Submitted:
17 November 2025
Posted:
18 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Literature Review
Berberine
Chlorogenic Acid
Curcumin
Epigallocatechin Gallate (EGCG)
Hesperidin
Quercetin
Rutin
Synthetic Drug Orforglipron
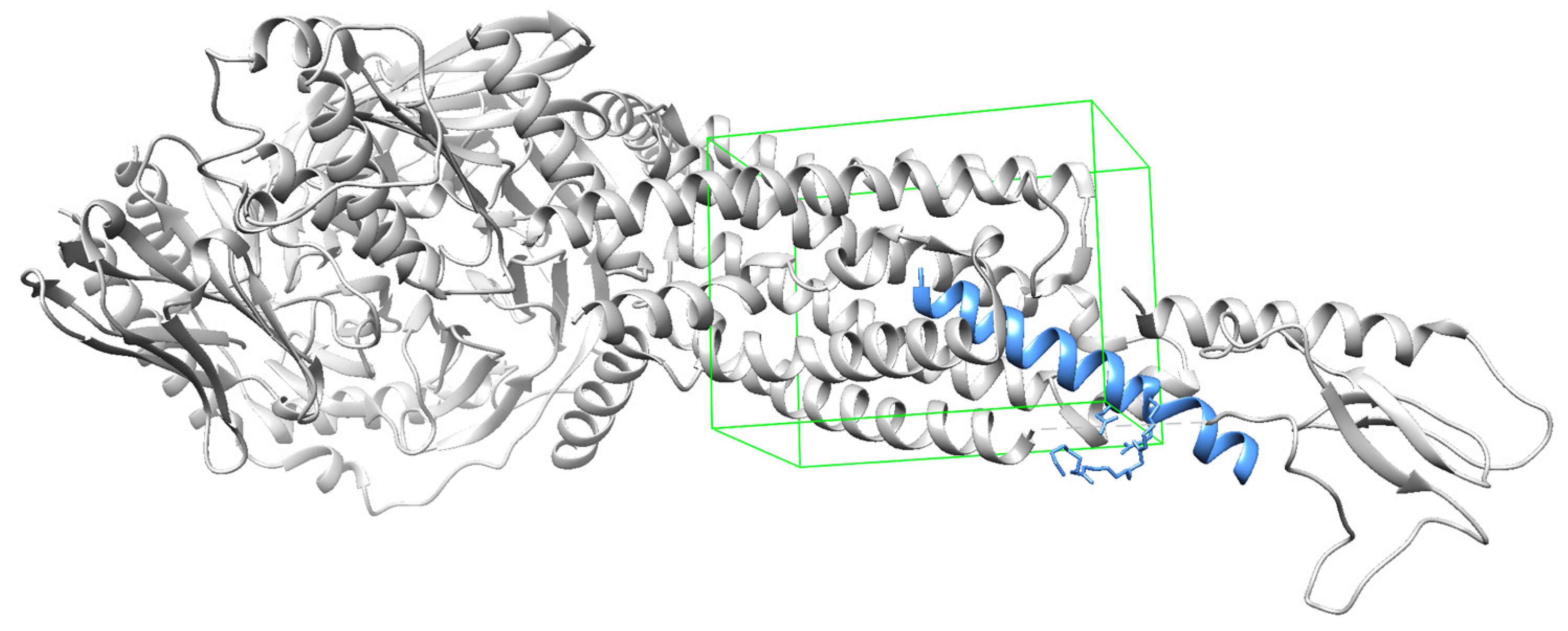
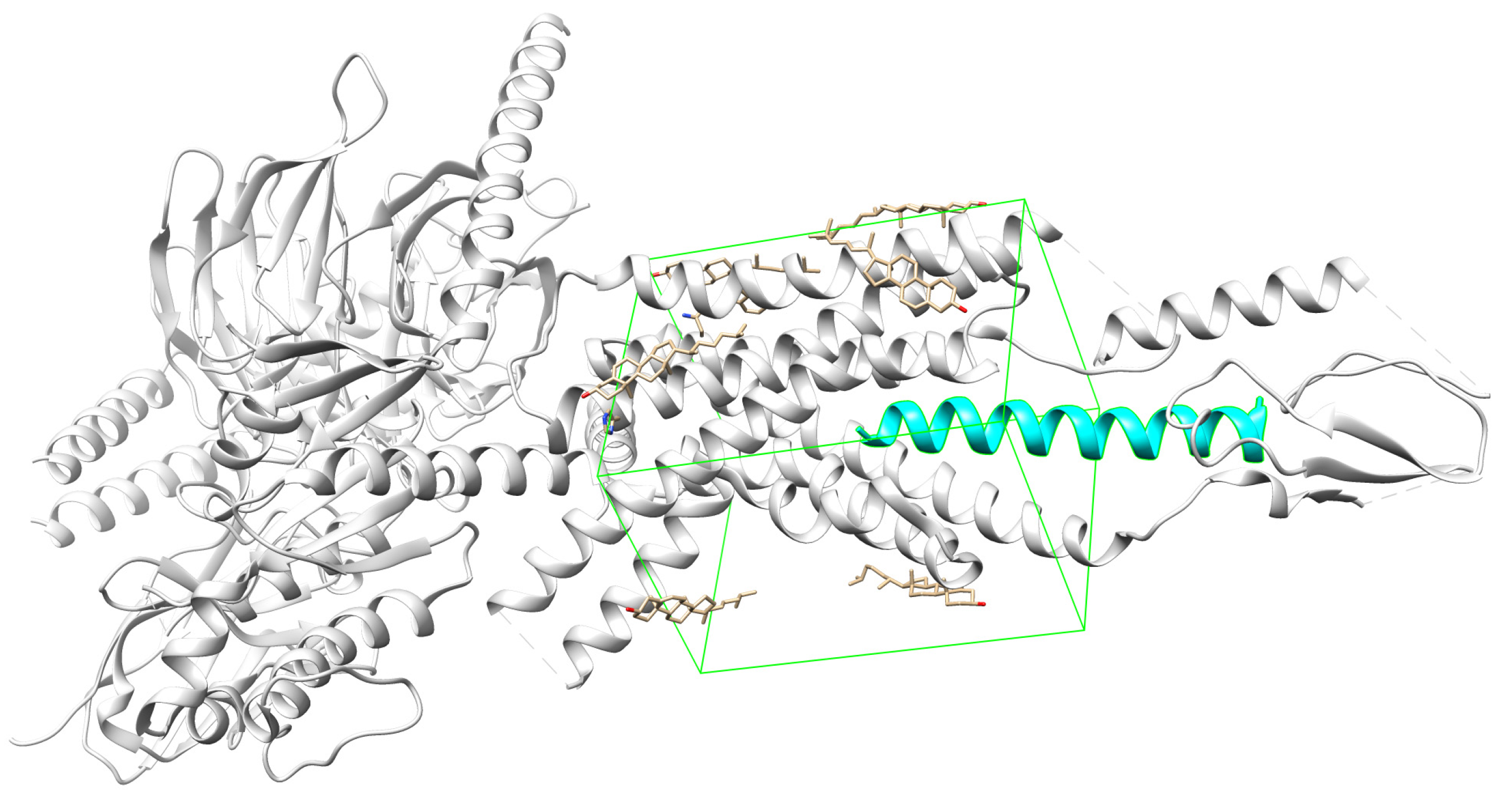
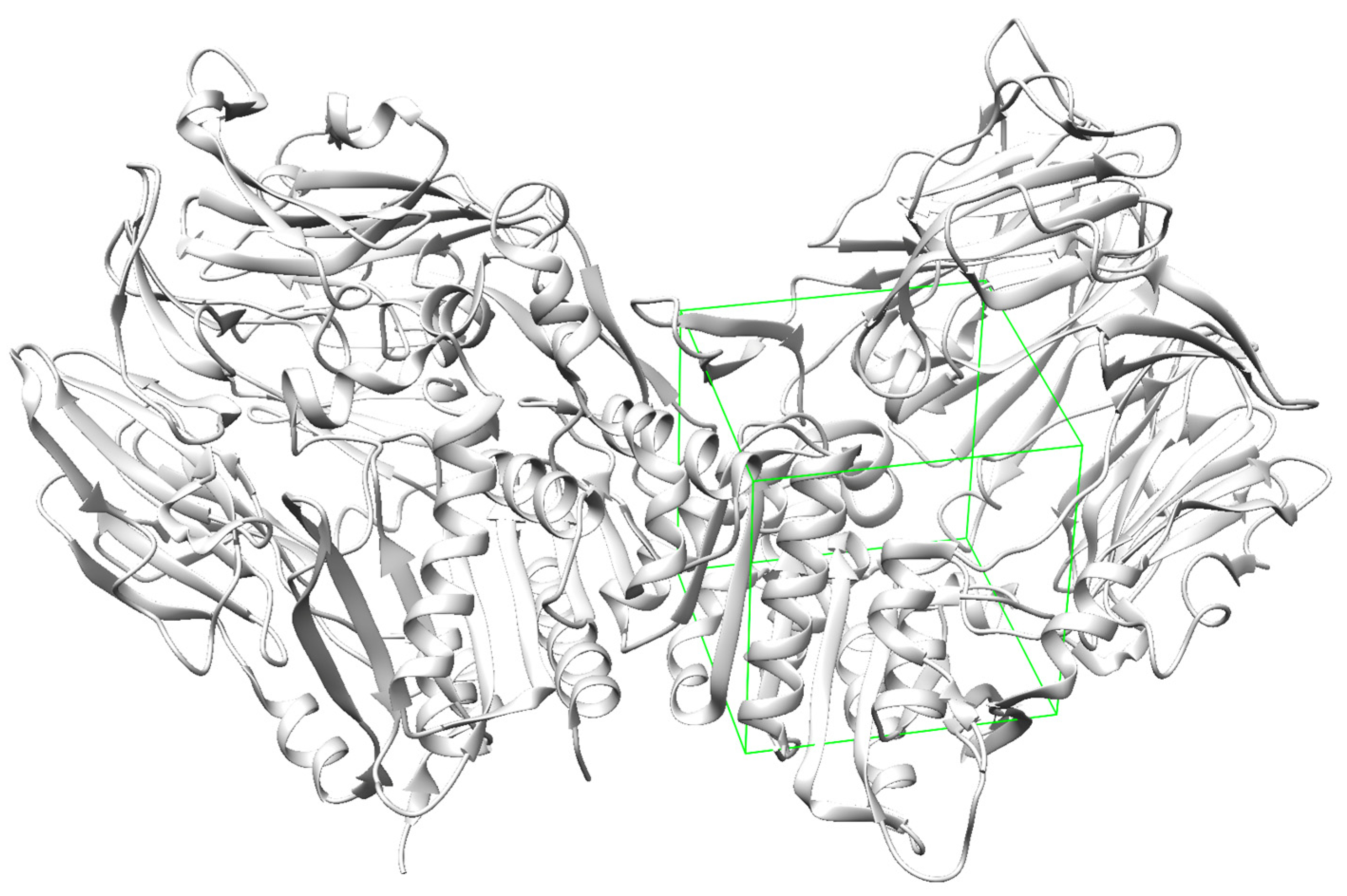
2.2. Molecular Docking
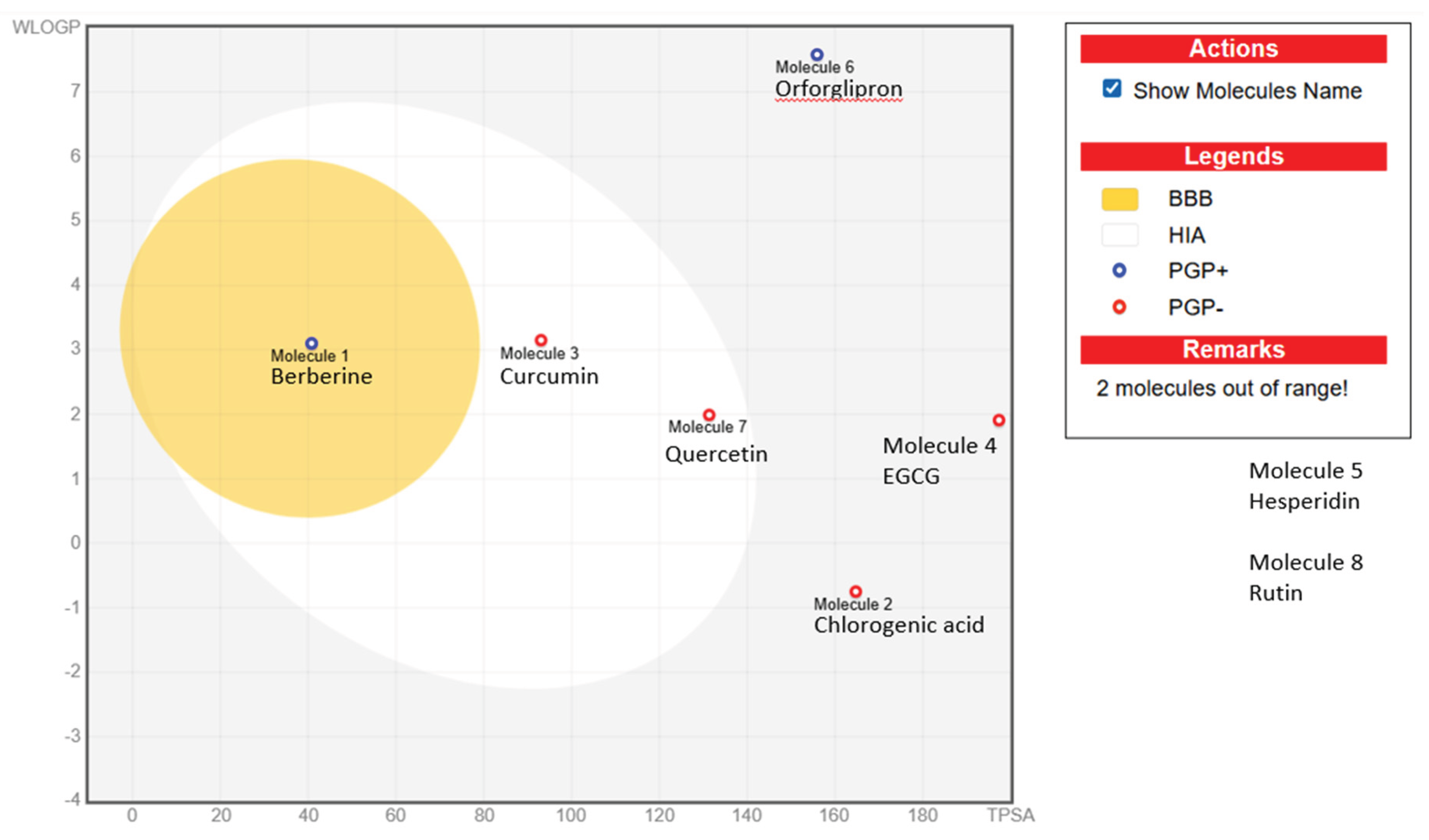
2.2. ADME Profiles
3. Discussion
4. Materials and Methods
Literature Review
Molecular Structures
Molecular Docking
ADME Profile
Artificial Intelligence (AI)
5. Conclusions
Supplementary Materials
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADME | Absorption, Distribution, Metabolism, and Excretion; the four key pharmacokinetic processes determining how a compound behaves in the body |
| AGI | Alpha-Glucosidase Inhibitor; a type of antidiabetic compound that slows carbohydrate digestion in the gut |
| AI | Artificial Intelligence |
| AMPK | Alpha-Glucosidase Inhibitor; a type of antidiabetic compound that slows carbohydrate digestion in the gut |
| BBB | Blood–Brain Barrier; a selective barrier between the bloodstream and brain tissue that restricts the passage of many molecules |
| BOILED-Egg model | Brain Or IntestinaL EstimateD permeation method; a visual predictor of gastrointestinal absorption and blood–brain barrier permeability used in SwissADME |
| CNS | Central Nervous System; includes the brain and spinal cord |
| CYP | Cytochrome P450; a family of liver enzymes responsible for metabolizing many drugs |
| DPP-4 | Dipeptidyl Peptidase-4; an enzyme that degrades incretin hormones such as GLP-1 and GIP |
| EC50 | Half Maximal Effective Concentration; the concentration of a compound that produces 50% of its maximal effect |
| EGCG | Epigallocatechin Gallate; a bioactive polyphenol found in green tea |
| GI | Gastro-Intestinal |
| GIP | Glucose-Dependent Insulinotropic Polypeptide; an incretin hormone that stimulates insulin release in response to food intake |
| GIPR | Glucose-Dependent Insulinotropic Polypeptide Receptor; the receptor that binds GIP |
| GLP-1 | Glucagon-Like Peptide-1; an incretin hormone involved in glucose metabolism and appetite regulation |
| GLP-1R | Glucagon-Like Peptide-1 Receptor; the receptor that mediates the effects of GLP-1 |
| HIA | Human Intestinal Absorption |
| Log P | Partition Coefficient; a measure of a compound’s lipophilicity or its tendency to dissolve in fats versus water |
| PGP+ | P-glycoprotein Substrate (Yes) |
| PGP− | P-glycoprotein Non-Substrate (No) |
| PK | Pharmacokinetics; the study of how a compound is absorbed, distributed, metabolized, and excreted by the body |
| SwissADME | Swiss Absorption, Distribution, Metabolism, and Excretion; a web tool for predicting pharmacokinetic and drug-likeness properties of small molecules |
| T2DM | Type 2 Diabetes Mellitus; a metabolic disorder characterized by insulin resistance and high blood glucose |
| TEER | Transepithelial Electrical Resistance; a measure of the integrity of cell layer barriers, such as intestinal or epithelial monolayers |
| TPSA | Topological Polar Surface Area |
| WLOGP | Wildman–Crippen LogP |
References
- World Health Organisation Obesity and overweight Available online:. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 13 November 2025).
- Zheng, Z.; Zong, Y.; Ma, Y.; Tian, Y.; Pang, Y.; Zhang, C.; Gao, J. Glucagon-like peptide-1 receptor: mechanisms and advances in therapy. Signal Transduct. Target. Ther. 2024, 9, 234. [Google Scholar] [CrossRef]
- Drucker, D.J. GLP-1-based therapies for diabetes, obesity and beyond. Nat. Rev. Drug Discov. 2025, 24, 631–650. [Google Scholar] [CrossRef]
- Shaikh, S.; Lee, E.-J.; Ahmad, K.; Ahmad, S.-S.; Lim, J.-H.; Choi, I. A Comprehensive Review and Perspective on Natural Sources as Dipeptidyl Peptidase-4 Inhibitors for Management of Diabetes. Pharmaceuticals 2021, 14, 591. [Google Scholar] [CrossRef] [PubMed]
- Sokary, S.; Bawadi, H. The promise of tirzepatide: A narrative review of metabolic benefits. Prim. Care Diabetes 2025, 19, 229–237. [Google Scholar] [CrossRef] [PubMed]
- Ansari, P.; Hannon-Fletcher, M.P.; Flatt, P.R.; Abdel-Wahab, Y.H.A. Effects of 22 traditional anti-diabetic medicinal plants on DPP-IV enzyme activity and glucose homeostasis in high-fat fed obese diabetic rats. Biosci. Rep. 2021, 41, BSR20203824. [Google Scholar] [CrossRef]
- Abiola, J.O.; Oluyemi, A.A.; Idowu, O.T.; Oyinloye, O.M.; Ubah, C.S.; Owolabi, O.V.; Somade, O.T.; Onikanni, S.A.; Ajiboye, B.O.; Osunsanmi, F.O.; et al. Potential Role of Phytochemicals as Glucagon-like Peptide 1 Receptor (GLP-1R) Agonists in the Treatment of Diabetes Mellitus. Pharmaceuticals 2024, 17, 736. [Google Scholar] [CrossRef]
- Chavan, A.; Daniel, K.; Patel, A.M. In-silico Exploration of Phytoconstituents of Gymnema sylvestre as PotentialGlucokinase Activators and DPP-IV Inhibitors for the Future Synthesisof Silver Nanoparticles for the Treatment of Type 2 Diabetes Mellitus. Curr. Enzyme Inhib. 2022, 18, 47–60. [Google Scholar] [CrossRef]
- Kong, Y.; Yang, H.; Nie, R.; Zhang, X.; Zhang, H.; Nian, X. Berberine as a multi-target therapeutic agent for obesity: from pharmacological mechanisms to clinical evidence. Eur. J. Med. Res. 2025, 30, 477. [Google Scholar] [CrossRef]
- Yanagimoto, A.; Matsui, Y.; Yamaguchi, T.; Hibi, M.; Kobayashi, S.; Osaki, N. Effects of Ingesting Both Catechins and Chlorogenic Acids on Glucose, Incretin, and Insulin Sensitivity in Healthy Men: A Randomized, Double-Blinded, Placebo-Controlled Crossover Trial. Nutrients 2022, 14, 5063. [Google Scholar] [CrossRef]
- Moetlediwa, M.T.; Ramashia, R.; Pheiffer, C.; Titinchi, S.J.J.; Mazibuko-Mbeje, S.E.; Jack, B.U. Therapeutic Effects of Curcumin Derivatives against Obesity and Associated Metabolic Complications: A Review of In Vitro and In Vivo Studies. Int. J. Mol. Sci. 2023, 24, 14366. [Google Scholar] [CrossRef]
- Mirzaei, A.; Mirzaei, A.; Najjar Khalilabad, S.; Askari, V.R.; Baradaran Rahimi, V. Promising influences of hesperidin and hesperetin against diabetes and its complications: a systematic review of molecular, cellular, and metabolic effects. EXCLI J. 22Doc1235 ISSN 1611-2156 2023. [Google Scholar] [CrossRef]
- Korkmaz, Y.; Dik, B. The comparison of the antidiabetic effects of exenatide, empagliflozin, quercetin, and combination of the drugs in type 2 diabetic rats. Fundam. Clin. Pharmacol. 2024, 38, 511–522. [Google Scholar] [CrossRef]
- Elekofehinti, O.O.; Molehin, O.R.; Akinjiyan, M.O.; Fakayode, A.E. Rutin modulates the TGR5/GLP1 pathway and downregulates proinflammatory cytokines genes in streptozotocin-induced diabetic rats. J. Food Bioact. 2024, 88–103. [Google Scholar] [CrossRef]
- Kavyani, Z.; Shahhosseini, E.; Moridpour, A.H.; Falahatzadeh, M.; Vajdi, M.; Musazadeh, V.; Askari, G. The effect of berberine supplementation on lipid profile and obesity indices: An umbrella review of meta-analysis. PharmaNutrition 2023, 26, 100364. [Google Scholar] [CrossRef]
- Askari, V.R.; Khosravi, K.; Baradaran Rahimi, V.; Garzoli, S. A Mechanistic Review on How Berberine Use Combats Diabetes and Related Complications: Molecular, Cellular, and Metabolic Effects. Pharmaceuticals 2023, 17, 7. [Google Scholar] [CrossRef] [PubMed]
- Shou, J.-W.; Shaw, P.-C. Berberine Reduces Lipid Accumulation in Obesity via Mediating Transcriptional Function of PPARδ. Int. J. Mol. Sci. 2023, 24, 11600. [Google Scholar] [CrossRef]
- Cho, A.-S.; Jeon, S.-M.; Kim, M.-J.; Yeo, J.; Seo, K.-I.; Choi, M.-S.; Lee, M.-K. Chlorogenic acid exhibits anti-obesity property and improves lipid metabolism in high-fat diet-induced-obese mice. Food Chem. Toxicol. 2010, 48, 937–943. [Google Scholar] [CrossRef]
- Zhong, Y.; Ding, Y.; Li, L.; Ge, M.; Ban, G.; Yang, H.; Dai, J.; Zhang, L. Effects and Mechanism of Chlorogenic Acid on Weight Loss. Curr. Pharm. Biotechnol. 2020, 21, 1099–1106. [Google Scholar] [CrossRef]
- Pimpley, V.; Patil, S.; Srinivasan, K.; Desai, N.; Murthy, P.S. The chemistry of chlorogenic acid from green coffee and its role in attenuation of obesity and diabetes. Prep. Biochem. Biotechnol. 2020, 50, 969–978. [Google Scholar] [CrossRef]
- Yan, Y.; Zhou, X.; Guo, K.; Zhou, F.; Yang, H. Use of Chlorogenic Acid against Diabetes Mellitus and Its Complications. J. Immunol. Res. 2020, 2020, 9680508. [Google Scholar] [CrossRef]
- Marton, L.T.; Pescinini-e-Salzedas, L.M.; Camargo, M.E.C.; Barbalho, S.M.; Haber, J.F.D.S.; Sinatora, R.V.; Detregiachi, C.R.P.; Girio, R.J.S.; Buchaim, D.V.; Cincotto Dos Santos Bueno, P. The Effects of Curcumin on Diabetes Mellitus: A Systematic Review. Front. Endocrinol. 2021, 12, 669448. [Google Scholar] [CrossRef]
- Pivari, F.; Mingione, A.; Brasacchio, C.; Soldati, L. Curcumin and Type 2 Diabetes Mellitus: Prevention and Treatment. Nutrients 2019, 11, 1837. [Google Scholar] [CrossRef]
- Lu, W.; Khatibi Shahidi, F.; Khorsandi, K.; Hosseinzadeh, R.; Gul, A.; Balick, V. An update on molecular mechanisms of curcumin effect on diabetes. J. Food Biochem. 2022, 46. [Google Scholar] [CrossRef]
- Neyrinck, A.M.; Sánchez, C.R.; Rodriguez, J.; Cani, P.D.; Bindels, L.B.; Delzenne, N.M. Prebiotic Effect of Berberine and Curcumin Is Associated with the Improvement of Obesity in Mice. Nutrients 2021, 13, 1436. [Google Scholar] [CrossRef] [PubMed]
- Zhu, T.; Li, M.; Zhu, M.; Liu, X.; Huang, K.; Li, W.; Wang, S.-X.; Yin, Y.; Li, P. Epigallocatechin-3-gallate alleviates type 2 diabetes mellitus via β-cell function improvement and insulin resistance reduction. Iran. J. Basic Med. Sci. 2022, 25. [Google Scholar] [CrossRef]
- Saadh, M.J.; Gataa, I.S.; Hussam, A.S.; Kaur, I.; Kumar, A.; Godara, P.; Zainul, R.; Muzammil, K.; Zahrani, Y. Effect of Epigallocatechin Gallate on Glycemic Index: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Clin. Ther. 2025, 47, 925–934. [Google Scholar] [CrossRef] [PubMed]
- Soussi, A.; Gargouri, M.; Magné, C.; Ben-Nasr, H.; Kausar, M.A.; Siddiqui, A.J.; Saeed, M.; Snoussi, M.; Adnan, M.; El-Feki, A.; et al. (−)-Epigallocatechin gallate (EGCG) pharmacokinetics and molecular interactions towards amelioration of hyperglycemia, hyperlipidemia associated hepatorenal oxidative injury in alloxan induced diabetic mice. Chem. Biol. Interact. 2022, 368, 110230. [Google Scholar] [CrossRef]
- Wan, C.; Ouyang, J.; Li, M.; Rengasamy, K.R.R.; Liu, Z. Effects of green tea polyphenol extract and epigallocatechin-3-O-gallate on diabetes mellitus and diabetic complications: Recent advances. Crit. Rev. Food Sci. Nutr. 2024, 64, 5719–5747. [Google Scholar] [CrossRef]
- Yıldızhan, K.; Bayir, M.H.; Huyut, Z.; Altındağ, F. Effect of Hesperidin on Lipid Profile, Inflammation and Apoptosis in Experimental Diabetes. Dokl. Biochem. Biophys. 2025, 521, 198–205. [Google Scholar] [CrossRef]
- Kaviani, F.; Baratpour, I.; Ghasemi, S. The Antidiabetic Mechanisms of Hesperidin: Hesperidin Nanocarriers as Promising Therapeutic Options for Diabetes. Curr. Mol. Med. 2024, 24, 1483–1493. [Google Scholar] [CrossRef]
- Peng, P.; Jin, J.; Zou, G.; Sui, Y.; Han, Y.; Zhao, D.; Liu, L. Hesperidin prevents hyperglycemia in diabetic rats by activating the insulin receptor pathway. Exp. Ther. Med. 2020, 21, 53. [Google Scholar] [CrossRef] [PubMed]
- Ansari, P.; Choudhury, S.T.; Seidel, V.; Rahman, A.B.; Aziz, Md.A.; Richi, A.E.; Rahman, A.; Jafrin, U.H.; Hannan, J.M.A.; Abdel-Wahab, Y.H.A. Therapeutic Potential of Quercetin in the Management of Type-2 Diabetes Mellitus. Life 2022, 12, 1146. [Google Scholar] [CrossRef] [PubMed]
- Dhanya, R. Quercetin for managing type 2 diabetes and its complications, an insight into multitarget therapy. Biomed. Pharmacother. 2022, 146, 112560. [Google Scholar] [CrossRef] [PubMed]
- Michala, A.-S.; Pritsa, A. Quercetin: A Molecule of Great Biochemical and Clinical Value and Its Beneficial Effect on Diabetes and Cancer. Diseases 2022, 10, 37. [Google Scholar] [CrossRef]
- Elekofehinti, O.O.; Molehin, O.R.; Akinjiyan, M.O.; Fakayode, A.E. Rutin modulates the TGR5/GLP1 pathway and downregulates proinflammatory cytokines genes in streptozotocin-induced diabetic rats. J. Food Bioact. 2024, 88–103. [Google Scholar] [CrossRef]
- Maradesha, T.; Patil, S.M.; Phanindra, B.; Achar, R.R.; Silina, E.; Stupin, V.; Ramu, R. Multiprotein Inhibitory Effect of Dietary Polyphenol Rutin from Whole Green Jackfruit Flour Targeting Different Stages of Diabetes Mellitus: Defining a Bio-Computational Stratagem. Separations 2022, 9, 262. [Google Scholar] [CrossRef]
- Zieniuk, B.; Pawełkowicz, M. Berberine as a Bioactive Alkaloid: Multi-Omics Perspectives on Its Role in Obesity Management. Metabolites 2025, 15, 467. [Google Scholar] [CrossRef]
- Gaba, S.; Saini, A.; Singh, G.; Monga, V. An insight into the medicinal attributes of berberine derivatives: A review. Bioorg. Med. Chem. 2021, 38, 116143. [Google Scholar] [CrossRef]
- Nguyen, V.; Taine, E.G.; Meng, D.; Cui, T.; Tan, W. Chlorogenic Acid: A Systematic Review on the Biological Functions, Mechanistic Actions, and Therapeutic Potentials. Nutrients 2024, 16, 924. [Google Scholar] [CrossRef]
- Urošević, M.; Nikolić, L.; Gajić, I.; Nikolić, V.; Dinić, A.; Miljković, V. Curcumin: Biological Activities and Modern Pharmaceutical Forms. Antibiotics 2022, 11, 135. [Google Scholar] [CrossRef]
- Mehmood, S.; Maqsood, M.; Mahtab, N.; Khan, M.I.; Sahar, A.; Zaib, S.; Gul, S. Epigallocatechin gallate: Phytochemistry, bioavailability, utilization challenges, and strategies. J. Food Biochem. 2022, 46. [Google Scholar] [CrossRef]
- Gamboa-Gómez, C.I.; Rocha-Guzmán, N.E.; Gallegos-Infante, J.A.; Moreno-Jiménez, M.R.; Vázquez-Cabral, B.D.; González-Laredo, R.F. Plants with potential use on obesity and its complications. EXCLI J. 14Doc809 ISSN 1611-2156 2015. [Google Scholar] [CrossRef]
- Pyrzynska, K. Hesperidin: A Review on Extraction Methods, Stability and Biological Activities. Nutrients 2022, 14, 2387. [Google Scholar] [CrossRef]
- Aghababaei, F.; Hadidi, M. Recent Advances in Potential Health Benefits of Quercetin. Pharmaceuticals 2023, 16, 1020. [Google Scholar] [CrossRef]
- Muvhulawa, N.; Dludla, P.V.; Ziqubu, K.; Mthembu, S.X.H.; Mthiyane, F.; Nkambule, B.B.; Mazibuko-Mbeje, S.E. Rutin ameliorates inflammation and improves metabolic function: A comprehensive analysis of scientific literature. Pharmacol. Res. 2022, 178, 106163. [Google Scholar] [CrossRef] [PubMed]
- Dutta, D.; Nagendra, L.; Anne, B.; Kumar, M.; Sharma, M.; Kamrul-Hasan, A.B.M. Orforglipron, a novel non-peptide oral daily glucagon-like peptide-1 receptor agonist as an anti-obesity medicine: A systematic review and meta-analysis. Obes. Sci. Pract. 2024, 10, e743. [Google Scholar] [CrossRef] [PubMed]
- Pratt, E.; Ma, X.; Liu, R.; Robins, D.; Haupt, A.; Coskun, T.; Sloop, K.W.; Benson, C. Orforglipron (LY3502970 ), a novel, oral non-peptide glucagon-like peptide-1 receptor agonist: A Phase 1a, blinded, placebo-controlled, randomized, single- and multiple-ascending-dose study in healthy participants. Diabetes Obes. Metab. 2023, 25, 2634–2641. [Google Scholar] [CrossRef] [PubMed]
- Daina, A.; Michielin, O.; Zoete, V. SwissADME: a free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci. Rep. 2017, 7, 42717. [Google Scholar] [CrossRef]
- Daina, A.; Zoete, V. A BOILED-Egg To Predict Gastrointestinal Absorption and Brain Penetration of Small Molecules. ChemMedChem 2016, 11, 1117–1121. [Google Scholar] [CrossRef]
- PubChem PubChem Available online:. Available online: https://pubchem.ncbi.nlm.nih.gov/ (accessed on 10 November 2025).
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef]
- Zhang, X.; Belousoff, M.J.; Danev, R.; Sexton, P.M.; Wootten, D. Semaglutide-bound Glucagon-Like Peptide-1 (GLP-1) Receptor in Complex with Gs protein: 7ki0 2021. [CrossRef]
- Zhao, F.H.; Zhou, Q.T.; Cong, Z.T.; Hang, K.N.; Zou, X.Y.; Zhang, C.; Chen, Y.; Dai, A.T.; Liang, A.Y.; Ming, Q.Q.; et al. Cryo-EM structure of the tirzepatide-bound human GIPR-Gs complex: 7fiy 2022. [CrossRef]
- Scapin, G. Human DPP4 in complex with a ligand 34a. Available: https://www.rcsb.
- Zhang, H.; Wen, H.Y. Crystal structure of E. coli NfsB in complex with berberine: 7x32 2022. [CrossRef]
- Pratap, S. Crystal structure of chorismate mutase like domain of bifunctional DAHP synthase of Bacillus subtilis in complex with Chlorogenic acid. Available. [CrossRef]
- Elkins, J.M.; Soundararajan, M.; Vollmar, M.; Krojer, T.; Bountra, C.; Edwards, A.M.; Arrowsmith, C.; Knapp, S. Human DYRK2 bound to Curcumin: 6hdr 2019. [CrossRef]
- Kowalinski, E.; Zubieta, C.; Wolkerstorfer, A.; Szolar, O.H.; Ruigrok, R.W.; Cusack, S. Influenza strain pH1N1 2009 polymerase subunit PA endonuclease in complex with (-)-epigallocatechin gallate from green tea: 4awm 2012. [CrossRef]
- Sun, B.; Kobilka, B.K.; Sloop, K.W.; Feng, D.; Kobilka, T.S. cryo-EM of human GLP-1R bound to non-peptide agonist LY3502970: 6xox 2020. [CrossRef]
- Arriaza, R.H.; Dermauw, W.; Wybouw, N.; Van Leeuwen, T.; Chruszcz, M. Crystal structure of TuUGT202A2 (Tetur22g00270) in complex with quercetin: 8sfw 2024. [CrossRef]
- Grygier, P. Crystal structure of DYRK1A in complex with rutin. Available: https://www.rcsb.
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef]
| Compound | Main Natural Sources | Reported Mechanism | References |
| Berberine | Berberis vulgaris, Coptis chinensis | Enhances GLP-1 secretion, improves insulin sensitivity | [9,15,16,17] |
| Chlorogenic acid | Coffee, sunflower, potatoes | Appetite suppression, glucose metabolism | [18,19,20,21] |
| Curcumin | Turmeric (Curcuma longa) | Anti-inflammatory, reported DPP-4 interaction | [22,23,24,25] |
| Epigallocatechin gallate (EGCG) | Green tea (Camellia sinensis) | Stimulates GLP-1 secretion, DPP-4 inhibition | [26,27,28,29] |
| Hesperidin | Citrus fruits | Incretin secretion enhancement, DPP-4 inhibition | [12,30,31,32] |
| Quercetin | Onions, apples, berries | Incretin modulation, anti-obesity activity | [33,34,35] |
| Rutin | Buckwheat, citrus, apples | GLP-1 modulation, antioxidant | [36,37] |
| Receptor affinity ((kcal/mol)1 | |||
| Ligand | GLP-1 R | GIP R | DPP4 |
| Berberine | -7.6 | -5,5 | -8,0 |
| Chlorogenic acid | -7.8 | -4,3 | -6,8 |
| Curcumin | -7,5 | -5,5 | -7,0 |
| Epigallocatechin gallate | -9,1 | -5,2 | -7,9 |
| Hesperidin | -9,2 | -5,8 | -8,3 |
| Orforglipron | -11,4 | -7.6 | -9.8 |
| Quercetin | -7,9 | -4,7 | -7,5 |
| Rutin | -9,2 | -5,5 | -8,0 |
| Molecule | GI Absorption | BBB* | Solubility | Log P** | BS*** | Notable Features |
| Berberine | High | Yes | Moderate | 2.53 | 0.55 | Balanced profile; multi-CYP inhibitor; good scaffold |
| Chlorogenic acid | Low | No | Very high | −0.39 | 0.11 | Highly polar; poor permeability |
| Curcumin | High | No | Moderate | 3.03 | 0.55 | Good oral potential; metabolic liabilities |
| Epigallocatechin gallate | Low | No | Moderate | 0.95 | 0.17 | Too polar; low bioavailability |
| Hesperidin | Low | No | Soluble | −1.06 | 0.17 | High MW; glycoside; poor permeability |
| Orforglipron | Low | No | Insoluble | 6.51 | 0.17 | Synthetic oral GLP-1 agonist; optimized despite poor properties |
| Quercetin | High | No | Soluble | 1.23 | 0.55 | Excellent scaffold; multiple CYP interactions |
| Rutin | Low | No | Soluble | −1.51 | 0.17 | Large, polar glycoside; likely inactive orally |
| Short name | Name | Role | ID* | Reference |
| GLP-1R | Glucagon-Like Peptide-1 (GLP-1) Receptor | receptor | 7KI0 | [53] |
| GIPR | Gastric inhibitory polypeptide receptor | receptor | 7FIY | [54] |
| DPP4 | Dipeptidyl peptidase-4 | receptor | 5T4B | [55] |
| Berberine | Berberine | ligand | 7X32 | [56] |
| CGA | Chlorogenic acid | ligand | 5GMU | [57] |
| Curcumin | Curcumin | ligand | 6HDR | [58] |
| EGCG | Epigallocatechin gallate | ligand | 4AWM | [59] |
| Hesperidin | Hesperidin | ligand | 10621*** | [51] |
| OFG** | Orforglipron | ligand | 6XOX | [60] |
| Quercetin | Quercetin | ligand | 8SFW | [61] |
| Rutin | Rutin | ligand | 8C3Q | [62] |
| Phase | Key Organs | Main Parameters | Determines |
| Absorption | GI tract, lungs, skin | Bioavailability (F) | How much drug enters circulation |
| Distribution | Blood, tissues | Volume of Distribution (Vd) | How widely the drug spreads |
| Metabolism | Liver (mainly) | Clearance, metabolic rate | How the drug is modified |
| Excretion | Kidneys, bile, lungs | Half-life (t½), Clearance (Cl) | How long the drug stays in the body |
| Molecule | PubChem CID | SMILES |
| Berberine | 2353 | COC1=C(C2=C[N+]3=C(C=C2C=C1)C4=CC5=C(C=C4CC3)OCO5)OC |
| Chlorogenic acid | 1794427 | C1[C@H]([C@H]([C@@H](C[C@@]1(C(=O)O)O)OC(=O)/C=C/C2=CC(=C(C=C2)O)O)O)O |
| Curcumin | 969516 | COC1=C(C=CC(=C1)/C=C/C(=O)CC(=O)/C=C/C2=CC(=C(C=C2)O)OC)O |
| Epigallocatechin gallate | 65064 | C1[C@H]([C@H](OC2=CC(=CC(=C21)O)O)C3=CC(=C(C(=C3)O)O)O)OC(=O)C4=CC(=C(C(=C4)O)O)O |
| Hesperidin | 10621 | C[C@H]1[C@@H]([C@H]([C@H]([C@@H](O1)OC[C@@H]2[C@H]([C@@H]([C@H]([C@@H](O2)OC3=CC(=C4C(=O)C[C@H](OC4=C3)C5=CC(=C(C=C5)OC)O)O)O)O)O)O)O)O |
| Orforglipron | 137319706 | C[C@H]1C[C@]1(C2=NOC(=O)N2)N3C4=C(C=C(C=C4)[C@H]5CCOC(C5)(C)C)C=C3C(=O)N6CCC7=NN(C(=C7[C@@H]6C)N8C=CN(C8=O)C9=C(C1=C(C=C9)N(N=C1)C)F)C1=CC(=C(C(=C1)C)F)C |
| Quercetin | 5280343 | C1=CC(=C(C=C1C2=C(C(=O)C3=C(C=C(C=C3O2)O)O)O)O)O |
| Rutin | 5280805 | C[C@H]1[C@@H]([C@H]([C@H]([C@@H](O1)OC[C@@H]2[C@H]([C@@H]([C@H]([C@@H](O2)OC3=C(OC4=CC(=CC(=C4C3=O)O)O)C5=CC(=C(C=C5)O)O)O)O)O)O)O)O |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




