Submitted:
24 November 2025
Posted:
24 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Ethical
2.3. Treatment of Tissues Samples
2.4. Structural and Immunohistochemical Diagnosis of Endometriosis
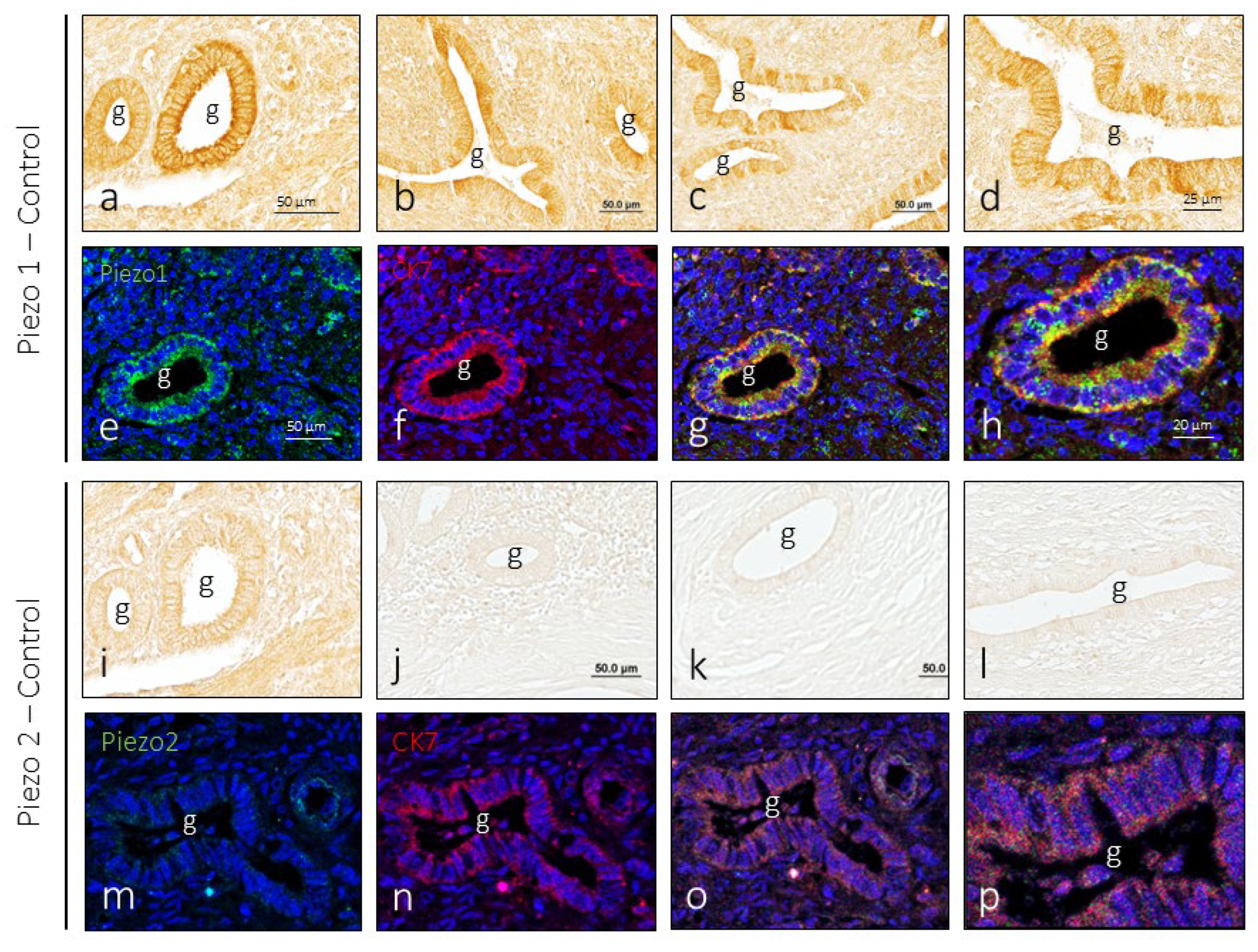
2.5. Simple Immunohistochemistry for PIEZOs
2.6. Evaluation of Intensity of Immunostaining for PIEZOs
2.6. Evaluation of Intensity of Immunostaining for PIEZOs
2.7. Double Immunofluorescence for Identification of PIEZO Positive Cells
3. Results
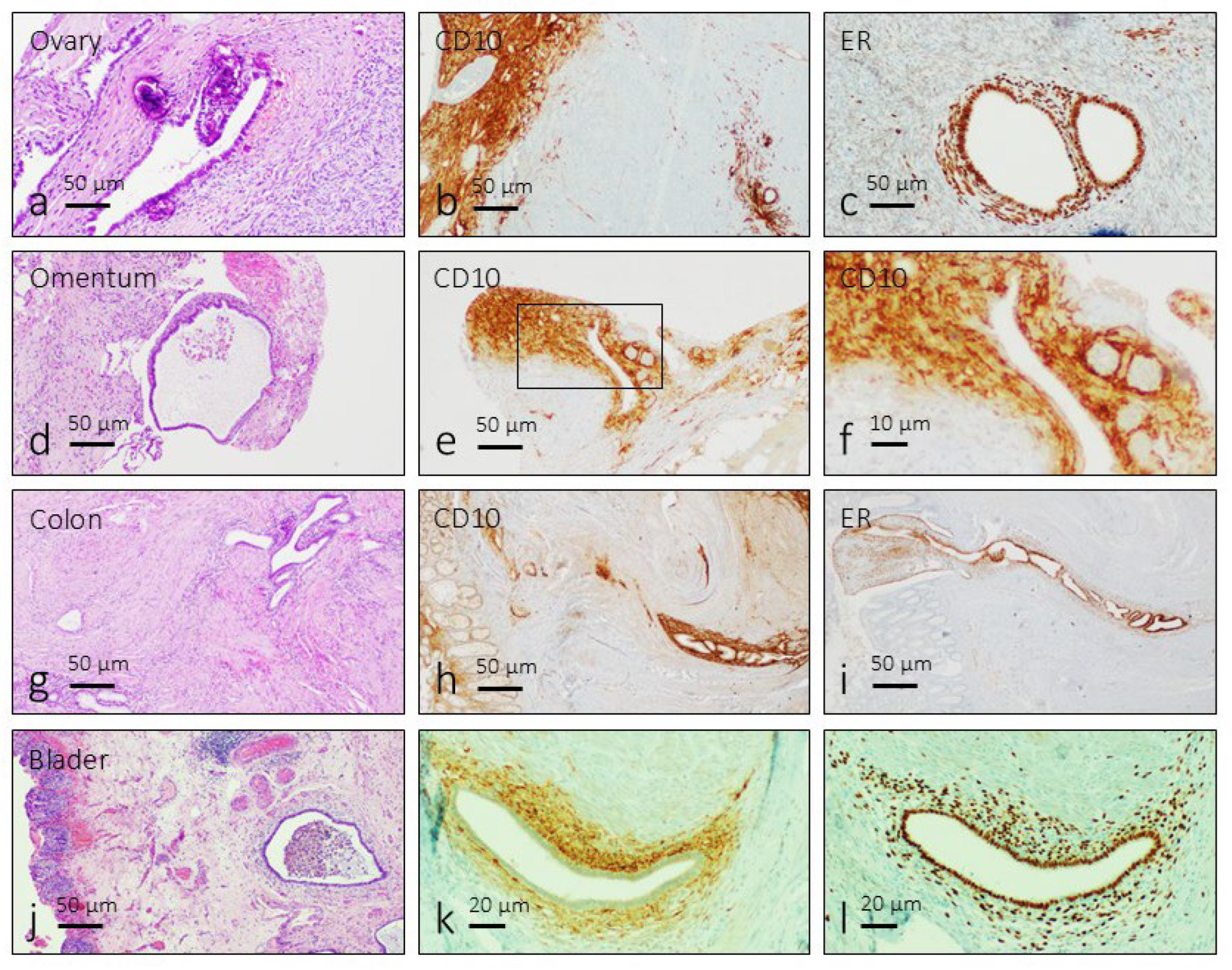
3.1. Identification of the Endometriosis Foci
3.2. Piezo1 and Piezo2 Immunoreactivity in the Endometrium
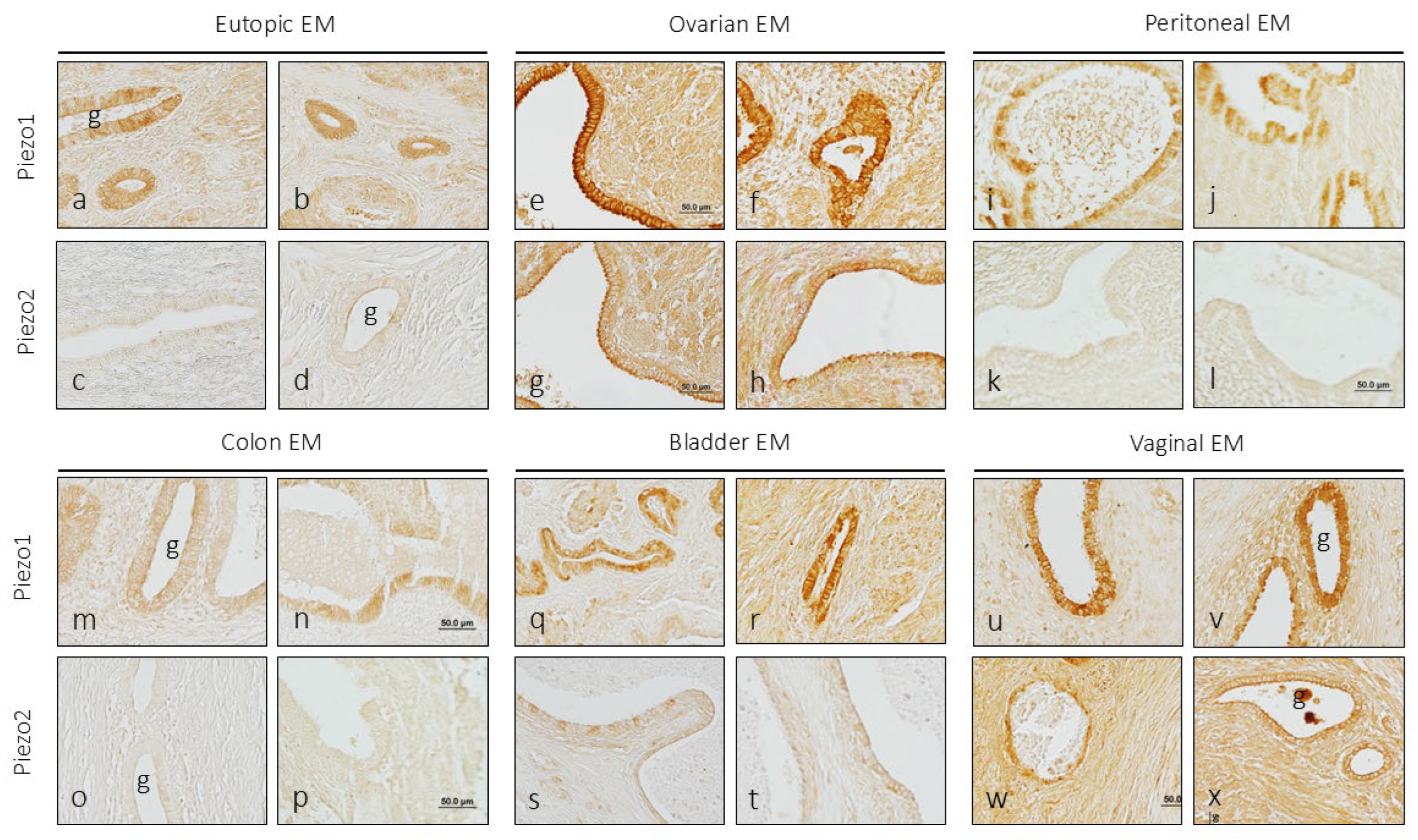
3.2. Localization of PIEZO1 and PIEZO2 in Endometriosis
3.2.1. Eutopic Endometriosis
3.2.2. Ovarian Endometriosis
3.2.3. Peritoneal Endometriosis
3.2.4. Intestinal Endometriosis
3.2.5. Vesical Endometriosis
3.2.6. Vaginal Endometriosis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- As-Sanie, S.; Black, R.; Giudice, L.C.; Gray Valbrun, T.; Gupta, J.; Jones, B.; Laufer, M.R.; Milspaw, A.T.; Missmer, S.A.; Norman, A.; et al. Assessing research gaps and unmet needs in endometriosis. Am. J. Obstet. Gynecol. 2019, 221, 86–94. [Google Scholar] [CrossRef]
- As-Sanie S, Mackenzie SC, Morrison L, Schrepf A, Zondervan KT, Horne AW, Missmer SA. Endometriosis: A Review. JAMA 2025, 334, 64–78. [Google Scholar] [CrossRef]
- Vannuccini, S.; Clemenza, S.; Rossi, M.; Petraglia, F. Hormonal treatments for endometriosis: The endocrine background. Rev. Endocr. Metab. Disord. 2022, 23, 333–355. [Google Scholar] [CrossRef]
- Bo, C.; Wang, Y. Angiogenesis signaling in endometriosis: Molecules, diagnosis and treatment (Review). Mol. Med. Rep. 2024, 29, 43. [Google Scholar] [CrossRef]
- Gruber, T.M.; Mechsner, S. Pathogenesis of Endometriosis: The Origin of Pain and Subfertility. Cells 2021, 10, 1381. [Google Scholar] [CrossRef]
- Velho, R.V.; Taube, E.; Sehouli, J.; Mechsner, S. Neurogenic Inflammation in the Context of Endometriosis-What Do We Know? Int. J. Mol. Sci. 2021, 22, 13102. [Google Scholar] [CrossRef]
- Mariadas, H.; Chen, J.H.; Chen, K.H. The Molecular and Cellular Mechanisms of Endometriosis: From Basic Pathophysiology to Clinical Implications. Int. J. Mol. Sci. 2025, 26, 2458. [Google Scholar] [CrossRef]
- Wang, Y.; Nicholes, K.; Shih, I.M. The Origin and Pathogenesis of Endometriosis. Annu. Rev. Pathol. 2020, 15, 71–95. [Google Scholar] [CrossRef]
- Kobayashi, H.; Imanaka, S.; Yoshimoto, C.; Matsubara, S.; Shigetomi, H. Rethinking the pathogenesis of endometriosis: Complex interactions of genomic, epigenetic, and environmental factors. J. Obstet. Gynaecol. Res. 2024, 50, 1771–1784. [Google Scholar] [CrossRef]
- Parasar, P.; Ozcan, P.; Terry, K.L. Endometriosis: Epidemiology, Diagnosis and Clinical Management. Curr. Obstet. Gynecol. Rep. 2017, 6, 34–41. [Google Scholar] [CrossRef]
- Kou, L.; Huang, C.; Xiao, D.; Liao, S.; Li, Y.; Wang, Q. Pharmacologic Interventions for Endometriosis-Related Pain: A Systematic Review and Meta-analysis. Obstet. Gynecol. 2025, 146, e23–e35. [Google Scholar] [CrossRef]
- Mick, I.; Freger, S.M.; van Keizerswaard, J.; Gholiof, M.; Leonardi, M. Comprehensive endometriosis care: a modern multimodal approach for the treatment of pelvic pain and endometriosis. Ther. Adv. Reprod. Health 2024, 18, 26334941241277759. [Google Scholar] [CrossRef]
- Coste, B.; Mathur, J.; Schmidt, M.; Earley, T.J.; Ranade, S.; Petrus, M.J.; Dubin, A.E.; Patapoutian, A. Piezo1 and Piezo2 are essential components of distinct mechanically activated cation channels. Science 2010, 330, 55–60. [Google Scholar] [CrossRef]
- Murthy, S.E.; Dubin, A.E.; Patapoutian, A. Piezos thrive under pressure: mechanically activated ion channels in health and disease. Nat. Rev. Mol. Cell Biol. 2017, 18, 771–783. [Google Scholar] [CrossRef]
- Delmas, P.; Parpaite, T.; Coste, B. PIEZO channels and newcomers in the mammalian mechanosensitive ion channel family. Neuron 2022, 110, 2713–2727. [Google Scholar] [CrossRef]
- Xiao, B. Mechanisms of mechanotransduction and physiological roles of PIEZO channels. Nat. Rev. Mol. Cell Biol. 2024, 25, 886–903. [Google Scholar] [CrossRef]
- Sforna, L.; Michelucci, A.; Morena, F.; Argentati, C.; Franciolini, F.; Vassalli, M.; Martino, S.; Catacuzzeno, L. Piezo1 controls cell volume and migration by modulating swelling-activated chloride current through Ca2+ influx. J. Cell Physiol. 2022, 237, 1857–1870. [Google Scholar] [CrossRef]
- Tang, Y.; Zhao, C.; Zhuang, Y.; Zhong, A.; Wang, M.; Zhang, W.; Zhu, L. Mechanosensitive Piezo1 protein as a novel regulator in macrophages and macrophage-mediated inflammatory diseases. Front. Immunol. 2023, 14, 1149336. [Google Scholar] [CrossRef]
- Du, S.; Liu, K. Mechanosensitive ion channels and inflammation: key links in cellular signal transduction. Inflamm. Res. 2025, 74, 104. [Google Scholar] [CrossRef]
- Pirri, C. PIEZO Channels in Mechano-Inflammation: Gatekeepers of Neuroimmune Crosstalk. Diseases 2025, 13, 263. [Google Scholar] [CrossRef]
- Alibrandi, S.; Rinaldi, C.; Vinci, S.L.; Conti, A.; Donato, L.; Scimone, C.; Sidoti, A.; D’Angelo, R. Mechanotransduction in Development: A Focus on Angiogenesis. Biology (Basel) 2025, 14, 346. [Google Scholar] [CrossRef]
- Di, X.; Gao, X.; Peng, L.; Ai, J.; Jin, X.; Qi, S.; Li, H.; Wang, K.; Luo, D. Cellular mechanotransduction in health and diseases: from molecular mechanism to therapeutic targets. Signal Transduct Target Ther. 2023, 8, 282. [Google Scholar] [CrossRef]
- Xu, Y.; Huang, Y.; Cheng, X.; Hu, B.; Jiang, D.; Wu, L.; Peng, S.; Hu, J. Mechanotransductive receptor Piezo1 as a promising target in the treatment of fibrosis diseases. Front. Mol. Biosci. 2023, 10, 1270979. [Google Scholar] [CrossRef] [PubMed]
- Zheng, M.; Borkar, N.A.; Yao, Y.; Ye, X.; Vogel, E.R.; Pabelick, C.M.; Prakash, Y.S. Mechanosensitive channels in lung disease. Front. Physiol. 2023, 14, 1302631. [Google Scholar] [CrossRef] [PubMed]
- Drobnik, M.; Smólski, J.; Grądalski, Ł.; Niemirka, S.; Młynarska, E.; Rysz, J.; Franczyk, B. Mechanosensitive Cation Channel Piezo1 Is Involved in Renal Fibrosis Induction. Int. J. Mol. Sci. 2024, 25, 1718. [Google Scholar] [CrossRef]
- Wan, Y.; Zhou, J.; Li, H. The Role of Mechanosensitive Piezo Channels in Chronic Pain. J. Pain Res. 2024, 17, 4199–4212. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, Y.; Mei, S.; Hu, J.; Wu, L.; Xu, L.; Bao, L.; Fang, X. The mechanism and potential therapeutic target of piezo channels in pain. Front. Pain Res. (Lausanne) 2024, 5, 1452389. [Google Scholar] [CrossRef]
- Liu, C.; Li, H.; Hang, L. The research progress into cellular mechanosensitive ion channels mediating cancer pain. Channels (Austin) 2025, 19, 2517109. [Google Scholar] [CrossRef]
- Burridge, K.; Monaghan-Benson, E.; Graham, D.M. Mechanotransduction: from the cell surface to the nucleus via RhoA. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2019, 374, 20180229. [Google Scholar] [CrossRef]
- Martino, S. Mechanobiology in Cells and Tissues. Int. J. Mol. Sci. 2023, 24. [Google Scholar] [CrossRef] [PubMed]
- Martino, F.; Perestrelo, A.R.; Vinarský, V.; Pagliari, S.; Forte, G. Cellular Mechanotransduction: From Tension to Function. Front. Physiol. 2018, 9, 824. [Google Scholar] [CrossRef]
- Lacroix, J.J.; Wijerathne, T.D. PIEZO channels as multimodal mechanotransducers. Biochem. Soc. Trans. 2025, 53, BST20240419. [Google Scholar] [CrossRef]
- Hennes, A.; Held, K.; Boretto, M.; De Clercq, K.; Van den Eynde, C.; Vanhie, A.; Van Ranst, N.; Benoit, M.; Luyten, C.; Peeraer, K.; Tomassetti, C.; Meuleman, C.; Voets, T.; Vankelecom, H.; Vriens, J. Functional expression of the mechanosensitive PIEZO1 channel in primary endometrial epithelial cells and endometrial organoids. Sci. Rep. 2019, 9, 1779. [Google Scholar] [CrossRef]
- Arishe, O.O.; Ebeigbe, A.B.; Webb, R.C. Mechanotransduction and Uterine Blood Flow in Preeclampsia: The Role of Mechanosensing Piezo 1 Ion Channels. Am. J. Hypertens. 2020, 33, 1–9. [Google Scholar] [CrossRef]
- Toki, T.; Shimizu, M.; Takagi, Y.; Ashida, T.; Konishi, I. CD10 is a marker for normal and neoplastic endometrial stromal cells. Int. J. Gynecol. Pathol. 2002, 21, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Potlog-Nahari, C.; Feldman, A.L.; Stratton, P.; Koziol, D.E.; Segars, J.; Merino, M.J.; Nieman, L.K. CD10 immunohistochemical staining enhances the histological detection of endometriosis. Fertil. Steril. 2004, 82, 86–92. [Google Scholar] [CrossRef]
- Chantalat, E.; Valera, M.C.; Vaysse, C.; Noirrit, E.; Rusidze, M.; Weyl, A.; Vergriete, K.; Buscail, E.; Lluel, P.; Fontaine, C.; Arnal, J.F.; Lenfant, F. Estrogen Receptors and Endometriosis. Int. J. Mol. Sci. 2020, 21, 2815. [Google Scholar] [CrossRef] [PubMed]
- Coroleucă, C.A.; Coroleucă, C.B.; Coroleucă, R.; Brătilă, P.C.; Nodiți, A.R.; Roșca, I.; Brîndușe, L.A.; Brătilă, E.; Boț, M. Molecular Profile (Estrogen Receptor, Progesterone Receptor, Bcl-2 and Ki-67) of the Ectopic Endometrium in Patients with Endometriosis. Int. J. Mol. Sci. 2025, 26, 2983. [Google Scholar] [CrossRef]
- Syeda, R. Physiology and Pathophysiology of Mechanically Activated PIEZO Channels. Annu Rev Neurosci. 2021, 44, 383–402. [Google Scholar] [CrossRef]
- Poole, K. The Diverse Physiological Functions of Mechanically Activated Ion Channels in Mammals. Annu. Rev. Physiol. 2022, 84, 307–329. [Google Scholar] [CrossRef] [PubMed]
- Di, X.; Gao, X.; Peng, L.; Ai, J.; Jin, X.; Qi, S.; Li, H.; Wang, K.; Luo, D. Cellular mechanotransduction in health and diseases: from molecular mechanism to therapeutic targets. Signal Transduct Target Ther. 2023, 8, 282. [Google Scholar] [CrossRef]
- Xin, Liu.; Weipin, Niu.; Shuqing, Zhao.; Wenjuan, Zhang.; Ying, Zhao.; Jing, Li. Piezo1. the potential new therapeutic target for fibrotic disease. Progress in Biophysics and molecular Biology 2023, 184, 42–49. [Google Scholar] [CrossRef]
- Karska, J.; Kowalski, S.; Saczko, J.; Moisescu, M.G.; Kulbacka, J. Mechanosensitive Ion Channels and Their Role in Cancer Cells. Membranes (Basel). 2023, 13, 167. [Google Scholar] [CrossRef]
- Alper, S.L. Genetic Diseases of PIEZO1 and PIEZO2 Dysfunction. Curr. Top. Membr. 2017, 79, 97–134. [Google Scholar] [CrossRef] [PubMed]
- Piddini, E. Epithelial Homeostasis: A Piezo of the Puzzle. Curr. Biol. 2017, 27, R232–R234. [Google Scholar] [CrossRef]
- García-Mesa, Y.; Cuendias, P.; Alonso-Guervós, M.; García-Piqueras, J.; Martín-Biedma, B.; Cobo, T.; García-Suárez, O.; Vega, J.A. Immunohistochemical detection of PIEZO1 and PIEZO2 in human digital Meissner’s corpuscles. Ann. Anat. 2024, 252, 152200. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Zhou, J.; Xu, X.; Zhou, P.; Zhong, H.; Liu, M. Piezo channels in the intestinal tract. Front. Physiol. 2024, 15, 1356317. [Google Scholar] [CrossRef] [PubMed]
- Pyo, I.H.; Yoon, Y.B.; Jeong, G.H.; Park, S.C.; Lee, G.W.; Aryal, Y.P.; Kwak, H.J.; Cho, S.J. Unveiling salivary gland-specific gene expression of Piezo1 and Neuroendocrine in the leech, Helobdella austinensis. Dev. Comp. Immunol. 2025, 168, 105391. [Google Scholar] [CrossRef]
- Sternberg, A.K.; Buck, V.U.; Classen-Linke, I.; Leube, R.E. How Mechanical Forces Change the Human Endometrium during the Menstrual Cycle in Preparation for Embryo Implantation. Cells. 2021, 10, 2008. [Google Scholar] [CrossRef]
- Davoodi, Nik. B.; Hashemi Karoii, D.; Favaedi, R.; Ramazanali, F.; Jahangiri, M.; Movaghar, B.; Shahhoseini, M. Differential expression of ion channel coding genes in the endometrium of women experiencing recurrent implantation failures. Sci. Rep. 2024, 14, 19822. [Google Scholar] [CrossRef]
- Lou, W.; Liu, J.; Ding, B.; Jin, L.; Xu, L.; Li, X.; Chen, J.; Fan, W. Five miRNAs-mediated PIEZO2 downregulation, accompanied with activation of Hedgehog signaling pathway, predicts poor prognosis of breast cancer. Aging (Albany NY). 2019, 11, 2628–2652. [Google Scholar] [CrossRef]
- Zondervan, K.T.; Becker, C.M.; Missmer, S.A. Endometriosis. N. Engl. J. Med. 2020, 382, 1244–1256. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Hang, L. Mechanical gated ion channel Piezo1: Function, and role in macrophage inflammatory response. Innate Immun 2024, 30, 32–39. [Google Scholar] [CrossRef]
- Dubin, A.E.; Schmidt, M.; Mathur, J.; Petrus, M.J.; Xiao, B.; Coste, B.; Patapoutian, A. Inflammatory signals enhance piezo2-mediated mechanosensitive currents. Cell Rep. 2012, 2, 511–517. [Google Scholar] [CrossRef]
- Szczot, M.; Liljencrantz, J.; Ghitani, N.; Barik, A.; Lam, R.; Thompson, J.H.; Bharucha-Goebel, D.; Saade, D.; Necaise, A.; Donkervoort, S.; Foley, A.R.; Gordon, T.; Case, L.; Bushnell, M.C.; Bönnemannn, C.G.; Chesler, A.T. PIEZO2 mediates injury-induced tactile pain in mice and humans. Sci. Transl. Med. 2018, 10, eaat9892. [Google Scholar] [CrossRef]
- Romero, L.O.; Caires, R.; Nickolls, A.R.; Chesler, A.T.; Cordero-Morales, J.F.; Vásquez, V. A dietary fatty acid counteracts neuronal mechanical sensitization. Nat Commun. 2020, 11, 2997, Erratum in: Nat Commun. 2020, 11, 3938. [Google Scholar] [CrossRef]
- De Logu, F.; Geppetti, P. Ion Channel Pharmacology for Pain Modulation. Handb. Exp. Pharmacol. 2019, 260, 161–186. [Google Scholar] [CrossRef]
- Kinsella, J.A.; Debant, M.; Parsonage, G.; Morley, L.C.; Bajarwan, M.; Revill, C.; Foster, R.; Beech, D.J. Pharmacology of PIEZO1 channels. Br. J. Pharmacol. 2024, 181, 4714–4732. [Google Scholar] [CrossRef] [PubMed]
- Thien, N.D.; Hai-Nam, N.; Anh, D.T.; Baecker, D. Piezo1 and its inhibitors: Overview and perspectives. Eur. J. Med. Chem. 2024, 273, 116502. [Google Scholar] [CrossRef] [PubMed]
| Age (mean ± SD) | 35,10 ± 8,30 | |
| Chief complaint | Dysmenorrhea | 30 (100%) |
| Dyspareunia | 7 (23,3%) | |
| Dyschezia | 6 (20%) | |
| Dysuria | 4 (13,3%) | |
| Sterility | 16 (53,3%) | |
| Visual analogue scale (VAS) | 0-3 | 3 (10%) |
| 4-7 | 10 (33,3%) | |
| >7 | 17 (56,66%) | |
| Laparoscopic and pathological findings | Ovarian endometriosis | 10 (37,93%) |
| Peritoneal and digestive endometriosis | 10 (34,48%) | |
| Other endometriosis localization (vesical, vaginal, abdominal wall) | 5 (13,79%) | |
| Eutopic endometrium from endometriosis | 5 (13,79%) | |
| Staging of endometriosis | III | 8 (27,58%) |
| IV | 22 (68,96)% |
| Antigen | Host/Origin | Dilution | Supplier |
|---|---|---|---|
| Piezo 1* | Rabbit polyclonal | 1:100 | Invitrogen1 |
| Piezo 2** | Rabbit polyclonal | 1:100 | Sigma-Aldrich2 |
| CD10 (SP67) | Rabbit monoclonal | Prediluted | Roche3 |
| anti-Estrogen receptor (ER) (SP1) | Rabbit monoclonal | Prediluted | Roche3 |
| anti-Progesterone receptor (PR) (1E2) | Rabbit monoclonal | Prediluted | Roche3 |
| NFP (2F11) | Mouse monoclonal | 1:200 | Roche3 |
| CK7 | Mouse monoclonal | Prediluted | Dako4 |
| S100P (S100A1) | Rabbit polyclonal | 1:500 | Dako4 |
| Antigen | PIEZO 1 | PIEZO2 | Stroma P1/P2 |
|---|---|---|---|
| Endometrium | +/++ | -/+ | +/+ |
| Eutopic EM | +/++ | -/+ | +/- |
| Ovarian EM | ++/+++ | ++ | ++/++ |
| Peritoneal EM | +/++ | -/+ | +/- |
| Intestinal EM | ++ | -/+ | |
| Vesical EM | +++ | -/+ | |
| Vaginal EM | ++/+++ | ++ | ++/++ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




