Submitted:
13 November 2025
Posted:
14 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and discussion
2.1. Cisplatin vs. cisPtI2 : NMR Investigation of Ligand Exchange with Histidine, Cysteine, and Methionine
2.2. Computational Assessment
4. Materials and Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Linares, J.; Sallent-Aragay, A.; Badia-Ramentol, J.; Recort-Bascuas, A.; Méndez, A.; Manero-Rupérez, N.; Re, D.L.; Rivas, E.I.; Guiu, M.; Zwick, M.; et al. Long-Term Platinum-Based Drug Accumulation in Cancer-Associated Fibroblasts Promotes Colorectal Cancer Progression and Resistance to Therapy. Nat Commun 2023, 14, 746. [Google Scholar] [CrossRef]
- Oun, R.; Moussa, Y.E.; Wheate, N.J. The Side Effects of Platinum-Based Chemotherapy Drugs: A Review for Chemists. Dalton Trans. 2018, 47, 6645–6653. [Google Scholar] [CrossRef]
- Cirri, D.; Chiaverini, L.; Pratesi, A.; Marzo, T. Is the Next Cisplatin Already in Our Laboratory? Comments on Inorganic Chemistry 2023. [Google Scholar] [CrossRef]
- Chiaverini, L.; Leo, R.D.; Famlonga, L.; Pacini, M.; Baglini, E.; Barresi, E.; Peana, M.F.; Tolbatov, I.; Marrone, A.; La Mendola, D.; et al. The Metal(Loid)s’ Dilemma. What’s the next Step for a New Era of Inorganic Molecules in Medicine? Metallomics. 2025, 17, mfaf013. [Google Scholar] [CrossRef]
- Musumeci, D.; Platella, C.; Riccardi, C.; Merlino, A.; Marzo, T.; Massai, L.; Messori, L.; Montesarchio, D. A First-in-Class and a Fished out Anticancer Platinum Compound: Cis-[PtCl2(NH3)2] and Cis-[PtI2(NH3)2] Compared for Their Reactivity towards DNA Model Systems. Dalton Trans 2016, 45, 8587–8600. [Google Scholar] [CrossRef]
- Marzo, T.; Pillozzi, S.; Hrabina, O.; Kasparkova, J.; Brabec, V.; Arcangeli, A.; Bartoli, G.; Severi, M.; Lunghi, A.; Totti, F.; et al. Cis-Pt I2(NH3)2: A Reappraisal. Dalton Trans. 2015, 44, 14896–14905. [Google Scholar] [CrossRef] [PubMed]
- Quiroga, A.G.; Cama, M.; Pajuelo-Lozano, N.; Álvarez-Valdés, A.; Perez, I.S. New Findings in the Signaling Pathways of Cis and Trans Platinum Iodido Complexes’ Interaction with DNA of Cancer Cells. ACS Omega 2019, 4, 21855–21861. [Google Scholar] [CrossRef]
- Tolbatov, I.; Marzo, T.; Cirri, D.; Gabbiani, C.; Coletti, C.; Marrone, A.; Paciotti, R.; Messori, L.; Re, N. Reactions of Cisplatin and Cis-[PtI2(NH3)2] with Molecular Models of Relevant Protein Sidechains: A Comparative Analysis. J Inorg Biochem 2020, 209, 111096. [Google Scholar] [CrossRef]
- Wang, J.; Tao, J.; Jia, S.; Wang, M.; Jiang, H.; Du, Z. The Protein-Binding Behavior of Platinum Anticancer Drugs in Blood Revealed by Mass Spectrometry. Pharmaceuticals (Basel) 2021, 14, 104. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, T.; Chval, Z.; Burda, J.V. Cisplatin Interaction with Cysteine and Methionine in Aqueous Solution: Computational DFT/PCM Study. J. Phys. Chem. B 2009, 113, 3139–3150. [Google Scholar] [CrossRef] [PubMed]
- Tolbatov, I.; Marzo, T.; Umari, P.; Mendola, D.L.; Marrone, A. Detailed Mechanism of a DNA/RNA Nucleobase Substituting Bridging Ligand in Diruthenium(II,III) and Dirhodium(II,II) Tetraacetato Paddlewheel Complexes: Protonation of the Leaving Acetate Is Crucial. Dalton Trans. 2025, 54, 662–673. [Google Scholar] [CrossRef]
- Tolbatov, I.; Umari, P.; Marrone, A. The Binding of Diruthenium (II,III) and Dirhodium (II,II) Paddlewheel Complexes at DNA/RNA Nucleobases: Computational Evidences of an Appreciable Selectivity toward the AU Base Pairs. J Mol Graph Model 2024, 131, 108806. [Google Scholar] [CrossRef]
- Tolbatov, I.; Marrone, A. Reactivity of N-Heterocyclic Carbene Half-Sandwich Ru-, Os-, Rh-, and Ir-Based Complexes with Cysteine and Selenocysteine: A Computational Study. Inorg. Chem. 2022, 61, 746–754. [Google Scholar] [CrossRef]
- Scoditti, S.; Dabbish, E.; Russo, N.; Mazzone, G.; Sicilia, E. Anticancer Activity, DNA Binding, and Photodynamic Properties of a N∧C∧N-Coordinated Pt(II) Complex. Inorg. Chem. 2021, 60, 10350–10360. [Google Scholar] [CrossRef]
- Tolbatov, I.; Cirri, D.; Tarchi, M.; Marzo, T.; Coletti, C.; Marrone, A.; Messori, L.; Re, N.; Massai, L. Reactions of Arsenoplatin-1 with Protein Targets: A Combined Experimental and Theoretical Study. Inorg. Chem. 2022, 61, 3240–3248. [Google Scholar] [CrossRef]
- Dunning, T.H. Gaussian Basis Sets Tor Molecular Calculations. Springer Vol.3.
- Chai, J.-D.; Head-Gordon, M. Long-Range Corrected Hybrid Density Functionals with Damped Atom–Atom Dispersion Corrections. Phys. Chem. Chem. Phys. 2008, 10, 6615–6620. [Google Scholar] [CrossRef]
- Remya, K.; Suresh, C.H. Which Density Functional Is Close to CCSD Accuracy to Describe Geometry and Interaction Energy of Small Noncovalent Dimers? A Benchmark Study Using Gaussian09. Journal of Computational Chemistry 2013, 34, 1341–1353. [Google Scholar] [CrossRef]
- Andrae, D.; Häußermann, U.; Dolg, M.; Stoll, H.; Preuß, H. Energy-Adjustedab Initio Pseudopotentials for the Second and Third Row Transition Elements. Theoret. Chim. Acta 1990, 77, 123–141. [Google Scholar] [CrossRef]
- Weigend, F.; Ahlrichs, R. Balanced Basis Sets of Split Valence, Triple Zeta Valence and Quadruple Zeta Valence Quality for H to Rn: Design and Assessment of Accuracy. Phys. Chem. Chem. Phys. 2005, 7, 3297–3305. [Google Scholar] [CrossRef] [PubMed]
- Barone, V.; Cossi, M.; Tomasi, J. A New Definition of Cavities for the Computation of Solvation Free Energies by the Polarizable Continuum Model. J. Chem. Phys. 1997, 107, 3210–3221. [Google Scholar] [CrossRef]
- Klamt, A.; Moya, C.; Palomar, J. A Comprehensive Comparison of the IEFPCM and SS(V)PE Continuum Solvation Methods with the COSMO Approach. J. Chem. Theory Comput. 2015, 11, 4220–4225. [Google Scholar] [CrossRef] [PubMed]
- Geerlings, P.; De Proft, F.; Langenaeker, W. Conceptual Density Functional Theory. Chem. Rev. 2003, 103, 1793–1874. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, D.; Chattaraj, P.K. Conceptual Density Functional Theory Based Electronic Structure Principles. Chem. Sci. 2021, 12, 6264–6279. [Google Scholar] [CrossRef] [PubMed]
- Yang, Weitao. ; Mortier, W.J. The Use of Global and Local Molecular Parameters for the Analysis of the Gas-Phase Basicity of Amines. J. Am. Chem. Soc. 1986, 108, 5708–5711. [Google Scholar] [CrossRef]

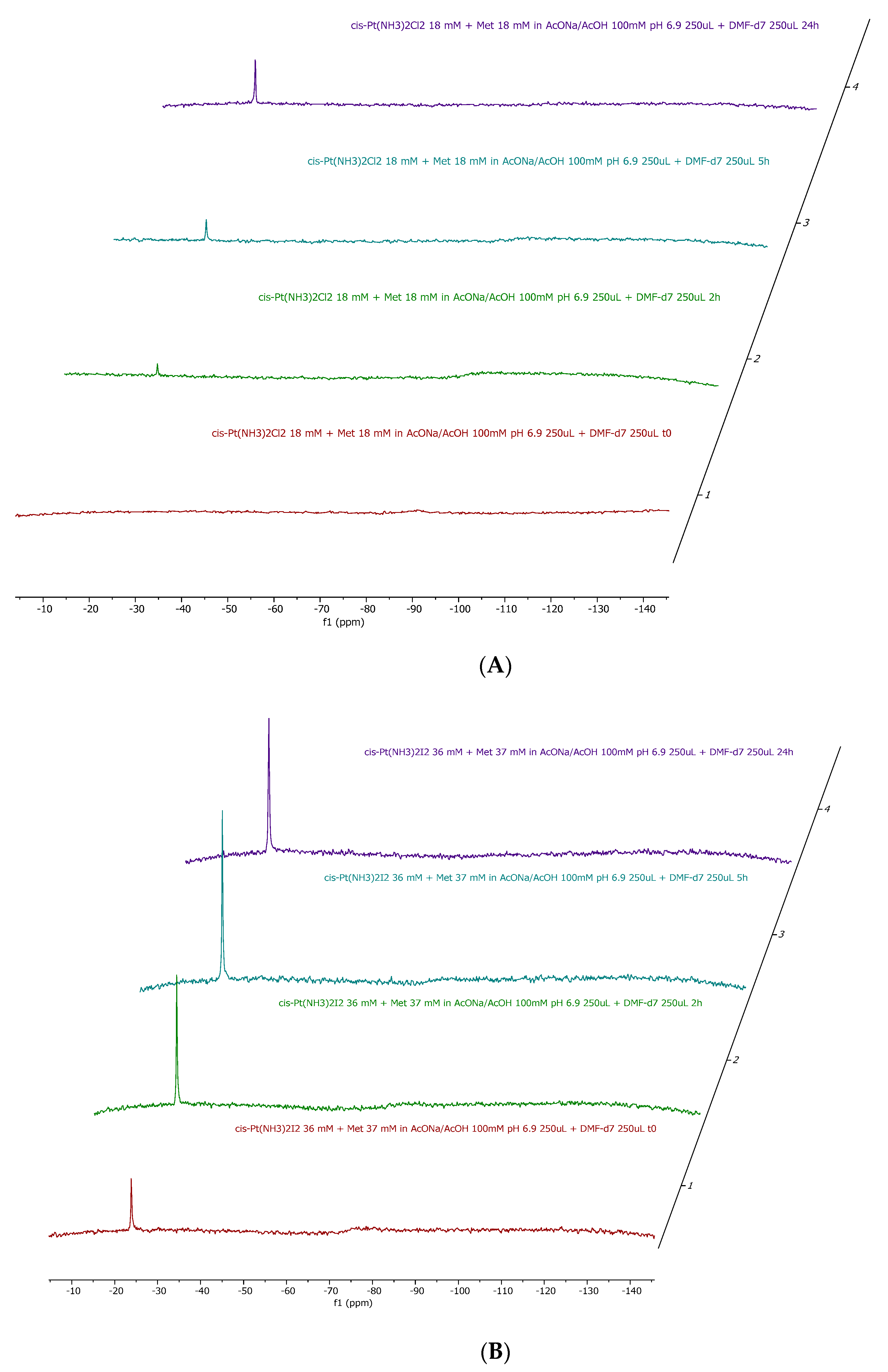
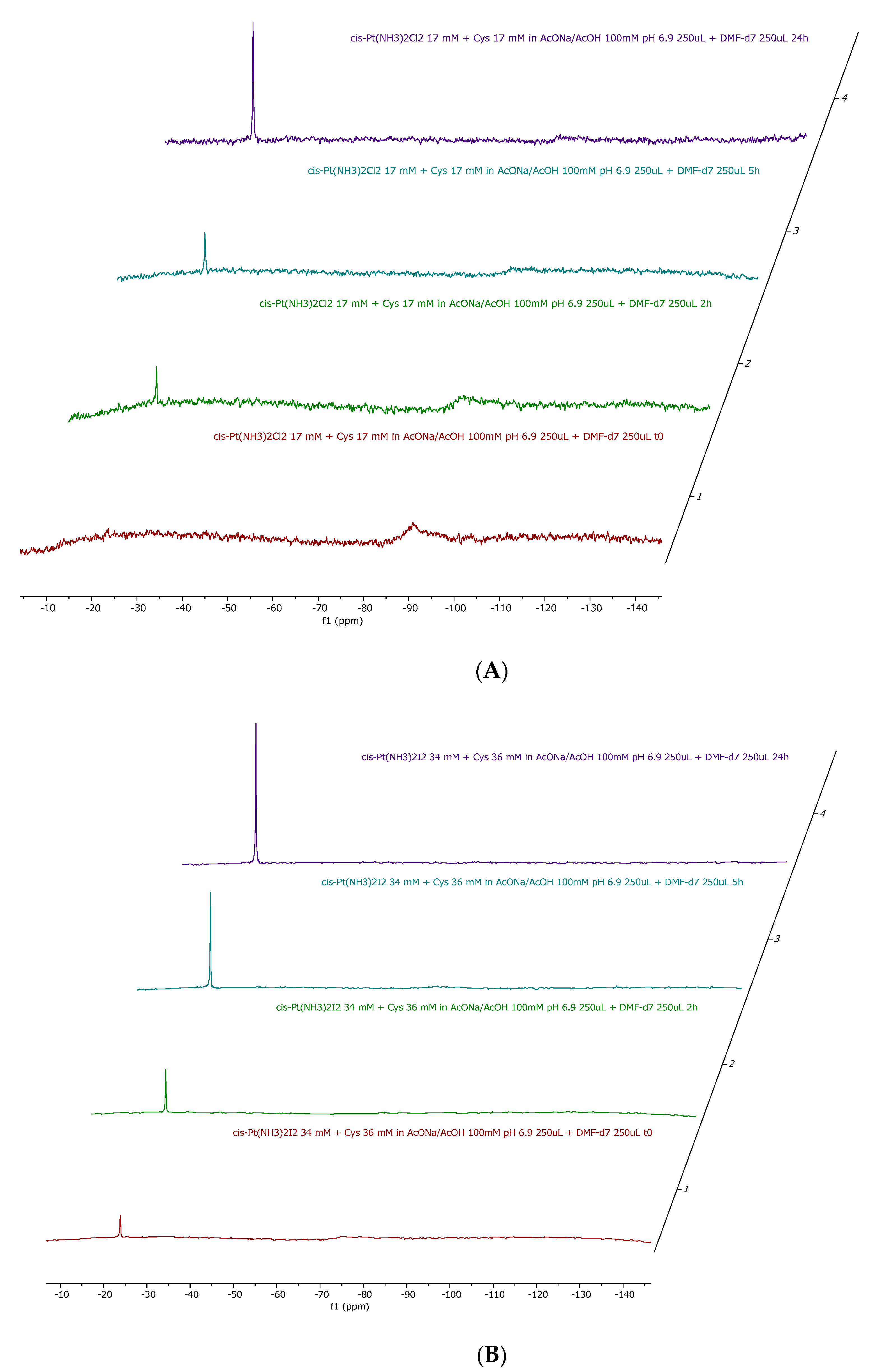
| Cisplatin + His | Cisplatin + Met | Cisplatin + Cys | cis-[PtI2(NH3)2] + His | cis-[PtI2(NH3)2] + Met | cis-[PtI2(NH3)2] + Cys |
|---|---|---|---|---|---|
| No release | 11% | 14% | 54% | 41% | 100% |
| Complex | GFE | ||||
|---|---|---|---|---|---|
| Pt(NH3)2Cl2 | 0 | -0.140 | 0.169 | 2.953 | 0.058 |
| Pt(NH3)2Cl(CH3SH) | 4.9 | -0.161 | 0.169 | 2.966 | 0.077 |
| Pt(NH3)2Cl(CH3SCH3) | 0.2 | -0.160 | 0.170 | 2.942 | 0.075 |
| Pt(NH3)2Cl(imi) | -4.2 | -0.144 | 0.177 | 2.819 | 0.059 |
| Pt(NH3)Cl2(CH3SH) | 5.1 | -0.154 | 0.163 | 3.071 | 0.072 |
| Pt(NH3)Cl2(CH3SCH3) | 1.0 | -0.153 | 0.164 | 3.053 | 0.071 |
| Pt(NH3)Cl2(imi) | -1.6 | -0.138 | 0.169 | 2.954 | 0.056 |
| Complex | GFE | μ | η | S | ω |
|---|---|---|---|---|---|
| Pt(NH3)2I2 | 0 | -0.149 | 0.148 | 3.386 | 0.075 |
| Pt(NH3)2I(CH3SH) | 2.2 | -0.161 | 0.152 | 3.279 | 0.085 |
| Pt(NH3)2I(CH3SCH3) | -2.3 | -0.159 | 0.153 | 3.259 | 0.082 |
| Pt(NH3)2I(imi) | -7.4 | -0.146 | 0.160 | 3.117 | 0.067 |
| Pt(NH3)I2(CH3SH) | 4.0 | -0.159 | 0.142 | 3.528 | 0.089 |
| Pt(NH3)I2(CH3SCH3) | -0.1 | -0.157 | 0.142 | 3.511 | 0.087 |
| Pt(NH3)I2(imi) | -2.9 | -0.146 | 0.149 | 3.352 | 0.071 |
| complex | Fukui index | Pt | N | N | Cl | Cl | N(imi) | S |
|---|---|---|---|---|---|---|---|---|
| Pt(NH3)2Cl2 |
-0.414 | -0.024 | -0.024 | -0.169 | -0.169 | n/a | n/a | |
| -0.517 | -0.001 | -0.001 | -0.172 | -0.172 | n/a | n/a | ||
| Pt(NH3)2Cl(CH3SH) | -0.361 | -0.024 | -0.031 | -0.172 | n/a | n/a | -0.136 | |
| -0.462 | -0.001 | -0.009 | -0.286 | n/a | n/a | -0.054 | ||
| Pt(NH3)2Cl(CH3SCH3) | -0.362 | -0.022 | -0.030 | -0.166 | n/a | n/a | -0.121 | |
| -0.459 | 0.000 | -0.009 | -0.274 | n/a | n/a | -0.053 | ||
| Pt(NH3)2Cl(imi) | -0.424 | -0.011 | -0.041 | -0.189 | n/a | 0.002 | n/a | |
| -0.485 | 0.004 | -0.024 | -0.292 | n/a | 0.016 | n/a | ||
| Pt(NH3)Cl2(CH3SH) | -0.340 | -0.024 | n/a | -0.161 | -0.158 | n/a | -0.134 | |
| -0.442 | 0.002 | n/a | -0.275 | -0.100 | n/a | -0.044 | ||
| Pt(NH3)Cl2(CH3SCH3) | -0.344 | -0.023 | n/a | -0.154 | -0.155 | n/a | -0.116 | |
| -0.443 | -0.003 | n/a | -0.258 | -0.104 | n/a | -0.043 | ||
| Pt(NH3)Cl2(imi) | -0.404 | -0.020 | n/a | -0.173 | -0.168 | 0.006 | n/a | |
| -0.470 | 0.000 | n/a | -0.112 | -0.247 | 0.026 | n/a |
| complex | Fukui index | Pt | N | N | I | I | N(imi) | S |
|---|---|---|---|---|---|---|---|---|
| Pt(NH3)2I2 |
-0.302 | -0.022 | -0.022 | -0.245 | -0.245 | n/a | n/a | |
| -0.359 | 0.004 | 0.004 | -0.278 | -0.278 | n/a | n/a | ||
| Pt(NH3)2I(CH3SH) | -0.308 | -0.025 | -0.028 | -0.278 | n/a | n/a | -0.116 | |
| -0.226 | -0.011 | 0.004 | -0.633 | n/a | n/a | -0.025 | ||
| Pt(NH3)2I(CH3SCH3) | -0.312 | -0.025 | -0.026 | -0.273 | n/a | n/a | -0.100 | |
| -0.226 | -0.010 | 0.004 | -0.630 | n/a | n/a | -0.023 | ||
| Pt(NH3)2I(imi) | -0.345 | -0.017 | -0.035 | -0.304 | n/a | 0.004 | n/a | |
| -0.249 | -0.009 | -0.002 | -0.635 | n/a | 0.029 | n/a | ||
| Pt(NH3)I2(CH3SH) | -0.254 | -0.021 | n/a | -0.232 | -0.232 | n/a | -0.107 | |
| -0.212 | -0.005 | n/a | -0.571 | -0.114 | n/a | -0.019 | ||
| Pt(NH3)I2(CH3SCH3) | -0.261 | -0.019 | n/a | -0.228 | -0.230 | n/a | -0.089 | |
| -0.216 | -0.004 | n/a | -0.562 | -0.118 | n/a | -0.018 | ||
| Pt(NH3)I2(imi) | -0.289 | -0.021 | n/a | -0.252 | -0.236 | 0.009 | n/a | |
| -0.097 | 0.000 | n/a | -0.354 | -0.482 | 0.014 | n/a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




