Submitted:
24 November 2025
Posted:
26 November 2025
You are already at the latest version
Abstract
Background And Objective: Changes in female sex hormone levels are closely linked to the development and progression of uterine fibroids (UFs). Clinical approaches to fibroid management vary according to guidelines and depend on patient symptoms, fibroid size, and clinician judgment. Despite available diagnostic tools, surgical decisions remain largely subjective. With the advancement of artificial intelligence (AI) and clinical decision support technologies, clinical experience can now be transferred into data-driven computational models trained with hormone-based parameters. To develop a clinical decision support algorithm that predicts surgical necessity for uterine fibroids by integrating fibroid characteristics and female sex hormone levels. Methods: This multicenter study included 618 women with UFs who presented to three hospitals; 238 underwent surgery. Statistical analyses and artificial intelligence–based modeling were performed to compare surgical and non-surgical groups. Training was conducted with each hormone—follicle-stimulating hormone (FSH), luteinizing hormone (LH), estrogen (E2), prolactin (PRL), and anti-Müllerian hormone (AMH)—and with 126 input combinations including hormonal and morphological variables. Five supervised learning algorithms—support vector machine, decision tree, random forest, and k-nearest neighbors—were applied, resulting in 630 trained models. In addition to this retrospective development phase, a prospective validation was conducted in which 20 independent clinical cases were evaluated in real time by a gynecologist blinded to both the model predictions and the surgical outcomes. Agreement between the clinician’s assessments and the model outputs was measured. Results: FSH, LH, and PRL levels were significantly lower in the surgery group (p < 0.001, 0.009, and < 0.001, respectively), while E2 and AMH were higher (p = 0.012 and 0.001). Fibroid volume was also greater among surgical cases (90.8 cc vs. 73.1 cc, p < 0.001). The random forest model using LH, FSH, E2, and AMH achieved the highest accuracy of 91 percent. In the external validation phase, the model’s predictions matched the blinded gynecologist’s decisions in 18 of 20 cases, corresponding to a 90% concordance rate. The two discordant cases were later identified as borderline scenarios with clinically ambiguous surgical indications. Conclusion: The decision support algorithm integrating hormonal and fibroid parameters offers an objective and data-driven approach to predicting surgical necessity in women with UFs. Beyond its strong internal performance metrics, the model demonstrated a high level of clinical concordance during external validation, achieving a 90% agreement rate with an independent, blinded gynecologist. This alignment underscores the model’s practical reliability and its potential to reduce subjective variability in surgical decision-making. By providing a reproducible and clinically consistent framework, the proposed AI-based system represents a meaningful advancement toward the validated integration of computational decision tools into routine gynecological practice.
Keywords:
Introduction
Materials and Methods
Ethical Consideration
Patients
Study Design
Validation Procedure
Statistical Analyses and Tools
Machine Learning Procedure and Pipeline
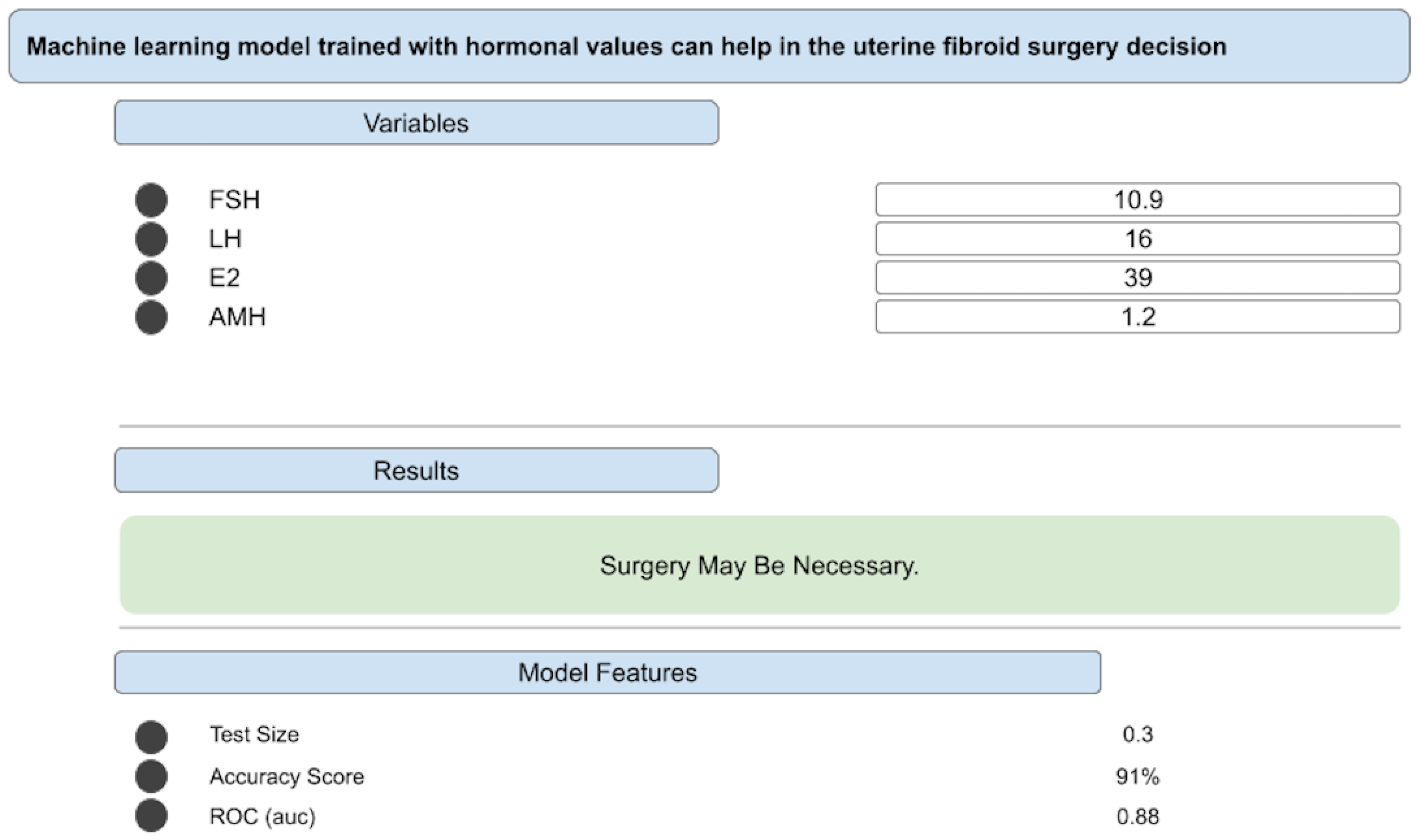
Deploying The Best Decision Support Algorithm on “JinekoAI.com” Web Application
Results
Statistical Results
Machine Learning Model Training Results
External Validation Results
Discussion
Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflict of Interest
Abbreviations
| AI | Artificial intelligence |
| AMH | Anti-Müllerian hormone |
| AUC | Area under the ROC curve |
| cc | Cubic centimeter |
| DT | Decision tree |
| E2 | Estradiol |
| FSH | Follicle-stimulating hormone |
| GnRH | Gonadotropin-releasing hormone |
| KNN | k-nearest neighbors |
| LH | Luteinizing hormone |
| LR | Logistic regression |
| ML | Machine learning |
| PRL | Prolactin |
| RF | Random forest |
| SVM | Support vector machine |
| UF | Uterine fibroid |
| UFs | Uterine fibroids |
| WAI | WisdomEra Artificial Intelligence |
References
- Yang Q, Ciebiera M, Bariani MV, Ali M, Elkafas H, Boyer TG, et al. Comprehensive Review of Uterine Fibroids: Developmental Origin, Pathogenesis, and Treatment. Endocr Rev 2022;43:678–719. [CrossRef]
- Giuliani E, As-Sanie S, Marsh EE. Epidemiology and management of uterine fibroids. Int J Gynaecol Obstet Off Organ Int Fed Gynaecol Obstet 2020;149:3–9. [CrossRef]
- Tinelli A, Morciano A, Sparic R, Hatirnaz S, Malgieri LE, Malvasi A, et al. Artificial Intelligence and Uterine Fibroids: A Useful Combination for Diagnosis and Treatment. J Clin Med 2025;14:3454. [CrossRef]
- Ahmad A, Kumar M, Bhoi NR, Badruddeen null, Akhtar J, Khan MI, et al. Diagnosis and management of uterine fibroids: current trends and future strategies. J Basic Clin Physiol Pharmacol 2023;34:291–310. [CrossRef]
- Micić J, Macura M, Andjić M, Ivanović K, Dotlić J, Micić DD, et al. Currently Available Treatment Modalities for Uterine Fibroids. Med Kaunas Lith 2024;60:868. [CrossRef]
- Uterine fibroids: Learn More – Hormone treatments for uterine fibroids. Inf. Internet, Institute for Quality and Efficiency in Health Care (IQWiG); 2025.
- Di Spiezio Sardo A, Ciccarone F, Muzii L, Scambia G, Vignali M. Use of oral GnRH antagonists combined therapy in the management of symptomatic uterine fibroids. Facts Views Vis ObGyn n.d.;15:29–33. [CrossRef]
- Mills S. Electronic Health Records and Use of Clinical Decision Support. Crit Care Nurs Clin North Am 2019;31:125–31. [CrossRef]
- Vetrivel S, Rexline D, Gowri MsTD. Decision Support Tool for Uterine Fibroids Treatment with Machine Learning Algorithms – A Study. Int J Sci Res Publ IJSRP 2022;12:442–9. [CrossRef]
- Wisdomera.io. wisdomera.io n.d. https://wisdomera.io/dashboard (accessed June 22, 2025).
- jinekoai.com n.d. https://jinekoai.com/dashboard (accessed June 27, 2025).
- jinekoai | /surgery-estimate-by-hormone-value-myoma-count n.d. https://jinekoai.com/surgery-estimate-by-hormone-value-myoma-count (accessed July 9, 2025).
- Vannuccini S, Petraglia F, Carmona F, Calaf J, Chapron C. The modern management of uterine fibroids-related abnormal uterine bleeding. Fertil Steril 2024;122:20–30. [CrossRef]
- Li Q, Zhong J, Yi D, Deng G, Liu Z, Wang W. Assessing the risk of rapid fibroid growth in patients with asymptomatic solitary uterine myoma using a multivariate prediction model. Ann Transl Med 2021;9:370. [CrossRef]
- Commandeur AE, Styer AK, Teixeira JM. Epidemiological and genetic clues for molecular mechanisms involved in uterine leiomyoma development and growth. Hum Reprod Update 2015;21:593–615. [CrossRef]
- Luo N, Guan Q, Zheng L, Qu X, Dai H, Cheng Z. Estrogen-mediated activation of fibroblasts and its effects on the fibroid cell proliferation. Transl Res 2014;163:232–41. [CrossRef]
- Polat G, Arslan HK. Artificial Intelligence in Clinical and Surgical Gynecology. İstanbul Gelişim Üniversitesi Sağlık Bilim Derg 2024:1232–41. [CrossRef]
- Huo T, Li L, Chen X, Wang Z, Zhang X, Liu S, et al. Artificial intelligence-aided method to detect uterine fibroids in ultrasound images: a retrospective study. Sci Rep 2023;13:3714. [CrossRef]
- Wright DE, Gregory AV, Anaam D, Yadollahi S, Ramanathan S, Oyemade KA, et al. Developing a Machine Learning-Based Clinical Decision Support Tool for Uterine Tumor Imaging 2023. [CrossRef]
- Chen M, Kong W, Li B, Tian Z, Yin C, Zhang M, et al. Revolutionizing hysteroscopy outcomes: AI-powered uterine myoma diagnosis algorithm shortens operation time and reduces blood loss. Front Oncol 2023;13:1325179. [CrossRef]
| Surgery Mean / % |
Non-Surgery Mean / % |
p | |
|---|---|---|---|
| Patient N, % | 238, (38.5) | 380, (61.5) | |
| Age | 35.7 | 35.4 | 0.613 |
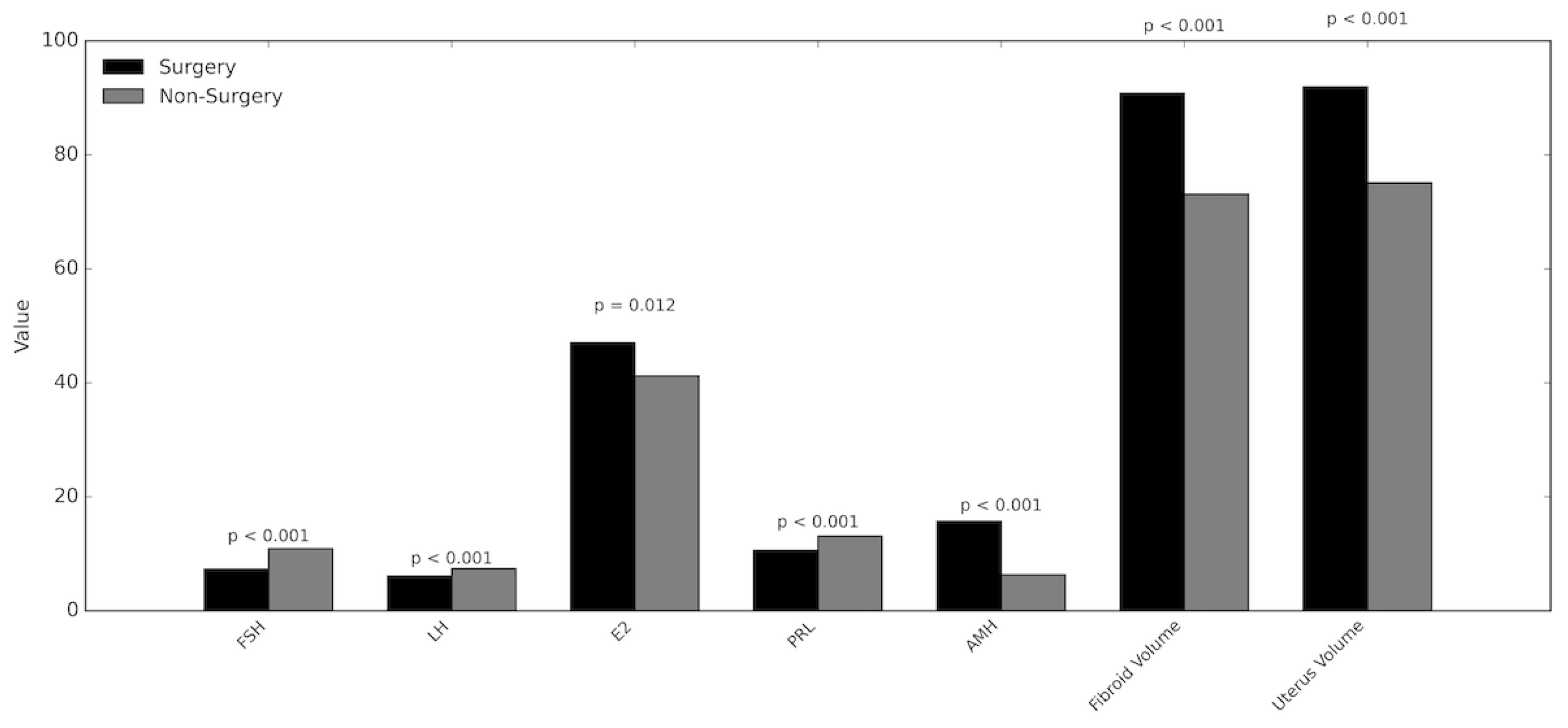
| FSH (mIU/mL) | 7.3 | 10.9 | < 0.001 |
| LH (mIU/mL) | 6.1 | 7.4 | < 0.001 |
| E2 (mIU/mL) | 47 | 41.2 | 0.012 |
| PRL (µg/L) | 10.6 | 13.1 | < 0.001 |
| AMH (ng/mL) | 15.7 | 6.3 | < 0.001 |
| Fibroid number | 4.7 | 4.6 | 0.384 |
| Fibroid volume (cc) | 90.8 | 73.1 | < 0.001 |
| Uterus volume (cc) | 91.9 | 75.1 | < 0.001 |
|
Disease Duration (years) 1-5 > 5 |
126 (53) 112 (47) |
216 (57) 164 (43) |
0.361 |
| Inputs | model | accuracy | roc | precision | recall | f score |
|---|---|---|---|---|---|---|
| LH, FSH, E2, AMH | RF | 0.91 | 0.88 | 0.91 | 0.91 | 0.91 |
| LH, FSH, E2, AMH | KNN | 0.86 | 0.84 | 0.86 | 0.86 | 0.86 |
| LH, FSH, PRL, E2, AMH | RF | 0.86 | 0.84 | 0.86 | 0.86 | 0.86 |
| LH, FSH, E2, UF number | RF | 0.85 | 0.82 | 0.85 | 0.85 | 0.85 |
| LH, FSH, E2, UF number | KNN | 0.85 | 0.83 | 0.85 | 0.85 | 0.85 |
| LH, FSH, E2, UF volume | RF | 0.85 | 0.82 | 0.85 | 0.85 | 0.84 |
| LH, FSH, PRL, E2, AMH | KNN | 0.85 | 0.83 | 0.85 | 0.85 | 0.85 |
| LH, FSH, E2, AMH, UF number | RF | 0.85 | 0.82 | 0.85 | 0.85 | 0.85 |
| LH, FSH, E2, AMH, UF number | KNN | 0.85 | 0.83 | 0.85 | 0.85 | 0.85 |
| LH, FSH, E2, AMH, UF volume | RF | 0.85 | 0.82 | 0.85 | 0.85 | 0.85 |
| LH, FSH, E2, UF volume, UF number | RF | 0.85 | 0.81 | 0.85 | 0.85 | 0.84 |
| LH, FSH, PRL, E2, AMH, UF number | RF | 0.85 | 0.82 | 0.85 | 0.85 | 0.85 |
| Accuracy Ratio Group | Count |
|---|---|
| >90% | 1 |
| 80-90% | 136 |
| 70-80% | 154 |
| 60-70% | 220 |
| 50-60% | 117 |
| <50% | 2 |
| Total | 630 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




