Submitted:
20 November 2025
Posted:
21 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
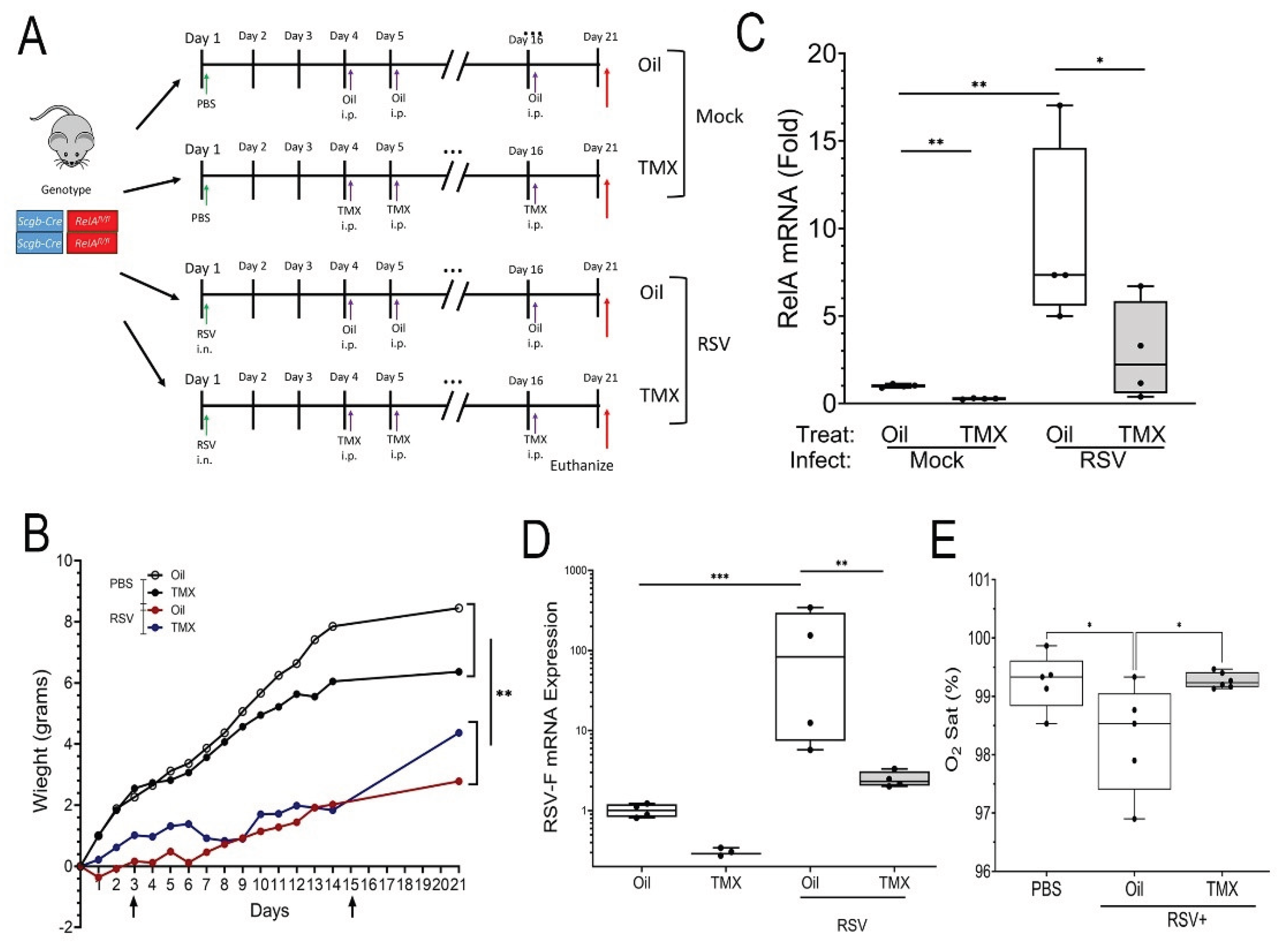
2.1. Acute RSV Infection
2.2. RSV-PVLD
2.3. Quantitative Real-Time PCR
2.4. Immunohistochemistry
2.5. Quantitation of Airway Features
2.6. Single Cell RNA Sequencing (scRNA-Seq)
2.7. scRNA Seq Analysis
2.8. Statistical Analysis
3. Results
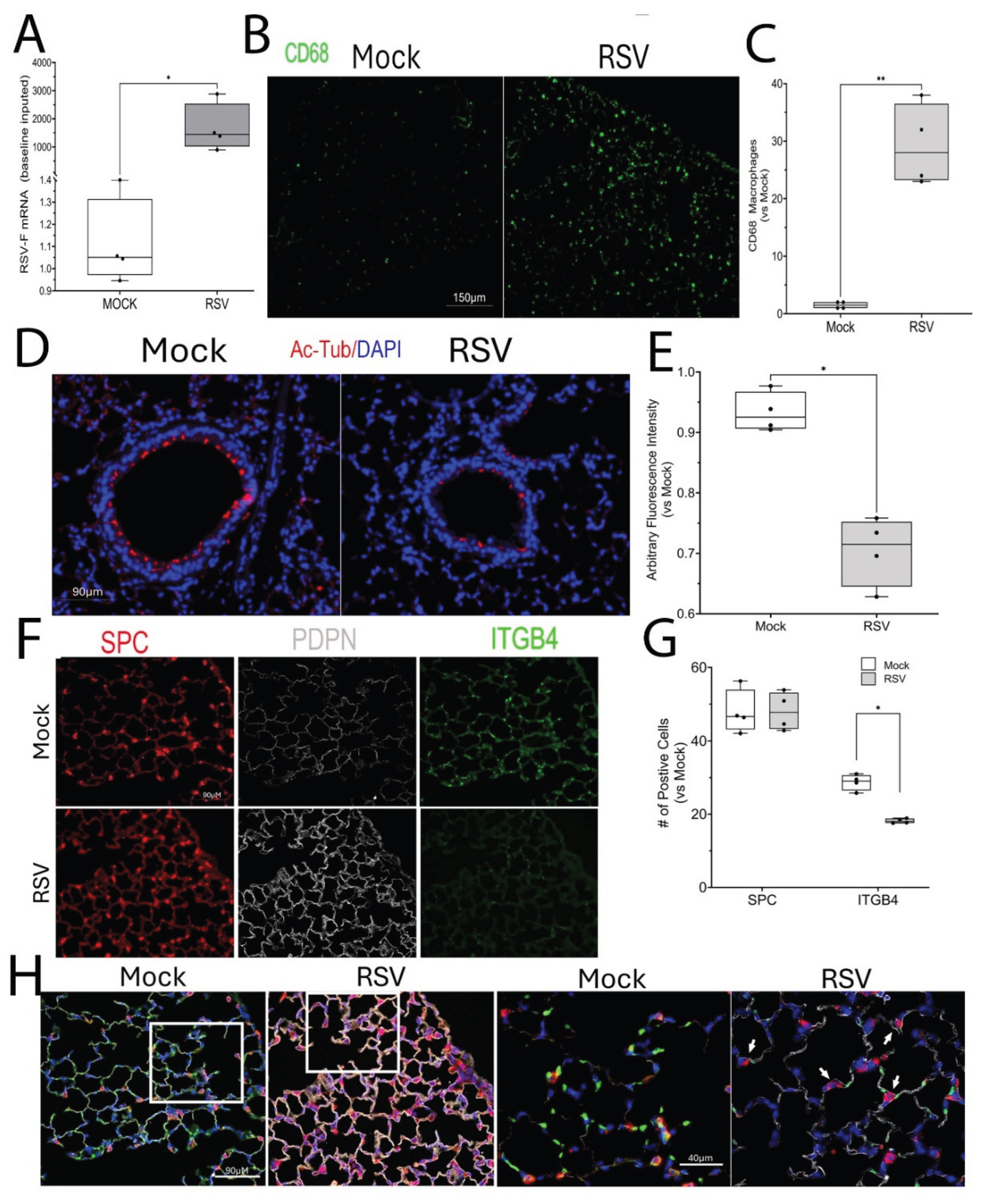
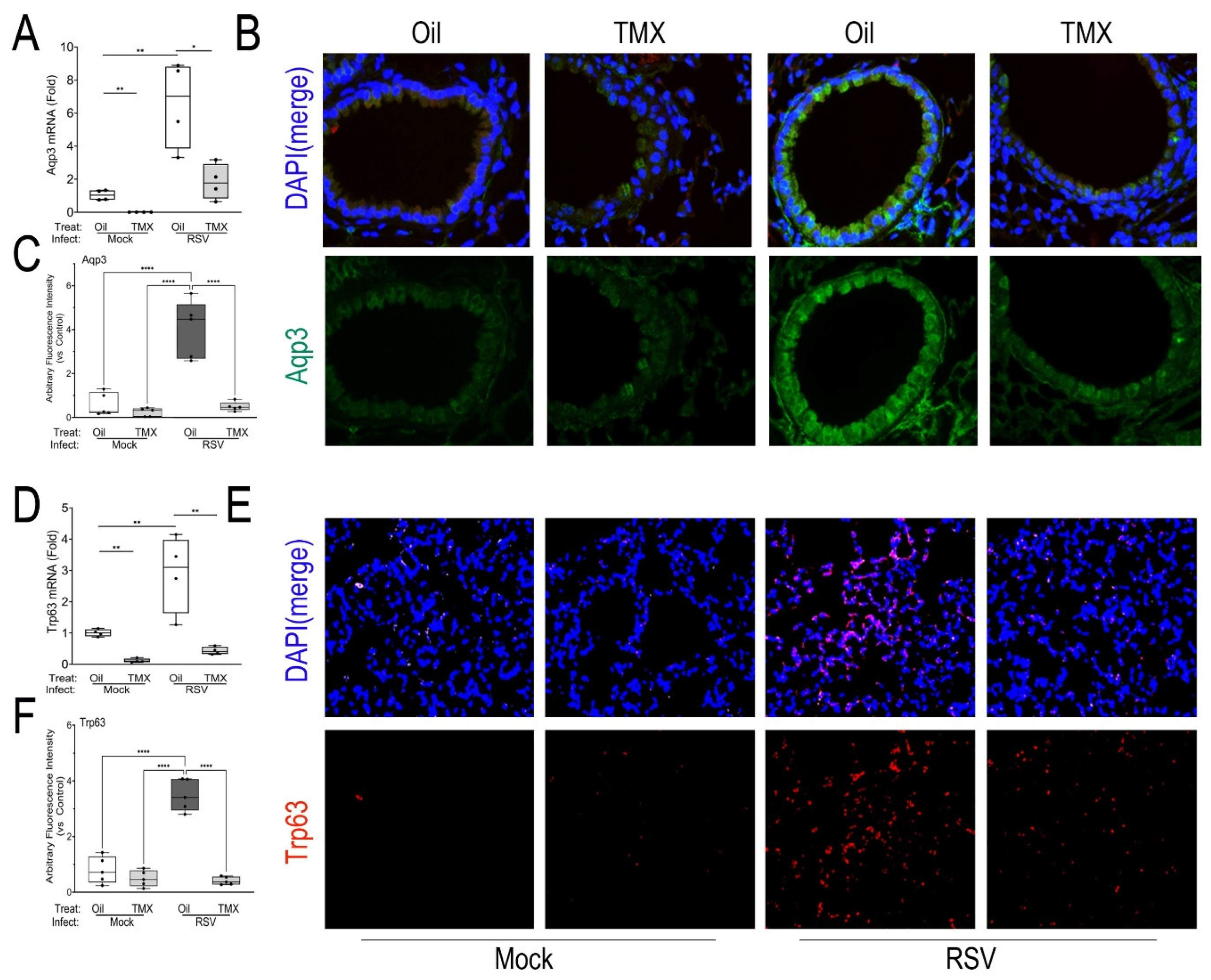
3.1. Acute RSV Infection Induces Phenotypic Changes in Distal Airway and Alveolus
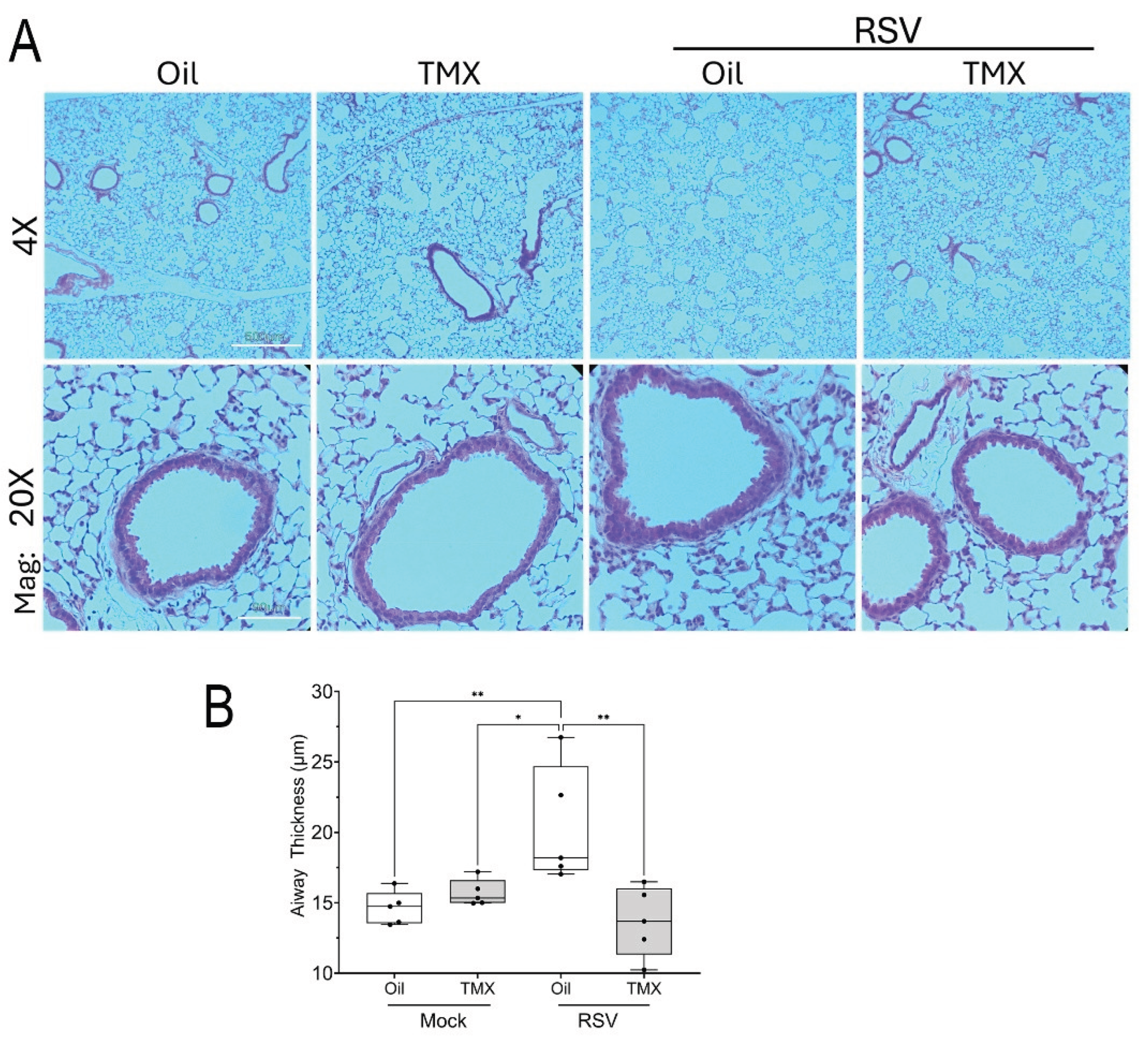
3.2. RSV-PVLD Induces Bronchiolar and Alveolar Remodeling
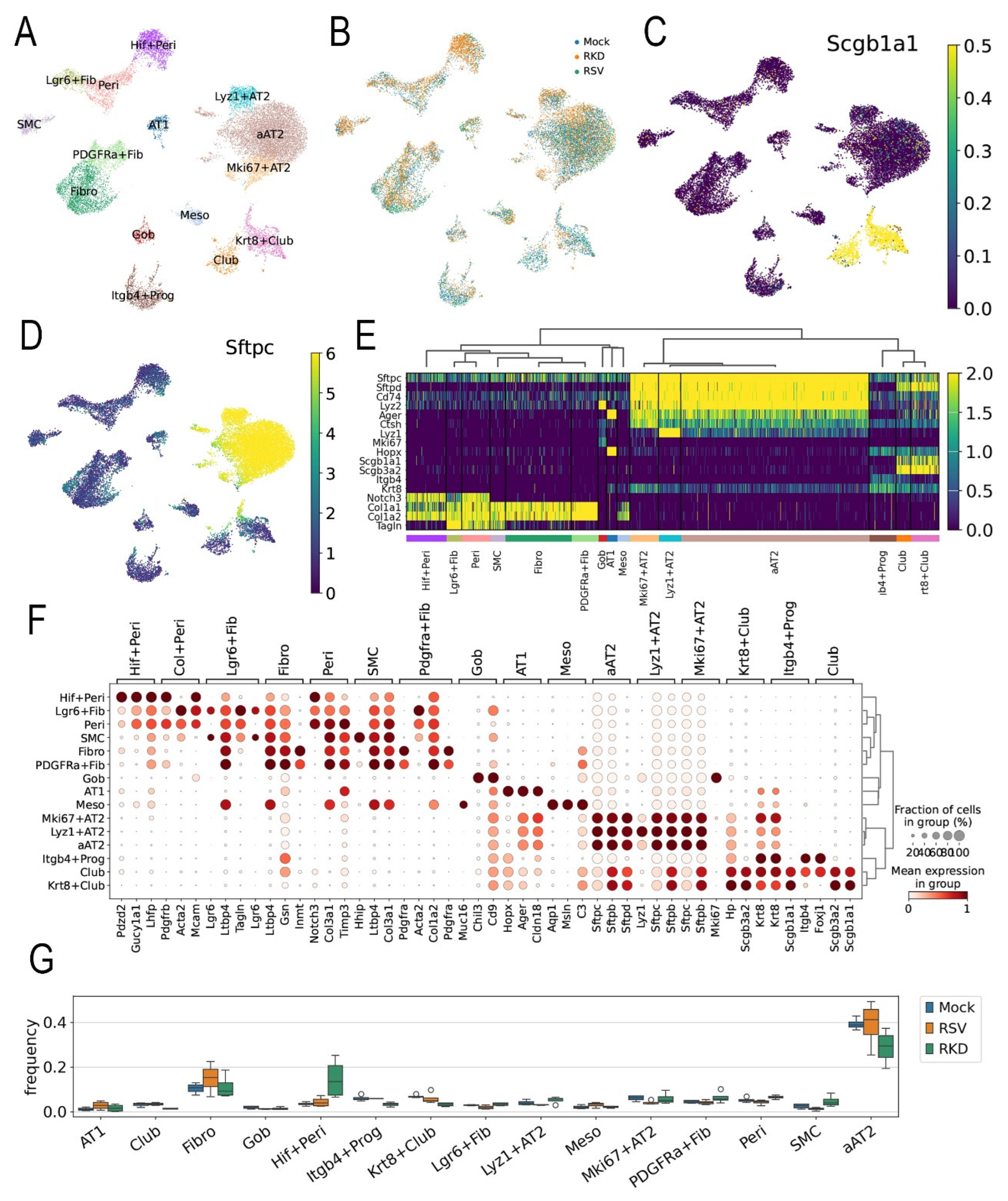
3.3. Expansion of Alveolar Progenitor Epithelial Populations in RSV-PVLD
3.4. scRNA Sequence Analysis of RSV-PVLD Shows Atypical AT2 Populations
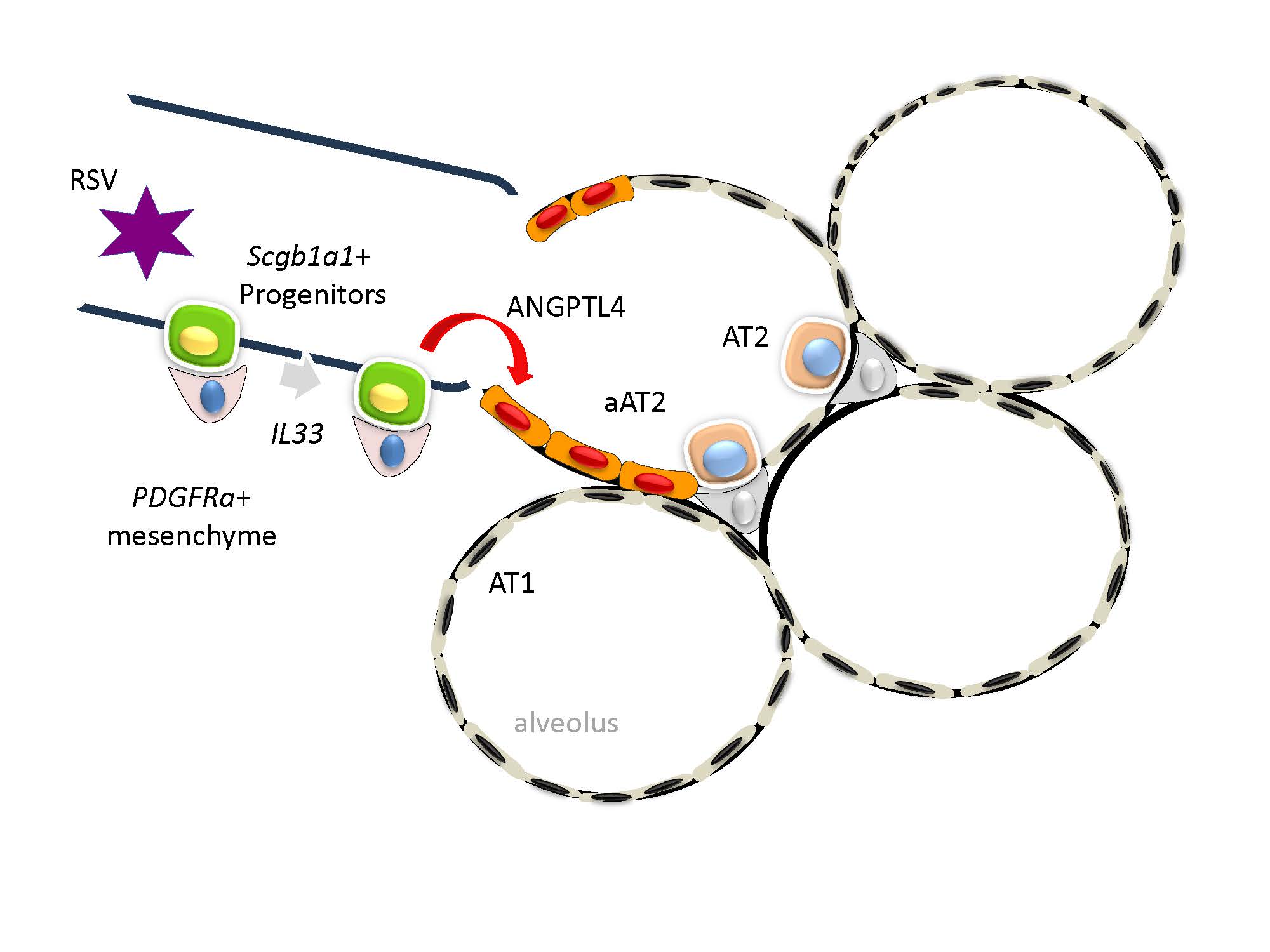
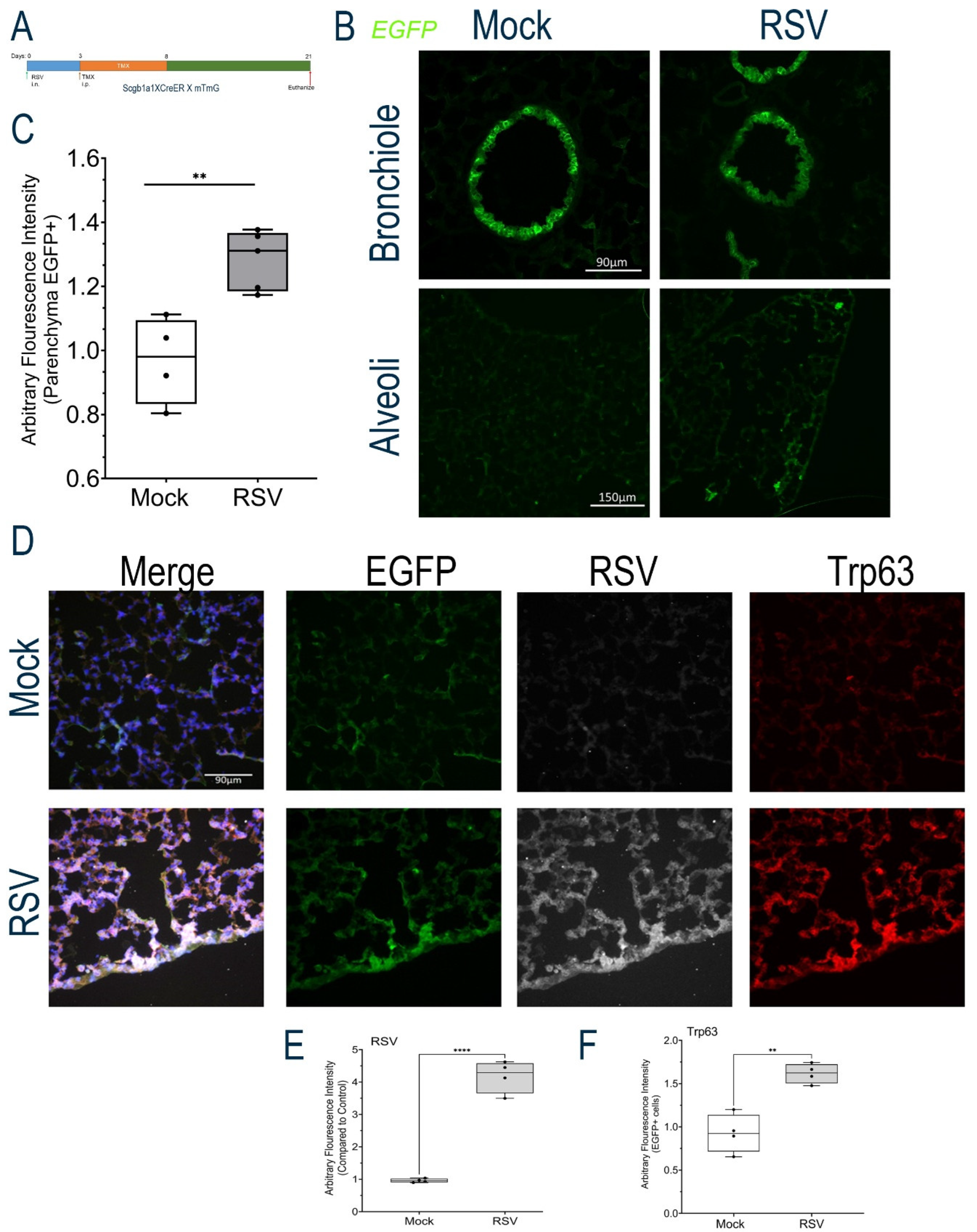
3.5. Scgba1a1 Progenitors Contribute to the aAT2 Population in RSV-PVLD
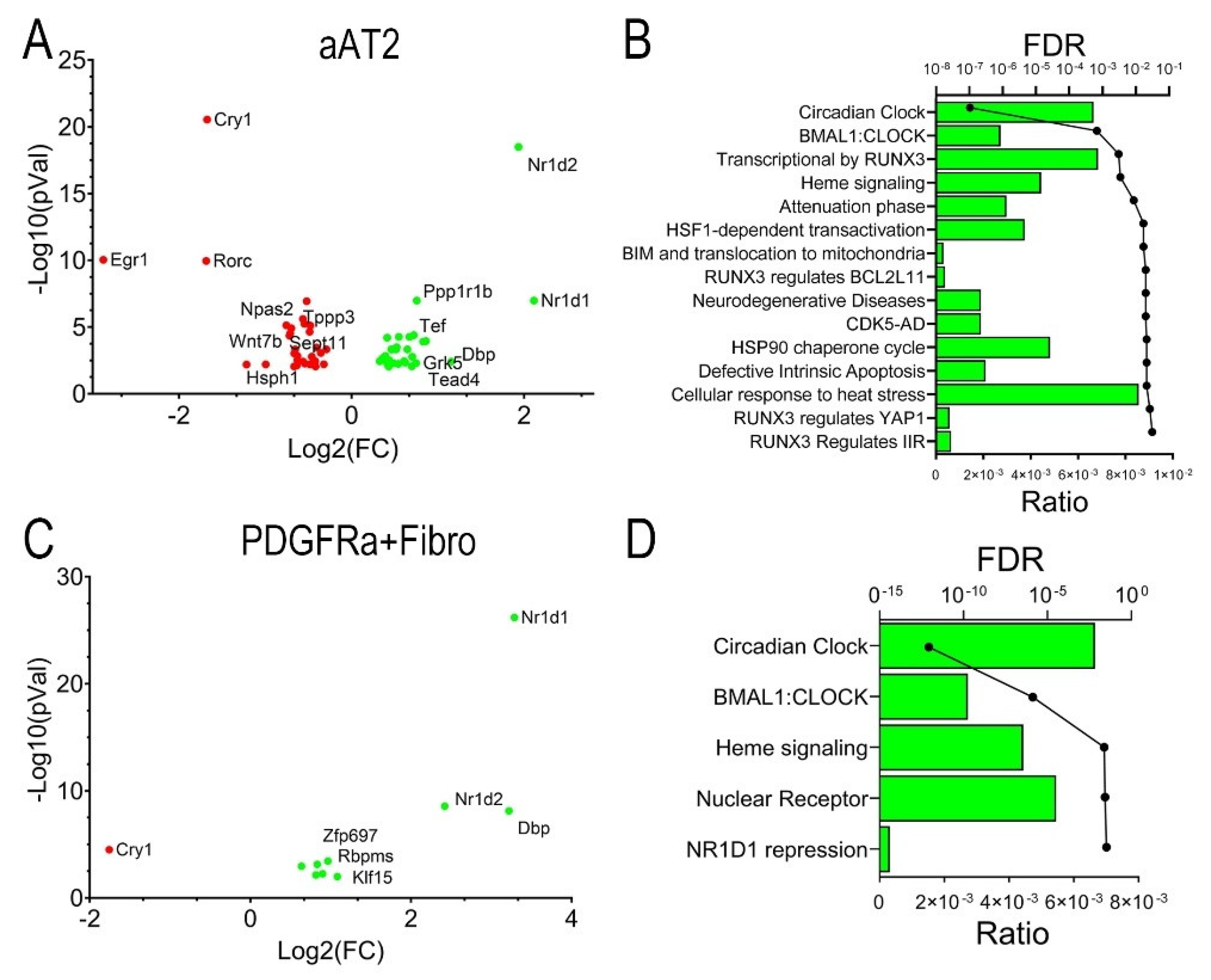
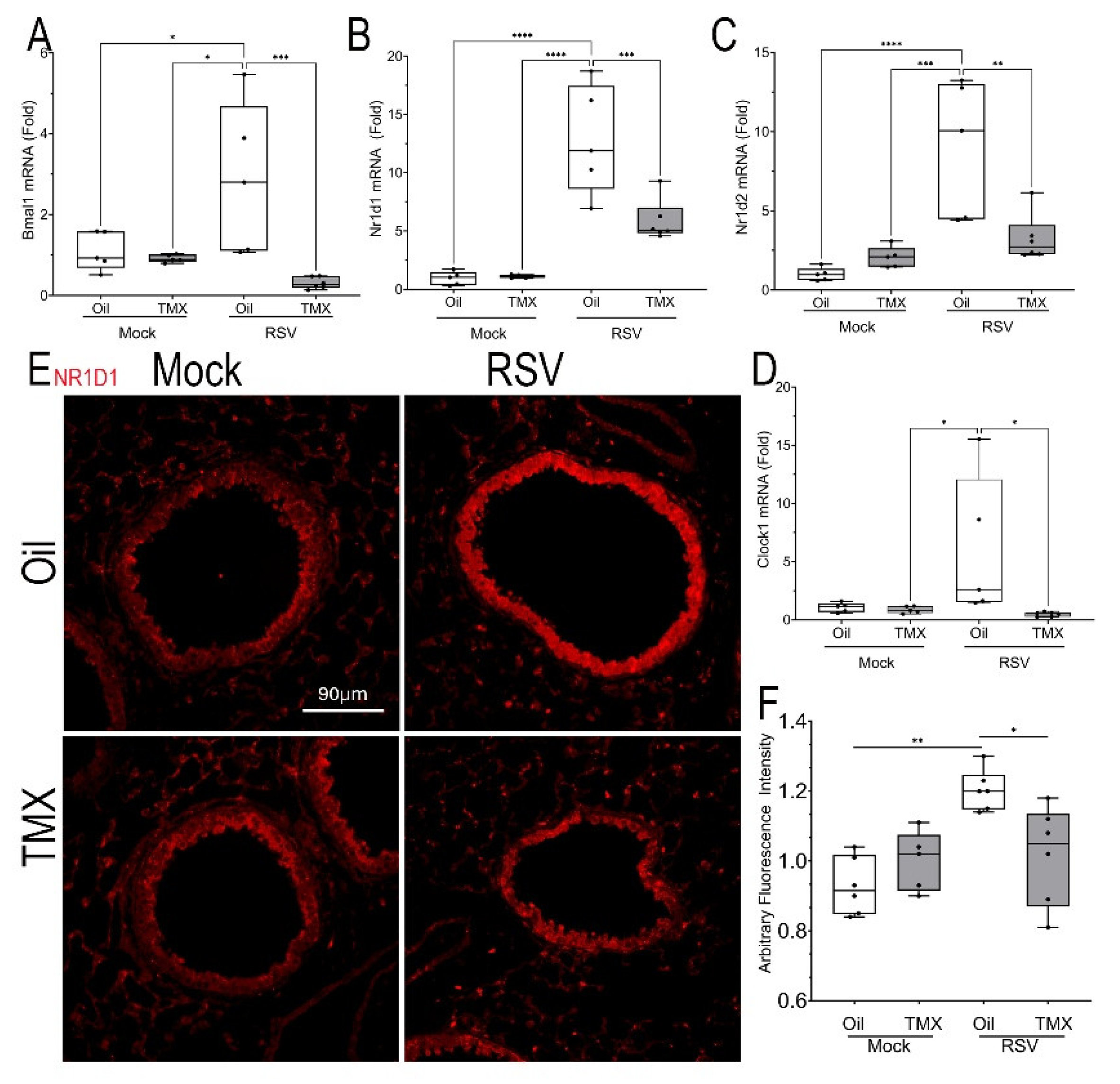
3.6. RSV-PVLD Dysregulates Inflammatory Clock Genes in the Alveolar-Mesenchymal Niche
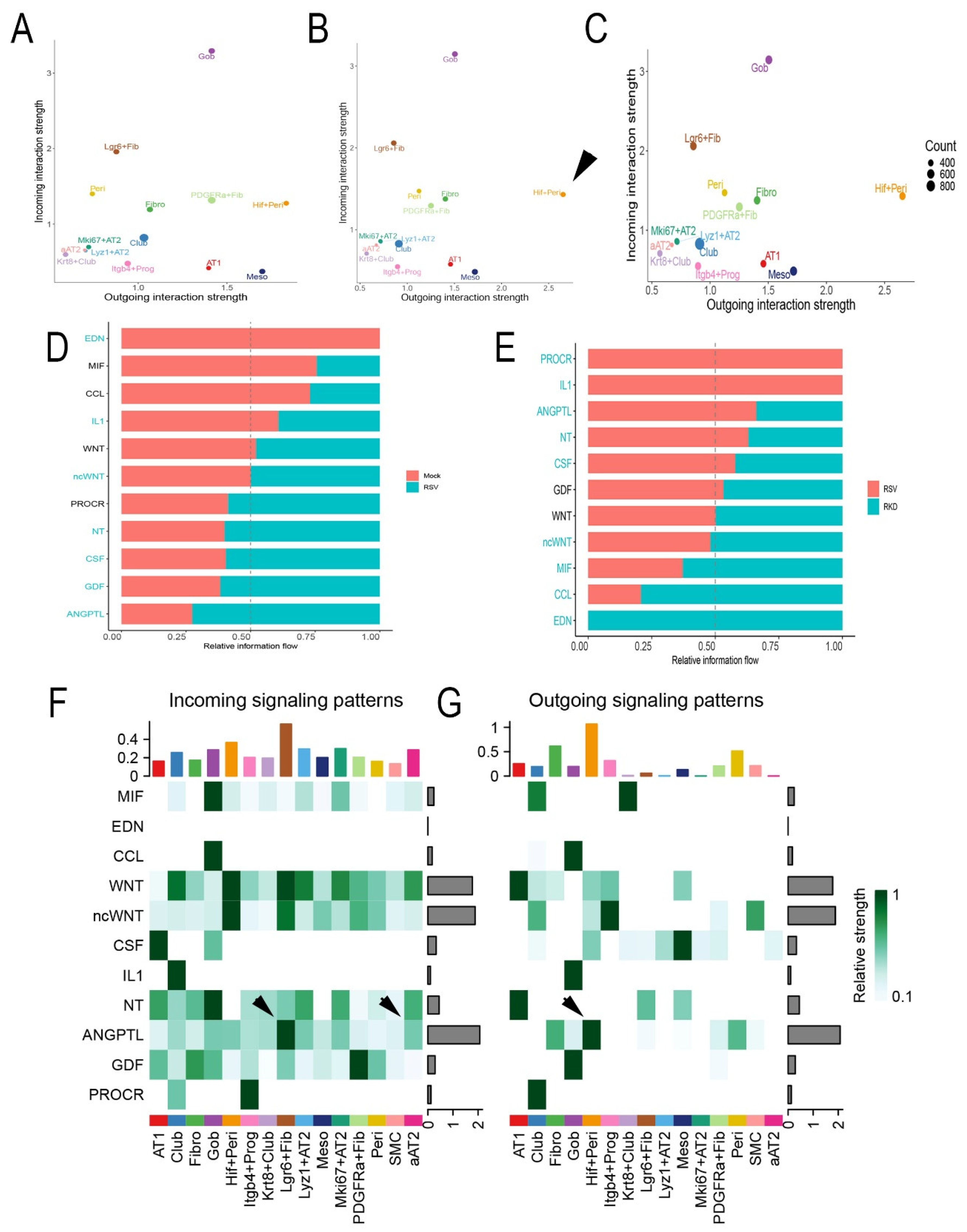
3.7. RSV Disrupts Intercellular Epithelial-Mesenchymal Signaling Networks
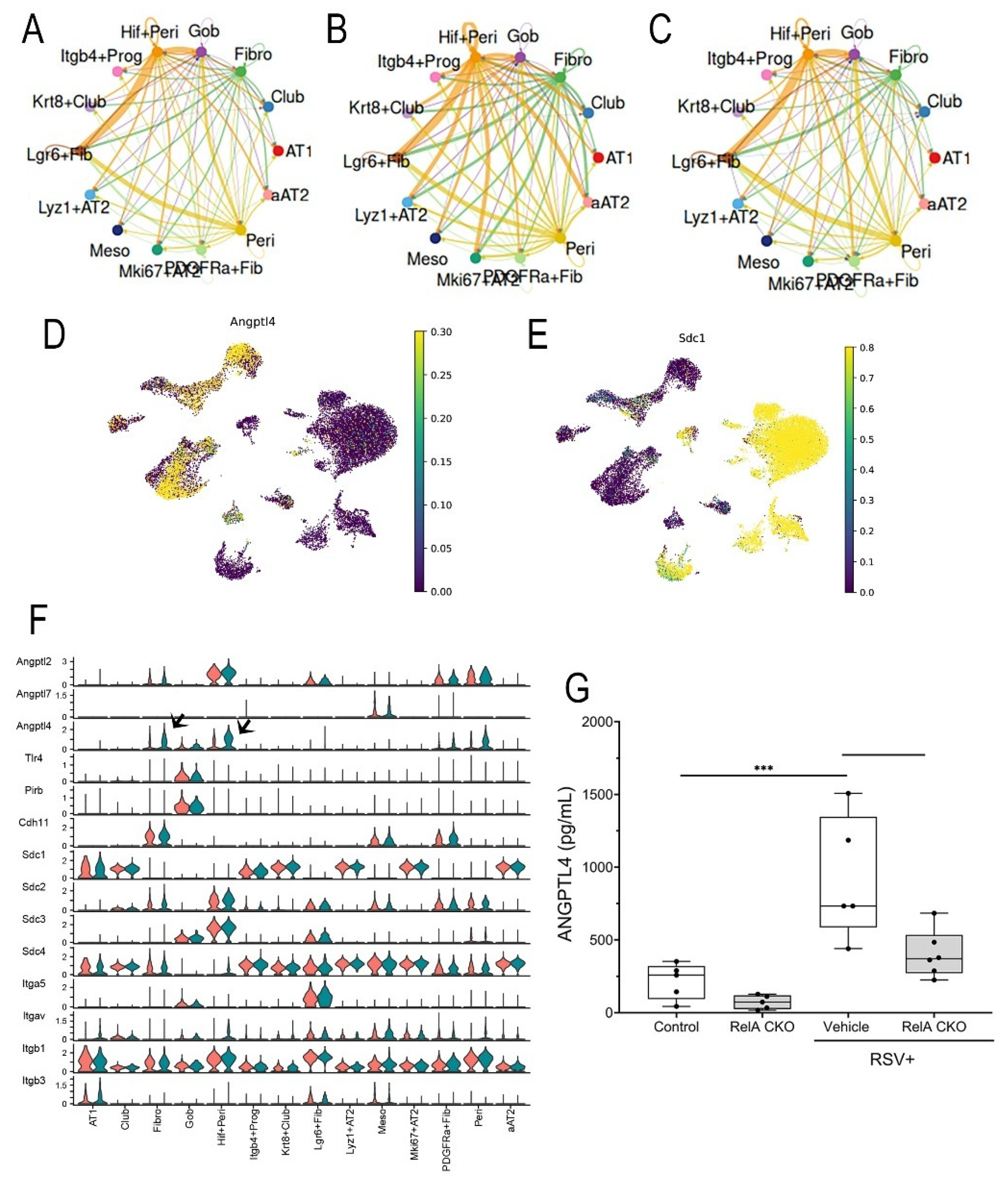
3.8. RSV Dysregulates Mesenchymal-Epithelial ANGPTL Signaling
4. Discussion
4.1. Early Life RSV Infections Induce Epithelial Atypia Through Epithelial Plasticity
4.2. Scgb1a1+ Progenitors Mediate Epithelial Atypia in RSV-PVLD
4.3. RSV Modulates Epithelial Plasticity and Differentiation Programs Through Innate Signaling
4.4. RSV Latency in RSV-PVLD
4.5. RSV Disrupts Circadian Clock Gene Expression in aAT2 Cells
4.6. RSV-PVLD Disrupts Epithelial-Mesenchymal Niches
4.7. A Potential Role for Angiopoietins in Post Viral Lung Disease
5. Conclusions
Author Contributions
Funding
Ethics Approval and Consent to Participate
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Shi T, McAllister DA, O’Brien KL, Simoes EAF, Madhi SA, Gessner BD, Polack FP, Balsells E, Acacio S, Aguayo C, et al.: Global, regional, and national disease burden estimates of acute lower respiratory infections due to respiratory syncytial virus in young children in 2015: a systematic review and modelling study. Lancet 2017, 390:946–958. [CrossRef]
- Stockman LJ, Curns AT, Anderson LJ, Fischer-Langley G: Respiratory syncytial virus-associated hospitalizations among infants and young children in the United States, 1997-2006. Pediatr Infect Dis J 2012, 31:5–9.
- Sigurs N, Aljassim F, Kjellman B, Robinson PD, Sigurbergsson F, Bjarnason R, Gustafsson PM: Asthma and allergy patterns over 18 years after severe RSV bronchiolitis in the first year of life. Thorax 2010, 65:1045–1052. [CrossRef]
- Fauroux B, Simoes EAF, Checchia PA, Paes B, Figueras-Aloy J, Manzoni P, Bont L, Carbonell-Estrany X: The Burden and Long-term Respiratory Morbidity Associated with Respiratory Syncytial Virus Infection in Early Childhood. Infect Dis Ther 2017, 6:173–197.
- Allinson JP, Chaturvedi N, Wong A, Shah I, Donaldson GC, Wedzicha JA, Hardy R: Early childhood lower respiratory tract infection and premature adult death from respiratory disease in Great Britain: a national birth cohort study. The Lancet 2023, 401:1183–1193. [CrossRef]
- Rosas-Salazar C, Chirkova T, Gebretsadik T, Chappell JD, Peebles RS, Jr., Dupont WD, Jadhao SJ, Gergen PJ, Anderson LJ, Hartert TV: Respiratory syncytial virus infection during infancy and asthma during childhood in the USA (INSPIRE): a population-based, prospective birth cohort study. The Lancet 2023, 401:1669–1680. [CrossRef]
- Saarikallio S, Riikonen R, Tikkakoski A, Laitinen S, Holster A, Korppi M, Lauhkonen E: Bronchiolitis in Early Infancy Is Associated With Irreversible Airway Obstruction in Young Adulthood. Pediatr Pulmonol 2025, 60:e71139. [CrossRef]
- Jozwik A, Habibi MS, Paras A, Zhu J, Guvenel A, Dhariwal J, Almond M, Wong EHC, Sykes A, Maybeno M, et al.: RSV-specific airway resident memory CD8+ T cells and differential disease severity after experimental human infection. Nat Commun 2015, 6:10224. [CrossRef]
- Johnson JE, Gonzales RA, Olson SJ, Wright PF, Graham BS: The histopathology of fatal untreated human respiratory syncytial virus infection. Mod Pathol 2007, 20:108–119. [CrossRef]
- Persson BD, Jaffe AB, Fearns R, Danahay H: Respiratory syncytial virus can infect basal cells and alter human airway epithelial differentiation. PLoS One 2014, 9:e102368. [CrossRef]
- El Saleeby CM, Bush AJ, Harrison LM, Aitken JA, Devincenzo JP: Respiratory syncytial virus load, viral dynamics, and disease severity in previously healthy naturally infected children. J Infect Dis 2011, 204:996–1002. [CrossRef]
- DeVincenzo JP, El Saleeby CM, Bush AJ: Respiratory syncytial virus load predicts disease severity in previously healthy infants. J Infect Dis 2005, 191:1861–1868.
- Mella C, Suarez-Arrabal MC, Lopez S, Stephens J, Fernandez S, Hall MW, Ramilo O, Mejias A: Innate immune dysfunction is associated with enhanced disease severity in infants with severe respiratory syncytial virus bronchiolitis. J Infect Dis 2013, 207:564–573. [CrossRef]
- García C, Soriano-Fallas A, Lozano J, Leos N, Gomez AM, Ramilo O, Mejias A: Decreased innate immune cytokine responses correlate with disease severity in children with respiratory syncytial virus and human rhinovirus bronchiolitis. Pediatr Infect Dis J 2012, 31:86–89. [CrossRef]
- Ganesan S, Comstock AT, Sajjan US: Barrier function of airway tract epithelium. Tissue Barriers 2013, 1:e24997.
- Lambrecht BN, Hammad H: Allergens and the airway epithelium response: gateway to allergic sensitization. J Allergy Clin Immunol 2014, 134:499–507. [CrossRef]
- Whitsett JA, Alenghat T: Respiratory epithelial cells orchestrate pulmonary innate immunity. Nat Immunol 2015, 16:27–35.
- Zhao C, Bai Y, Wang W, Amonkar GM, Mou H, Olejnik J, Hume AJ, Mühlberger E, Lukacs NW, Fearns R, et al.: Activation of STAT3-mediated ciliated cell survival protects against severe infection by respiratory syncytial virus. J Clin Invest 2024, 134. [CrossRef]
- Hogan BL, Barkauskas CE, Chapman HA, Epstein JA, Jain R, Hsia CC, Niklason L, Calle E, Le A, Randell SH, et al.: Repair and regeneration of the respiratory system: complexity, plasticity, and mechanisms of lung stem cell function. Cell Stem Cell 2014, 15:123–138. [CrossRef]
- Strunz M, Simon LM, Ansari M, Kathiriya JJ, Angelidis I, Mayr CH, Tsidiridis G, Lange M, Mattner LF, Yee M, et al.: Alveolar regeneration through a Krt8+ transitional stem cell state that persists in human lung fibrosis. Nat Commun 2020, 11:3559. [CrossRef]
- Zepp JA, Zacharias WJ, Frank DB, Cavanaugh CA, Zhou S, Morley MP, Morrisey EE: Distinct Mesenchymal Lineages and Niches Promote Epithelial Self-Renewal and Myofibrogenesis in the Lung. Cell 2017, 170:1134–1148.e1110. [CrossRef]
- Barkauskas CE, Cronce MJ, Rackley CR, Bowie EJ, Keene DR, Stripp BR, Randell SH, Noble PW, Hogan BL: Type 2 alveolar cells are stem cells in adult lung. The Journal of clinical investigation 2013, 123:3025–3036. [CrossRef]
- Tian B, Liu Z, Yang J, Sun H, Zhao Y, Wakamiya M, Chen H, Rytting E, Zhou J, Brasier AR: Selective Antagonists of the Bronchiolar Epithelial NF-kappaB-Bromodomain-Containing Protein 4 Pathway in Viral-Induced Airway Inflammation. Cell Rep 2018, 23:1138–1151. [CrossRef]
- Tian B, Yang J, Zhao Y, Ivanciuc T, Sun H, Wakamiya M, Garofalo RP, Brasier AR: Central Role of the NF-kappaB Pathway in the Scgb1a1-Expressing Epithelium in Mediating Respiratory Syncytial Virus-Induced Airway Inflammation. J Virol 2018, 92. [CrossRef]
- Skibba ME, Brasier AR: NFkB/RelA Signaling in Secretoglobin Progenitors Mediates Plasticity and MMP-Induced Barrier Disruption In House Dust Mite-Induced Allergic Asthma. American Journal of Physiology-Lung Cellular and Molecular Physiology 2024, 327:L86–L101. [CrossRef]
- Skibba ME, Xu X, Weiss K, Huisken J, Brasier AR: Role of Secretoglobin(+) (club cell) NFκB/RelA-TGFβ signaling in aero-allergen-induced epithelial plasticity and subepithelial myofibroblast transdifferentiation. Respir Res 2021, 22:315.
- Tully JE, Hoffman SM, Lahue KG, Nolin JD, Anathy V, Lundblad LKA, Daphtary N, Aliyeva M, Black KE, Dixon AE, et al.: Epithelial NF-κB Orchestrates House Dust Mite–Induced Airway Inflammation, Hyperresponsiveness, and Fibrotic Remodeling. The Journal of Immunology 2013. [CrossRef]
- Zhao Y, Jamaluddin M, Zhang Y, Sun H, Ivanciuc T, Garofalo RP, Brasier AR: Systematic Analysis of Cell-Type Differences in the Epithelial Secretome Reveals Insights into the Pathogenesis of Respiratory Syncytial Virus-Induced Lower Respiratory Tract Infections. J Immunol 2017, 198:3345–3364.
- Calhoun WJ, Dick EC, Schwartz LB, Busse WW: A common cold virus, rhinovirus 16, potentiates airway inflammation after segmental antigen bronchoprovocation in allergic subjects. J Clin Invest 1994, 94:2200–2208.
- Message SD, Laza-Stanca V, Mallia P, Parker HL, Zhu J, Kebadze T, Contoli M, Sanderson G, Kon OM, Papi A, et al.: Rhinovirus-induced lower respiratory illness is increased in asthma and related to virus load and Th1/2 cytokine and IL-10 production. Proc Natl Acad Sci U S A 2008, 105:13562–13567.
- Olzewska B, Casola A, Saito T, Alam R, Crowe S, Mei F, Ogra PL, Garofalo R: Cell-specific expression of RANTES, MCP-1, and MIP-1a by lower airway epithelial cells and eosinophils infected with respiratory syncytial virus. Journal of Virology 1998, 72:4756–4764. [CrossRef]
- Malinczak CA, Fonseca W, Hrycaj SM, Morris SB, Rasky AJ, Yagi K, Wellik DM, Ziegler SF, Zemans RL, Lukacs NW: Early-life pulmonary viral infection leads to long-term functional and lower airway structural changes in the lungs. Am J Physiol Lung Cell Mol Physiol 2024, 326:L280–L291. [CrossRef]
- Wu K, Kamimoto K, Zhang Y, Yang K, Keeler SP, Gerovac BJ, Agapov EV, Austin SP, Yantis J, Gissy KA, et al.: Basal epithelial stem cells cross an alarmin checkpoint for postviral lung disease. The Journal of Clinical Investigation 2021, 131.
- Garcia GL, Valenzuela A, Manzoni T, Vaughan AE, López CB: Distinct Chronic Post-Viral Lung Diseases upon Infection with Influenza or Parainfluenza Viruses Differentially Impact Superinfection Outcome. Am J Pathol 2020, 190:543–553. [CrossRef]
- Wolock SL, Lopez R, Klein AM: Scrublet: Computational Identification of Cell Doublets in Single-Cell Transcriptomic Data. Cell Systems 2019, 8:281–291.e289. [CrossRef]
- Stuart T, Butler A, Hoffman P, Hafemeister C, Papalexi E, Mauck WM, 3rd, Hao Y, Stoeckius M, Smibert P, Satija R: Comprehensive Integration of Single-Cell Data. Cell 2019, 177:1888–1902.e1821.
- Zeng H: What is a cell type and how to define it? Cell 2022, 185:2739–2755.
- Franzén O, Gan L-M, Björkegren JLM: PanglaoDB: a web server for exploration of mouse and human single-cell RNA sequencing data. Database 2019, 2019:baz046. [CrossRef]
- Love MI, Huber W, Anders S: Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 2014, 15:550.
- Liesman RM, Buchholz UJ, Luongo CL, Yang L, Proia AD, DeVincenzo JP, Collins PL, Pickles RJ: RSV-encoded NS2 promotes epithelial cell shedding and distal airway obstruction. J Clin Invest 2014, 124:2219–2233.
- Watanabe H, Numata K, Ito T, Takagi K, Matsukawa A: Innate immune response in Th1- and Th2-dominant mouse strains. Shock 2004, 22:460–466. [CrossRef]
- Sucre JMS, Bock F, Negretti NM, Benjamin JT, Gulleman PM, Dong X, Ferguson KT, Jetter CS, Han W, Liu Y, et al.: Alveolar repair following LPS-induced injury requires cell-ECM interactions. JCI Insight 2023, 8. [CrossRef]
- Chi Y, Wang X, Zhang D, Han J, Shao X, Xiang Y, Deng L: Deficiency of integrin β4 contributes bronchopulmonary dysplasia by compromising cellular stability through the activation of RhoA-(ZO-1) signaling pathways. Scientific Reports 2025, 15:20985. [CrossRef]
- Kobayashi Y, Tata A, Konkimalla A, Katsura H, Lee RF, Ou J, Banovich NE, Kropski JA, Tata PR: Persistence of a regeneration-associated, transitional alveolar epithelial cell state in pulmonary fibrosis. Nat Cell Biol 2020, 22:934–946.
- Li J, Yang D, Lin L, Yu L, Chen L, Lu K, Lan J, Zeng Y, Xu Y: Important functions and molecular mechanisms of aquaporins family on respiratory diseases: potential translational values. J Cancer 2024, 15:6073–6085. [CrossRef]
- Lv Z, Liu Z, Liu K, Lin X, Pu W, Li Y, Zhao H, Xi Y, Sui P, Vaughan AE, et al.: Alveolar regeneration by airway secretory-cell-derived p63(+) progenitors. Cell Stem Cell 2024, 31:1685–1700 e1686. [CrossRef]
- Zuo W, Zhang T, Wu DZ, Guan SP, Liew AA, Yamamoto Y, Wang X, Lim SJ, Vincent M, Lessard M, et al.: p63(+)Krt5(+) distal airway stem cells are essential for lung regeneration. Nature 2015, 517:616–620.
- Carroll DK, Carroll JS, Leong CO, Cheng F, Brown M, Mills AA, Brugge JS, Ellisen LW: p63 regulates an adhesion programme and cell survival in epithelial cells. Nat Cell Biol 2006, 8:551–561.
- Chapman HA, Li X, Alexander JP, Brumwell A, Lorizio W, Tan K, Sonnenberg A, Wei Y, Vu TH: Integrin α6β4 identifies an adult distal lung epithelial population with regenerative potential in mice. J Clin Invest 2011, 121:2855–2862.
- Liu K, Meng X, Liu Z, Tang M, Lv Z, Huang X, Jin H, Han X, Liu X, Pu W, et al.: Tracing the origin of alveolar stem cells in lung repair and regeneration. Cell 2024, 187:2428–2445.e2420. [CrossRef]
- Aros CJ, Vijayaraj P, Pantoja CJ, Bisht B, Meneses LK, Sandlin JM, Tse JA, Chen MW, Purkayastha A, Shia DW, et al.: Distinct Spatiotemporally Dynamic Wnt-Secreting Niches Regulate Proximal Airway Regeneration and Aging. Cell Stem Cell 2020, 27:413–429.e414. [CrossRef]
- Habermann AC, Gutierrez AJ, Bui LT, Yahn SL, Winters NI, Calvi CL, Peter L, Chung MI, Taylor CJ, Jetter C, et al.: Single-cell RNA sequencing reveals profibrotic roles of distinct epithelial and mesenchymal lineages in pulmonary fibrosis. Sci Adv 2020, 6:eaba1972. [CrossRef]
- Rawlins EL, Okubo T, Xue Y, Brass DM, Auten RL, Hasegawa H, Wang F, Hogan BL: The role of Scgb1a1+ Clara cells in the long-term maintenance and repair of lung airway, but not alveolar, epithelium. Cell Stem Cell 2009, 4:525–534.
- Squair JW, Gautier M, Kathe C, Anderson MA, James ND, Hutson TH, Hudelle R, Qaiser T, Matson KJE, Barraud Q, et al.: Confronting false discoveries in single-cell differential expression. Nature Communications 2021, 12:5692. [CrossRef]
- Jin S, Guerrero-Juarez CF, Zhang L, Chang I, Ramos R, Kuan C-H, Myung P, Plikus MV, Nie Q: Inference and analysis of cell-cell communication using CellChat. Nature Communications 2021, 12:1088. [CrossRef]
- Saito S, Kitabatake M, Ouji-Sageshima N, Ogawa T, Oda A, Nishimura T, Nishioka T, Fushimi S, Hara A, Shichino S, et al.: Angiopoietin-like 4 Is a Critical Regulator of Fibroblasts during Pulmonary Fibrosis Development. Am J Respir Cell Mol Biol 2023, 69:328–339. [CrossRef]
- Horiguchi H, Endo M, Kawane K, Kadomatsu T, Terada K, Morinaga J, Araki K, Miyata K, Oike Y: ANGPTL2 expression in the intestinal stem cell niche controls epithelial regeneration and homeostasis. Embo j 2017, 36:409–424.
- Jafri HS, Chavez-Bueno S, Mejias A, Gomez AM, Rios AM, Nassi SS, Yusuf M, Kapur P, Hardy RD, Hatfield J, et al.: Respiratory syncytial virus induces pneumonia, cytokine response, airway obstruction, and chronic inflammatory infiltrates associated with long-term airway hyperresponsiveness in mice. J Infect Dis 2004, 189:1856–1865. [CrossRef]
- You D, Becnel D, Wang K, Ripple M, Daly M, Cormier SA: Exposure of neonates to respiratory syncytial virus is critical in determining subsequent airway response in adults. Respir Res 2006, 7:107. [CrossRef]
- Morris SB, Ocadiz-Ruiz R, Asai N, Malinczak C-A, Rasky AJ, Lombardo GK, Velarde EM, Ptaschinski C, Zemans RL, Lukacs NW, Fonseca W: Long-term alterations in lung epithelial cells after EL-RSV infection exacerbate allergic responses through IL-1β-induced pathways. Mucosal Immunology 2024, 17:1072–1088. [CrossRef]
- Berdnikovs S, Newcomb DC, Haruna NF, McKernan KE, Kuehnle SN, Gebretsadik T, McKennan C, Ma S, Cephus JY, Rosas-Salazar C, et al.: Single-cell profiling demonstrates the combined effect of wheeze phenotype and infant viral infection on airway epithelial development. Sci Adv 2025, 11:eadr9995. [CrossRef]
- Vaughan AE, Brumwell AN, Xi Y, Gotts JE, Brownfield DG, Treutlein B, Tan K, Tan V, Liu FC, Looney MR, et al.: Lineage-negative progenitors mobilize to regenerate lung epithelium after major injury. Nature 2015, 517:621–625.
- Wu K, Zhang Y, Austin SR, Yin-Declue H, Byers DE, Crouch EC, Holtzman MJ: Lung Remodeling Regions in Long-Term Coronavirus Disease 2019 Feature Basal Epithelial Cell Reprogramming. Am J Pathol 2023, 193:680–689.
- Kumar PA, Hu Y, Yamamoto Y, Hoe NB, Wei TS, Mu D, Sun Y, Joo LS, Dagher R, Zielonka EM, et al.: Distal airway stem cells yield alveoli in vitro and during lung regeneration following H1N1 influenza infection. Cell 2011, 147:525–538.
- Zheng D, Yin L, Chen J: Evidence for Scgb1a1(+) cells in the generation of p63(+) cells in the damaged lung parenchyma. Am J Respir Cell Mol Biol 2014, 50:595–604.
- Hernandez BJ, Cain MP, Lynch AM, Flores JR, Tuvim MJ, Dickey BF, Chen J: Intermediary Role of Lung Alveolar Type 1 Cells in Epithelial Repair upon Sendai Virus Infection. Am J Respir Cell Mol Biol 2022, 67:389–401.
- Choi J, Park JE, Tsagkogeorga G, Yanagita M, Koo BK, Han N, Lee JH: Inflammatory Signals Induce AT2 Cell-Derived Damage-Associated Transient Progenitors that Mediate Alveolar Regeneration. Cell Stem Cell 2020, 27:366–382.e367. [CrossRef]
- Zheng D, Limmon GV, Yin L, Leung NH, Yu H, Chow VT, Chen J: Regeneration of alveolar type I and II cells from Scgb1a1-expressing cells following severe pulmonary damage induced by bleomycin and influenza. PLoS One 2012, 7:e48451. [CrossRef]
- Kathiriya JJ, Brumwell AN, Jackson JR, Tang X, Chapman HA: Distinct Airway Epithelial Stem Cells Hide among Club Cells but Mobilize to Promote Alveolar Regeneration. Cell Stem Cell 2020, 26:346–358.e344.
- Tian B, Patrikeev I, Ochoa L, Vargas G, Belanger KK, Litvinov J, Boldogh I, Ameredes BT, Motamedi M, Brasier AR: NF-kappaB Mediates Mesenchymal Transition, Remodeling, and Pulmonary Fibrosis in Response to Chronic Inflammation by Viral RNA Patterns. Am J Respir Cell Mol Biol 2017, 56:506–520.
- Zhang J, Jamaluddin M, Zhang Y, Widen SG, Sun H, Brasier AR, Zhao Y: Type II Epithelial-Mesenchymal Transition Upregulates Protein N-Glycosylation To Maintain Proteostasis and Extracellular Matrix Production. J Proteome Res 2019, 18:3447–3460.
- Auyeung VC, Downey MS, Thamsen M, Wenger TA, Backes BJ, Sheppard D, Papa FR: IRE1α drives lung epithelial progenitor dysfunction to establish a niche for pulmonary fibrosis. Am J Physiol Lung Cell Mol Physiol 2022, 322:L564–l580.
- Qiao D, Skibba M, Xu X, Garofalo RP, Zhao Y, Brasier AR: Paramyxovirus replication induces the hexosamine biosynthetic pathway and mesenchymal transition via the IRE1alpha-XBP1s arm of the unfolded protein response. Am J Physiol Lung Cell Mol Physiol 2021, 321:L576–L594. [CrossRef]
- Tian B, Widen SG, Yang J, Wood TG, Kudlicki A, Zhao Y, Brasier AR: The NFkappaB subunit RELA is a master transcriptional regulator of the committed epithelial-mesenchymal transition in airway epithelial cells. J Biol Chem 2018, 293:16528–16545. [CrossRef]
- Erjefalt JS, Erjefalt I, Sundler F, Persson CG: In vivo restitution of airway epithelium. Cell Tissue Res 1995, 281:305–316.
- Zhang J, Tian XJ, Zhang H, Teng Y, Li R, Bai F, Elankumaran S, Xing J: TGF-beta-induced epithelial-to-mesenchymal transition proceeds through stepwise activation of multiple feedback loops. Sci Signal 2014, 7:ra91.
- Tian B, Yang J, Zhao Y, Ivanciuc T, Sun H, Garofalo RP, Brasier AR: BRD4 Couples NF-kappaB/RelA with Airway Inflammation and the IRF-RIG-I Amplification Loop in Respiratory Syncytial Virus Infection. J Virol 2017, 91:e00007–00017. [CrossRef]
- Shilovskiy IP, Yumashev KV, Nikolsky AA, Vishnyakova LI, Khaitov MR: Molecular and Cellular Mechanisms of Respiratory Syncytial Viral Infection: Using Murine Models to Understand Human Pathology. Biochemistry (Moscow) 2021, 86:290–306.
- Rameix-Welti MA, Le Goffic R, Hervé PL, Sourimant J, Rémot A, Riffault S, Yu Q, Galloux M, Gault E, Eléouët JF: Visualizing the replication of respiratory syncytial virus in cells and in living mice. Nat Commun 2014, 5:5104. [CrossRef]
- Schwarze J, O’Donnell DR, Rohwedder A, Openshaw PJM: Latency and Persistence of Respiratory Syncytial Virus Despite T Cell Immunity. American Journal of Respiratory and Critical Care Medicine 2004, 169:801–805.
- Duez H, Staels B: Rev-erb-alpha: an integrator of circadian rhythms and metabolism. J Appl Physiol (1985) 2009, 107:1972–1980.
- Gibbs JE, Blaikley J, Beesley S, Matthews L, Simpson KD, Boyce SH, Farrow SN, Else KJ, Singh D, Ray DW, Loudon ASI: The nuclear receptor REV-ERBα mediates circadian regulation of innate immunity through selective regulation of inflammatory cytokines. Proceedings of the National Academy of Sciences 2012, 109:582–587. [CrossRef]
- Gibbs J, Ince L, Matthews L, Mei J, Bell T, Yang N, Saer B, Begley N, Poolman T, Pariollaud M, et al.: An epithelial circadian clock controls pulmonary inflammation and glucocorticoid action. Nat Med 2014, 20:919–926.
- Ehlers A, Xie W, Agapov E, Brown S, Steinberg D, Tidwell R, Sajol G, Schutz R, Weaver R, Yu H, et al.: BMAL1 links the circadian clock to viral airway pathology and asthma phenotypes. Mucosal Immunol 2018, 11:97–111.
- Bilodeau C, Shojaie S, Goltsis O, Wang J, Luo D, Ackerley C, M Rogers I, Cox B, Post M: TP63 basal cells are indispensable during endoderm differentiation into proximal airway cells on acellular lung scaffolds. npj Regenerative Medicine 2021, 6:12. [CrossRef]
- Desai U, Lee EC, Chung K, Gao C, Gay J, Key B, Hansen G, Machajewski D, Platt KA, Sands AT, et al.: Lipid-lowering effects of anti-angiopoietin-like 4 antibody recapitulate the lipid phenotype found in angiopoietin-like 4 knockout mice. Proc Natl Acad Sci U S A 2007, 104:11766–11771.
- Goh YY, Pal M, Chong HC, Zhu P, Tan MJ, Punugu L, Lam CR, Yau YH, Tan CK, Huang RL, et al.: Angiopoietin-like 4 interacts with integrins beta1 and beta5 to modulate keratinocyte migration. Am J Pathol 2010, 177:2791–2803.
- Kirsch N, Chang L-S, Koch S, Glinka A, Dolde C, Colozza G, Benitez MDJ, De Robertis EM, Niehrs C: Angiopoietin-like 4 Is a Wnt Signaling Antagonist that Promotes LRP6 Turnover. Developmental Cell 2017, 43:71–82.e76.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



