Submitted:
17 November 2025
Posted:
18 November 2025
You are already at the latest version
Abstract
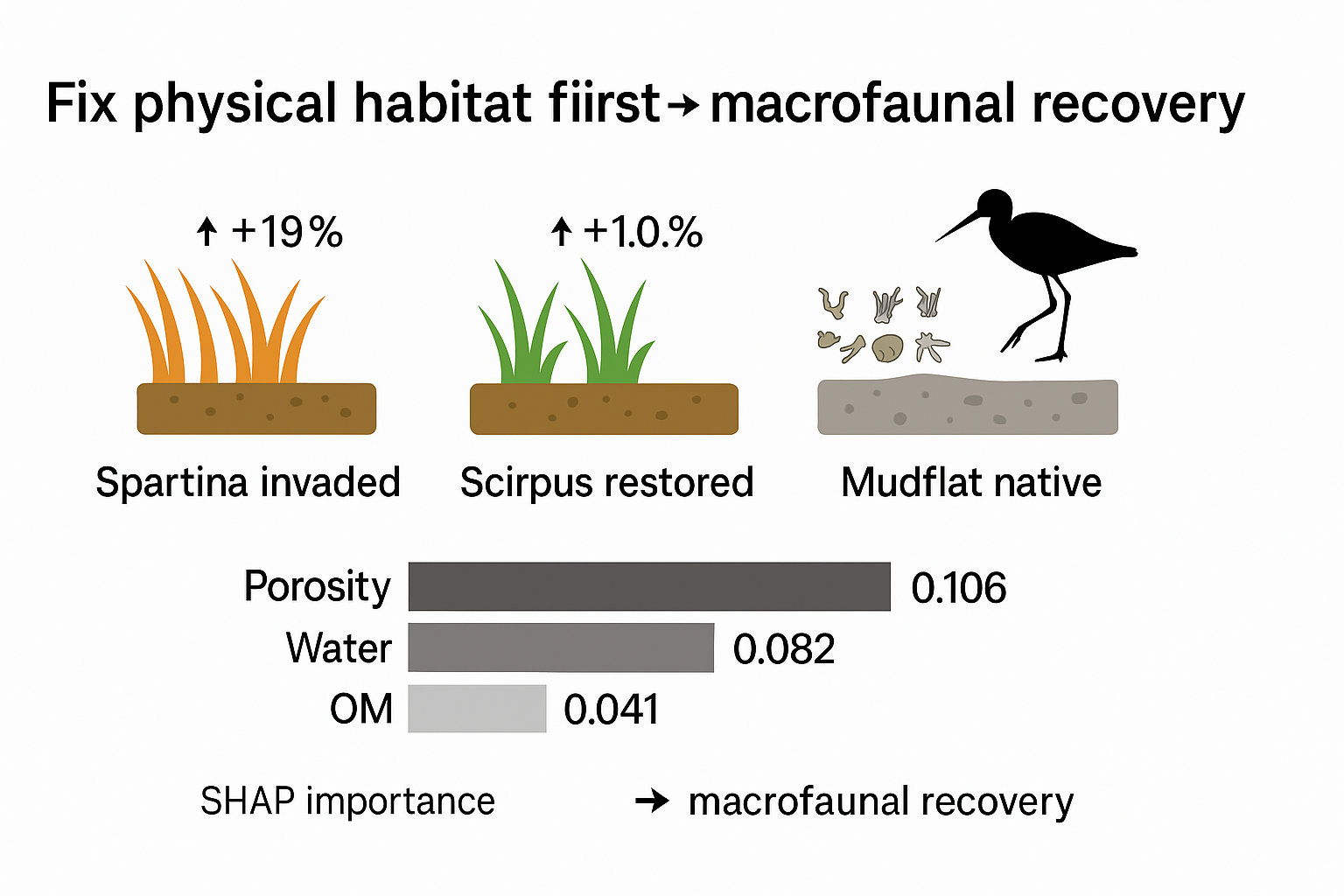
Restoring an ecosystem after a biological invasion is a huge challenge. A key question for ecologists is whether it can truly bring back what was lost. In coastal areas, the invasive cordgrass Spartina alterniflora is a major problem. It takes over tidal flats, changing the very structure of the sediment and devastating the small creatures—the macrobenthos—that live within it. This, in turn, disrupts the food supply for migrating waterbirds that depend on these flats. While digging up the invasive grass is a common solution, we still don't fully understand what drives the recovery. Does the habitat bounce back because the physical environment is fixed, or is it because the food sources (nutrients) return? To figure this out, we ran a field study in Hangzhou Bay, an area known for its large tides. We compared three different zones: patches of the invasive Spartina, mudflats where native Scirpus mariqueter sedge was restored after clearing the invader, and untouched native mudflats. We simultaneously tracked changes in the sediment's physical makeup (like porosity, water content, and grain size), its nutritional content (organic matter), and the community of bottom-dwelling creatures. Using a combination of Gradient Boosting Decision Trees (XGBoost) and SHAP analysis to sort through the data, we found that removing the invasive grass worked. The median number of species jumped from 5 to 9, and the total biomass shot up from a meager 1.36 to 11.75 g/m².Interestingly, this comeback was directly tied to improvements in the sediment's physical condition. After removal, total porosity increased by 19%, water content went up by nearly 11%, and the fraction of coarser particles grew by over 41%. The SHAP analysis confirmed what we saw: total porosity and water content were the most important factors predicting recovery (SHAP importance: 0.106 and 0.082). Their influence was more than double that of organic matter (0.041).What this tells us is that, at least in Hangzhou Bay, fixing the physical structure of the habitat is the first and most critical step. Rebuilding the sediment foundation kickstarts the return of the native benthic community, more so than any immediate change in nutrient levels. Our work shows that successful coastal restoration hinges on getting the physical substrate right first, giving managers a clearer target for assessing and managing invaded wetlands.
Keywords:
1. Introduction
2. Materials and Methods
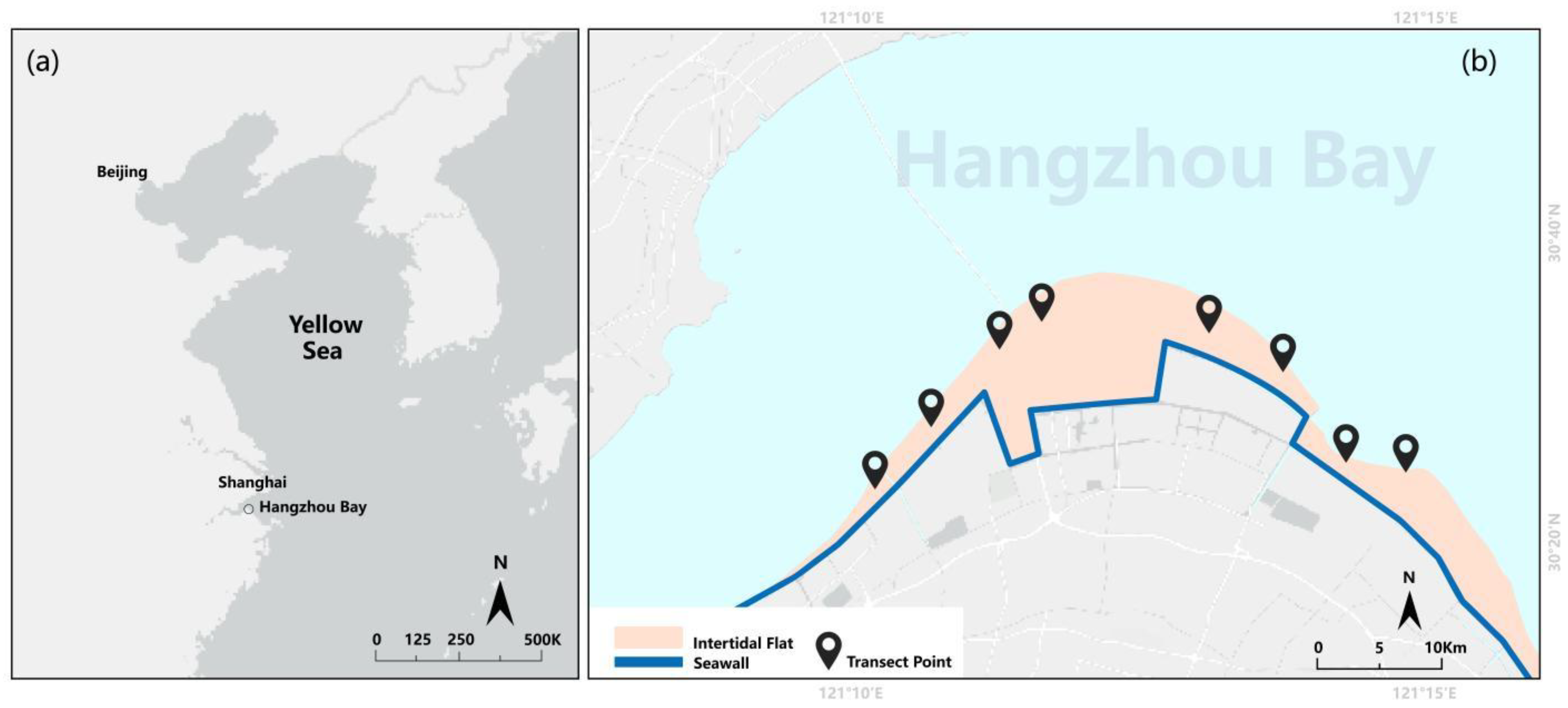
2.1. Study Area and Experimental Design
2.2. Sediment Collection and Laboratory Analysis
2.3. Macrobenthic Fauna Sampling and Biomass Determination
2.4. Statistical Analysis
3. Results
3.1. Variation in Sediment Physicochemical Properties Among Habitats
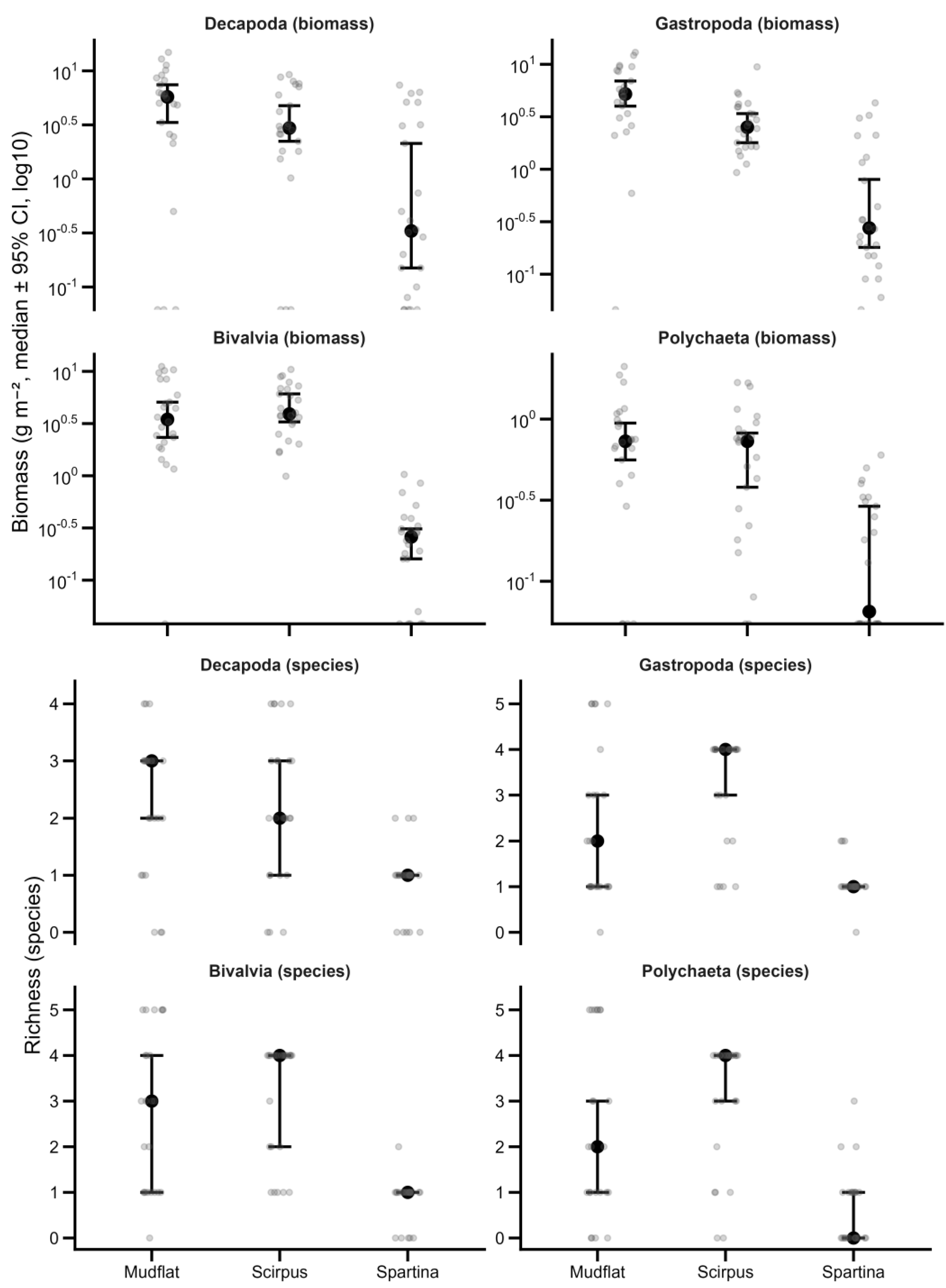
3.2. Variation in Benthic Community Richness and Biomass Across Habitats
3.3. Key Sediment Properties
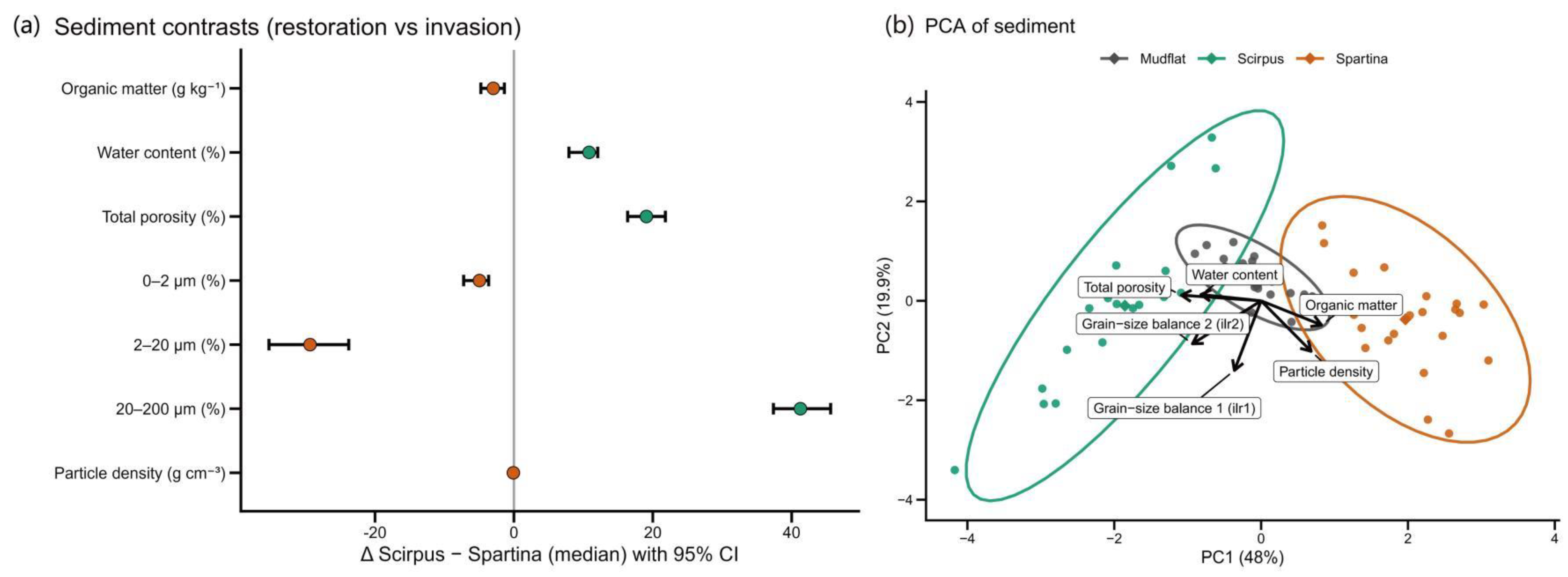
3.4. Comparison of Sediment Properties and Principal Component Structure
- 1)
- Contrasting Scirpus and Spartina Habitats
- 2)
- Environmental Gradients Separating the Three Habitats
3.5. Benthic Community: Median Distribution by Habitat and Management-Induced Gains (Δ Scirpus − Spartina)
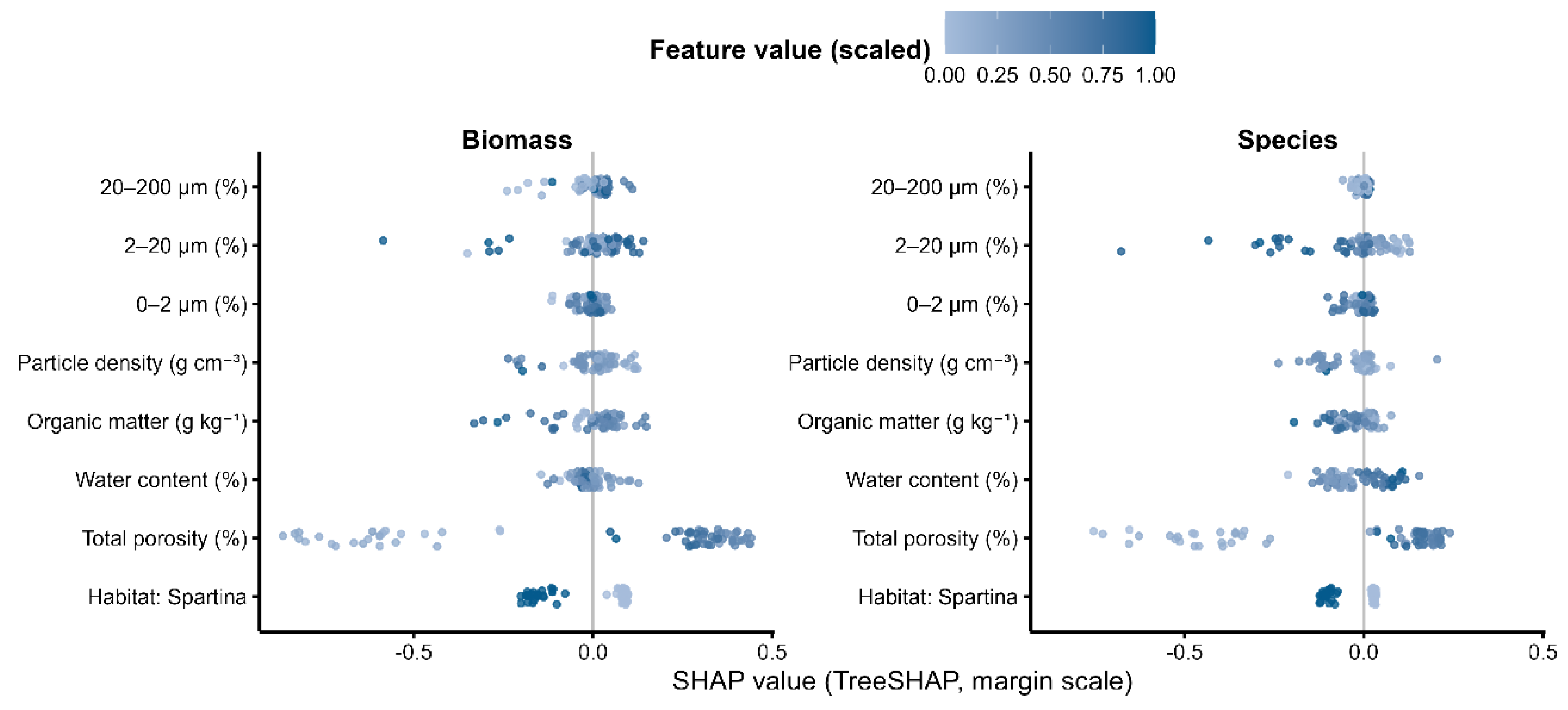
3.6. Key Drivers of Benthic Recovery (XGBoost–SHAP)
4. Discussion
4.1. Fundamental Shifts in Habitat Physical Structure
4.2. Differential Response Mechanisms of Macrobenthic Communities
4.3. Understanding the Ecological Process of Invasion and Recovery from a Physical Structure Perspective
5. Management Implications and Research Limitations
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Murray, N. J.; Worthington, T. A.; Bunting, P.; Duce, S.; Hagger, V.; Lovelock, C. E.; Lyons, M. B. The global distribution and trajectory of tidal flats. Nature 2019, 565, 222–225. [Google Scholar] [CrossRef] [PubMed]
- Murray, N. J.; Worthington, T. A.; Bunting, P.; Duce, S.; Hagger, V.; Lovelock, C. E.; Lyons, M. B. High-resolution mapping of losses and gains of Earth’s tidal wetlands. Science 2022, 376, 744–749. [Google Scholar] [CrossRef] [PubMed]
- Studds, C. E.; Kendall, B. E.; Murray, N. J.; Wilson, H. B.; Rogers, D. I.; Clemens, R. S.; Fuller, R. A. Rapid population decline in migratory shorebirds relying on Yellow Sea tidal mudflats as stopover sites. Nat. Commun. 2017, 8, 14895. [Google Scholar] [CrossRef]
- Cai, S.; Mu, T.; Peng, H.-B.; Ma, Z.; Wilcove, D. S. Importance of habitat heterogeneity in tidal flats to the conservation of migratory shorebirds. Conserv. Biol. 2024, 38, e14153. [Google Scholar] [CrossRef]
- Piersma, T. Production by intertidal benthic animals and limits to their predation by shorebirds: A heuristic model. Mar. Ecol. Prog. Ser. 1987, 38, 187–196. [Google Scholar] [CrossRef]
- Piersma, T.; Beukema, J. J. Population dynamics of benthic species on tidal flats: The possible roles of shorebird predation. In Tidal Flat Ecology; Reise, K., Ed.; Springer: Berlin/Heidelberg, Germany, 1993; pp. 197–209. [Google Scholar]
- Simberloff, D.; Martin, J.-L.; Genovesi, P.; Maris, V.; Wardle, D. A.; Aronson, J.; Courchamp, F.; Galil, B.; García-Berthou, E.; Pascal, M.; Pyšek, P.; Sousa, R.; Tabacchi, E.; Vilà, M. Impacts of biological invasions: what’s what and the way forward. Trends Ecol. Evol. 2013, 28, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Javed, Z.; Liu, B.; Zhong, S.; Cheng, Z.; Rehman, A.; Li, J. Impact of Spartina alterniflora invasion in coastal wetlands of China: Boon or bane? Biology 2023, 12, 1057. [Google Scholar] [CrossRef]
- Strong, D. R.; Ayres, D. R. Ecological and evolutionary misadventures of Spartina. Annu. Rev. Ecol. Evol. Syst. 2013, 44, 389–410. [Google Scholar] [CrossRef]
- Li, Y.; Hua, J.; He, C.; Wang, D.; Zhao, Z.; Wang, F.; Liu, X. Spartina alterniflora raised sediment sulfide in a tidal environment and buffered it with iron in the Jiuduansha wetland. J. Soils Sediments 2024, 24, 657–669. [Google Scholar] [CrossRef]
- Chen, Q.; Yu, Z.; Wang, M. Effects of Spartina alterniflora invasion on macrobenthic faunal community in different habitats of a mangrove wetland in Zhanjiang, South China. Reg. Stud. Mar. Sci. 2023, 66, 103148. [Google Scholar] [CrossRef]
- Lin, Y. Z.; Chen, Q. Q.; Qiu, Y. F.; Xie, R. R.; Zhang, H.; Zhang, Y.; Han, Y. H. Spartina alterniflora invasion altered phosphorus retention and microbial phosphate solubilization of the Minjiang estuary wetland in southeastern China. J. Environ. Manag. 2024, 358, 120817. [Google Scholar] [CrossRef]
- Xie, B.; Han, G.; Qiao, P.; Mei, B.; Wang, Q.; Zhou, Y.; Guan, B. Effects of mechanical and chemical control on invasive Spartina alterniflora in the Yellow River Delta, China. PeerJ 2019, 7, e7655. [Google Scholar] [CrossRef]
- Nie, M.; Liu, W.; Pennings, S. C.; Li, B. Lessons from the invasion of Spartina alterniflora in coastal China. Ecology 2023, 104, e3874. [Google Scholar] [CrossRef]
- APHA; AWWA; WEF. Standard Methods for the Examination of Water and Wastewater; Method 2540 (Solids), 23rd ed.; APHA: Washington, DC, USA, 2017. [Google Scholar]
- Heiri, O.; Lotter, A. F.; Lemcke, G. Loss on ignition as a method for estimating organic and carbonate content of lake sediments. J. Paleolimnol. 2001, 25, 101–110. [Google Scholar] [CrossRef]
- ISO. ISO 13320:2020—Particle Size Analysis: Laser Diffraction Methods; ISO: Geneva, Switzerland, 2020. [Google Scholar]
- ASTM International. ASTM D854—Standard Test Methods for Specific Gravity of Soil Solids by Water Pycnometer; ASTM International: West Conshohocken, PA, USA, 2014 (or latest).
- USGS. Techniques of Water-Resources Investigations (TWRI), Book 9—AFDM/biomass procedures; U.S. Geological Survey: Reston, VA, USA.
- Efron, B.; Tibshirani, R. J. An Introduction to the Bootstrap; Chapman & Hall/CRC: New York, NY, USA, 1993. [Google Scholar] [CrossRef]
- Pearson, K. LIII. On lines and planes of closest fit to systems of points in space. Philos. Mag. 1901, 2, 559–572. [Google Scholar] [CrossRef]
- Egozcue, J. J.; Pawlowsky-Glahn, V.; Mateu-Figueras, G.; Barceló-Vidal, C. Isometric logratio transformations for compositional data analysis. Math. Geol. 2003, 35, 279–300. [Google Scholar] [CrossRef]
- Anderson, M. J. A new method for non-parametric multivariate analysis of variance. Austral Ecol. 2001, 26, 32–46. [Google Scholar] [CrossRef]
- Chen, T.; Guestrin, C. XGBoost: A scalable tree boosting system. In Proceedings of the 22nd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining (KDD ’16); ACM: New York, NY, USA, 2016; pp. 785–794. [Google Scholar] [CrossRef]
- Lundberg, S. M.; Lee, S.-I. A unified approach to interpreting model predictions. In Advances in Neural Information Processing Systems 30 (NeurIPS 2017); Curran Associates: Red Hook, NY, USA, 2017. [Google Scholar]
- Lundberg, S. M.; Erion, G.; Chen, H.; DeGrave, A.; Prutkin, J. M.; Nair, B.; Katz, R.; Himmelfarb, J.; Bansal, N.; Lee, S.-I. From local explanations to global understanding with explainable AI for trees. Nat. Mach. Intell. 2020, 2, 56–67. [Google Scholar] [CrossRef] [PubMed]
- Runion, K. D.; Alber, M.; Mishra, D. R.; Lever, M. A.; Hladik, C. M.; O’Connell, J. L. Early warning signs of salt marsh drowning indicated by widespread vulnerability from declining belowground plant biomass. Proc. Natl. Acad. Sci. USA 2025, 122, e2425501122. [Google Scholar] [CrossRef]
- Donatelli, C.; Zhang, X.; Ganju, N. K.; Aretxabaleta, A. L.; Fagherazzi, S.; Leonardi, N. Marsh size has a nonlinear effect on sediment fluxes and accretion. Geology 2020, 48, 966–970. [Google Scholar] [CrossRef]
- Ganju, N. K.; Brosnahan, S. M.; Gibeaut, J.; Hagen, S. C.; Leonardi, N.; Nowacki, D. J.; Santos, F.; Sherwood, C. R.; Alin, S. R. Linking tidal-creek sediment fluxes to vertical sediment accretion in a restored salt marsh. Earth Surf. Process. Landf. 2025, 50, e70053. [Google Scholar] [CrossRef]
- Smith, A. J.; Guntenspergen, G. R.; Carr, J. A.; Walters, D. C.; Kirwan, M. L. Microtopographic variation offers an early warning indicator of salt marsh vulnerability. Estuaries Coasts 2024, 47, 2120–2134. [Google Scholar] [CrossRef]
- Wang, D.; Labra, F. A.; Yang, H.; Hu, Y.; Zhao, Z.; Zhou, W.; Yuan, L. Restoration of native saltmarshes enhances carbon sequestration and mitigates warming effects following Spartina alterniflora removal. J. Appl. Ecol. 2025, Early View, article pending. [CrossRef]
- Rilov, G.; Canning-Clode, J.; Guy-Haim, T. Ecological impacts of invasive ecosystem engineers: A global perspective across terrestrial and aquatic systems. Funct. Ecol. 2023, 37, 1918–1933. [Google Scholar] [CrossRef]
- Sevilgen, D. S.; Mehmetoglu, H.; Suthhof, A.; Severyn, J. P.; Maerz, J. C.; LeRoy Poff, N.; Nilsson, C.; Lake, P. S.; Nies, T. Planning hydrological restoration in wetlands: A model-based approach to orchestrate water flow and habitat functions. Sci. Total Environ. 2024, 927, 171620. [Google Scholar] [CrossRef]
- Du, H.; Yu, J.; Wang, J.; Fu, X.; Liu, G.; Zhang, X. Plant- and microbial-mediated soil organic carbon accumulation in coastal wetlands: Mechanisms, processes and ecological restoration. Catena 2023, 228, 107082. [Google Scholar] [CrossRef]
- Cook, P. L. M.; Eyre, B. D.; Huettel, M.; Glud, R. N.; Rao, A. M. F.; Kana, T. M.; Middelburg, J. J.; Kristensen, E. Macrofaunal control on benthic biogeochemistry in permeable sands. Mar. Ecol. Prog. Ser. 2018, 596, 1–18. [Google Scholar] [CrossRef]
- Jiang, Y.; Shao, Y.; Huang, J.; Du, Y.; Wen, Y.; Tang, H.; Xu, J.; Gao, D.; Lin, X.; Sun, D. Spartina alterniflora invasion enhances greenhouse gas emissions via altered soil physicochemistry and microbial networks. Front. Microbiol. 2024, 15, 1420924. [Google Scholar] [CrossRef]
- Liang, W.; Chen, X.; Chen, Z.-L.; Zhu, P.; Huang, Z. ;Unraveling the impact of Spartina alterniflora invasion on greenhouse gases: DOM chemistry and surface water–porewater interactions. Water Res. 2024, 266, 122120. [Google Scholar] [CrossRef]
- Lin, Y.-T. V.; Château, P.-A.; Nozawa, Y.; Wei, C.-L.; Wunderlich, R. F.; Denis, V. Drivers of coastal benthic communities in a complex environmental setting. Mar. Pollut. Bull. 2024, 203, 116462. [Google Scholar] [CrossRef] [PubMed]
- Shuai, P.; Ravelo, A. M.; Divine, L. M.; Jewett, S. C.; Iken, K. ; Environmental filtering influences functional community assembly of epibenthic communities. Front. Mar. Sci. 2021, 8, 736917. [Google Scholar] [CrossRef]
- Song, J.; Farhadi, A.; Tan, K.; Lim, L.; Tan, K. Impact of anthropogenic global hypoxia on the physiological response of bivalves. Sci. Total Environ. 2024, 927, 172056. [Google Scholar] [CrossRef]
- Fang, J.; Meng, S.; Escobar Lux, R. H.; Jiang, W.; Jiang, Z.; Mao, Y.; Jansen, H.; Fang, J.; Strand, Ø. Why and how is burrow ventilation initiated? A case study of polychaete behavior at different temperatures. Front. Mar. Sci. 2022, 9, 910781. [Google Scholar] [CrossRef]
- Pearson, T. H.; Rosenberg, R. Macrobenthic succession in relation to organic enrichment and pollution of the marine environment. Oceanogr. Mar. Biol. Annu. Rev. 1978, 16, 229–311. [Google Scholar]
- Zhang, L.; Zhang, D.; Su, X.; Yue, S.; Xie, Z. ; Effects of burrow architecture and ventilation on biogeochemical exchange in cohesive sediments. Front. Mar. Sci. 2022, 9, 911366. [Google Scholar] [CrossRef]
- Liu, Z.; Fagherazzi, S.; Cui, B. Success of coastal wetlands restoration is driven by sediment availability. Commun. Earth Environ. 2021, 2, 74. [Google Scholar] [CrossRef]
- Jia, X.; Yang, J.; Wang, C.; Liu, B.; Zheng, H.; Zou, Y.; Wang, H.; Zhao, H. Predicting the regime shift and bistable ecological states in coastal wetlands under hurricane impacts. Front. Mar. Sci. 2023, 10, 1126682. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, H.; Zhan, L.; Lao, C.; Xin, P. Salt marsh morphological evolution under plant species invasion. Estuaries Coasts 2024, 47, 949–962. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhou, Y.; Liu, H.; Li, Y.; Wang, C.; Wright, A. Coastal wetland restoration framework based on hydrogeomorphic units of the coastal wetland of Yancheng, China. Ocean Coast. Manag. 2023, 243, 106737. [Google Scholar] [CrossRef]
- Rilov, G.; Canning-Clode, J.; Guy-Haim, T. Ecological impacts of invasive ecosystem engineers: A global perspective across terrestrial and aquatic systems. Funct. Ecol. 2023, 37, 1918–1933. [Google Scholar] [CrossRef]
- Peller, T.; Côté, I. M. Invasive species drive cross-ecosystem effects worldwide. Nat. Ecol. Evol. 2024, 8, 1087–1097. [Google Scholar] [CrossRef]
- Li, F.; Ma, Y.; Song, X.; Li, S.; Zhang, X.; Wang, X.; Wang, T.; Sun, Z. Community structure and ecological quality assessment of macrobenthos in the coastal sea areas of Northern Yantai, China. Front. Mar. Sci. 2022, 9, 989034. [Google Scholar] [CrossRef]
- Tian, J.; Zhang, W.; Wu, J.; Chen, Q.; Huang, J. Effects of hypoxia on community structure of macrobenthos in the Zhujiang River Estuary. Acta Oceanol. Sin. 2025, 44, 14–23. [Google Scholar] [CrossRef]
- Dong, J.; Zhao, L.; Yang, X.; Sun, X.; Zhang, X. Functional trait responses of macrobenthos to anthropogenic pressure in three temperate intertidal communities. Front. Mar. Sci. 2021, 8, 756814. [Google Scholar] [CrossRef]
- Wang, X.; Xu, P.; Yang, B.; Zheng, L.; Xu, B.; Ge, S.; Wang, B.; Liu, C.; Meng, Q.; Xiong, X. ; Numerical models of bio-geomorphological processes in tidal marshes: Applications and challenges. Int. J. Sediment Res. 2024, 39, 451–472. [Google Scholar] [CrossRef]
- Sevilgen, D. S.; Mehmetoglu, H.; Suthhof, A.; Severyn, J. P.; Maerz, J. C.; LeRoy Poff, N.; Nilsson, C.; Lake, P. S.; Nies, T. Planning hydrological restoration in wetlands: A model-based approach to orchestrate water flow and habitat functions. Sci. Total Environ. 2024, 927, 171620. [Google Scholar] [CrossRef]
| Variable | Spartina | Scirpus | Mudflat | F | p | η² |
| Total porosity (%) | 42.53 ± 2.28ᶜ | 62.21 ± 3.85ᵇ | 54.26 ± 2.16ᵃ | 285.39 | <0.001 | 0.892 |
| Water content (%) | 25.67 ± 2.22ᵃ | 35.30 ± 2.63ᵇ | 26.32 ± 4.92ᵃ | 57.92 | <0.001 | 0.627 |
| Organic matter (g kg⁻¹) | 10.38 ± 1.83ᵇ | 7.27 ± 1.95ᵃ | 7.15 ± 1.05ᵃ | 29.36 | <0.001 | 0.460 |
| Particle density (g cm⁻³) | 2.76 ± 0.09ᵇ | 2.66 ± 0.05ᵃ | 2.65 ± 0.05ᵃ | 22.23 | <0.001 | 0.392 |
| 0–2 μm (%) | 8.41 ± 2.16ᶜ | 2.96 ± 2.05ᵇ | 6.95 ± 0.84ᵃ | 59.82 | <0.001 | 0.634 |
| 2–20 μm (%) | 56.23 ± 11.14ᵃ | 29.10 ± 5.31ᵇ | 55.08 ± 6.11ᵃ | 89.44 | <0.001 | 0.722 |
| 20–200 μm (%) | 31.39 ± 13.74ᵃ | 67.52 ± 6.79ᵇ | 37.67 ± 6.44ᵃ | 97.10 | <0.001 | 0.738 |
| Variable | Spartina | Scirpus | Mudflat | F | p | η² |
| Decapoda (species) | 1.62 ± 1.21ᵇ | 2.62 ± 1.71ᵇ | 4.58 ± 2.21ᵃ | 17.60 | <0.001 | 0.338 |
| Gastropoda (species) | 1.00 ± 1.10ᵇ | 1.79 ± 1.35ᵃ | 2.46 ± 1.59ᵃ | 6.90 | 0.0019 | 0.167 |
| Bivalvia (species) | 0.96 ± 1.00ᵇ | 2.17 ± 1.20ᵃ | 2.00 ± 1.50ᵃ | 6.56 | 0.0025 | 0.160 |
| Polychaeta (species) | 0.83 ± 0.92ᵇ | 1.71 ± 1.30ᵇ | 3.54 ± 2.25ᵃ | 18.15 | <0.001 | 0.345 |
| Total species | 4.42 ± 2.10ᶜ | 8.29 ± 2.46ᵇ | 12.58 ± 5.02ᵃ | 33.68 | <0.001 | 0.494 |
| Decapoda biomass (g m⁻²) | 0.49 ± 0.70ᶜ | 4.25 ± 3.24ᵇ | 2.11 ± 1.49ᵃ | 19.37 | <0.001 | 0.360 |
| Gastropoda biomass (g m⁻²) | 0.61 ± 0.89ᶜ | 4.98 ± 2.94ᵇ | 2.43 ± 1.58ᵃ | 29.14 | <0.001 | 0.458 |
| Bivalvia biomass (g m⁻²) | 0.24 ± 0.22ᶜ | 5.77 ± 3.19ᵇ | 3.39 ± 2.44ᵃ | 34.30 | <0.001 | 0.499 |
| Polychaeta biomass (g m⁻²) | 0.15 ± 0.18ᵇ | 6.96 ± 5.58ᵃ | 4.95 ± 4.14ᵃ | 18.24 | <0.001 | 0.346 |
| Total biomass (g m⁻²) | 1.49 ± 1.21ᶜ | 21.97 ± 7.99ᵇ | 12.89 ± 5.35ᵃ | 80.59 | <0.001 | 0.700 |
| Variable | Spartina (median) | Scirpus (median) | Mudflat (median) | Δ Scirpus–Spartina (95% CI) |
| Organic matter (g kg^-1) | 10.70 | 7.73 | 6.83 | −2.96 (−4.24–−1.77) |
| Water content (%) | 24.80 | 35.62 | 27.64 | 10.82 (9.24–12.15) |
| Total porosity (%) | 42.48 | 61.56 | 54.98 | 19.08 (17.59–20.46) |
| 0–2 μm (%) | 8.41 | 4.49 | 6.73 | −3.92 (−5.24–−2.82) |
| 2–20 μm (%) | 60.53 | 40.63 | 54.89 | −19.90 (−23.37–−16.35) |
| 20–200 μm (%) | 25.56 | 66.82 | 38.16 | 41.26 (37.01–45.50) |
| Particle density (g cm^-3) | 2.75 | 2.67 | 2.65 | −0.08 (−0.10–−0.06) |
| Variable | Spartina (median) | Scirpus (median) | Mudflat (median) | Δ Scirpus–Spartina |
| Richness (species) | ||||
| Decapoda (species) | 1 | 3 | 5 | +2 |
| Gastropoda (species) | 1 | 2 | 3 | +1 |
| Bivalvia (species) | 1 | 2 | 2 | +1 |
| Polychaeta (species) | 0 | 2 | 4 | +2 |
| Total species | 5 | 9 | 14 | +4 |
| Biomass (g m⁻²) | ||||
| Decapoda biomass | 0.33 | 2.96 | 5.74 | 2.62 |
| Gastropoda biomass | 0.28 | 2.51 | 5.21 | 2.24 |
| Bivalvia biomass | 0.26 | 3.9 | 3.46 | 3.64 |
| Polychaeta biomass | 0.07 | 0.73 | 0.73 | 0.67 |
| Total biomass | 1.36 | 11.75 | 16.43 | 10.38 |
| Variable | Biomass importance (SHAP ± SE) | Biomass rank | Biomass effect (SHAP ± SE) | Species importance (SHAP ± SE) | Species rank | Species effect (SHAP ± SE) |
| Total porosity (%) | 0.106 ± 0.013 | 1 | 0.102 ± 0.016 | 0.092 ± 0.014 | 1 | 0.091 ± 0.017 |
| Water content (%) | 0.082 ± 0.011 | 2 | 0.079 ± 0.014 | 0.084 ± 0.013 | 2 | 0.081 ± 0.015 |
| Organic matter (g kg⁻¹) | 0.041 ± 0.009 | 6 | 0.018 ± 0.011 | 0.049 ± 0.010 | 4 | 0.023 ± 0.012 |
| Particle density (g cm⁻³) | 0.036 ± 0.008 | 7 | −0.028 ± 0.010 | 0.040 ± 0.009 | 6 | −0.031 ± 0.011 |
| 0–2 μm (%) | 0.058 ± 0.011 | 4 | −0.019 ± 0.013 | 0.053 ± 0.011 | 5 | −0.016 ± 0.013 |
| 2–20 μm (%) | 0.047 ± 0.010 | 5 | −0.006 ± 0.010 | 0.061 ± 0.012 | 5 | −0.011 ± 0.014 |
| 20–200 μm (%) | 0.028 ± 0.007 | 8 | 0.007 ± 0.007 | 0.061 ± 0.012 | 5 | −0.011 ± 0.014 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




