Submitted:
25 November 2025
Posted:
28 November 2025
You are already at the latest version
Abstract
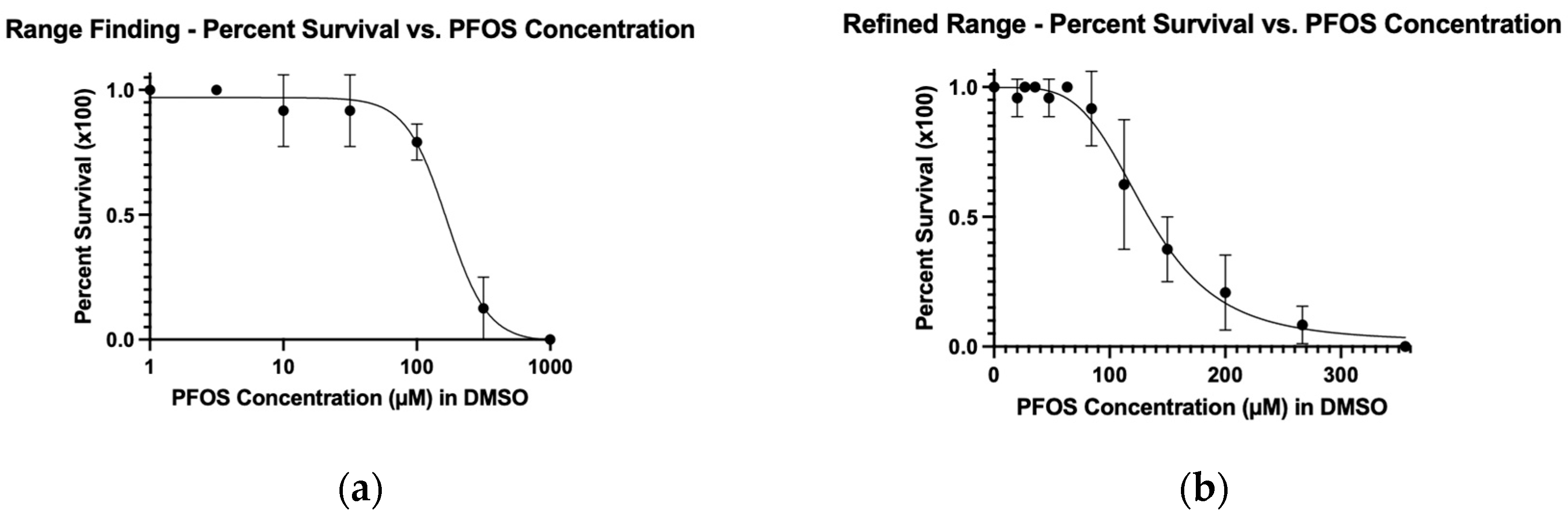
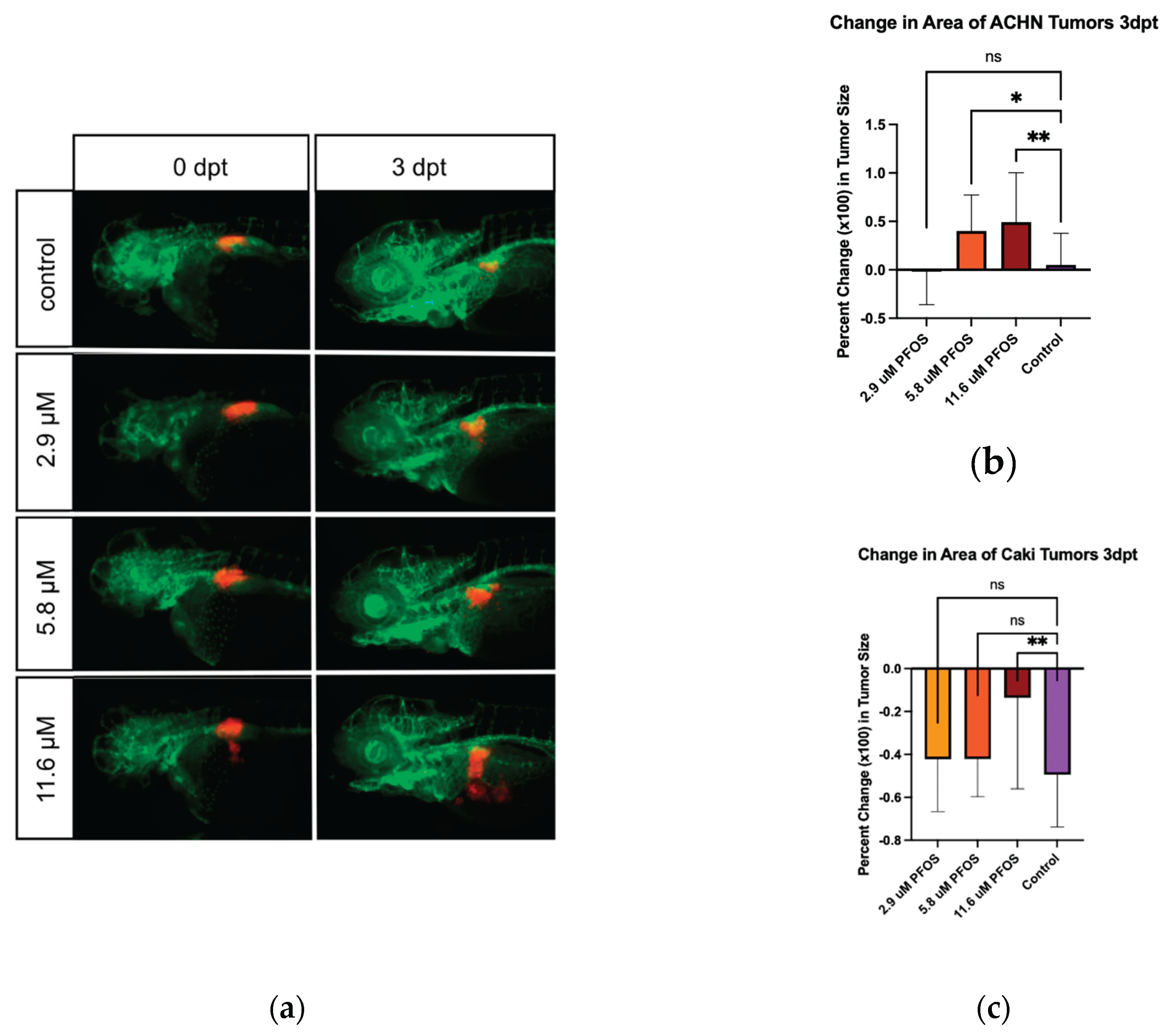
Persistent environmental pollutants such as per- and poly-fluoroalkyl substances (PFAS) have been associated with a wide range of toxic effects, including cancer. There are over 12,000 PFAS compounds, which may act as carcinogens individually or in combinations. Therefore, efficient in vivo models of carcinogenicity are needed for evaluating environmental contaminant compounds and chemical mixtures. Here, we use the larval zebrafish xenograft assay to identify tumor growth activity of perfluorooctanesulfonic acid (PFOS), a known carcinogenic PFAS. Dose response curves for PFOS exposure were used to identify the Maximum Tolerated Concentration (MTC) and Lethal Concentration causing 50% death (LC50) under xenograft conditions. Zebrafish xenografts were established by injecting fluorescently labeled kidney cancer cells into the embryonic body cavity near the developing kidney, followed by treatment with PFOS at a concentration of 5%, 10% and 20% of the MTC. When treated with PFOS, zebrafish xenografts using renal cell carcinoma (ACHN) cells and clear renal cell carcinoma (Caki-1) cells show dose-dependent changes in tumor area. This study is the first to directly show cancer-promoting activity of a PFAS, using a rapid in vivo zebrafish xenograft assay, and demonstrates the utility of this model for validation of predicted cancer-promoting properties of environmental contaminants.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Zebrafish
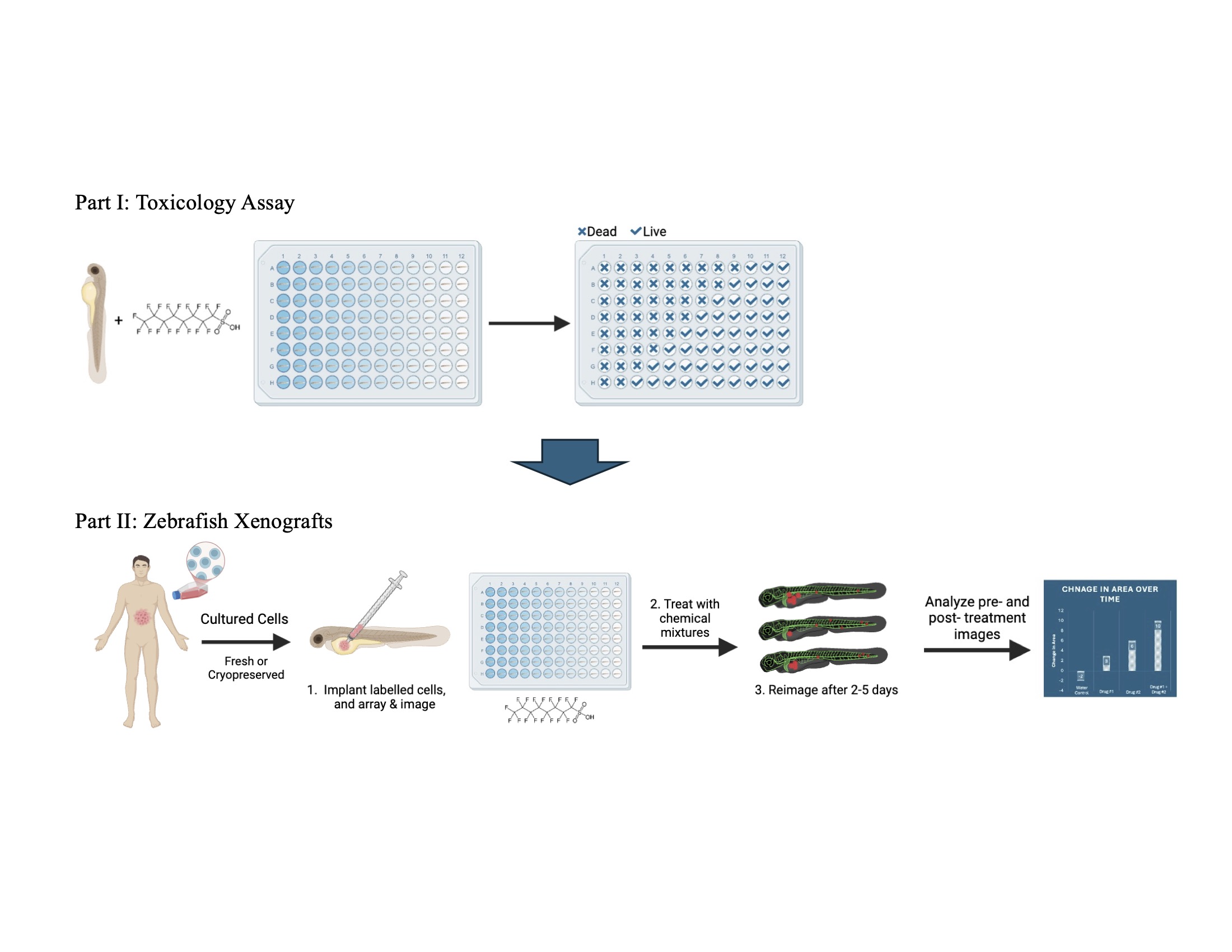
2.2. Toxicology Assays
2.3. Cell Culture
2.4. Zebrafish Xenografts
2.5. Statistical Analysis
3. Results
3.2. Xenograft Experiments
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PFOS | Perfluorooctanesulfonic Acid |
| PFAS | Per- and Poly-fluoroalkyl substances |
| LC50 | Lethal Concetration causing 50% death |
| MTC | Maximum Tolerated Concentration |
| ACHN | Renal cell carcinoma |
| Caki-1 | Clear renal cell carcinoma cells |
| NAM | New Approach Methodology |
| hpf | Hours post fertilization |
| dpf | Days post fertilization |
| dpt | Days post treatment |
| PTU | Phenylthiourea |
| ANOVA | One-way analysis of variance |
References
- Zeng, Z.; Song, B.; Xiao, R.; Zeng, G.; Gong, J.; Chen, M.; Xu, P.; Zhang, P.; Shen, M.; Yi, H. Assessing the Human Health Risks of Perfluorooctane Sulfonate by in Vivo and in Vitro Studies. Environ Int 2019, 126, 598–610. [Google Scholar] [CrossRef]
- Buck, R.C.; Franklin, J.; Berger, U.; Conder, J.M.; Cousins, I.T.; de Voogt, P.; Jensen, A.A.; Kannan, K.; Mabury, S.A.; van Leeuwen, S.P. Perfluoroalkyl and Polyfluoroalkyl Substances in the Environment: Terminology, Classification, and Origins. Integr Environ Assess Manag 2011, 7, 513–41. [Google Scholar] [CrossRef]
- Paul, A.G.; Jones, K.C.; Sweetman, A.J. A First Global Production, Emission, and Environmental Inventory for Perfluorooctane Sulfonate. Environ Sci Technol 2009, 43, 386–92. [Google Scholar] [CrossRef]
- Williams, A.J.; Gaines, L.G.T.; Grulke, C.M.; Lowe, C.N.; Sinclair, G.F.B.; Samano, V.; Thillainadarajah, I.; Meyer, B.; Patlewicz, G.; Richard, A.M. Assembly and Curation of Lists of Per- and Polyfluoroalkyl Substances (Pfas) to Support Environmental Science Research. Front Environ Sci 2022, 10, 1–13. [Google Scholar] [CrossRef]
- Blanc, M.; Karrman, A.; Kukucka, P.; Scherbak, N.; Keiter, S. Mixture-Specific Gene Expression in Zebrafish (Danio Rerio) Embryos Exposed to Perfluorooctane Sulfonic Acid (Pfos), Perfluorohexanoic Acid (Pfhxa) and 3,3',4,4',5-Pentachlorobiphenyl (Pcb126). Sci Total Environ 2017, 590-591, 249-57. [Google Scholar] [CrossRef]
- Ding, G.; Zhang, J.; Chen, Y.; Wang, L.; Wang, M.; Xiong, D.; Sun, Y. Combined Effects of Pfos and Pfoa on Zebrafish (Danio Rerio) Embryos. Arch Environ Contam Toxicol 2013, 64, 668–75. [Google Scholar] [CrossRef] [PubMed]
- Fey, M. E., P. E. Goodrum, N. R. Razavi, C. M. Whipps, S. Fernando, and J. K. Anderson. Is Mixtures' Additivity Supported by Empirical Data? A Case Study of Developmental Toxicity of Pfos and 6:2 Fts in Wildtype Zebrafish Embryos. Toxics 10, no. 8 (2022).
- Khezri, A., T. W. Fraser, R. Nourizadeh-Lillabadi, J. H. Kamstra, V. Berg, K. E. Zimmer, and E. Ropstad. A Mixture of Persistent Organic Pollutants and Perfluorooctanesulfonic Acid Induces Similar Behavioural Responses, but Different Gene Expression Profiles in Zebrafish Larvae. Int J Mol Sci 18, no. 2 (2017).
- Arrieta-Cortes, R.; Farias, P.; Hoyo-Vadillo, C.; Kleiche-Dray, M. Carcinogenic Risk of Emerging Persistent Organic Pollutant Perfluorooctane Sulfonate (Pfos): A Proposal of Classification. Regul Toxicol Pharmacol 2017, 83, 66–80. [Google Scholar] [CrossRef] [PubMed]
- Steenland, K.; Winquist, A. Pfas and Cancer, a Scoping Review of the Epidemiologic Evidence. Environ Res 2021, 194, 110690. [Google Scholar] [CrossRef]
- Tingley, K. Forever Chemicals Are Everywhere. What Are They Doing to Us? New York Times, August 16, 2023 2023.
- Li, Y.; Fletcher, T.; Mucs, D.; Scott, K.; Lindh, C.H.; Tallving, P.; Jakobsson, K. Half-Lives of Pfos, Pfhxs and Pfoa after End of Exposure to Contaminated Drinking Water. Occup Environ Med 2018, 75, 46–51. [Google Scholar] [CrossRef]
- Niu, S.; Cao, Y.; Chen, R.; Bedi, M.; Sanders, A.P.; Ducatman, A.; Ng, C. A State-of-the-Science Review of Interactions of Per- and Polyfluoroalkyl Substances (Pfas) with Renal Transporters in Health and Disease: Implications for Population Variability in Pfas Toxicokinetics. Environ Health Perspect 2023, 131, 76002. [Google Scholar] [CrossRef]
- Wen, Y.; Rashid, F.; Fazal, Z.; Singh, R.; Spinella, M.J.; Irudayaraj, J. Nephrotoxicity of Perfluorooctane Sulfonate (Pfos)-Effect on Transcription and Epigenetic Factors. Environ Epigenet 2022, 8, dvac010. [Google Scholar] [CrossRef]
- Durham, J.; Tessmann, J.W.; Deng, P.; Hennig, B.; Zaytseva, Y.Y. The Role of Perfluorooctane Sulfonic Acid (Pfos) Exposure in Inflammation of Intestinal Tissues and Intestinal Carcinogenesis. Front Toxicol 2023, 5, 1244457. [Google Scholar] [CrossRef]
- Zuo, X.; Tan, S.; Zhang, Y.; Zhang, C.; Ma, L.; Hou, X.; Wang, W.; Sun, R.; Yin, L.; Pu, Y.; Zhang, J. Linking Pfos Exposure to Chronic Kidney Disease: A Multimodal Study Integrating Epidemiology, Network Toxicology, and Experimental Validation. Ecotoxicol Environ Saf 2025, 302, 118770. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Glasgow, E.; Agarwal, S. Zebrafish Xenografts for Drug Discovery and Personalized Medicine. Trends Cancer 2020, 6, 569–79. [Google Scholar] [CrossRef] [PubMed]
- Martínez-López, M.F.; López-Gil, J.F. Small Fish, Big Answers: Zebrafish and the Molecular Drivers of Metastasis. Int J Mol Sci 26, no. 3 (2025).
- Siddiqui, S., H. Siddiqui, E. Riguene, and M. Nomikos. Zebrafish: A Versatile and Powerful Model for Biomedical Research. Bioessays (2025): e70080.
- Subcommittee, New Alternative Methods. Potential Approaches to Drive Future Integration of New Alternative Methods for Regulatory Decision-Making: A Report to the Science Board to the Food and Drug Administration from the New Alternative Methods Subcommittee. Food and Drug Administration, 2024.
- Morris-Schaffer, Keith, and Michael J. McCoy. A Review of the Ld50 and Its Current Role in Hazard Communication. ACS Chemical Health & Safety 2021, 28, 25–33. [Google Scholar]
- Hutchinson, T.H.; Bögi, C.; Winter, M.J.; Owens, J.W. Benefits of the Maximum Tolerated Dose (Mtd) and Maximum Tolerated Concentration (Mtc) Concept in Aquatic Toxicology. Aquat Toxicol 2009, 91, 197–202. [Google Scholar] [CrossRef]
- Obaidi, I., H. Cassidy, V. I. Gaspar, J. McCaul, M. Higgins, M. Halász, A. L. Reynolds, B. N. Kennedy, and T. McMorrow. Curcumin Sensitizes Kidney Cancer Cells to Trail-Induced Apoptosis Via Ros Mediated Activation of Jnk-Chop Pathway and Upregulation of Dr4. Biology (Basel) 9, no. 5 (2020).
- Kimmel, C.B.; Ballard, W.W.; Kimmel, S.R.; Ullmann, B.; Schilling, T.F. Stages of Embryonic-Development of the Zebrafish. Developmental Dynamics 1995, 203, 253–310. [Google Scholar] [CrossRef]
- Schindelin, Johannes, Ignacio Arganda-Carreras, Erwin Frise, Verena Kaynig, Mark Longair, Tobias Pietzsch, Stephan Preibisch, Curtis Rueden, Stephan Saalfeld, Benjamin Schmid, Jean-Yves Tinevez, Daniel James White, Volker Hartenstein, Kevin Eliceiri, Pavel Tomancak, and Albert Cardona. Fiji: An Open-Source Platform for Biological-Image Analysis. Nat Methods 2012, 9, 676–82. [Google Scholar] [CrossRef]
- Menger, F.; Pohl, J.; Ahrens, L.; Carlsson, G.; Orn, S. Behavioural Effects and Bioconcentration of Per- and Polyfluoroalkyl Substances (Pfass) in Zebrafish (Danio Rerio) Embryos. Chemosphere 2020, 245, 125573. [Google Scholar] [CrossRef] [PubMed]
- Hagenaars, A.; Vergauwen, L.; De Coen, W.; Knapen, D. Structure-Activity Relationship Assessment of Four Perfluorinated Chemicals Using a Prolonged Zebrafish Early Life Stage Test. Chemosphere 2011, 82, 764–72. [Google Scholar] [CrossRef]
- Shi, X.; Yeung, L.W.; Lam, P.K.; Wu, R.S.; Zhou, B. Protein Profiles in Zebrafish (Danio Rerio) Embryos Exposed to Perfluorooctane Sulfonate. Toxicol Sci 2009, 110, 334–40. [Google Scholar] [CrossRef]
- Shi, X.; Zhou, B. The Role of Nrf2 and Mapk Pathways in Pfos-Induced Oxidative Stress in Zebrafish Embryos. Toxicol Sci 2010, 115, 391–400. [Google Scholar] [CrossRef] [PubMed]
- Jantzen, C.E.; Annunziato, K.A.; Bugel, S.M.; Cooper, K.R. Pfos, Pfna, and Pfoa Sub-Lethal Exposure to Embryonic Zebrafish Have Different Toxicity Profiles in Terms of Morphometrics, Behavior and Gene Expression. Aquat Toxicol 2016, 175, 160–70. [Google Scholar] [CrossRef] [PubMed]
- Yang, Dan, Xiaohui Li, Shasha Dong, Xiaohui Zhao, Xiaoying Li, Meng Zhang, Yawei Shi, and Guanghui Ding. Developmental Toxicity and Cardiotoxicity Induced by Pfos and Its Novel Alternative Obs in Early Life Stage of Zebrafish (Danio Rerio). Water, Air, & Soil Pollution 2023, 234, 481. [Google Scholar] [CrossRef]
- Ding, Guang Hui, Jing Zhang, Yi Hong Chen, Guo Yi Luo, and Chao Hong Mao. Acute Toxicity Effect of Pfos on Zebrafish Embryo. Advanced Materials Research 2011, 356-360, 603-06.
- Zheng, Xin-Mei, Hong-Ling Liu, Wei Shi, Si Wei, John P. Giesy, and Hong-Xia Yu. Effects of Perfluorinated Compounds on Development of Zebrafish Embryos. Environmental Science and Pollution Research 2012, 19, 2498–505. [Google Scholar] [CrossRef]
- Cao, D.; Rericha, Y.; Powley, C.; Truong, L.; Tanguay, R.L.; Field, J.A. Background Per- and Polyfluoroalkyl Substances (Pfas) in Laboratory Fish Diet: Implications for Zebrafish Toxicological Studies. Sci Total Environ 2022, 842, 156831. [Google Scholar] [CrossRef]
- Rushing, Rosie, Christopher Schmokel, Bryan W. Brooks, and Matt F. Simcik. Occurrence of Per- and Polyfluoroalkyl Substance Contamination of Food Sources and Aquaculture Organisms Used in Aquatic Laboratory Experiments. Environ Toxicol Chem 2023, 42, 1463–71. [Google Scholar] [CrossRef]
- Lindahl, G.; Fjellander, S.; Selvaraj, K.; Vildeval, M.; Ali, Z.; Almter, R.; Erkstam, A.; Rodriguez, G.V.; Abrahamsson, A.; Kersley Å, R.; Fahlgren, A.; Kjølhede, P.; Linder, S.; Dabrosin, C.; Jensen, L. Zebrafish Tumour Xenograft Models: A Prognostic Approach to Epithelial Ovarian Cancer. NPJ Precis Oncol 2024, 8, 53. [Google Scholar] [CrossRef]
- Mendes, R.V.; Ribeiro, J.M.; Gouveia, H.; de Almeida, C.R.; Castillo-Martin, M.; Brito, M.J.; Canas-Marques, R.; Batista, E.; Alves, C.; Sousa, B.; Gouveia, P.; Ferreira, M.G.; Cardoso, M.J.; Cardoso, F.; Fior, R. Zebrafish Avatar Testing Preclinical Study Predicts Chemotherapy Response in Breast Cancer. NPJ Precis Oncol 2025, 9, 94. [Google Scholar] [CrossRef]
- Song, F.; Yi, X.; Zheng, X.; Zhang, Z.; Zhao, L.; Shen, Y.; Zhi, Y.; Liu, T.; Liu, X.; Xu, T.; Hu, X.; Zhang, Y.; Shou, H.; Huang, P. Zebrafish Patient-Derived Xenograft System for Predicting Carboplatin Resistance and Metastasis of Ovarian Cancer. Drug Resist Updat 2025, 78, 101162. [Google Scholar] [CrossRef] [PubMed]
- Costa, B.; Estrada, M.F.; Gomes, A.; Fernandez, L.M.; Azevedo, J.M.; Povoa, V.; Fontes, M.; Alves, A.; Galzerano, A.; Castillo-Martin, M.; Herrando, I.; Brandao, S.; Carneiro, C.; Nunes, V.; Carvalho, C.; Parvaiz, A.; Marreiros, A.; Fior, R. Zebrafish Avatar-Test Forecasts Clinical Response to Chemotherapy in Patients with Colorectal Cancer. Nat Commun 2024, 15, 4771. [Google Scholar] [CrossRef]
- Hua, X.; Wu, X.; Xu, K.; Zhan, P.; Liu, H.; Zhang, F.; Lv, T.; Song, Y. Zebrafish Patient-Derived Xenografts Accurately and Quickly Reproduce Treatment Outcomes in Non-Small Cell Lung Cancer Patients. Exp Biol Med (Maywood) 2023, 248, 361–69. [Google Scholar] [CrossRef]
- Azzam, N.; Fletcher, J.I.; Melong, N.; Lau, L.M.S.; Dolman, E.M.; Mao, J.; Tax, G.; Cadiz, R.; Tuzi, L.; Kamili, A.; Dumevska, B.; Xie, J.; Chan, J.A.; Senger, D.L.; Grover, S.A.; Malkin, D.; Haber, M.; Berman, J.N. Modeling High-Risk Pediatric Cancers in Zebrafish to Inform Precision Therapy. Cancer Res Commun 2025, 5, 1215–27. [Google Scholar] [CrossRef] [PubMed]
- Pierozan, P.; Karlsson, O. Pfos Induces Proliferation, Cell-Cycle Progression, and Malignant Phenotype in Human Breast Epithelial Cells. Arch Toxicol 2018, 92, 705–16. [Google Scholar] [CrossRef]
- Zheng, J.; Sun, B.; Berardi, D.; Lu, L.; Yan, H.; Zheng, S.; Aladelokun, O.; Xie, Y.; Cai, Y.; Pollitt, K.J.G.; Khan, S.A.; Johnson, C.H. Perfluorooctanesulfonic Acid and Perfluorooctanoic Acid Promote Migration of Three-Dimensional Colorectal Cancer Spheroids. Environ Sci Technol 2023, 57, 21016–28. [Google Scholar] [CrossRef]
- Beccacece, L., F. Costa, J. P. Pascali, and F. M. Giorgi. Cross-Species Transcriptomics Analysis Highlights Conserved Molecular Responses to Per- and Polyfluoroalkyl Substances. Toxics 11, no. 7 (2023).
- Limbu, S., and S. Dakshanamurthy. Predicting Dose-Dependent Carcinogenicity of Chemical Mixtures Using a Novel Hybrid Neural Network Framework and Mathematical Approach. Toxics 11, no. 7 (2023).
- Limbu, S., E. Glasgow, T. Block, and S. Dakshanamurthy. A Machine-Learning-Driven Pathophysiology-Based New Approach Method for the Dose-Dependent Assessment of Hazardous Chemical Mixtures and Experimental Validations. Toxics 12, no. 7 (2024).
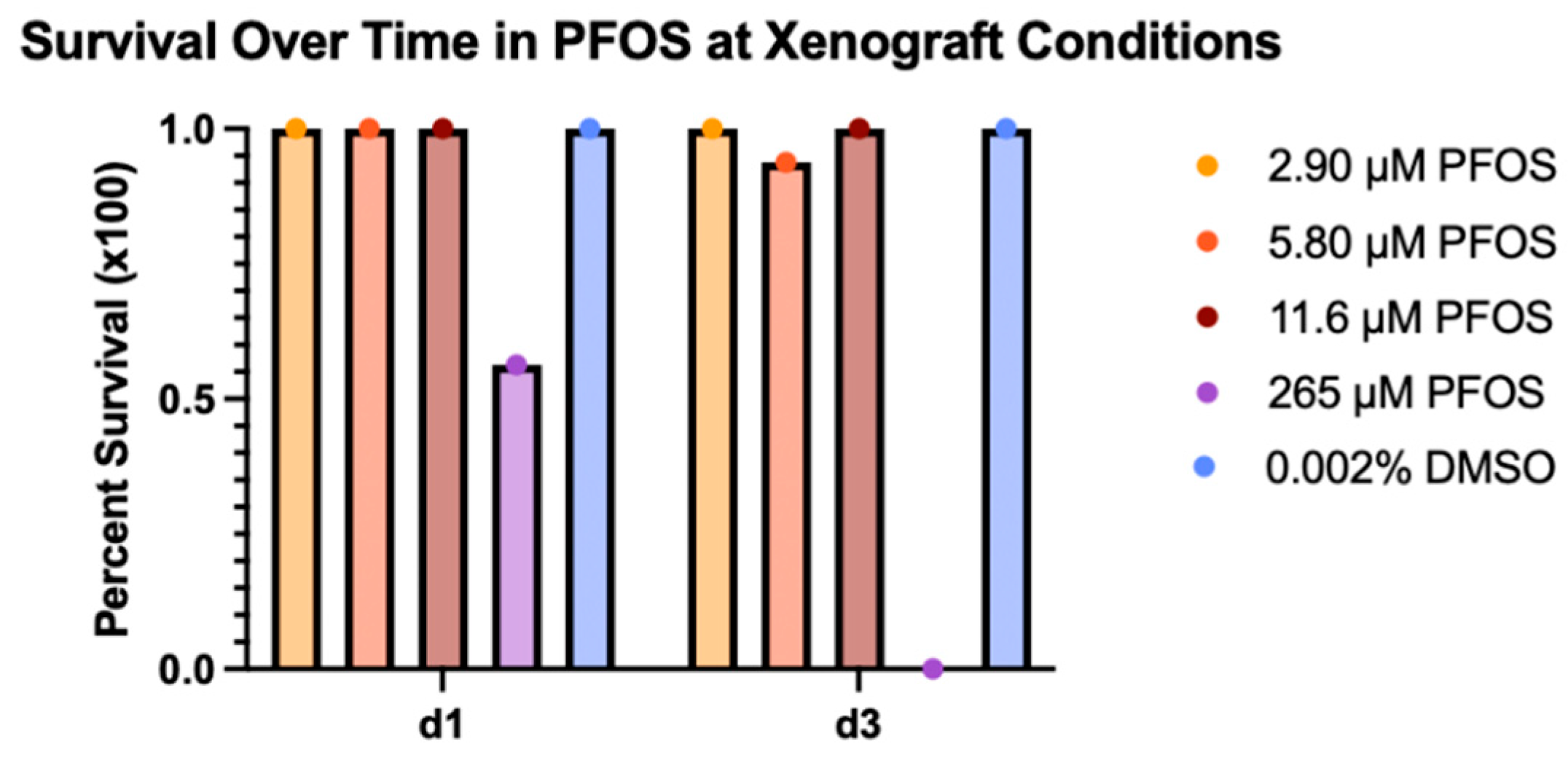
| Sample Set | LC50 (μM) | MTC (μM) | 5% of MTC (μM) | 10% of MTC (μM) | 20% of MTC (μM) |
| Range Finding | 167 | 73 | - | - | - |
| Refined Range | 132 | 58 | 2.9 | 5.8 | 11.6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




