Submitted:
27 November 2025
Posted:
28 November 2025
You are already at the latest version
Abstract
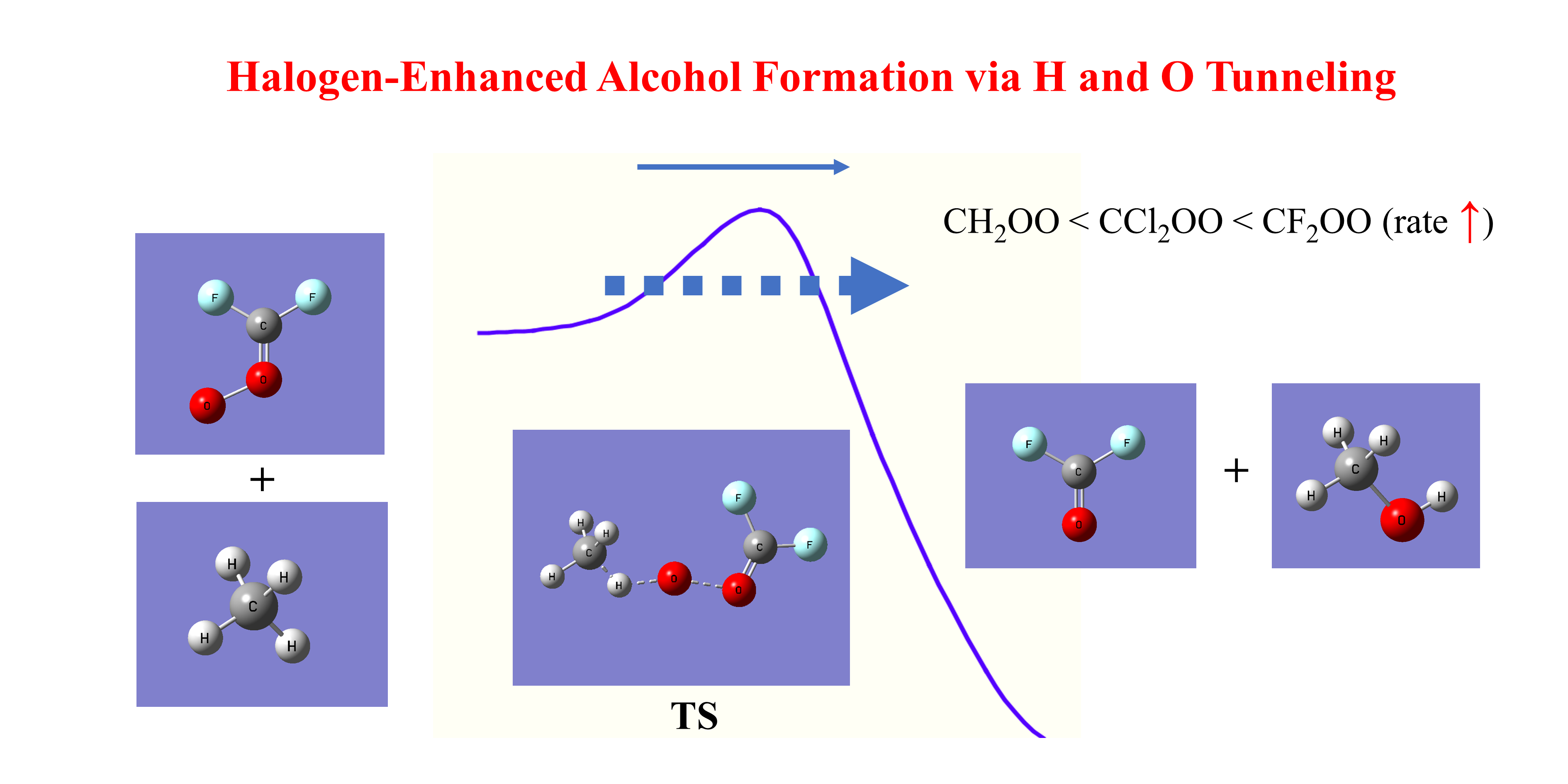
The reactions of formaldehyde oxide (CH2OO) with methane and ethane that yield alcohol products were investigated using dual-level variational transition state theory with multidimensional tunneling corrections (VTST/MT). Additional systems—including halogenated formaldehyde oxides (CF2OO and CCl2OO), deuterated alkanes (CD4, C2D6), and isotopically substituted formaldehyde oxide (CH218O18O)—were also examined to explore substituent and isotope effects. Bimolecular rate constants and kinetic isotope effects (KIEs) were computed over the temperature range of 100–600 K. Significant tunneling contributions were predicted, especially below room temperature, where tunneling increases the rate constants of the CH2OO + alkane reactions by up to two orders of magnitude. The computed H/D KIEs are approximately 3 at 300 K and rise to ~10 at 200 K. Notably, pronounced oxygen tunneling was also observed, giving 18O KIEs of ~1.2 at 300 K and ~2.2 at 200 K. Halogen substitution was predicted to substantially reduce reaction barriers due to the weakening of the O–O bond, leading to rate constants for CF2OO reactions that exceed those of CH2OO by more than ten orders of magnitude at 300 K. The mechanisms underlying the strong tunneling effects, the individual contributions to the calculated KIEs, and the implications of these findings for atmospheric chemistry are discussed.
Keywords:
1. Introduction
2. Computational Details
3. Results and Discussion
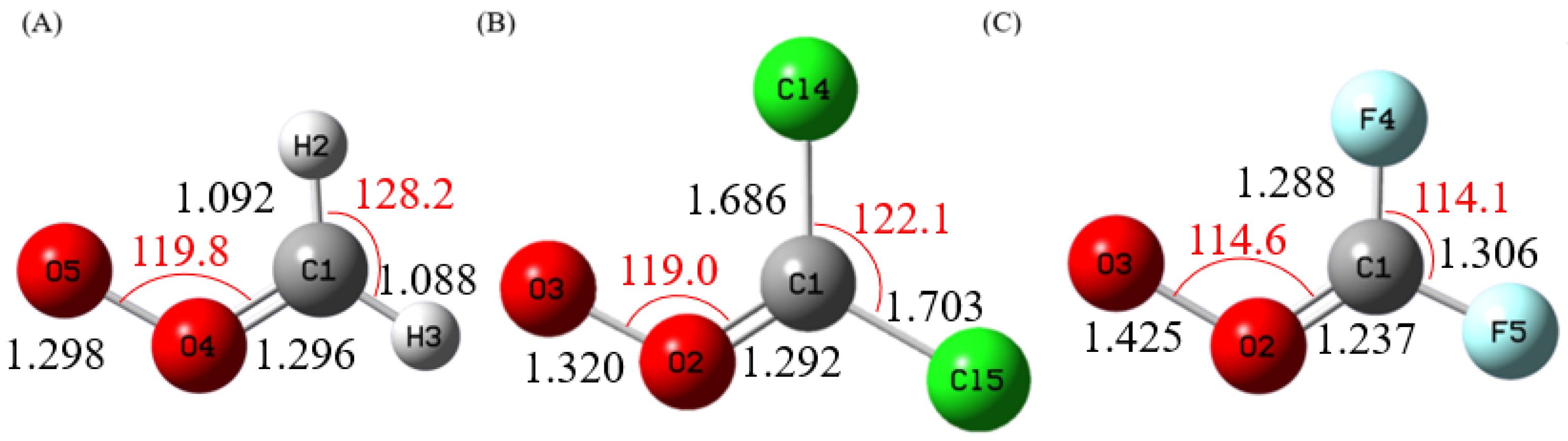
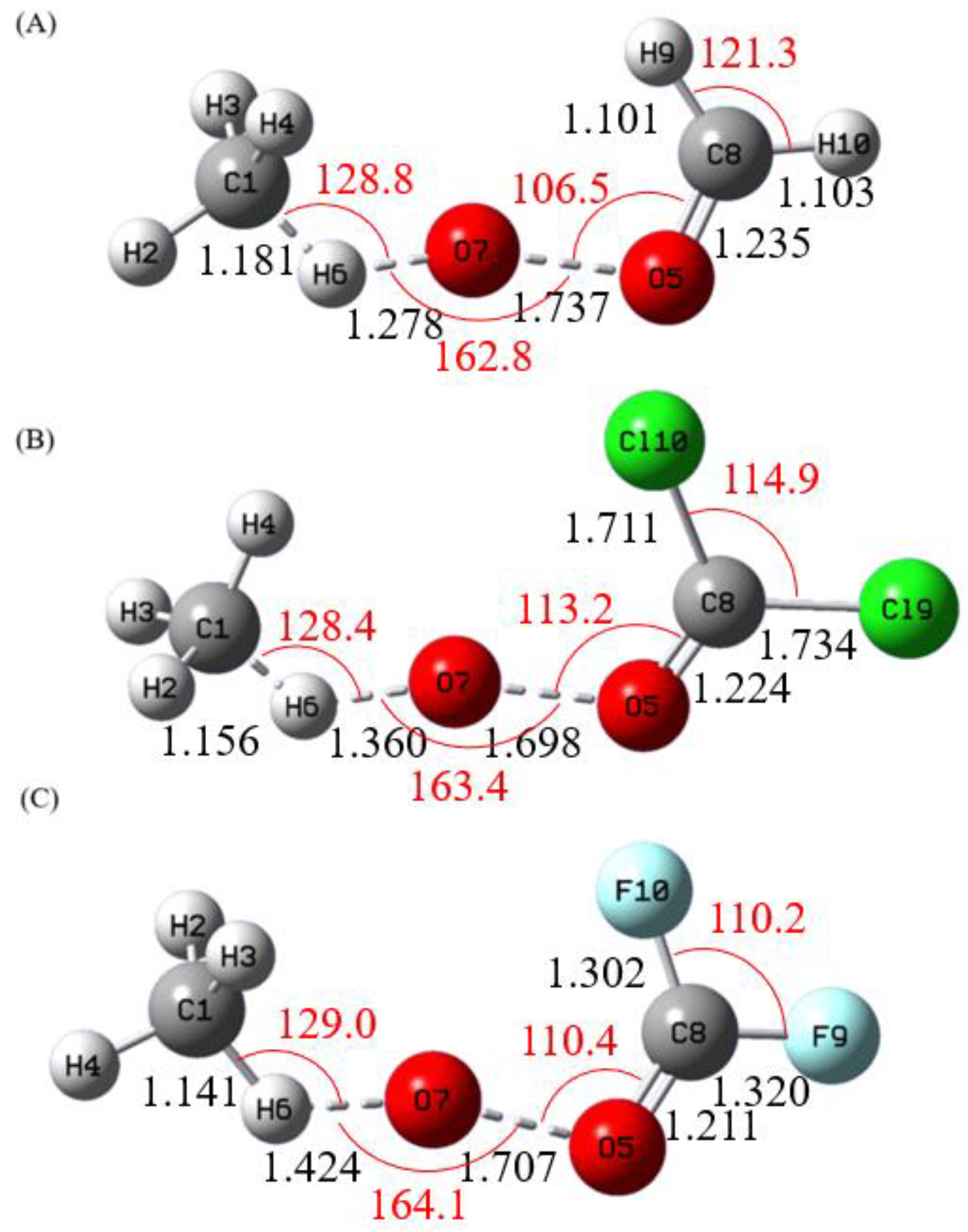
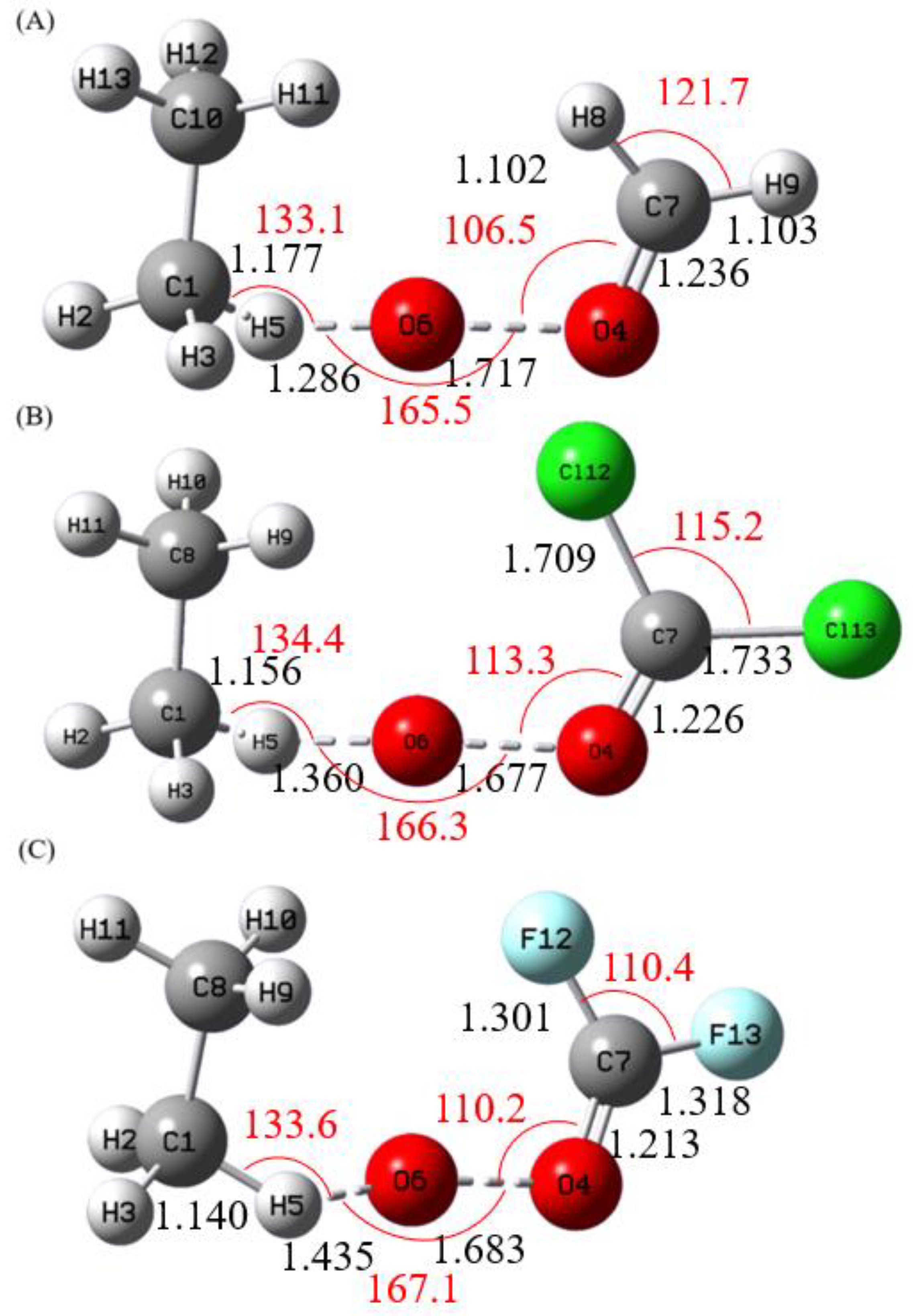
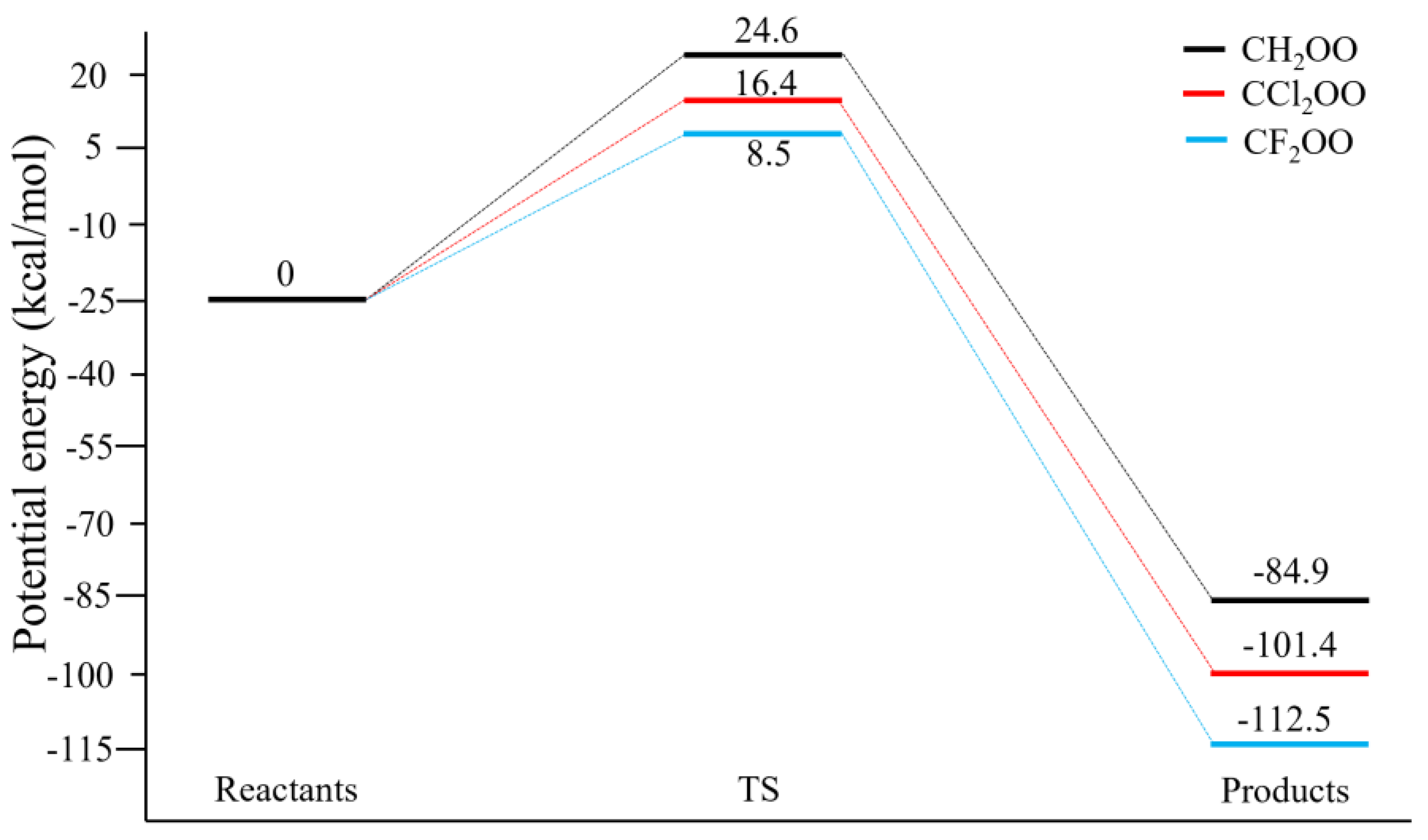
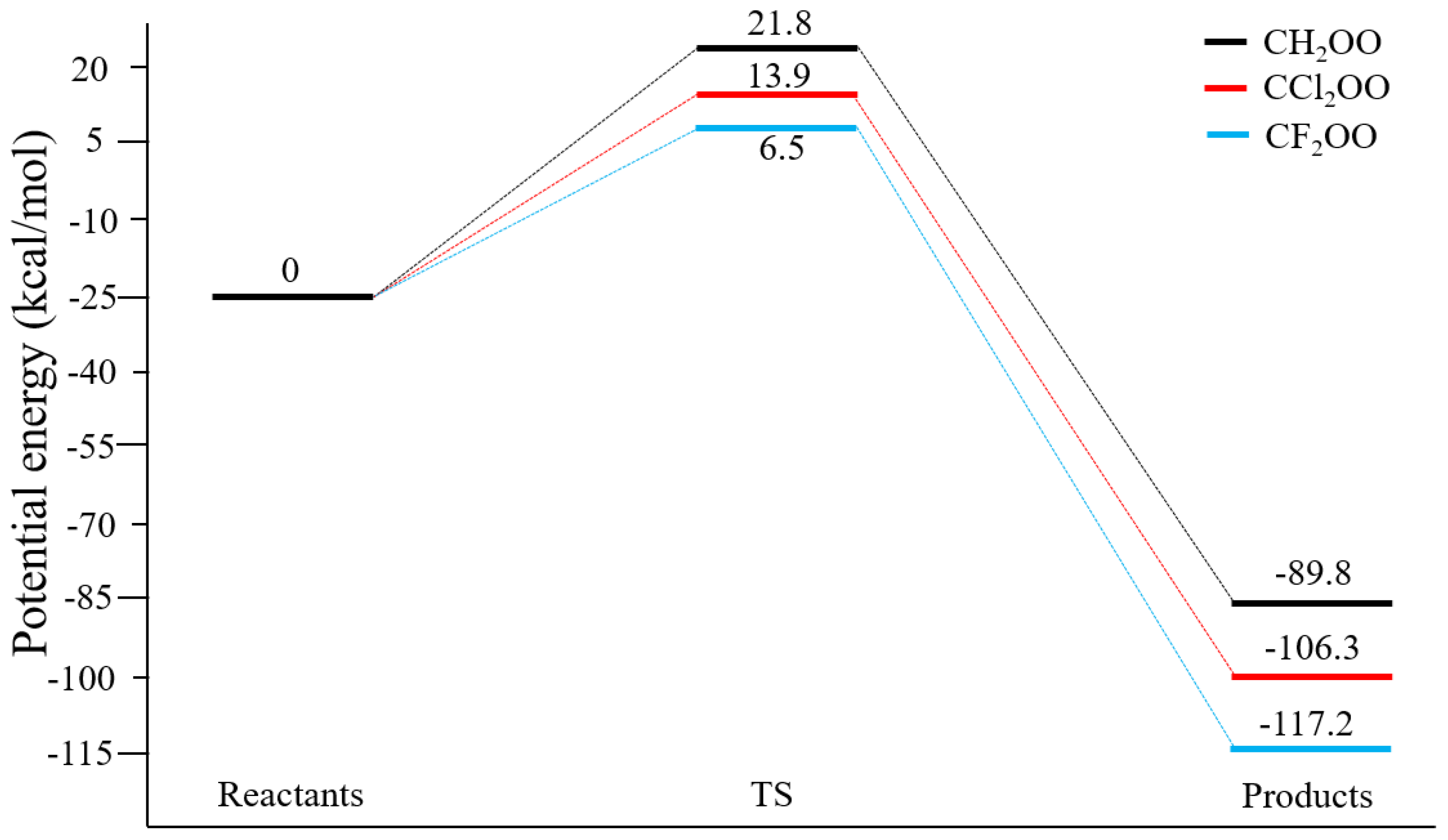
3.1. Structures and Energetics
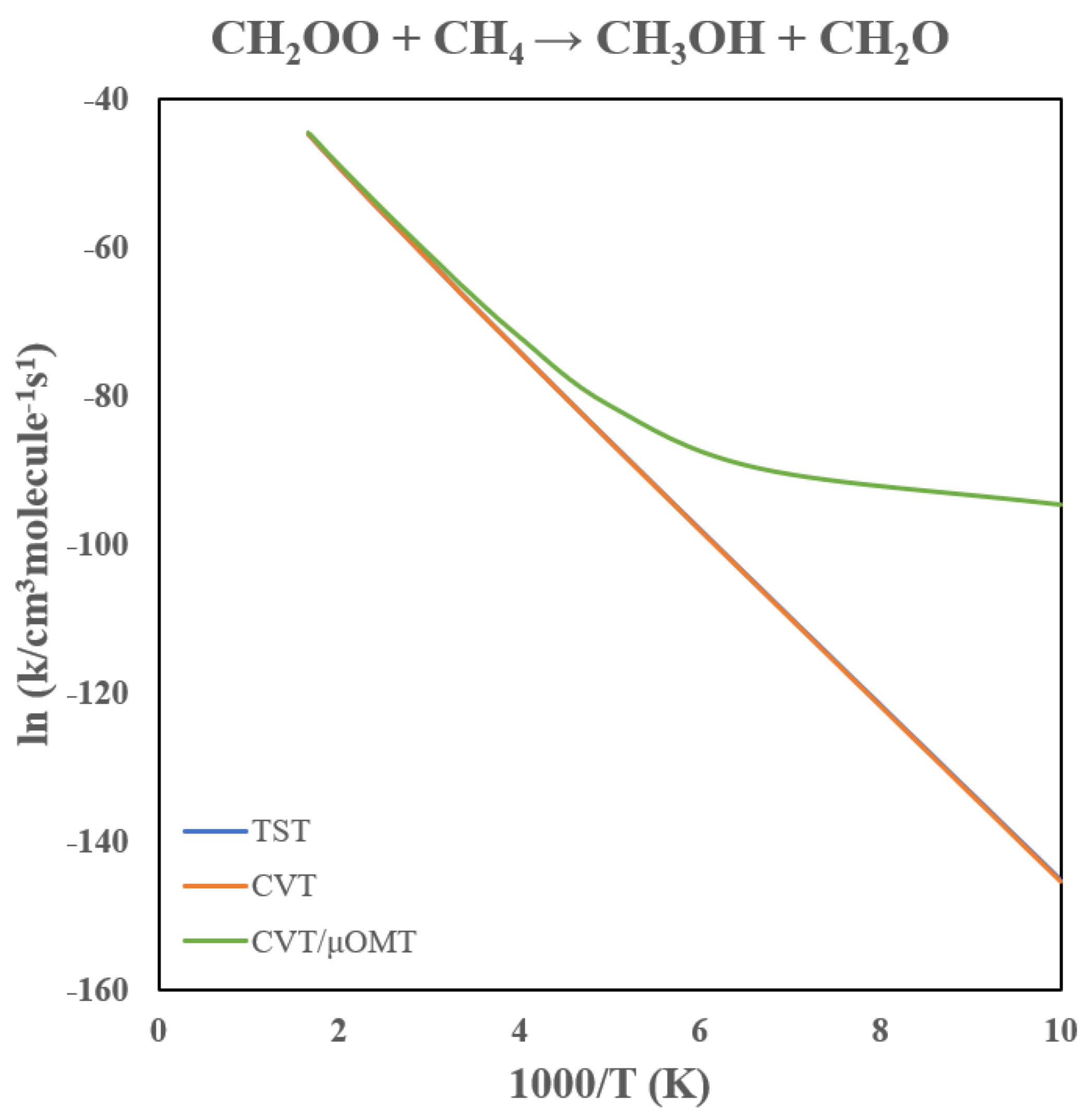
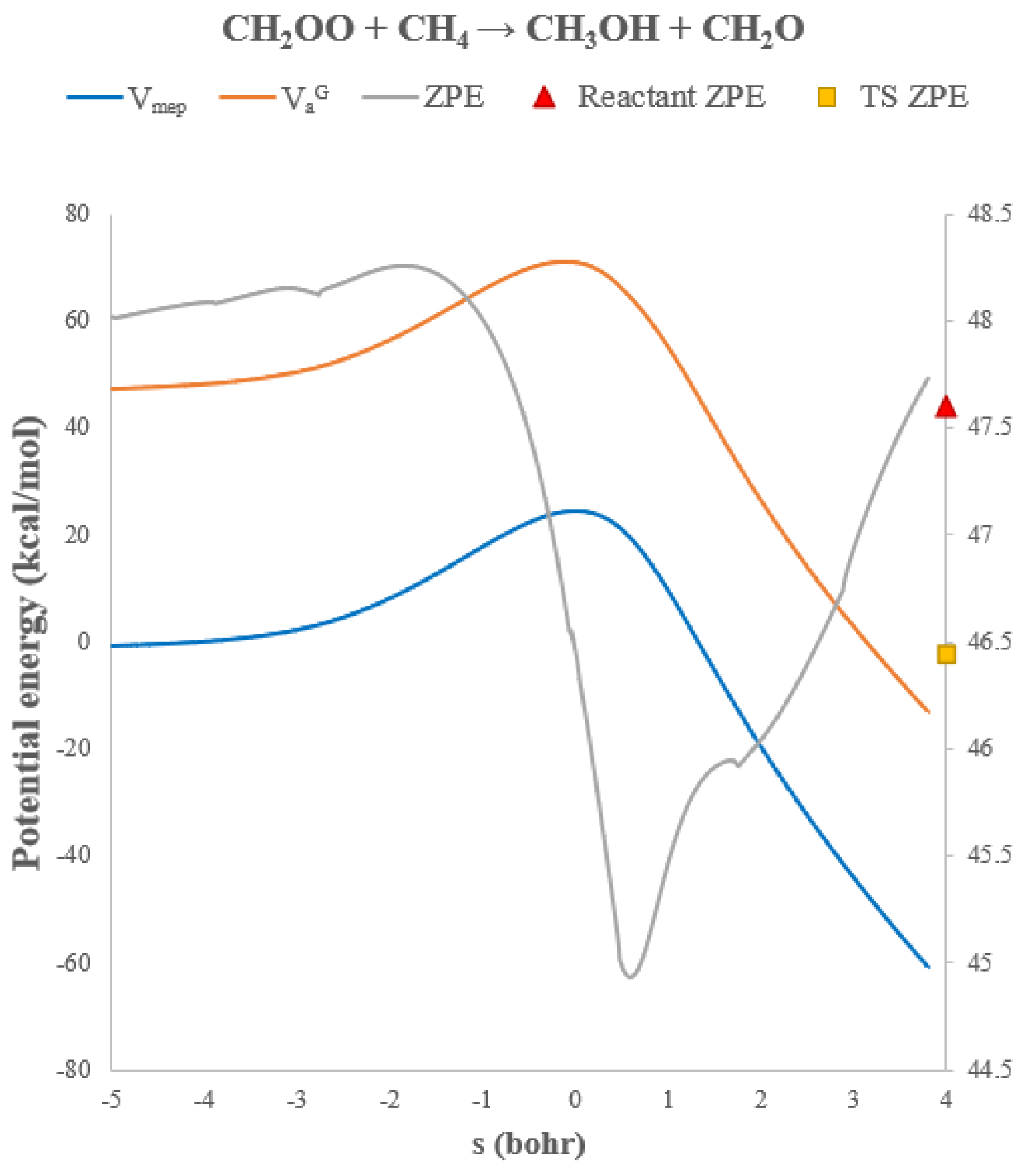
3.2. Rate Constants of CH2OO + CH4
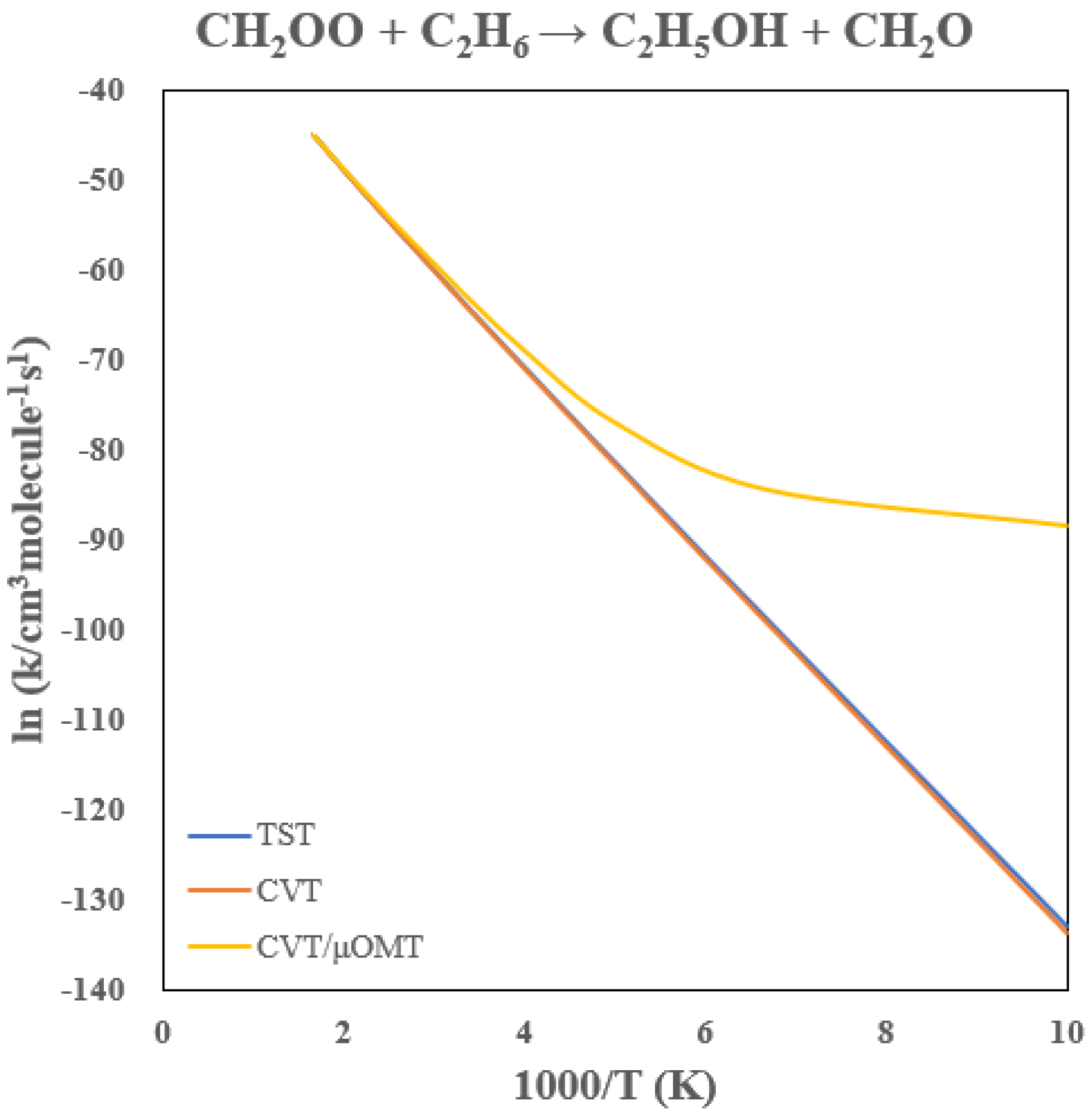
3.3. Rate Constants of CH2OO + C2H6
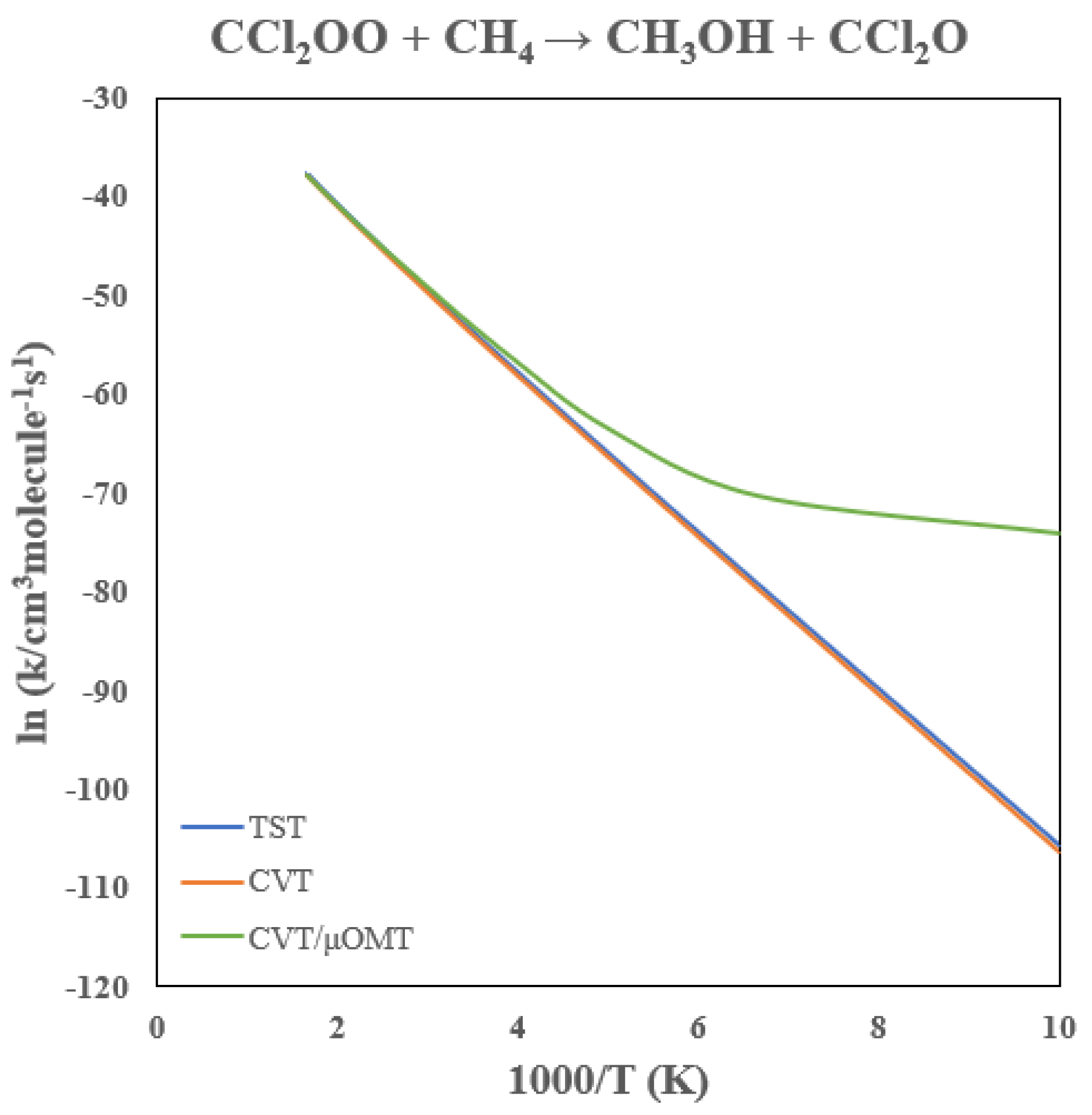
3.4. Rate Constants of CCl2OO + CH4
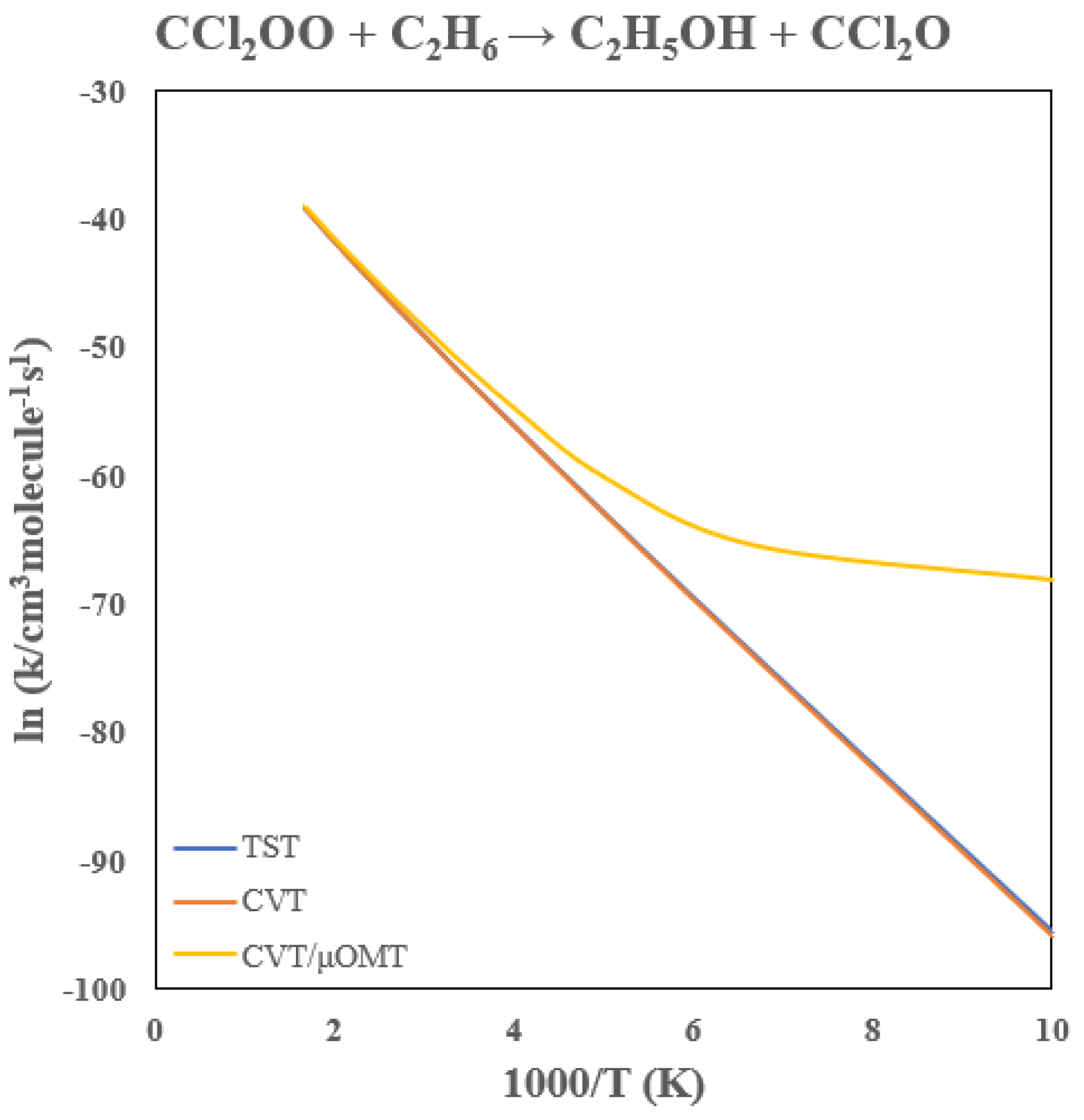
3.5. Rate Constants of CCl2OO + C2H6
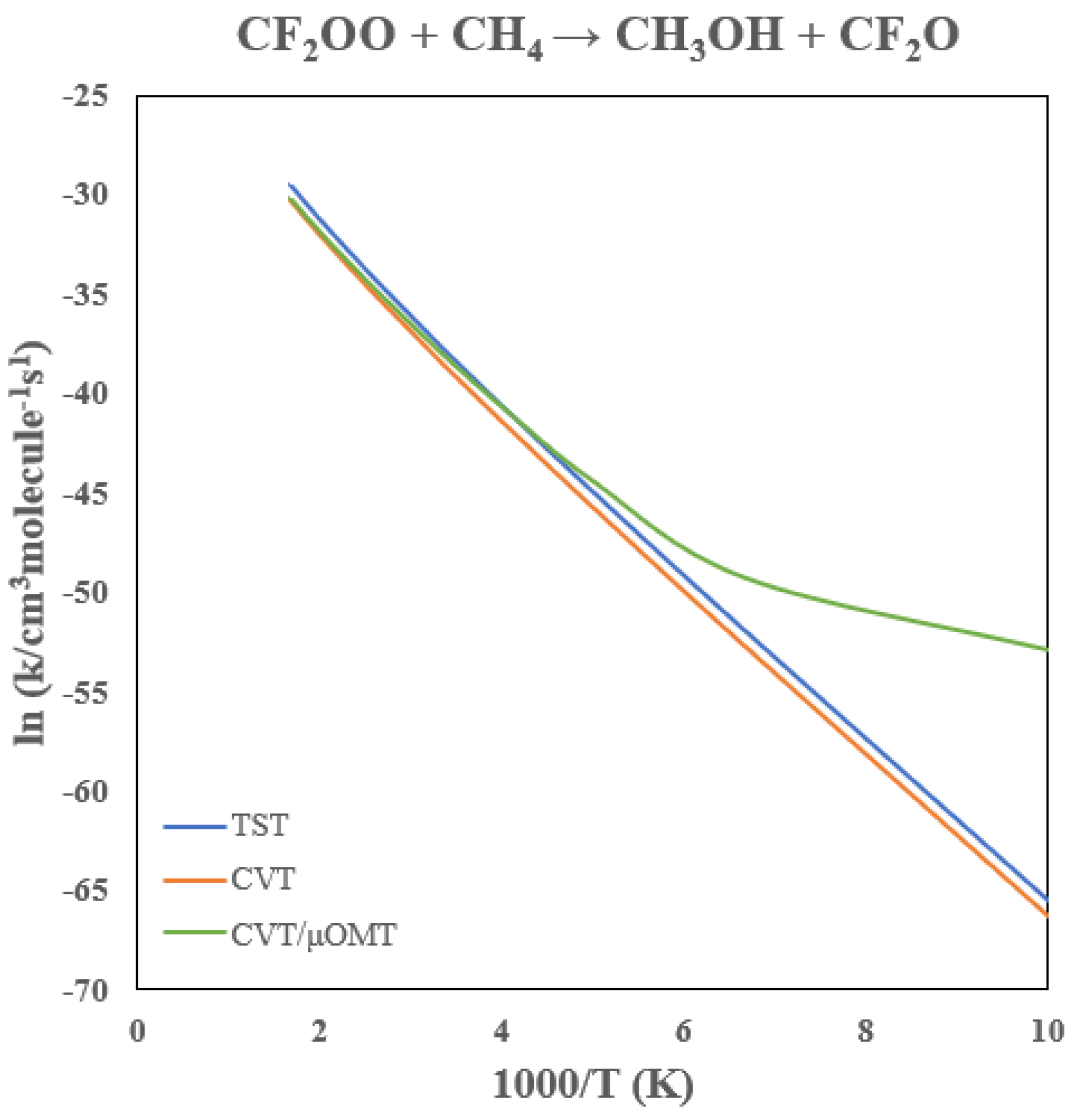
3.6. Rate Constants of CF2OO + CH4
3.7. Rate Constants of CF2OO + C2H6
3.8. Kinetic Isotope Effects
3.8.1. Deuterium Kinetic Isotope Effects (H/D)
3.8.2. Heavy-Atom Kinetic Isotope Effects (16O/18O)
3.8.3. Mechanistic Implications
4. Summary
Supplementary Materials
Ackowlegement
References
- Criegee, R. Mechanism of Ozonolysis. Angew. Chem. Int. Ed. Engl. 1975, 14, 745–752. [Google Scholar] [CrossRef]
- Johnson, D.; Marston, G. The Gas-Phase Ozonolysis of Unsaturated Volatile Organic Compounds in the Troposphere. Chem. Soc. Rev. 2008, 37, 699–716. [Google Scholar] [CrossRef]
- Vereecken, L.; Harder, H.; Novelli, A. The Reaction of Criegee Intermediates with NO, RO2, and SO2, and Their Fate in the Atmosphere. Phys. Chem. Chem. Phys. 2012, 14, 14682–14695. [Google Scholar] [CrossRef] [PubMed]
- Percival, C.J.; Welz, O.; Eskola, A.J.; Savee, J.D.; Osborn, D.L.; Topping, D.O.; Lowe, D.; Utembe, S.R.; Bacak, A.; McFiggans, G.; Cooke, M.C.; Xiao, P.; Archibald, A.T.; Jenkin, M.E.; Derwent, R.G.; Riipinen, I.; Mok, D.W.K.; Lee, E.P.F.; Dyke, J.M.; Taatjes, C.A.; Shallcross, D.E. Regional and Global Impacts of Criegee Intermediates on Atmospheric Sulphuric Acid Concentrations and First Steps of Aerosol Formation. Faraday Discuss. 2013, 165, 45–73. [Google Scholar] [CrossRef] [PubMed]
- Kurtén, T.; Lane, J.R.; Jørgensen, S.; Kjaergaard, H.G. A Computational Study of the Oxidation of SO2 to SO3 by Gas-Phase Organic Oxidants. J. Phys. Chem. A 2011, 115, 8669–8681. [Google Scholar] [CrossRef] [PubMed]
- Ziemann, P.J.; Atkinson, R. Kinetics, Products, and Mechanisms of Secondary Organic Aerosol Formation. Chem. Soc. Rev. 2012, 41, 6582–6605. [Google Scholar] [CrossRef]
- Gong, Y.; Chen, Z. Quantification of the Role of Stabilized Criegee Intermediates in the Formation of Aerosols in Limonene Ozonolysis. Atmospheric Chem. Phys. 2021, 21, 813–829. [Google Scholar] [CrossRef]
- Gutbrod, R.; Kraka, E.; Schindler, R.N.; Cremer, D. Kinetic and Theoretical Investigation of the Gas-Phase Ozonolysis of Isoprene: Carbonyl Oxides as an Important Source for OH Radicals in the Atmosphere. J. Am. Chem. Soc. 1997, 119, 7330–7342. [Google Scholar] [CrossRef]
- Novelli, A.; Vereecken, L.; Lelieveld, J.; Harder, H. Direct Observation of OH Formation from Stabilised Criegee Intermediates. Phys. Chem. Chem. Phys. 2014, 16, 19941–19951. [Google Scholar] [CrossRef] [PubMed]
- Long, B.; Bao, J.L.; Truhlar, D.G. Atmospheric Chemistry of Criegee Intermediates: Unimolecular Reactions and Reactions with Water. J. Am. Chem. Soc. 2016, 138, 14409–14422. [Google Scholar] [CrossRef]
- Long, B.; Wang, Y.; Xia, Y.; He, X.; Bao, J.L.; Truhlar, D.G. Atmospheric Kinetics: Bimolecular Reactions of Carbonyl Oxide by a Triple-Level Strategy. J. Am. Chem. Soc. 2021, 143, 8402–8413. [Google Scholar] [CrossRef]
- Chao, W.; Hsieh, J.-T.; Chang, C.-H.; Lin, J.J.-M. Direct Kinetic Measurement of the Reaction of the Simplest Criegee Intermediate with Water Vapor. Science 2015, 347, 751–754. [Google Scholar] [CrossRef]
- Smith, M.C.; Chang, C.-H.; Chao, W.; Lin, L.-C.; Takahashi, K.; Boering, K.A.; Lin, J.J.-M. Strong Negative Temperature Dependence of the Simplest Criegee Intermediate CH2OO Reaction with Water Dimer. J. Phys. Chem. Lett. 2015, 6, 2708–2713. [Google Scholar] [CrossRef] [PubMed]
- Vereecken, L. The Reaction of Criegee Intermediates with Acids and Enols. Phys. Chem. Chem. Phys. 2016, 18, 21–46. [Google Scholar] [CrossRef]
- Tratnyek, P.G.; Edwards, E.; Carpenter, L.; Blossom, S. Environmental Occurrence, Fate, Effects, and Remediation of Halogenated (Semi)Volatile Organic Compounds. Environ. Sci. Process. Impacts 2020, 22, 465–471. [Google Scholar] [CrossRef]
- U.S. EPA. Technical Overview of Volatile Organic Compounds (VOCs); U.S. Environmental Protection Agency: Washington, DC, 2023. [Google Scholar]
- Varga, B.; Csenki, J.T.; Tóth, B.L.; Béke, F.; Novák, Z.; Kotschy, A. Application of Industrially Relevant HydroFluoroOlefin (HFO) Gases in Organic Syntheses. Synthesis 2021, 53, 4313–4326. [Google Scholar] [CrossRef]
- Ceballos, D.M.; Fellows, K.; Evans, A.E.; Janulewicz, P.A.; Lee, E.G.; Whittaker, S.G. Perchloroethylene and Dry Cleaning: It’s Time to Move the Industry to Safer Alternatives. Front. Public Health 2021, 9, 638082. [Google Scholar] [CrossRef]
- Modenese, A.; Bisegna, F.; Fabbri, G.; Cozza, V. Evaluation of Occupational Exposure to Perchlorethylene in the Dry-Cleaning Industry. Int. J. Environ. Res. Public Health 2019, 16, 2832. [Google Scholar] [CrossRef]
- Tanaka, K.; Higashi, Y. Thermodynamic Properties of HFO-1234yf (2,3,3,3-Tetrafluoropropene). Int. J. Refrigeration 2010, 33, 474–479. [Google Scholar] [CrossRef]
- Puts, G.J.; Crouse, P.; Ameduri, B.M. Polytetrafluoroethylene: Synthesis and Characterization of the Original Extreme Polymer. Chem. Rev. 2019, 119, 1763–1805. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Kumar, S.; Mall, A. A Comprehensive Review Regarding Condensation of Low-GWP Refrigerants (Including R-1234yf, R-1234ze) in HVAC Systems. Processes 2022, 10, 1882. [Google Scholar] [CrossRef]
- Zhang, X.; Li, Y. A Review of Recent Research on Hydrofluoroolefin (HFO) and Hydrochlorofluoroolefin (HCFO) Refrigerants. Energy 2024, 311, 133423. [Google Scholar] [CrossRef]
- Caravan, R.L.; Vansco, M.F.; Lester, M.I. Open questions on the reactivity of Criegee intermediates . Commun. Chem. 2021, 4, 44. [Google Scholar] [CrossRef]
- Karlsson, E.; Rabayah, R.; Liu, T.; Cruz, E.M.; Kozlowski, M.C.; Karsili, T.N.V.; Lester, M.I. Electronic Spectroscopy of the Halogenated Criegee Intermediate, ClCHOO: Experiment and Theory. J. Phys. Chem. A 2024, 128, 10949–10956. [Google Scholar] [CrossRef]
- Taatjes, C.A.; Shallcross, D.E.; Percival, C.J. Research frontiers in the chemistry of Criegee intermediates and tropospheric ozonolysis. Phys. Chem. Chem. Phys. 2014, 16, 1704–1718. [Google Scholar] [CrossRef]
- Welz, O.; Savee, J.D.; Osborn, D.L.; Vasu, S.; Reisenauer, H.P.; Lester, M.I.; Taatjes, C.A. Direct Kinetic Measurements of Criegee Intermediate (CH2OO) Formed by Reaction of CH2I with O2. Science 2012, 335, 204–207. [Google Scholar] [CrossRef]
- Novelli, A.; Vereecken, L.; Lelieveld, J.; Harder, H. Direct Observation of OH Formation from Stabilised Criegee Intermediates. Phys. Chem. 2014, 16, 19941–19951. [Google Scholar] [CrossRef]
- Watson, N.A.I.; Beames, J.M. Bimolecular Sinks of Criegee Intermediates Derived from Hydrofluoroolefins — A Computational Analysis. Environ. Sci.: Atmos. 2023, 3, 1460–1484. [Google Scholar] [CrossRef]
- Holland, R.E.T.; Khan, M.A.H.; Driscoll, I.; Chhantyal-Pun, R.; Derwent, R.G.; Taatjes, C.A.; Orr-Ewing, A.J.; Percival, C.J.; Shallcross, D.E. Investigation of the Production of Trifluoroacetic Acid from Two Halocarbons, HFC-134a and HFO-1234yf, and Its Fates Using a Global 3-D Chemical Transport Model. ACS Earth Space Chem. 2021, 5, 849–857. [Google Scholar] [CrossRef]
- McGillen, M.R.; Fried, Z.T.P.; Khan, M.A.H.; Zhang, K.Z. Ozonolysis Can Produce Long-Lived Greenhouse Gases from Commercial Refrigerants. Proc. Natl. Acad. Sci. U.S.A. 2023, 120, e2312714120. [Google Scholar] [CrossRef] [PubMed]
- Hearn, J.D.; Harding, L.B.; Sheps, L.; et al. Electronic Spectroscopy of the Halogenated Criegee Intermediates: Photochemical Generation and VUV Photoionization Detection of ClCHOO. J. Phys. Chem. A 2024, 128, 10949–10956. [Google Scholar] [CrossRef]
- Welz, O.; Savee, J.D.; Osborn, D.L.; Vasu, S.S.; Percival, C.J.; Shallcross, D.E.; Taatjes, C.A. Direct Kinetic Measurements of Criegee Intermediate (CH2OO) Formed by Reaction of CH2I with O2. Science 2012, 335, 204–207. [Google Scholar] [CrossRef] [PubMed]
- Ting, W.L.; Chang, C.H.; Chao, W.; Smith, M.C.; Lin, J.J.M. Direct Kinetic Measurement of the Reaction of the Simplest Criegee Intermediate CH2OO with Water Vapor. J. Phys. Chem. A 2014, 118, 10116–10125. [Google Scholar] [CrossRef]
- Lee, Y.-P. Perspective: Spectroscopy and kinetics of small gaseous Criegee intermediates. J. Chem. Phys. 2015, 143, 020901. [Google Scholar] [CrossRef]
- Taatjes, C.A. Criegee intermediates: what direct production and detection can teach us about reactions of carbonyl oxides. Annu. Rev. Phys. Chem. 2017, 68, 183–207. [Google Scholar] [CrossRef]
- Gong, S.; Shi, Y. Evaluation of Comprehensive Monthly-Gridded Methane Emissions from Natural and Anthropogenic Sources in China. Sci. Total Environ. 2021, 784, 147116. [Google Scholar] [CrossRef]
- Lee, B.H.; Munger, W.; Wofsy, S.C.; Goldstein, A.H. Anthropogenic emissions of nonmethane hydrocarbons in the United States: Measured seasonal variations from 1992–1996 and 1999–2001. J. Geophys. Res. Atmos. 2006, 111, D20307. [Google Scholar] [CrossRef]
- Peischl, J.; Ryerson, T.B.; Aikin, K.C.; et al. Quantifying Methane and Ethane Emissions to the Atmosphere from Central and Western U.S. Oil and Gas Production Regions. J. Geophys. Res. Atmos. 2018, 123, 7725–7740. [Google Scholar] [CrossRef]
- Allen, D.T.; Torres, V.M.; Thomas, J.; Sullivan, D.W.; Harrison, M.; Hendler, A.; Herndon, S.C.; Kolb, C.E.; Fraser, M.P.; Hill, A.D.; Lamb, B. Attributing Atmospheric Methane to Anthropogenic Emission Sources. Acc. Chem. Res. 2016, 49, 1494–1502. [Google Scholar] [CrossRef]
- Bourtsoukidis, E.; et al. Non-methane hydrocarbon (C2–C8) sources and sinks in and around the Arabian Gulf. Atmos. Chem. Phys. 2019, 19, 7209–7232. [Google Scholar] [CrossRef]
- Li, M.; Pozzer, A.; Lelieveld, J.; Williams, J. Northern hemispheric atmospheric ethane trends (2006–2016) with reference to methane and propane. Earth Syst. Sci. Data 2022, 14, 4351–4364. [Google Scholar] [CrossRef]
- Filonchyk, M.; Peterson, M.P.; Zhang, L.; Hurynovich, V.; He, Y. Greenhouse Gases Emissions and Global Climate Change: Examining the Influence of CO2, CH4, and N2O. Sci. Total Environ. 2024, 935, 173359. [Google Scholar] [CrossRef]
- Dorofeenko, S.O.; Polianczyk, E.V.; Tsvetkov, M.V. Toward the Ultimate Efficiency of Methane to Syngas Conversion by Partial Oxidation: A Moving Bed Reactor with Parallel Preheating of Reactants. Fuel 2024, 363, 131005. [Google Scholar] [CrossRef]
- Rajeswari, S.; Baskaran, D.; Saravanan, P.; Rajasimman, M.; Rajamohan, N.; Vasseghian, Y. Production of Ethanol from Biomass – Recent Research, Scientometric Review and Future Perspectives. Fuel 2022, 317, 123448. [Google Scholar] [CrossRef]
- Gao, J.; Li, Z.; Dong, M.; Fan, W.; Wang, J. Thermodynamic Analysis of Ethanol Synthesis from Hydration of Ethylene Coupled with a Sequential Reaction. Front. Chem. Sci. Eng. 2020, 14, 847–856. [Google Scholar] [CrossRef]
- Xu, K.; Wang, W.; Wei, W.; Feng, W.; Sun, Q.; Li, P. Insights into the Reaction Mechanism of Criegee Intermediate CH2OO with Methane and Implications for the Formation of Methanol. J. Phys. Chem. A 2017, 121, 7236–7245. [Google Scholar] [CrossRef]
- Schreiner, P.R. Quantum Mechanical Tunneling Is Essential to Understanding Chemical Reactivity. Trends Chem. 2020, 2, 980–989. [Google Scholar] [CrossRef]
- Cha, Y.; Murray, C.J.; Klinman, J.P. Hydrogen Tunneling in Enzyme Reactions. Science 1989, 243, 1325–1330. [Google Scholar] [CrossRef]
- Zhou, Y.; Fang, W.; Wang, L.; Zeng, X.; Zhang, D.H.; Zhou, M. Quantum Tunneling in Peroxide O–O Bond Breaking Reaction. J. Am. Chem. Soc. 2023, 145, 8817–8821. [Google Scholar] [CrossRef]
- Truhlar, D.G.; Isaacson, A.D.; Garrett, B.C. In Theory of Chemical Reaction Dynamics; Baer, M., Ed.; CRC Press: Boca Raton, FL, 1985; Vol. 4, p 65.
- Truhlar, D.G.; Garrett, B.C. Variational Transition-State Theory . Annu. Rev. Phys. Chem. 1984, 35, 159–189. [Google Scholar] [CrossRef]
- Truhlar, D.G.; Garrett, B.C.; Klippenstein, S.J. Current Status of Transition-State Theory. J. Phys. Chem. 1996, 100, 12771–12800. [Google Scholar] [CrossRef]
- Fernandez-Ramos, A.; Ellingson, B.A.; Garrett, B.C.; Truhlar, D.G. Variational Transition State Theory with Multidimensional Tunneling. In Reviews in Computational Chemistry; Lipkowitz, K.B., Cundari, T.R., Eds.; Wiley-VCH: Hoboken, NJ, 2007; Vol. 23, pp. 125–232. [Google Scholar]
- Lu, D.-h.; Truong, T.N.; Melissas, V.S.; Lynch, G.C.; Liu, Y.-P.; Garrett, B.C.; Steckler, R.; Isaacson, A.D.; Rai, S.N.; Hancock, G.C.; Lauderdale, J.G.; Joseph, T.; Truhlar, D.G. POLYRATE 4: A New Version of a Computer Program for the Calculation of Chemical Reaction Rates for Polyatomics. Comput. Phys. Commun. 1992, 71, 235–262. [Google Scholar] [CrossRef]
- Yu, T.; Truhlar, D.G. Multipath Variational Transition State Theory: Multiple Reaction Paths and Path-Dependent Tunneling Effects . J. Phys. Chem. A 2012, 116, 297–308. [Google Scholar] [CrossRef]
- Kohen, A. Kinetic Isotope Effects as Probes for Hydrogen Tunneling, Coupled Motion and Dynamics Contributions to Enzyme Catalysis. Prog. React. Kinet. Mech. 2003, 28, 119–156. [Google Scholar] [CrossRef]
- Sen, A.; Kohen, A. Enzymatic Tunneling and Kinetic Isotope Effects: Chemistry at the Crossroads. J. Phys. Org. Chem. 2010, 23, 613–619. [Google Scholar] [CrossRef]
- Møller, Chr; Plesset, M.S. Note on an Approximation Treatment for Many-Electron Systems. Phys. Rev. 1934, 46, 618–622. [CrossRef]
- Woon, D.E.; Dunning, T.H. Gaussian Basis Sets for Use in Correlated Molecular Calculations. III. The Atoms Aluminum through Argon. J. Chem. Phys. 1993, 98, 1358–1371. [Google Scholar] [CrossRef]
- Dunning, T.H., Jr. Gaussian Basis Sets for Use in Correlated Molecular Calculations. I. The Atoms Boron through Neon and Hydrogen. J. Chem. Phys. 1989, 90, 1007–1023. [Google Scholar] [CrossRef]
- Helgaker, T.; Klopper, W.; Koch, H.; Noga, J. Basis-Set Convergence of Correlated Calculations on Water. J. Chem. Phys. 1997, 106, 9639–9646. [Google Scholar] [CrossRef]
- Sorathia, K.; Frantzov, D.; Tew, D.P. Improved CPS and CBS Extrapolation of PNO-CCSD(T) Energies: The MOBH35 and ISOL24 Data Sets . J. Chem. Theory Comput. 2024, 20, 2740–2750. [Google Scholar] [CrossRef] [PubMed]
- Náfrádi, D.; Kállay, M. Self-consistent Basis Set Extrapolation of Hartree–Fock Energies . Struct. Chem. 2025, 36, 1539–1546. [Google Scholar] [CrossRef]
- Karton, A. Effective Basis Set Extrapolations for CCSDT, CCSDT(Q), and CCSDT(Q) Correlation Energies . J. Chem. Phys. 2020, 153, 024102. [Google Scholar] [CrossRef]
- Page, M.; Doubleday, C.; McIver, J.W. Jr. Following steepest descent reaction paths. The use of higher energy derivatives with ab initio electronic structure methods. J. Chem. Phys. 1990, 93, 5634–5642. [Google Scholar] [CrossRef]
- Melissas, V.S.; Truhlar, D.G.; Garrett, B.C. Optimized Calculations of Reaction Paths and Reaction-Path Functions for Chemical Reactions. J. Chem. Phys. 1992, 96, 5758–5772. [Google Scholar] [CrossRef]
- Hu, W.-P.; Liu, Y.-P.; Truhlar, D.G. Variational Transition-State Theory and Semiclassical Tunnelling Calculations with Interpolated Corrections: A New Approach to Interfacing Electronic Structure Theory and Dynamics for Organic Reactions. J. Chem. Soc. Faraday Trans. 1994, 90, 1715. [Google Scholar] [CrossRef]
- Huang, C.-H.; You, R.-M.; Lian, P.-Y.; Hu, W.-P. Improved Interpolated Correction Schemes for Dual-Level Direct Dynamics Calculation. J. Phys. Chem. A 2000, 104, 7200–7208. [Google Scholar] [CrossRef]
- Corchado, J.C.; Espinosa-García, J.; Hu, W.-P.; Rossi, I.; Truhlar, D.G. Dual-Level Reaction-Path Dynamics (the III Approach to VTST with Semiclassical Tunneling). Application to OH + NH3 → H2O + NH2. J. Phys. Chem. 1995, 99, 687–694. [Google Scholar] [CrossRef]
- Truhlar, D.G.; Garrett, B.C. Variational Transition-State Theory. Acc. Chem. Res. 1980, 13, 440–448. [Google Scholar] [CrossRef]
- Liu, Y.P.; Lu, D.H.; Gonzalez-Lafont, A.; Truhlar, D.G.; Garrett, B.C. Direct Dynamics Calculation of the Kinetic Isotope Effect for an Organic Hydrogen-Transfer Reaction, Including Corner-Cutting Tunneling in 21 Dimensions. J. Am. Chem. Soc. 1993, 115, 7806–7817. [Google Scholar] [CrossRef]
- Liu, Y.P.; Lynch, G.C.; Truong, T.N.; Lu, D.H.; Truhlar, D.G.; Garrett, B.C. Molecular modeling of the kinetic isotope effect for the [1,5]-sigmatropic rearrangement of cis-1,3-pentadiene. J. Am. Chem. Soc. 1993, 115, 2408–2415. [Google Scholar] [CrossRef]
- Fernandez-Ramos, A.; Truhlar, D.G. Improved algorithm for corner-cutting tunneling calculations. J. Chem. Phys. 2001, 114, 1491–1496. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; et al.Gaussian 16, Revision B.01 Gaussian, Inc., Wallingford CT, 2016.
- Zheng, J.; Bao, J.L.; Meana-Pañeda, R.; Zhang, S.; Lynch, B.J.; Corchado, J.C.; Chuang, Y.-Y.; Fast, P.L.; Hu, W.-P.; Liu, Y.-P.; Lynch, G.C.; Nguyen, K.A.; Jackels, C.F.; Fernandez Ramos, A.; Ellingson, B.A.; Melissas, V.S.; Villà, J.; Rossi, I.; Coitiño, E.L.; Pu, J.; Albu, T.V.; Ratkiewicz, A.; Steckler, R.; Garrett, B.C.; Isaacson, A.D.; Truhlar, D.G. Polyrate-version 2017-C; University of Minnesota: Minneapolis, 2017. [Google Scholar]
- Zheng, J.; Bao, J.L.; Zhang, S.; Corchado, J.C.; Meana-Pañeda, R.; Chuang, Y.-Y.; Coitiño, E.L.; Ellingson, B.A.; Truhlar, D.G. Gaussrate 17; University of Minnesota: Minneapolis, 2017. [Google Scholar]
- Li, J.; Carter, S.; Bowman, J.M.; Dawes, R.; Xie, D.; Guo, H. High-Level, First-Principles, Full-Dimensional Quantum Calculation of the Ro-vibrational Spectrum of the Simplest Criegee Intermediate (CH2OO). J. Phys. Chem. Lett. 2014, 5, 2364–2369. [Google Scholar] [CrossRef]
- Wagner, J.P. Gauging stability and reactivity of carbonyl O-oxide Criegee intermediates. Phys. Chem. Chem. Phys. 2019, 21, 21530. [Google Scholar] [CrossRef] [PubMed]
- Howard, C.J.; Evenson, K.M. Rate constants for the reactions of OH with CH4 and fluorine, chlorine, and bromine substituted methanes at 296 K. J. Chem. Phys. 1976, 64, 197–202. [Google Scholar] [CrossRef]
- Berg, F.; Novelli, A.; Dubus, R.; Hofzumahaus, A.; Holland, F.; Wahner, A.; Fuchs, H. Temperature-dependent rate coefficients for the reactions of OH radicals with selected alkanes, aromatic compounds, and monoterpenes. Atmos. Chem. Phys. 2024, 24, 13715–13731. [Google Scholar]
- Sage, A.M.; Donahue, N.M. Deconstructing Experimental Rate Constant Measurements: Obtaining Intrinsic Reaction Parameters, Kinetic Isotope Effects, and Tunneling Coefficients from Kinetic Data for OH + Methane, Ethane and Cyclohexane. J. Photochem. Photobiol. A: Chem. 2005, 176, 238–249. [Google Scholar] [CrossRef]
- Hu, W.-P.; Truhlar, D.G. Factors Affecting Competitive Ion–Molecule Reactions: ClO– + C2H5Cl and C2D5Cl via E2 and SN2 Channels. J. Am. Chem. Soc. 1996, 118, 860–869. [Google Scholar] [CrossRef]
- Wu, Y.-R.; Hu, W.-P. Reaction Dynamics Study on the Tunneling Effects of a Microsolvated E2 Reaction: FO–(H2O) + C2H5Cl → HOF(H2O) + C2H4 + Cl–. J. Am. Chem. Soc. 1999, 121, 10168–10177. [Google Scholar] [CrossRef]
- Tsai, W.-C.; Hu, W.-P. Theoretical Analysis on the Kinetic Isotope Effects of Bimolecular Nucleophilic Substitution (SN2) Reactions and Their Temperature Dependence. Molecules 2013, 18, 4816–4843. [Google Scholar] [CrossRef]
- Kang, B.; Radosavich, A.T. 18O Kinetic Isotope Effect Evidence for a Concerted [3 + 1] Cycloaddition in Phosphetane-Catalyzed Reductive N-Arylation of Nitroarenes. Tetrahedron 2025, 186, 134892. [Google Scholar] [CrossRef]
- Karmakar, S.; Datta, A. Heavy-Atom Tunneling in Organic Transformations. J. Chem. Sci. 2020, 132, 127. [Google Scholar] [CrossRef]
- Zuev, P.S.; Sheridan, R.S.; Albu, T.V.; Truhlar, D.G.; Hrovat, D.A.; Borden, W.T. Carbon Tunneling from a Single Quantum State. Science 2003, 299, 867. [Google Scholar] [CrossRef]
- Gonzalez-James, O.M.; Zhang, X.; Datta, A.; Hrovat, D.A.; Borden, W.T.; Singleton, D.A. Experimental Evidence for Heavy-Atom Tunneling in the Ring-Opening of Cyclopropylcarbinyl Radical from Intramolecular 12C/13C Kinetic Isotope Effects. J. Am. Chem. Soc. 2010, 132, 12548. [Google Scholar] [CrossRef]
- Chen, J. -L.; Hu, W. -P. Theoretical Prediction on the Thermal Stability of Cyclic Ozone and Strong Oxygen Tunneling. J. Am. Chem. Soc. 2011, 133, 16045-16053. [CrossRef]
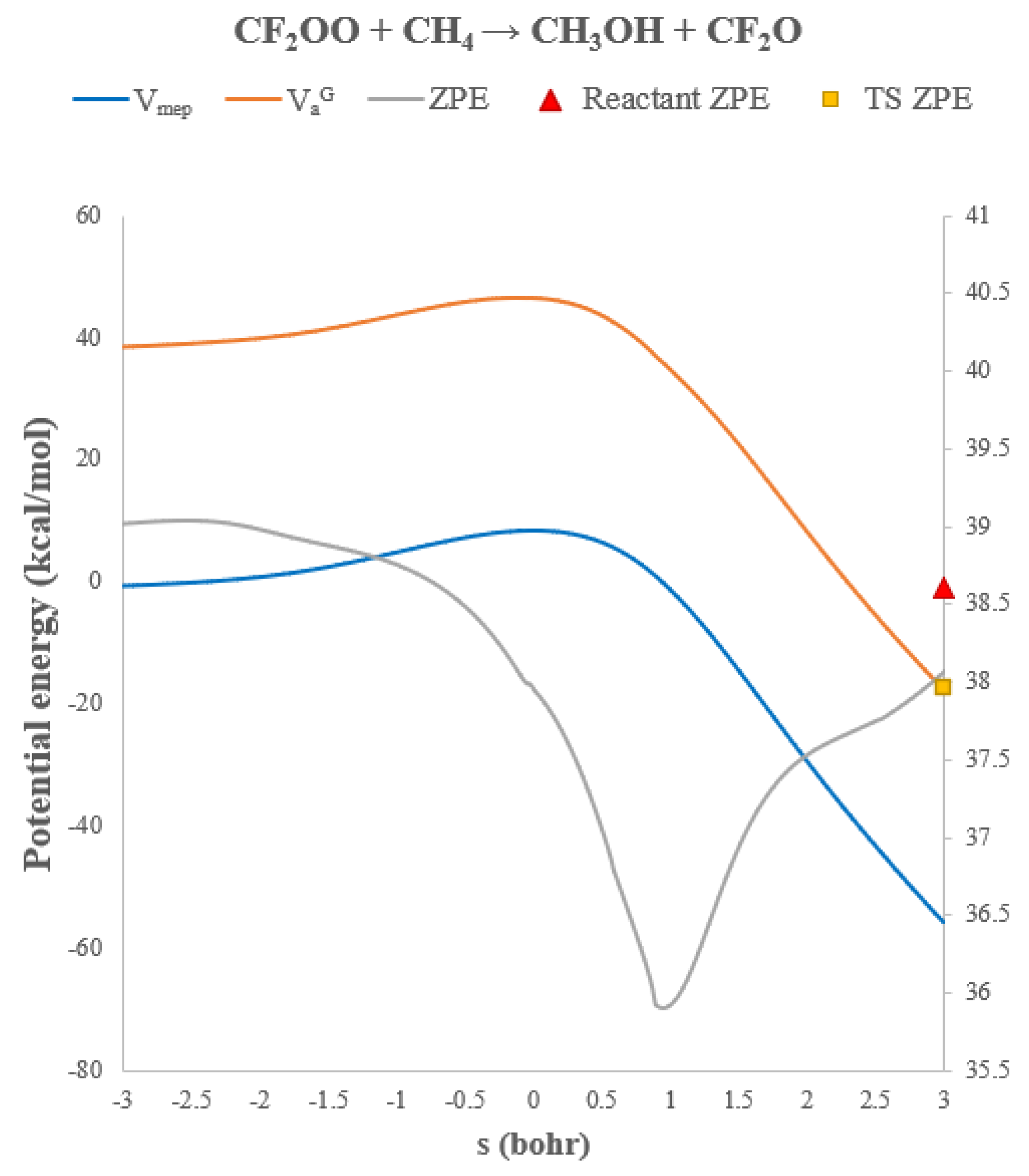
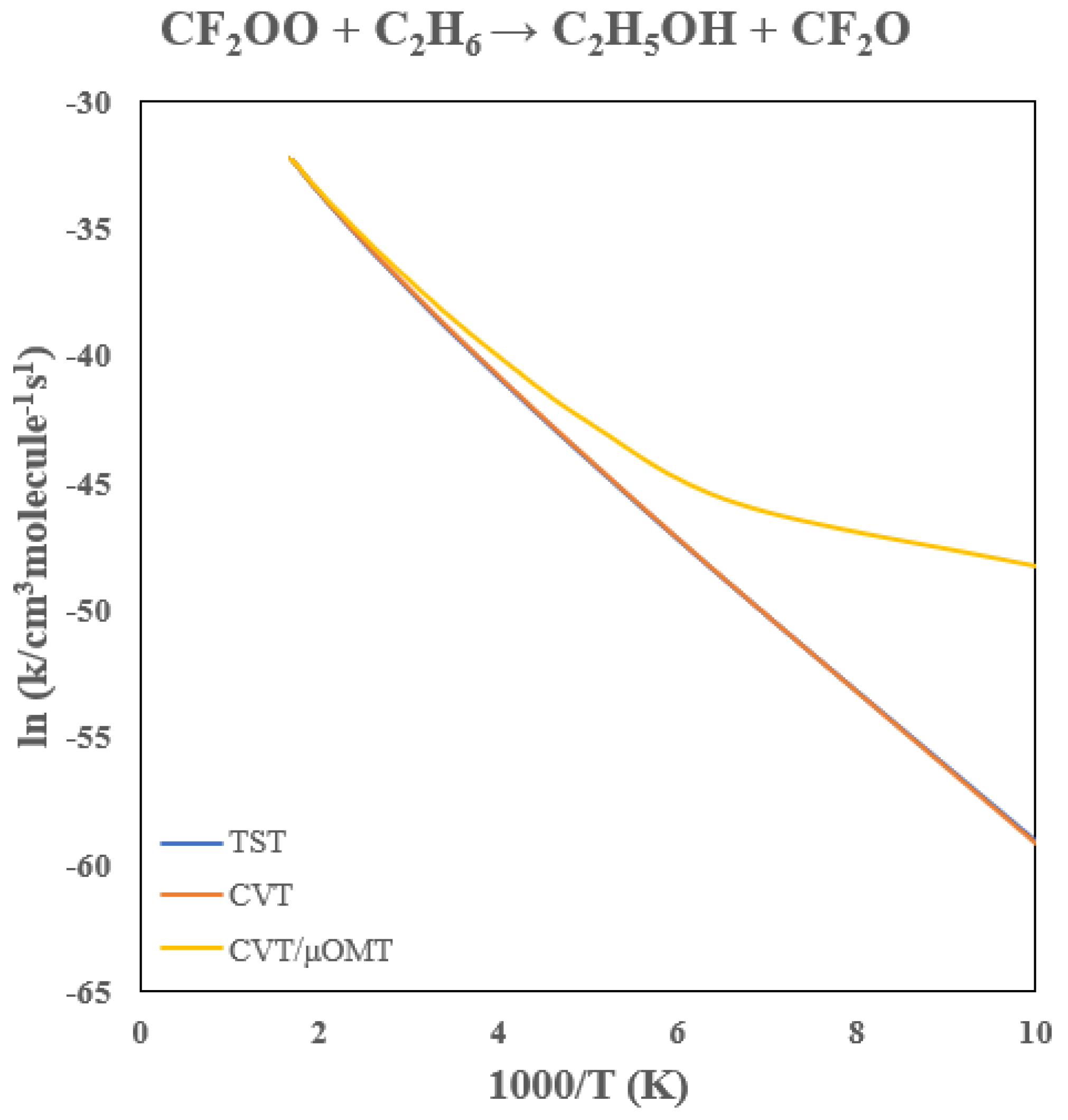
| T(K) | TST | CVT | CVT/μOMT |
|---|---|---|---|
| 100 | 8.29 × 10−64 | 6.33 × 10−64 | 7.65 × 10−42 |
| 150 | 1.06 × 10−46 | 8.99 × 10−47 | 9.97 × 10−40 |
| 200 | 4.55 × 10−38 | 4.06 × 10−38 | 5.12 × 10−36 |
| 250 | 7.91 × 10−33 | 7.25 × 10−33 | 5.43 × 10−32 |
| 298.15 | 2.11 × 10−29 | 1.97 × 10−29 | 6.31 × 10−29 |
| 300 | 2.73 × 10−29 | 2.54 × 10−29 | 7.98 × 10−29 |
| 350 | 9.87 × 10−27 | 9.30 × 10−27 | 1.97 × 10−29 |
| 400 | 8.66 × 10−25 | 8.22 × 10−25 | 1.40 × 10−24 |
| 500 | 5.09 × 10−22 | 4.88 × 10−22 | 6.65 × 10−22 |
| 600 | 3.97 × 10−20 | 3.83 × 10−20 | 4.67 × 10−20 |
| T(K) | TST | CVT | CVT/μOMT |
|---|---|---|---|
| 100 | 3.62 × 10−58 | 2.02 × 10−58 | 8.85 × 10−39 |
| 150 | 2.70 × 10−43 | 1.98 × 10−43 | 5.01 × 10−37 |
| 200 | 9.10 × 10−36 | 7.60 × 10−36 | 8.68 × 10−34 |
| 250 | 3.48 × 10−31 | 3.12 × 10−31 | 2.60 × 10−30 |
| 298.15 | 3.52 × 10−28 | 3.28 × 10−28 | 1.15 × 10−27 |
| 300 | 4.40 × 10−28 | 4.11 × 10−28 | 1.41 × 10−27 |
| 350 | 7.82 × 10−26 | 7.49 × 10−26 | 1.69 × 10−25 |
| 400 | 4.03 × 10−24 | 3.93 × 10−24 | 6.92 × 10−24 |
| 500 | 1.14 × 10−21 | 1.12 × 10−21 | 1.52 × 10−21 |
| 600 | 5.44 × 10−20 | 5.42 × 10−20 | 6.43 × 10−20 |
| T(K) | TST | CVT | CVT/μOMT |
|---|---|---|---|
| 100 | 1.30 × 10−46 | 6.91 × 10−47 | 6.01 × 10−33 |
| 150 | 3.80 × 10−35 | 2.22 × 10−35 | 2.71 × 10−31 |
| 200 | 2.48 × 10−29 | 1.52 × 10−29 | 2.69 × 10−28 |
| 250 | 8.74 × 10−26 | 5.53 × 10−26 | 2.22 × 10−25 |
| 298.15 | 1.87 × 10−23 | 1.21 × 10−23 | 2.90 × 10−23 |
| 300 | 2.23 × 10−23 | 1.44 × 10−23 | 3.40 × 10−23 |
| 350 | 1.25 × 10−21 | 8.19 × 10−22 | 1.49 × 10−21 |
| 400 | 2.72 × 10−20 | 1.80 × 10−20 | 2.80 × 10−20 |
| 500 | 2.28 × 10−18 | 1.53 × 10−18 | 2.00 × 10−18 |
| 600 | 4.85 × 10−17 | 3.28 × 10−17 | 3.95 × 10−17 |
| T(K) | TST | CVT | CVT/μOMT |
|---|---|---|---|
| 100 | 6.40 × 10−42 | 4.46 × 10−42 | 5.72 × 10−30 |
| 150 | 1.67 × 10−32 | 1.39 × 10−32 | 8.50 × 10−29 |
| 200 | 1.05 × 10−27 | 9.51 × 10−28 | 1.73 × 10−26 |
| 250 | 9.25 × 10−25 | 8.73 × 10−25 | 3.70 × 10−24 |
| 298.15 | 8.14 × 10−23 | 7.88 × 10−23 | 1.93 × 10−22 |
| 300 | 9.41 × 10−23 | 9.12 × 10−23 | 2.20 × 10−22 |
| 350 | 2.76 × 10−21 | 2.72 × 10−21 | 4.91 × 10−21 |
| 400 | 3.70 × 10−20 | 3.67 × 10−20 | 5.59 × 10−20 |
| 500 | 1.58 × 10−18 | 1.58 × 10−18 | 1.95 × 10−18 |
| 600 | 2.16 × 10−17 | 2.16 × 10−17 | 2.45 × 10−17 |
| T(K) | TST | CVT | CVT/μOMT |
| 100 | 3.56 × 10−29 | 1.58 × 10−29 | 1.08 × 10−23 |
| 150 | 2.69 × 10−23 | 1.25 × 10−23 | 4.06 × 10−22 |
| 200 | 2.87 × 10−20 | 1.37 × 10−20 | 5.26 × 10−20 |
| 250 | 2.15 × 10−18 | 1.04 × 10−18 | 2.19 × 10−18 |
| 298.15 | 3.84 × 10−17 | 1.87 × 10−17 | 3.05 × 10−17 |
| 300 | 4.21 × 10−17 | 2.06 × 10−17 | 3.33 × 10−17 |
| 350 | 3.78 × 10−16 | 1.86 × 10−16 | 2.61 × 10−16 |
| 400 | 2.07 × 10−15 | 1.03 × 10−15 | 1.32 × 10−15 |
| 500 | 2.50 × 10−14 | 1.25 × 10−14 | 1.45 × 10−14 |
| 600 | 1.47 × 10−14 | 7.35 × 10−14 | 8.14 × 10−14 |
| T(K) | TST | CVT | CVT/μOMT |
|---|---|---|---|
| 100 | 4.55 × 10−26 | 3.85 × 10−26 | 2.10 × 10−21 |
| 150 | 8.54 × 10−22 | 8.09 × 10−22 | 2.45 × 10−20 |
| 200 | 1.47 × 10−19 | 1.45 × 10−19 | 6.00 × 10−19 |
| 250 | 3.72 × 10−18 | 3.72 × 10−18 | 7.92 × 10−18 |
| 298.15 | 3.31 × 10−17 | 3.31 × 10−17 | 5.20 × 10−17 |
| 300 | 3.56 × 10−17 | 3.56 × 10−17 | 5.54 × 10−17 |
| 350 | 1.92 × 10−16 | 1.91 × 10−16 | 2.50 × 10−16 |
| 400 | 7.21 × 10−16 | 7.12 × 10−16 | 8.35 × 10−16 |
| 500 | 5.16 × 10−15 | 5.00 × 10−15 | 5.19 × 10−15 |
| 600 | 2.13 × 10−14 | 2.03 × 10−14 | 1.98 × 10−14 |
| T(K) | TST | CVT | CVT/μOMT | |||||
|---|---|---|---|---|---|---|---|---|
| 100 | 10.4 | 9.34 | 1.95 × 103 | 3.63 | 0.897 | 209 | ||
| 150 | 5.39 | 5.06 | 159 | 1.88 | 0.939 | 31.4 | ||
| 200 | 3.80 | 3.64 | 10.7 | 1.32 | 0.958 | 2.95 | ||
| 250 | 3.04 | 2.95 | 3.91 | 1.06 | 0.969 | 1.33 | ||
| 298.15 | 2.61 | 2.55 | 2.84 | 1.27 | 2.26 | 0.909 | 0.976 | 1.12 |
| 300 | 2.60 | 2.53 | 2.82 | 0.904 | 0.976 | 1.11 | ||
| 350 | 2.31 | 2.26 | 2.37 | 0.803 | 0.980 | 1.05 | ||
| 400 | 2.10 | 2.03 | 2.11 | 0.731 | 0.984 | 1.02 | ||
| 500 | 1.82 | 1.80 | 1.80 | 0.635 | 0.989 | 0.998 | ||
| 600 | 1.65 | 1.64 | 1.62 | 0.575 | 0.991 | 0.991 |
| T(K) | TST | CVT | CVT/μOMT | |||||
|---|---|---|---|---|---|---|---|---|
| 100 | 17.0 | 12.2 | 1.00 × 103 | 8.86 | 0.714 | 82.8 | ||
| 150 | 7.13 | 5.92 | 137.5 | 3.71 | 0.831 | 23.2 | ||
| 200 | 4.55 | 4.06 | 14.0 | 2.37 | 0.894 | 3.45 | ||
| 250 | 3.44 | 3.20 | 5.03 | 1.79 | 0.931 | 1.57 | ||
| 298.15 | 2.85 | 2.72 | 3.42 | 1.17 | 1.64 | 1.49 | 0.953 | 1.26 |
| 300 | 2.84 | 2.71 | 3.38 | 1.48 | 0.954 | 1.25 | ||
| 350 | 2.46 | 2.39 | 2.71 | 1.28 | 0.970 | 1.14 | ||
| 400 | 2.20 | 2.16 | 2.33 | 1.15 | 0.980 | 1.08 | ||
| 500 | 1.88 | 1.87 | 1.91 | 0.979 | 0.992 | 1.02 | ||
| 600 | 1.69 | 1.68 | 1.68 | 0.878 | 0.998 | 0.999 |
| T(K) | TST | CVT | CVT/μOMT | |||||
|---|---|---|---|---|---|---|---|---|
| 100 | 1.23 | 1.24 | 6.40 | 1.13 | 1.00 | 5.18 | ||
| 150 | 1.16 | 1.16 | 4.57 | 1.07 | 0.999 | 3.94 | ||
| 200 | 1.12 | 1.12 | 2.09 | 1.03 | 0.998 | 1.87 | ||
| 250 | 1.10 | 1.10 | 1.35 | 1.01 | 0.997 | 1.23 | ||
| 298.15 | 1.08 | 1.08 | 1.19 | 1.03 | 1.05 | 0.997 | 0.997 | 1.10 |
| 300 | 1.08 | 1.08 | 1.19 | 0.997 | 0.997 | 1.10 | ||
| 350 | 1.07 | 1.07 | 1.14 | 0.987 | 0.997 | 1.06 | ||
| 400 | 1.06 | 1.06 | 1.11 | 0.980 | 0.997 | 1.04 | ||
| 500 | 1.06 | 1.05 | 1.08 | 0.971 | 0.998 | 1.03 | ||
| 600 | 1.05 | 1.05 | 1.07 | 0.967 | 0.998 | 1.02 |
| T(K) | TST | CVT | CVT/μOMT | |||||
|---|---|---|---|---|---|---|---|---|
| 100 | 1.23 | 1.27 | 10.1 | 1.11 | 1.04 | 7.91 | ||
| 150 | 1.16 | 1.19 | 5.84 | 1.05 | 1.03 | 4.92 | ||
| 200 | 1.12 | 1.14 | 2.15 | 1.01 | 1.02 | 1.89 | ||
| 250 | 1.10 | 1.11 | 1.42 | 0.991 | 1.01 | 1.27 | ||
| 298.15 | 1.08 | 1.09 | 1.24 | 1.05 | 1.06 | 0.978 | 1.01 | 1.14 |
| 300 | 1.08 | 1.09 | 1.24 | 0.977 | 1.01 | 1.14 | ||
| 350 | 1.07 | 1.08 | 1.17 | 0.968 | 1.01 | 1.08 | ||
| 400 | 1.06 | 1.07 | 1.13 | 0.961 | 1.01 | 1.06 | ||
| 500 | 1.06 | 1.06 | 1.09 | 0.953 | 1.00 | 1.03 | ||
| 600 | 1.05 | 1.05 | 1.08 | 0.948 | 1.00 | 1.02 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




