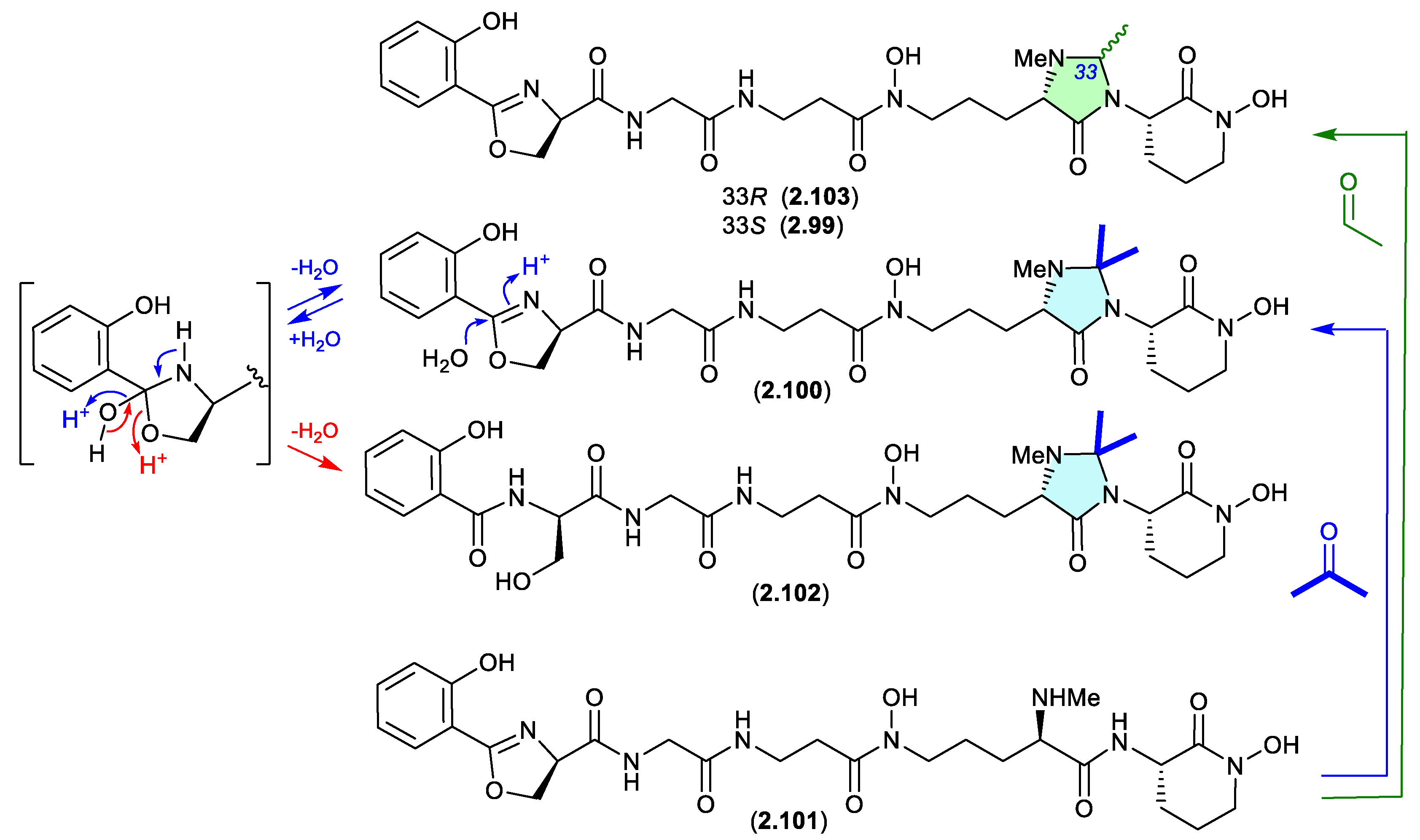
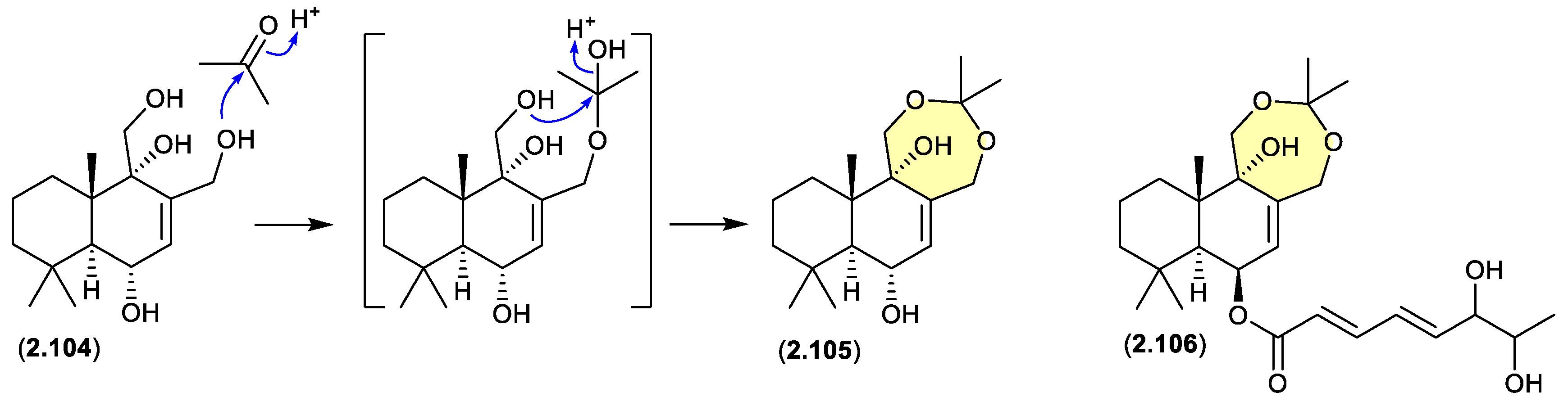
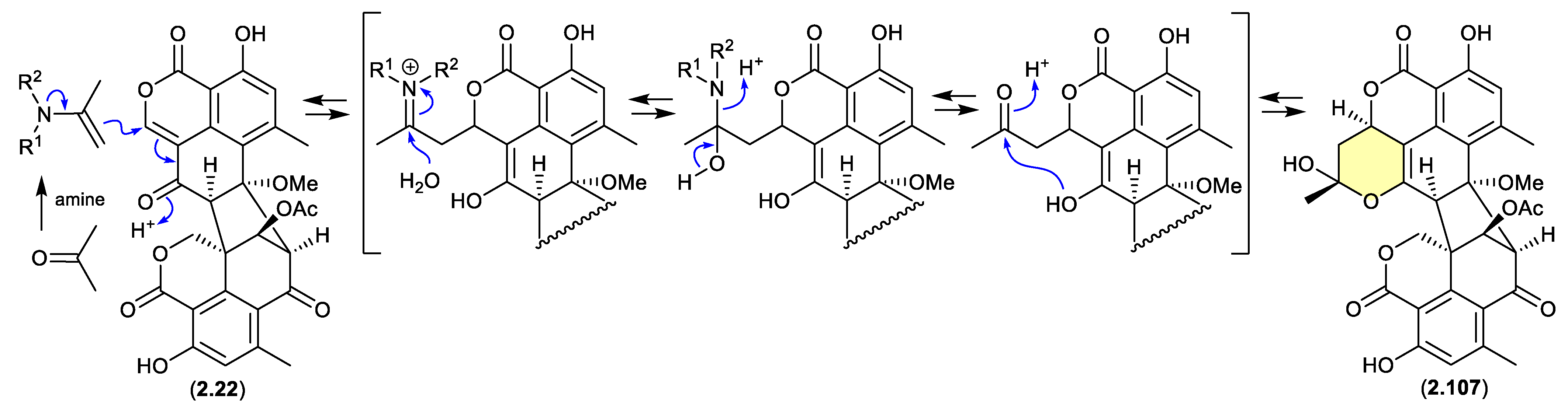
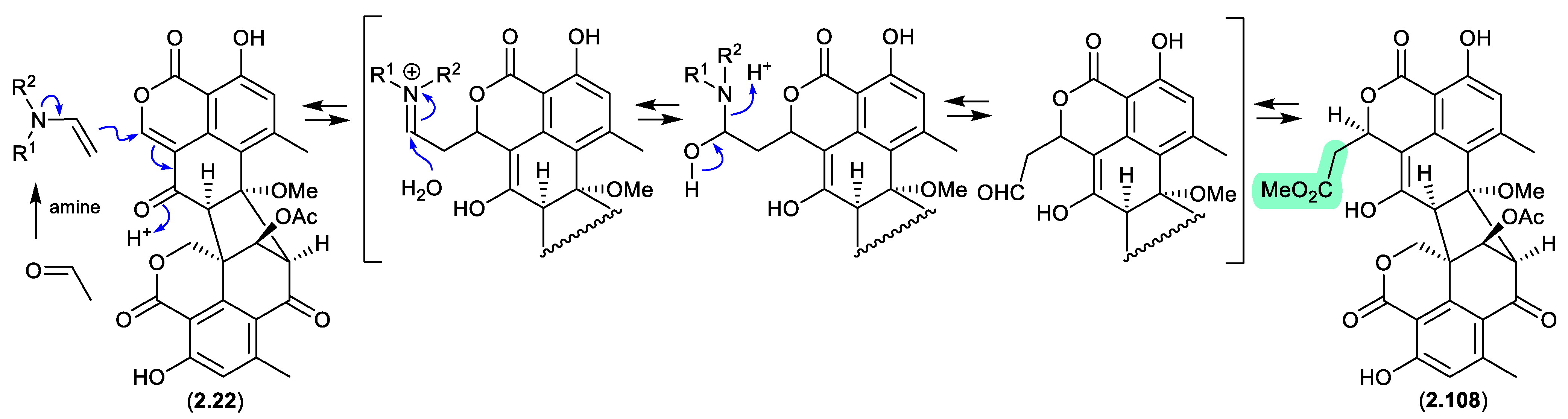
2.5. Acetonitrile
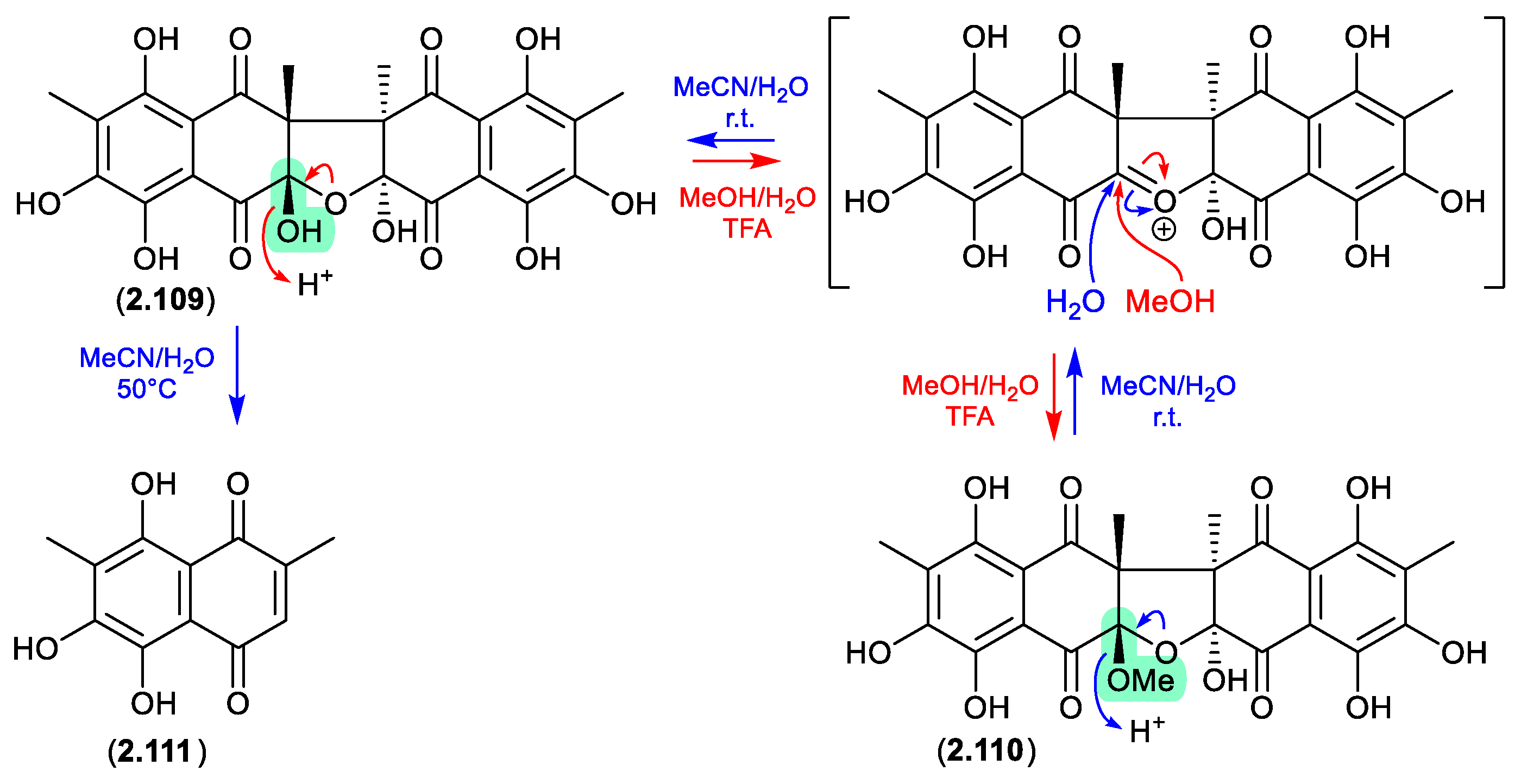
2.6. Chloroform
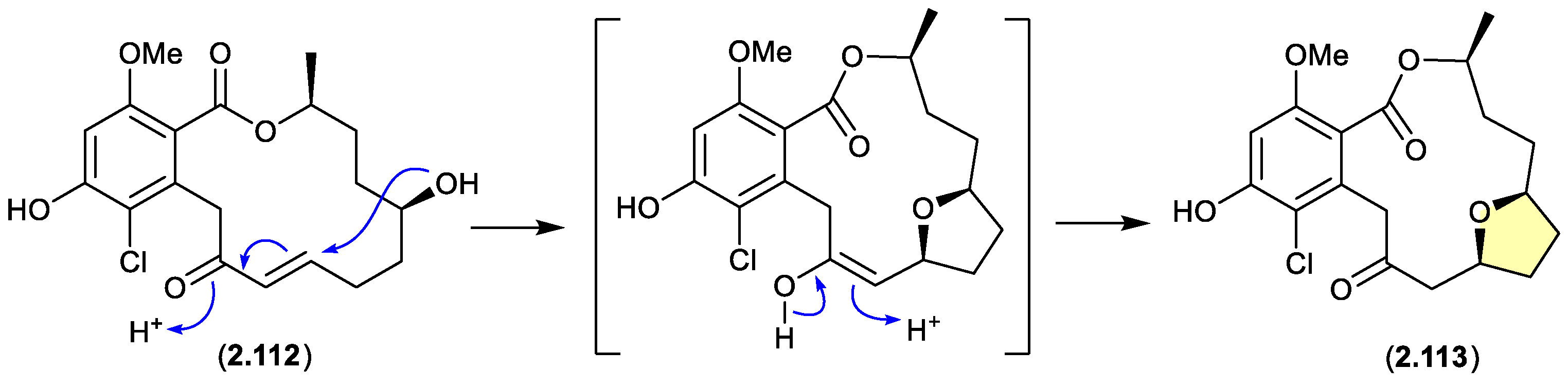
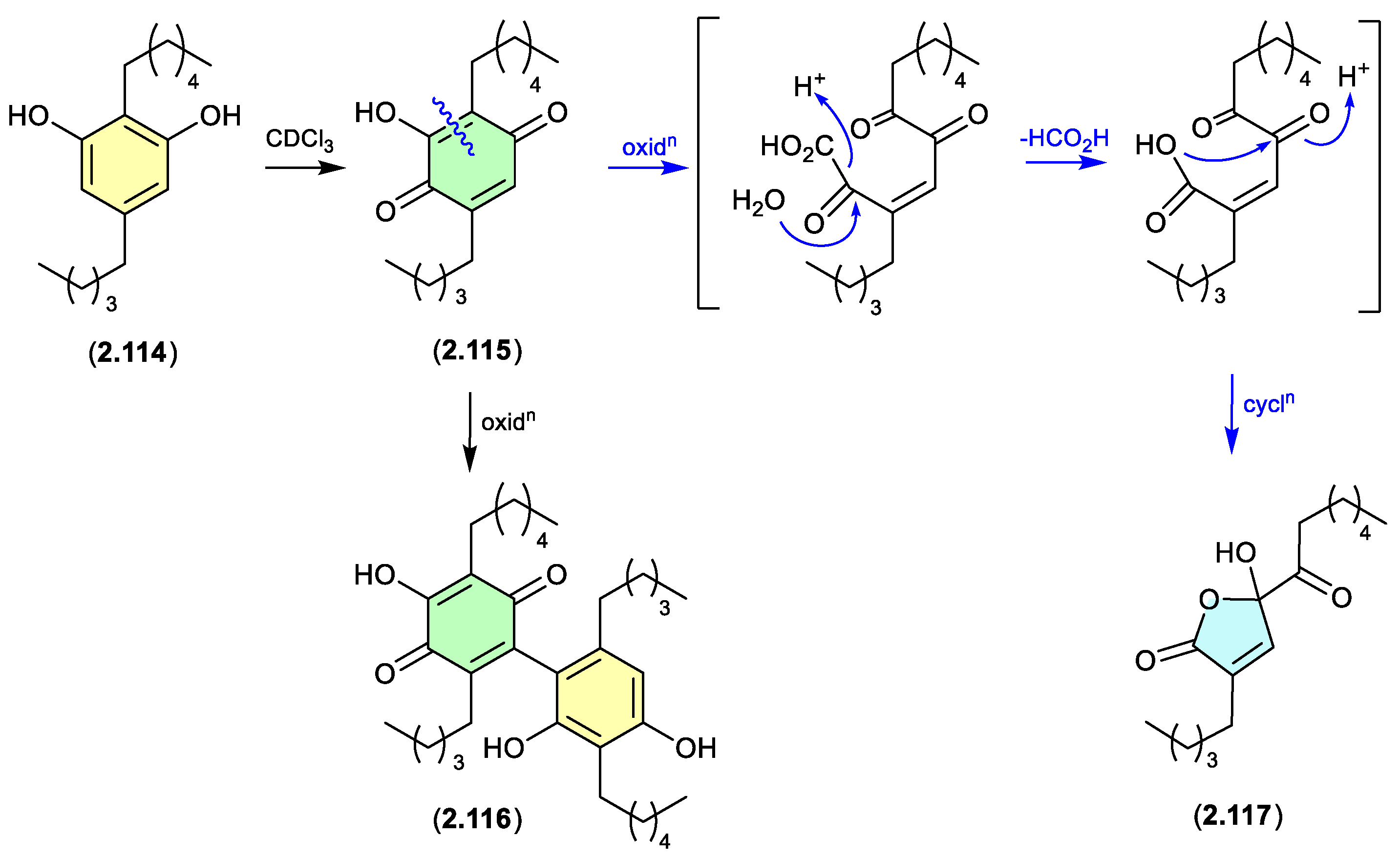
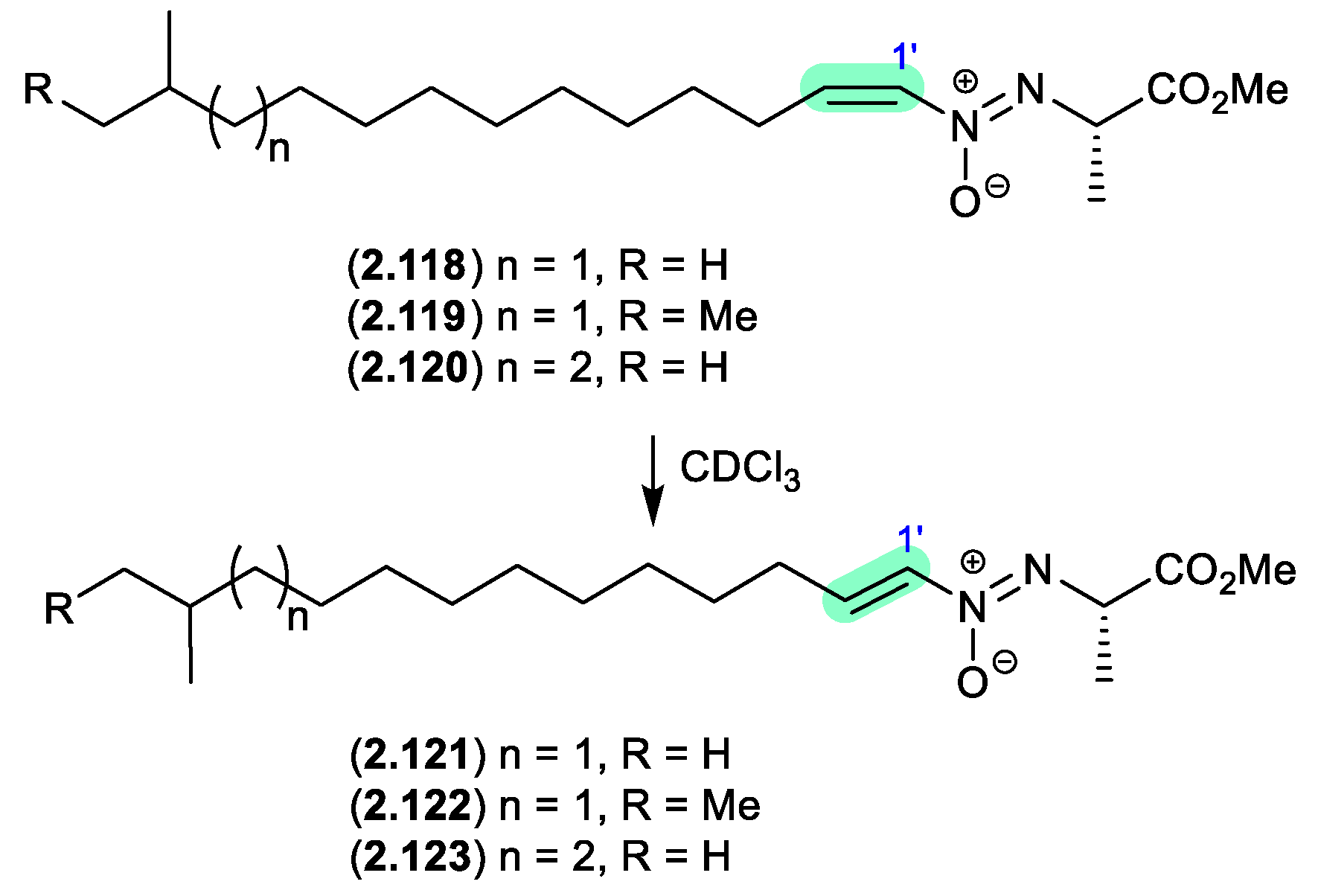
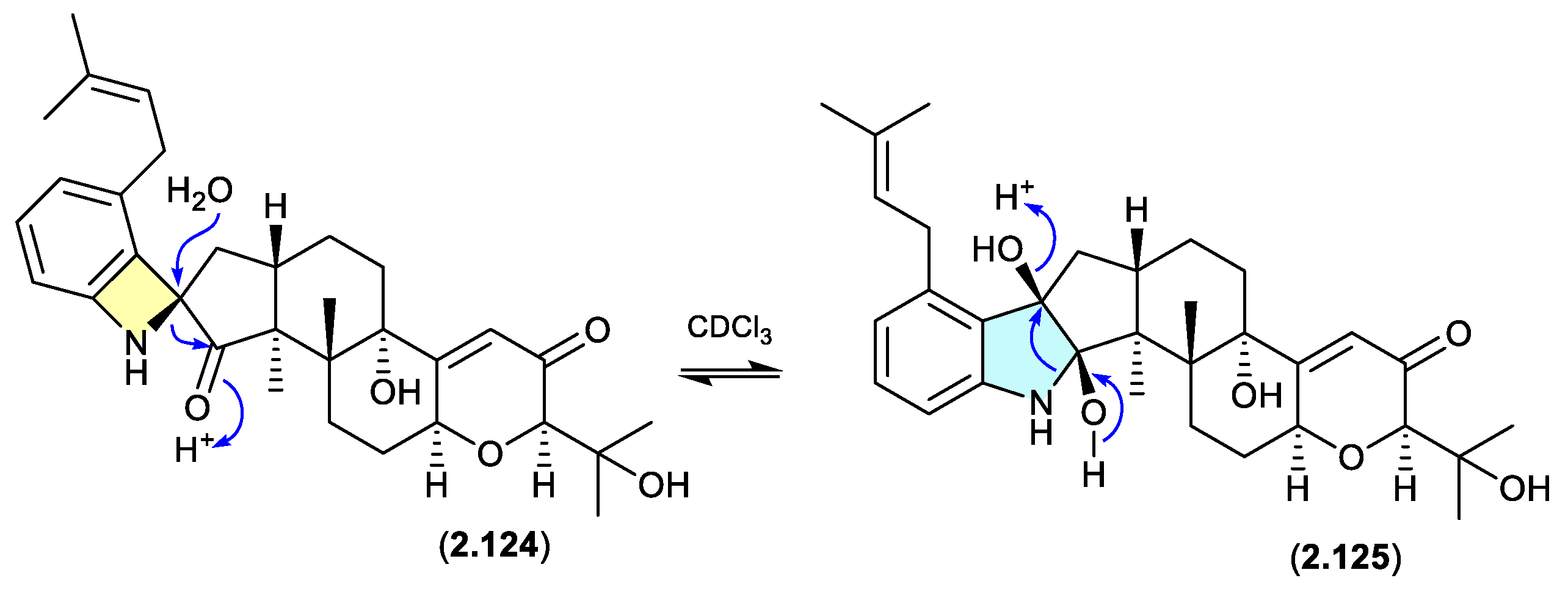
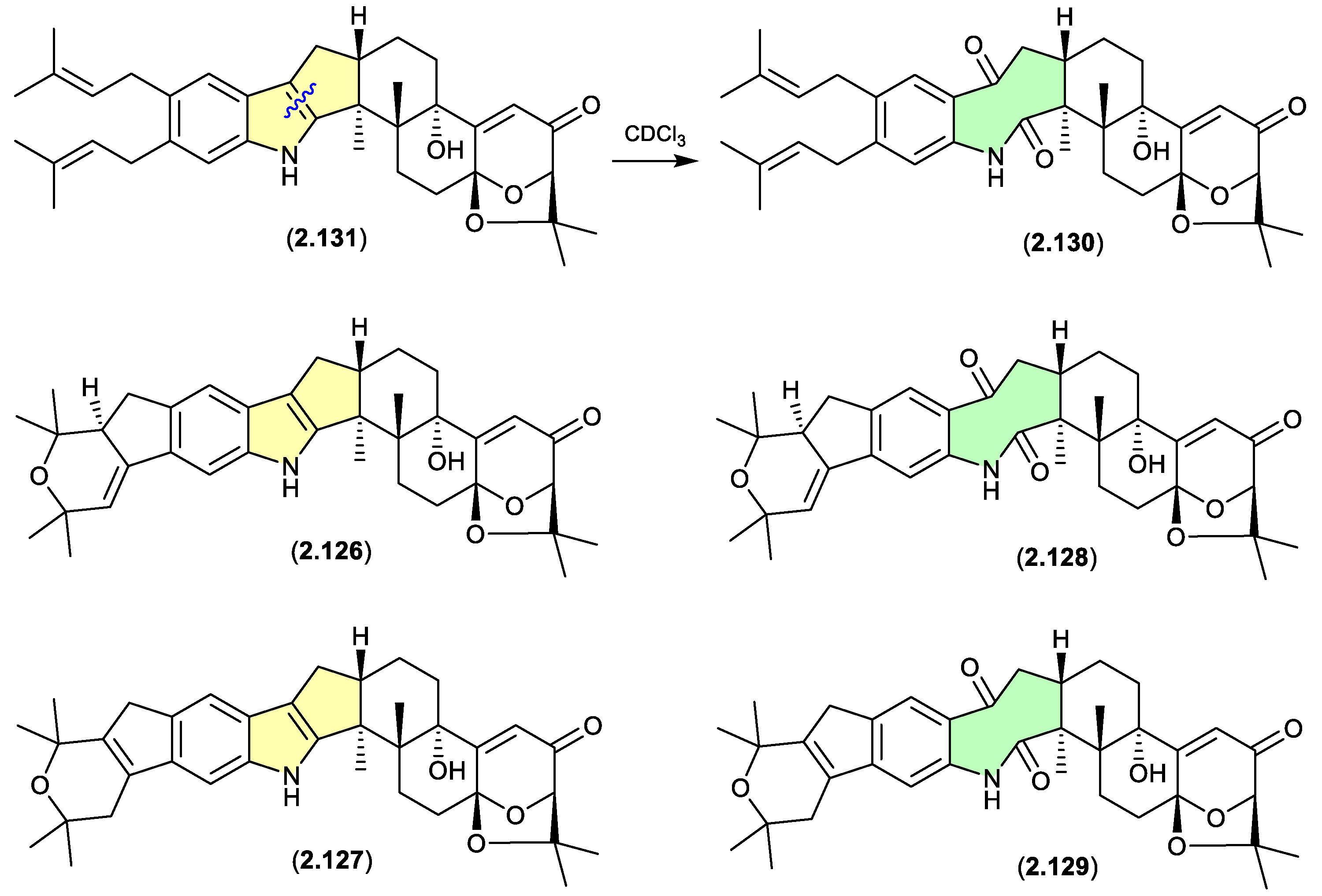
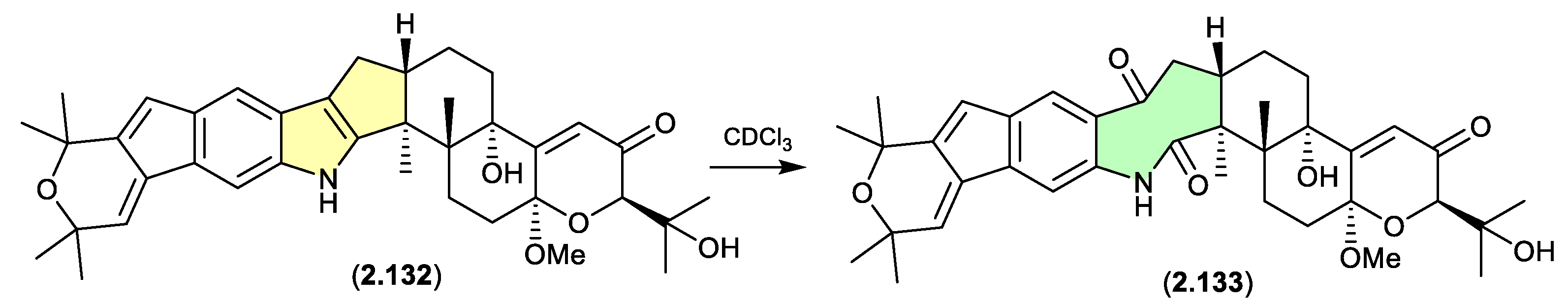
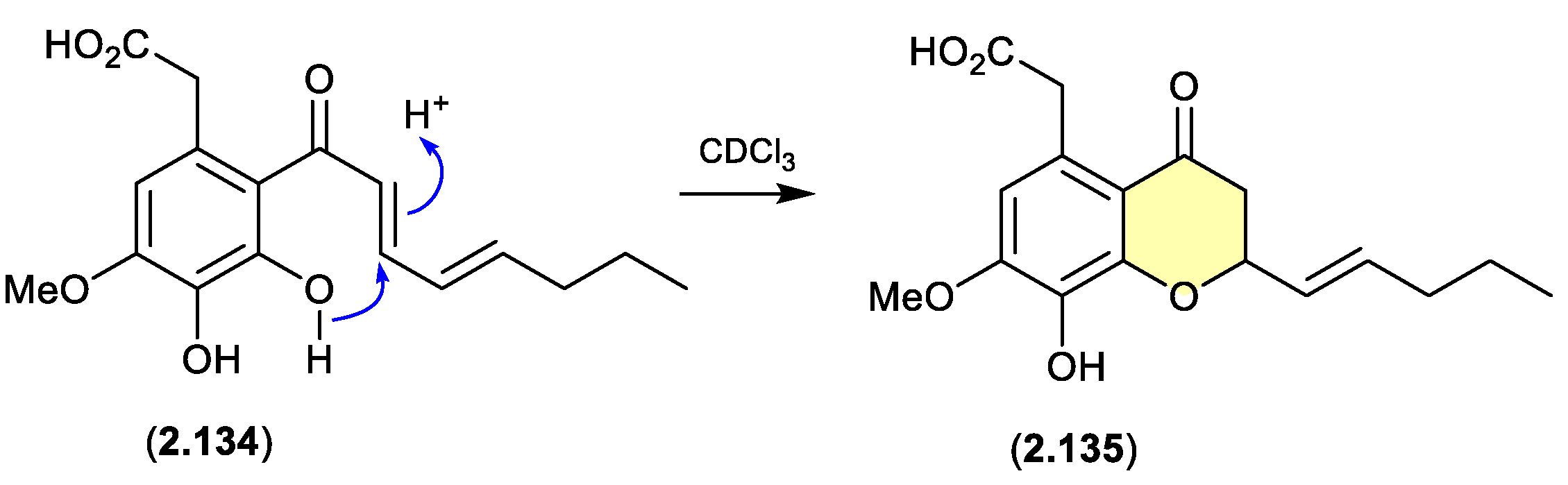
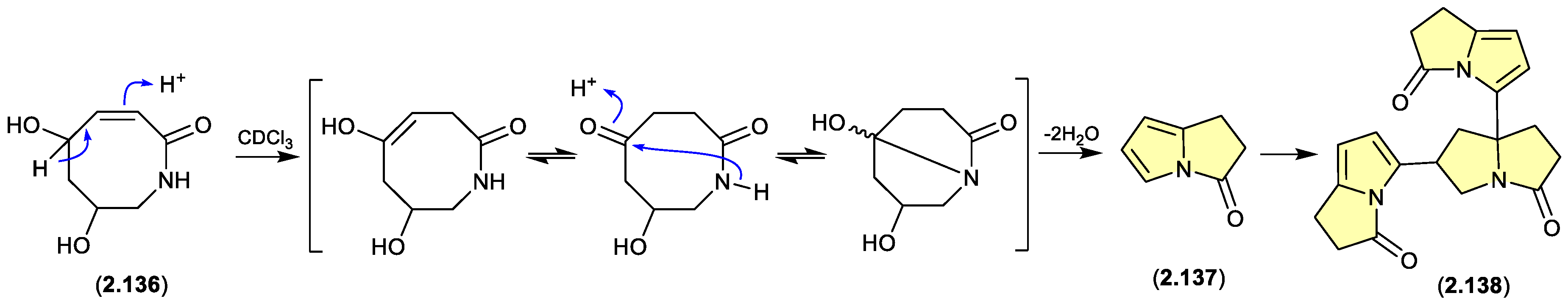
2.7. Dichloromethane
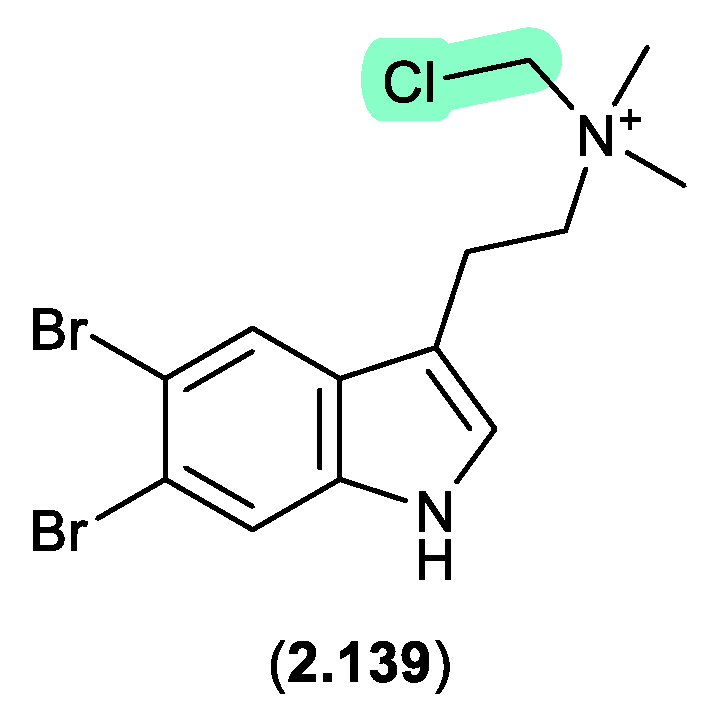
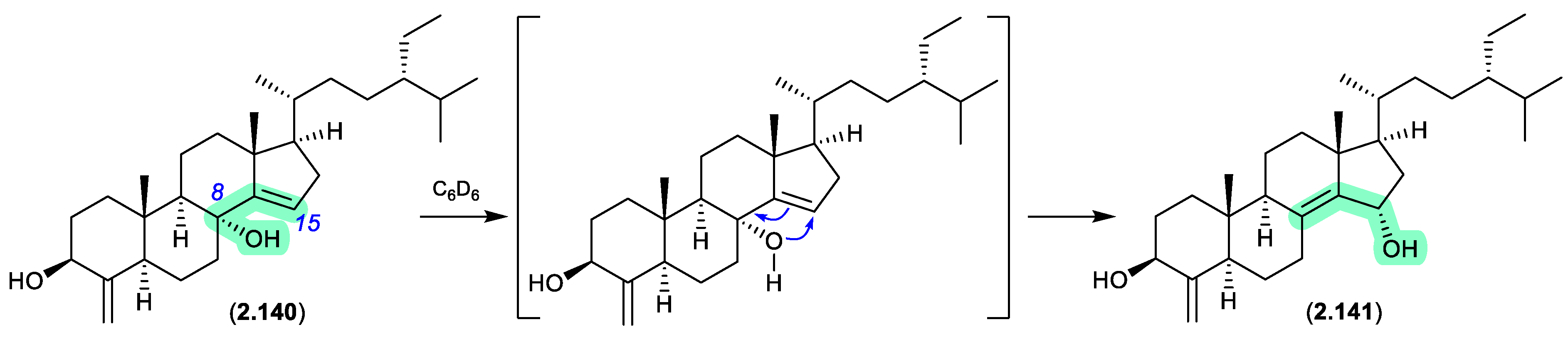
2.8. Benzene
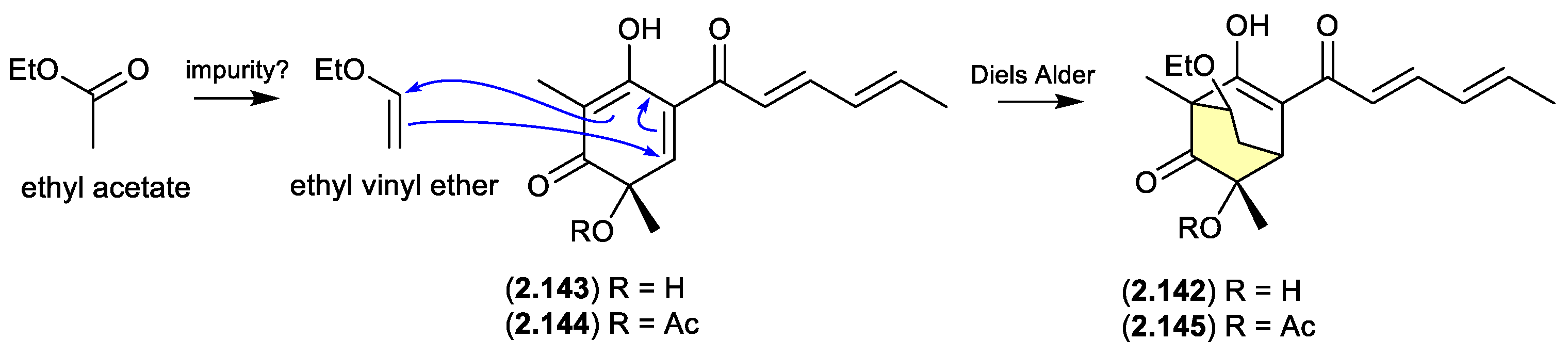
2.9. Ethyl Acetate
2.10. Aprotic vs Protic
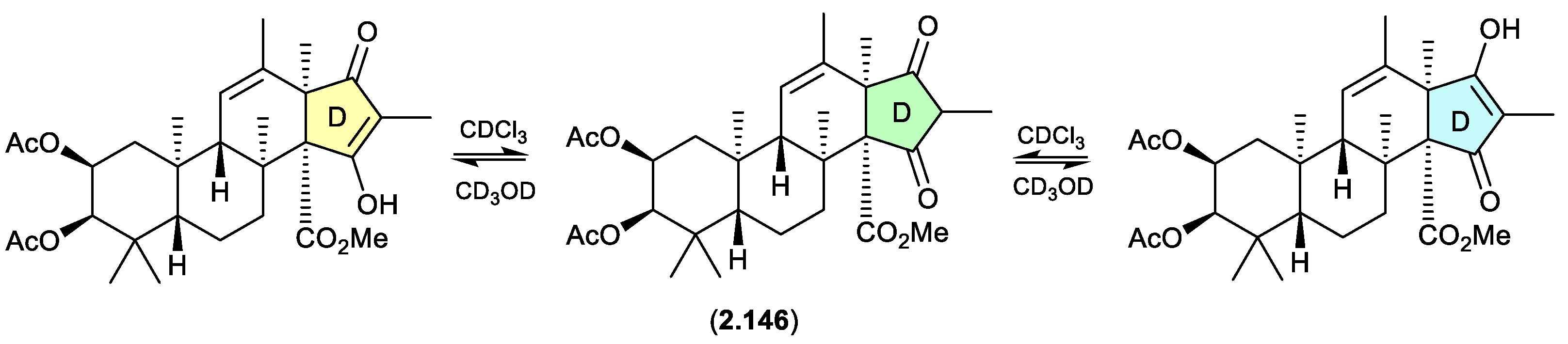
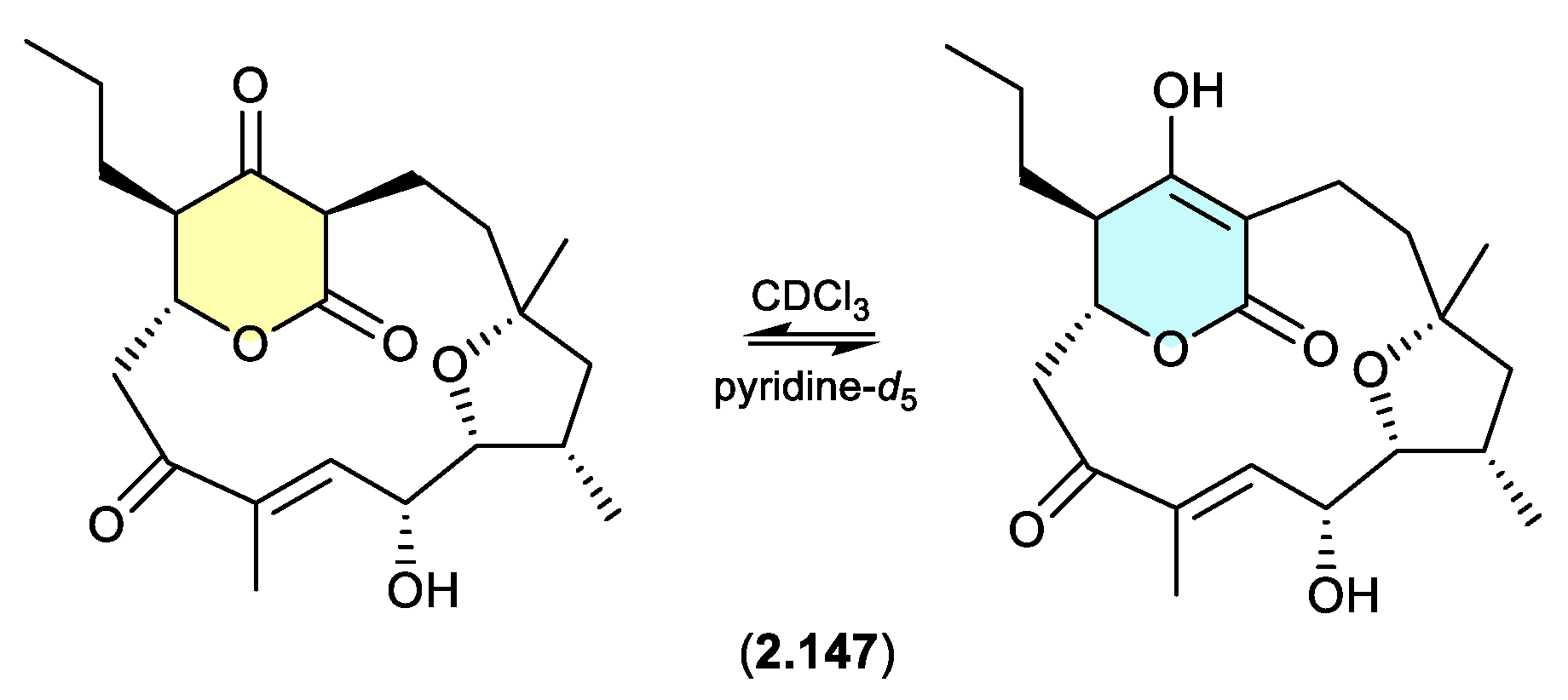
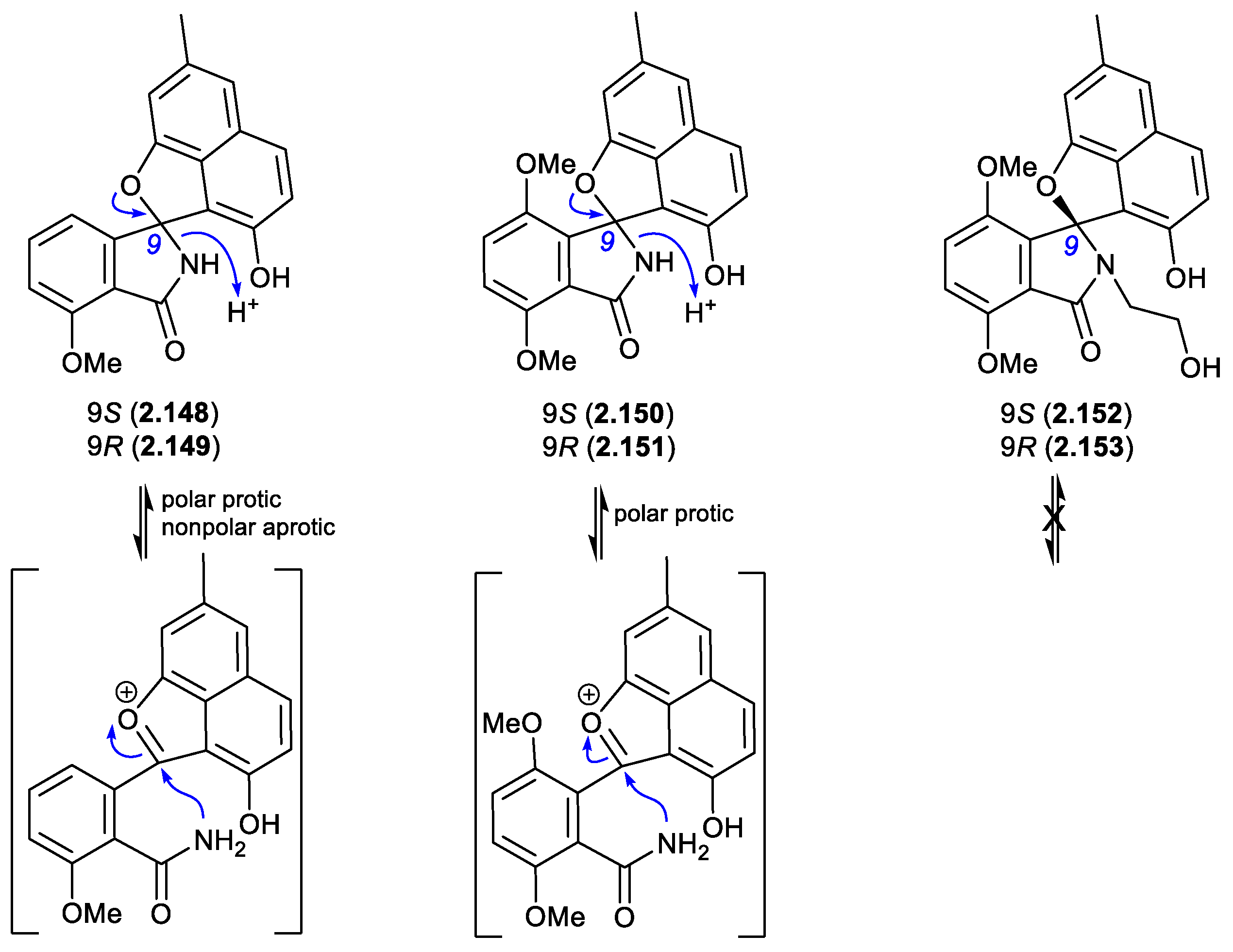
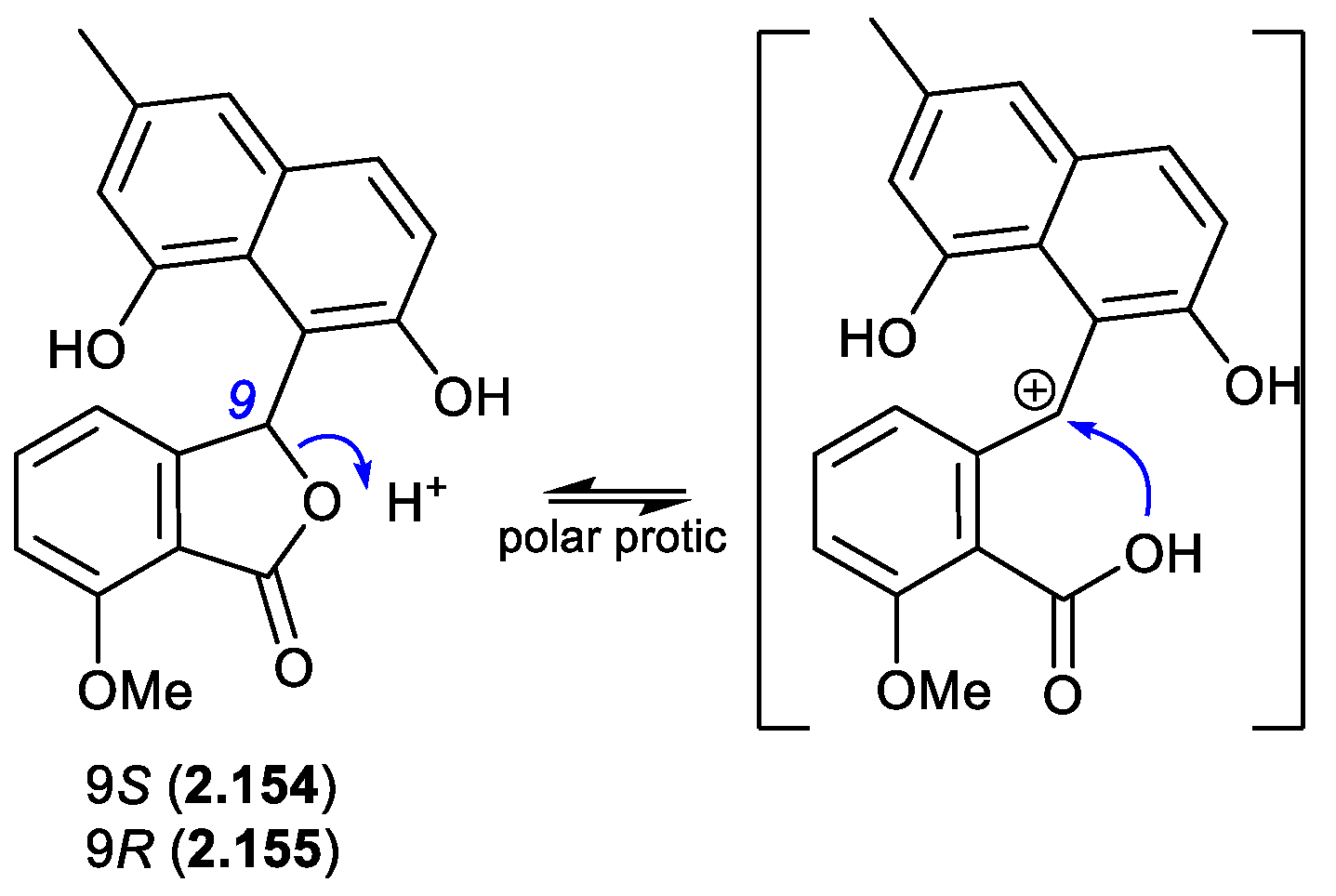
3. Heat
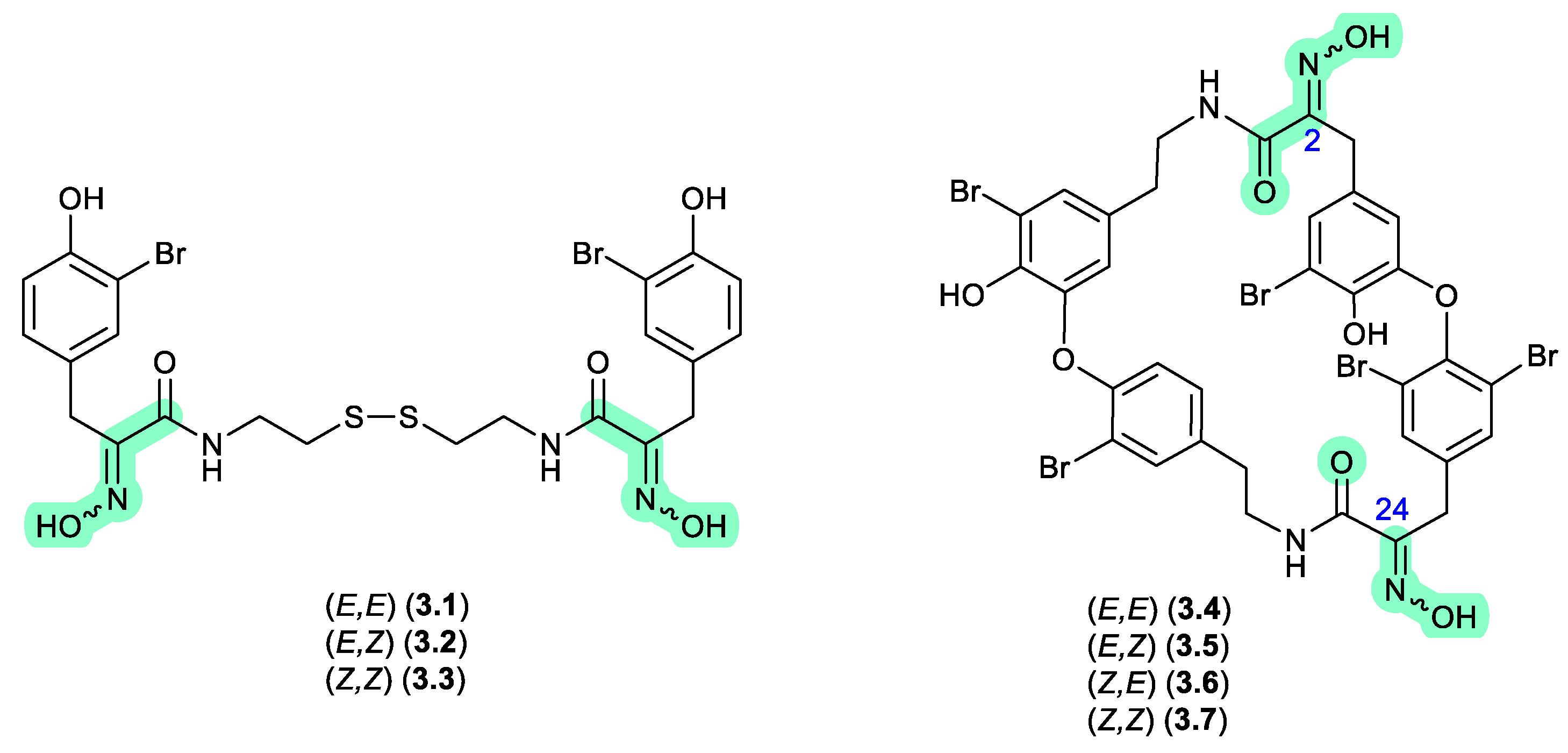
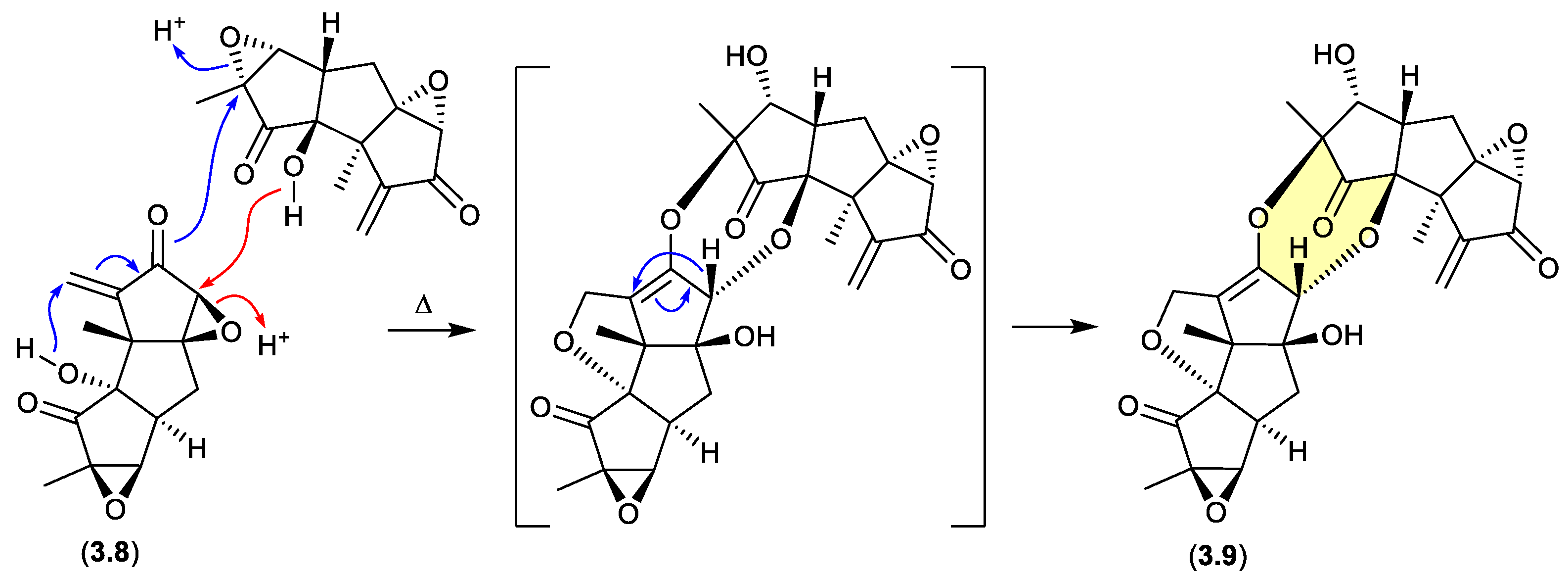
4. pH
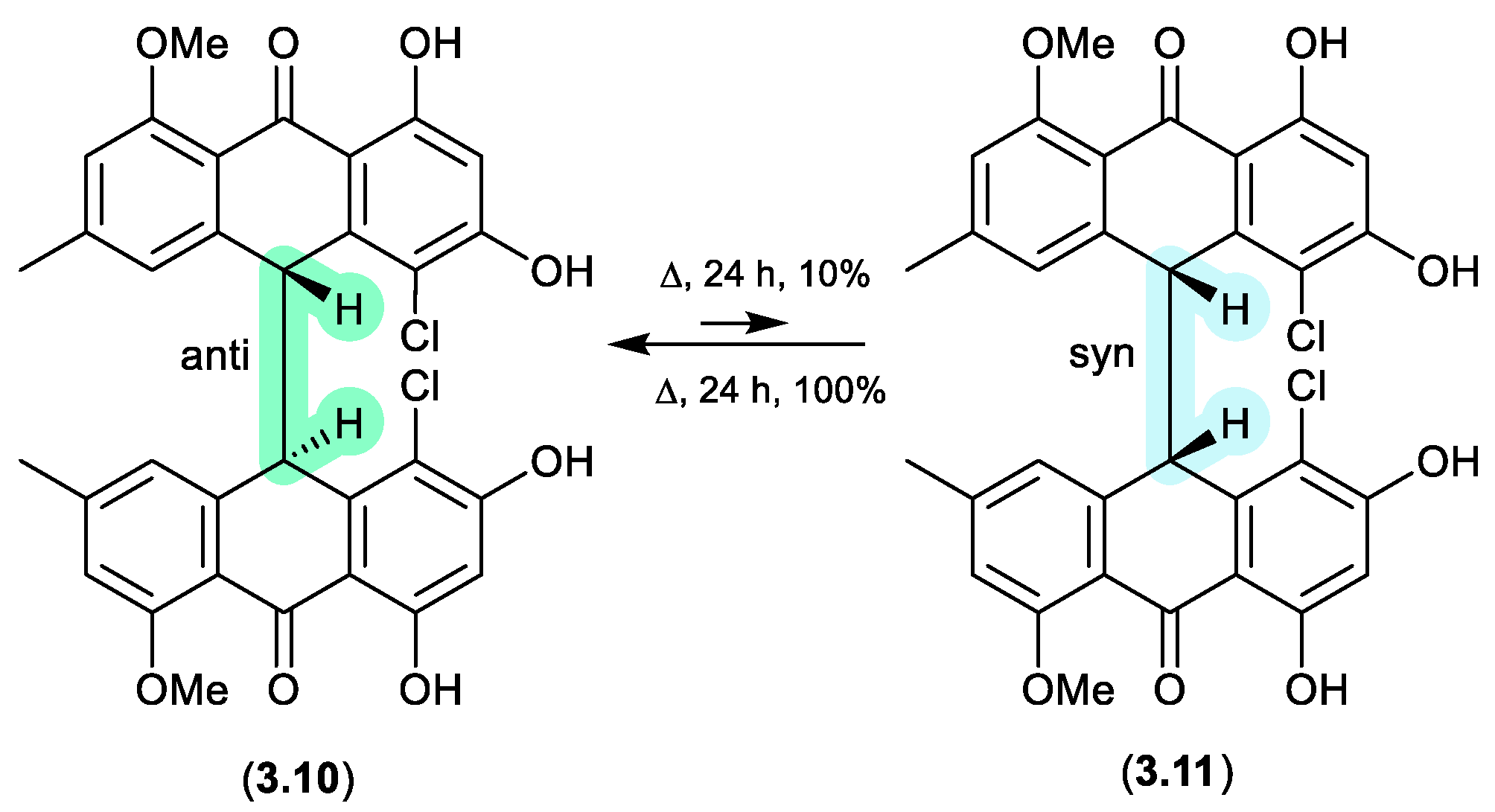
4.1. Basic
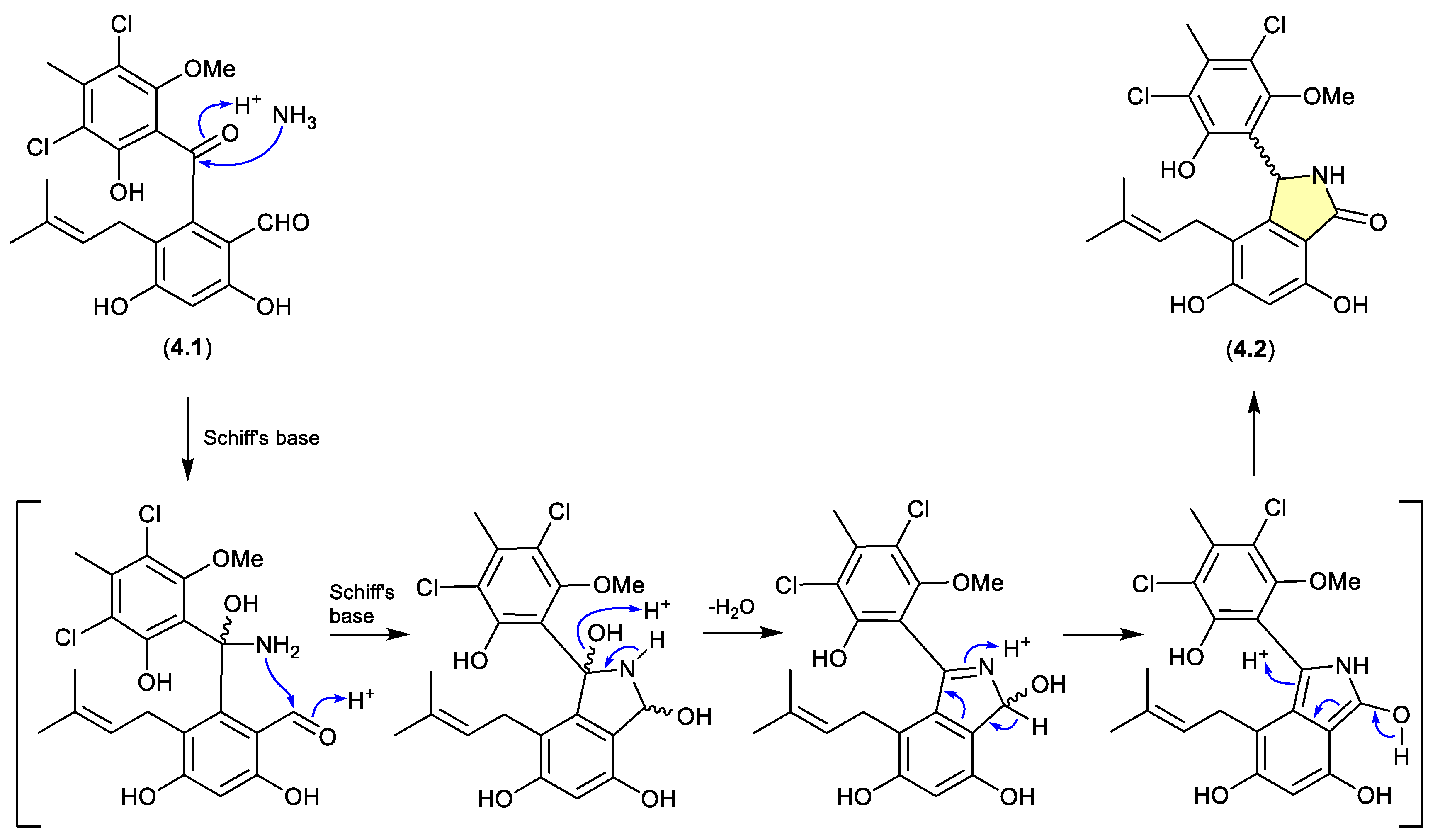
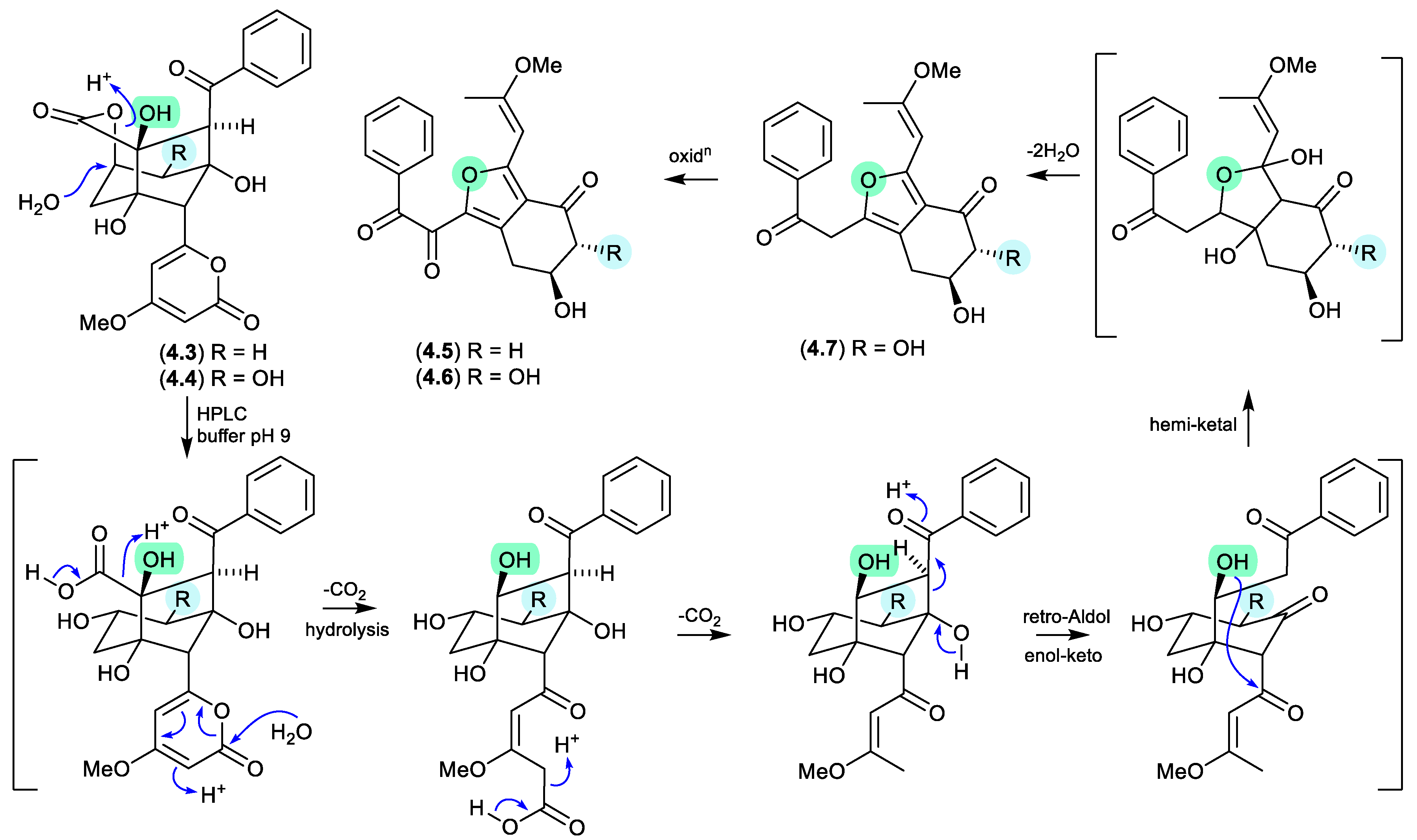
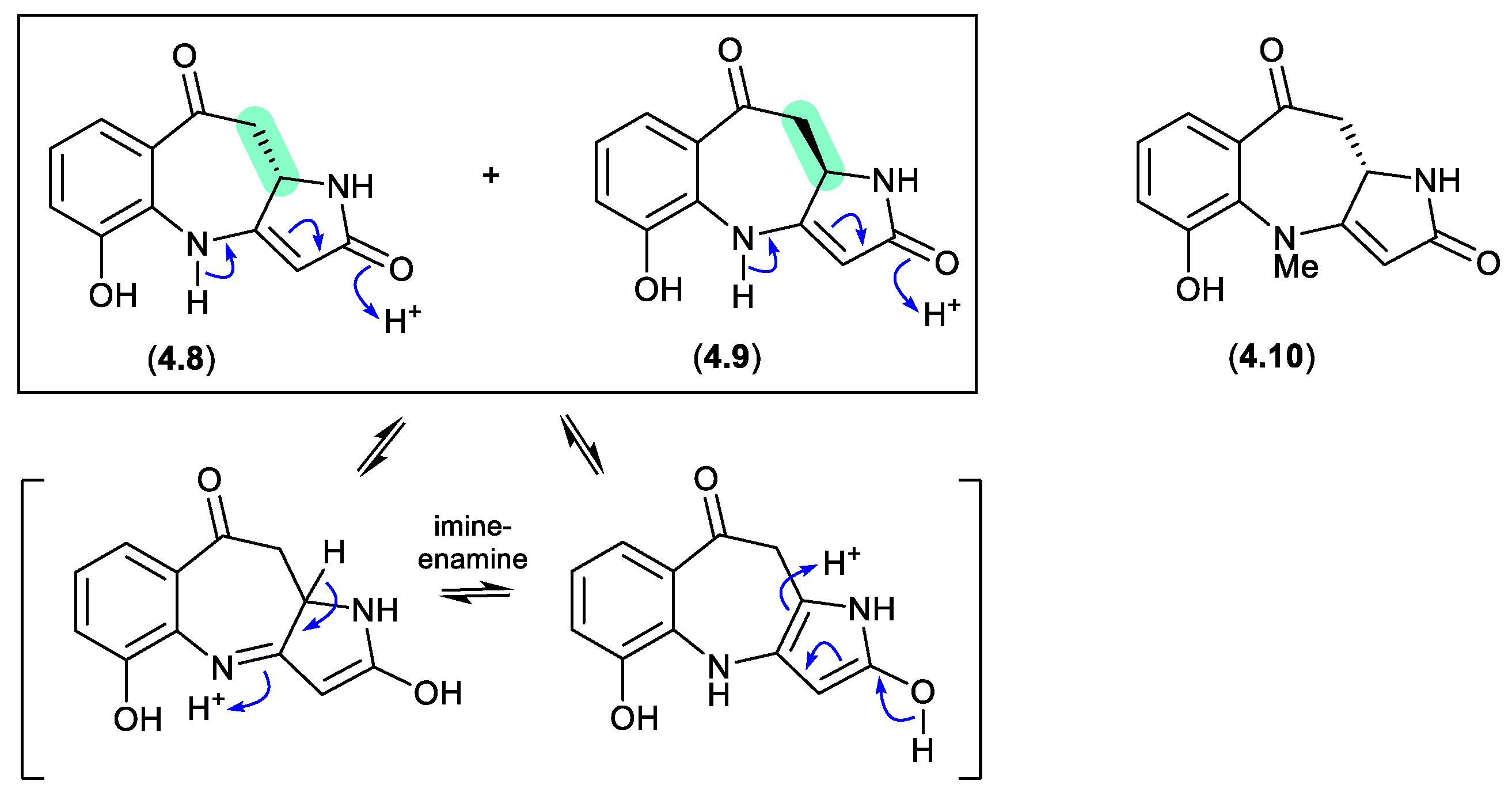
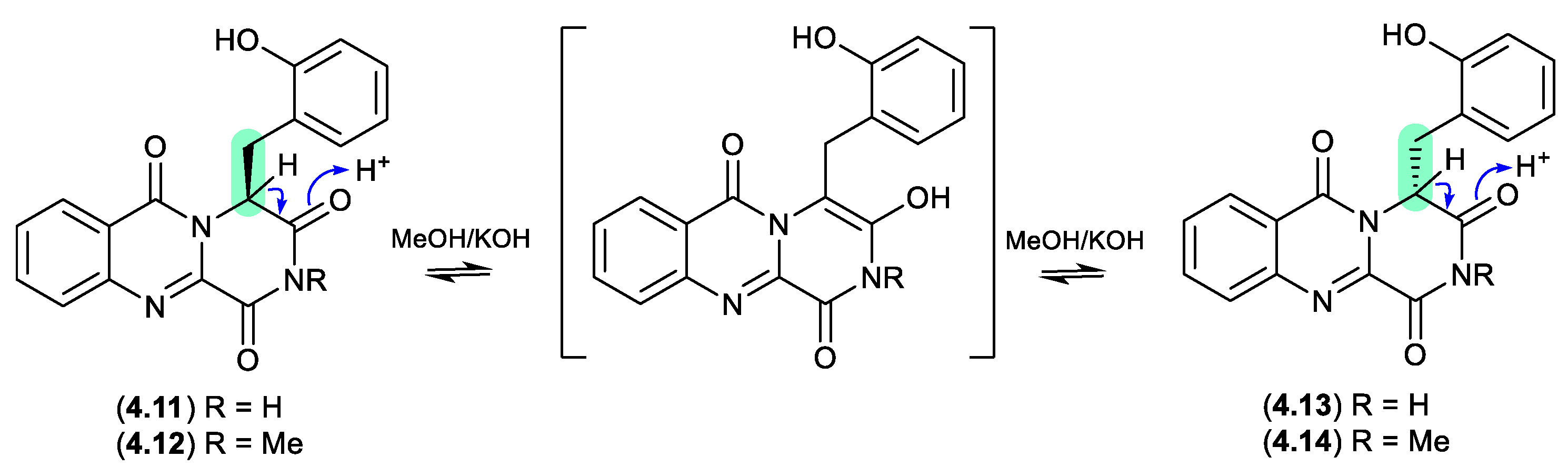
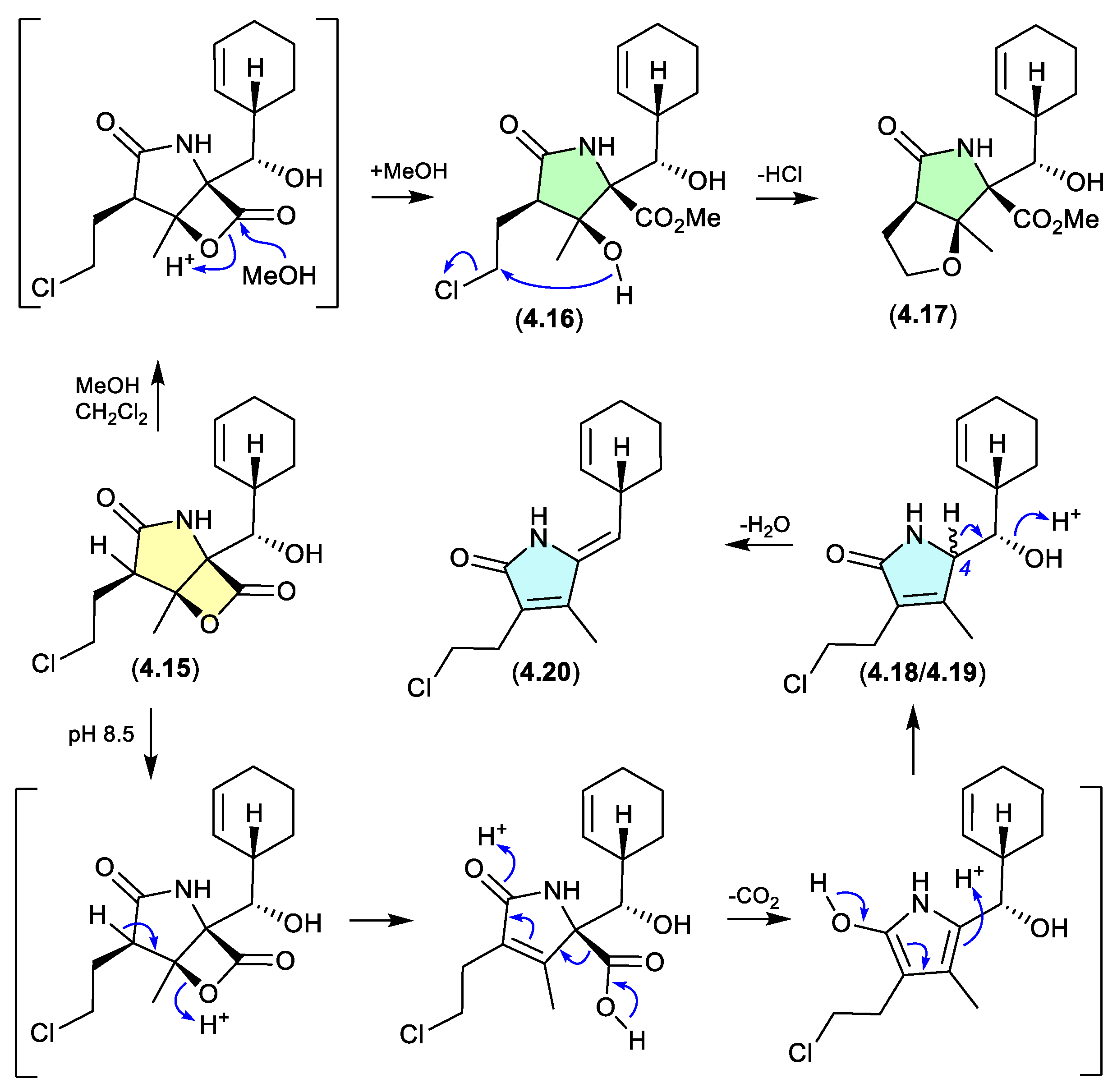
4.2. Acidic
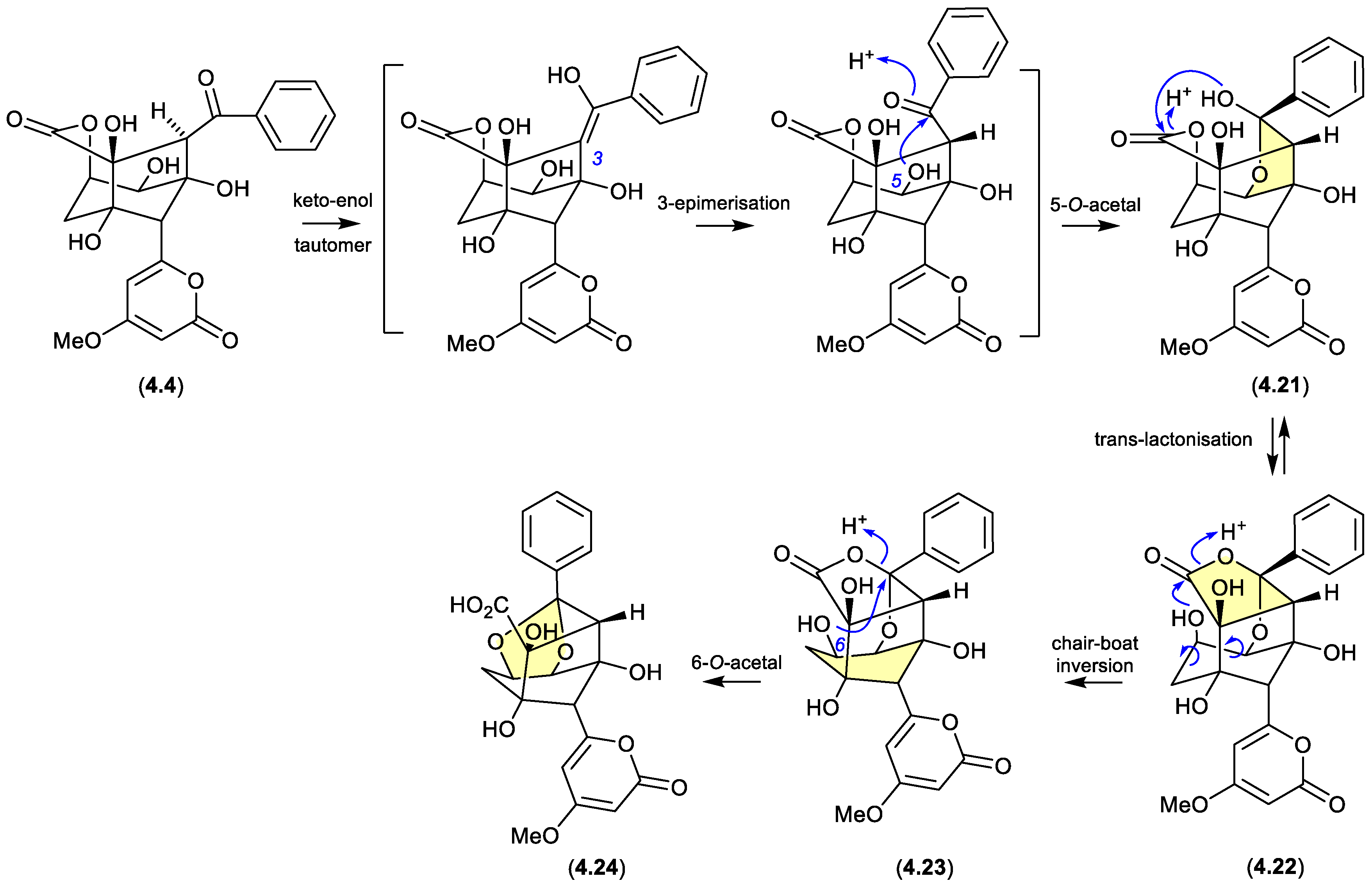
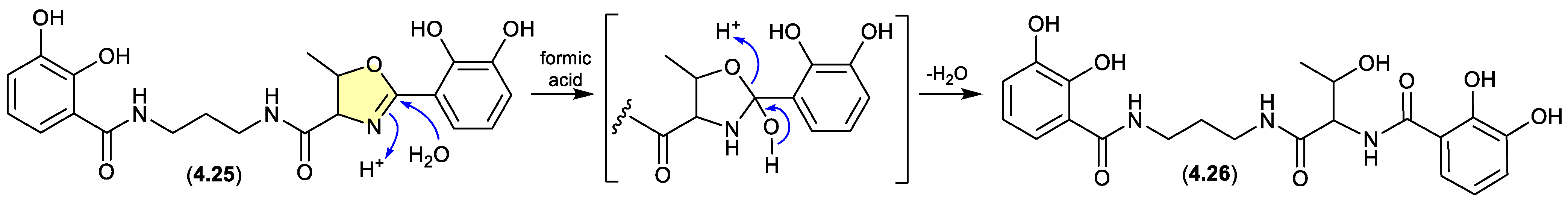
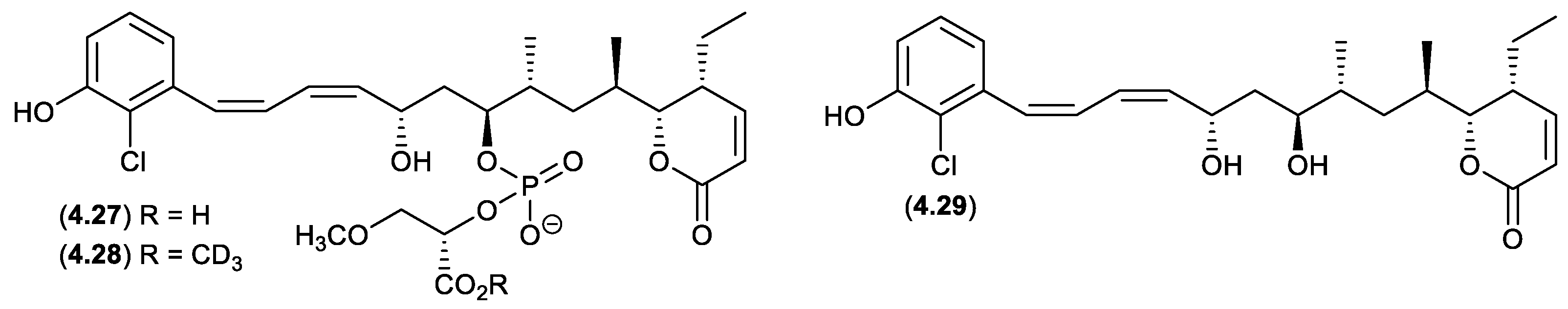
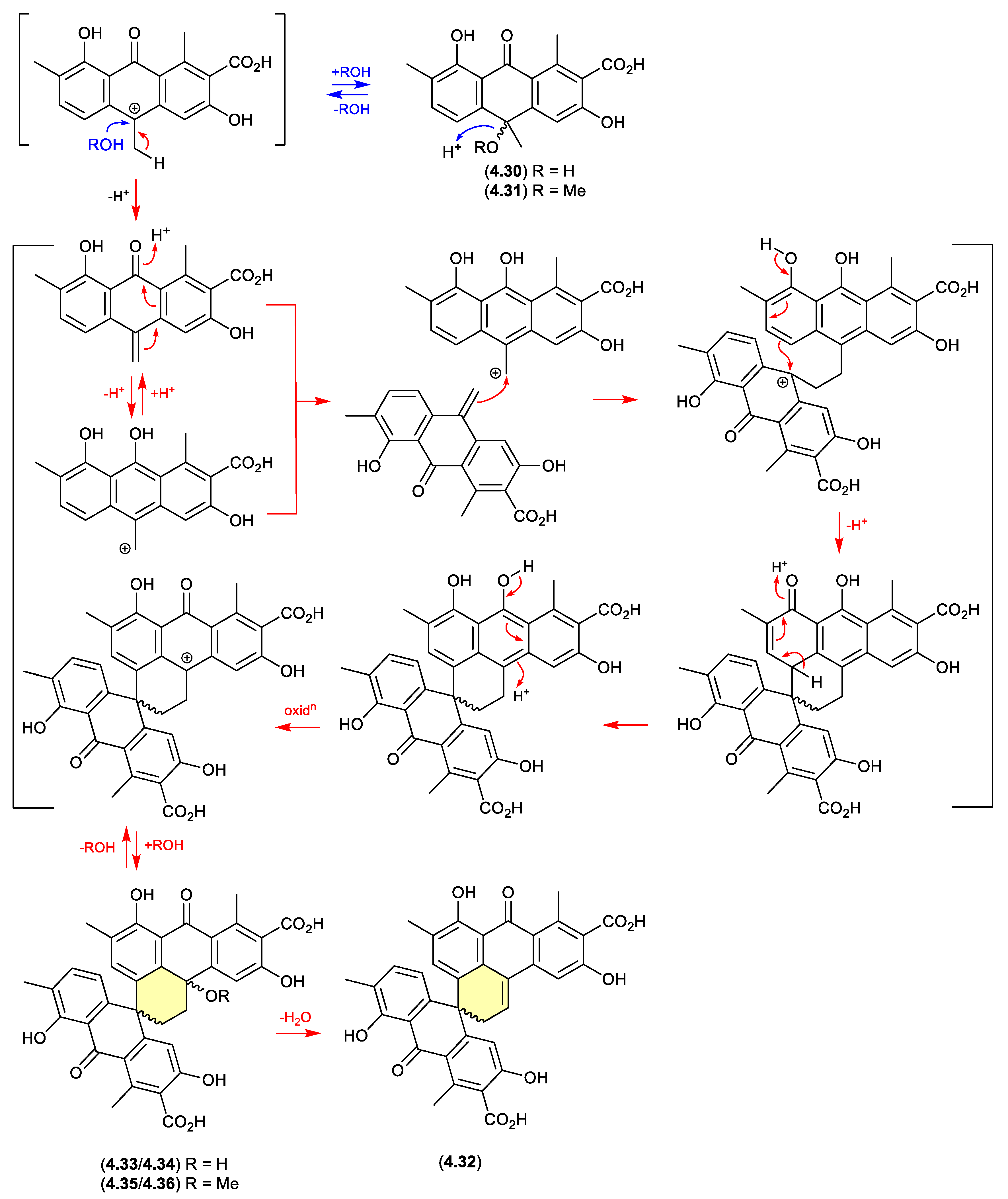
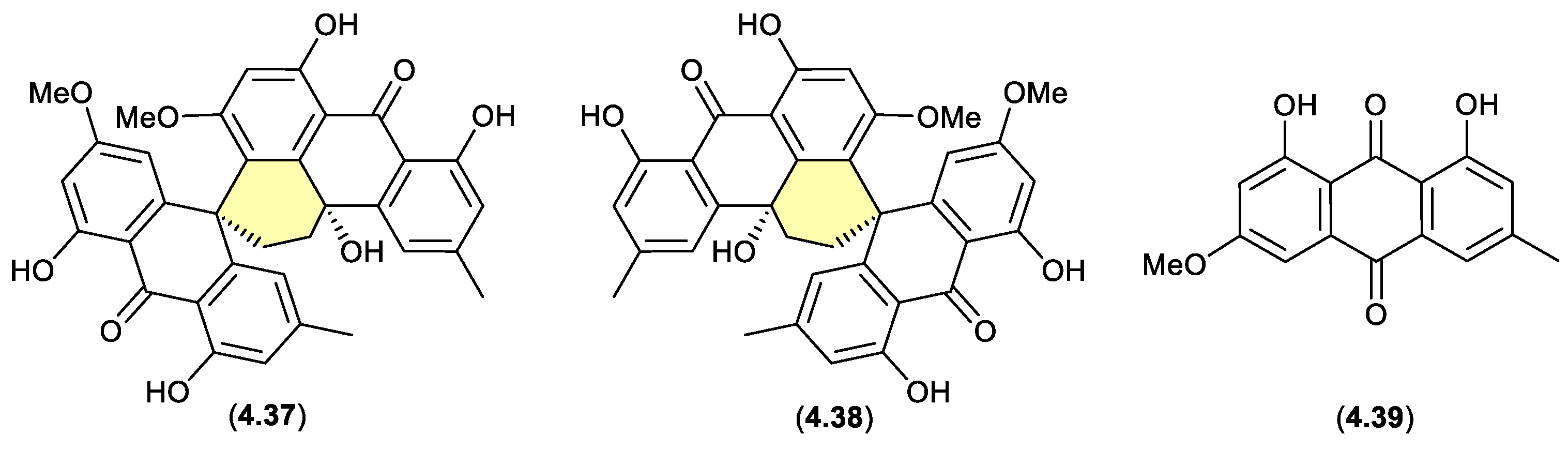
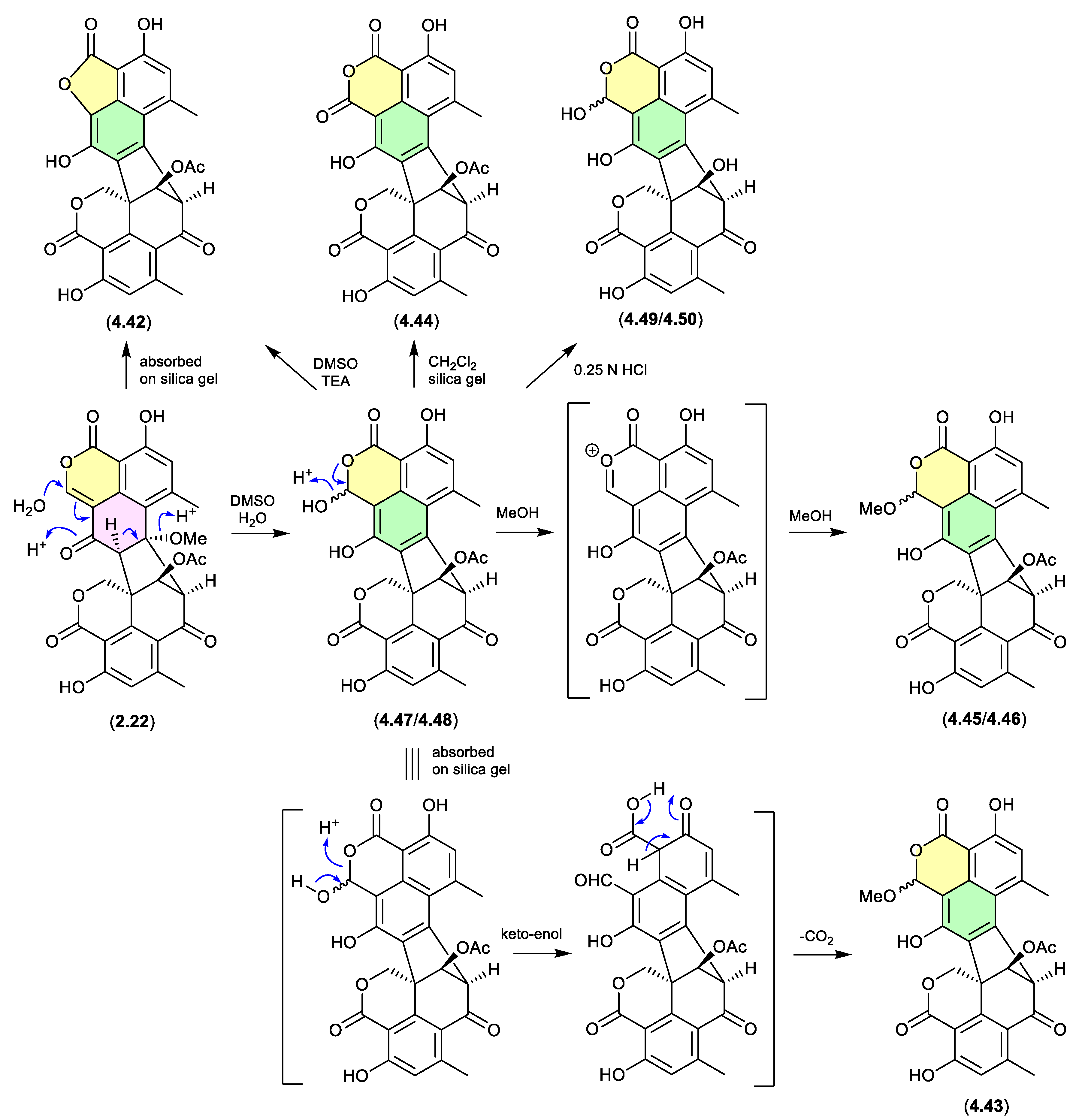
4.3. Silica Gel
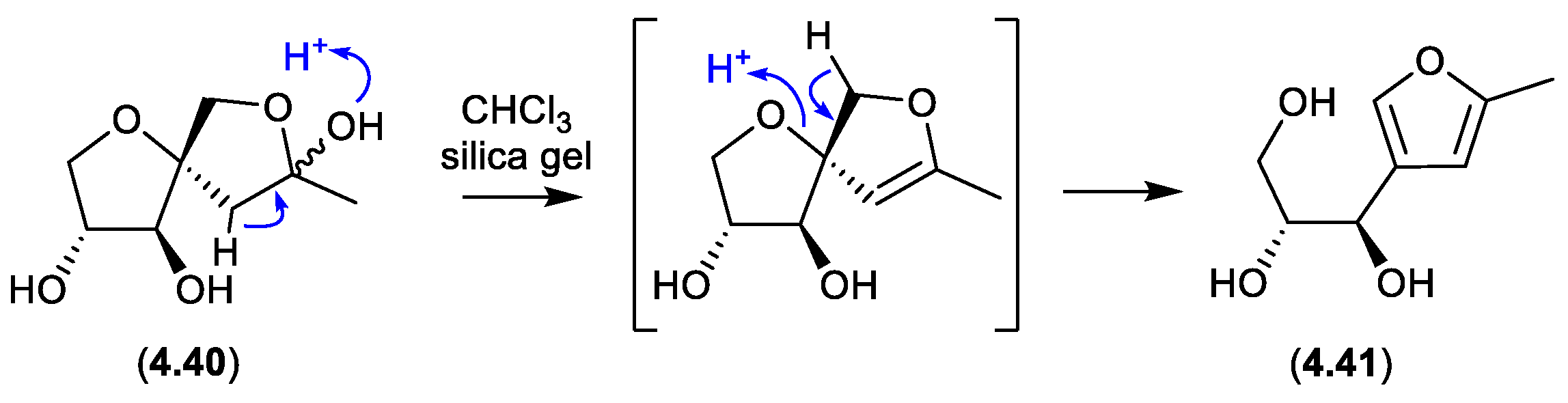
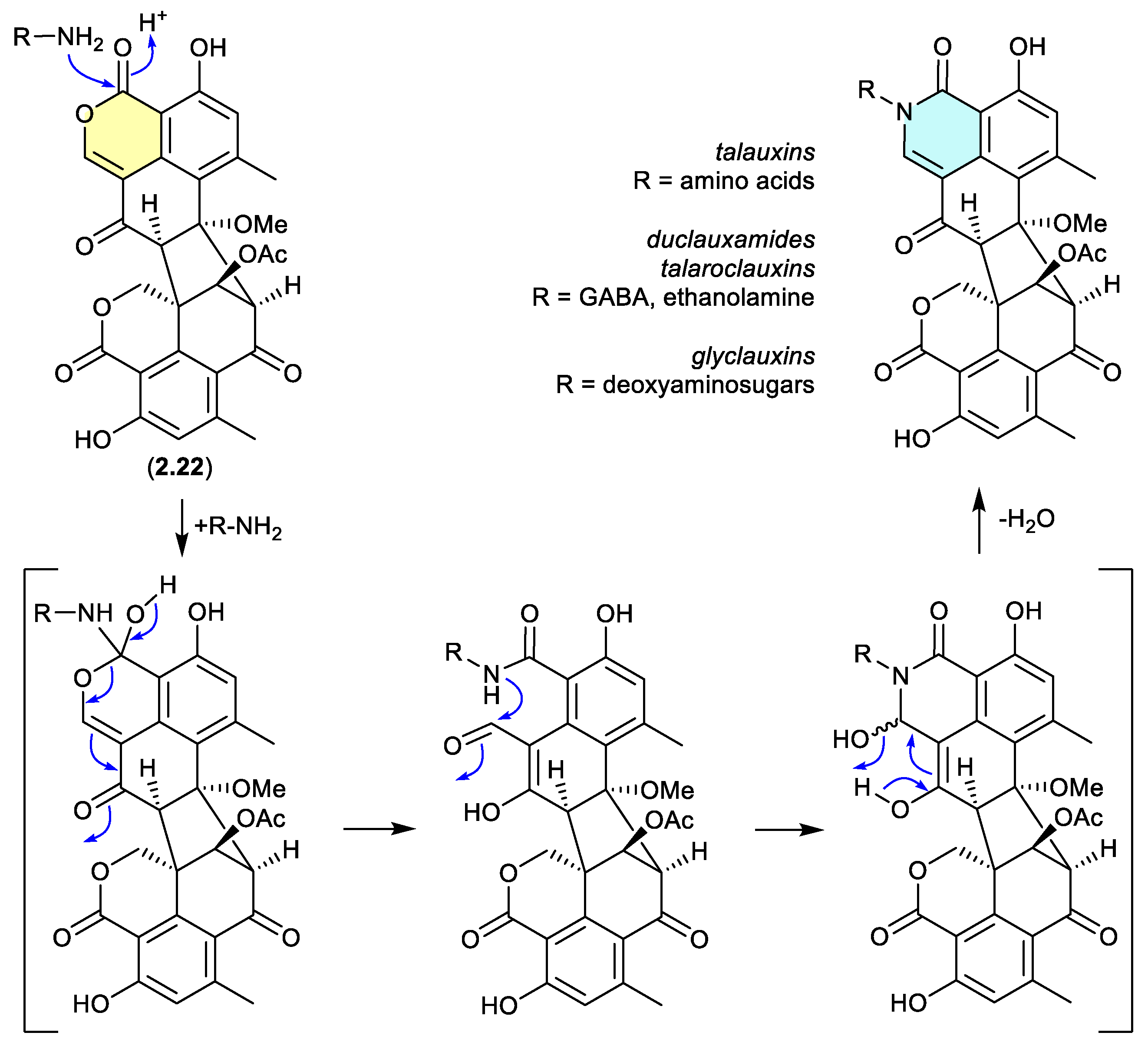
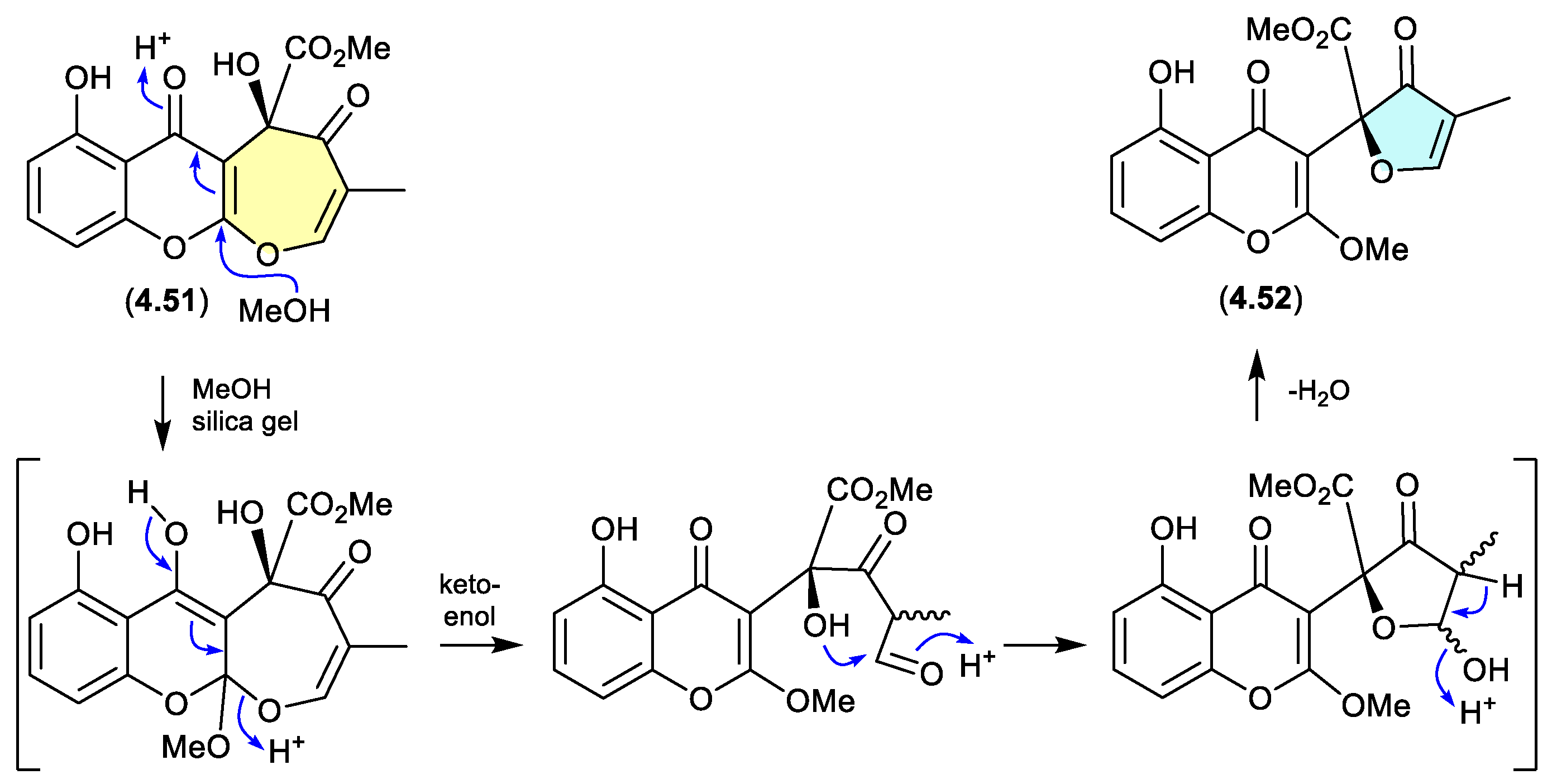
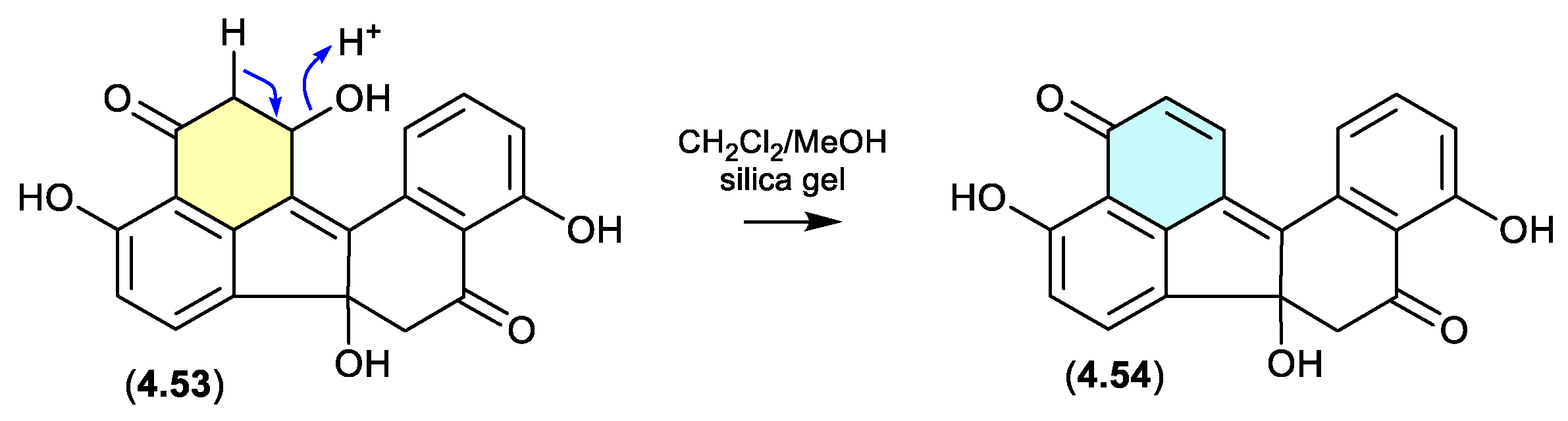
5. Light
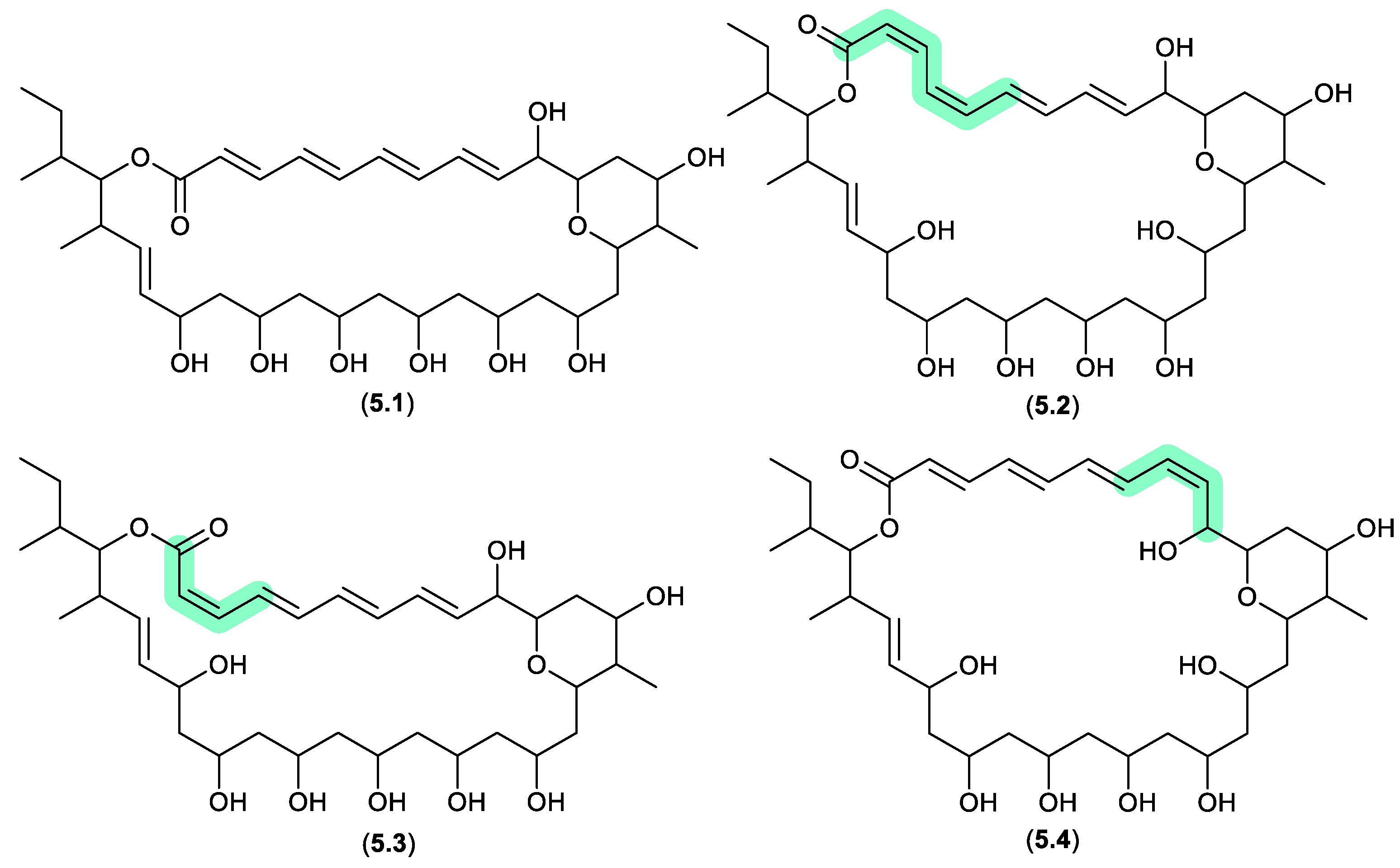
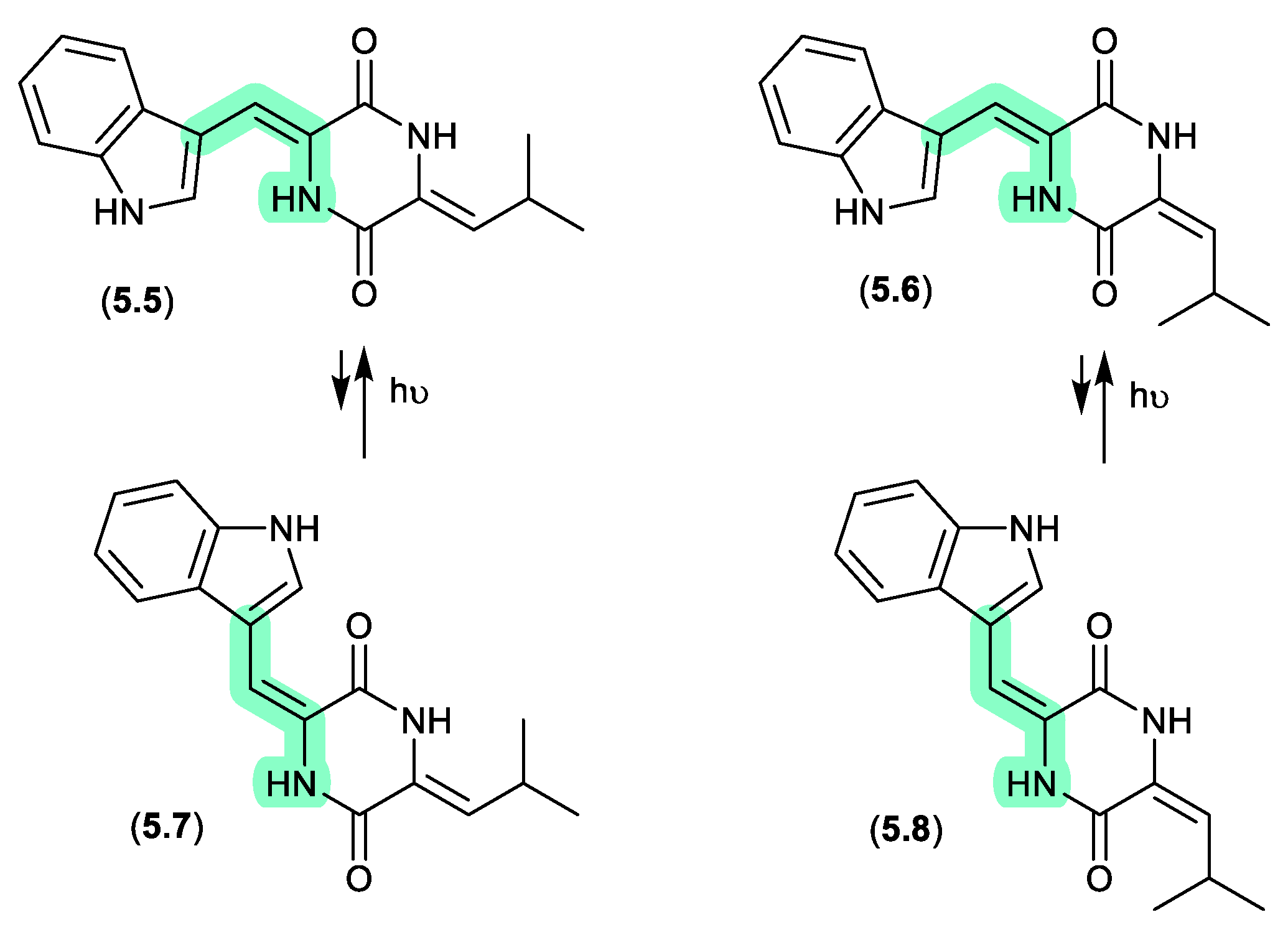
5.1. Photoisomerization
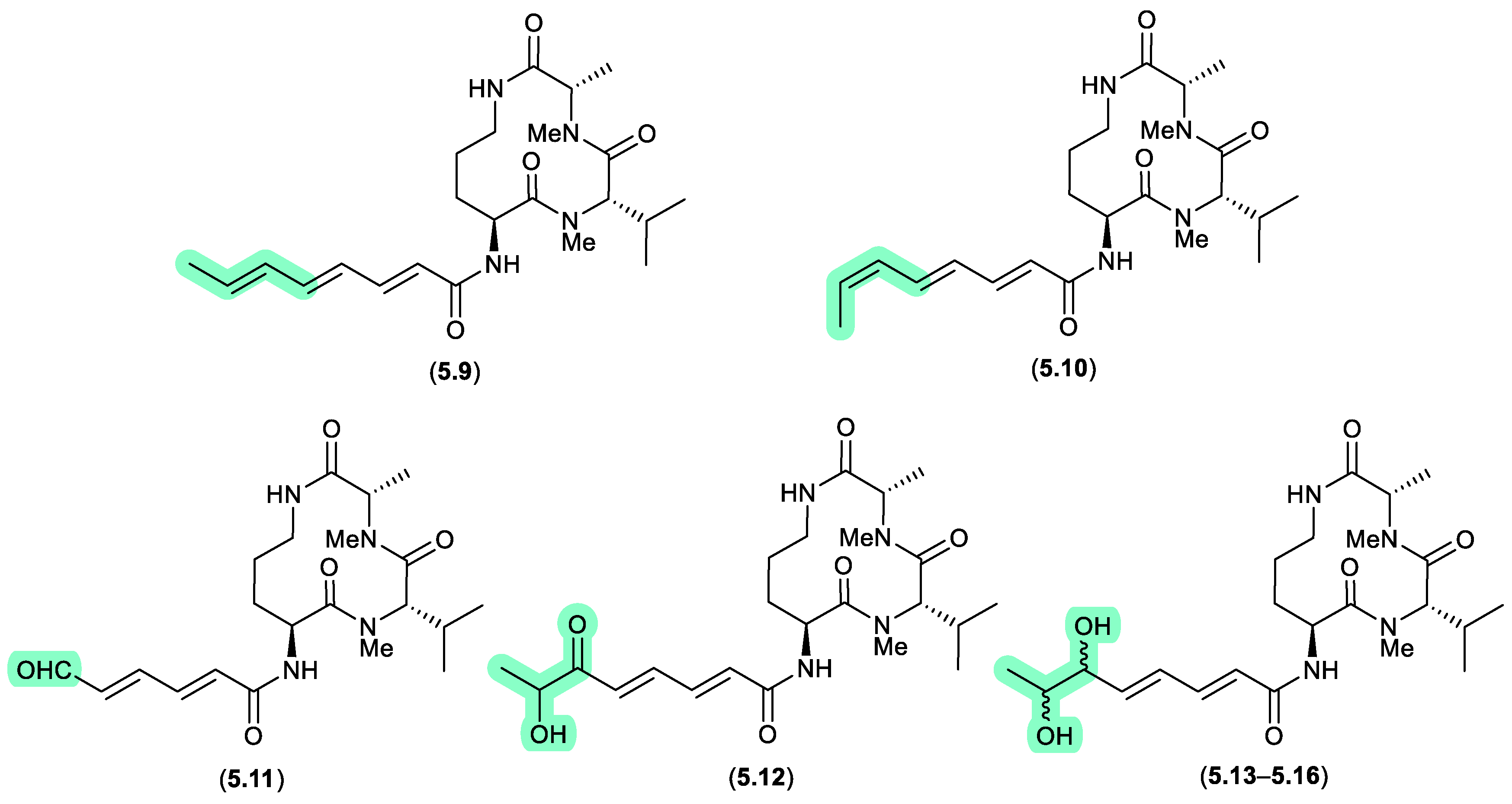
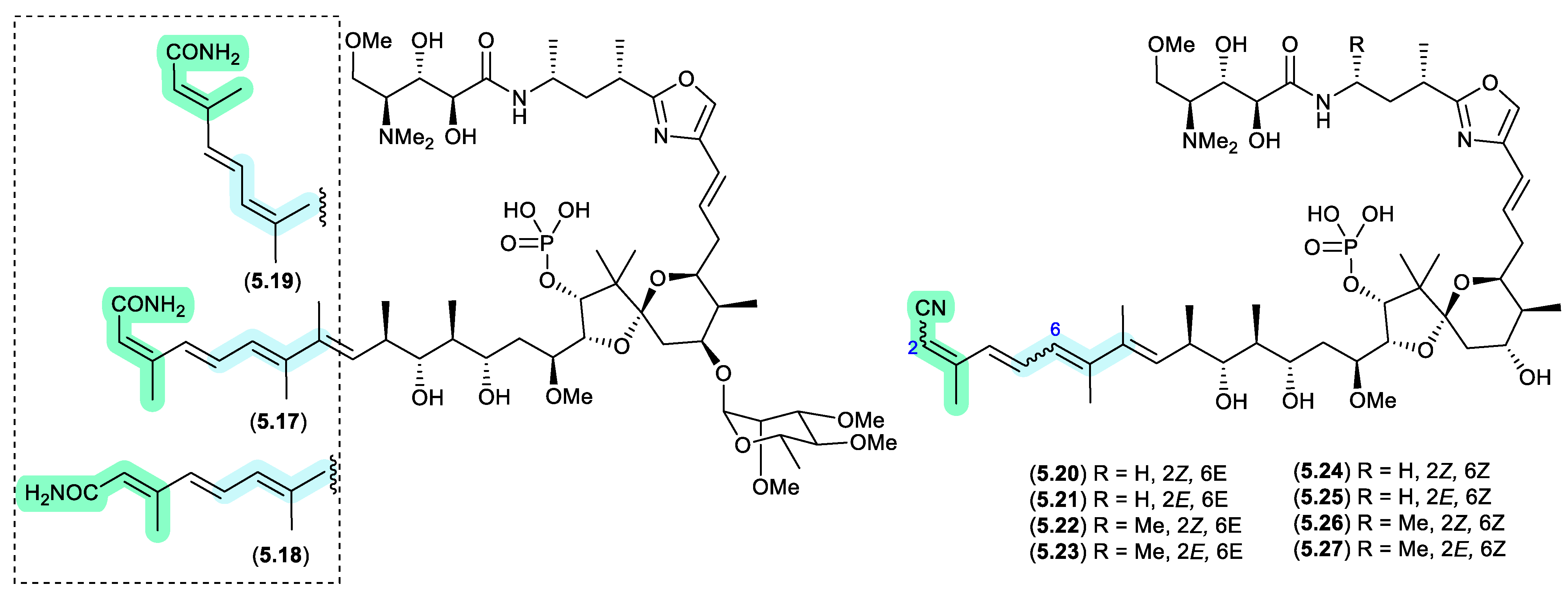
5.2. Photooxidation
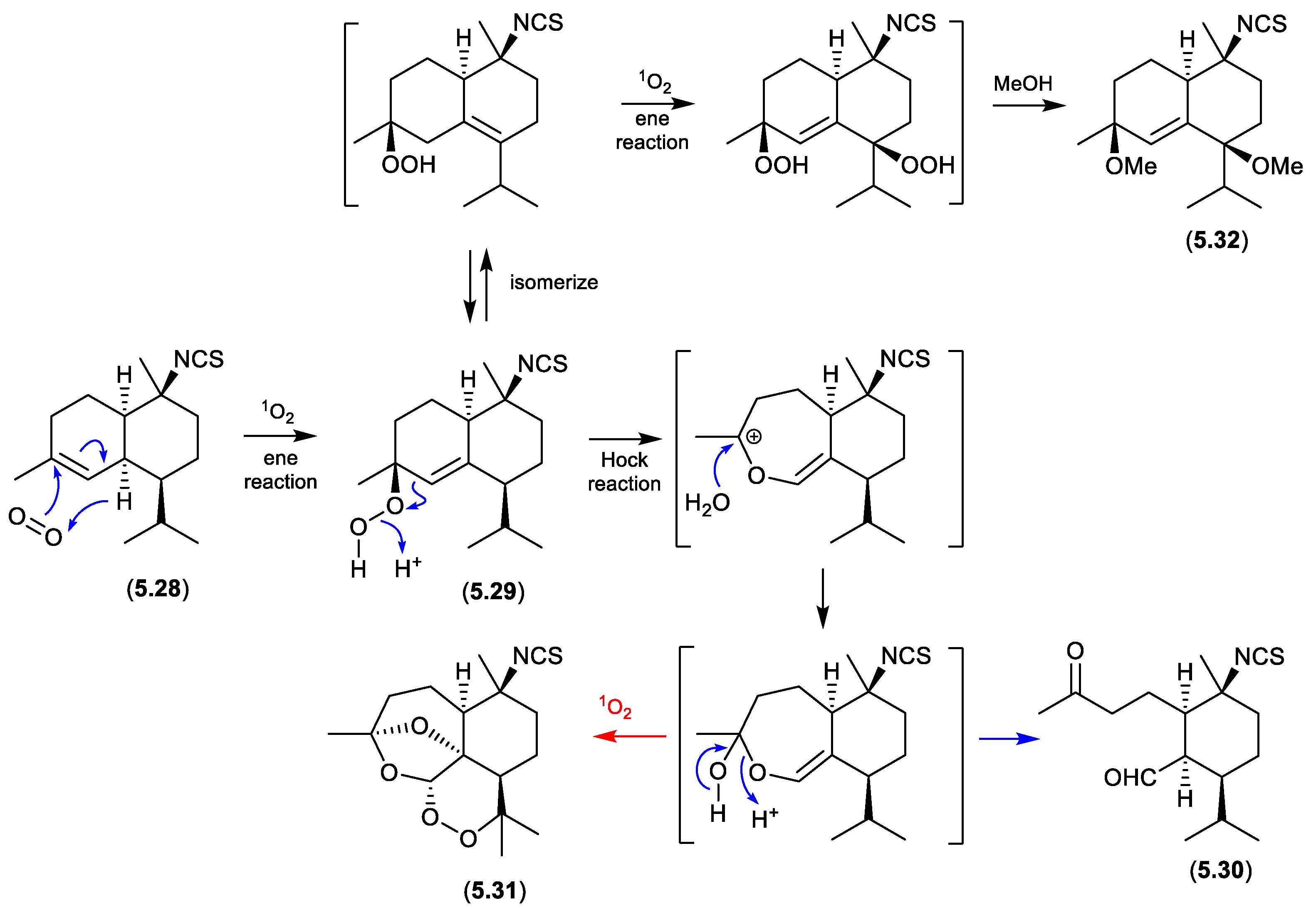
5.3. Photoreactivity
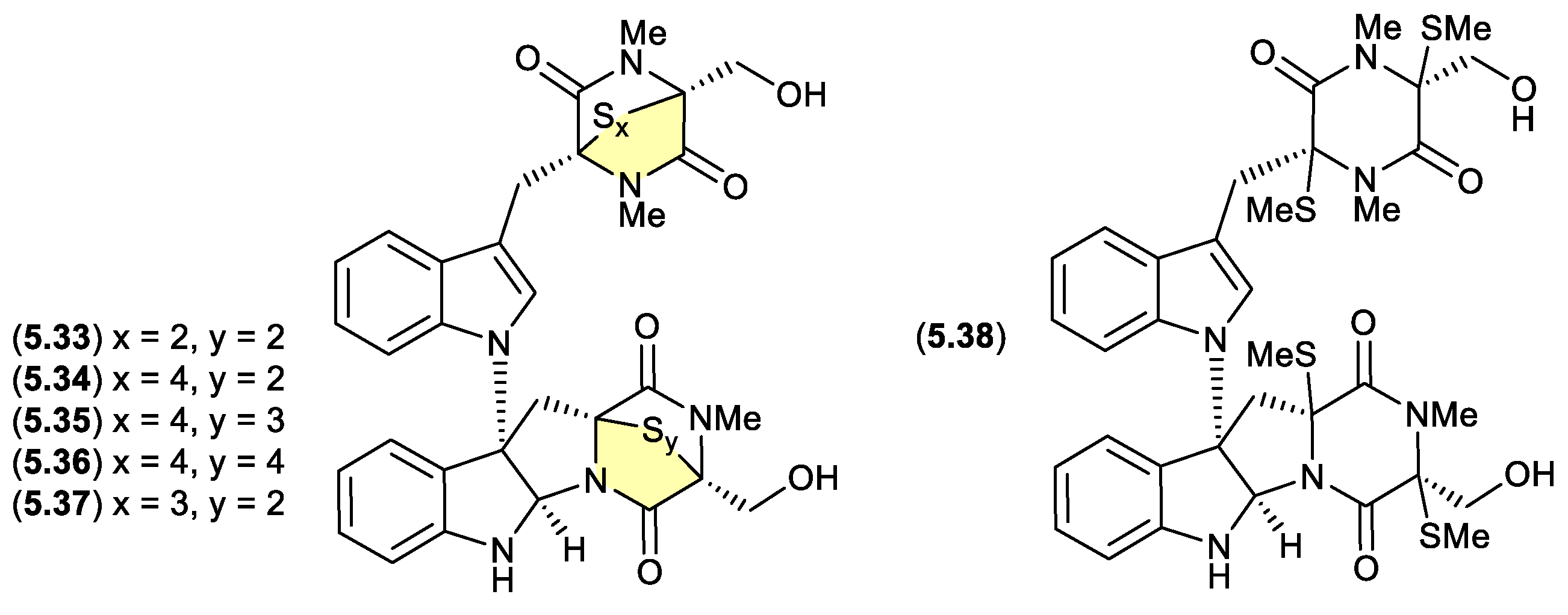
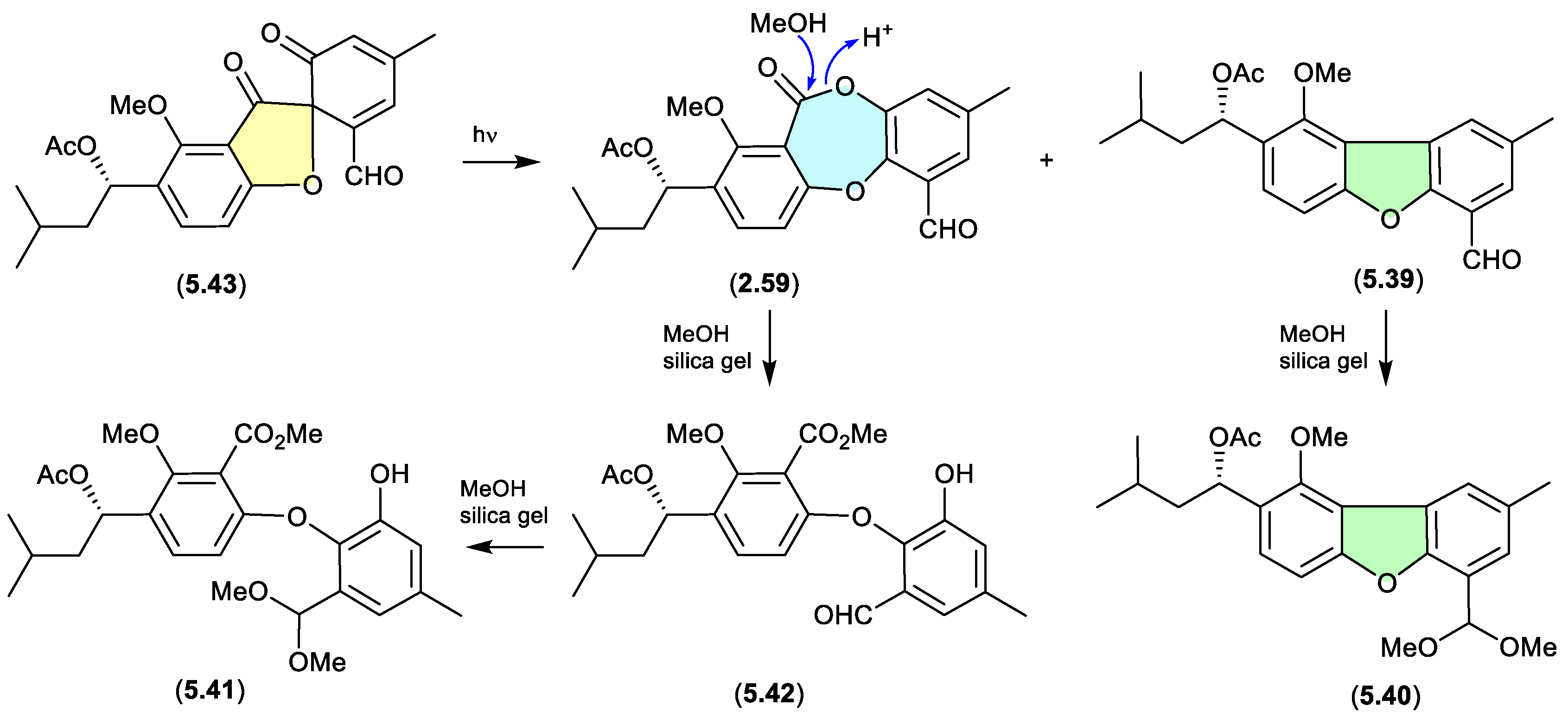
6. Air Oxidation
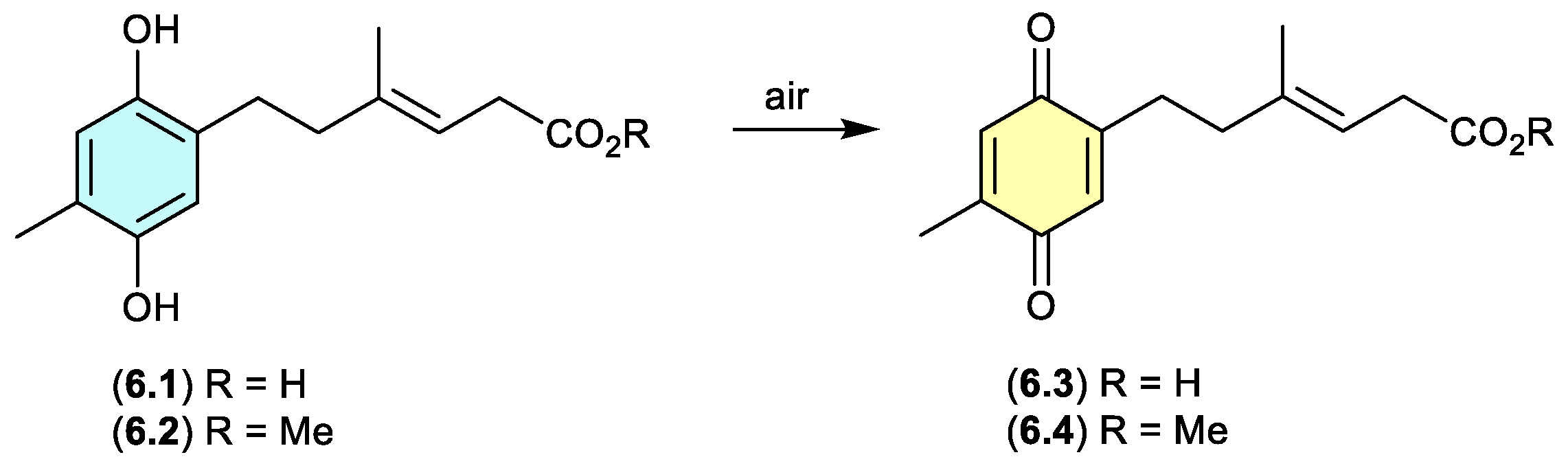
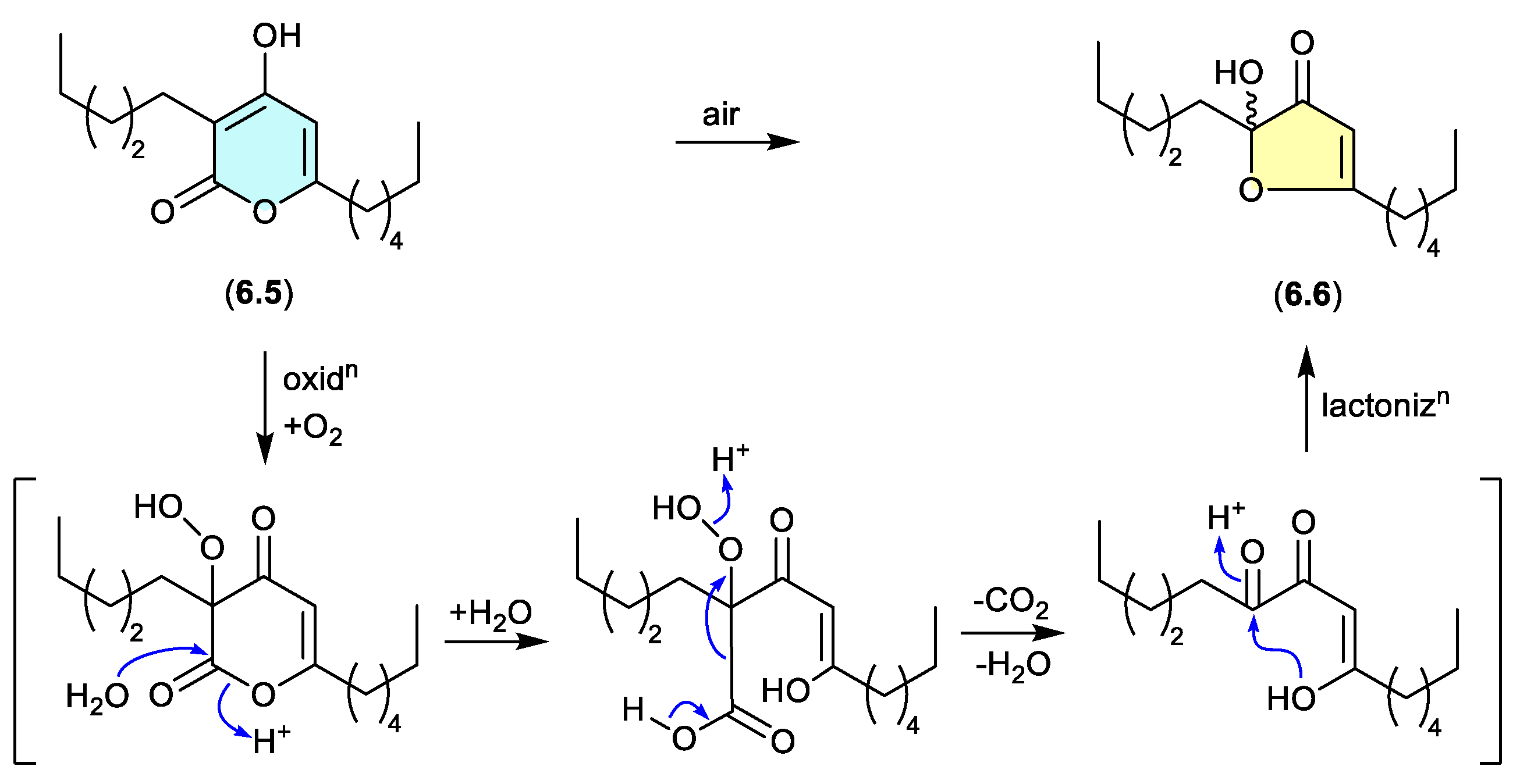
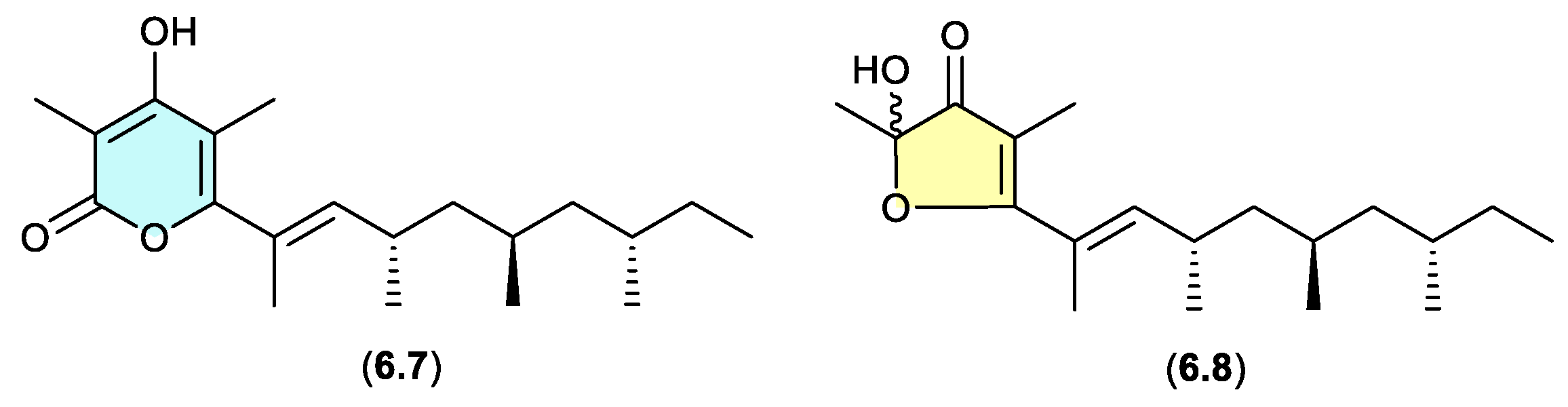
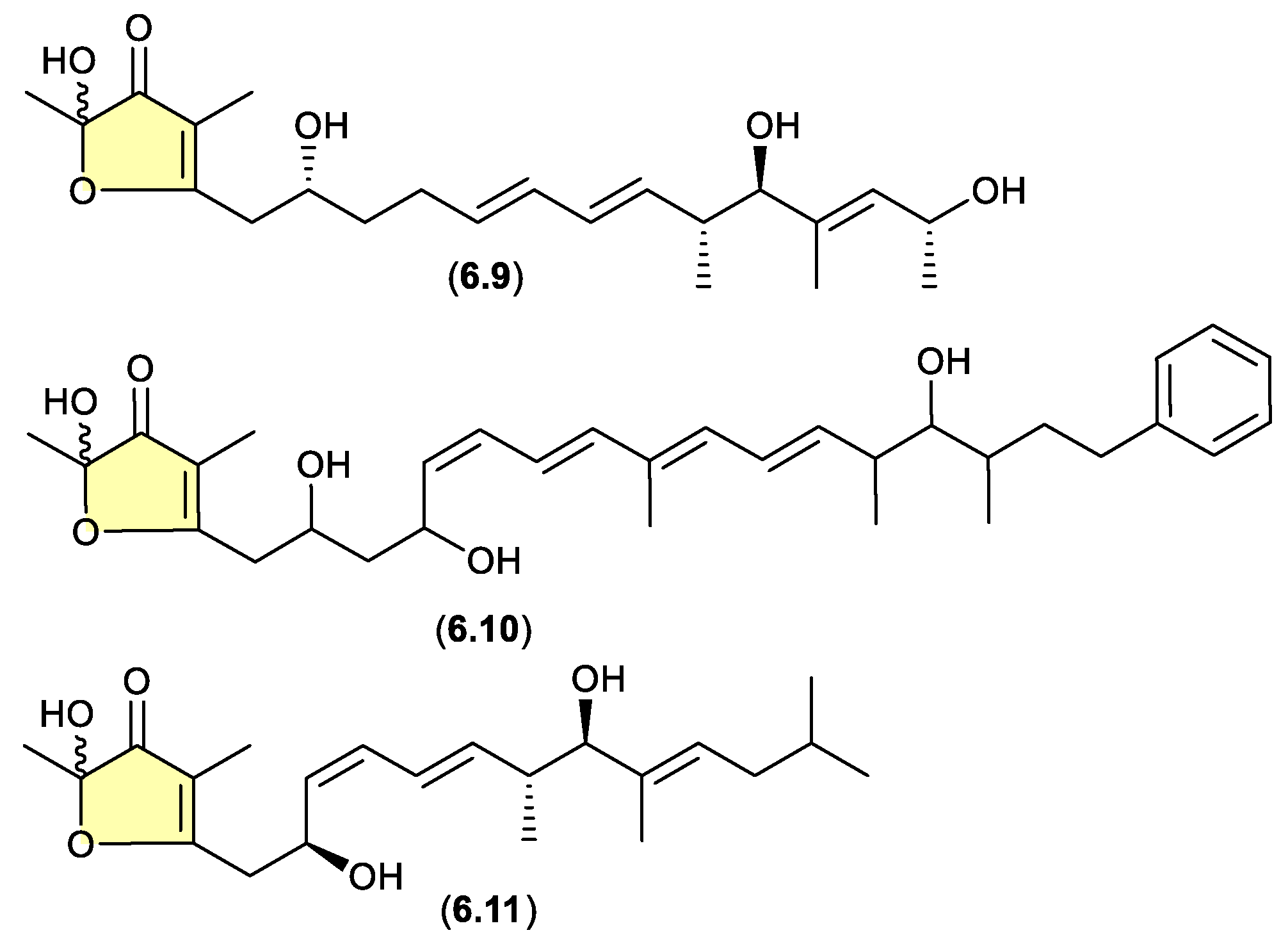
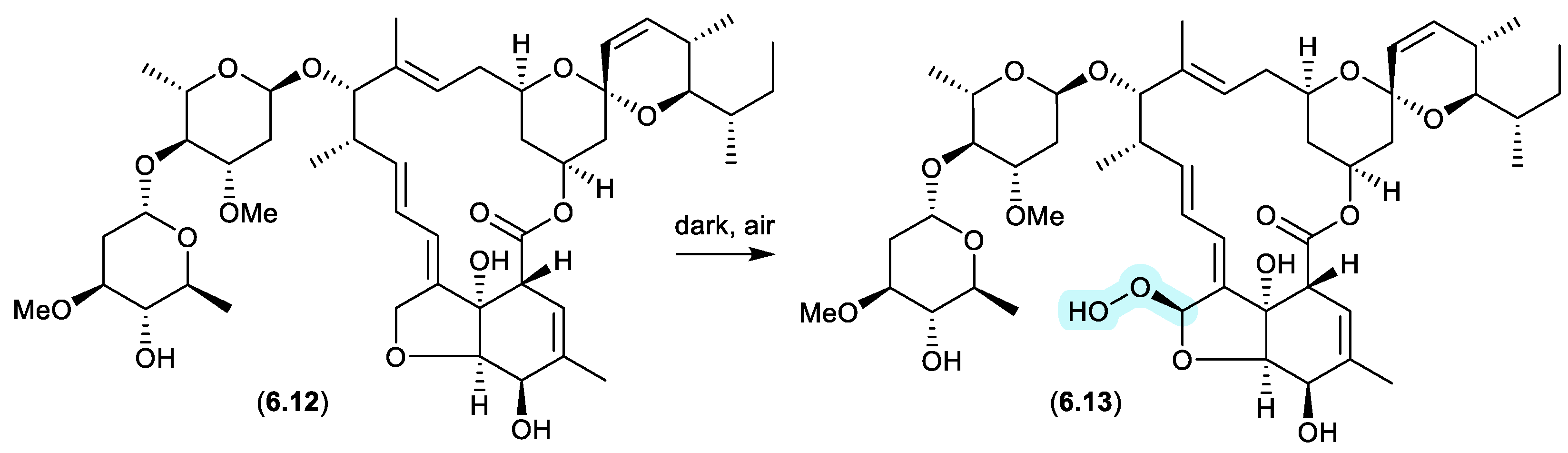
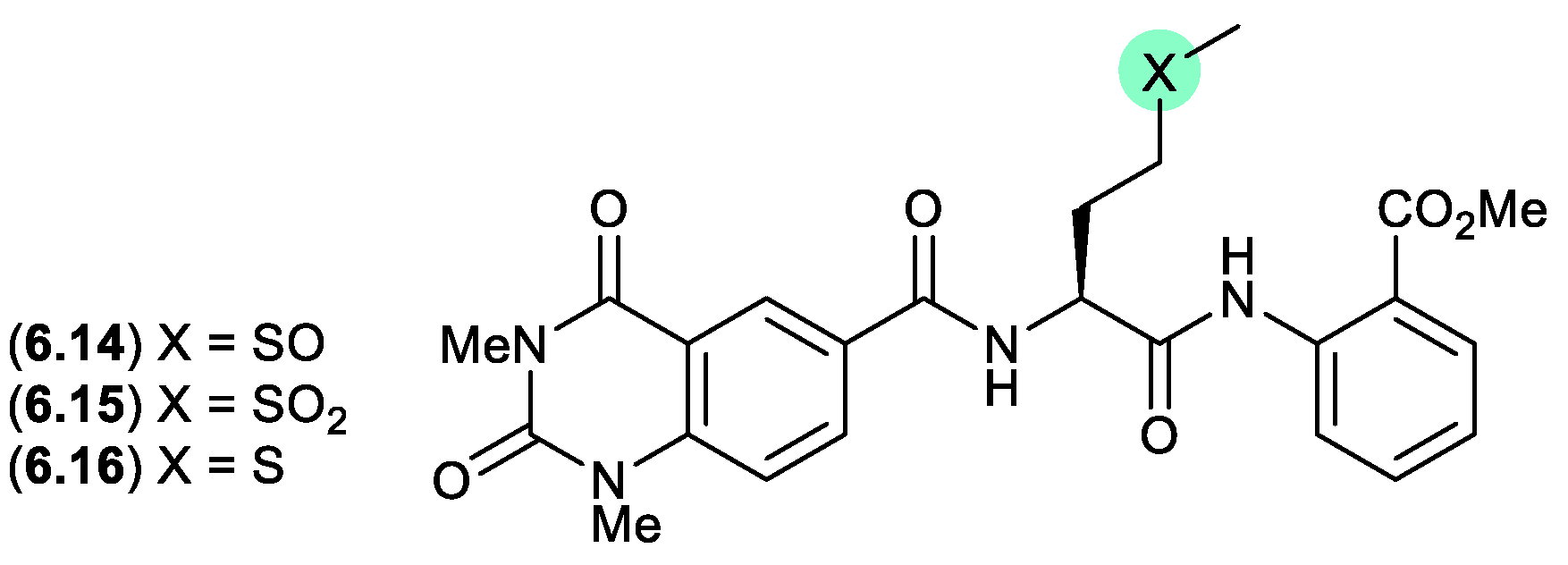
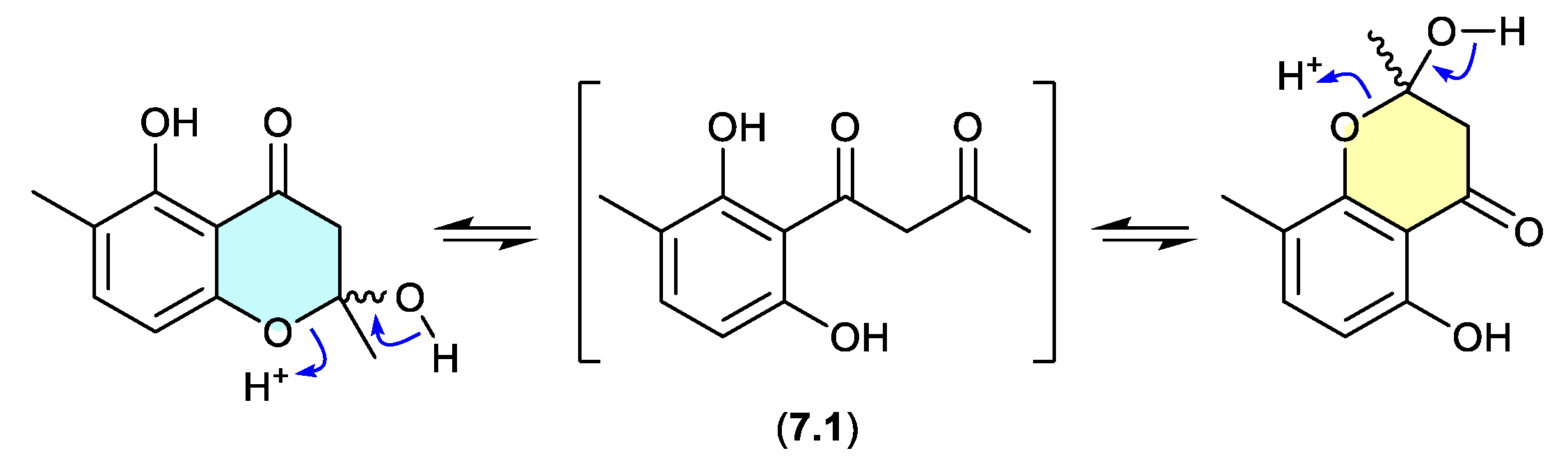
7. Acetal/ketal Equilibration
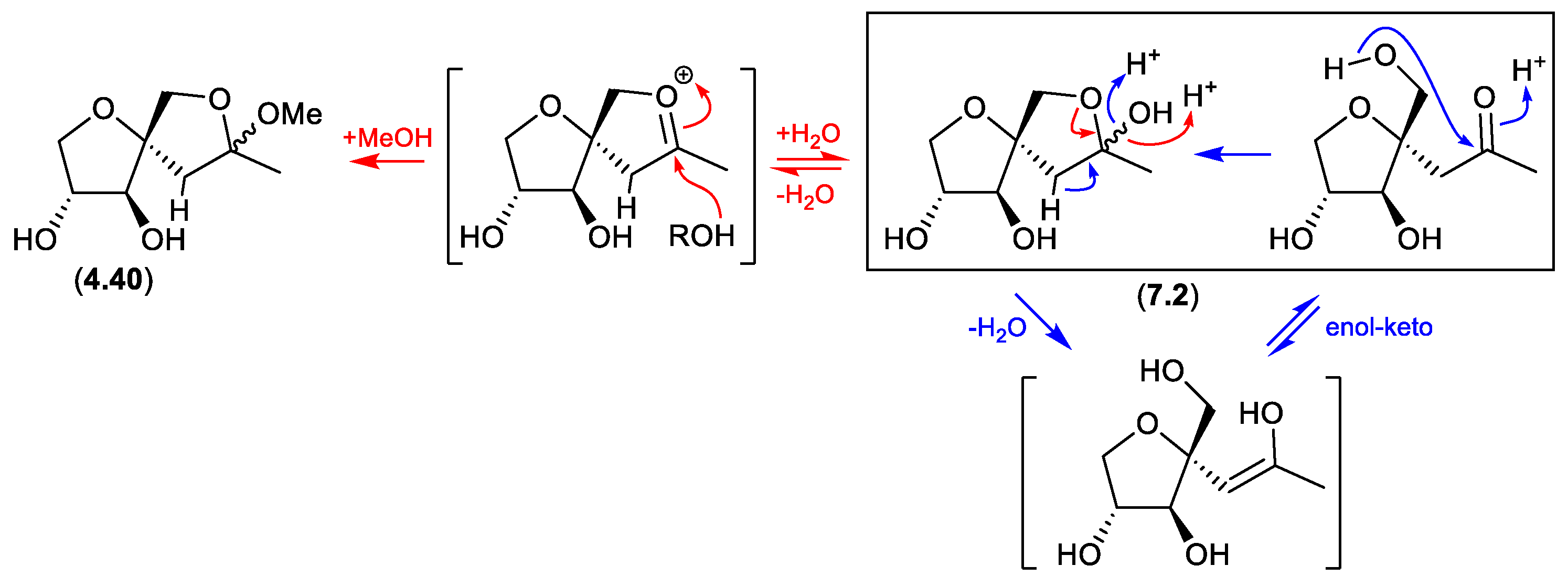
8. Trans-Esterification
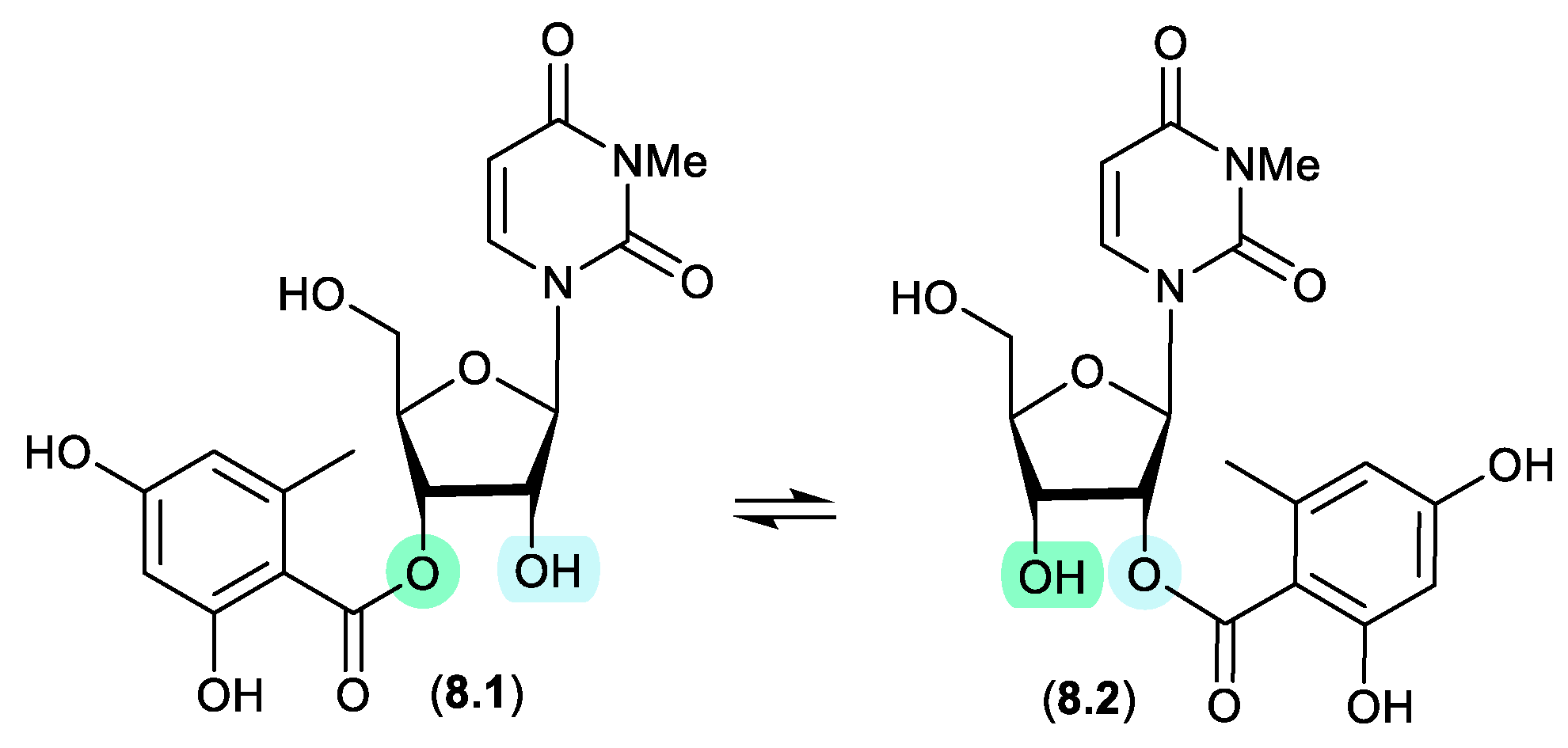
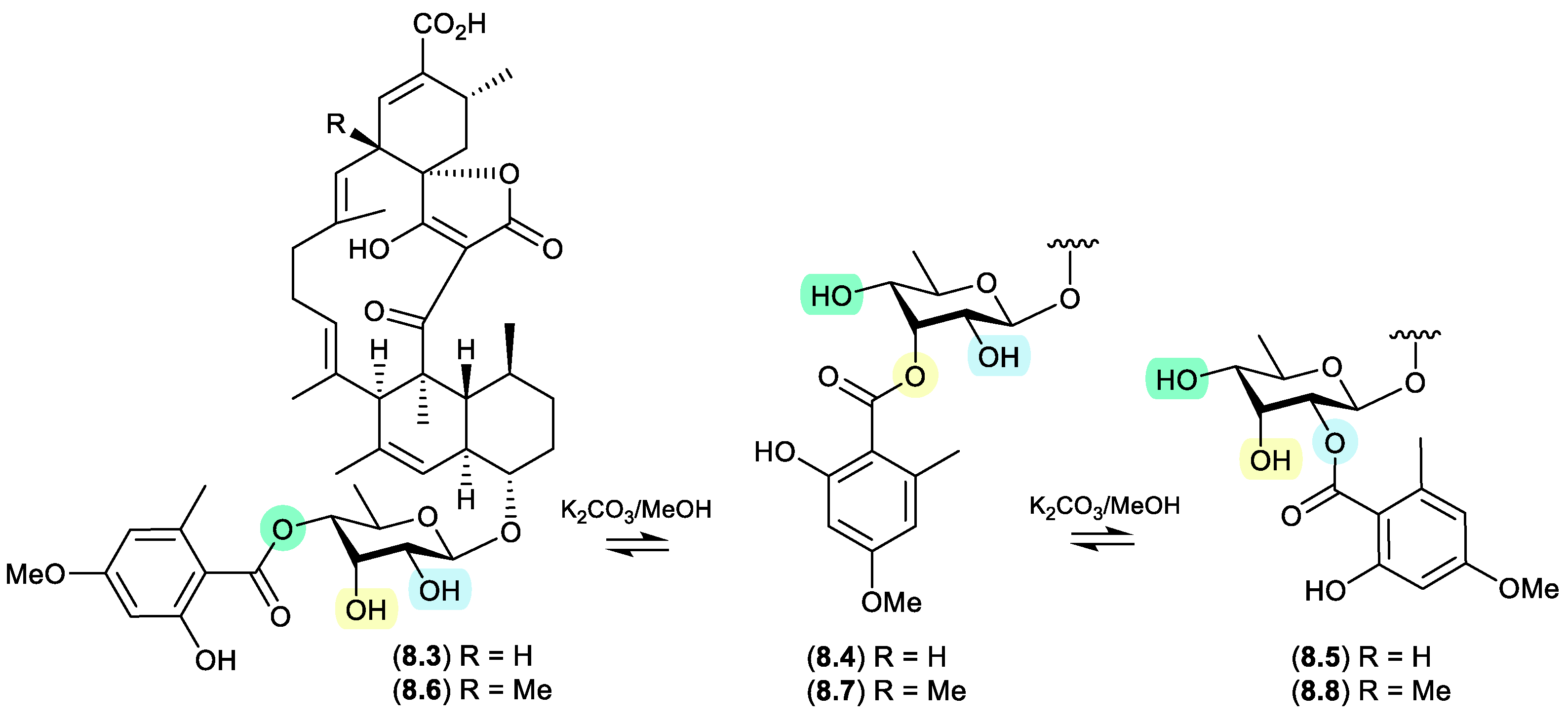
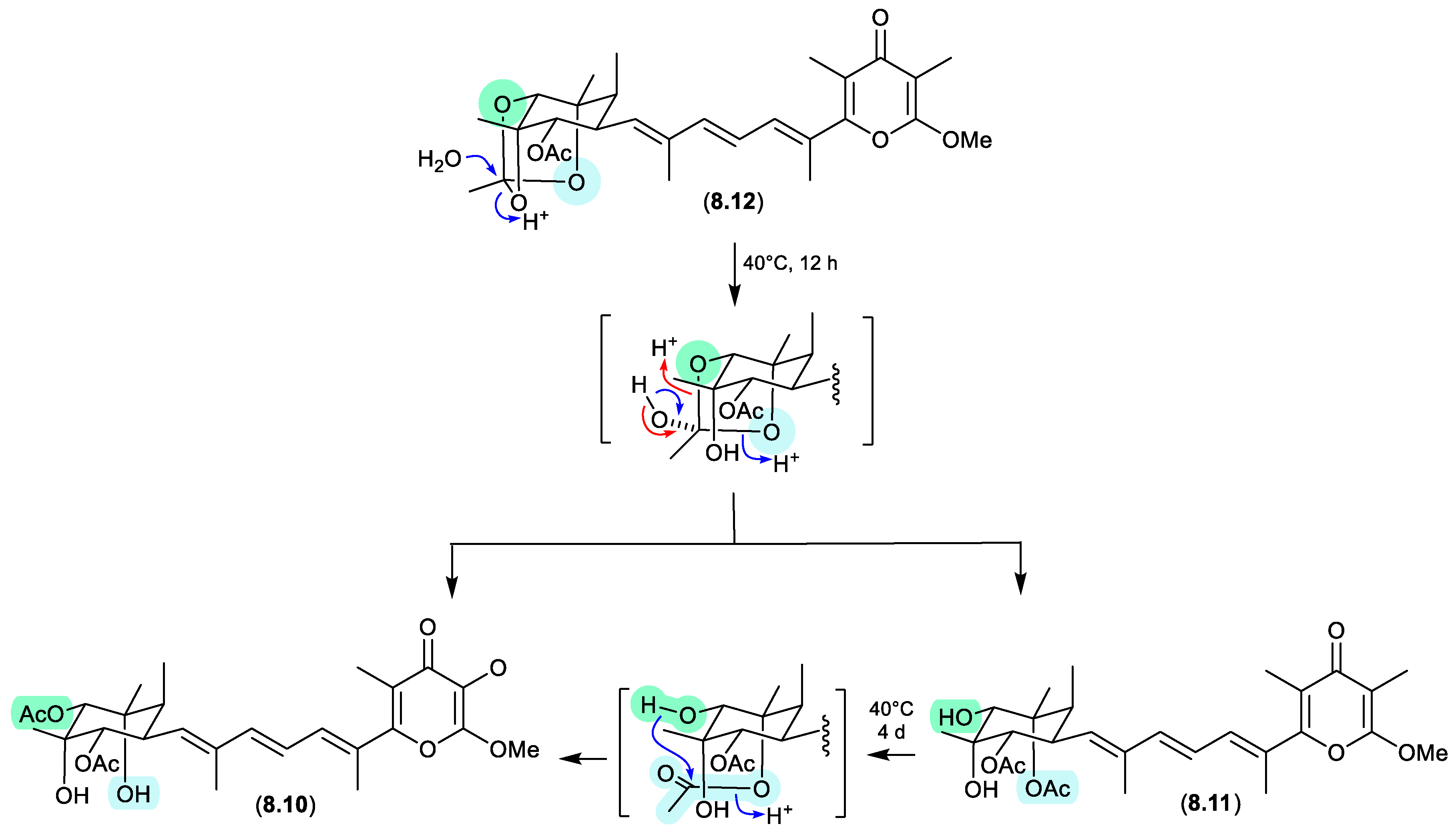
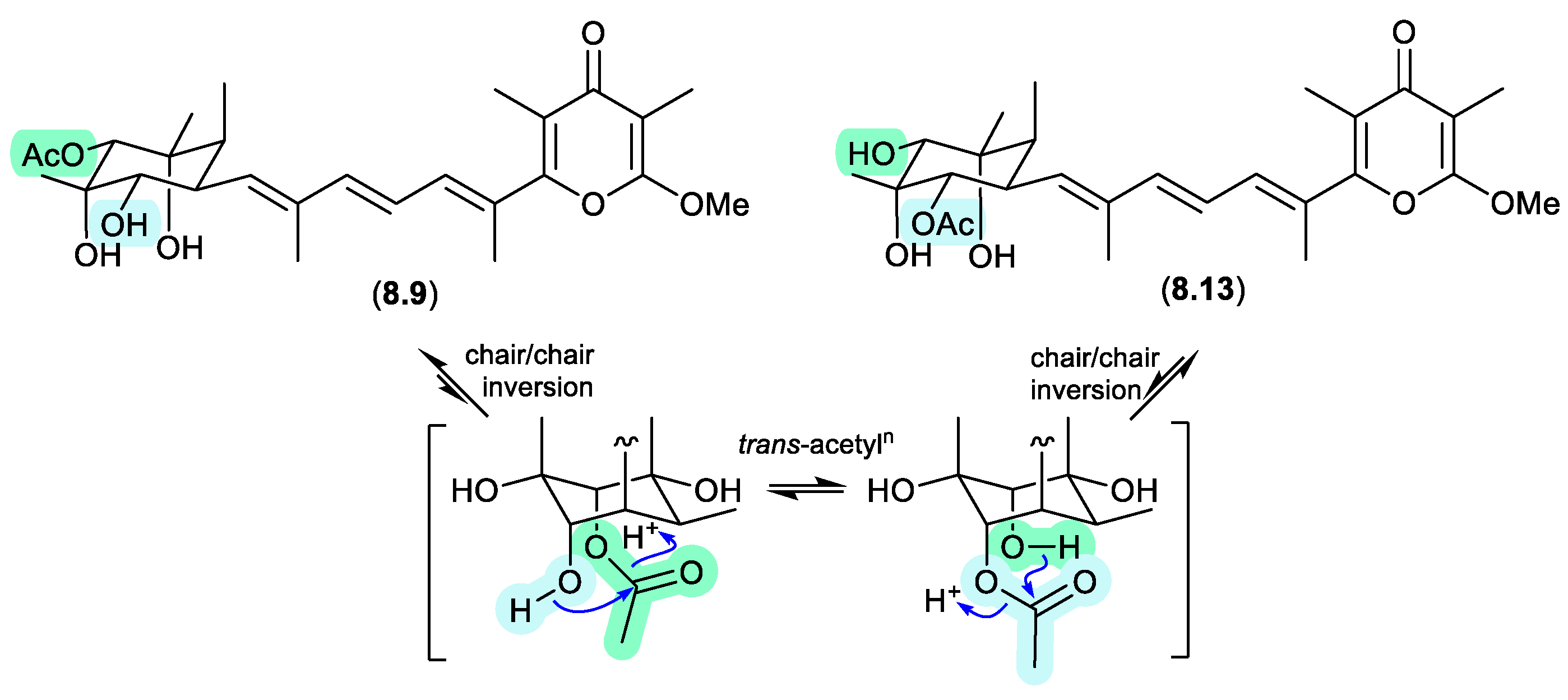
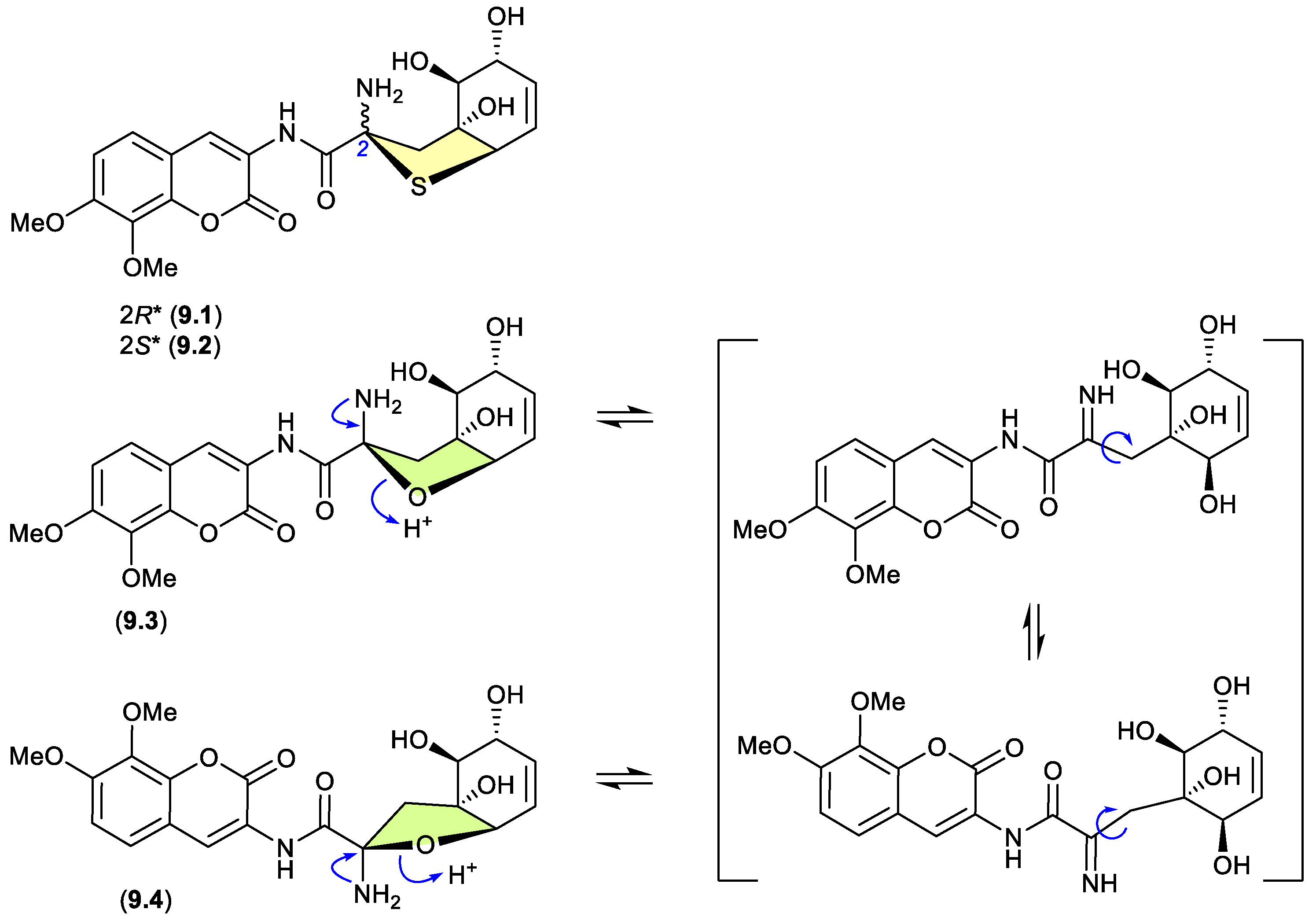
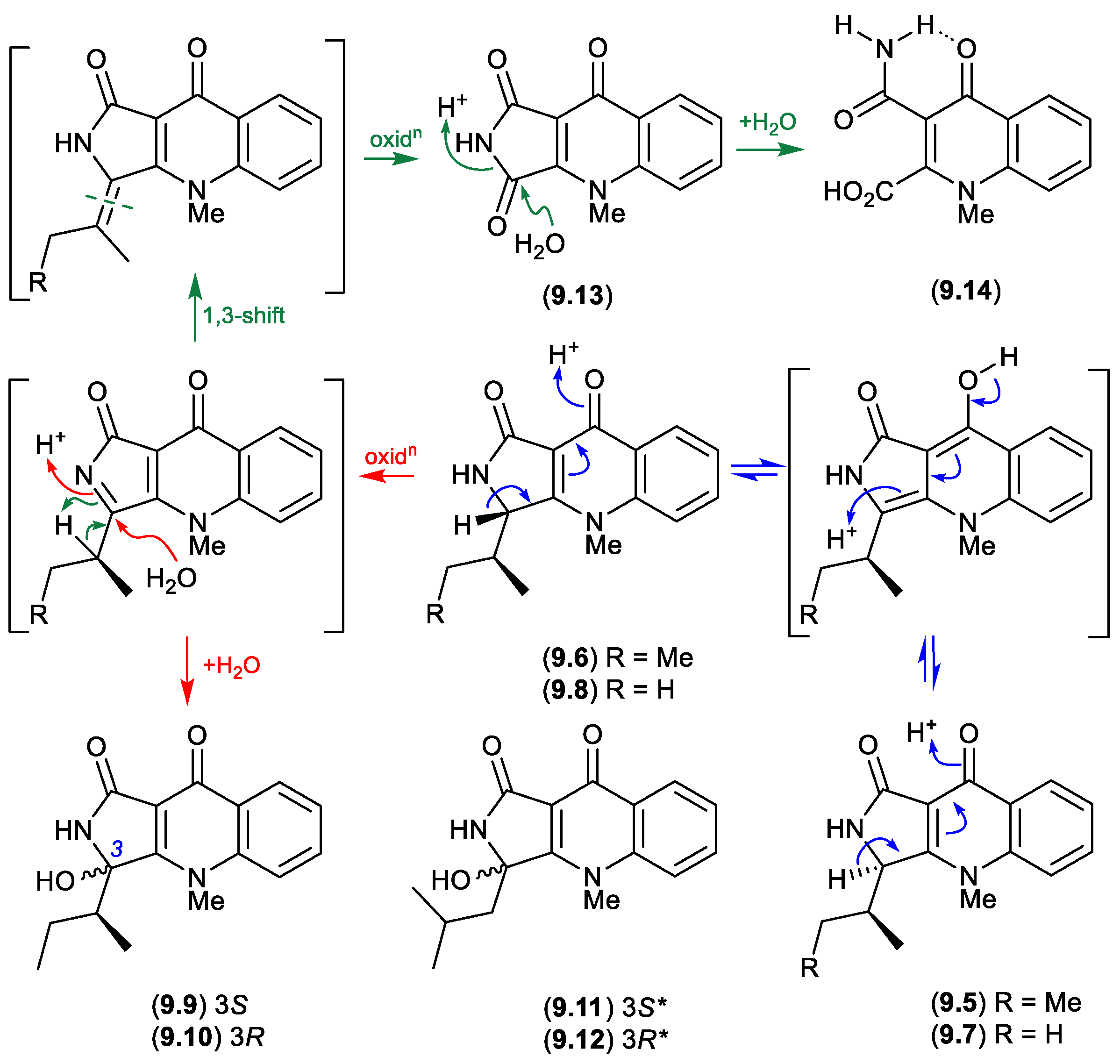
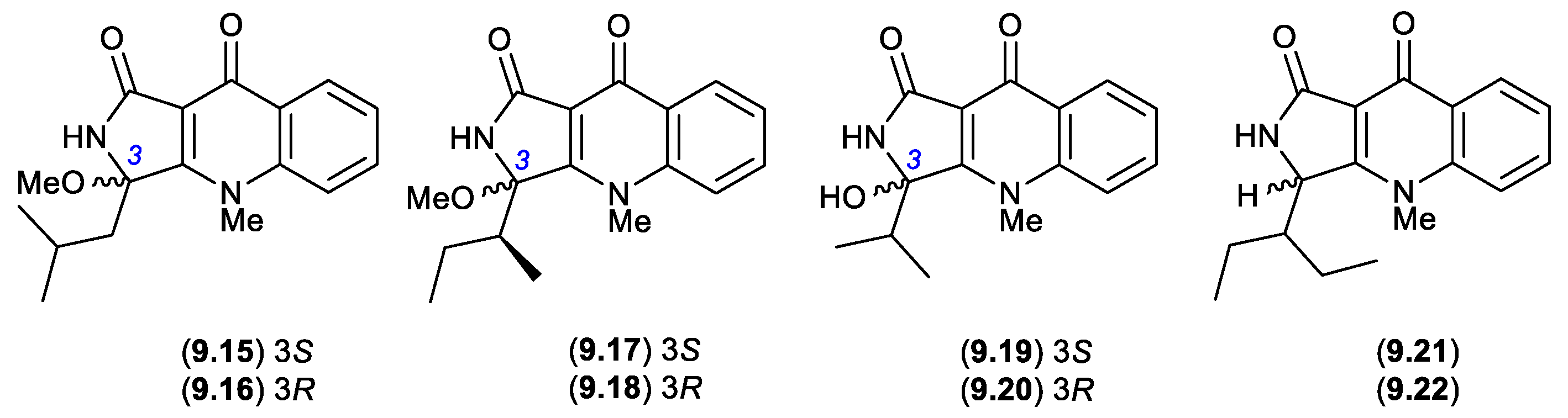
9. Epimerization
10. Cryptic Natural Products
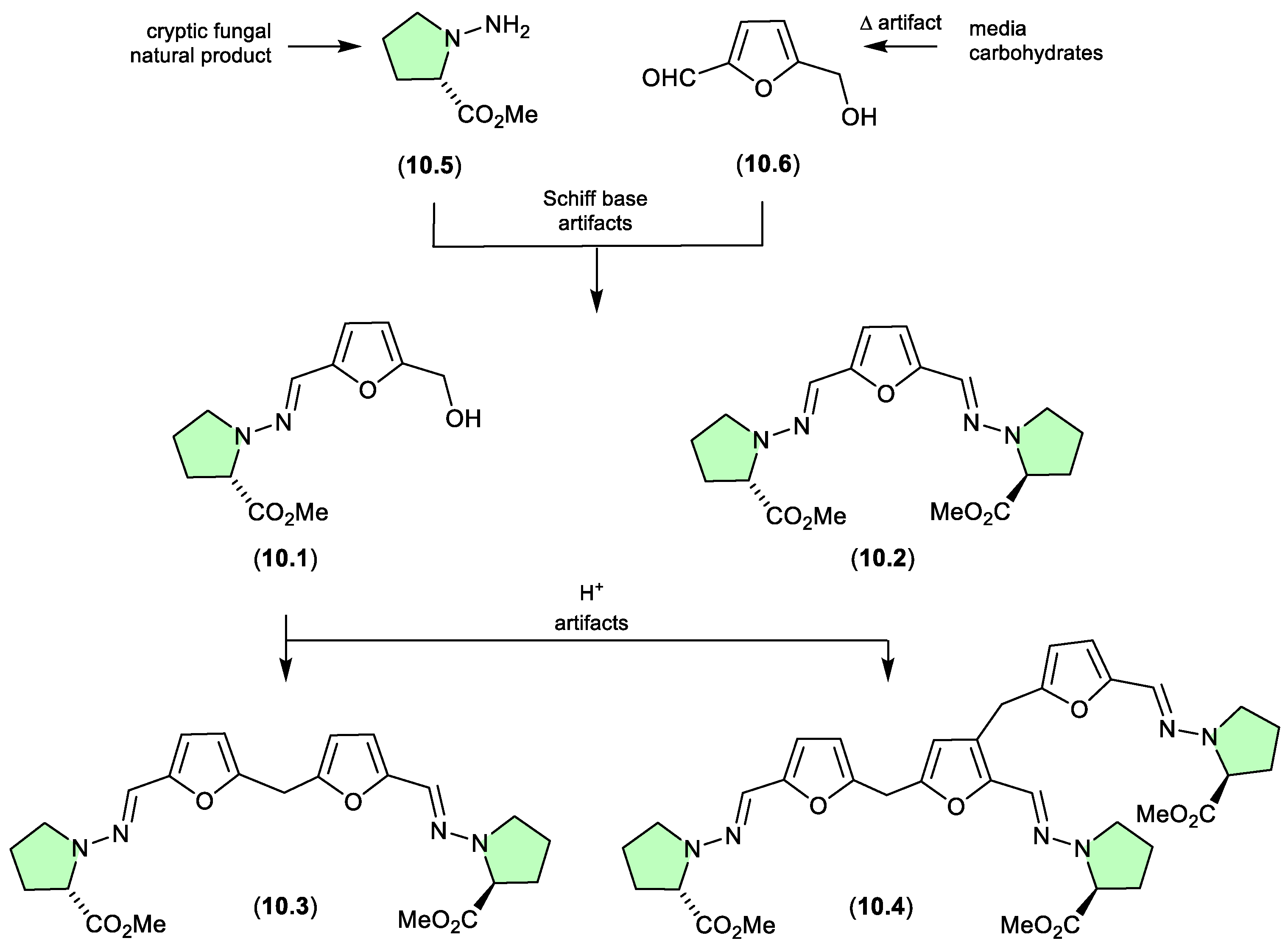
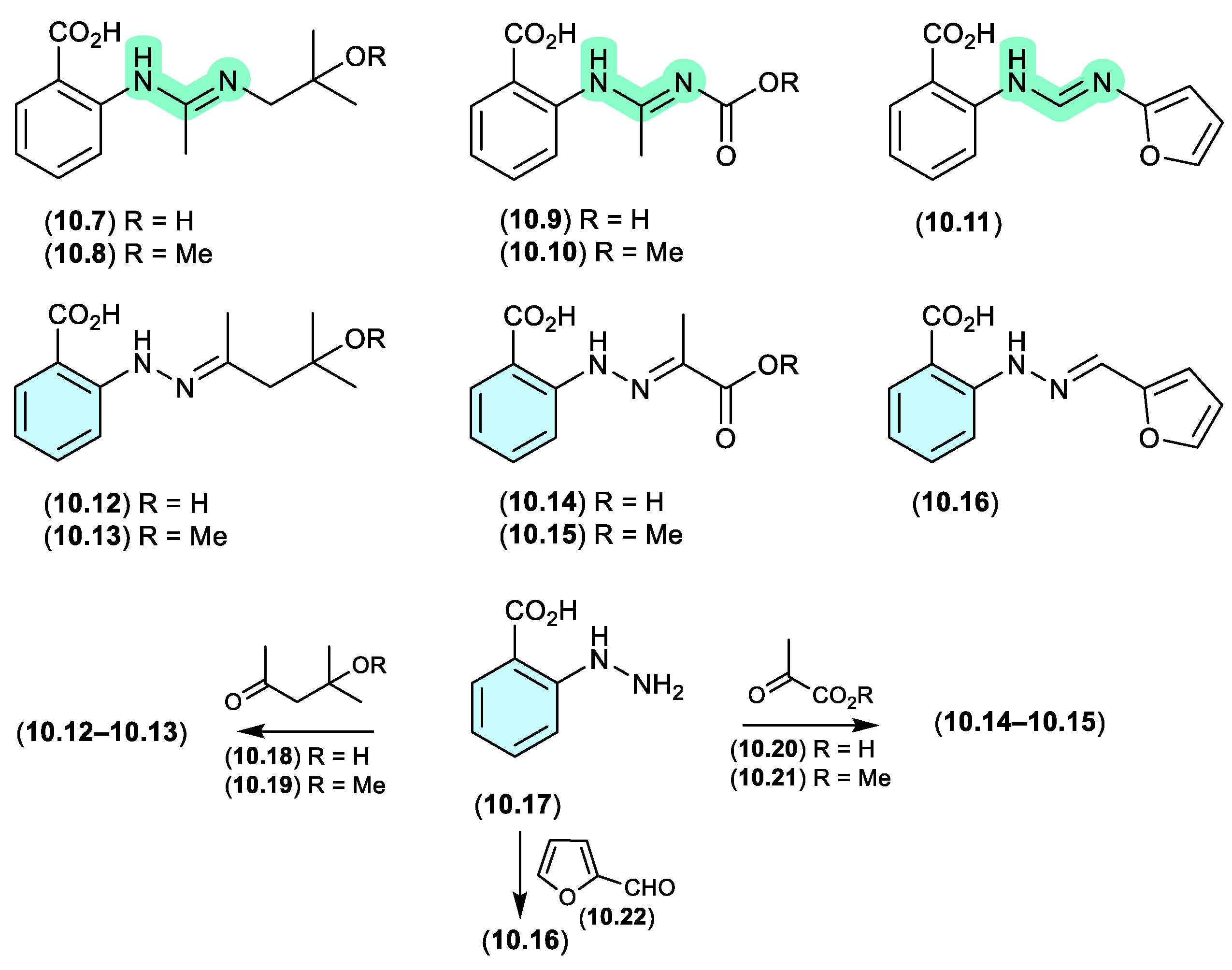
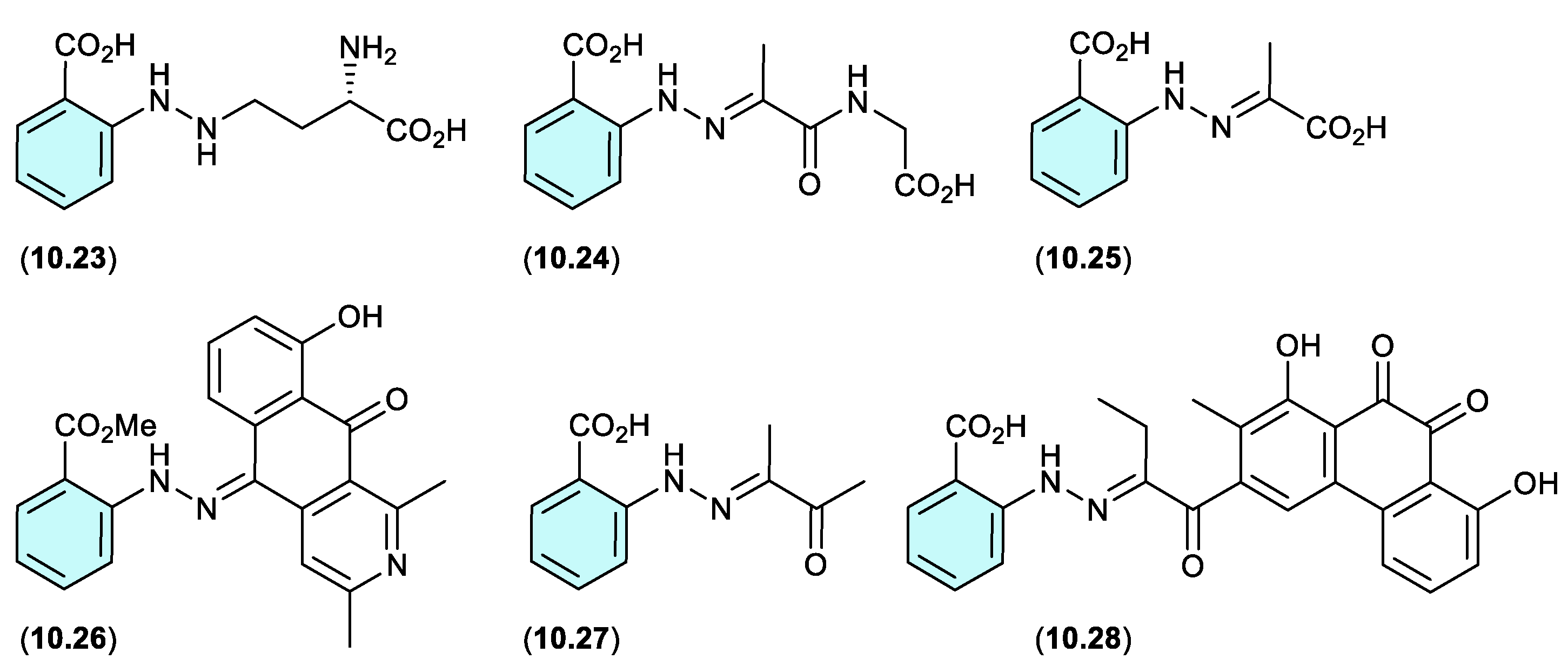
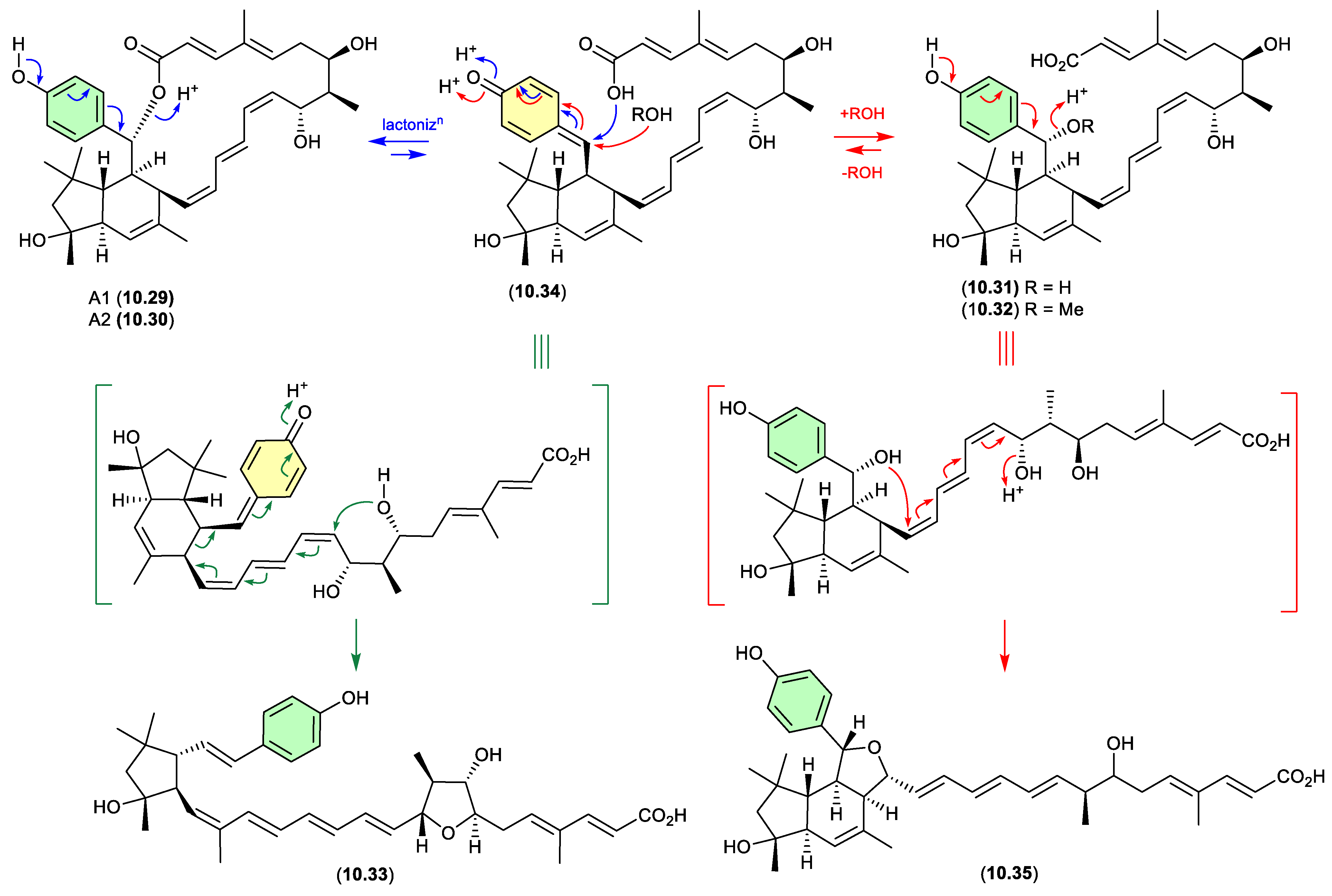
11. Biotransformation
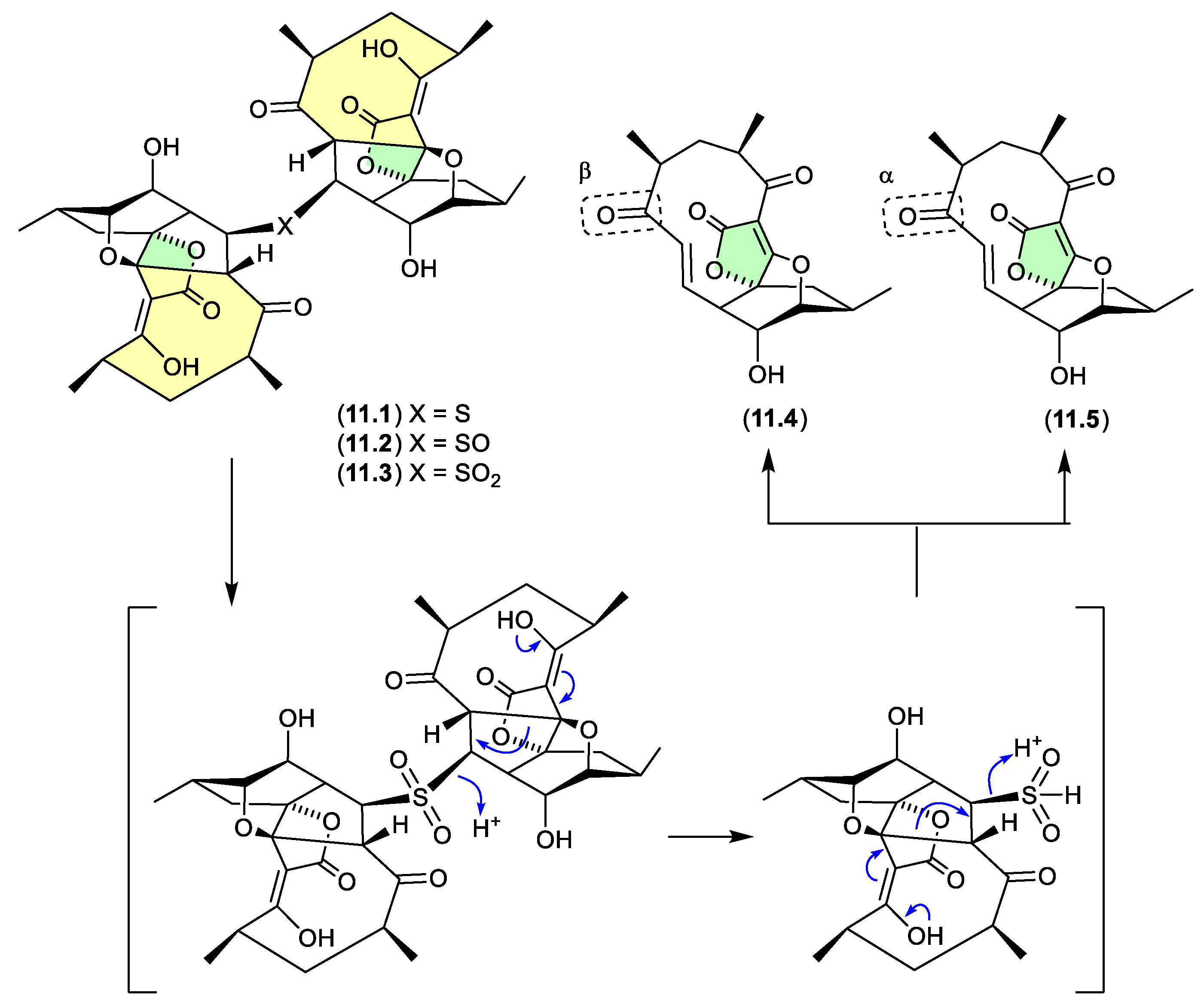
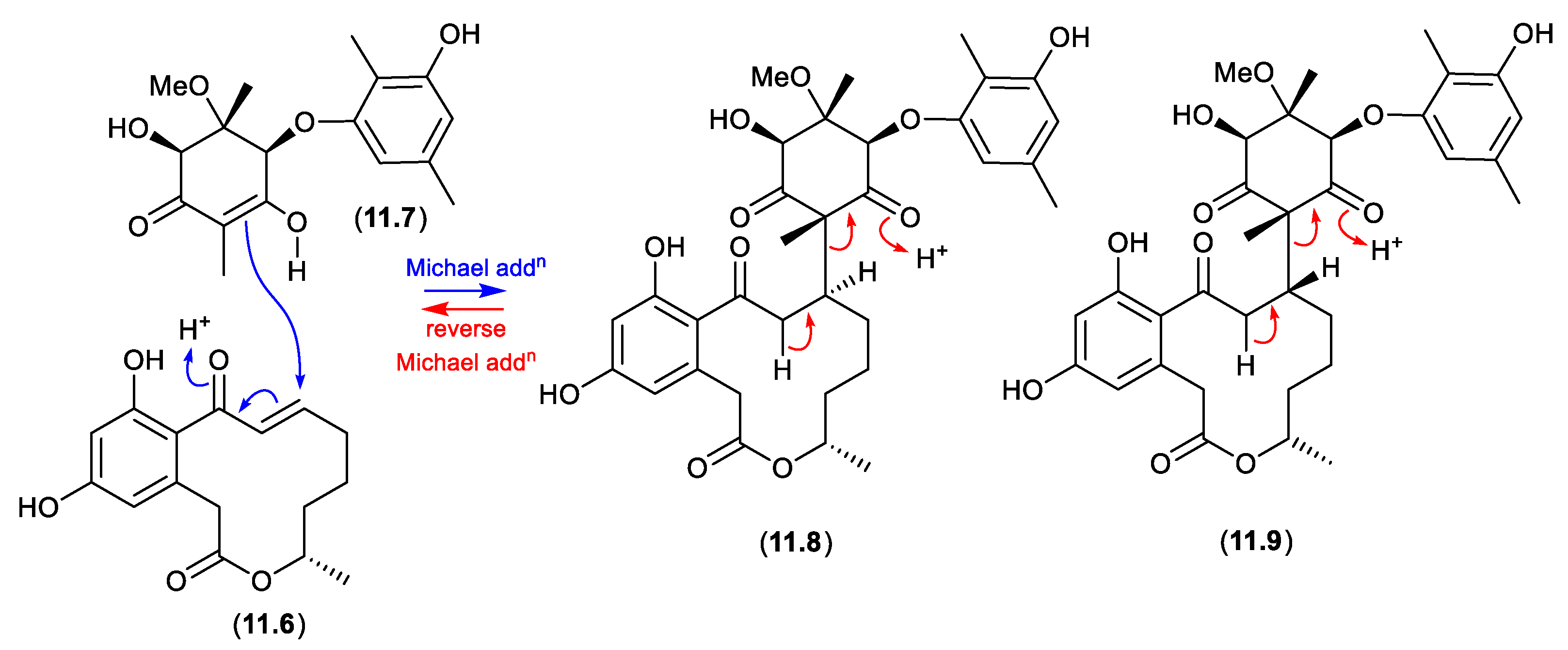
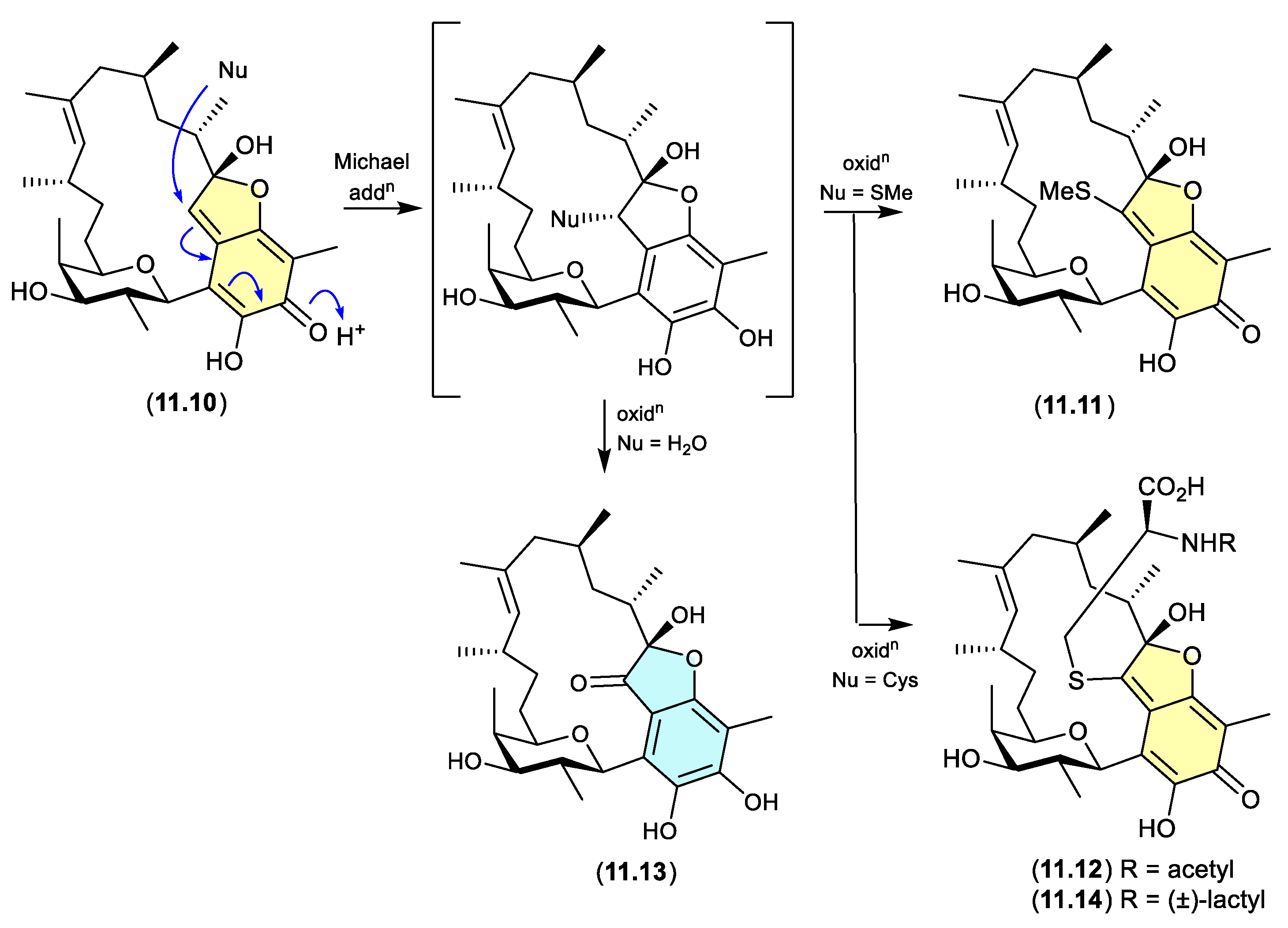
12. Conclusions
12.1. Distinction Between a Natural Product and an Artifact
12.2. How to Recognise the Presence of Artifacts
12.3. How to Control the Formation of Artifacts
12.4. Why and How to Take Advantage of Artifacts
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hanson, J.R. Pseudo-Natural Products, Some Artefacts Formed during the Isolation of Terpenoids. J. Chem. Res. 2017, 41, 497–503. [CrossRef]
- Xu, T.; Chen, W.; Zhou, J.; Dai, J.; Li, Y.; Zhao, Y. Virtual Screening for Reactive Natural Products and Their Probable Artifacts of Solvolysis and Oxidation. Biomolecules 2020, 10, 1486. [CrossRef]
- Tang, Y.; Friesen, J.; Nikolić, D.; Lankin, D.; McAlpine, J.; Chen, S.-N.; Pauli, G. Silica Gel-Mediated Oxidation of Prenyl Motifs Generates Natural Product-Like Artifacts. Planta Med. 2021, 87, 998–1007. [CrossRef]
- Verpoorte, R.; Kim, H.K.; Choi, Y.H. Trivialities in Metabolomics: Artifacts in Extraction and Analysis. Front. Mol Biosci. 2022, 9, 972190. [CrossRef]
- Sauerschnig, C.; Doppler, M.; Bueschl, C.; Schuhmacher, R. Methanol Generates Numerous Artifacts during Sample Extraction and Storage of Extracts in Metabolomics Research. Metabolites 2017, 8, 1. [CrossRef]
- Venditti, A. What Is and What Should Never Be: Artifacts, Improbable Phytochemicals, Contaminants and Natural Products. Nat. Prod. Res. 2020, 34, 1014–1031. [CrossRef]
- Capon, R.J. Extracting Value: Mechanistic Insights into the Formation of Natural Product Artifacts – Case Studies in Marine Natural Products. Nat. Prod. Rep. 2020, 37, 55–79. [CrossRef]
- Woo, E.J.; Starks, C.M.; Carney, J.R.; Arslanian, R.; Cadapan, L.; Zavala, S.; Licari, P. Migrastatin and a New Compound, Isomigrastatin, from Streptomyces platensis. J. Antibiot. 2002, 55, 141–146. [CrossRef]
- Hochlowski, J.E.; Whittern, D.N.; Hill, P.; McAlpine, J.B. Dorrigocins: Novel Antifungal Antibiotics That Change the Morphology of Ras-Transformed NIH/3T3 Cells to That of Normal Cells. II. Isolation and Elucidation of Structures. J. Antibiot. 1994, 47, 870–874. [CrossRef]
- Ju, J.; Lim, S.-K.; Jiang, H.; Shen, B. Migrastatin and Dorrigocins Are Shunt Metabolites of Iso-Migrastatin. J. Am. Chem. Soc. 2005, 127, 1622–1623. [CrossRef]
- Ju, J.; Lim, S.-K.; Jiang, H.; Seo, J.-W.; Shen, B. Iso-Migrastatin Congeners from Streptomyces platensis and Generation of a Glutarimide Polyketide Library Featuring the Dorrigocin, Lactimidomycin, Migrastatin, and NK30424 Scaffolds. J. Am. Chem. Soc. 2005, 127, 11930–11931. [CrossRef]
- Ju, J.; Lim, S.-K.; Jiang, H.; Seo, J.-W.; Her, Y.; Shen, B. Thermolysis of Isomigrastatin and Its Congeners via [3,3]-Sigmatropic Rearrangement: A New Route to the Synthesis of Migrastatin and Its Analogues. Org. Lett. 2006, 8, 5865–5868. [CrossRef]
- Orfanoudaki, M.; Akee, R.K.; Martínez-Fructuoso, L.; Wang, D.; Kelley, J.A.; Smith, E.A.; Henrich, C.J.; Schnermann, M.J.; O’Keefe, B.R.; Grkovic, T. Formation of Trideuteromethylated Artifacts of Pyrrole-Containing Natural Products. J. Nat. Prod. 2024, 87, 415–423. [CrossRef]
- Sala, S.; James, P.J.C.; Nealon, G.L.; Fromont, J.; Gomez, O.; Vuong, D.; Lacey, E.; Flematti, G.R. Dendrillic Acids A and B: Nitrogenous, Rearranged Spongian Nor-Diterpenes from a Dendrilla sp. Marine Sponge. J. Nat. Prod. 2023, 86, 482–489. [CrossRef]
- Yu, G.; Wang, Q.; Liu, S.; Zhang, X.; Che, Q.; Zhang, G.; Zhu, T.; Gu, Q.; Li, D. Methylsulfonylated Polyketides Produced by Neosartorya udagawae HDN13-313 via Exogenous Addition of Small Molecules. J. Nat. Prod. 2019, 82, 998–1001. [CrossRef]
- Shang, Z.; Arishi, A.A.; Wu, C.; Lao, F.; Gilchrist, C.L.M.; Moggach, S.A.; Lacey, E.; Piggott, A.M.; Chooi, Y. Self-Resistance Gene-Guided Discovery of the Molecular Basis for Biosynthesis of the Fatty Acid Synthase Inhibitor Cerulenin. Angew. Chem. 2024, 137, e20241494. [CrossRef]
- Socha, A.M.; Garcia, D.; Sheffer, R.; Rowley, D.C. Antibiotic Bisanthraquinones Produced by a Streptomycete Isolated from a Cyanobacterium Associated with Ecteinascidia turbinata. J. Nat. Prod. 2006, 69, 1070–1073. [CrossRef]
- Samarasekera, K.; Hussein, W.M.; Wu, T.; Salim, A.A.; Capon, R.J. Glyclauxins A-E: Dimeric Oxaphenalenone Aminoglycosides from an Australian Wasp Nest-Derived Fungus Talaromyces sp. CMB-MW102. J. Nat. Prod. 86, 517–525, 2023. [CrossRef]
- Wu, J.; Shui, H.; Zhang, M.; Zeng, Y.; Zheng, M.; Zhu, K.-K.; Wang, S.-B.; Bi, H.; Hong, K.; Cai, Y.-S. Aculeaxanthones A–E, New Xanthones from the Marine-Derived Fungus Aspergillus aculeatinus WHUF0198. Front. Microbiol. 2023, 14, 1138830. [CrossRef]
- Zhen, X.; Gong, T.; Wen, Y.-H.; Yan, D.-J.; Chen, J.-J.; Zhu, P. Chrysoxanthones A–C, Three New Xanthone–Chromanone Heterdimers from Sponge-Associated Penicillium chrysogenum HLS111 Treated with Histone Deacetylase Inhibitor. Mar. Drugs 2018, 16, 357. [CrossRef]
- Wu, G.; Yu, G.; Kurtán, T.; Mándi, A.; Peng, J.; Mo, X.; Liu, M.; Li, H.; Sun, X.; Li, J.; et al. Versixanthones A–F, Cytotoxic Xanthone–Chromanone Dimers from the Marine-Derived Fungus Aspergillus versicolor HDN1009. J. Nat. Prod. 2015, 78, 2691–2698. [CrossRef]
- Lünne, F.; Köhler, J.; Stroh, C.; Müller, L.; Daniliuc, C.G.; Mück-Lichtenfeld, C.; Würthwein, E.-U.; Esselen, M.; Humpf, H.-U.; Kalinina, S.A. Insights into Ergochromes of the Plant Pathogen Claviceps purpurea. J. Nat. Prod. 2021, 84, 2630–2643. [CrossRef]
- Qin, T.; Iwata, T.; Ransom, T.T.; Beutler, J.A.; Porco, J.A. Syntheses of Dimeric Tetrahydroxanthones with Varied Linkages: Investigation of “Shapeshifting” Properties. J. Am. Chem. Soc. 2015, 137, 15225–15233. [CrossRef]
- Parish, C.A.; Smith, S.K.; Calati, K.; Zink, D.; Wilson, K.; Roemer, T.; Jiang, B.; Xu, D.; Bills, G.; Platas, G.; et al. Isolation and Structure Elucidation of Parnafungins, Antifungal Natural Products That Inhibit mRNA Polyadenylation. J. Am. Chem. Soc. 2008, 130, 7060–7066. [CrossRef]
- Guan, J.; Zhang, P.-P.; Wang, X.-H.; Guo, Y.-T.; Zhang, Z.-J.; Li, P.; Lin, L.-P. Structure-Guided Discovery of Diverse Cytotoxic Dimeric Xanthones/Chromanones from Penicillium chrysogenum C-7-2-1 and Their Interconversion Properties. J. Nat. Prod. 2024, 87, 238–257. [CrossRef]
- Ayers, S.; Graf, T.N.; Adcock, A.F.; Kroll, D.J.; Shen, Q.; Swanson, S.M.; Matthew, S.; Blanco, E.J.C. de; Wani, M.C.; Darveaux, B.A.; et al. Cytotoxic Xanthone–Anthraquinone Heterodimers from an Unidentified Fungus of the Order Hypocreales (MSX 17022). J. Antibiot. 2012, 65, 3–8. [CrossRef]
- Khong, Q.T.; Smith, E.A.; Wendt, K.L.; Dalilian, M.; Goncharova, E.I.; Brownell, I.; Cichewicz, R.H.; Henrich, C.J.; Beutler, J.A.; O’Keefe, B.R.; et al. Chemoreactive 2,5-Diketopiperazines from a Penicillium sp., Structure Revision of Reported Analogues and Proposed Facile Transformation Pathways. J. Nat. Prod. 2024, 87, 1826–1837. [CrossRef]
- Lv, H.; Su, H.; Xue, Y.; Jia, J.; Bi, H.; Wang, S.; Zhang, J.; Zhu, M.; Emam, M.; Wang, H.; et al. Polyketides with Potential Bioactivities from the Mangrove-Derived Fungus Talaromyces sp. WHUF0362. Mar. Life Sci. Technol. 2023, 5, 232–241. [CrossRef]
- Bang, S.; Kwon, H.E.; Baek, J.Y.; Jang, D.S.; Kim, S.; Nam, S.-J.; Lee, D.; Kang, K.S.; Shim, S.H. Colletotrichalactones A-Ca, Unusual 5/6/10-Fused Tricyclic Polyketides Produced by an Endophytic Fungus, Colletotrichum sp. JS-0361. Bioorg. Chem. 2020, 105, 104449. [CrossRef]
- Pang, X.; Lin, X.; Wang, J.; Liang, R.; Tian, Y.; Salendra, L.; Luo, X.; Zhou, X.; Yang, B.; Tu, Z.; et al. Three New Highly Oxygenated Sterols and One New Dihydroisocoumarin from the Marine Sponge-Derived Fungus Cladosporium sp. SCSIO41007. Steroids 2018, 129, 41–46. [CrossRef]
- Chen, M.; Han, L.; Shao, C.-L.; She, Z.-G.; Wang, C.-Y. Bioactive Diphenyl Ether Derivatives from a Gorgonian-Derived Fungus Talaromyces sp. Chem. Biodivers. 2015, 12, 443–450. [CrossRef]
- Li, F.; Sun, C.; Che, Q.; Zhu, T.; Gu, Q.; Guan, H.; Zhang, G.; Li, D. Pyrazinopyrimidine Alkaloids from a Mangrove-Derived Fungus Aspergillus versicolor HDN11-84. Phytochemistry 2021, 188, 112817. [CrossRef]
- Wu, C.-Z.; Li, G.; Zhang, Y.-H.; Yuan, S.-Z.; Dong, K.-M.; Lou, H.-X.; Peng, X.-P. Interconvertible Pyridone Alkaloids from the Marine-Derived Fungus Penicillium oxalicum QDU1. J. Nat. Prod. 2023, 86, 739–750. [CrossRef]
- Davidson, B.S.; Molinski, T.F.; Barrows, L.R.; Ireland, C.M. Varacin: A Novel Benzopentathiepin from Lissoclinum vareau That Is Cytotoxic toward a Human Colon Tumor. J. Am. Chem. Soc. 1991, 113, 4709–4710. [CrossRef]
- Makarieva, T.N.; Stonik, V.A.; Dmitrenok, A.S.; Grebnev, B.B.; Isakov, V.V.; Rebachyk, N.M.; Rashkes, Y.W. Varacin and Three New Marine Antimicrobial Polysulfides from the Far-Eastern Ascidian Polycitor sp. J. Nat. Prod. 1995, 58, 254–258. [CrossRef]
- Jiang, G.; Zhang, P.; Ratnayake, R.; Yang, G.; Zhang, Y.; Zuo, R.; Powell, M.; Huguet-Tapia, J.C.; Abboud, K.A.; Dang, L.H.; et al. Fungal Epithiodiketopiperazines Carrying α,β-Polysulfide Bridges from Penicillium steckii YE, and Their Chemical Interconversion. Chembiochem 2020, 22, 416–422. [CrossRef]
- Lindel, T.; Jensen, P.R.; Fenical, W.; Long, B.H.; Casazza, A.M.; Carboni, J.; Fairchild, C.R. Eleutherobin, a New Cytotoxin That Mimics Paclitaxel (Taxol) by Stabilizing Microtubules. J. Am. Chem. Soc. 1997, 119, 8744–8745. [CrossRef]
- Cinel, B.; Roberge, M.; Behrisch, H.; Ofwegen, L. van; Castro, C.B.; Andersen, R.J. Antimitotic Diterpenes from Erythropodium caribaeorum Test Pharmacophore Models for Microtubule Stabilization. Org. Lett. 2000, 2, 257–260. [CrossRef]
- Britton, R.; Roberge, M.; Berisch, H.; Andersen, R.J. Antimitotic Diterpenoids from Erythropodium caribaeorum : Isolation Artifacts and Putative Biosynthetic Intermediates. Tetrahedron Lett. 2001, 42, 2953–2956. [CrossRef]
- Ortiz-López, F.J.; Oves-Costales, D.; Garzón, J.F.G.; Gren, T.; Sterndorff, E.B.; Jiang, X.; Jo̷rgensen, T.S.; Blin, K.; Fernández-Pastor, I.; Tormo, J.R.; et al. Genome-Led Discovery of the Antibacterial Cyclic Lipopeptide Kutzneridine A and Its Silent Biosynthetic Gene Cluster from Kutzneria Species. J. Nat. Prod. 2024, 87, 2515–2522. [CrossRef]
- Koshino, S.; Koshino, H.; Matsuura, N.; Kobinata, K.; Onose, R.; Isono, K.; Osada, H. A New Cyclic Lipopeptide Antibiotic, Enamidonin. J. Antibiot. 1995, 48, 185–187. [CrossRef]
- Son, S.; Ko, S.-K.; Kim, S.M.; Kim, E.; Kim, G.S.; Lee, B.; Ryoo, I.-J.; Kim, W.-G.; Lee, J.-S.; Hong, Y.-S.; et al. Antibacterial Cyclic Lipopeptide Enamidonins with an Enamide-Linked Acyl Chain from a Streptomyces Species. J. Nat. Prod. 2018, 81, 2462–2469. [CrossRef]
- Namatame, I.; Tomoda, H.; Matsuda, D.; Tabata, N.; Kobayashi, S.; Omura, S. K97-0239A and B, New Inhibitors of Macrophage Foam Cell Formation, Produced by Streptomyces sp. K97-0239. Proc. Jpn. Acad., Ser. B 2002, 78, 45–50. [CrossRef]
- Zhang, H.; Zhang, C.; Li, Q.; Ma, J.; Ju, J. Metabolic Blockade-Based Genome Mining Reveals Lipochain-Linked Dihydro-β-Alanine Synthetases Involved in Autucedine Biosynthesis. Org. Lett. 2022, 24, 5535–5540. [CrossRef]
- Yan, J.-X.; Chevrette, M.G.; Braun, D.R.; Harper, M.K.; Currie, C.R.; Bugni, T.S. Madurastatin D1 and D2, Oxazoline Containing Siderophores Isolated from an Actinomadura sp. Org. Lett. 2019, 21, 6275–6279. [CrossRef]
- Pérez-Bonilla, M.; Sánchez-Hidalgo, M.; González, I.; Oves-Costales, D.; Martín, J.; Murillo-Alba, J.; Tormo, J.R.; Cho, A.; Byun, S.-Y.; No, J.-H.; et al. Madurastatins with Imidazolidinone Rings: Natural Products or Side-Reaction Products from Extraction Solvents? Int. J. Mol. Sci. 2023, 25, 301. [CrossRef]
- Oleinikova, G.K.; Zhuravleva, O.I.; Berdyshev, D.V.; Menzorova, N.I.; Popov, R.S.; Denisenko, V.A.; Kirichuk, N.N.; Afiyatullov, S.S. New Dihydrobenzofuranoid from the Marine-Derived Fungus Aspergillus ustus KMM 4664. Nat. Prod. Res. 2019, 35, 3332–3335. [CrossRef]
- Aguilar-Ramírez, E.; Rivera-Chávez, J.; Alvarado-Zacarías, B.D.; Barquera-Lozada, J.E. Exploring the Nonenzymatic Origin of Duclauxin-like Natural Products. J. Nat. Prod. 2024, 87, 2230–2242. [CrossRef]
- Wang, M.; Yang, L.; Feng, L.; Hu, F.; Zhang, F.; Ren, J.; Qiu, Y.; Wang, Z. Verruculosins A–B, New Oligophenalenone Dimers from the Soft Coral-Derived Fungus Talaromyces verruculosus. Mar. Drugs 2019, 17, 516. [CrossRef]
- Lacey, A.E.; Minns, S.A.; Chen, R.; Vuong, D.; Lacey, E.; Kalaitzis, J.A.; Tan, Y.P.; Shivas, R.G.; Butler, M.S.; Piggott, A.M. Talcarpones A and B: Bisnaphthazarin-Derived Metabolites from the Australian Fungus Talaromyces johnpittii sp. Nov. MST-FP2594. J. Antibiot. 2023, 77, 147–155. [CrossRef]
- El-Elimat, T.; Raja, H.A.; Day, C.S.; Chen, W.-L.; Swanson, S.M.; Oberlies, N.H. Greensporones: Resorcylic Acid Lactones from an Aquatic Halenospora sp. J. Nat. Prod. 2014, 77, 2088–2098. [CrossRef]
- Shi, Y.; Zaleta-Pinet, D.A.; Clark, B.R. Isolation, Identification, and Decomposition of Antibacterial Dialkylresorcinols from a Chinese Pseudomonas aurantiaca Strain. J. Nat. Prod. 2020, 83, 194–201. [CrossRef]
- Wibowo, M.; Gotfredsen, C.H.; Sassetti, E.; Melchiorsen, J.; Clausen, M.H.; Gram, L.; Ding, L. Azodyrecins A–C: Azoxides from a Soil-Derived Streptomyces Species. J. Nat. Prod. 2020, 83, 3519–3525. [CrossRef]
- Su, X.; Zhang, L.; Hu, K.; An, Y.; Zhang, Q.; Tang, J.; Yan, B.; Li, X.; Cai, J.; Li, X.; et al. Discovery of Natural Potent HMG-CoA Reductase Degraders for Lowering Cholesterol. Angew. Chem. Int. Ed. 2024, 63, e202313859. [CrossRef]
- Xu, M.; Gessner, G.; Groth, I.; Lange, C.; Christner, A.; Bruhn, T.; Deng, Z.; Li, X.; Heinemann, S.H.; Grabley, S.; et al. Shearinines D–K, New Indole Triterpenoids from an Endophytic Penicillium sp. (Strain HKI0459) with Blocking Activity on Large-Conductance Calcium-Activated Potassium Channels. Tetrahedron 2007, 63, 435–444. [CrossRef]
- Chen, M.; Hao, B.-C.; Zhu, X.-H.; Zhang, L.-K.; Zheng, Y.-Y.; Zhou, X.-J.; Schäberle, T.F.; Shen, L.; Wang, C.-Y.; Liu, Y. Molecular Networking Reveals Indole Diterpenoids from the Marine-Derived Fungus Penicillium sp. N4-3. Mar. Life Sci. Technol. 2025, 7, 302–312. [CrossRef]
- Tuzi, A.; Carbone, M.; Ciavatta, M.L.; Evidente, A. Structure Revision of the Fungal Phytotoxin Cavoxin and of Its Corresponding Chroman-4-One Cavoxone by X-ray Crystallography. J. Nat. Prod. 2024, 87, 1888–1892. [CrossRef]
- Fotso, S.; Wu, S.J.; Qin, S.; Laatsch, H. 5,7-Dihydroxy-5,6,7,8-Tetrahydro-1H-Azocin-2-One from a Marine-Derived Streptomyces sp. Nat. Prod. Commun. 2006, 1, 1934578X0600100102. [CrossRef]
- Miguel-Gordo, M.; Gegunde, S.; Calabro, K.; Jennings, L.K.; Alfonso, A.; Genta-Jouve, G.; Vacelet, J.; Botana, L.M.; Thomas, O.P. Bromotryptamine and Bromotyramine Derivatives from the Tropical Southwestern Pacific Sponge Narrabeena nigra. Mar. Drugs 2019, 17, 319. [CrossRef]
- Shin, A.-Y.; Lee, H.-S.; Lee, Y.-J.; Lee, J.S.; Son, A.; Choi, C.; Lee, J. Oxygenated Theonellastrols: Interpretation of Unusual Chemical Behaviors Using Quantum Mechanical Calculations and Stereochemical Reassignment of 7α-Hydroxytheonellasterol. Mar. Drugs 2020, 18, 607. [CrossRef]
- Bringmann, G.; Lang, G.; Gulder, T.A.M.; Tsuruta, H.; Mühlbacher, J.; Maksimenka, K.; Steffens, S.; Schaumann, K.; Stöhr, R.; Wiese, J.; et al. The First Sorbicillinoid Alkaloids, the Antileukemic Sorbicillactones A and B, from a Sponge-Derived Penicillium chrysogenum Strain. Tetrahedron 2005, 61, 7252–7265. [CrossRef]
- Abe, N.; Sugimoto, O.; Tanji, K.; Hirota, A. Identification of the Quinol Metabolite “Sorbicillinol”, a Key Intermediate Postulated in Bisorbicillinoid Biosynthesis. J. Am. Chem. Soc. 2000, 122, 12606–12607. [CrossRef]
- Elbanna, A.H.; Khalil, Z.G.; Capon, R.J. Oxandrastins: Antibacterial Meroterpenes from an Australian Mud Dauber Wasp Nest-Associated Fungus, Penicillium sp. CMB-MD14. Molecules 2021, 26, 7144. [CrossRef]
- Igarashi, Y.; Zhou, T.; Sato, S.; Matsumoto, T.; Yu, L.; Oku, N. Akaeolide, a Carbocyclic Polyketide from Marine-Derived Streptomyces. Org. Lett. 2013, 15, 5678–5681. [CrossRef]
- Zhang, S.; Yang, Q.; Guo, L.; Zhang, Y.; Feng, L.; Zhou, L.; Yang, S.; Yao, Q.; Pescitelli, G.; Xie, Z. Isolation, Structure Elucidation and Racemization of (+)- and (-)-Pratensilins A-C: Unprecedented Spiro Indolinone-Naphthofuran Alkaloids from a Marine Streptomyces sp. Chem. Comm. 2017, 53, 10066–10069. [CrossRef]
- Zhang, S.; Zhang, L.; Kou, L.-J.; Yang, Q.-L.; Qu, B.; Pescitelli, G.; Xie, Z. Isolation, Stereochemical Study, and Racemization of (±)-Pratenone A, the First Naturally Occurring 3-(1-Naphthyl)-2-Benzofuran-1(3H)-One Polyketide from a Marine-Derived Actinobacterium. Chirality 2020, 32, 299–307. [CrossRef]
- Arabshahi, L.; Schmitz, F.J. Brominated Tyrosine Metabolites from an Unidentified Sponge. J. Org. Chem. 1987, 52, 3584–3586. [CrossRef]
- Calcul, L.; Inman, W.D.; Morris, A.A.; Tenney, K.; Ratnam, J.; McKerrow, J.H.; Valeriote, F.A.; Crews, P. Additional Insights on the Bastadins: Isolation of Analogues from the Sponge Ianthella cf. reticulata and Exploration of the Oxime Configurations. J. Nat. Prod. 2010, 73, 365–372. [CrossRef]
- Birnbacher, J.; Schüffler, A.; Deininger, F.; Opatz, T.; Anke, T. Isolation and Biological Activity of New Norhirsutanes from Creolophus cirrhatus. Z. für Naturforsch. C 2008, 63, 203–206. [CrossRef]
- Elbanna, A.H.; Khalil, Z.G.; Bernhardt, P.V.; Capon, R.J. Neobulgarones Revisited: Anti and Syn Bianthrones from an Australian Mud Dauber Wasp Nest-Associated Fungus, Penicillium sp. CMB-MD22. J. Nat. Prod. 2021, 84, 762–770. [CrossRef]
- Cueto, M.; Jensen, P.R.; Kauffman, C.; Fenical, W.; Lobkovsky, E.; Clardy, J. Pestalone, a New Antibiotic Produced by a Marine Fungus in Response to Bacterial Challenge. J. Nat. Prod. 2001, 64, 1444–1446. [CrossRef]
- Li, E.; Jiang, L.; Guo, L.; Zhang, H.; Che, Y. Pestalachlorides A-C, Antifungal Metabolites from the Plant Endophytic Fungus Pestalotiopsis adusta. Bioorg. Med. Chem. 2008, 16, 7894–7899. [CrossRef]
- Slavov, N.; Cvengroš, J.; Neudörfl, J.; Schmalz, H. Total Synthesis of the Marine Antibiotic Pestalone and Its Surprisingly Facile Conversion into Pestalalactone and Pestalachloride A. Angew. Chem. Int. Ed. 2010, 49, 7588–7591. [CrossRef]
- Zheng, L.; Jiang, X.; Zhang, Q.; Zhu, Y.; Zhang, H.; Zhang, W.; Saurav, K.; Liu, J.; Zhang, C. Discovery and Biosynthesis of Neoenterocins Indicate a Skeleton Rearrangement of Enterocin. Org. Lett. 2019, 21, 9066–9070. [CrossRef]
- Xu, L.; Guo, F.-W.; Zhang, X.-Q.; Zhou, T.-Y.; Wang, C.-J.; Wei, M.-Y.; Gu, Y.-C.; Wang, C.-Y.; Shao, C.-L. Discovery, Total Syntheses and Potent Anti-Inflammatory Activity of Pyrrolinone-Fused Benzoazepine Alkaloids Asperazepanones A and B from Aspergillus candidus. Commun. Chem. 2022, 5, 80. [CrossRef]
- Xu, W.-F.; Chao, R.; Hai, Y.; Guo, Y.-Y.; Wei, M.-Y.; Wang, C.-Y.; Shao, C.-L. 17-Hydroxybrevianamide N and Its N1-Methyl Derivative, Quinazolinones from a Soft-Coral-Derived Aspergillus sp. Fungus: 13 S Enantiomers as the True Natural Products. J. Nat. Prod. 2021, 84, 1353–1358. [CrossRef]
- Williams, P.G.; Buchanan, G.O.; Feling, R.H.; Kauffman, C.A.; Jensen, P.R.; Fenical, W. New Cytotoxic Salinosporamides from the Marine Actinomycete Salinispora tropica. J. Org. Chem. 2005, 70, 6196–6203. [CrossRef]
- Kang, H.; Jensen, P.R.; Fenical, W. Isolation of Microbial Antibiotics from a Marine Ascidian of the Genus Didemnum. J. Org. Chem. 1996, 61, 1543–1546. [CrossRef]
- Liu, H.; Chen, Z.; Zhu, G.; Wang, L.; Du, Y.; Wang, Y.; Zhu, W. Phenolic Polyketides from the Marine Alga-Derived Streptomyces sp. OUCMDZ-3434. Tetrahedron 2017, 73, 5451–5455. [CrossRef]
- Xu, D.-B.; Ma, M.; Deng, Z.-X.; Hong, K. Genotype-Driven Isolation of Enterocin with Novel Bioactivities from Mangrove-Derived Streptomyces qinglanensis 172205. Appl. Microbiol. Biotechnol. 2015, 99, 5825–5832. [CrossRef]
- Salim, A.A.; Samarasekera, K.; Khalil, Z.G.; Capon, R.J. Exploring Natural Product Artifacts: The Polyketide Enterocin Warms to a Ballet of Isomers. Org. Lett. 2020, 22, 4828–4832. [CrossRef]
- Schneider, Y.; Jenssen, M.; Isaksson, J.; Hansen, K.Ø.; Andersen, J.H.; Hansen, E.H. Bioactivity of Serratiochelin A, a Siderophore Isolated from a Co-Culture of Serratia sp. and Shewanella sp. Microorganisms 2020, 8, 1042. [CrossRef]
- Zhang, H.; Conte, M.M.; Capon, R.J. Franklinolides A-C from an Australian Marine Sponge Complex: Phosphodiesters Strongly Enhance Polyketide Cytotoxicity. Angew. Chem. Int. Ed. 2010, 49, 9904–9906. [CrossRef]
- Sirirath, S.; Tanaka, J.; Ohtani, I.I.; Ichiba, T.; Rachmat, R.; Ueda, K.; Usui, T.; Osada, H.; Higa, T. Bitungolides A-F, New Polyketides from the Indonesian Sponge Theonella cf. swinhoei. J. Nat. Prod. 2002, 65, 1820–1823. [CrossRef]
- Salim, A.A.; Xiao, X.; Cho, K.-J.; Piggott, A.M.; Lacey, E.; Hancock, J.F.; Capon, R.J. Rare Streptomyces sp. Polyketides as Modulators of K-Ras Localisation. Org. Biomol. Chem. 2014, 12, 4872–4878. [CrossRef]
- Zhong, W.-M.; Wang, J.-F.; Wei, X.-Y.; Zeng, Q.; Chen, X.-Y.; Xiang, Y.; Tian, X.-P.; Zhang, S.; Long, L.J.; Wang, F.-Z. (+)- and (−)-Eurotone A: A Pair of Enantiomeric Polyketide Dimers from a Marine-Derived Fungus Eurotium sp. SCSIO F452. Tetrahedron Lett. 2019, 60, 1600–1603. [CrossRef]
- Bindseil, K.U.; Henkel, T.; Zeeck, A.; Bur, D.; Niederer, D.; Séquin, U. Metabolic Products of Microorganisms. Part 262. The Absolute Configuration of Sphydrofuran, a Widespread Metabolite from Streptomycetes. Helv. Chim. Acta 1991, 74, 1281–1286. [CrossRef]
- Shibata, S.; Ogihara, Y.; Tokutake, N.; Tanaka, O. Duclauxin, a Metabolite of Penicillium duclauxi (Delacroix). Tetrahedron Lett. 1965, 6, 1287–1288. [CrossRef]
- Zheng, M.; Li, Q.; Liao, H.; Li, Y.; Zhou, C.; Zhao, X.; Chen, C.; Sun, W.; Zhang, Y.; Zhu, H. Adpressins A-G: Oligophenalenone Dimers from Talaromyces adpressus. J. Nat. Prod. 2024, 87, 1921–1929. [CrossRef]
- Zang, Y.; Genta-Jouve, G.; Retailleau, P.; Escargueil, A.; Mann, S.; Nay, B.; Prado, S. Talaroketals A and B, Unusual Bis(Oxaphenalenone) Spiro and Fused Ketals from the Soil Fungus Talaromyces stipitatus ATCC 10500. Org. Biomol. Chem. 2016, 14, 2691–2697. [CrossRef]
- Li, Q.; Zhang, M.; Zhang, X.; Li, L.; Zheng, M.; Kang, J.; Liu, F.; Zhou, Q.; Li, X.; Sun, W.; et al. Talaroclauxins A and B: Duclauxin-Ergosterol and Duclauxin-Polyketide Hybrid Metabolites with Complicated Skeletons from Talaromyces stipitatus. Chin. Chem. Lett. 2024, 35, 108193. [CrossRef]
- Chaiyosang, B.; Kanokmedhakul, K.; Sanmanoch, W.; Boonlue, S.; Hadsadee, S.; Jungsuttiwong, S.; Kanokmedhakul, S. Bioactive Oxaphenalenone Dimers from the Fungus Talaromyces macrosporus KKU-1NK8. Fitoterapia 2019, 134, 429–434. [CrossRef]
- Dethoup, T.; Manoch, L.; Kijjoa, A.; Nascimento, M.S.; Puaparoj, P.; Silva, A.M.; Eaton, G.; Herz, W. Bacillisporins D and E, New Oxyphenalenone Dimers from Talaromyces bacillisporus. Planta Med. 2006, 72, 957–960. [CrossRef]
- Chaudhary, N.K.; Crombie, A.; Vuong, D.; Lacey, E.; Piggott, A.M.; Karuso, P. Talauxins: Hybrid Phenalenone Dimers from Talaromyces stipitatus. J. Nat. Prod. 2020, 83, 1051–1060. [CrossRef]
- Dramae, A.; Intaraudom, C.; Bunbamrung, N.; Saortep, W.; Srichomthong, K.; Pittayakhajonwut, P. Heptacyclic Oligophenalenones from the Soil Fungus Talaromyces bacillisporus BCC17645. Tetrahedron 2020, 76, 130980. [CrossRef]
- Cao, P.; Yang, J.; Miao, C.-P.; Yan, Y.; Ma, Y.-T.; Li, X.-N.; Zhao, L.-X.; Huang, S.-X. New Duclauxamide from Penicillium manginii YIM PH30375 and Structure Revision of the Duclauxin Family. Org. Lett. 2015, 17, 1146–1149. [CrossRef]
- Isaka, M.; Sappan, M.; Auncharoen, P.; Srikitikulchai, P. Chromone Derivatives from the Wood-Decay Fungus Rhizina sp. BCC 12292. Phytochem. Lett. 2010, 3, 152–155. [CrossRef]
- Liu, Y.; Stuhldreier, F.; Kurtán, T.; Mándi, A.; Arumugam, S.; Lin, W.; Stork, B.; Wesselborg, S.; Weber, H.; Henrich, B.; et al. Daldinone Derivatives from the Mangrove-Derived Endophytic Fungus Annulohypoxylon sp. RSC Adv. 2017, 7, 5381–5393. [CrossRef]
- Jiang, L.; Huang, P.; Ren, B.; Song, Z.; Zhu, G.; He, W.; Zhang, J.; Oyeleye, A.; Dai, H.; Zhang, L.; et al. Antibacterial Polyene-Polyol Macrolides and Cyclic Peptides from the Marine-Derived Streptomyces sp. MS110128. Appl. Microbiol. Biotechnol. 2021, 105, 4975–4986. [CrossRef]
- Kim, M.C.; Cullum, R.; Machado, H.; Smith, A.J.; Yang, I.; Rodvold, J.J.; Fenical, W. Photopiperazines A-D, Photosensitive Interconverting Diketopiperazines with Significant and Selective Activity against U87 Glioblastoma Cells, from a Rare, Marine-Derived Actinomycete of the Family Streptomycetaceae. J. Nat. Prod. 2019, 82, 2262–2267. [CrossRef]
- Zheng, J.; Xu, Z.; Wang, Y.; Hong, K.; Liu, P.; Zhu, W. Cyclic Tripeptides from the Halotolerant Fungus Aspergillus sclerotiorum PT06-1. J. Nat. Prod. 2010, 73, 1133–1137. [CrossRef]
- Fu, X.; Schmitz, F.J.; Kelly-Borges, M.; McCready, T.L.; Holmes, C.F.B. Clavosines A−C from the Marine Sponge Myriastra clavosa: Potent Cytotoxins and Inhibitors of Protein Phosphatases 1 and 2A. J. Org. Chem. 1998, 63, 7957–7963. [CrossRef]
- Fusetani, N.; Matsunaga, S. Bioactive Sponge Peptides. Chem. Rev. 1993, 93, 1793–1806. [CrossRef]
- Kurnianda, V.; Fujimura, H.; Kanna, Y.; Tanaka, J. Photooxidation Products from a Marine Cadinane Sesquiterpenoid. Chem. Lett. 2021, 50, 220–222. [CrossRef]
- Waksman, S.A.; Bugie, E. Chaetomin, a New Antibiotic Substance Produced by Chaetomium cochliodes. J. Bacteriol. 1944, 48, 527–530. [CrossRef]
- Wang, M.-H.; Hu, Y.-C.; Sun, B.-D.; Yu, M.; Niu, S.-B.; Guo, Z.; Zhang, X.-Y.; Zhang, T.; Ding, G.; Zou, Z.-M. Highly Photosensitive Poly-Sulfur-Bridged Chetomin Analogues from Chaetomium cochliodes. Org. Lett. 2018, 20, 1806–1809. [CrossRef]
- Shah, M.; Sun, C.; Sun, Z.; Zhang, G.; Che, Q.; Gu, Q.; Zhu, T.; Li, D. Antibacterial Polyketides from Antarctica Sponge-Derived Fungus Penicillium sp. HDN151272. Mar. Drugs 2020, 18, 71. [CrossRef]
- Kong, F.; Singh, M.P.; Carter, G.T. Pseudopyronines A and B, α-Pyrones Produced by a Marine Pseudomonas sp. F92S91, and Evidence for the Conversion of 4-Hydroxy-α-Pyrone to 3-Furanone. J. Nat. Prod. 2005, 68, 920–923. [CrossRef]
- Capon, R.J.; Faulkner, D.J. Metabolites of the Pulmonate Siphonaria lessoni. J. Org. Chem. 1984, 49, 2506–2508. [CrossRef]
- Akiyama, H.; Indananda, C.; Thamchaipenet, A.; Motojima, A.; Oikawa, T.; Komaki, H.; Hosoyama, A.; Kimura, A.; Oku, N.; Igarashi, Y. Linfuranones B and C, Furanone-Containing Polyketides from a Plant-Associated Sphaerimonospora mesophila. J. Nat. Prod. 2018, 81, 1561–1569. [CrossRef]
- Indananda, C.; Igarashi, Y.; Ikeda, M.; Oikawa, T.; Thamchaipenet, A. Linfuranone A, a New Polyketide from Plant-Derived Microbispora sp. GMKU 363. J. Antibiot. 2013, 66, 675–677. [CrossRef]
- Okanya, P.W.; Mohr, K.I.; Gerth, K.; Kessler, W.; Jansen, R.; Stadler, M.; Müller, R. Hyafurones, Hyapyrrolines, and Hyapyrones: Polyketides from Hyalangium minutum. J. Nat. Prod. 2014, 77, 1420–1429. [CrossRef]
- Frank, B.; Wenzel, S.C.; Bode, H.B.; Scharfe, M.; Blöcker, H.; Müller, R. From Genetic Diversity to Metabolic Unity: Studies on the Biosynthesis of Aurafurones and Aurafuron-like Structures in Myxobacteria and Streptomycetes. J. Mol. Biol. 2007, 374, 24–38. [CrossRef]
- Gliński, J.A.; Proudfoot, J.; Madura, I.; Zhang, H.; Gleńsk, M.; Day, V.; Dudek, M.K. Spontaneous Stereoselective Oxidation of Crystalline Avermectin B1a to Its C-8a-(S)-Hydroperoxide. J. Nat. Prod. 2019, 82, 3477–3481. [CrossRef]
- Meyer, S.W.; Mordhorst, T.F.; Lee, C.; Jensen, P.R.; Fenical, W.; Köck, M. Penilumamide, a Novel Lumazine Peptide Isolated from the Marine-Derived Fungus, Penicillium sp. CNL-338. Org. Biomol. Chem. 2010, 8, 2158–2163. [CrossRef]
- Zheng, C.; Wu, L.; Li, X.; Song, X.; Niu, Z.; Song, X.; Chen, G.; Wang, C. Structure and Absolute Configuration of Aspergilumamide A, a Novel Lumazine Peptide from the Mangrove-Derived Fungus Aspergillus sp. Helv. Chim. Acta 2015, 98, 368–373. [CrossRef]
- Chen, M.; Shao, C.-L.; Fu, X.-M.; Kong, C.-J.; She, Z.-G.; Wang, C.-Y. Lumazine Peptides Penilumamides B-D and the Cyclic Pentapeptide Asperpeptide A from a Gorgonian-Derived Aspergillus sp. Fungus. J. Nat. Prod. 2014, 77, 1601–1606. [CrossRef]
- Elsbaey, M.; Jomori, T.; Tanaka, J.; Oku, N.; Igarashi, Y. Okichromanone, a New Antiviral Chromanone from a Marine-Derived Microbispora. J. Antibiot. 2024, 77, 389–392. [CrossRef]
- Li, H.; Guo, J.; Zhang, R.; Wang, J.; Hu, Z.; Zhang, Y. Two New Nucleoside Derivatives Isolated from the Marine-Derived Aspergillus versicolor and Their Intramolecular Transesterification. Nat. Prod. Res. 2022, 36, 3346–3352. [CrossRef]
- Wu, T.; Salim, A.A.; Khalil, Z.G.; Bernhardt, P.V.; Capon, R.J. Glenthmycins A-M: Macrocyclic Spirotetronate Polyketide Antibacterials from the Australian Pasture Plant-Derived Streptomyces sp. CMB-PB041. J. Nat. Prod. 2022, 85, 1641–1657. [CrossRef]
- Wu, T.; Salim, A.A.; Bernhardt, P.V.; Capon, R.J. Amaurones A–K: Polyketides from the Fish Gut-Derived Fungus Amauroascus sp. CMB-F713. J. Nat. Prod. 2021, 84, 474–482. [CrossRef]
- Capon, R.J.; Ratnayake, R.; Stewart, M.; Lacey, E.; Tennant, S.; Gill, J.H. Aspergillazines A-E: Novel Heterocyclic Dipeptides from an Australian Strain of Aspergillus unilateralis. Org. Biomol. Chem. 2005, 3, 123–129. [CrossRef]
- Takahashi, S.; Kakinuma, N.; Iwai, H.; Yanagisawa, T.; Nagai, K.; Suzuki, K.; Tokunaga, T.; Nakagawa, A. Quinolactacins A, B and C. Novel Quinolone Compounds from Penicillium sp. EPF-6. II. Physico-Chemical Properties and Structure Elucidation. J. Antibiot. 2000, 53, 1252–1256. [CrossRef]
- Kim, W.-G.; Song, N.-K.; Yoo, I.-D. Quinolactacins Al and A2, New Acetylcholinesterase Inhibitors from Penicillium citrinum. J. Antibiot. 2001, 54, 831–835. [CrossRef]
- Tatsuta, K.; Misawa, H.; Chikauchi, K. Biomimetric Total Synthesis of Quinolactacin B, TNF Production Inhibitor, and Its Analogs. J. Antibiot. 2001, 54, 109–112. [CrossRef]
- Zhang, X.; Jiang, W.; Sui, Z. Concise Enantioselective Syntheses of Quinolactacins A and B through Alternative Winterfeldt Oxidation. J. Org. Chem. 2003, 68, 4523–4526. [CrossRef]
- Clark, B.; Capon, R.J.; Lacey, E.; Tennant, S.; Gill, J.H. Quinolactacins Revisited: From Lactams to Imide and Beyond. Org. Biomol. Chem. 2006, 4, 1512–1519. [CrossRef]
- Guo, T.-T.; Song, M.-M.; Han, W.-R.; Zhu, J.-H.; Liu, Q.-C.; Wang, J.-F. New N-Methyl-4-Quinolone Alkaloid and Citrinin Dimer Derivatives from the Sponge-Derived Fungus Penicillium sp. SCSIO 41303. Phytochem. Lett. 2021, 46, 29–35. [CrossRef]
- Zhu, J.X.; Lu, Y.; Chen, J.; Chen, J.; Zhang, H.; Bao, X.; Ye, X.; Wang, H. Total Synthesis of Quinolactacin-H from Marine-Derived: Penicillium sp. ENP701 and Biological Activities. RSC Advances 2020, 10, 24251–24254. [CrossRef]
- Liu, Y.; Xue, X.; Zhou, L.; Yang, W.; She, Z.; Liao, Q.; Feng, Y.; Chen, X.; Zhang, Y. Quinolinones Alkaloids with AChE Inhibitory Activity from Mangrove Endophytic Fungus Penicillium citrinum YX-002. Chem. Biodivers. 2023, 20, e202300735. [CrossRef]
- Mohamed, O.G.; Mohamed, O.G.; Khalil, Z.G.; Capon, R.J. Prolinimines: N-Amino-L-Pro-Methyl Ester (Hydrazine) Schiff Bases from a Fish Gastrointestinal Tract-Derived Fungus, Trichoderma sp. CMB-F563. Org. Lett. 2018, 20, 377–380. [CrossRef]
- Mohamed, O.G.; Khalil, Z.G.; Capon, R.J. N-Amino-L-Proline Methyl Ester from an Australian Fish Gut-Derived Fungus: Challenging the Distinction between Natural Product and Artifact. Mar. Drugs 2021, 19, 151. [CrossRef]
- Li, C.-S.; Li, X.-M.; Gao, S.-S.; Lu, Y.H.; Wang, B.-G. Cytotoxic Anthranilic Acid Derivatives from Deep Sea Sediment-Derived Fungus Penicillium paneum SD-44. Mar. Drugs 2013, 11, 3068–3076. [CrossRef]
- Khalil, Z.G.; Kankanamge, S.; Capon, R.J. Structure Revision of Penipacids A–E Reveals a Putative New Cryptic Natural Product, N-Aminoanthranilic Acid, with Potential as a Transcriptional Regulator of Silent Secondary Metabolism. Mar. Drugs 2022, 20, 339. [CrossRef]
- Minato, S. Isolation of Anthglutin, an Inhibitor of γ-Glutamyl Transpeptidase from Penicillum oxalicum. Arch. Biochem. Biophys. 1979, 192, 235–240. [CrossRef]
- Ma, C.; Li, Y.; Niu, S.; Zhang, H.; Liu, X.; Che, Y. N-Hydroxypyridones, Phenylhydrazones, and a Quinazolinone from Isaria farinosa. J. Nat. Prod. 2011, 74, 32–37. [CrossRef]
- Abdelfattah, M.S.; Toume, K.; Arai, M.A.; Masu, H.; Ishibashi, M. Katorazone, a New Yellow Pigment with a 2-Azaquinone-Phenylhydrazone Structure Produced by Streptomyces sp. IFM 11299. Tetrahedron Lett. 2012, 53, 3346–3348. [CrossRef]
- Zhang, T.; Zhu, M.-L.; Sun, G.-Y.; Li, N.; Gu, Q.-Q.; Li, D.-H.; Che, Q.; Zhu, T.-J. Exopisiod B and Farylhydrazone C, Two New Alkaloids from the Antarctic-Derived Fungus Penicillium sp. HDN14-431. J. Asian Nat. Prod. Res. 2016, 18, 959–965. [CrossRef]
- Cheng, P.; Xu, K.; Chen, Y.C.; Wang, T.T.; Chen, Y.; Yang, C.L.; Ma, S.Y.; Liang, Y.; Ge, H.M.; Jiao, R.H. Cytotoxic Aromatic Polyketides from an Insect Derived Streptomyces sp. NA4286. Tetrahedron Lett. 2019, 60, 1706–1709. [CrossRef]
- Steinmetz, H.; Gerth, K.; Jansen, R.; Schläger, N.; Dehn, R.; Reinecke, S.; Kirschning, A.; Müller, R. Elansolid A, a Unique Macrolide Antibiotic from Chitinophaga sancti Isolated as Two Stable Atropisomers. Angew. Chem. Int. Ed. 2011, 50, 532–536. [CrossRef]
- Jansen, R.; Gerth, K.; Steinmetz, H.; Reinecke, S.; Kessler, W.; Kirschning, A.; Müller, R. Elansolid A3, a Unique p-Quinone Methide Antibiotic from Chitinophaga sancti. Chem. A Eur. J. 2011, 17, 7739–7744. [CrossRef]
- Teta, R.; Gurgui, M.; Helfrich, E.J.N.; Künne, S.; Schneider, A.; Echten-Deckert, G.V.; Mangoni, A.; Piel, J. Genome Mining Reveals trans-AT Polyketide Synthase Directed Antibiotic Biosynthesis in the Bacterial Phylum Bacteroidetes. ChemBioChem 2010, 11, 2506–2512. [CrossRef]
- Wang, Q.; Song, F.; Xiao, X.; Huang, P.; Li, L.; Monte, A.; Abdel-Mageed, W.M.; Wang, J.; Guo, H.; He, W.; et al. Abyssomicins from the South China Sea Deep-Sea Sediment Verrucosispora sp.: Natural Thioether Michael Addition Adducts as Antitubercular Prodrugs. Angew. Chem. Int. Ed. 2013, 52, 1231–1234. [CrossRef]
- Bister, B.; Bischoff, D.; Ströbele, M.; Riedlinger, J.; Reicke, A.; Wolter, F.; Bull, A.T.; Zähner, H.; Fiedler, H.-P.; Süssmuth, R.D. Abyssomicin C - A Polycyclic Antibiotic from a Marine Verrucosispora Strain as an Inhibitor of the p-Aminobenzoic Acid/Tetrahydrofolate Biosynthesis Pathway. Angew. Chem. Int. Ed. 2004, 43, 2574–2576. [CrossRef]
- Shang, Z.; Khalil, Z.; Li, L.; Salim, A.A.; Quezada, M.; Kalansuriya, P.; Capon, R.J. Roseopurpurins: Chemical Diversity Enhanced by Convergent Biosynthesis and Forward and Reverse Michael Additions. Org. Lett. 2016, 18, 4340–4343. [CrossRef]
- Han, J.; Bruhn, D.F.; Roberts, D.C.; Burkman, E.; Moreno, Y.; Salim, A.A.; Capon, R.J. Goondomycins A–H: Carbocyclic Ansa-Polyketides from an Australian Pasture Streptomyces with Selective Activity against Dirofilaria immitis. J. Nat. Prod. 2024, 87, 2810–2821. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




