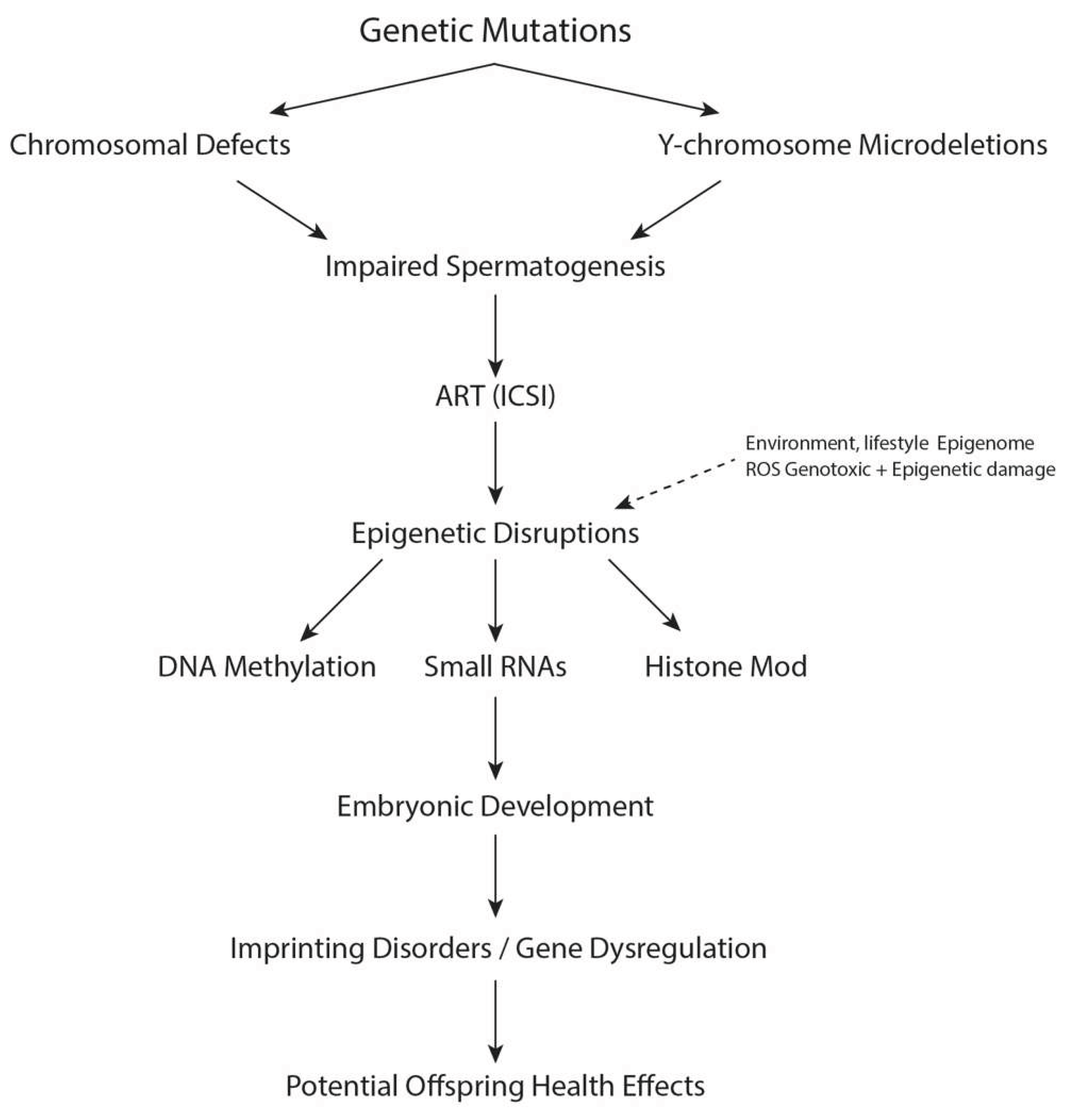
1. Introduction
Infertility occurs in about 15% of couples of reproductive age across the globe, and male factors are responsible for about half of these [
1]. The advent of assisted reproductive technologies, more importantly intracytoplasmic sperm injection (ICSI), has transformed treatment options for severe male-factor infertility. Through fertilization with a single spermatozoon, ICSI overcomes a number of physiological hurdles such as sperm selection and natural defence mechanisms of the oocyte. Although this approach has dramatically increased pregnancy rates, it has also solicited concern regarding the potential transmission of genetic and epigenetic abnormalities carried in the paternal germline [
2,
3].
Male infertility encompasses a heterogeneous assortment of aetiologies, ranging from single-gene defects and chromosomal abnormalities to complex polygenic and environmental interactions [
4]. Up to 30% of cases are idiopathic, often with subtle genomic or epigenomic dysregulation that is not detectable with conventional testing [
5]. Whole-genome and whole-exome sequencing genomic medicine advances have uncovered a growing list of genes implicated in spermatogenic failure, sperm motility disorders, and endocrine regulation. The integration of these findings with transcriptomic and epigenomic research has demonstrated that genetic and epigenetic causes of male infertility are interconnected and often co-regulated [
6,
7].
Apart from genetic aetiology, recent evidence points to the role of epigenetic mechanisms (DNA methylation, histone modification, and small noncoding RNAs) in the regulation of sperm function and embryonic development. Exposures to the environment, paternal age, obesity, and oxidative stress are all linked to epigenetic alterations in spermatozoa that can be trans-generationally heritable. The process of assisted reproduction itself can also play a role through in vitro manipulation, cryopreservation, and culture media. As such, ART offspring do have a marginally increased frequency of imprinting disorders and epigenetic dysregulation, though the absolute risk is low [
8,
9,
10].
The aim of this review is to provide an updated synthesis of the molecular mechanisms of the genetic and epigenetic risk of male infertility in the ART setting. Through the integration of data from the past twenty years, this review highlights the mechanisms of heritable and acquired defects of spermatogenesis, the impact of ART on germline integrity, and the implications for reproductive counselling, screening, and long-term offspring health [
11,
12].
2. Genetic Basis of Male Infertility
2.1. Overview of Genetic Contributions to Male Infertility
Male infertility is a complex disorder with monogenic, chromosomal, and polygenic aetiology that has long been recognized (
Figure 1). Early research estimated that 15-30% of male infertility is accounted for by recognized genetic factors; however, emerging multi-omics research demonstrates that the proportion may be much higher. Genomic technologies revealed that a significant proportion of male infertility cases that were previously idiopathic are underpinned by mutations in genes that regulate spermatogonial proliferation, meiosis, or sperm structural integrity. They include genes that regulate flagellar assembly (
DNAH1,
CFAP43,
CFAP44), meiosis (
TEX11,
SYCP3), and chromatin remodelling (
TNP1,
PRM2). Use of next-generation sequencing (NGS) panels now enables high-resolution genetic characterization of infertile men, with diagnostic yields of up to 40% in non-obstructive azoospermia (NOA) and severe oligozoospermia [
6,
13,
14].
Genetic defects found in infertile men often extend beyond classical chromosomal abnormalities. Copy number variations (CNVs), small insertions or deletions, and point mutations of important genes on autosomes and sex chromosomes have been described [
5]. The increasing availability of population-level reference datasets, such as gnomAD, has facilitated variant interpretation and genotype-phenotype correlation. Functional validation through animal models and patient-derived induced pluripotent stem cells has also delineated causal links between gene defects and spermatogenic arrest. Accumulating evidence highlights that infertility itself may be a sentinel phenotype for generalized genomic instability and predisposition to somatic disease, such as cancer and metabolic dysfunctions [
15].
2.2. Single-Gene Disorders and Monogenic Infertility
2.2.1. Cftr Mutations and Congenital Bilateral Absence of the Vas Deferens (Cbavd)
CFTR gene mutations remain the most frequent monogenic origin of obstructive azoospermia, leading to congenital bilateral absence of the vas deferens (CBAVD). The prevalence of
CFTR mutations among CBAVD patients is 60-90%, and compound heterozygosity for severe and mild alleles will result in partial or complete obstruction of the excurrent ducts [
16].
High-throughput sequencing, since 2006, has expanded the inventory of
CFTR variants, identifying over 2000 alleles with variable penetrance. Polymorphisms in intron 8 (5T, 7T, 9T repeats) and mutations such as R117H modulate splicing efficiency and protein function, influencing the clinical phenotype. Current recommendations include
CFTR mutation screening and genetic counselling for all men with obstructive azoospermia prior to ART, with preimplantation genetic testing (PGT) when both partners are carriers of pathogenic variants [
17,
18,
19].
2.2.2. Ciliary Dyskinesia and Flagellar Disorders
Primary ciliary dyskinesia (PCD), including Kartagener syndrome, is caused by mutations in genes encoding axonemal dynein arms and radial spoke proteins. Since the initial descriptions of DNAH5 and DNAI1, over 40 causative genes have now been described, including
CCDC39,
CCDC40,
DNAH11, and
CFAP300. These mutations cause defective sperm motility and chronic respiratory disease due to impaired ciliary motility. Whole-exome sequencing has facilitated the discovery of rare, family-specific mutations that account for subtle sperm motility phenotypes such as multiple morphological abnormalities of the sperm flagella (MMAF). ICSI remains a highly successful treatment, even though there is always the danger of transmission of disease-causing variants if the disorder has an autosomal recessive inheritance. Genetic diagnosis allows for personalized counselling and targeted therapy [
20,
21,
22].
2.2.3. Endocrine and Neurologic Genetic Disorders
Primary and secondary hypogonadism are caused by genetic disorders of gonadotropin signalling, androgen sensitivity, or hypothalamic–pituitary development. Mutations of
KAL1 and
FGFR1 lead to Kallmann syndrome, and mutations of the
GNRHR and
FSHR genes can result in isolated hypogonadotropic hypogonadism. Mutations of the androgen receptor (AR) gene result in varied entities of androgen insensitivity syndrome (AIS), from subtle spermatogenic failure to complete testicular feminization.
CYP21A2 and
SRD5A2 defects affect steroid biosynthesis and 5α-reductase activity. These disorders highlight the intersection of endocrinology and genetics in male infertility, with the necessity of molecular evaluation before ART [
23,
24].
2.2.4. Neuromuscular Repeat Expansion Disorders
Trinucleotide repeat expansion disorders such as Kennedy's disease (spinal bulbar muscular atrophy; AR gene CAG expansion) and myotonic dystrophy type 1 (
DMPK gene CTG expansion) are increasingly being recognized as etiologies of subfertility. These illnesses have spermatogenic failure and may transmit unstable repeat expansions to offspring through ART. Ethical considerations and genetic counseling are crucial because affected males may transmit neurodegenerative or systemic illness. Preimplantation genetic testing for monogenic disorders (PGT-M) is strongly indicated for such couples [
25,
26].
2.3. Chromosomal Abnormalities
Chromosomal abnormalities underlie 5–10% of the causes of genetic male infertility, with sex chromosome aneuploidies being common. The most common karyotypic abnormality is Klinefelter syndrome (47,XXY), seen in approximately 10% of azoospermic men. In mosaic forms (46,XY/47,XXY), restricted spermatogenesis is often preserved, and viable sperm can be recovered for ICSI. Although children of Klinefelter patients conceived by ICSI are usually chromosomally normal, preimplantation genetic testing (PGT-A) is recommended to prevent the transmission of aneuploid embryos. On the other hand, 47,XYY syndrome is generally associated with milder spermatogenic defect but a larger proportion of disomic spermatozoa. The application of fluorescence in situ hybridization (FISH) and single-cell genomic analysis has also enhanced risk assessment for chromosomal transmission by ART [
27,
28,
29].
Structural chromosome rearrangements, including reciprocal and Robertsonian translocations, inversions, and deletions, can disrupt meiotic segregation and cause infertility. Balanced translocations occur in approximately 1% of infertile men—10 times the frequency in the normal population. High-resolution breakpoint mapping and identification of disrupted genes are now possible with current cytogenetic and sequencing techniques. Couples carrying balanced translocations benefit from PGT-SR (structural rearrangement testing) in preventing the implantation of unbalanced embryos [
30,
31,
32].
2.4. Y-Chromosome Microdeletions
Microdeletions in the azoospermia factor (AZF) regions of the Y chromosome are a major cause of non-obstructive azoospermia and severe oligozoospermia. The AZF locus, which spans AZFa, AZFb, and AZFc, contains a number of gene families that are important for spermatogenesis, including
USP9Y,
DBY,
RBMY,
DAZ, and
CDY1. Complete deletions of AZFa or AZFb result in Sertoli cell-only syndrome or spermatogenic arrest, while deletions of AZFc have variable phenotypes ranging from hypospermatogenesis to azoospermia. Current research has now also described partial deletions such as gr/gr and b2/b3, with variable effects depending on the Y haplogroup background. Men with AZFc deletions can, on the odd occasion, produce viable sperm and successful ICSI can be obtained, but all male offspring will inherit the same deletion, transmitting infertility through generations [
33,
34,
35].
Technology has enhanced detection from PCR-based assays to high-throughput sequencing and digital droplet PCR, enabling more accurate delineation of the boundaries of microdeletions and structural polymorphisms. Guidelines now suggest Y-chromosome microdeletion testing in all men with sperm concentrations below 5 million/mL. Cryopreservation of sperm from AZFc-deleted patients is advised due to ongoing testicular degeneration. Genetic counseling should explicitly address the certainty of transmission to the male offspring and potential associations with chromosomal instability and aneuploid risk [
36].
2.5. Sperm Chromosomal Aneuploidy and Genetic Instability
Sperm from infertile men carry significantly greater frequencies of chromosomal aneuploidy than sperm from fertile controls. Those who have oligozoospermia, testicular failure, or chromosomal rearrangements have increased disomy for sex chromosomes and autosomes 13, 18, and 21. ICSI with these sperm may result in embryos with mosaicism or aneuploidy that may be detected by preimplantation genetic diagnosis. Single-cell high-throughput sequencing has confirmed that the degree of sperm aneuploidy correlates with the severity of impairment of spermatogenesis. These findings emphasize the need for gamete genetic testing in ART to minimize the transmission of chromosomal abnormalities [
37].
3. Epigenetic Control and Dangers in Assisted Reproductive Technologies
3.1. Epigenetic Reprogramming in the Male Germ Line
Spermatogenesis entails extensive epigenetic remodeling that ensures correct transmission of paternal genomic information while re-establishing epigenetic marks for embryonic development. During spermatogonial differentiation, histones are replaced sequentially by transition proteins and protamines, condensing the paternal genome and establishing a highly specialized chromatin state. Concurrently, DNA methylation patterns and histone modifications such as H3K4me3 and H3K27me3 coordinate gene silencing and activation. Defects in this exquisitely coordinated process undermine sperm function and embryonic developmental potential. Defects in DNA methyltransferases (DNMT1, DNMT3A/B) or histone-modifying enzymes have been associated with spermatogenic arrest, defective chromatin packaging, and impaired fertilizing ability. The dynamic nature of these epigenetic marks makes them vulnerable to environmental, metabolic, and age insults, with the potential to cause heritable epimutations [
38,
39].
3.2. Environmental and Lifestyle Impacts on the Sperm Epigenome
Emerging evidence indicates that environmental exposures can reshape the sperm epigenome and influence offspring health. Smoking, obesity, endocrine-disrupting chemicals, air pollution, and advanced paternal age have been associated with abnormal DNA methylation and histone retention in spermatozoa. Oxidative stress, which represents a common final denominator for many of these exposures, results in base oxidation, protamination disruption, and imprinted gene methylation disturbance at loci such as H19 and IGF2. Folate, zinc, and omega-3 fatty acids represent some of the nutritional influences that impact one-carbon metabolism and can be utilized to buffer epigenetic defects partially. Animal models demonstrate that high-fat or low-protein paternal diets reprogram sperm small-RNAs content, transmitting metabolic phenotypes to subsequent generations. The results support the concept that pre-conceptional paternal lifestyle contributes to the epigenetic quality of gametes and may influence ART outcomes [
40,
41].
3.3. Art-Induced Epigenetic Perturbations
While ART procedures have been highly successful, they involve non-physiological manipulations, which may interfere with natural epigenetic reprogramming. Ovarian hyperstimulation, in vitro gamete manipulation, composition of the culture medium, cryopreservation, and micromanipulation have all been involved in subtle epigenetic defects [
42]. ART-conceived versus naturally conceived offspring comparisons reveal minor but significant differences in DNA methylation status, particularly at imprinted genes. The incidence of imprinting disorders such as Beckwith–Wiedemann, Silver–Russell, and Angelman syndromes is slightly increased in children conceived by ICSI or in vitro fertilization, which suggests that epigenetic remodeling in early embryos is ex vivo condition-sensitive. These effects, nevertheless, appear to be stochastic rather than deterministic and are influenced by the embryo culture duration, oxygen tension, and parental epigenetic status [
43,
44].
3.4. Genomic Imprinting and Parent-of-Origin Effects
Genomic imprinting ensures parent-specific monoallelic expression of key developmental genes, and its failure results in congenital malformations and abnormal growth patterns. Methylation imprints in the male germ line are established during fetal and perinatal life and maintained throughout spermatogenesis. ART bypasses natural mechanisms of sperm selection for epigenetically stable gametes, and concern exists that the zygote will inherit flawed imprinting. Aberrant methylation of
H19,
KCNQ1OT1, and
MEG3 has been described in infertile men's sperm and in embryos from ICSI cycles. These imprinting alterations can also be found in somatic tissues of the offspring, pointing to the transgenerational effect of paternal epigenetic integrity. Despite the generally low risk, clinical vigilance and long-term follow-up studies are warranted [
45,
46].
3.5. Sperm Small Rnas and Transgenerational Inheritance
Along with DNA and histone modifications, spermatozoa carry a sophisticated inventory of small noncoding RNAs—including microRNAs (miRNAs), PIWI-interacting RNAs (piRNAs), and tRNA-derived fragments (tsRNAs)—that contribute to early embryonic gene regulation. The RNA cargo can be reconfigured in response to environmental or metabolic insults, thereby reprogramming embryonic transcriptional programs after fertilization. Experimental models have shown that father's stress, diet, and toxin exposure can reprogram offspring metabolism and behavior via sperm-borne small RNAs, without changing DNA sequence. These findings have redefined paradigms of inheritance, positioning the sperm epigenome as a vector of environmental memory. Human data, though limited, support the relevance of sperm small-RNA signatures as biomarkers of fertility potential and ART outcome [
47,
48].
3.6. Interaction Between Epigenetic and Genetic Mechanisms
Genetic and epigenetic mechanisms interact strongly in the pathophysiology of male infertility. Mutations in genes that regulate chromatin remodeling, such as
TDRD9,
DNMT3L, and
KAT8, result in secondary epigenetic disruption, while epigenetic instability can predispose to genomic damage and aneuploidy. Oxidative DNA damage and defective DNA repair generate both sequence mutation and abnormal methylation, establishing a vicious cycle of genomic and epigenomic instability. In ART, these mechanisms can converge: paternal germline defects predispose to culture-induced epimutations, and in vitro stress exacerbates the expression of occult genetic susceptibilities. Combined multi-omics approaches now permit simultaneous assessment of sperm DNA integrity, chromatin accessibility, and methylation, with unprecedented resolution of this interplay [
6,
49].
3.7. Clinical Implications and Risk Mitigation
Detection of epigenetic susceptibility in ART has prompted laboratory protocol optimization to minimize environmental stress during gamete and embryo handling. Optimized oxygen concentrations (5%), antioxidant supplementation, and shorter durations of in vitro culture have been shown to preserve normal methylation patterns. Epigenetic testing of sperm prospectively—through methylation arrays or small-RNA profiling—is being refined as an adjunct to conventional semen analysis. Besides, preconception lifestyle modification and antioxidant therapy can improve sperm epigenetic stability. Preconceptional counselling of the couples regarding these factors is a feasible strategy for reducing the epigenetic burden transmitted through ART [
50] (
Table 1).
4. Molecular Mechanisms Bridging Genetics and Epigenetics in Male Infertility
4.1. Oxidative Stress as A Common Pathogenic Driver
Oxidative stress is perhaps the most common mechanism coupling genetic and epigenetic damage in the male germ line. Reactive oxygen species (ROS) generated following inflammation, testicular heat stress, or environmental exposures target sperm DNA, lipids, and proteins. With mature spermatozoa possessing minimal antioxidant defences and lacking DNA repair capability, oxidative lesions such as 8-hydroxy-2′-deoxyguanosine (8-OHdG) accumulate with high velocity, causing single- and double-strand breaks. These lesions cause mutations, chromosomal rearrangements, and aberrant methylation of CpG islands. ROS also oxidize methylcytosine to hydroxymethylcytosine, interfering with DNA methyltransferase binding and leading to epigenetic drift. The occurrence of elevated oxidative markers in seminal plasma is strongly associated with sperm DNA fragmentation, global hypomethylation, and decreased fertilization rates following ICSI. Cumulatively, these findings position oxidative stress as a molecular bridge between genotoxic and epigenetic instability in male infertility [
51,
52].
4.2. Dna Damage Response and Chromatin Remodelling
The histone- to protamine-based chromatin remodelling in spermiogenesis entails the precise coordination of topoisomerases, nucleases, and chromatin-remodelling enzymes. Disruption of this process is manifested by incomplete protamination, residual nucleosomal retention, and an open chromatin structure prone to DNA breaks. Mutations or dysregulation of histone acetyltransferases (
KAT8,
CREBBP), deacetylases (
HDAC1,
SIRT1), and ATP-dependent remodelers (
SMARCA4,
CHD5) have been associated with faulty sperm chromatin packaging. Abnormal protamine P1/P2 ratios are always associated with increased DNA fragmentation and aberrant methylation of paternally imprinted loci. Under ART, spermatozoa bearing such chromatin defects are still fertilizing but may transmit DNA lesions not repaired after fertilization, resulting in embryo arrest or de novo mutations. Chromatin remodelling defects thus constitute a molecular interface through which epigenetic deregulation is translated into genetic instability [
53,
54].
4.3. Sperm Energy Metabolism and Mitochondrial Dysfunction
Mitochondria are important determinants of sperm motility and redox balance. Accumulation of mtDNA mutations, either inherited or developed as a result of oxidative damage, derails ATP synthesis and enhances ROS production. Sperm with greater mtDNA copy number and deletions have compromised motility and fertilizing capacity. Furthermore, mtDNA mutations can indirectly influence nuclear epigenetics by virtue of altered NAD⁺/NADH ratios and modulation of one-carbon metabolism, thereby controlling histone and DNA methylation patterns. Recent metabolomic studies demonstrate that mitochondrial dysfunction accounts for global sperm hypomethylation and defective imprint establishment, and mitochondrial–epigenetic cross-talk is implicated in the core of male infertility and ART success [
55,
56].
4.4. Dna Methylation Drift and Epimutation Propagation
Male germ cells undergo sequential waves of de- and re-methylation during development to establish sex-specific imprinting and maintain genomic stability. Aberrant methylation patterns, either due to mutations in methyltransferases (
DNMT3A,
DNMT3L) or environmental stress, cause localized or global hypomethylation. This destabilizes repetitive elements, promoting transposon activation and double-strand breaks. Methylation drift stochastically accrues across spermatogenic generations, predisposing individuals to meiotic errors and sperm aneuploidy with time. In ART, the use of sperm from older men or those experiencing chronic oxidative stress exacerbates such epigenetic erosion, with the possibility of inducing imprinting instability in embryos. The perseverance of such methylation errors into offspring somatic tissues underscores their heritability [
57].
4.5. Dysregulation of Noncoding Rna and Post-Transcriptional Regulation
Testicular epigenetic regulation is not confined to chromatin marks but also includes small and long noncoding RNAs (ncRNAs). These molecules govern transcriptional activity and genome protection during spermatogenesis. piRNAs, for instance, silence transposable elements, while tRNA-derived fragments modulate translation efficiency. Mutations in piRNA biogenesis genes (
PIWIL1,
TDRD9,
MOV10L1) cause failure of spermatogenesis with widespread transposon de-repression and epigenetic collapse. Environmental insults and ART conditions can alter sperm small-RNA content, reshaping early embryonic transcriptomes and developmental trajectories. This intersection of RNA-mediated and chromatin-based control is a hallmark of how genetic and epigenetic pathways interlock to safeguard germline integrity [
58].
4.6. Oxidative-Epigenetic Feedback Loops in Art
The ART laboratory environment can intensify oxidative–epigenetic interactions. Sperm centrifugation, prolonged incubation, and cryopreservation procedures induce ROS and disrupt sperm nuclear architecture. Oxidized guanine residues within promoter CpG islands compromise methylation maintenance, inducing localized epimutations. Conversely, aberrant methylation of antioxidant defence genes such as
SOD2 and
GPX4 further reduces the tolerance of the sperm to oxidative stress, generating a self-sustaining cycle. Clinical evidence indicates that men with high sperm DNA fragmentation and aberrant methylation patterns have lower blastocyst formation rates and increased early miscarriage following ICSI. Antioxidant-containing media and optimized oxygen tension (5%) have been shown to buffer such effects, emphasizing the need for molecularly driven ART optimization [
59].
4.7. Multi-Omics Integration and Systems-Level Insights
Emerging multi-omics technologies have enabled the in-depth profiling of the sperm epigenome, transcriptome, proteome, and metabolome. Integrative investigations demonstrate that infertile men exhibit a concerted disruption across these molecular layers, reflecting convergent pathophysiologic processes. For example, hypomethylation of chromatin remodeler promoters (CHD5, BRDT) is linked to altered histone retention and downstream proteomic defects. Such multi-level derangement suggests that male infertility is not a failure of a single molecular process but a systems failure of genome maintenance. These findings form the basis of a paradigm shift towards personalized, molecularly driven reproductive medicine, with sperm "omics" signatures guiding diagnosis, prognosis, and ART strategy [
60,
61] (
Table 2).
5. Clinical and Ethical Implications in Art
5.1. Integration of Molecular Diagnostics in Infertility Investigation
It has been recognized that epigenetic and genetic abnormalities are a significant aetiology of male infertility, and this has transformed diagnostic paradigms. Traditional semen analysis, while helpful in assessing sperm count, motility, and morphology, won't detect molecular abnormalities underlying possible negative reproductive outcomes. The incorporation of advanced diagnostics—karyotyping, Y-chromosome microdeletion analysis,
CFTR mutation screening, and next-generation sequencing panels for targeted genes—has become the norm in tertiary fertility centres. Most recently, epigenetic profiling tests, including sperm DNA methylation arrays and small-RNA signatures, have emerged as highly promising biomarkers of sperm quality and embryo developmental potential. These molecular assays not only improve etiologic diagnosis but also inform patient counselling, allowing clinicians to tailor ART strategies and foresee potential heritable risks [
14,
62].
5.2. Preimplantation Genetic and Epigenetic Testing
Preimplantation genetic testing (PGT) is a cornerstone of risk reduction in ART for those with known genetic disease. PGT for aneuploidy (PGT-A) improves implantation efficiency and reduces miscarriage risk by selecting euploid embryos. PGT for monogenic disorders (PGT-M) allows detection of single-gene defects such as
CFTR mutations or repeat expansions, while PGT for structural rearrangements (PGT-SR) is applicable in translocation carriers. Recent developments in genome-wide haplotyping and low-input sequencing enable the simultaneous identification of chromosomal and point mutations from small embryo biopsy samples. Although standard epigenetic testing is not yet routinized, pilot studies using bisulfite sequencing and methylation arrays show potential for identifying embryos with abnormal imprinting or methylation instability. These integrated approaches herald a new age of precision embryology in which genomic and epigenomic integrity drive embryo choice [
63,
64].
5.3. Genetic Counselling and Risk Communication
Genetic counselling is necessary for the explanation of test results, transmission risk discussion, and managing ethical concerns in ART. Men who have Y-chromosome microdeletions, for instance, must be informed that any male child born by ICSI will also inherit the deletion and may be infertile. Similarly, couples where both partners are carriers of a
CFTR mutation or chromosomal rearrangement must be counselled about the potential need for PGT or donor gametes. Counselling needs to move beyond deterministic risk to cover probabilistic and epigenetic influences, as well as the possible but immeasurable risk of imprinting disorders. Increasing molecular complexity demands multidisciplinary teams—clinical geneticists, reproductive endocrinologists, and bioethicists—to promote open and even-handed communication with patients [
65,
66].
5.4. Sperm Selection and Processing Advances
On the basis of the proven inter relationships between sperm DNA fragmentation, chromatin instability, and ART outcomes, new sperm selection methods have been developed to identify gametes with improved molecular integrity. Hyaluronic acid (HA) binding assays, magnetic-activated cell sorting (MACS), and microfluidic sperm sorting systems are a few of the methods that exploit biophysical and biochemical markers of sperm maturity and DNA integrity. Raman spectroscopy and micro-optical tweezers are being investigated for non-invasive molecular screening of spermatozoa before ICSI. These technologies can enrich for sperm with intact chromatin and healthy epigenetic marks, leading to improved fertilization and embryo quality. Although still experimental, their integration into ART workflows can be anticipated to reduce the burden of genetic and epigenetic defects transmitted to offspring [
67,
68].
5.5. Lifestyle, Nutritional, and Pharmacologic Interventions
The epigenome of sperm is liable to dynamic alteration, and its stability could be reinforced through counteractive measures against oxidative and metabolic stress. Life-style modification (i.e., smoking, weight, and alcohol consumption reduction) optimises semen parameters and reduces DNA fragmentation. Nutritional therapy with antioxidants (vitamin C, vitamin E, coenzyme Q10, selenium, zinc) and methyl donors (folate, betaine) supports one-carbon metabolism and methylation homeostasis. Novel pharmacologic therapies targeting mitochondrial activity and redox status are also promising to restore sperm epigenetic integrity. Randomized trials have demonstrated that preconception antioxidant treatment improves fertilization and live birth rates in couples undergoing ART, likely through reduction of oxidative DNA damage and normalization of imprinted gene methylation [
69].
5.6. Offspring Outcomes and Long-Term Follow-Up
Concerns about the health of ART offspring have motivated epigenomic and epidemiologic research on a grand scale over the past two decades. While most ART children experience normal growth and mental development, there has been a small increase in imprinting disorders and cardiometabolic risk factors. Methylation profiling of cord blood and placental tissue discloses subtle but consistent differences in epigenetic marks, namely at imprinted gene clusters and developmental genes. Long-term follow-up studies are beginning to establish whether these differences persist into adulthood or predispose to susceptibility to disease in later life. Current evidence suggests that while ART does not carry major health risk, continued monitoring of epigenetic outcomes is crucial to intergenerational safety [
70].
5.7. Ethical and Regulatory Considerations
The potential of ART to circumvent natural selection raises ethical and societal questions regarding germline integrity and the boundaries of medical intervention. Ethical considerations are focused on the transmission risk of genetic or epigenetic defects to subsequent generations and the role of the clinician in minimizing such risks. International guidelines by ESHRE and ASRM emphasize informed consent, disclosure of results of genetic tests, and the avoidance of experimental methods in the absence of satisfactory safety data. The introduction of genome editing technologies, such as CRISPR-Cas9, has only intensified ethical debate, particularly for the hypothetical correction of inherited mutations in embryos or gametes. Consensus strongly prohibits germline editing for reproductive purposes outside well-controlled research environments. Continued debate among researchers, ethicists, regulators, and the public is still required to balance reproduction autonomy with genomic stewardship in the long term [
71,
72].
5.8. Future Directions in Clinical Translation
The advent of artificial intelligence and multi-omics diagnostics in reproductive medicine has created new opportunities for personalized medicine. Machine learning algorithms have the potential to integrate genetic, epigenetic, and phenotypic datasets for the prediction of ART outcomes and embryo selection. Single-cell sequencing and spatial transcriptomics will offer a glimpse into gamete and embryo development at a more profound level, guiding individualized intervention strategies. Meanwhile, ethical standards must evolve to address data privacy, algorithmic bias, and equitable access to emerging technologies. The confluence of molecular biology, computer science, and bioethics will form the future of precision reproductive medicine, leading ART from empirical to data-driven science [
73,
74].
6. Future Perspectives
6.1. the Era of Multi-Omics and Systems Reproductive Biology
The integration of genomics, epigenomics, transcriptomics, proteomics, and metabolomics is reshaping the field of male infertility research. Multi-omics investigations reveal that sperm pathology does not result from a single molecular defect but is an expression of systemic dysregulation at the biological strata levels. Concurrent DNA methylation, chromatin accessibility, and RNA expression profiling is feasible now and allows for the identification of molecular signatures for sperm quality prediction and ART outcome. Recent studies with single-cell RNA sequencing have characterized transcriptional heterogeneity among spermatogonial and Sertoli cell populations and revealed new candidate genes involved in spermatogenic arrest. Systems biology approaches linking omics data to clinical phenotypes are at the forefront of developing integrative diagnostic models that can personalize infertility treatment and simplify ART protocols [
1,
6].
6.2. Artificial Gametogenesis and in Vitro Spermatogenesis
One of the active frontiers in reproductive biology is the differentiation of functional gametes from stem cells. The advent of induced pluripotent stem cell (iPSC) technology and organoid culture systems has made it possible to conduct in vitro spermatogenesis with increasing feasibility [
75]. These systems recapitulate key stages of germ cell differentiation, epigenetic reprogramming, and meiosis, providing experimental models for investigation of infertility mechanisms and testing of therapeutic interventions. Artificial gametogenesis can potentially be useful for those with complete germline failure, i.e., AZFa/b deletions or gonadotoxic damage. However, the establishment of appropriate imprinting and chromatin structure is a major hurdle. Preclinical data show that in vitro-derived gametes exhibit aberrant methylation at imprinted loci, emphasizing the importance of stringent epigenetic testing before clinical application. Ethical and regulatory frameworks must evolve alongside to govern responsible use of such technologies [
76].
6.3. Crispr-Based Functional Genomics and Gene Correction
Genome editing tools such as CRISPR-Cas9, base editors, and prime editors have emerged as integral to elucidating gene function in spermatogenesis and infertility. Precise correction of
CFTR,
TEX11, and
SYCP3 mutations in animal models has reinstated fertility, showcasing therapeutic promise. In human germline research, however, stringent ethical restrictions prevent reproductive applications. Even so, CRISPR-based functional genomics enables modelling of patient-derived mutations in vitro with unprecedented molecular mechanism resolution. The emergence of high-fidelity and epigenome-targeting CRISPR variants can one day enable reprogramming of aberrant sperm epigenomes with precision or rescue of developmentally arrested spermatogenesis without altering the DNA sequence. These technologies are slated to be utilized exclusively in research environments for the foreseeable future but do mark the convergence of mechanistic understanding and translational potential within reproductive genetics [
77].
6.4. Artificial Intelligence in Reproductive Genomics
Artificial intelligence (AI) and machine learning (ML) are increasingly being applied to high-dimensional biological data analysis and ART outcome prediction for some time to come. Deep learning models trained on sperm imagery, embryo morphokinetics, and molecular profiles have the ability to recognize subtle patterns that are not visible to the human eye. The combination of genomic and epigenomic information with AI-based decision systems allows for embryo selection and personalized stimulation protocols to be more objective. Predictive models using sperm methylation patterns and fragmentation indices are being established to predict fertilization potential and implantation success. With improved computational precision, AI systems will find a place in ART laboratories, complementing the abilities of embryologists and geneticists. Transparency of algorithms and data ethics will be crucial in ensuring fair and unbiased application of such technologies [
78].
6.5. Epigenetic Therapies and Reproductive Precision Medicine
A novel goal in reproductive medicine is the correction or stabilization of epigenetic defects before conception. Epigenetic therapies—ranging from small-molecule modulators of DNMTs and HDACs and dietary interventions to antioxidant supplementation, are being investigated to restore sperm epigenome integrity. Pharmacologic agents that enhance NAD⁺ metabolism and sirtuin activity have demonstrated potential in optimizing chromatin compaction and mitochondrial function. The pre-conceptional window is a critical period for such therapies since sperm epigenetic marks are continuously remodelled during spermatogenesis. The future model is likely to involve molecular stratification of patients according to omics-based biomarkers, then targeted lifestyle and pharmacologic strategies to optimize germline health prior to ART [
8].
6.6. Transgenerational Health and Longitudinal Cohorts
Ascertaining the long-term impact of ART on offspring health remains an active research area. Large-scale, multi-generational cohort studies are required to assess subtle genetic and epigenetic impacts over decades. Integration of biobank data, electronic health records, and molecular profiling will enable follow-up of developmental, metabolic, and neurocognitive outcomes in ART-conceived children. Recent evidence suggests that certain epigenetic alterations induced by ART are reversible or compensated post-natally, highlighting the plasticity of the epigenome. Longitudinal studies should therefore seek to identify permanent versus transient changes and their correlation with diseases of adult onset. These data will inform future ART optimization for reproductive success without intergenerational health compromise [
11,
79].
6.7. the Path Toward Molecularly Guided Reproduction
The introduction of molecular data into clinical decision-making marks a new era for reproductive medicine. Global preconception evaluation of the male germline (integrating genetic, epigenetic, and environmental considerations) will guide individualized ART strategies. AI-driven analysis of omics data will assist clinicians in a trade-off between reproductive effectiveness and genomic protection. Future ART programs will incorporate molecular diagnostics, predictive modelling, and precision intervention to establish a closed-loop system of "molecularly guided reproduction." Such development will require collaboration among geneticists, embryologists, data scientists, and ethicists and foster a framework that maximizes reproductive freedom while guarding against heritable genomic integrity [
80].
7. Conclusions
In the last two decades, molecular genetic and epigenomic advances have revolutionized the field of male infertility and its relevance to assisted reproduction. What was previously considered to be largely endocrine or structural in nature is now a multifactorial process with complex interactions between genomic integrity, chromatin structure, and environmental regulation. Single-gene defects, Y-chromosome deletions, chromosomal abnormalities, and copy-number variations explain but one dimension of pathology; superimposed epigenetic derangements, at the level of aberrant methylation, disrupted histone retention, and altered small-RNA cargo-describe a second, dynamic dimension of reproductive risk [
81].
Widespread use of ICSI and other ART modalities has circumvented natural selection processes, enabling fatherhood in men carrying severe spermatogenic defects. However, such technological success brings responsibility: ART has the potential to inadvertently pass on genetic or epigenetic flaws to the next generation. While the absolute risk for clinically manifest disorders is low, molecular abnormalities have been described in ART offspring. These reports stress that the integration of molecular diagnostics, preimplantation testing, and long-term epidemiologic follow-up is warranted in reproductive medicine [
82].
A comprehensive approach to male infertility must therefore traverse the range from genome to phenotype, encompassing oxidative stress, DNA fragmentation, chromatin remodeling, and imprinting maintenance. Multidisciplinary evaluation (welcoming molecular biologists, genetic counselors, embryologists, and clinicians) is paramount for the translation of omics results into actionable protocols. Lifestyle optimization, antioxidant therapy, and precision sperm selection can already pad against certain molecular risks, and emerging technologies such as single-cell sequencing and multi-omics integration are poised to revolutionize diagnosis and treatment [
83].
In the future, the field will move into an age of molecularly guided reproduction. Artificial intelligence will also assist clinicians in the future to interpret complex genomic and epigenomic data, ART outcome prediction, and determination of optimal embryos for transfer. The future of reproductive medicine will be in balancing technological capability with ethical responsibility, so that the pursuit of fertility is not at the cost of undermining the integrity of the human germline. Achieving this balance will require sustained debate among scientists, ethicists, and the public, based on a shared interest in safe and equitable reproductive innovation.
Author Contributions
Conceptualization, A.ZIK. and I.G.; writing—original draft preparation, A.ZIK., P.K., M.F., A.ZAC., A.S., I.B., C.K., S.M., I.G.; writing—review and editing, A.ZIK., P.K., M.F., A.ZAC., A.S., I.B., C.K., S.M., I.G.; supervision, I.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data was created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| AI |
Artificial intelligence |
| AIS |
Androgen insensitivity syndrome |
| AR |
Androgen receptor |
| ART |
Assisted reproductive technologies |
| AZF |
Azoospermia factor |
| CBAVD |
Congenital bilateral absence of the vas deferens |
| CNVs |
Copy number variations |
| FISH |
Fluorescence in situ hybridization |
| HA |
Hyaluronic acid |
| ICSI |
Intracytoplasmic sperm injection |
| iPSC |
Induced pluripotent stem cell |
| MACS |
Magnetic-activated cell sorting |
| miRNAs |
microRNAs |
| ML |
Machine learning |
| MMAF |
Multiple morphological abnormalities of the sperm flagella |
| ncRNAs |
Noncoding RNAs |
| NGS |
Next-generation sequencing |
| NOA |
Non-obstructive azoospermia |
| PCD |
Primary ciliary dyskinesia |
| PGT |
Preimplantation genetic testing |
| PGT-A |
Preimplantation genetic testing for aneuploidy |
| PGT-M |
Preimplantation genetic testing for monogenic disorders |
| PGT-SR |
Preimplantation genetic testing for structural rearrangements |
| piRNAs |
PIWI-interacting RNAs |
| ROS |
Reactive oxygen species |
| tsRNAs |
tRNA-derived fragments |
| 8-OHdG |
8-hydroxy-2’-deoxyguanosine |
| |
|
References
- Assidi, M. Infertility in Men: Advances towards a Comprehensive and Integrative Strategy for Precision Theranostics. Cells 2022, 11, 1711. [Google Scholar] [CrossRef]
- Henningsen, A.-K.A.; Opdahl, S.; Wennerholm, U.-B.; Tiitinen, A.; Rasmussen, S.; Romundstad, L.B.; Bergh, C.; Gissler, M.; Forman, J.L.; Pinborg, A. Risk of Congenital Malformations in Live-Born Singletons Conceived after Intracytoplasmic Sperm Injection: A Nordic Study from the CoNARTaS Group. Fertil Steril 2023, 120, 1033–1041. [Google Scholar] [CrossRef] [PubMed]
- Sciorio, R.; Esteves, S.C. Contemporary Use of ICSI and Epigenetic Risks to Future Generations. J Clin Med 2022, 11, 2135. [Google Scholar] [CrossRef] [PubMed]
- Tesarik, J. Lifestyle and Environmental Factors Affecting Male Fertility, Individual Predisposition, Prevention, and Intervention. Int J Mol Sci 2025, 26, 2797. [Google Scholar] [CrossRef]
- Sudhakar, D.V.S.; Shah, R.; Gajbhiye, R.K. Genetics of Male Infertility - Present and Future: A Narrative Review. J Hum Reprod Sci 2021, 14, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Wagner, A.O.; Turk, A.; Kunej, T. Towards a Multi-Omics of Male Infertility. World J Mens Health 2023, 41, 272–288. [Google Scholar] [CrossRef]
- Huang, X.-Y.; Sha, J.-H. Proteomics of Spermatogenesis: From Protein Lists to Understanding the Regulation of Male Fertility and Infertility. Asian J Androl 2011, 13, 18–23. [Google Scholar] [CrossRef]
- Kaltsas, A.; Markou, E.; Kyrgiafini, M.-A.; Zikopoulos, A.; Symeonidis, E.N.; Dimitriadis, F.; Zachariou, A.; Sofikitis, N.; Chrisofos, M. Oxidative-Stress-Mediated Epigenetic Dysregulation in Spermatogenesis: Implications for Male Infertility and Offspring Health. Genes (Basel) 2025, 16, 93. [Google Scholar] [CrossRef]
- Donkin, I.; Barrès, R. Sperm Epigenetics and Influence of Environmental Factors. Mol Metab 2018, 14, 1–11. [Google Scholar] [CrossRef]
- Hattori, H.; Hiura, H.; Kitamura, A.; Miyauchi, N.; Kobayashi, N.; Takahashi, S.; Okae, H.; Kyono, K.; Kagami, M.; Ogata, T.; et al. Association of Four Imprinting Disorders and ART. Clin Epigenetics 2019, 11, 21. [Google Scholar] [CrossRef]
- Ahmadi, H.; Aghebati-Maleki, L.; Rashidiani, S.; Csabai, T.; Nnaemeka, O.B.; Szekeres-Bartho, J. Long-Term Effects of ART on the Health of the Offspring. Int J Mol Sci 2023, 24, 13564. [Google Scholar] [CrossRef]
- Graziani, A.; Rocca, M.S.; Vinanzi, C.; Masi, G.; Grande, G.; De Toni, L.; Ferlin, A. Genetic Causes of Qualitative Sperm Defects: A Narrative Review of Clinical Evidence. Genes (Basel) 2024, 15, 600. [Google Scholar] [CrossRef] [PubMed]
- Montjean, D.; Beaumont, M.; Natiq, A.; Louanjli, N.; Hazout, A.; Miron, P.; Liehr, T.; Cabry, R.; Ratbi, I.; Benkhalifa, M. Genome and Epigenome Disorders and Male Infertility: Feedback from 15 Years of Clinical and Research Experience. Genes (Basel) 2024, 15, 377. [Google Scholar] [CrossRef] [PubMed]
- Ikbal Atli, E.; Yalcintepe, S.; Atli, E.; Demir, S.; Gurkan, H. Next-Generation Sequencing Infertility Panel in Turkey: First Results. Balkan J Med Genet 2024, 27, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Gudmundsson, S.; Singer-Berk, M.; Watts, N.A.; Phu, W.; Goodrich, J.K.; Solomonson, M.; Genome Aggregation Database Consortium; Rehm, H.L.; MacArthur, D.G.; O’Donnell-Luria, A. Variant Interpretation Using Population Databases: Lessons from gnomAD. Hum Mutat 2022, 43, 1012–1030. [Google Scholar] [CrossRef]
- Cui, X.; Wu, X.; Li, Q.; Jing, X. Mutations of the Cystic Fibrosis Transmembrane Conductance Regulator Gene in Males with Congenital Bilateral Absence of the Vas Deferens: Reproductive Implications and Genetic Counseling (Review). Mol Med Rep 2020, 22, 3587–3596. [Google Scholar] [CrossRef]
- Sorrentino, U.; Menegazzo, M.; Gabbiato, I.; Calosci, D.; Zambon, C.F.; Zuccarello, D. Challenges of Preimplantation Genetic Counselling in the Context of Cystic Fibrosis and Other CFTR-Related Disorders: A Monocentric Experience in a Cohort of 92 Couples. Genes (Basel) 2024, 15, 937. [Google Scholar] [CrossRef]
- Anjankar, N.; More, A.; Anjankar, A.P.; Mahajan, S.S.; Nawale, N. CFTR Gene Mutations and Their Role in Male Infertility: A Case Study. J Pharm Bioallied Sci 2025, 17, S1008–S1010. [Google Scholar] [CrossRef]
- Wosnitzer, M.S. Genetic Evaluation of Male Infertility. Transl Androl Urol 2014, 3, 17–26. [Google Scholar] [CrossRef]
- Castleman, V.H.; Romio, L.; Chodhari, R.; Hirst, R.A.; de Castro, S.C.P.; Parker, K.A.; Ybot-Gonzalez, P.; Emes, R.D.; Wilson, S.W.; Wallis, C.; et al. Mutations in Radial Spoke Head Protein Genes RSPH9 and RSPH4A Cause Primary Ciliary Dyskinesia with Central-Microtubular-Pair Abnormalities. Am J Hum Genet 2009, 84, 197–209. [Google Scholar] [CrossRef]
- Gileles-Hillel, A.; Mor-Shaked, H.; Shoseyov, D.; Reiter, J.; Tsabari, R.; Hevroni, A.; Cohen-Cymberknoh, M.; Amirav, I.; Brammli-Greenberg, S.; Horani, A.; et al. Whole-Exome Sequencing Accuracy in the Diagnosis of Primary Ciliary Dyskinesia. ERJ Open Res 2020, 6, 00213–02020. [Google Scholar] [CrossRef] [PubMed]
- Newman, L.; Chopra, J.; Dossett, C.; Shepherd, E.; Bercusson, A.; Carroll, M.; Walker, W.; Lucas, J.S.; Cheong, Y. The Impact of Primary Ciliary Dyskinesia on Female and Male Fertility: A Narrative Review. Hum Reprod Update 2023, 29, 347–367. [Google Scholar] [CrossRef]
- Hwang, K.; Yatsenko, A.N.; Jorgez, C.J.; Mukherjee, S.; Nalam, R.L.; Matzuk, M.M.; Lamb, D.J. Mendelian Genetics of Male Infertility. Ann N Y Acad Sci 2010, 1214, E1–E17. [Google Scholar] [CrossRef] [PubMed]
- Millar, A.C.; Faghfoury, H.; Bieniek, J.M. Genetics of Hypogonadotropic Hypogonadism. Transl Androl Urol 2021, 10, 1401–1409. [Google Scholar] [CrossRef]
- Kim, W.B.; Jeong, J.Y.; Doo, S.W.; Yang, W.J.; Song, Y.S.; Lee, S.R.; Park, J.W.; Kim, D.W. Myotonic Dystrophy Type 1 Presenting as Male Infertility. Korean J Urol 2012, 53, 134–136. [Google Scholar] [CrossRef]
- Lian, M.; Lee, C.G.; Chong, S.S. Robust Preimplantation Genetic Testing Strategy for Myotonic Dystrophy Type 1 by Bidirectional Triplet-Primed Polymerase Chain Reaction Combined With Multi-Microsatellite Haplotyping Following Whole-Genome Amplification. Front Genet 2019, 10, 589. [Google Scholar] [CrossRef]
- Kim, I.W.; Khadilkar, A.C.; Ko, E.Y.; Sabanegh, E.S. 47,XYY Syndrome and Male Infertility. Rev Urol 2013, 15, 188–196. [Google Scholar] [PubMed]
- Maiburg, M.; Repping, S.; Giltay, J. The Genetic Origin of Klinefelter Syndrome and Its Effect on Spermatogenesis. Fertil Steril 2012, 98, 253–260. [Google Scholar] [CrossRef]
- Lamb, D.J. Chromosome Defects and Male Factor Infertility. Fertil Steril 2025, 123, 933–942. [Google Scholar] [CrossRef]
- Verdoni, A.; Hu, J.; Surti, U.; Babcock, M.; Sheehan, E.; Clemens, M.; Drewes, S.; Walsh, L.; Clark, R.; Katari, S.; et al. Reproductive Outcomes in Individuals with Chromosomal Reciprocal Translocations. Genet Med 2021, 23, 1753–1760. [Google Scholar] [CrossRef]
- Wang, H.; Jia, Z.; Mao, A.; Xu, B.; Wang, S.; Wang, L.; Liu, S.; Zhang, H.; Zhang, X.; Yu, T.; et al. Analysis of Balanced Reciprocal Translocations in Patients with Subfertility Using Single-Molecule Optical Mapping. J Assist Reprod Genet 2020, 37, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Shetty, S.; Nair, J.; Johnson, J.; Shetty, N.; J, A.K.; Thondehalmath, N.; Ganesh, D.; Bhat, V.R.; M, S.; R, A.; et al. Preimplantation Genetic Testing for Couples with Balanced Chromosomal Rearrangements. J Reprod Infertil 2022, 23, 213–223. [Google Scholar] [CrossRef]
- Rabinowitz, M.J.; Huffman, P.J.; Haney, N.M.; Kohn, T.P. Y-Chromosome Microdeletions: A Review of Prevalence, Screening, and Clinical Considerations. Appl Clin Genet 2021, 14, 51–59. [Google Scholar] [CrossRef]
- Liu, X.; Qiao, J.; Li, R.; Yan, L.; Chen, L. Y Chromosome AZFc Microdeletion May Not Affect the Outcomes of ICSI for Infertile Males with Fresh Ejaculated Sperm. J Assist Reprod Genet 2013, 30, 813–819. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Costa, P.; Plancha, C.E.; Gonçalves, J. Genetic Dissection of the AZF Regions of the Human Y Chromosome: Thriller or Filler for Male (in)Fertility? J Biomed Biotechnol 2010, 2010, 936569. [Google Scholar] [CrossRef]
- Dai, B.; Zhao, D.; Sha, R.-N.; Cang, M. The Selection of Y Chromosome Microdeletion Detection Methods Based on Seminal Analysis Results: A Comparison of High-Throughput Sequencing and Fluorescence Quantitative Polymerase Chain Reaction (qPCR) Applications. Transl Androl Urol 2025, 14, 619–626. [Google Scholar] [CrossRef]
- Elnahas, R.F.; Behery, A.K.; Kholeif, S.; Orief, Y.I.; Elhady, G.M. Sperm Chromosomal Abnormalities in Infertile Men with Failed Intracytoplasmic Sperm Injection (ICSI). Middle East Fertil Soc J 2023, 28, 18. [Google Scholar] [CrossRef]
- Ren, W.; Gao, L.; Song, J. Structural Basis of DNMT1 and DNMT3A-Mediated DNA Methylation. Genes (Basel) 2018, 9, 620. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Deng, J.; Zhang, Y.; Du, L.; Jiang, F.; Li, C.; Chen, W.; Zhang, H.; He, Z. Epigenetic Regulation by DNA Methylation, Histone Modifications and Chromatin Remodeling Complexes in Controlling Spermatogenesis and Their Dysfunction with Male Infertility. Cell Mol Life Sci 2025, 82, 343. [Google Scholar] [CrossRef]
- Akhatova, A.; Jones, C.; Coward, K.; Yeste, M. How Do Lifestyle and Environmental Factors Influence the Sperm Epigenome? Effects on Sperm Fertilising Ability, Embryo Development, and Offspring Health. Clin Epigenetics 2025, 17, 7. [Google Scholar] [CrossRef]
- Sudhakaran, G.; Kesavan, D.; Kandaswamy, K.; Guru, A.; Arockiaraj, J. Unravelling the Epigenetic Impact: Oxidative Stress and Its Role in Male Infertility-Associated Sperm Dysfunction. Reprod Toxicol 2024, 124, 108531. [Google Scholar] [CrossRef]
- Sciorio, R.; Cantatore, C.; D’Amato, G.; Smith, G.D. Cryopreservation, Cryoprotectants, and Potential Risk of Epigenetic Alteration. J Assist Reprod Genet 2024, 41, 2953–2967. [Google Scholar] [CrossRef]
- Kopca, T.; Tulay, P. Association of Assisted Reproductive Technology Treatments with Imprinting Disorders. Glob Med Genet 2021, 8, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Håberg, S.E.; Page, C.M.; Lee, Y.; Nustad, H.E.; Magnus, M.C.; Haftorn, K.L.; Carlsen, E.Ø.; Denault, W.R.P.; Bohlin, J.; Jugessur, A.; et al. DNA Methylation in Newborns Conceived by Assisted Reproductive Technology. Nat Commun 2022, 13, 1896. [Google Scholar] [CrossRef]
- Rotondo, J.C.; Lanzillotti, C.; Mazziotta, C.; Tognon, M.; Martini, F. Epigenetics of Male Infertility: The Role of DNA Methylation. Front Cell Dev Biol 2021, 9, 689624. [Google Scholar] [CrossRef]
- Rahimi, S.; Shao, X.; Chan, D.; Martel, J.; Bérard, A.; Fraser, W.D.; Simon, M.-M.; Kwan, T.; Bourque, G.; Trasler, J. Capturing Sex-Specific and Hypofertility-Linked Effects of Assisted Reproductive Technologies on the Cord Blood DNA Methylome. Clin Epigenetics 2023, 15, 82. [Google Scholar] [CrossRef]
- Yang, C.; Zeng, Q.-X.; Liu, J.-C.; Yeung, W.S.-B.; Zhang, J.V.; Duan, Y.-G. Role of Small RNAs Harbored by Sperm in Embryonic Development and Offspring Phenotype. Andrology 2023, 11, 770–782. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Sharma, U. Sperm RNA Payload: Implications for Intergenerational Epigenetic Inheritance. Int J Mol Sci 2023, 24, 5889. [Google Scholar] [CrossRef]
- Hosseini, M.; Khalafiyan, A.; Zare, M.; Karimzadeh, H.; Bahrami, B.; Hammami, B.; Kazemi, M. Sperm Epigenetics and Male Infertility: Unraveling the Molecular Puzzle. Hum Genomics 2024, 18, 57. [Google Scholar] [CrossRef] [PubMed]
- Menezo, Y.; Dale, B.; Elder, K. Time to Re-Evaluate ART Protocols in the Light of Advances in Knowledge about Methylation and Epigenetics: An Opinion Paper. Hum Fertil (Camb) 2018, 21, 156–162. [Google Scholar] [CrossRef]
- Aitken, R.J. Impact of Oxidative Stress on Male and Female Germ Cells: Implications for Fertility. Reproduction 2020, 159, R189–R201. [Google Scholar] [CrossRef]
- Chakraborty, S.; Roychoudhury, S. Pathological Roles of Reactive Oxygen Species in Male Reproduction. Adv Exp Med Biol 2022, 1358, 41–62. [Google Scholar] [CrossRef]
- Deng, T.; Xiao, Y.; Dai, Y.; Xie, L.; Li, X. Roles of Key Epigenetic Regulators in the Gene Transcription and Progression of Prostate Cancer. Front Mol Biosci 2021, 8, 743376. [Google Scholar] [CrossRef]
- Hart, H.M.; Nixon, B.; Martin, J.H.; Aitken, R.J.; De Iuliis, G.N. Improving Sperm Selection Strategies for Assisted Reproduction through Closing the Knowledge Gap in Sperm Maturation Mechanics. Hum Reprod Open 2025, 2025, hoaf040. [Google Scholar] [CrossRef]
- Mai, Z.; Yang, D.; Wang, D.; Zhang, J.; Zhou, Q.; Han, B.; Sun, Z. A Narrative Review of Mitochondrial Dysfunction and Male Infertility. Transl Androl Urol 2024, 13, 2134–2145. [Google Scholar] [CrossRef]
- Vahedi Raad, M.; Firouzabadi, A.M.; Tofighi Niaki, M.; Henkel, R.; Fesahat, F. The Impact of Mitochondrial Impairments on Sperm Function and Male Fertility: A Systematic Review. Reprod Biol Endocrinol 2024, 22, 83. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Chen, T. DNA Methylation Reprogramming during Mammalian Development. Genes (Basel) 2019, 10, 257. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.Y.; Wan, C.W.T.; Law, T.Y.S.; Chan, D.Y.L.; Fok, E.K.L. The Sperm Small RNA Transcriptome: Implications beyond Reproductive Disorder. Int J Mol Sci 2022, 23, 15716. [Google Scholar] [CrossRef]
- Agarwal, A.; Maldonado Rosas, I.; Anagnostopoulou, C.; Cannarella, R.; Boitrelle, F.; Munoz, L.V.; Finelli, R.; Durairajanayagam, D.; Henkel, R.; Saleh, R. Oxidative Stress and Assisted Reproduction: A Comprehensive Review of Its Pathophysiological Role and Strategies for Optimizing Embryo Culture Environment. Antioxidants (Basel) 2022, 11, 477. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wu, J.; Kim, S.-Y.; Zhao, M.; Hearn, S.A.; Zhang, M.Q.; Meistrich, M.L.; Mills, A.A. Chd5 Orchestrates Chromatin Remodelling during Sperm Development. Nat Commun 2014, 5, 3812. [Google Scholar] [CrossRef]
- Jin, J.; Li, K.; Du, Y.; Gao, F.; Wang, Z.; Li, W. Multi-Omics Study Identifies That PICK1 Deficiency Causes Male Infertility by Inhibiting Vesicle Trafficking in Sertoli Cells. Reprod Biol Endocrinol 2023, 21, 114. [Google Scholar] [CrossRef]
- Wang, C.; Swerdloff, R.S. Limitations of Semen Analysis as a Test of Male Fertility and Anticipated Needs from Newer Tests. Fertil Steril 2014, 102, 1502–1507. [Google Scholar] [CrossRef]
- Giuliano, R.; Maione, A.; Vallefuoco, A.; Sorrentino, U.; Zuccarello, D. Preimplantation Genetic Testing for Genetic Diseases: Limits and Review of Current Literature. Genes (Basel) 2023, 14, 2095. [Google Scholar] [CrossRef]
- Del Arco de la Paz, A.; Giménez-Rodríguez, C.; Selntigia, A.; Meseguer, M.; Galliano, D. Advancements and Challenges in Preimplantation Genetic Testing for Aneuploidies: In the Pathway to Non-Invasive Techniques. Genes (Basel) 2024, 15, 1613. [Google Scholar] [CrossRef]
- De Die-Smulders, C.; Van Golde, R. Genetic Counseling in Assisted Reproductive Treatment. In Textbook of Human Reproductive Genetics; Viville, S., Sermon, K.D., Eds.; Cambridge University Press, 2023; pp. 155–168. ISBN 978-1-009-19770-0. [Google Scholar]
- Muthuswamy, V. Ethical Issues in Genetic Counselling with Special Reference to Haemoglobinopathies. Indian J Med Res 2011, 134, 547–551. [Google Scholar]
- Lepine, S.; McDowell, S.; Searle, L.M.; Kroon, B.; Glujovsky, D.; Yazdani, A. Advanced Sperm Selection Techniques for Assisted Reproduction. Cochrane Database Syst Rev 2019, 7, CD010461. [Google Scholar] [CrossRef]
- Pacheco, A.; Blanco, A.; Bronet, F.; Cruz, M.; García-Fernández, J.; García-Velasco, J.A. Magnetic-Activated Cell Sorting (MACS): A Useful Sperm-Selection Technique in Cases of High Levels of Sperm DNA Fragmentation. J Clin Med 2020, 9, 3976. [Google Scholar] [CrossRef] [PubMed]
- Skoracka, K.; Eder, P.; Łykowska-Szuber, L.; Dobrowolska, A.; Krela-Kaźmierczak, I. Diet and Nutritional Factors in Male (In)Fertility-Underestimated Factors. J Clin Med 2020, 9, 1400. [Google Scholar] [CrossRef] [PubMed]
- Hart, R.J.; Wijs, L.A. The Longer-Term Effects of IVF on Offspring from Childhood to Adolescence. Front Reprod Health 2022, 4, 1045762. [Google Scholar] [CrossRef] [PubMed]
- Guttinger, S. Trust in Science: CRISPR-Cas9 and the Ban on Human Germline Editing. Sci Eng Ethics 2018, 24, 1077–1096. [Google Scholar] [CrossRef]
- Ayanoğlu, F.B.; Elçin, A.E.; Elçin, Y.M. Bioethical Issues in Genome Editing by CRISPR-Cas9 Technology. Turk J Biol 2020, 44, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Koplin, J.J.; Johnston, M.; Webb, A.N.S.; Whittaker, A.; Mills, C. Ethics of Artificial Intelligence in Embryo Assessment: Mapping the Terrain. Hum Reprod 2025, 40, 179–185. [Google Scholar] [CrossRef]
- Rolfes, V.; Bittner, U.; Gerhards, H.; Krüssel, J.-S.; Fehm, T.; Ranisch, R.; Fangerau, H. Artificial Intelligence in Reproductive Medicine - An Ethical Perspective. Geburtshilfe Frauenheilkd 2023, 83, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Ishii, T. Human iPS Cell-Derived Germ Cells: Current Status and Clinical Potential. J Clin Med 2014, 3, 1064–1083. [Google Scholar] [CrossRef]
- Stewart, K.R.; Veselovska, L.; Kelsey, G. Establishment and Functions of DNA Methylation in the Germline. Epigenomics 2016, 8, 1399–1413. [Google Scholar] [CrossRef]
- Wang, H.-Q.; Wang, T.; Gao, F.; Ren, W.-Z. Application of CRISPR/Cas Technology in Spermatogenesis Research and Male Infertility Treatment. Genes (Basel) 2022, 13, 1000. [Google Scholar] [CrossRef]
- More, A.; Chimurkar, V.; Mahajan, S.; Dakre, S.; Anjankar, N.; More, D. The Integration of Artificial Intelligence in In Vitro Fertilization: A Comprehensive Narrative Review. J Pharm Bioallied Sci 2025, 17, S155–S158. [Google Scholar] [CrossRef]
- Schroeder, M.; Badini, G.; Sferruzzi-Perri, A.N.; Albrecht, C. The Consequences of Assisted Reproduction Technologies on the Offspring Health Throughout Life: A Placental Contribution. Front Cell Dev Biol 2022, 10, 906240. [Google Scholar] [CrossRef]
- Orovou, E.; Tzimourta, K.D.; Tzitiridou-Chatzopoulou, M.; Kakatosi, A.; Sarantaki, A. Artificial Intelligence in Assisted Reproductive Technology: A New Era in Fertility Treatment. Cureus 2025, 17, e81568. [Google Scholar] [CrossRef]
- Dada, R.; Kumar, M.; Jesudasan, R.; Fernández, J.L.; Gosálvez, J.; Agarwal, A. Epigenetics and Its Role in Male Infertility. J Assist Reprod Genet 2012, 29, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Alukal, J.P.; Lamb, D.J. Intracytoplasmic Sperm Injection (ICSI)--What Are the Risks? Urol Clin North Am 2008, 35, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Sinha, A.; Singh, V.; Yadav, S. Multi-Omics and Male Infertility: Status, Integration and Future Prospects. Front Biosci (Schol Ed) 2017, 9, 375–394. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).