1. Introduction
Stress in animals is a complex physiological reaction with far-reaching implications for animal welfare, scientific research validity (Bailey, 2018), and ethical considerations (Baumans, 2004). Stress refers to the physiological and psychological responses to environmental challenges. Stress is differentiated in eustress and distress (Selye, 1975). Eustress describes a positive emotional state associated with activation and engagement. Distress is a destructive type of stress and reflects a state of negative emotional arousal associated with dissatisfaction and disengagement (Pluut et al., 2022). In contrast to eustress associated with successful coping strategies, distress occurs when animal face a stressor exceeding their coping resources.
In animal experiments under the framework outlined in Annex VIII of the EU 2010/63 directive, distress is relevant for classification of the experiment in the four severity categories "non-recovery", "mild", "moderate", and "severe". These categories help researchers to assess the severity of their experiment and to address the impact on the welfare of laboratory animals. In the EU in 2022, almost half (3,921,024) of all animals used in experiments experienced stress of mild severity (European Commission, 2024). Examples for procedures that are assessed with mild severity are administration of anesthesia, non-invasive imaging or short-term deprivation of social partner (Directive 2010/63/EU). A mild severity can still result in short term stress for an animal and that impacts animal well-being. For example, even routine procedures such as blood sampling, as a part in many research protocols, may cause stress in animals (Balcombe et al., 2004).
Blood sampling or venepuncture are standard procedures in animal experiments, e.g., for the detection of metabolic changes (Tosto et al., 2021) or environmental influences on the animals homeostasis (Li et al., 2024). Apart from experimental studies, “practices [that are] not likely to cause pain, suffering, distress or lasting harm equivalent to, or higher than, that caused by the introduction of a needle in accordance with good veterinary practice” can serve as a threshold in determining whether a procedure falls under the scope of the EU Directive 2010/63 and is therefore classified as an animal experiment requiring approval (Directive 2010/63/EU). In farm animals such as goats, blood sampling is commonly accompanied with restraining the animal. Therefore, not only the short/mild pain caused by punctuation of the skin but also restraining and short-term isolation may cause stress in the animal (Kruger et al., 2016, Price and Thos, 1980). When this procedure is repeated several times during a running experiment, negative experience can aggravate the affective state of animals (Wang et al., 2008, Imfeld-Mueller et al., 2011) or human patients (Bisogni et al., 2014) in this situation. In contrast, repetition of a stressful procedure such as transportation can reduce stress through habituation (Dai et al., 2020). This dichotomy also occurs in the severity assessment framework by the European Commission as it mentions that “there is no direct link between frequency and severity. […] When interventions are repeated, there is the potential for acclimatization, which may reduce severity, […]. Conversely, repetition may increase severity, e.g., due to anticipation of a stressful procedure […]” (European Commission, 2018). In conclusion, it is likely that a lower severity could take place when the acclimatization phase is optimized while the sensation of a procedure is minimized.
Assessment of stress of certain procedures in an experiment helps to categorize the severity of the experiment. Stress can be assessed with physiological and ethological parameters. Hydbring-Sandberg et al. (2022) highlighted, that stress measurement must be multifaceted to allow a reliable prediction. They measured and compared the stress reaction in goats in different handling situations (loose holding vs. tethered). Serum cortisol level did not differ between both groups, but other physiological parameters, such as blood pressure or heart rate were higher in the tethered group. However, the measurement of glucocorticoids as a crucial part of the hypothalamus-pituitary-adrenal (HPA) axis, is frequently used for stress evaluation (Kruger et al., 2016, Kannan et al., 2000). Cortisol, a glucocorticoid, serves as a reliable biomarker of stress reactions, yet its measurement may present challenges in interpretation and standardization especially when it comes to the assessment of animal welfare (Ralph and Tilbrook, 2016). Infrared thermography is promoted as a non- invasive method to evaluate stress in farm animals (Jerem et al., 2019, Arfuso et al., 2022, Bartolomé et al., 2019). By activation of the HPA axis and the sympathetic nervous system, peripheral vessels are constricted. This leads to a decrease in peripheral skin temperature e.g., on the ears of rabbits (Blessing, 1997) as well as an increase in eye region surface temperature (Stewart et al., 2007). Accordingly, Arfuso et al. (2022) found eye infrared thermography as a useful immediate and non-invasive physiological tool to assess stress response in sheep. This is in accordance with the study of Bartolomé et al. (2019). They described eye thermography to be an appropriate and non-invasive tool to explore stress levels in goats during routine management practices.
In addition, observing stress-related behaviors provide valuable insights into animal welfare. This is particularly relevant as it is difficult to distinguish distress and eustress as reaction to a mild aversive stimulus when compared with a rewarding scenario e.g., through food. Therefore, behavior responses, such as vocalization, or escaping attempts (Siebert et al., 2011, Briefer et al., 2015), can help to assess the emotional state of the animal. Siebert et al. (2011), observed that completely isolated goats exhibited lower activity levels, reared less frequently, and emitted fewer high bleats compared to partially isolated animals. In contrast the latter showed more active behaviors such as increased locomotion, rearing, jumping, and high bleats, and motivated attempts to re-establish social contact.
Due to our knowledge, there is a lack of information on the affective state of goats undergoing repeated blood sampling over a longer period. Filling that gap could help to refine severity assessment for animals in experimental settings. Therefore, this study observes the influence of repeated blood sampling on the affective state of the animals. We wanted to observe how repeated blood sampling would effect stress parameters in goats in a experiment with repeated blood sampling that underwent an intensive cooperative vet care training and were handled with focus on gentle handling.
2. Materials and Methods
The data were collected as part of a study on the transfer of per- and polyfluoroalkyl substances (PFAS) in hay to milk from dairy goats (transfer study; (Just et al., 2025)). This study was approved by the Competent Authority (Landesamt für Gesundheit und Soziales Berlin - LAGeSo) under the reference number G0050/22.
2.1. Animals and Housing
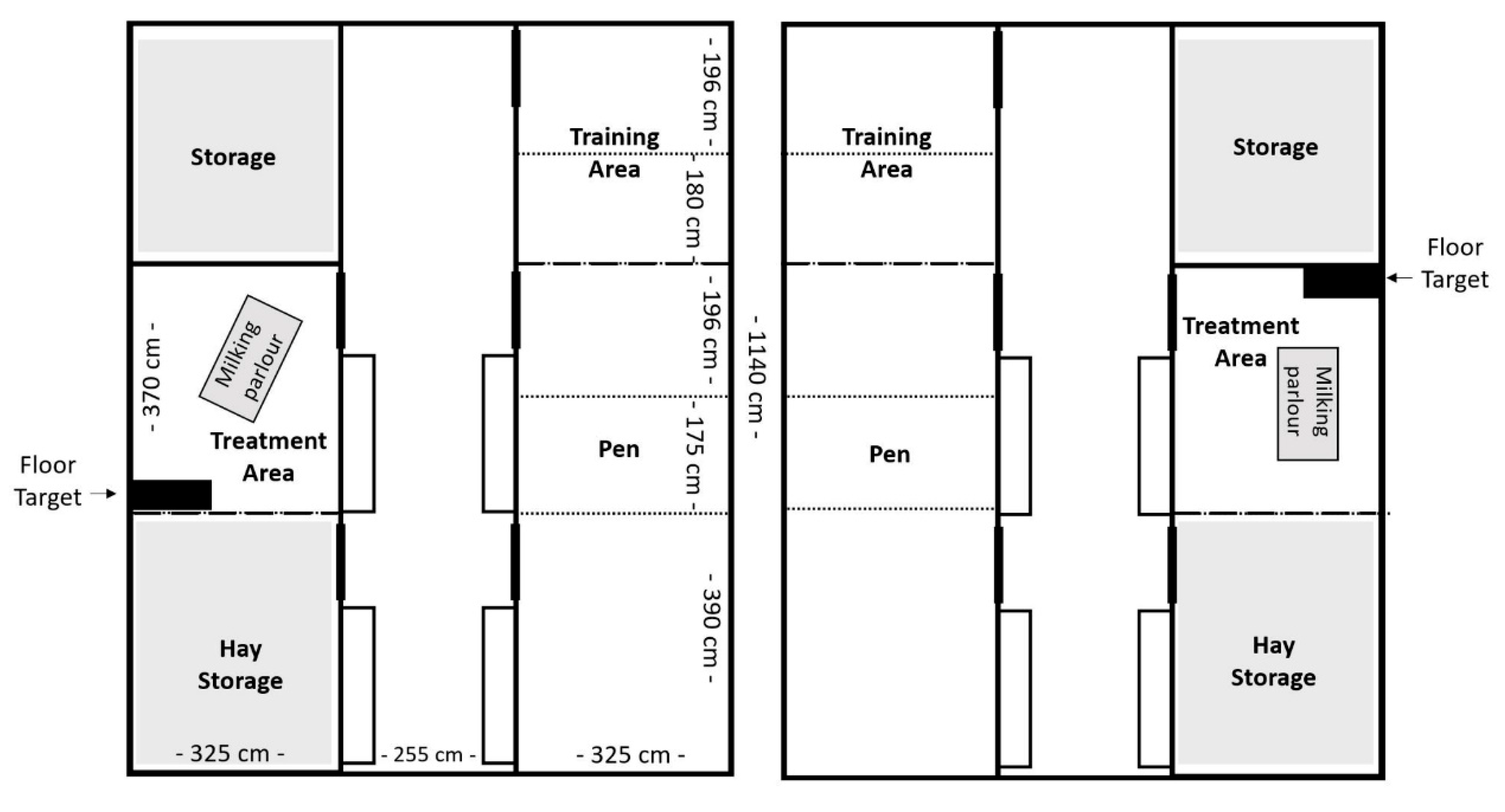
Ten lactating German Improved White female goats (capra aegagrus hircus; first or second lactation) were purchased from a commercial dairy goat farm and were included in the transfer experiment. They were housed at the German Federal Institute for Risk Assessment (BfR) for a period of 24 weeks (24th April 2023 to 9th October 2023). The goats were divided into two groups of five and housed in two separate pens under matched conditions. Each pen contained the housing pen, a training area, a hay storage area and a treatment area (
Figure 1). The housing pen had a floor area of 25 m² and was enriched with deep straw bedding and four to five platforms for the goats to jump on or seek shelter under. At weeks 5, 10 and 13 the housing pen was subdivided with mobile fencing, to house the goats individually and to collect goat-specific feces and urine samples for the PFAS transfer study. Data of four goats of each group (n=8) were used in this study, excluding the two surrogate animals, as backup. There were not used for blood sampling and date collection but remained in the group during the hole time. No goat had to be excluded during this study.
Figure 1.
Pen design for Group E (left) and Group C (right). Dotted lines: fences, only during weeks of individual housing.
Figure 1.
Pen design for Group E (left) and Group C (right). Dotted lines: fences, only during weeks of individual housing.
Goats had ad libitum access to hay in slow feeders (HeuToy, Udo Röck GmbH, Bad Saulgau, Germany), water, and mineral stones. Each goat received milk performance-oriented concentrate feed (M 18-4 Green pelleted, Agravis Ost GmbH & CO.KG, Fürstenwalde, GER) tailored to its milk yield and additionally during training or handling procedures as a reward. During the transfer study, five goats were consistently fed PFAS-free hay throughout the study (group C), while the remaining five goats were exposed to PFAS-contaminated hay for a period of time (Week 5 – 12; group E). All goats were checked daily for any signs of reduced general condition. No discernible adverse effects on the general health status of the goats were observed during the study. Yet, group E had on average a significant lower body weight (during the complete period) and lower milk yield (during PFAS contaminated hay exposure period) compared to group C.
The treatment pens (one in each barn, see Fig. 1) were located opposite of the housing pens and were measured approximately 12 m² in size. This area contained a milking parlour with a portable milking machine (Milk trolley for goats, Milkline, Italy), a table and, on days with blood sampling, a black rubber mat measuring 85 x 45 cm (floor target). Goats were individually let into the treatment pen for milking (1x daily at 1 p.m.), blood sampling (1-3x weekly) and positive reinforcement training (PRT). During milking, each goat received concentrated feed adjusted to milk yield.
2.2. Training
The ethical approval according to 2010/63/EU included a training protocol to prepare all goats for the blood sampling as a refinement measure, as part of the 3R concept to reduce stress during an experiment (Russell and Burch, 1959). Therefore, after two weeks of habituation all goats underwent positive reinforcement training to enhance voluntary behavior. In this study, PRT serves as an umbrella term for positive reinforces training methods such as cooperative vet care, husbandry training or clicker training and shaping when used to refine experimental procedures. Before arrival at the BfR, the goats had not been trained. The training started at week 3 and continued until week 17, conducted by JM. JM had 2 two years of specific experience in training goats with PRT in an experimental context. On average, each goat underwent eight training sessions of 13 minutes each. The training followed the principles of PRT, which means animals receive a reward (food) for a desired behavior, to enhance the probability that the rewarded behavior is repeated (Skinner, 1951). The initial phase of training was to habituate both groups in the housing pen to accept food from the hand. This was achieved after 120 minutes in Group C (in four Sessions) and 105 minutes (in three Sessions) in Group E. The next goal was to train each goat to follow a hand as a leading target (hand target) in the training area. For this, the goat had to touch the trainer’s presented hand with its nose. The distance of the trainer’s hand was increased during training, as a low-stress method of guiding the animal, for example to the training area. All goats learned to follow the hand target for at least 5 meters with minimal distraction. The floor target, represented by a black rubber mat (85 x 45 cm), was then introduced. Training with the floor target was conducted in both, the training and the treatment areas. The goats were trained to stand with all four claws on the target and maintain this position with minimal distraction. This helps to position the goats in a good position for handling with minimal physical restraint. The goats were then trained to tolerate a hand under the chin, a crucial position for blood sampling. The number of training sessions and the performance of each goat are listed in table 1. Achievements during the training sessions were: following a hand target for at least 5 meters (hand target), finding a position on floor target (with or without a supporting signal such as a hand target), voluntarily placing the chin on the trainer’s hand, tolerating of touches.
Table 1.
Summary of training sessions, latency and achievements. Hand target: following the hand target; Floor target: positioning all four claws on floor target; Touch: tolerating touch while maintaining position on the floor target; Chin on hand: placing the chin on the trainer’s hand, while remaining on the floor target.
Table 1.
Summary of training sessions, latency and achievements. Hand target: following the hand target; Floor target: positioning all four claws on floor target; Touch: tolerating touch while maintaining position on the floor target; Chin on hand: placing the chin on the trainer’s hand, while remaining on the floor target.
| Goat |
Training Sessions |
Duration (min) |
Achievement |
| 1 |
10 |
120.1 |
Hand target (> 5 meter)
Floor target (without additional signals)
+ Touch (> 5 sec) & Chin on Hand (2 sec) |
| 2 |
10 |
107.7 |
Hand target (> 5 meter)
Floor target (with additional signals)
+ Touch (> 5 sec) & Chin on Hand (2 sec) |
| 3 |
8 |
99.0 |
Hand target (> 5 meter)
Floor target (with additional signals)
+ Touch (< 5 sec) & Chin on Hand (7 sec) |
| 4 |
8 |
92.6 |
Hand target (> 5 meter)
Floor target (with additional signals)
+ Touch (> 5 sec) & Chin on Hand (2 sec) |
| 5 |
8 |
111.7 |
Hand target (> 5 meter)
Floor target (without additional signals)
+ Touch (< 5 sec) & Chin on Hand (3 sec) |
| 6 |
7 |
90.7 |
Hand target (> 5 meter)
Floor target (with additional signals)
+ Touch (< 5 sec) & Chin on Hand (1 sec) |
| 7 |
7 |
95.1 |
Hand target (> 5 meter)
Floor target (with additional signals)
+ Touch (< 5 sec) & Chin on Hand (2 sec) |
| 8 |
7 |
114.2 |
Hand target (> 5 meter)
Floor target (with additional signals)
+ Touch (< 5 sec) & Chin on Hand (1 sec) |
2.3. Blood Sampling
In the context of the PFAS study, blood samples were collected on Mondays, Wednesdays and Fridays during weeks 5, 6, 10, 11, 13, and 14, and once a week on Wednesdays during all other weeks of the experiment with a total of 33 blood samplings per goat over the entire duration of the experiment. They were collected from the jugular vein (alternate sides) in the treatment area (Fig. 1) using a "BD Vacutainer Safety-Lok" cannula (21G, 0.8x19mm) and BD Vacutainers (Becton Dickinson AG, Allschwil, Switzerland). Blood sampling was performed as follows: the goat was led to the treatment area and onto the floor target by hand target/ luring or, if necessary, by light force on a leash. While the goat was being rewarded in this position, a caretaker stepped to the left side of the goat and lightly restrained the goat with their leg next to the goat’s chest, with one hand under the chin, and the other hand, if necessary, on the goat’s head. A second person knelt beside the goat’s left shoulder to compress the goat’s jugular vein by hand. A third person (HJ) disinfected the fur and skin over the jugular vein with alcohol and punctured the vein with the cannula. The goat was rewarded the moment the cannula exited the vein. While the goat was receiving the reward, the caretaker released the chin restraint. The vein was then compressed with a gauze at the site of the previous cannula insertion. If the vein did not bleed, the staff member removed the gauze and stepped back from the goat. After a final reward, the goat was returned to the pen.
To evaluate stress levels during blood sampling, the following parameters were assessed: (1) blood serum cortisol levels, (2) temperature change at the inner corner of the eye, and (3) behavior, observed using an ethogram. All parameters were recorded at nine designated time points throughout the study (time points 1-9 correspond to Wednesdays in study weeks 6, 7, 10, 12, 13, 15, 17, 21, and 23).
2.3.1. Blood Serum Cortisol
Blood serum samples were analyzed at the Federal Research Institute for Animal Health, Friedrich-Loeffler-Institut (FLI), Braunschweig, Germany, and serum cortisol concentrations were determined using a commercial ELISA kit (Enzo Life Sciences, Farmingdale, NY, USA). Three 10 ml BD Serum Vacutainers (Becton Dickinson GmbH, Heidelberg, Germany) were centrifuged at 2100 rpm for 15 seconds and the serum was pipetted. Aliquots of about 2 ml were stored of -21 C until analysis. In addition to the nine specified time points, the zero sample of the transfer study (week 2) was included for the analysis. When the zero sample was taken, training with the goats had not yet started.
2.3.2. Eye Temperature
Eye temperature images were captured using an infrared thermography (IRT) camera (VarioCAM HD, InfraTec, Dresden, Germany) and analyzed using IRBIS3 plus thermography software (version 3.1, InfraTec, Dresden, Germany). According to Sutherland et al. (2020) the images were taken from a distance of approximately 50 to 100 cm at an angle of approximately 90 degrees to the left eye of the goat. With 25 frames per second five images were taken for each image set. The first image in focus with the eye open was used for measurement. The maximum temperature was measured in the area of the medial canthus (see
Figure 2). Two image sets per animal per
time point were taken: the first with the restrained goat before the skin puncture (T1), the second approximately 5 seconds after the puncture during blood sampling (T2). The difference T2 - T1 was used for statistical analysis. Relative humidity (%) and ambient temperature (°C) were measured before the procedure.
Figure 2.
Thermographic image of a goat’s head shot in lateral view during blood sampling. The circled area is the medical canthus of the animal’s left eye.
Figure 2.
Thermographic image of a goat’s head shot in lateral view during blood sampling. The circled area is the medical canthus of the animal’s left eye.
2.3.3. Behavior
The blood sampling procedure was continuously recorded per group using a GoPro 7 White (GoPRo, San Mateo, USA). Behavior was evaluated retrospective from video material. The recording started with the first goat leaving the pen and ended when the last goat exited the treatment area. The videos were then evaluated by JM using an ethogram consisting of the four behavioral characteristics of latency, compliance, escape and defensive behavior, defined and characterized in Tab. 2. The program BORIS (29). was used for the ethogram evaluation.
2.4. Statistical Analysis
Data analysis and visualization were performed using R 4.2.2 (R Core Team, 2022) and Excel (Microsoft, 2021). The potential predictor variables and the outcome variables are listed in Tab. 2. Linear models and linear mixed models were calculated to assess the effect of
time point on the variables
cortisol,
thermo and
latency. These models consist of
cortisol,
thermo, and
latency as response or dependent variables,
time point as independent continuous variables or fixed factors,
humidity and
temperature as possible additional fixed factors,
goat as random factor and
pen,
person 1 and
person 2 were included as possible additional random factors (see
Table 2). We compared the Akaike Information Criteria (AIC) across these different models for each of the three continuous dependent variables. The model with the lowest AIC value was selected as most suitable and therefore optimal model (using the lme function from the nlme package in R, and the lm function from the R package stats).
For analysis of the binary outcomes
compliance and
escape a similar approach was used (see
Table 2). However, instead of linear and linear mixed models, generalized linear models (glm) and generalized linear mixed models applied (glmm). The R function glmmTMB from the package glmmTMB and glm from the package stats were used. For the ordinal variable
defensive behavior an ordered logistic regression with the R function polr from the package MASS was applied with
time point as independent variable (see
Table 2).
3. Results
3.1. Blood Serum Cortisol
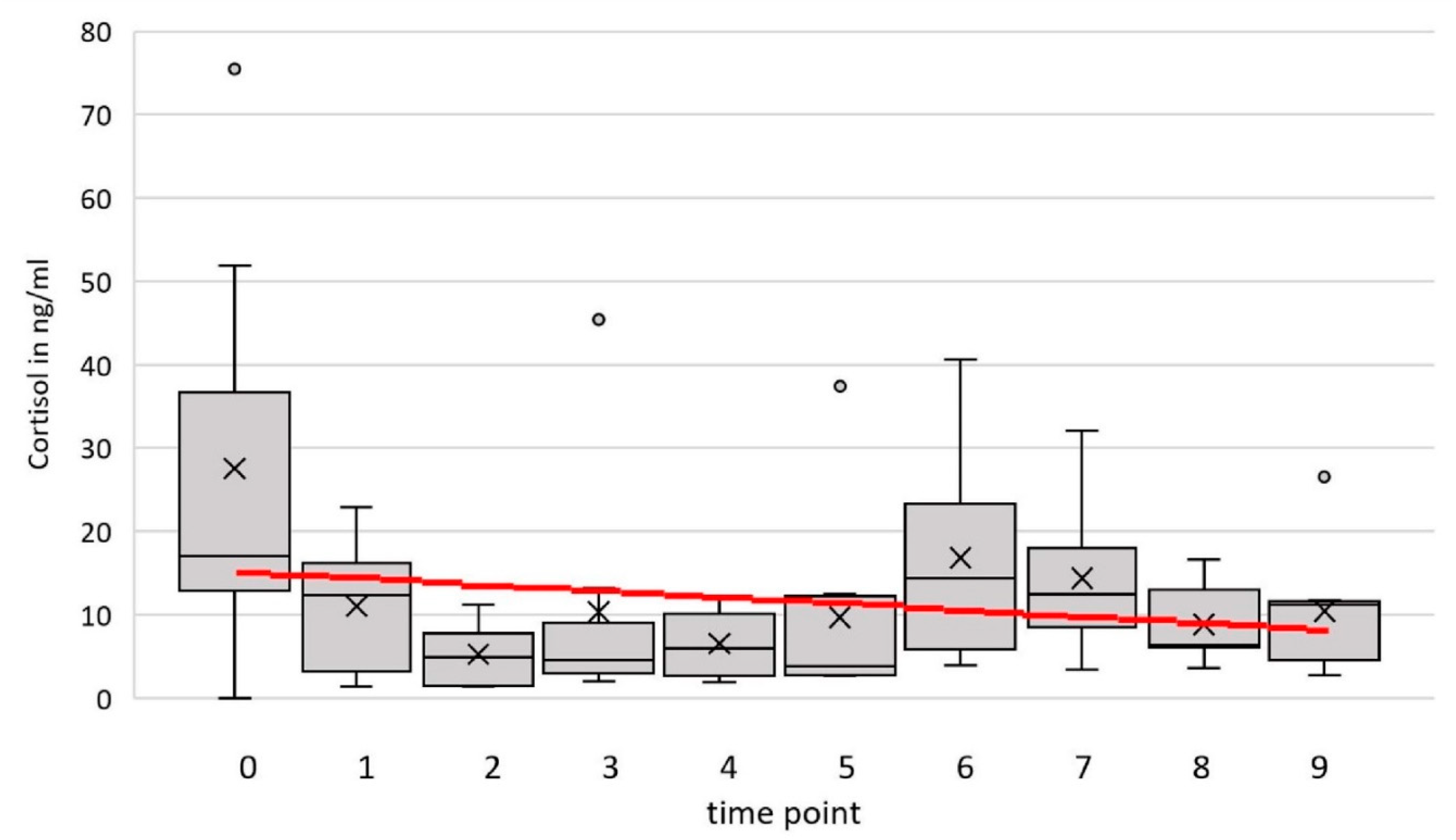
The cortisol concentration in the blood serum ranged between 1.02 and 75.49 ng/ml over all animals and time points. The optimal model (time point and temperature as fixed effects and goat and person 2 as random factors) estimated a slope of m= -0.125ng/ml/ time point. The difference at the end of the period in comparison to the cortisol level in the zero-sample estimated by the model is -0.003ng/ml. There was no significant influence of the time point on cortisol (F(1, 30) = 1.01, p = .32, 95% confidence interval (CI) [–0.855, 0.604]).
Figure 3.
Cortisol level (ng/ml) of 8 goats at 9 time points and the additional zero sample (0). The red line represents the prediction by the simple linear model with time point as the only independent variable.
Figure 3.
Cortisol level (ng/ml) of 8 goats at 9 time points and the additional zero sample (0). The red line represents the prediction by the simple linear model with time point as the only independent variable.
3.2. Eye Temperature
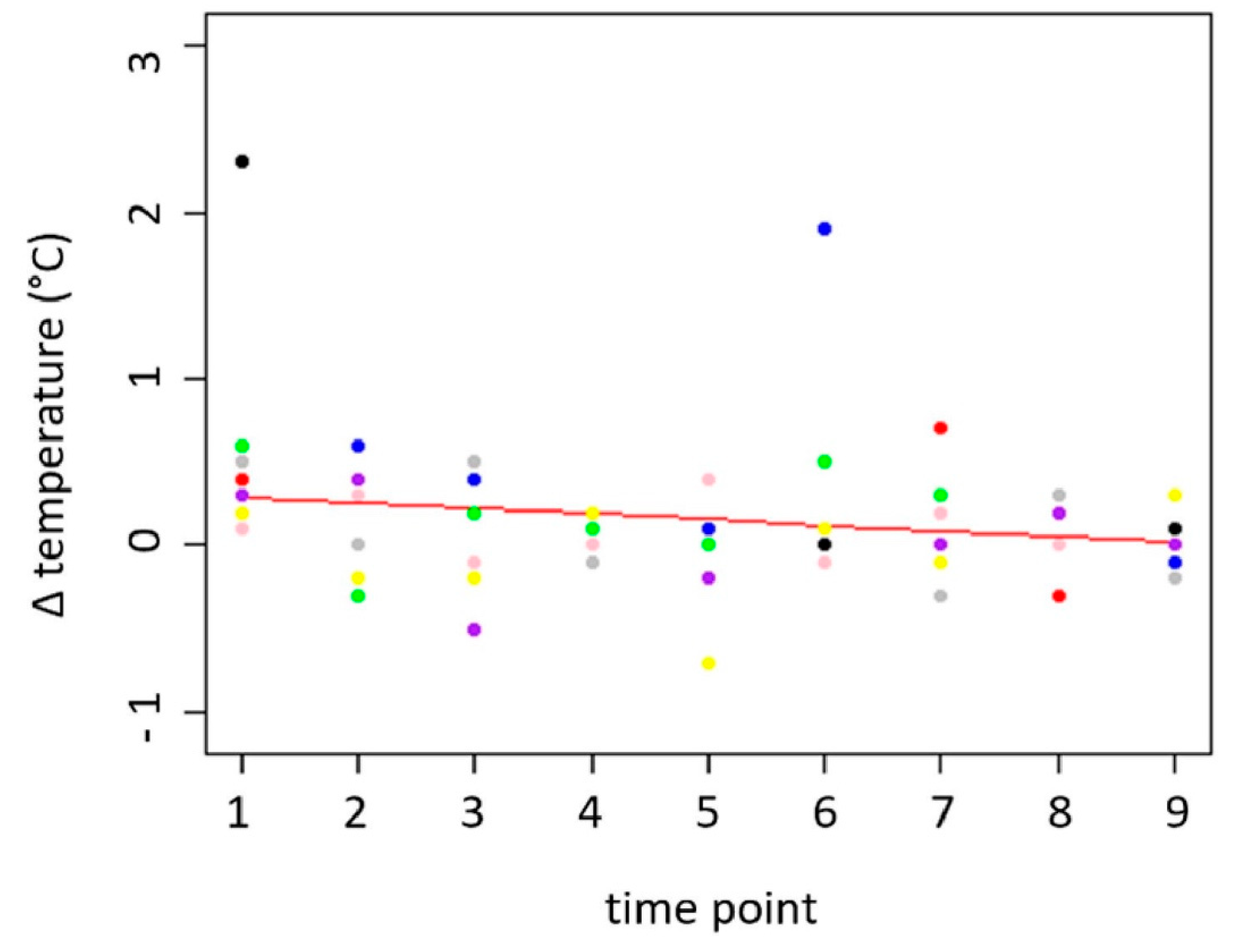
The eye temperature differences between “before” (Z1) and “after” (Z2) the needle insertion (thermo) were between -0.7 and 2.3 °C. The average temperatures before and after insertion of the needle were 37.30 °C (before; SD= 0.497 °C) and 37.40 °C (after; SD= 0.544 °C). For the variable
thermo, the optimal model consists of the
time point as the only fixed effect and
Person 1 and
Person 2 as random variables (
F(1, 2) = 93.34,
p = .01, 95% CI [–0.075, –0.029] °C per day). P value and CI for the change of eye temperature difference over time suggest that the difference decreases by habituation to the procedure. The temperature difference decreased between 0.32 °C and 0.6 °C over the measurement period. This means that the difference almost disappeared at the end of the measurement period, indicating that there was no effect of the needle insertion on the eye temperature at the end of the measurement period (see
Figure 4).
Figure 4.
Temperature difference (°C) at the medial canthus before and after the insertion of the needle.
Figure 4.
Temperature difference (°C) at the medial canthus before and after the insertion of the needle.
3.3. Behavior
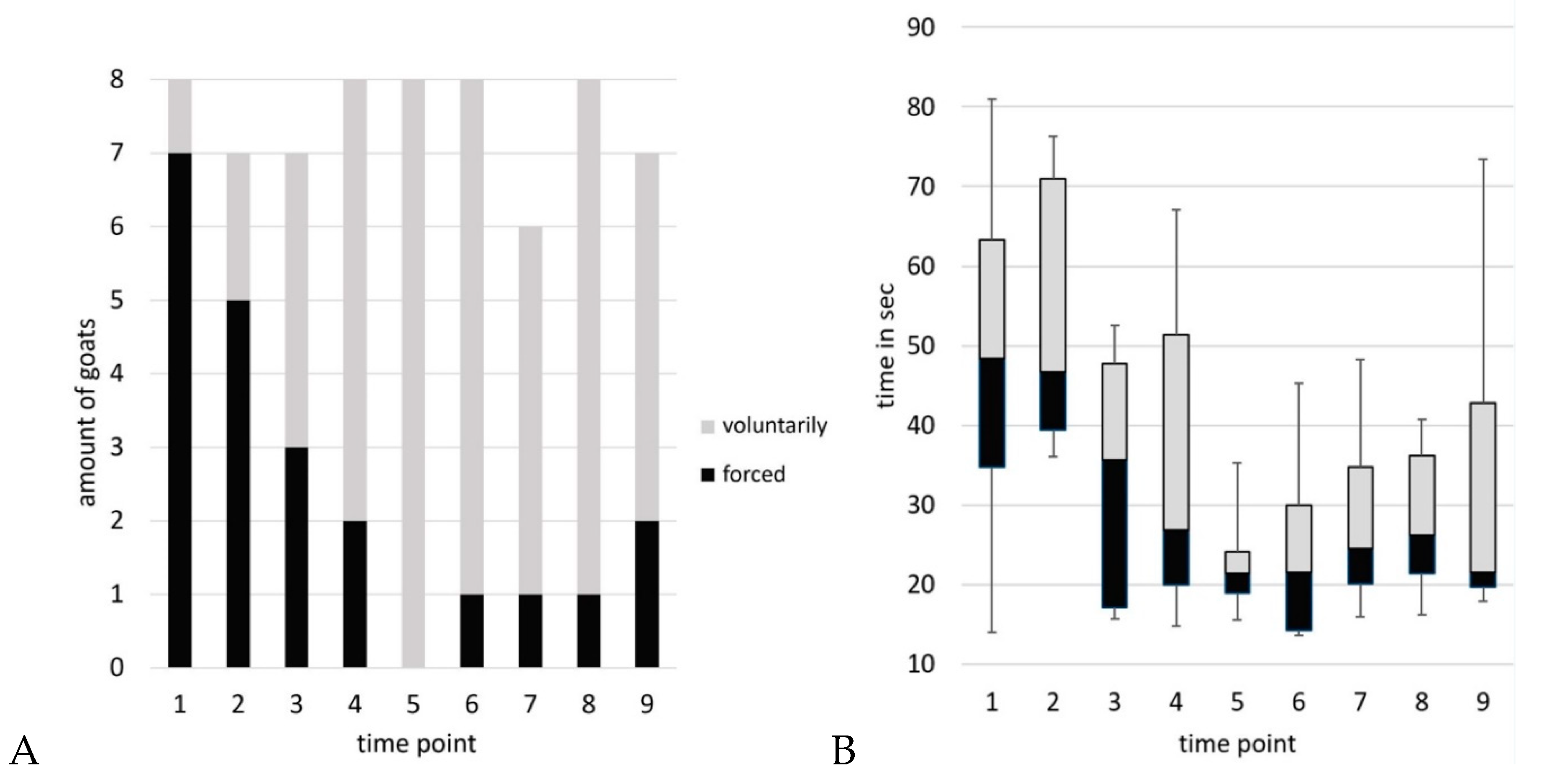
Blood sampling sessions took 3 to 10 min from separation from the group to the end of blood sampling. A maximum of three goats per time point attempted to escape from the position. No goat exhibited this behavior more than three times during the experiment. Per time point, two to a maximum of five animals showed defensive reactions. Goat 2 showed no defensive reactions at any time, whereas goat 8 showed defensive behavior at all times. Kicking behavior was observed 35 times, 25 of which were by goat 8. The number of goats voluntarily walking on the floor target varied from one (time point 1) to all (n=8, time point 5). On average, they needed 17.4 sec (time point 6) to 51.3 sec (time point 2) to enter the position.
Within the optimal model for compliance (time point, temperature and humidity as independent variables), there was a significant decrease in the number of goats that had to be forced to walk on the floor target (z(66) = –2.78, p = .005). For the variable latency (optimal model: linear model with temperature and time point as independent variables) a decrease of - 3.41 sec/time point was estimated. The decrease at the end of the period compared to the zero sample is -31 %. There was a significant decrease of time the goats needed to step on the floor target (F(1, 64) = 26.03, p < .001, 95% CI [–4.92, –1.90] s per time point).
Figure 5.
A) Compliance (category of positioning on the floor target: voluntarily or forced) and B) Latency (time the goats took to enter the position on the floor target) over time.
Figure 5.
A) Compliance (category of positioning on the floor target: voluntarily or forced) and B) Latency (time the goats took to enter the position on the floor target) over time.
For the variable escape (optimal model: generalized linear model with the time point as the independent variable) there is no clear indication of whether the value of the indicator is increasing or decreasing (z(71) = –1.17, p = .24). For the variable defensive behavior, no significant increase or decrease over time point was found (z(69) = –0.89, p = .37). The Pearson Correlation between cortisol and latency is low (r = –0.055; t(65) = –0.44, p = .66). There is also no statistically significant relationship between cortisol and compliance (t(65) = 0.98, p = .329) or cortisol and escape behavior (t(65) = –0.26, p = .795).
4. Discussion
The aim of the study was to observe stress parameters in goats in repeated blood sampling. We did not see an increase over the time in any stress parameter during the experiment . First, blood serum cortisol showed no significant change over time. A possible interpretation might be that repeated blood sampling did not lead to a higher stress level. This and the model estimation of a slight decrease in cortisol concentration over time, aligns with the findings of Andanson et al. (2020). In their study the serum cortisol level of lambs (blood taken by catheters) where compared after saliva sampling, venepuncture and no intervention. Handling was described as “carefully” and the animals were “gentle” moved to restrain for sample collection. On the first day, the group of lambs with venepuncture had the highest serum cortisol level. After one week of blood sampling twice daily, there was a decrease in serum cortisol levels amongst the venepuncture group and no significant differences amongst all three groups. Another plausible scenario could have been that stress levels have increased over time but appeared constant due to the overlay of stress reducing factors, such as habituation. This is in consistency to the EU guideline’s hypothesis that stress levels during a repeated intervention may decrease due to habituation and desensitization (European Commission, 2018). In our study, the procedure of blood sampling was quick, with 3 to 10 min from separation from the group to the end of blood sampling. Kannan et al. (2021). found that when goats were isolated from the group, cortisol levels peaked immediately. However, Kruger et al. (2016) found a peak in cortisol levels 10 minutes after handling stress. We may would have observed a change in blood serum cortisol, if an additional measurement had been taken 20-30 minutes after the first intervention. Due to the design of the underlying experiment, this was not feasible, and therefore the results of the cortisol measurement must be regarded as preliminary and interpreted with caution. However, in this study PRT and gentle handling may have contributed to the tendency for decreasing cortisol levels. This would be consistent with the findings of other studies where, for example, PRT with grizzly bears (Joyce-Zuniga et al., 2016) or minipigs (Fiderer et al., 2024) or habituation in mice (Marin et al., 2023) led to a decrease in cortisol level during blood sampling. The positive effects of habituation and PRT may be difficult to disentangle, as both can happen at the same time.
Eye temperatures showed no significant change over time. This suggests that the act of puncturing the skin did not have an immediate, consistent effect on the peripheral vasculature. As shown by several studies eye temperature increased after a stressor like veterinary treatments (Bartolomé et al., 2019), transportation (Aragona et al., 2024) or shearing (Arfuso et al., 2022). In contrast to the study by Arfuso et al. (2022), the stress stimuli in our study may have been too mild, and the intervals between measurements too short, to induce a significant and consistent change in eye temperature. Therefore, thermographic measurements seem not suitable for assessing stress during short-term blood sampling procedures and could not address the research question of this study. In future studies on this topic, the baseline values of the goats before isolation from the group should be measured to assess the effects of handling and blood sampling compared to neutral or relaxed states without interventions. Here, baseline values were not considered necessary to assess the effects of the repetitive nature of these procedures and, secondly, not feasible to measure with a consistent angle and distance.
The Ethological analysis of behavior during blood sampling did not reveal any relevant changes in escape or defensive behavior. However, there were notable differences between individual animals. The defensive behavior “kicking” was primarily exhibited by one goat (no. 8) throughout all sessions. This behavior may have been unintentionally reinforced during the procedure, potentially contributing to its consistency. Despite this, the goat appeared attentive and engaged during training sessions. Beyond these individual, animal-specific effects, the force and technique of restraining influences the occurrence of this behavior as well. Importantly, the absence of defensive behaviors should not automatically be interpreted as lower stress, as not showing such reactions may represent a freeze response within the four stress response patterns (fight, flight, freeze, or fawn). Therefore, no defensive behavior does not necessarily calmness or vuluntariness, particularly under forceful or tight restraint. It would remain unclear whether this is due to cooperation and voluntariness or to a passive stress response such as freezing (Rosenberger et al., 2022). However, a significant reduction in escape or defensive behavior with less force during restraining would have been expected with intensified training, since there are numerous examples in animal training, particularly in zoo animals, where voluntary participation in blood sampling is achieved, e.g., in macaques and chimpanzees (Coleman et al., 2008), or lions (Callealta et al., 2020). Specifically, training to maintain the position for longer and under more distraction would have been required. Stimuli such as the touch of a second person, the presence of additional staff in the proximity of the animal, or the application of disinfectant at the injection site would have had to be incorporated into the training to facilitate the generalization of the learned behavior.
The significant decrease in the latency to reach the floor target argues against a possible anticipation of a negative stressor, as delayed locomotion is associated with fear in common behavioral tests (open-field test, novel-object test) (Forkman et al., 2007). In our study, the goats were specifically trained for this behavior. This is in line with the findings of Hutson (1985) that trained sheep enter a sheep-handling machine more quickly than untrained sheep. Furthermore, the goats stepped more often voluntarily on the floor target and, therefore, showed more compliance. Again, we resume that this is a training effect, as they were trained to step on the floor target, with or without additional cues (such as the leading hand target). To determine the exact influence of PRT on the decreasing latency and increasing voluntary behavior, an untrained control group would have been required. The use of the floor target reduced the need for coercion in positioning. The decrease of time is in line with the findings of Sankey et al. (2010), where ponies trained with positive reinforcement showed increased interest in humans and approached them with a shorter latency than ponies trained with negative reinforcement. This highlights that PRT can improve the human- animal-relationship. Furthermore, contact with calmer goats was perceived as pleasant by the staff and may therefore also contribute to a reduction of human stress, potentially improving safety in the workplace. The voluntary behavior in the blood sampling process may have resulted in a lower overall stress level for the goats. However, other parameters did not confirm this. For example, there was no significant correlation between cortisol levels and behavioral parameters, as well as no significant change in escape or aversive behavior. Therefore, this study cannot address the emotional state of the goats during the procedures.
We assume that mainly three factors may explain the stable stress parameters during repeated blood sampling in this study: First, habituation and PRT. This is in line with the EU recommendation, which identifies habituation as a mitigating factor in repeated procedures (Environment, 2018). Second, optimized work procedures, such as a well-coordinated team, thorough preparation, and, importantly, a calm approach to handling the animals. In addition to gentle handling, it is likely that, the animals developed a positive expectation towards the trainer, who was present at all time points. In this study goats were rewarded with food therefore classically conditioned took place for a positive association with the trainer, and also testing location and procedures. A third reason may be that the stressor of blood sampling itself has been reduced by choosing the smallest possible needle size to minimize the pain associated with the injection (Okamoto et al., 2012). In the context of animal experimentation, the introduction of a needle is the threshold for a regulated animal experiment (Directive 2010/63/EU). However, there is evidence that even routine handling procedures, including subcutaneous vaccination causes stress in goats (Kruger et al., 2016). As repeated blood sampling can be considered a relatively low-stress procedure for goats, the influence of repetition on other species or of more stressful procedures remains to be investigated. However, it is likely that with adequate PRT, habituation and optimized protocols, repetition does not necessarily lead to increased stress levels.
Major limitation of this study is a missing control group. There it is a strictly descriptive study that was performed as a satellite in a PFAS transfer study, the ethical approval allowed only sufficient animals for this experiment, but not extra animals for a control group. It was therefore not possible to include an untrained group of goats or a group that was included in only one or two time points of blood sampling. Furthermore, the measurement of further stress-relevant parameters, like heart-rate-variability or blood pressure (Hydbring-Sandberg et al., 2022) was not possible, since the animals must not receive any other treatment than approved for the transfer study. However, as our observations were made during a specific stressor (blood sampling), we cannot determine why the stress parameters remained unchanged over time; nevertheless, the measurements provide valuable information about the animals’ responses under these conditions.
Furthermore, the ethical approval included all animal to be trained. Therefore, we could not evaluate the effect of training with a control group. Furthermore, the positive influence of PRT might have been more pronounced with a higher number of training sessions and an even more experienced trainer. However, even without the full additional training and desensitization, many animals showed clear benefits, as they were noticeably calmer and more affiliative in their interactions with humans compared to the beginning of the study.
5. Conclusions
In summary, under well-controlled and positively reinforced handling conditions, repeated exposure to blood sampling as a potential stressful procedure not necessary as an effect on stress parameters. This observation can be attributed to three key factors: first, habituation and PRT for cooperative behavior. Second, optimal organizational practices, such as gentle handling, the use of minimally invasive needles and a well-coordinated team, and third, the insertion of the needle for blood sampling as a relative mild aversive stimulus. If the first two factors are effectively implemented, it is possible that even more painful stimuli may not necessarily lead to an increase of stress over time. These observations highlight the potential benefits of habituation and PRT as refinement strategies, but confirm that definitive conclusions require studies designed specifically to address this question, including control groups and repeated sampling within shorter intervals, are needed to confirm whether habituation or other factors underlie the stable cortisol levels observed here.
Author Contributions
Conceptualization, J.M. and C.F-T.; Methodology, J.M. and C.F-T.; Software, M.S.;.; Formal Analysis, J.M. and M.S; Investigation, J.M. and C.F-T.; Resources, J.M., H.J. and C.F-T.; Data Curation, J.M.; Writing – Original Draft Preparation, J.M.; Writing – Review & Editing, J.M., H.J., M.S and C.F-T; Visualization, J.M. and M.S.; Supervision, C.F-T.; Project Administration, J.M. and C.F-T. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
In this section, you should add the Institutional Review Board Statement and approval number, if relevant to your study. You might choose to exclude this statement if the study did not require ethical approval. Please note that the Editorial Office might ask you for further information. Please add “The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board (or Ethics Committee) of NAME OF INSTITUTE (protocol code XXX and date of approval).” for studies involving humans. OR “The animal study protocol was approved by the Institutional Review Board (or Ethics Committee) of NAME OF INSTITUTE (protocol code XXX and date of approval).” for studies involving animals. OR “Ethical review and approval were waived for this study due to REASON (please provide a detailed justification).” OR “Not applicable” for studies not involving humans or animals.
Informed Consent Statement
Any research article describing a study involving humans should contain this statement. Please add “Informed consent was obtained from all subjects involved in the study.” OR “Not applicable.” for studies that did not involve humans. You might also choose to exclude this statement if the study did not involve humans. Written informed consent for publication must be obtained from identifiable human participants. For studies involving client-owned animals written informed consent must be obtained from the owner of the animals (or an authorized agent for the owner).
Data Availability Statement
All relevant data are provided upon request by e-mail to carola.fischer-tenhagen@bfr.bund.de.
Acknowledgments
We would like to thank Susanne Kersten, Lisa Bode and Karen Semela from the Friedrich-Löffler-Institute for the project partnership in the transfer experiment and for the analysis of the serum samples. We would also like to thank all the animal caretakers for their competent and caring care of the animals.
Conflicts of Interest
The authors declare no conflict of interest.
References
- ANDANSON, S., BOISSY, A. & VEISSIER, I. 2020. Conditions for assessing cortisol in sheep: the total form in blood v. the free form in saliva. Animal, 14, 1916-1922. [CrossRef]
- ARAGONA, F., RIZZO, M., ARFUSO, F., ACRI, G., FAZIO, F., PICCIONE, G. & GIANNETTO, C. 2024. Eye Temperature Measured with Infrared Thermography to Assess Stress Responses to Road Transport in Horses. Animals, 14, 1877, https://www.mdpi.com/2076-2615/14/13/1877.
- ARFUSO, F., ACRI, G., PICCIONE, G., SANSOTTA, C., FAZIO, F., GIUDICE, E. & GIANNETTO, C. 2022. Eye surface infrared thermography usefulness as a noninvasive method of measuring stress response in sheep during shearing: Correlations with serum cortisol and rectal temperature values. Physiology & Behavior, 250, 113781. [CrossRef]
- BAILEY, J. 2018. Does the stress of laboratory life and experimentation on animals adversely affect research data? A critical review. Altern Lab Anim, 46, 291-305.10.1177/026119291804600501.
- BALCOMBE, J. P., BARNARD, N. D. & SANDUSKY, C. 2004. Laboratory routines cause animal stress. Contemp Top Lab Anim Sci, 43, 42-51,.
- BARTOLOMÉ, E., AZCONA, F., CAÑETE-ARANDA, M., PERDOMO-GONZÁLEZ, D. I., RIBES-PONS, J. & TERÁN, E. M. 2019. Testing eye temperature assessed with infrared thermography to evaluate stress in meat goats raised in a semi-intensive farming system: a pilot study. Arch Anim Breed, 62, 199-204.10.5194/aab-62-199-2019.
- BAUMANS, V. 2004. Use of animals in experimental research: an ethical dilemma? Gene Therapy, 11, S64-S66.10.1038/sj.gt.3302371.
- BISOGNI, S., DINI, C., OLIVINI, N., CIOFI, D., GIUSTI, F., CAPRILLI, S., GONZALEZ LOPEZ, J. R. & FESTINI, F. 2014. Perception of venipuncture pain in children suffering from chronic diseases. BMC Res Notes, 7, 735.10.1186/1756-0500-7-735.
- BLESSING, W. W. The Lower Brainstem and Bodily Homeostasis. 1997.
- BRIEFER, E. F., TETTAMANTI, F. & MCELLIGOTT, A. G. 2015. Emotions in goats: mapping physiological, behavioural and vocal profiles. Animal Behaviour, 99, 131-143. [CrossRef]
- CALLEALTA, I., LUEDERS, I., LUTHER-BINOIR, I. & GANSWINDT, A. 2020. Positive Reinforcement Conditioning as a Tool for Frequent Minimally Invasive Blood and Vaginal Swab Sampling in African Lions (Panthera Leo). J Appl Anim Welf Sci, 23, 508-519.10.1080/10888705.2019.1709066.
- COLEMAN, K., PRANGER, L., MAIER, A., LAMBETH, S. P., PERLMAN, J. E., THIELE, E. & SCHAPIRO, S. J. 2008. Training rhesus macaques for venipuncture using positive reinforcement techniques: a comparison with chimpanzees. J Am Assoc Lab Anim Sci, 47, 37-41,.
- DAI, F., MAZZOLA, S., CANNAS, S., HEINZL, E. U. L., PADALINO, B., MINERO, M. & DALLA COSTA, E. 2020. Habituation to Transport Helps Reducing Stress-Related Behavior in Donkeys During Loading. Front Vet Sci, 7, 593138.10.3389/fvets.2020.593138.
- DIRECTIVE 2010/63/EU of the European Parliament and of the Council on the protection of animals used for scientific purposes, Official Journal of the European Union, 2010.
- EUROPEAN COMMISSION: 2018. Directorate-General for Environment, Caring for animals aiming for better science – Directive 2010/63/EU on protection of animals used for scientific purposes – Severity assessment framework, Publications Office, https://data.europa.eu/doi/10.2779/068620.
- EUROPEAN COMMISSION, 2024. Summary Report on the statistics on the use of animals for scientific purposes in the Member States of the European Union and Norway in 2022. Commission Staff Working Document SWD(2024) 185 final. Available at: https://d8aaf127-0203-427a-b8b6-1f1b942cd1af.usrfiles.com/ugd/d8aaf1_cf831339d46f4fe4bfcfd0b0d1eb1f37.pdf [Accessed February 18, 2025].FIDERER, D., THOENE-REINEKE, C. & WIEGARD, M. 2024. Clicker Training in Minipigs to Reduce Stress during Blood Collection-An Example of Applied Refinement. Animals (Basel), 14.10.3390/ani14192819.
- FORKMAN, B., BOISSY, A., MEUNIER-SALAÜN, M. C., CANALI, E. & JONES, R. B. 2007. A critical review of fear tests used on cattle, pigs, sheep, poultry and horses. Physiology & Behavior, 92, 340-374. [CrossRef]
- HUTSON, G. D. 1985. The influence of barley food rewards on sheep movement through a handling system. Applied Animal Behaviour Science, 14, 263-273. [CrossRef]
- HYDBRING-SANDBERG, E., VON WALTER, L. W. & FORKMAN, B. 2022. Cortisol is not enough: A complex stress reaction in tethered goats. Animal Welfare, 31, 91-98.10.7120/09627286.31.1.008.
- IMFELD-MUELLER, S., VAN WEZEMAEL, L., STAUFFACHER, M., GYGAX, L. & HILLMANN, E. 2011. Do pigs distinguish between situations of different emotional valences during anticipation? Applied Animal Behaviour Science, 131, 86-93.10.1016/j.applanim.2011.02.009.
- JEREM, P., JENNI-EIERMANN, S., MCKEEGAN, D., MCCAFFERTY, D. J. & NAGER, R. G. 2019. Eye region surface temperature dynamics during acute stress relate to baseline glucocorticoids independently of environmental conditions. Physiol Behav, 210, 112627.10.1016/j.physbeh.2019.112627.
- JOYCE-ZUNIGA, N. M., NEWBERRY, R. C., ROBBINS, C. T., WARE, J. V., JANSEN, H. T. & NELSON, O. L. 2016. Positive Reinforcement Training for Blood Collection in Grizzly Bears (Ursus arctos horribilis) Results in Undetectable Elevations in Serum Cortisol Levels: A Preliminary Investigation. Journal of Applied Animal Welfare Science, 19, 210-215.10.1080/10888705.2015.1126523.
- JUST, H., BODE, L., WAGNER, B., MEIER, J., KERSTEN, S., FISCHER-TENHAGEN, C., JAHNKE, A., KÜRBIS, C., LÜTH, A., ., PIEPER, R., DÄNICKE, S. & KOWALCZYK, J. 2025. Linear and branched Poly- and Perfluoroalkyl Acids: Transfer from oral exposure via hay into the milk of dairy goats (Capra aegagrus hircus) [Unpublished manuscript].
- KANNAN, G., ESTRADA-REYES, Z. M., BATCHU, P., KOUAKOU, B., TERRILL, T. H. & NALDURTIKER, A. 2021. Social isolation of goats: significance of visual contact with conspecifics on behavioral and physiological responses. J Anim Sci, 99.10.1093/jas/skab150.
- KANNAN, G., TERRILL, T. H., KOUAKOU, B., GAZAL, O. S., GELAYE, S., AMOAH, E. A. & SAMAKÉ, S. 2000. Transportation of goats: effects on physiological stress responses and live weight loss. J Anim Sci, 78, 1450-7.10.2527/2000.7861450x.
- KRUGER, L. P., NEDAMBALE, T. L., SCHOLTZ, M. M. & WEBB, E. C. 2016. The effect of environmental factors and husbandry practices on stress in goats. Small Ruminant Research, 141, 1-4. [CrossRef]
- LI, R., WANG, L., CHEN, B., ZHANG, Y. & QI, P. 2024. Effects of Transportation on Blood Indices, Oxidative Stress, Rumen Fermentation Parameters and Rumen Microbiota in Goats. Animals (Basel), 14.10.3390/ani14111616.
- MARIN, N., MORAGON, A., GIL, D., GARCIA-GARCIA, F. & BISBAL, V. 2023. Acclimation and Blood Sampling: Effects on Stress Markers in C57Bl/6J Mice. Animals (Basel), 13.10.3390/ani13182816.
- OKAMOTO, K., AMI, N. & OSHIMA, H. 2012. Assessment of needle insertion pain with flexor reflex responses in anesthetized rats PAIN RESEARCH, 27, 215-225.10.11154/pain.27.215.
- PLUUT, H., CURȘEU, P. L. & FODOR, O. C. 2022. Development and Validation of a Short Measure of Emotional, Physical, and Behavioral Markers of Eustress and Distress (MEDS). Healthcare (Basel), 10.10.3390/healthcare10020339.
- PRICE, E. O. & THOS, J. 1980. Behavioral responses to short-term social isolation in sheep and goats. Applied Animal Ethology, 6, 331-339. [CrossRef]
- RALPH, C. R. & TILBROOK, A. J. 2016. INVITED REVIEW: The usefulness of measuring glucocorticoids for assessing animal welfare. J Anim Sci, 94, 457-70.10.2527/jas.2015-9645.
- ROSENBERGER, K., SIMMLER, M., LANGBEIN, J., NAWROTH, C. & KEIL, N. 2022. Responsiveness of domesticated goats towards various stressors following long-term cognitive test exposure. PeerJ, 10, e12893.10.7717/peerj.12893.
- RUSSELL, W. M. S. & BURCH, R. L. 1959. The principles of humane experimental technique, Methuen,.
- SANKEY, C., RICHARD-YRIS, M.-A., HENRY, S., FUREIX, C., NASSUR, F. & HAUSBERGER, M. 2010. Reinforcement as a mediator of the perception of humans by horses (Equus caballus). Animal Cognition, 13, 753-764.10.1007/s10071-010-0326-9.
- SELYE, H. 1975. Stress and distress. Compr Ther, 1, 9-13,.
- SIEBERT, K., LANGBEIN, J., SCHÖN, P.-C., TUCHSCHERER, A. & PUPPE, B. 2011. Degree of social isolation affects behavioural and vocal response patterns in dwarf goats (Capra hircus). Applied Animal Behaviour Science, 131, 53-62. [CrossRef]
- SKINNER, B. F. 1951. How to Teach Animals. Scientific American, 185, 26-29, http://www.jstor.org/stable/24950550.
- STEWART, M., WEBSTER, J. R., VERKERK, G. A., SCHAEFER, A. L., COLYN, J. J. & STAFFORD, K. J. 2007. Non-invasive measurement of stress in dairy cows using infrared thermography. Physiology & Behavior, 92, 520-525. [CrossRef]
- SUTHERLAND, M. A., WORTH, G. M., DOWLING, S. K., LOWE, G. L., CAVE, V. M. & STEWART, M. 2020. Evaluation of infrared thermography as a non-invasive method of measuring the autonomic nervous response in sheep. PLOS ONE, 15, e0233558.10.1371/journal.pone.0233558.
- TOSTO, M. S. L., SANTOS, S. A., FILHO, R., RODRIGUES, T., NICORY, I. M. C., DE CARVALHO, G. G. P., BITTENCOURT, R. F., AYRES, M. C. C. & PEREIRA, T. C. J. 2021. Metabolic and behavior changings during the transition period as predictors of calving proximity and welfare of dairy goats. Vet Anim Sci, 11, 100168.10.1016/j.vas.2021.100168.
- WANG, J. Y., ZHANG, H. T., CHANG, J. Y., WOODWARD, D. J., BACCALÁ, L. A. & LUO, F. 2008. Anticipation of pain enhances the nociceptive transmission and functional connectivity within pain network in rats. Mol Pain, 4, 34.10.1186/1744-8069-4-34.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).