Submitted:
21 November 2025
Posted:
24 November 2025
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
Cell Culture
Protein Expression via Flow Cytometry
Proliferation and Apoptosis
Statistical Analysis
Results
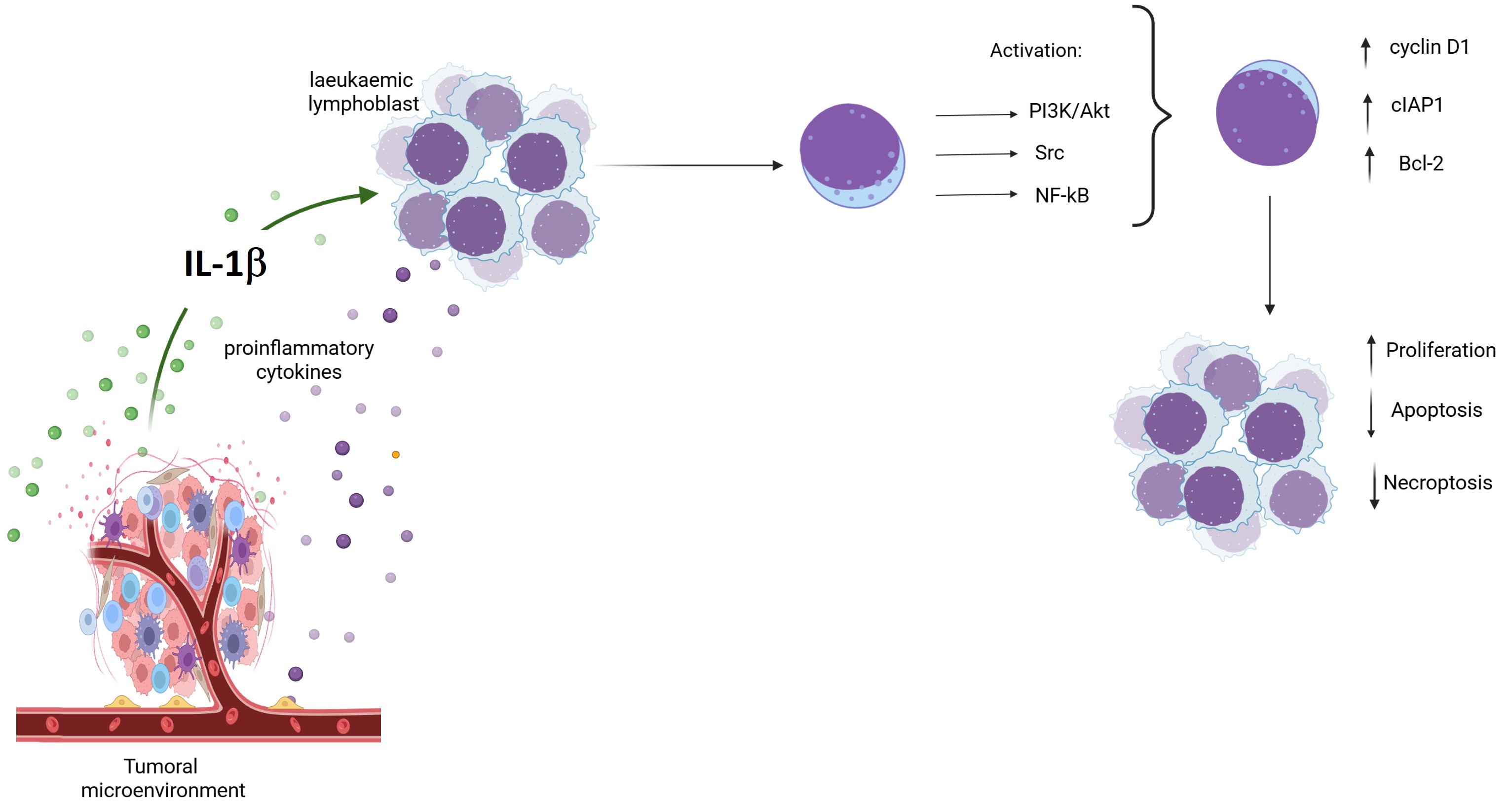
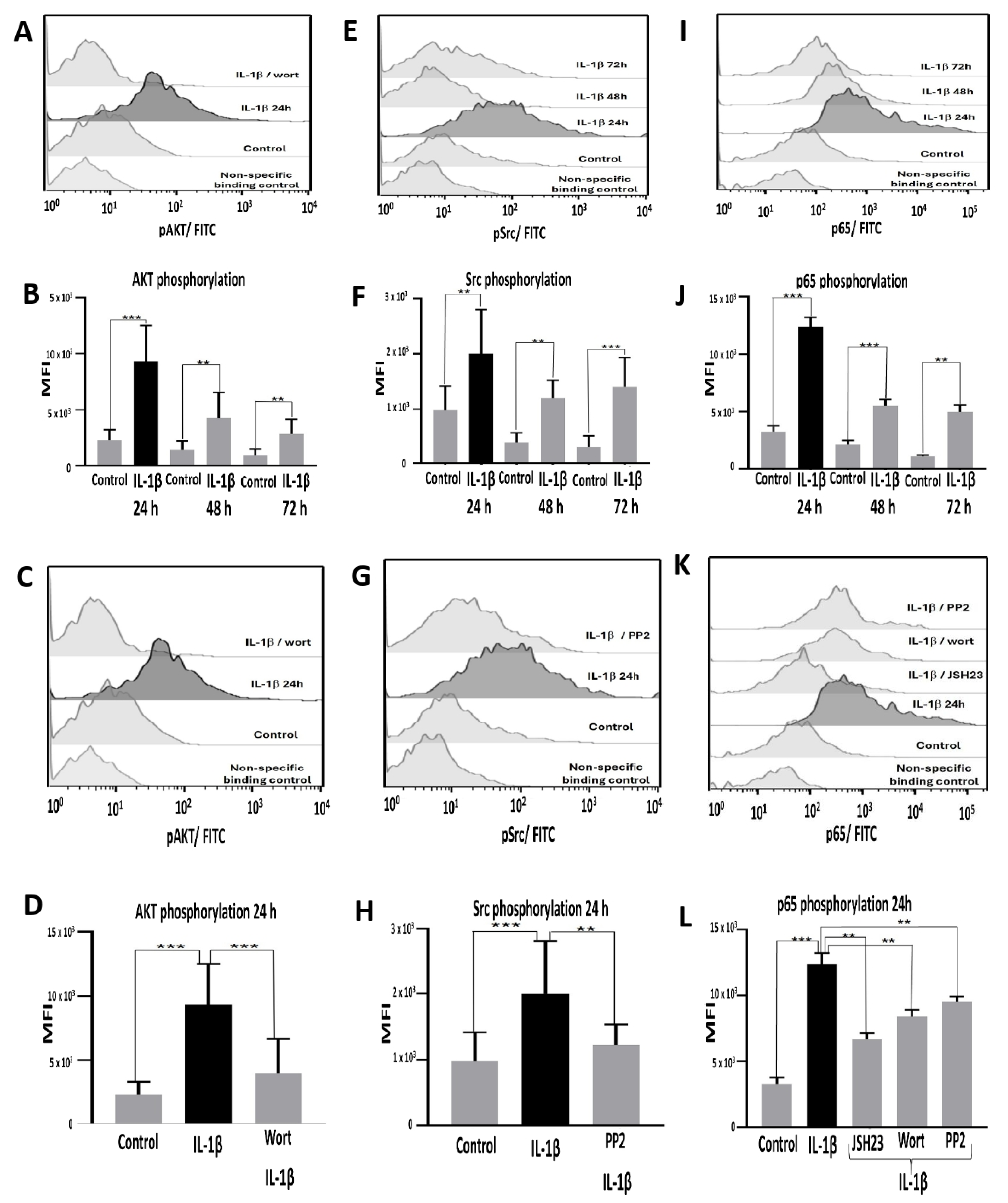
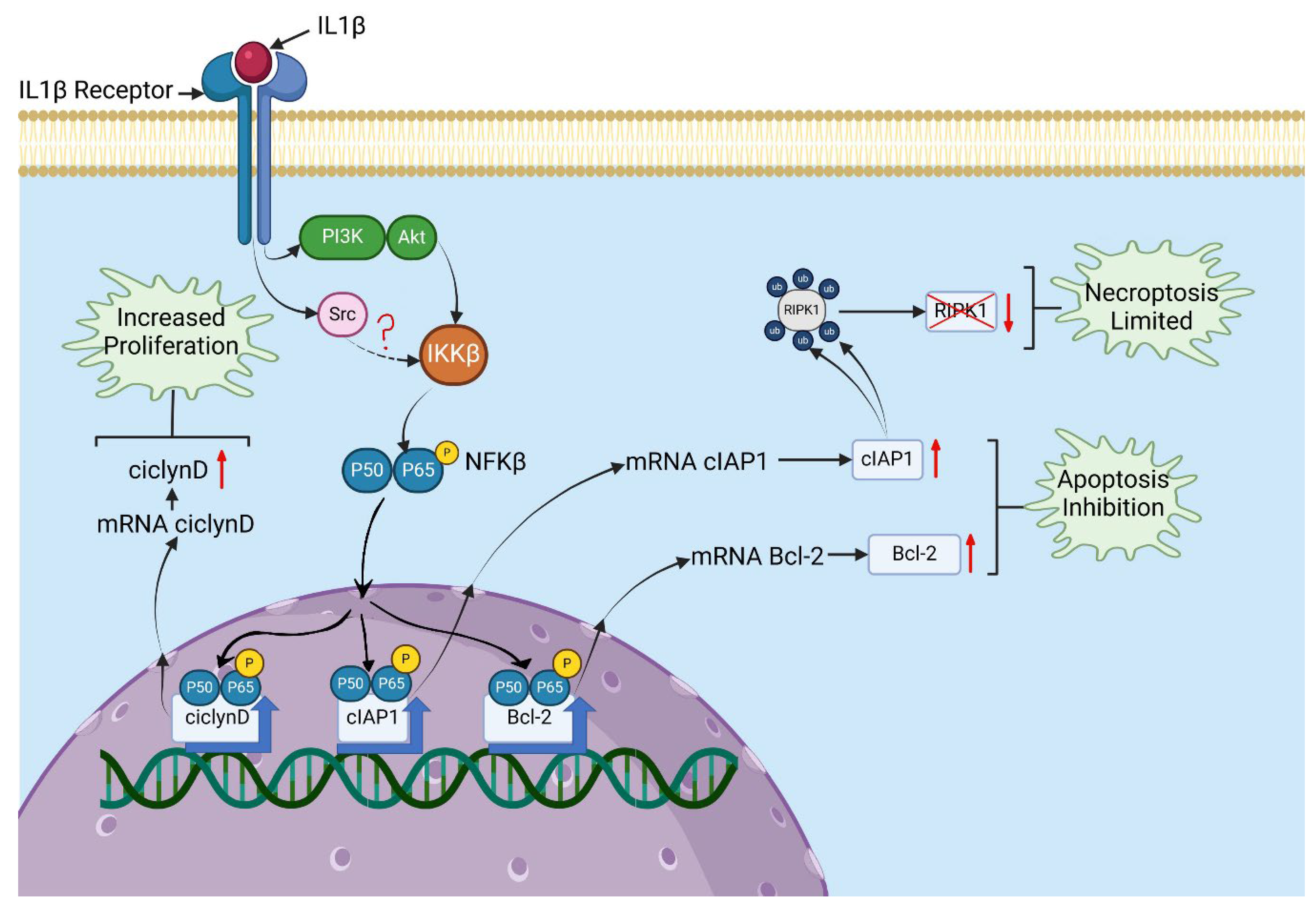
IL-1β Induces AKT, Src, and NF-κB Activation in RS4:11 Leukaemic Lymphoblasts
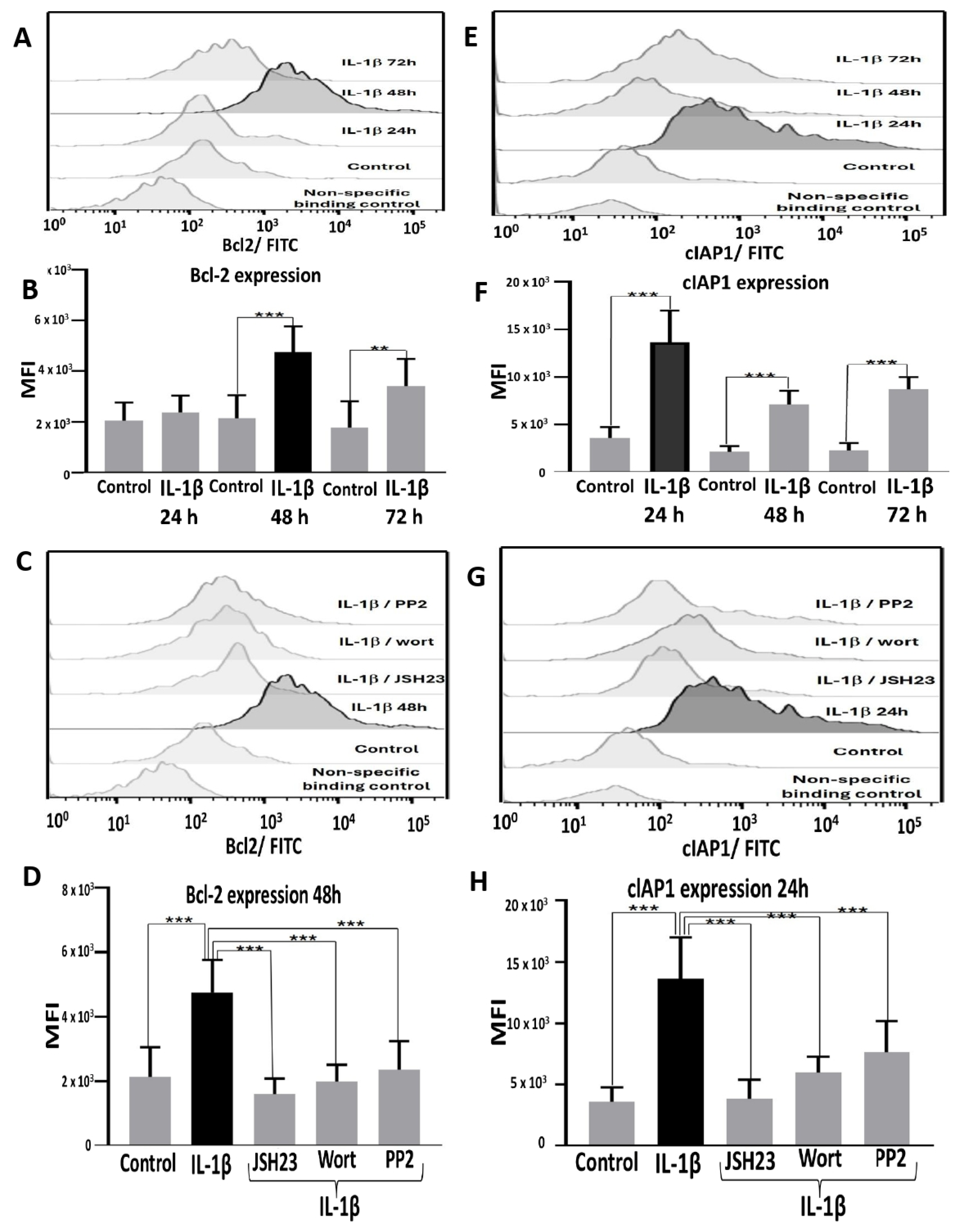
IL-1β Influences Expression of Bcl-2 and cIAP1 via PI3K/AKT/NF-κB Pathway in RS4:11 Leukaemic Lymphoblasts
IL-1β Decreases Apoptosis and Limits Necroptosis via the PI3K/AKT /Src/NF-κB Pathway in RS4:11 Leukaemic Lymphoblasts
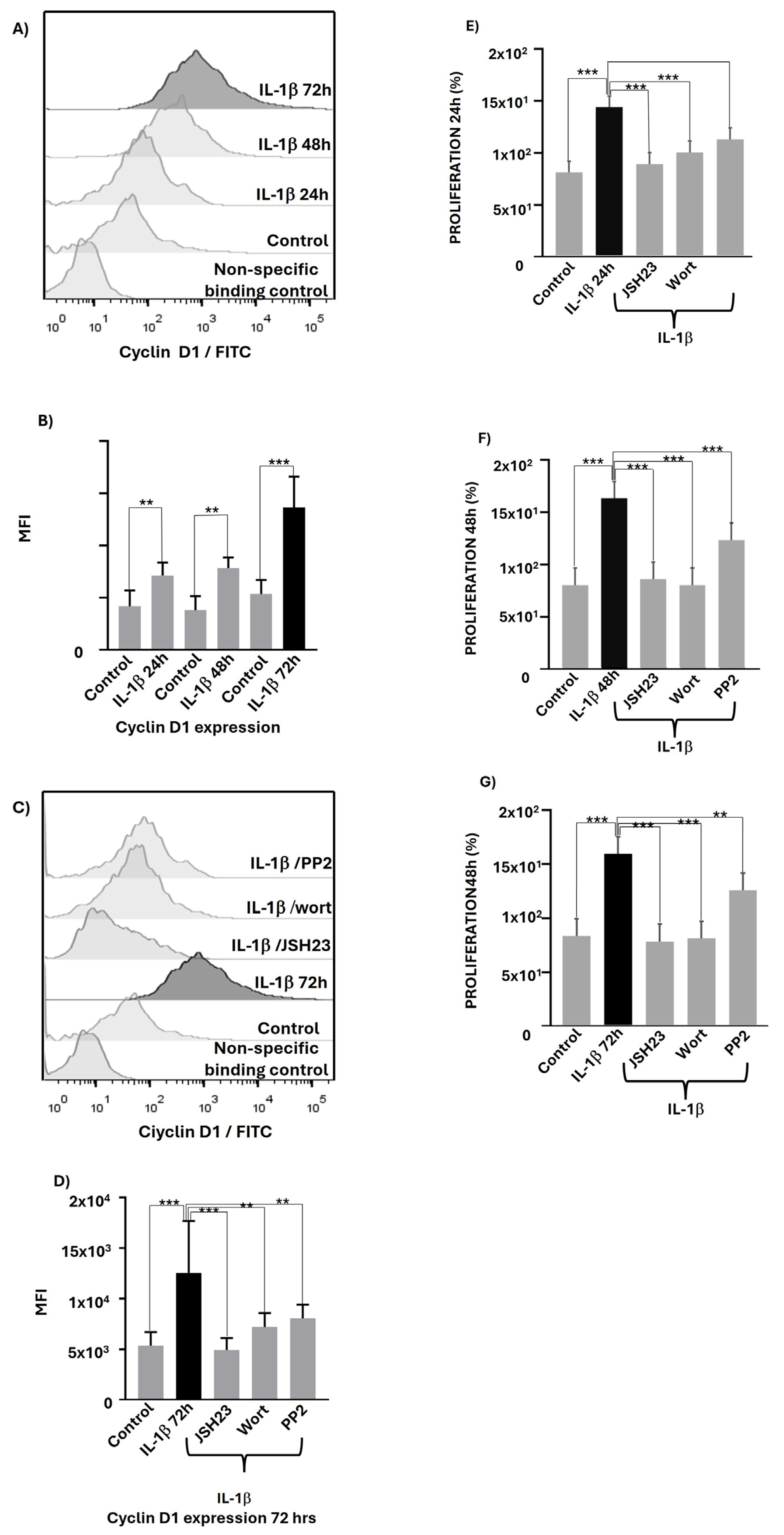
IL-1β via PI3K/AKT /Src/NF-κB Influences Cyclin D1 Expression and Proliferation of RS4:11 Leukaemic Lymphoblasts
Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
References
- Braun T., Carvalho G., et al. Targeting NF-kB in hematologic malignancies. Cell Death Differ. 2006. 13, 748-758. [CrossRef]
- Naugler WE, Karin M. NF-kappaB and cancer-identifying targets and mechanisms. Curr Opin Genet Dev. 2008.18:19-26. [CrossRef]
- Grivennikov SI, Greten FR., et al. Immunity, inflammation, and cancer. Cell. 2010. 140:883-99. [CrossRef]
- Darnay, B. G., Aggarwal, B. Human cytokines. In Advances in Oncobiology. Pergamon Press.UK. ISBN 0-7623-016-5. 1996. Vol. 1, pp. 179–205.
- Bent R, Moll L., et al. Interleukin-1 Beta—A Friend or Foe in Malignancies. International Journal of Molecular Sciences. 2018. 8: 2155. [CrossRef]
- Krzystek-Korpacka M, Diakowska D., et al. Profiles of circulating inflammatory cytokines in colorectal cancer (CRC), high cancer risk conditions, and health are distinct. Possible implications for CRC screening and surveillance. Cancer Lett. 2013.337:107-14. [CrossRef]
- Matti BF, Saleem MA., et al. Assessment of interleukin 1β serum level in different responder groups and stages of chronic myeloid leukemia patients on imatinb mesylate therapy. Indian J Hematol Blood Transfus. 2014. 30:247-52. [CrossRef]
- Dolcet X, Llobet D, et al. NF-kB in development and progression of human cancer. Virchows Arch. 2005. 446:475-82. [CrossRef]
- Chan KW. Acute lymphoblastic leukemia, Current Problems in Pediatric and Adolescent Health Care. 2002. 32:40-49. [CrossRef]
- Sizemore N, Leung S., et al. Activation of phosphatidylinositol 3-kinase in response to interleukin-1 leads to phosphorylation and activation of the NF-kappaB p65/RelA subunit. Mol Cell Biol. 1999. 19:4798-805. [CrossRef]
- Iliopoulos D, Hirsch HA., et al. An epigenetic switch involving NF-kappaB, Lin28, Let-7 MicroRNA, and IL6 links inflammation to cell transformation. Cell. 2009. 139:693-706. [CrossRef]
- American Type Culture Collection. (2025). RS4;11 cell line (CRL-1873). Manassas, VA, USA.https://www.atcc.org/products/crl-1873-.
- Cyton. (2025). RS4:11 cells. Available at: https://www.cytion.com/es/RS4-11-Celulas/305360.
- Nakamura I, Takahashi N, et al. Wortmannin, a specific inhibitor of phosphatidylinositol-3 kinase, blocks osteoclastic bone resorption.FEBS Lett. 1995 Mar 13;361(1):79-84. [CrossRef]
- Dai X, Wang LJ, et al. Src kinase inhibitor PP2 regulates the biological characteristics of A549 cells via the PI3K/AKT signaling pathway.Oncol Lett. 2018.16 (4):5059-5065. [CrossRef]
- Chen X., Liu G., et al. NEK7 interacts with NLRP3 to modulate the pyroptosis in inflammatory bowel disease via NF-κB signaling. Cell Death Dis. 2019;10:906. [CrossRef]
- Wang CY, Guttridge DC., et al. NF-kappaB induces expression of the Bcl-2 homologue A1/Bfl-1 to preferentially suppress chemotherapy-induced apoptosis. Mol Cell Biol. 1999. 19:5923-9. [CrossRef]
- Ahmad I, Irfan S., et al. The SMAC mimetic AT-101 exhibits anti-tumor in lung adenocarcinoma cells by the IAPs/ caspase-dependent apoptosis and p65-NFƙB cross-talk. Iran J Basic Med Sci. 2021. 24:969-977. [CrossRef]
- Cai Z, Wang J., et al. Overexpressed Cyclin D1 and CDK4 proteins are responsible for the resistance to CDK4/6 inhibitor in breast cancer that can be reversed by PI3K/mTOR inhibitors. Sci China Life Sci. 2023. 66:94-109. [CrossRef]
- Coussens LM, Werb Z. Inflammation and cancer. Nature. 2002.420:860-7. [CrossRef]
- Turzanski J, Grundy M., et al. Interleukin-1beta maintains an apoptosis-resistant phenotype in the blast cells of acute myeloid leukaemia via multiple pathways. Leukemia. 2004. 18:1662-70. [CrossRef]
- Beaupre DM, Talpaz M., et al. Autocrine interleukin-1beta production in leukemia: evidence for the involvement of mutated RAS. Cancer Res. 1999. 15:2971-80. PMID: 10383163.
- Reddy SA, Huang JH., et al. Phosphatidylinositol 3-kinase in interleukin 1 signaling. Physical interaction with the interleukin 1 receptor and requirement in NFkappaB and AP-1 activation. J Biol Chem. 1997. 272:29167-73. [CrossRef]
- Lin MT, Lin BR., et al. IL-6 induces AGS gastric cancer cell invasion via activation of the c-Src/RhoA/ROCK signaling pathway. Int J Cancer. 2007. 120: 2600-8. [CrossRef]
- Diep S, Maddukuri M., et al. Interleukin-1 and Nuclear Factor Kappa B Signaling Promote Breast Cancer Progression and Treatment Resistance. Cells. 2022. 11:1673. [CrossRef]
- Zhang W, Ding W., et al. Up-regulation of breast cancer resistance protein plays a role in HER2-mediated chemoresistance through PI3K/AKT and nuclear factor-kappa B signaling pathways in MCF7 breast cancer cells. Acta Biochim Biophys Sin (Shanghai). 2011. 43:647-53. [CrossRef]
- Ghoneum A, Said N. PI3K-AKT -mTOR and NFκB Pathways in Ovarian Cancer: Implications for Targeted Therapeutics. Cancers (Basel). 2019. 11:949. [CrossRef]
- Wheeler DL, Iida M., et al. The role of Src in solid tumors. Oncologist. 2009. 14:667-78. [CrossRef]
- Mantovani A, Allavena P., et al. Cancer-related inflammation. Nature. 2008. 454:436-44. [CrossRef]
- Cory S, Huang DC., et al. The Bcl-2 family: roles in cell survival and oncogenesis. Oncogene. 2003. 22:8590-607. [CrossRef]
- Rahmani M, Aust MM., et al. Dual inhibition of Bcl-2 and Bcl-xL strikingly enhances PI3K inhibition-induced apoptosis in human myeloid leukemia cells through a GSK3- and Bim-dependent mechanism. Cancer Res. 2013. 73:1340-51. [CrossRef]
- Nachmias B, Ashhab Y., et al. The inhibitor of apoptosis protein family (IAPs): an emerging therapeutic target in cancer. Semin Cancer Biol. 2004. 14:231-43. [CrossRef]
- Spets H, Strömberg T., et al. Expression of the bcl-2 family of pro- and anti-apoptotic genes in multiple myeloma and normal plasma cells: regulation during interleukin-6(IL-6)-induced growth and survival. Eur J Haematol. 2002. 69:76-89. [CrossRef]
- Martin SJ, Reutelingsperger CP., et al. Early redistribution of plasma membrane phosphatidylserine is a general feature of apoptosis regardless of the initiating stimulus: inhibition by overexpression of Bcl-2 and Abl. J Exp Med. 1995. 82:1545-56. [CrossRef]
- Earnshaw WC, Martins LM., et al. Mammalian caspases: structure, activation, substrates, and functions during apoptosis. Annu Rev Biochem. 1999. 68:383-424. [CrossRef]
- Kayagaki N, Webster JD., et al. Control of Cell Death in Health and Disease. Annu Rev Pathol. 2024.19:157-180. [CrossRef]
- Kocab AJ, Duckett CS. Inhibitor of apoptosis proteins as intracellular signaling intermediates. FEBS J. 2016. 283:221-31. [CrossRef]
- McComb S, Cheung HH., et al. cIAP1 and cIAP2 limit macrophage necroptosis by inhibiting Rip1 and Rip3 activation. Cell Death Differ. 2012.19:1791-801. [CrossRef]
- Gerges S, Rohde K., et al. Cotreatment with Smac mimetics and demethylating agents induces both apoptotic and necroptotic cell death pathways in acute lymphoblastic leukemia cells. Cancer Lett. 2016. 375:127-132. [CrossRef]
- Feldmann F, Schenk B., et al. Sorafenib inhibits therapeutic induction of necroptosis in acute leukemia cells. Oncotarget. 2017. 8:68208-68220. [CrossRef]
- Bousserouel S, Raymondjean M, et al. Modulation of cyclin D1 and early growth response factor-1 gene expression in interleukin-1beta-treated rat smooth muscle cells by n-6 and n-3 polyunsaturated fatty acids. Eur J Biochem.2004. 271(22):4462-73. [CrossRef]
- Ming J, Jiang G., et al. Interleukin-7 up-regulates cyclin D1 via activator protein-1 to promote proliferation of cell in lung cancer. Cancer Immunol Immunother. 2012. 61(1):79-88. [CrossRef]
- Baumann P, Schneider L., et al. Simultaneous targeting of PI3K and mTOR with NVP-BGT226 is highly effective in multiple myeloma. Anticancer Drugs. 2012. 23(1):131-8. [CrossRef]
- Shishodia S, Aggarwal BB. Nuclear factor-kappaB: a friend or a foe in cancer? Biochem Pharmacol. 2004. 68:1071-80. [CrossRef]
- Liu C, Wu K., et al. SPP1+ macrophages promote head and neck squamous cell carcinoma progression by secreting TNF-α and IL-1β. J Exp Clin Cancer Res. 2024. 43(1):332. [CrossRef]
- Teixeira LFS, Peron JPS., et al. Silencing of nuclear factor kappa b 1 gene expression inhibits colony formation, cell migration and invasion via the downregulation of interleukin 1 beta and matrix metallopeptidase 9 in renal cell carcinoma. Mol Biol Rep. 2020. 47(2):1143-1151. [CrossRef]
- Viatour P, Merville MP., et al. Phosphorylation of NF-kappa B and Ikappa B proteins: implications in cancer and inflammation. Trends Biochem Sci. 2005. 30:43-52. [CrossRef]
- Gilmore TD. Introduction to NF-kappaB: players, pathways, perspectives. Oncogene. 2006. 25:6680-4. PMID: 17072321. [CrossRef]
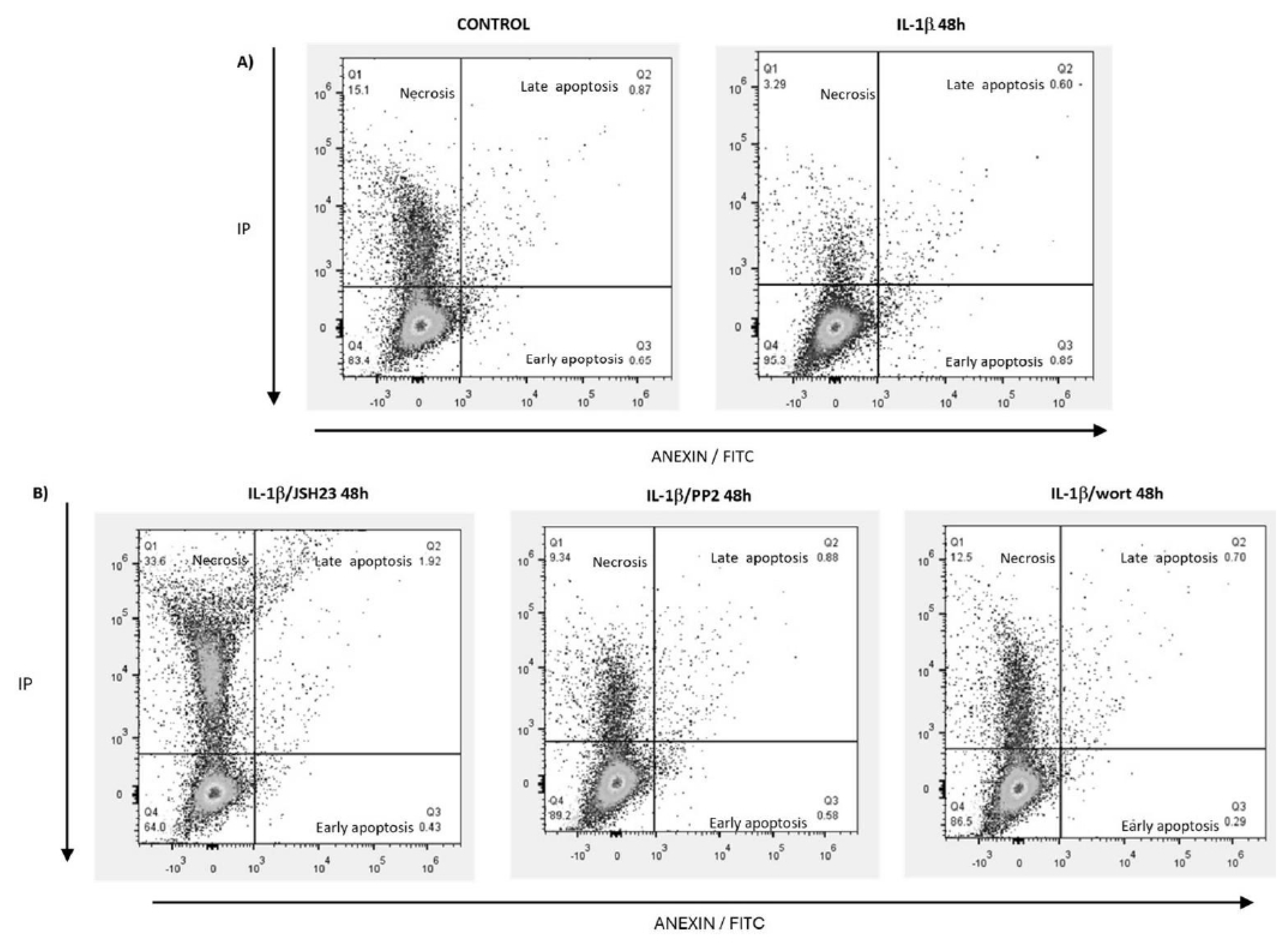
| Treatment | Viable cells (%) | Early apoptosis (%) | Late apoptosis (%) | Necrosis (%) |
|---|---|---|---|---|
| Control | 66.11+/- 7.0 | 5.0 +/- 0.08 | 0.84 +/- 0.10 | 13.54 +/- 2.7 |
| IL-1β | 89.76 +/- 5.0 | 0.90 +/- 0.06 | 0.91 +/- 0.33 | 5.0 +/- 1.9 |
| IL-1β+JSH23 | 66.47 +/- 7.3 | 0.53 +/- 0.12 | 1.9 +/- 6.16 | 30.5 +/- 3.8 |
| IL-1β + PP2 | 84.90 +/- 6.4 | 0.62 +/- 0.10 | 0.83 +/- 0.09 | 10.53 +/- 2.3 |
| IL-1β + Wort | 85.50 +/- 5.4 | 0.24 +/- 0.05 | 0.75 +/- 0.07 | 12.7 +/- 1.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




