Submitted:
26 November 2025
Posted:
28 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sample Preparation
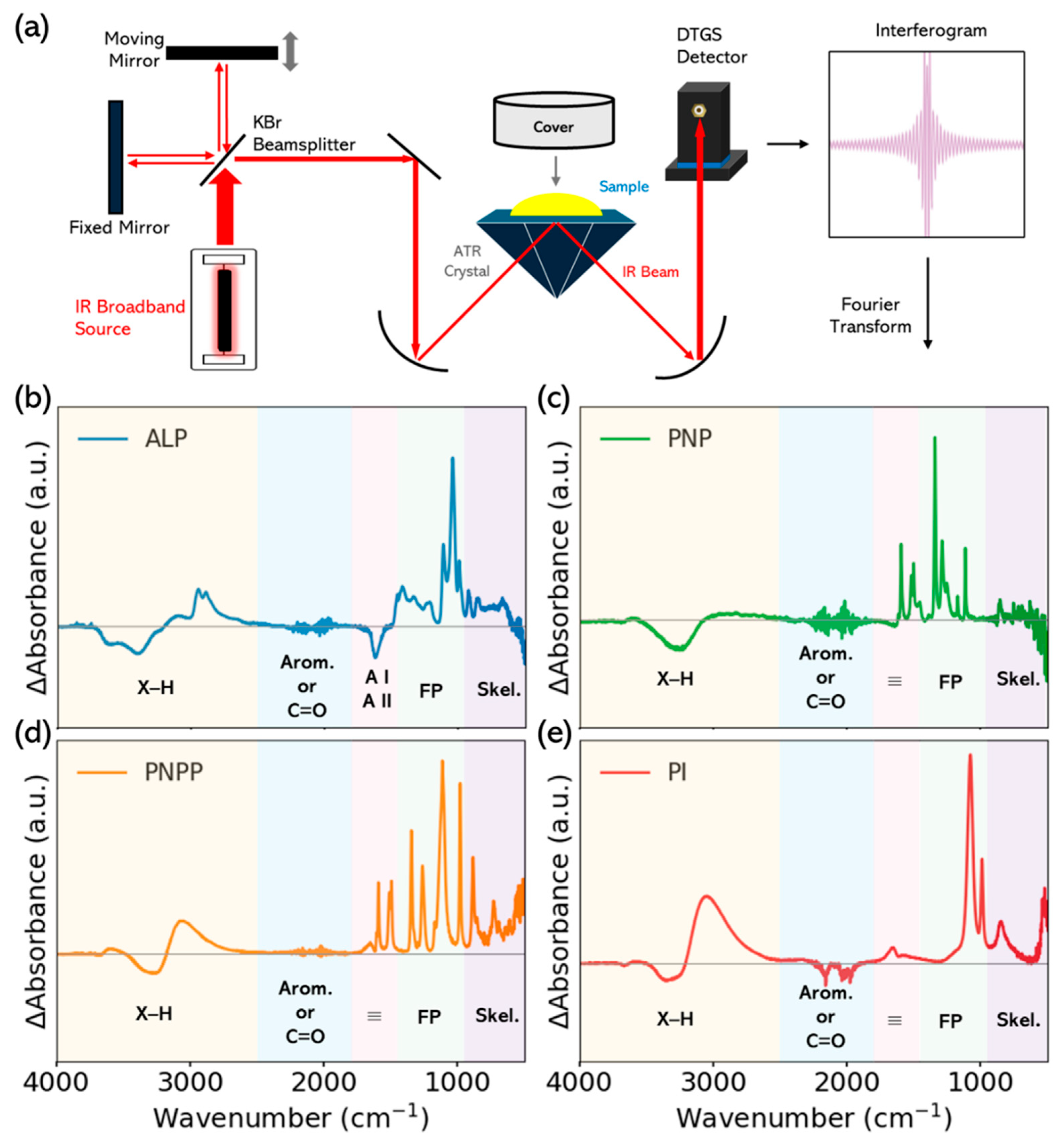
2.3. FTIR Measurements
2.4. Data Analysis
3. Results
3.1. Static Absorbance of ALP, PNPP, PNP and Pi
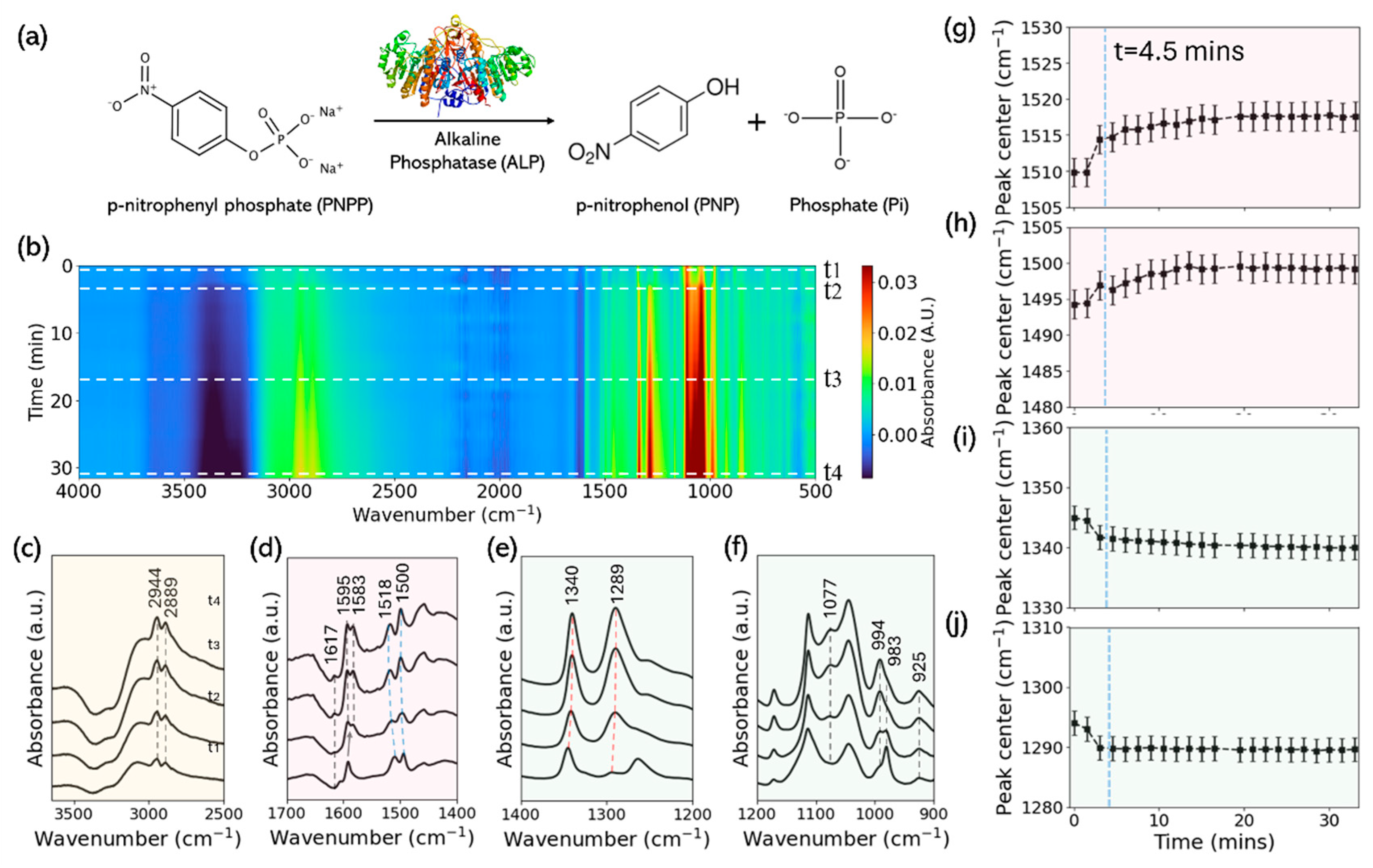
3.2. In Situ Spectra of PNPP Reaction Under High ALP
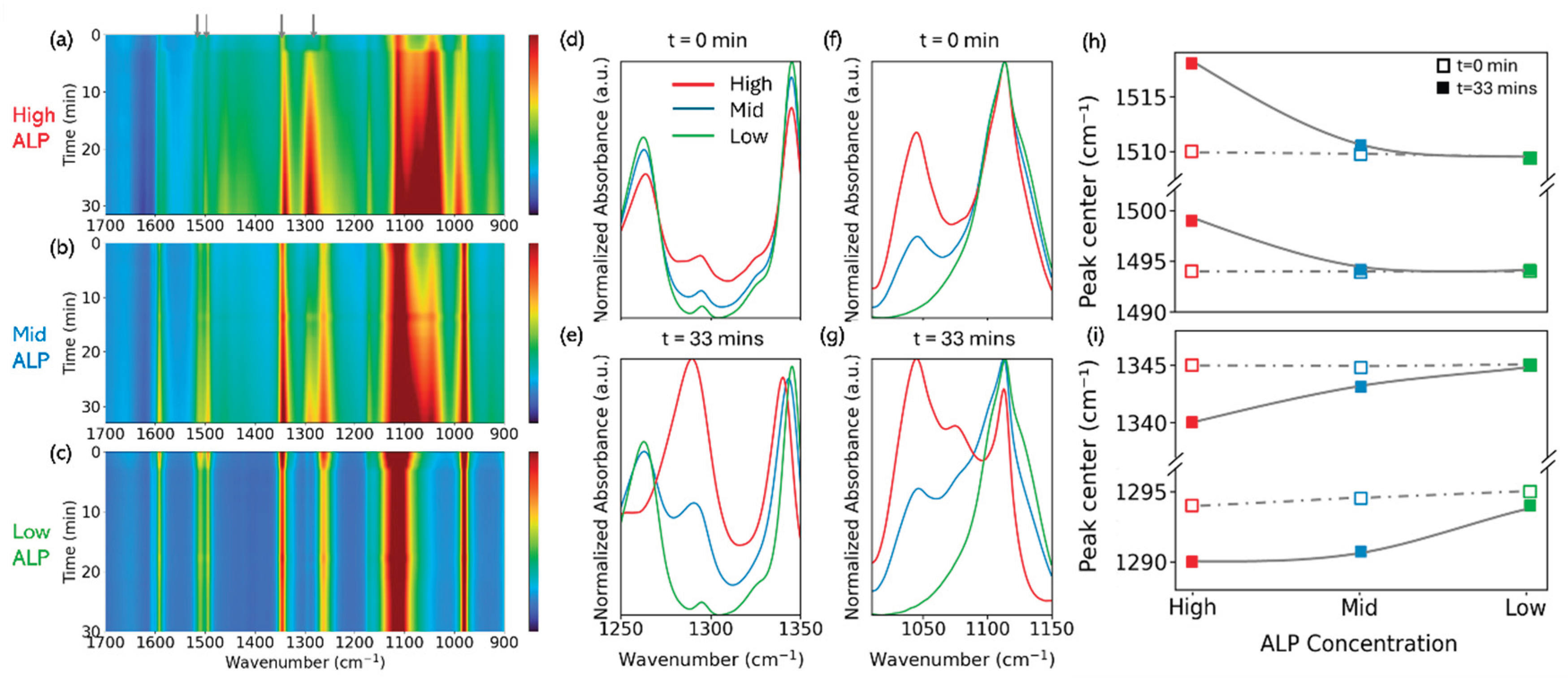
3.3. ALP Concentration-Dependence
4. Discussion
5. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
References
- Coleman, J.E. Structure and Mechanism of Alkaline Phosphatase. Annual Review of Biophysics and Biomolecular Structure 1992, 21, 441–483. [Google Scholar] [CrossRef] [PubMed]
- Harris, H. The Human Alkaline Phosphatases: What We Know and What We Don’t Know. Clinica Chimica Acta 1990, 186, 133–150. [Google Scholar] [CrossRef]
- Harris, H. The Harvey Lectures: Series 76; Academic Press: New York, 1986; pp. 95–123. [Google Scholar]
- Millán, J.L. The Role of Phosphatases in the Initiation of Skeletal Mineralization. Calcified Tissue International 2013, 93, 299–306. [Google Scholar] [CrossRef]
- Millán, J.L.; Whyte, M.P. Alkaline Phosphatase and Hypophosphatasia. Calcified Tissue International 2016, 98, 398–416. [Google Scholar] [CrossRef]
- Makris, K.; Mousa, C.; Cavalier, E. Alkaline phosphatases: Biochemistry, functions, and measurement. Calcif. Tissue Int. 2023, 112, 233–242. [Google Scholar] [CrossRef]
- Minisola, S.; Cipriani, C.; Colangelo, L.; Labbadia, G.; Pepe, J.; Magnusson, P. Diagnostic Approach to Abnormal Alkaline Phosphatase Value. Mayo Clinic Proceedings 2025, 100, 712–728. [Google Scholar] [CrossRef]
- Sharma, U.; Pal, D.; Prasad, R. Alkaline phosphatase: an overview. Indian journal of clinical biochemistry, 2014, 29, 269–278. [Google Scholar] [CrossRef]
- McKenna, M.J.; Hamilton, T.A.; Sussman, H.H. Comparison of human alkaline phosphatase isoenzymes: structural evidence for three protein classes. Biochem. J. 1979, 181, 67–73. [Google Scholar] [CrossRef]
- Mornet, E.; Stura, E.; Lia-Baldini, A.-S.; Stigbrand, T.; Ménez, A.; Le Du, M.-H. Structural evidence for a functional role of human tissue nonspecific alkaline phosphatase in bone mineralization. J. Biol. Chem. 2001, 276, 31171–31178. [Google Scholar] [CrossRef]
- Drechsler, C.; Verduijn, M.; Pilz, S.; Krediet, R.T.; Dekker, F.W.; Wanner, C.; Ketteler, M.; Boeschoten, E.W.; Brandenburg, V. Bone Alkaline Phosphatase and Mortality in Dialysis Patients. Clin. J. Am. Soc. Nephrol. 2011, 6, 1752–1759. [Google Scholar] [CrossRef]
- Haarhaus, M.; Monier-Faugere, M.-C.; Magnusson, P.; Malluche, H.H. Bone Alkaline Phosphatase Isoforms in Hemodialysis Patients with Low versus Non-Low Bone Turnover: A Diagnostic Test Study. Am. J. Kidney Dis. 2015, 65, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Roston, D.; Cui, Q. Substrate and Transition State Binding in Alkaline Phosphatase Analyzed by Computation of Oxygen Isotope Effects. J. Am. Chem. Soc. 2016, 138, 11946–11957. [Google Scholar] [CrossRef]
- Igunnu, A.; Singh, J.; Flanagan, J.U. Distinct Metal Ion Requirements for the Phosphomonoesterase and Phosphodiesterase Activities of Calf Intestinal Alkaline Phosphatase. Open Biochem. J. 2011, 5, 67–75. [Google Scholar] [CrossRef]
- Marhuenda-Egea, F.C.; Piera-Velázquez, S.; Cadenas, C.; Cadenas, E. Kinetic Regulation of an Alkaline p-Nitrophenylphosphate Phosphatase from Halobacterium salinarum in Low Water System by Mn2+ and Monovalent Cations. FEMS Microbiol. Lett. 2001, 198, 111–115. [Google Scholar] [CrossRef]
- Holtz, K.M.; Kantrowitz, E.R. The mechanism of the alkaline phosphatase reaction: insights from NMR, crystallography and site-specific mutagenesis. FEBS Lett. 1999, 462, 7–11. [Google Scholar] [CrossRef]
- Dean, R.L. Kinetic studies with alkaline phosphatase in the presence and absence of inhibitors and divalent cations. Biochem. Mol. Biol. Educ. 2002, 30, 401–407. [Google Scholar] [CrossRef]
- Barth, A. Infrared Spectroscopy of Proteins. Biochim. Biophys. Acta Bioenerg. 2007, 1767, 1073–1101. [Google Scholar] [CrossRef]
- Barth, A.; Zscherp, C. What Vibrations Tell about Proteins. Q. Rev. Biophys. 2002, 35, 369–430. [Google Scholar] [CrossRef] [PubMed]
- Jackson, M.; Mantsch, H.H. The Use and Misuse of FTIR Spectroscopy in the Determination of Protein Structure. Crit. Rev. Biochem. Mol. Biol. 1995, 30, 95–120. [Google Scholar] [CrossRef] [PubMed]
- Goormaghtigh, E.; Ruysschaert, J.-M.; Raussens, V. Evaluation of the Information Content in Infrared Spectra for Protein Secondary Structure Determination. Biophys. J. 2006, 90, 2946–2957. [Google Scholar] [CrossRef]
- Kumar, S.; Barth, A. Following Enzyme Activity with Infrared Spectroscopy. Sensors 2010, 10, 2626–2637. [Google Scholar] [CrossRef] [PubMed]
- Fabian, H.; Mäntele, W. Infrared spectroscopy of proteins. In Handbook of Vibrational Spectroscopy; John Wiley & Sons: Chichester, 2002. [Google Scholar] [CrossRef]
- Shi, H.; Lercher, J.A.; Yu, X. Sailing into uncharted waters: recent advances in the in situ monitoring of catalytic processes in aqueous environments. Catal. Sci. Technol. 2015, 5, 3035–3060. [Google Scholar] [CrossRef]
- Montis, C.; Berti, D. Controlling the kinetics of an enzymatic reaction through enzyme or substrate confinement into lipid mesophases with tunable structural parameters. Int. J. Mol. Sci. 2020, 21, 5116. [Google Scholar] [CrossRef]
- Zhao, J.; Cui, J.-K.; Chen, R.-X.; Tang, Z.-Z.; Tan, Z.-L.; Jiang, L.-Y.; Liu, F. Real-time in-situ quantification of protein secondary structures in aqueous solution based on ATR-FTIR subtraction spectrum. Biochem. Eng. J. 2021, 176, 108225. [Google Scholar] [CrossRef]
- Lin-Vien, D.; Colthup, N.B.; Fateley, W.G.; Grasselli, J.G. The Handbook of Infrared and Raman Characteristic Frequencies of Organic Molecules; Academic Press: San Diego, CA, USA, 1991. [Google Scholar]
- Ferraro, J.R.; Nakamoto, K.; Brown, C.W. Introductory Raman Spectroscopy, 2nd ed.; Academic Press: Burlington, MA, USA, 2003. [Google Scholar]
- Nakamoto, K. Infrared and Raman Spectra of Inorganic and Coordination Compounds, 6th ed.; Wiley: Hoboken, NJ, USA, 2009. [Google Scholar]
- Movasaghi, Z.; Rehman, S.; Rehman, I.U. Fourier Transform Infrared (FTIR) Spectroscopy of Biological Tissues. Appl. Spectrosc. Rev. 2008, 43, 134–179. [Google Scholar] [CrossRef]
- Ji, Y. , Yang, X., Ji, Z., Zhu, L., Ma, N., Chen, D., Jia, X., Tang, J., Cao, Y. DFT-calculated IR spectrum amide I, II, and III band contributions of N-methylacetamide fine components. ACS omega 2020, 5, 8572–8578. [Google Scholar] [CrossRef] [PubMed]
- Usoltsev, D.; Sitnikova, V.; Kajava, A.; Uspenskaya, M. Systematic FTIR Spectroscopy Study of the Secondary Structure Changes in Human Serum Albumin under Various Denaturation Conditions. Biomolecules 2019, 9, 359. [Google Scholar] [CrossRef] [PubMed]
- Bonnin, S.; Besson, F.; Gelhausen, M.; Chierici, S.; Roux, B. A FTIR Spectroscopy Evidence of the Interactions between Wheat Germ Agglutinin and N-Acetylglucosamine Residues. FEBS Lett. 1999, 456, 361–364. [Google Scholar] [CrossRef]
- WebSpectra. IR Absorption Table. 2000. Available online: https://webspectra.chem.ucla.edu/irtable.html (accessed on 1 October 2025).
- Vidal, B. de C.; Mello, M.L.S. Collagen Type I Amide I Band Infrared Spectroscopy. Micron 2011, 42, 283–289. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, K.; Li, Y.; Feng, Q. Fourier transform infrared spectroscopy analysis of the active components in serum of rats treated with Zuogui Pill. J. Tradit. Chin. Med. Sci. 2015, 2, 264–269. [Google Scholar] [CrossRef]
- Smith, B.C. The carbonyl group, part V: Carboxylates—coming clean. Spectroscopy Online 2018. Available at: https://www.spectroscopyonline.
- Chia, N.C.; Mendelson, R. Conformational disorder in unsaturated phospholipids by FTIR spectroscopy. Biochim. Biophys. Acta 1996, 1283, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Jastrzębski, W.; Sitarz, M.; Rokita, M.; Bułat, K. Infrared spectroscopy of different phosphates structures. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2011, 79, 722–727. [Google Scholar] [CrossRef]
- Linstrom, P.J.; Mallard, W.G. (Eds.) NIST Chemistry WebBook, NIST Standard Reference Database Number 69; National Institute of Standards and Technology: Gaithersburg, MD, USA. Available online: https://webbook.nist.gov/chemistry/ (accessed on 1 October 2025).
- OrgChemBoulder. IR Spectroscopy Tutorial; University of Colorado Boulder, Department of Chemistry and Biochemistry. Available online: https://orgchemboulder.com/Spectroscopy/irtutor/tutorial.shtml (accessed on 1 October 2025).
- LibreTexts. Infrared Spectroscopy. Chemistry LibreTexts. Available online: https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Supplemental_Modules_%28Physical_and_Theoretical_Chemistry%29/Spectroscopy/Vibrational_Spectroscopy/Infrared_Spectroscopy (accessed on 1 October 2025).
- Socrates, G. Infrared and Raman Characteristic Group Frequencies: Tables and Charts, 3rd ed.; Wiley: Chichester, UK, 2001. [Google Scholar]
- Larkin, P. Infrared and Raman Spectroscopy: Principles and Spectral Interpretation, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Abkowicz-Bieńko, A.J.; Latajka, Z.; Bieńko, D.C.; Michalska, D. Theoretical infrared spectrum and revised assignment for para-nitrophenol: density functional theory studies. Chem. Phys. 1999, 250, 123–129. [Google Scholar] [CrossRef]
- Koleva, V.; Stefov, V.; Cahil, A.; Najdoski, M.; Šoptrajanov, B.; Engelen, B.; Lutz, H.-D. Infrared and Raman studies of manganese dihydrogen phosphate dihydrate, Mn(H2PO4)2·2H2O. Part I: Region of the vibrations of the phosphate ions and external modes of the water molecules. J. Mol. Struct. 2009, 917, (2–3). [Google Scholar] [CrossRef]
- Sun, S.T.; et al. Microhydrated dihydrogen phosphate clusters probed by gas phase vibrational spectroscopy and first principles calculations. Phys. Chem. Chem. Phys. 2015, 17, 25714–25724. [Google Scholar] [CrossRef] [PubMed]
| Peak position (cm−1) |
Assignment | Bond description |
|---|---|---|
| 3398 | Amide A | N–H stretching (hydrogen-bond sensitive) |
| 2944 | C–H stretch | Alkyl side chains |
| 2889 | C–H stretch | Alkyl side chains |
| 1708 | Amide I (β-turn) | C=O stretching + H-bonding |
| 1624 | Amide I (β-sheet) | C=O stretching, intermolecular β-sheet |
| 1551 | Amide II | NH bending + CN stretching |
| 1531 | Amide II | NH bending + CN stretching |
| 1459 | CH2 scissoring | CH2 bending vibrations |
| 1418 | COO− symmetric stretch | Carboxylates |
| 1334 | CH2 wagging | Side-chain CH2 vibration |
| 1215 | Amide III | NH bending + CN stretching (H-bond sensitive) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





