Submitted:
13 November 2025
Posted:
14 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
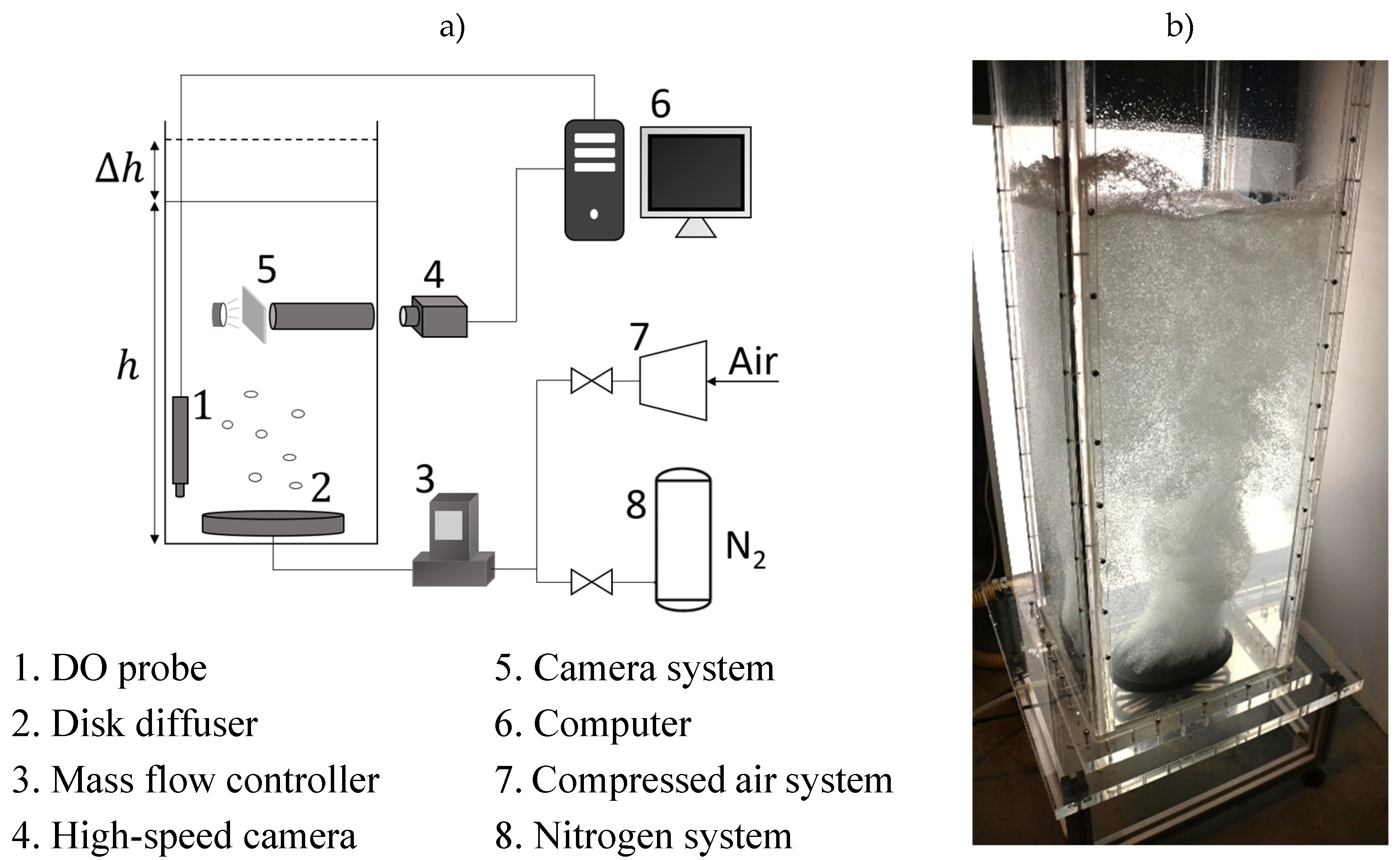
2.2. Experimental Setup
2.3. Method
2.3.1. Volumetric Mass Transfer Coefficient
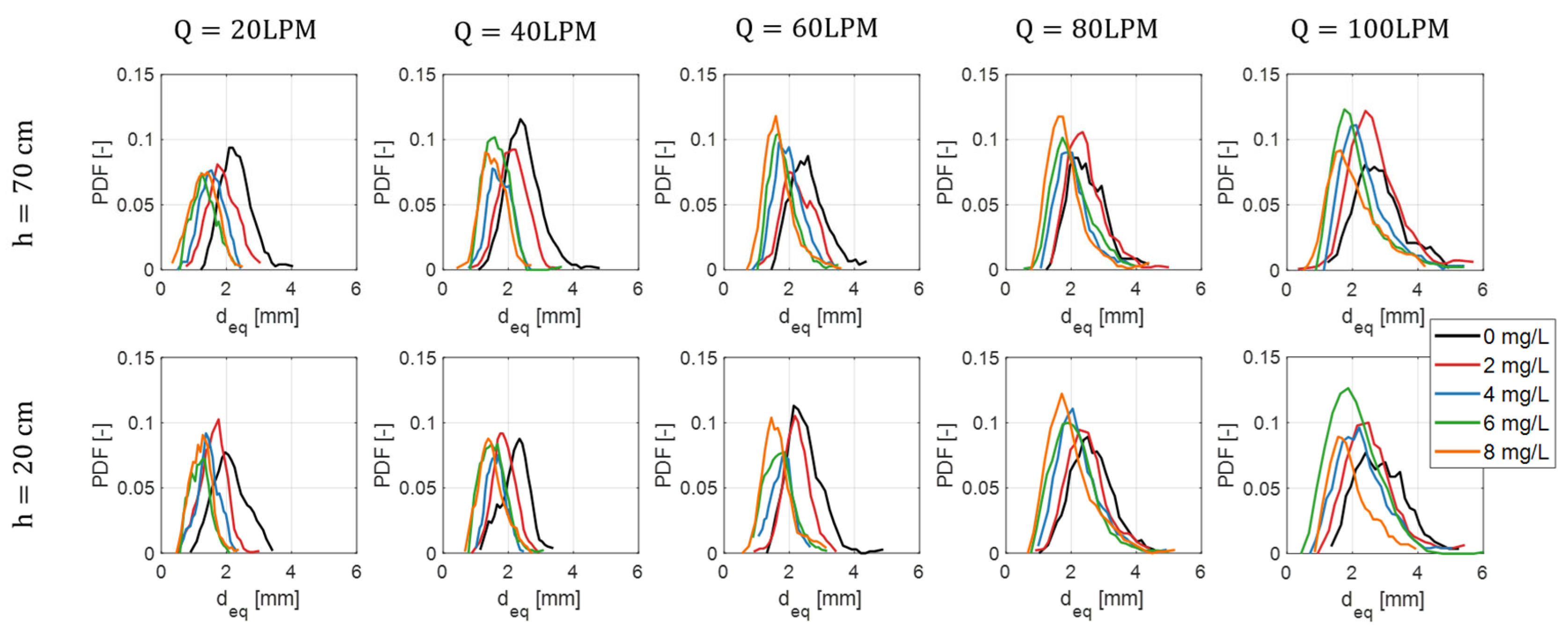
2.3.2. Bubble Equivalent Diameter
2.3.3. Gas Holdup
2.3.4. Interfacial Area
2.3.5. Mass Transfer Coefficient
3. Results
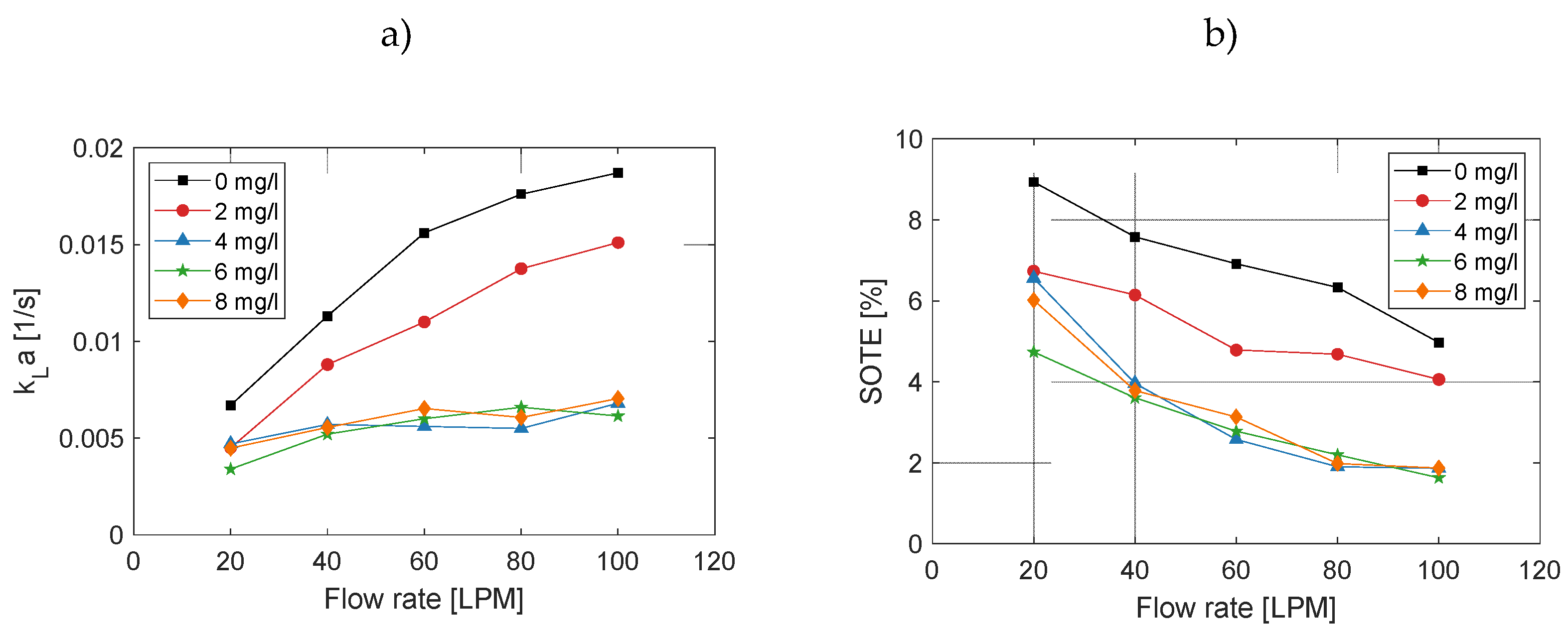
3.1. Effect on Volumetric Mass Transfer Coefficient
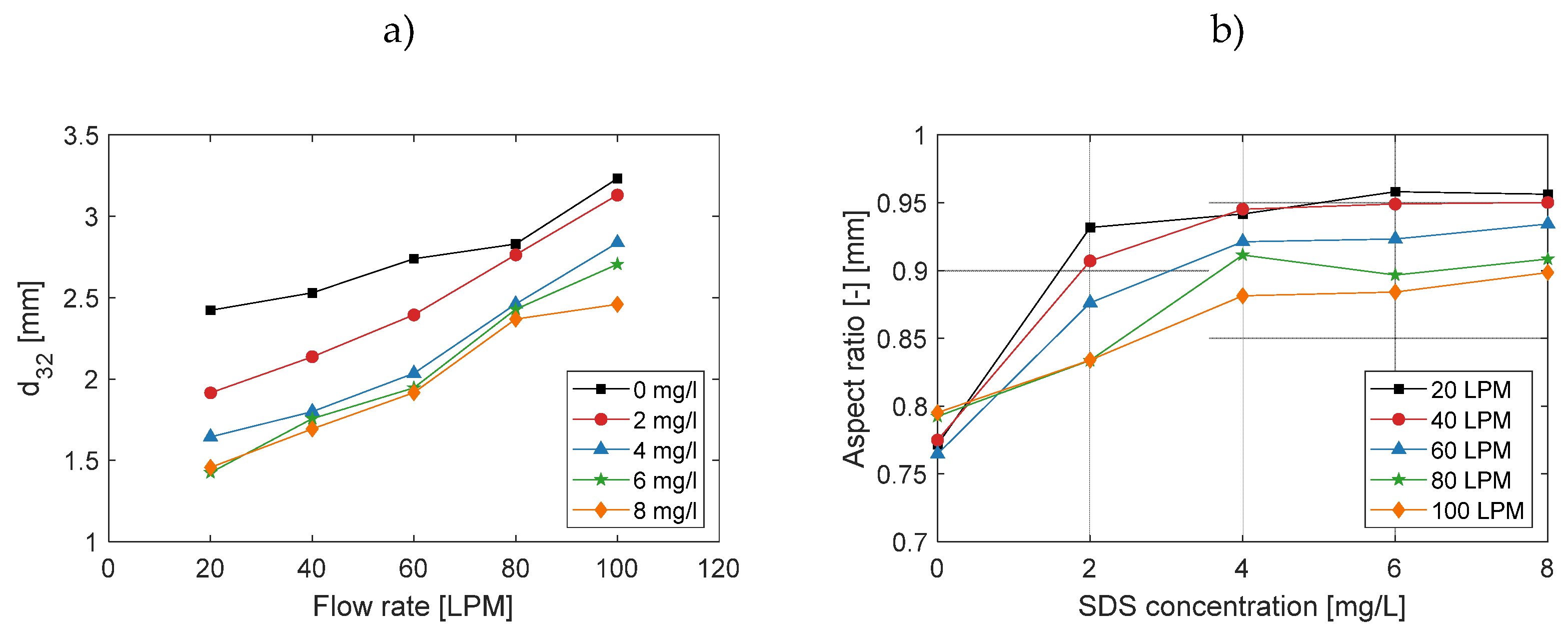
3.2. Effect on Bubble Size and Shape
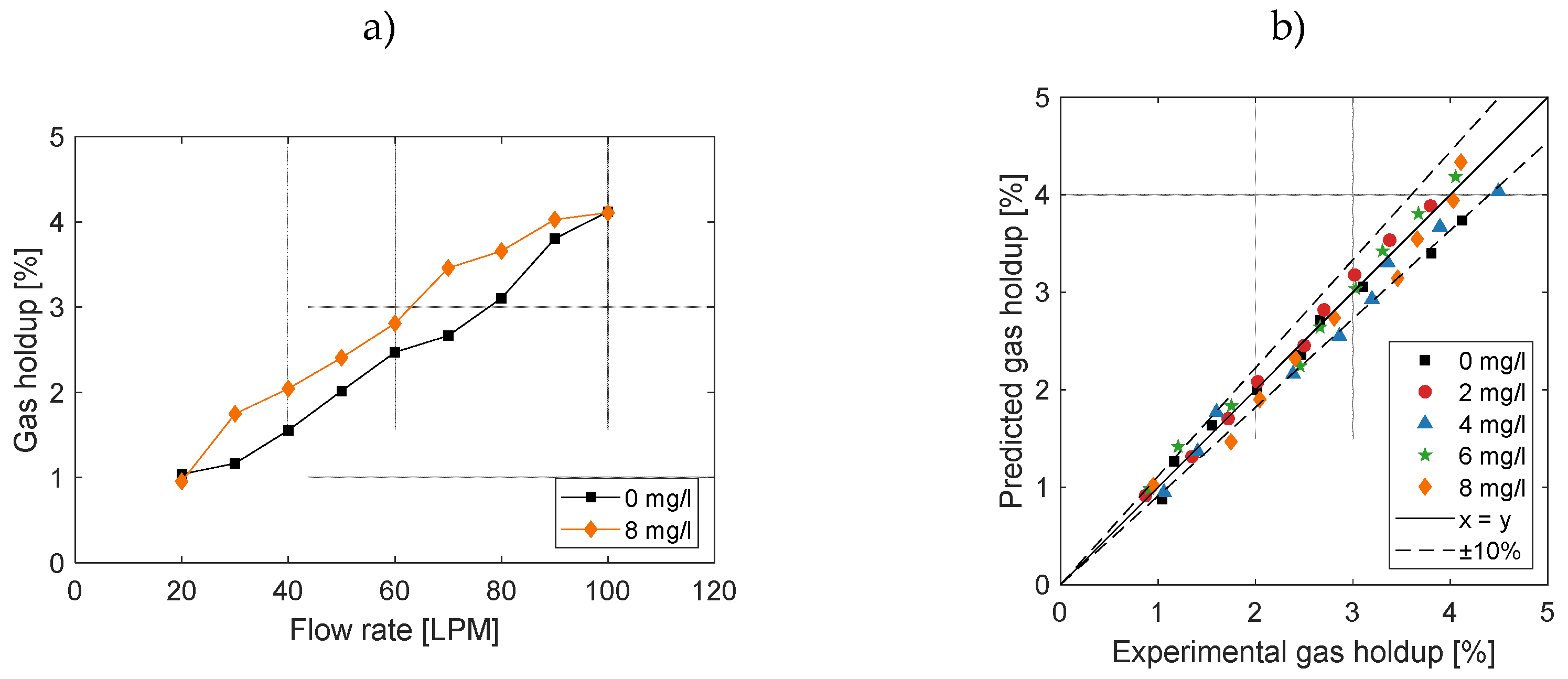
3.3. Effect on Gas Holdup
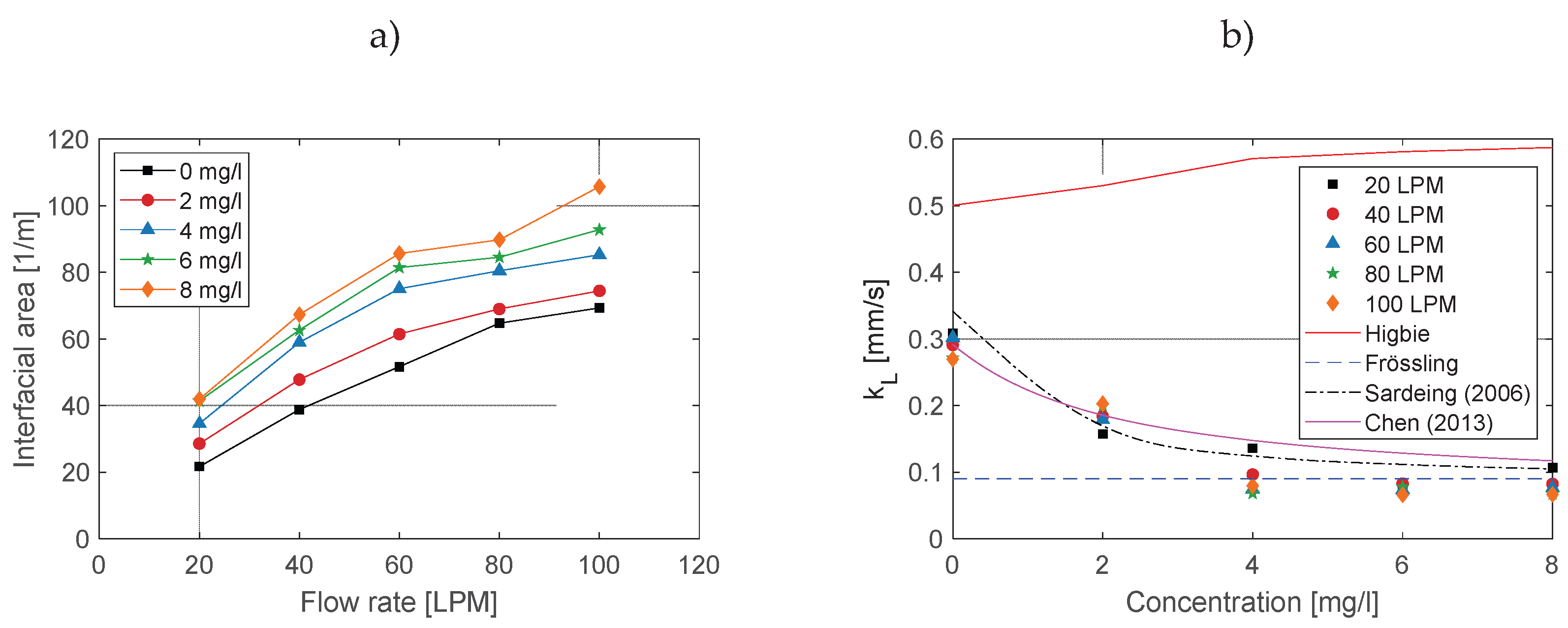
3.4. Effect on Interfacial Area
3.5. Effect on Mass Transfer Coefficient
- For bubble diameters smaller than 1.5 mm. In this range, is independent of surfactant concentration and corresponds to the mass transfer coefficient for rigid bubbles. In this regime, can be described using the Calderbank and Moo-Young’s correlation [45] or the Frössling correlation (Equation (11)).
- For bubble diameters between 1.5 mm and 3.5 mm. In this range, increases with bubble diameter, but in the presence of surfactants, this increase is significantly reduced. Consequently, varies approximately linearly between the limits corresponding to bubbles smaller than 1.5 mm and larger than 3.5 mm. In this study, we are within this range, which is the most critical due to the variability of with the bubble diameter. For this reason, it is essential to accurately measure the bubble diameter.
- For bubble diameters larger than 3.5 mm. For this range, does not depend on bubble diameter. The constant is determined by the interfacial coverage (), the mass transfer coefficient for a clean (surfactant-free) interface (, given by Higbie’s correlation, Eq. (9)), and the mass transfer coefficient for a fully surfactant-saturated interface (, given by Eq. (13)).
4. Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Obaideen, K.; Shehata, N.; Sayed, E.T.; Abdelkareem, M.A.; Mahmoud, M.S.; Olabi, A.G. The Role of Wastewater Treatment in Achieving Sustainable Development Goals (SDGs) and Sustainability Guideline. Energy Nexus 2022, 7. [Google Scholar] [CrossRef]
- Fayolle, Y.; Cockx, A.; Gillot, S.; Roustan, M.; Héduit, A. Oxygen Transfer Prediction in Aeration Tanks Using CFD. Chem Eng Sci 2007, 62, 7163–7171. [Google Scholar] [CrossRef]
- Wagner, M.; Pöpel, J. Surface Active Agents and Their Influence on Oxygen Transfer. Wat. Sci. Tech. 1996, 34, 249–256. [Google Scholar] [CrossRef]
- Rosso, D.; Stenstrom, M.K. Surfactant Effects on α-Factors in Aeration Systems. Water Res 2006, 40, 1397–1404. [Google Scholar] [CrossRef]
- Alves, S.S.; Orvalho, S.P.; Vasconcelos, J.M.T. Effect of Bubble Contamination on Rise Velocity and Mass Transfer. Chem Eng Sci 2005, 60, 1–9. [Google Scholar] [CrossRef]
- Jamnongwong, M.; Loubiere, K.; Dietrich, N.; Hébrard, G. Experimental Study of Oxygen Diffusion Coefficients in Clean Water Containing Salt, Glucose or Surfactant: Consequences on the Liquid-Side Mass Transfer Coefficients. Chemical Engineering Journal 2010, 165, 758–768. [Google Scholar] [CrossRef]
- Nekoeian, S.; Aghajani, M.; Alavi, S.M.; Sotoudeh, F. Effect of Surfactants on Mass Transfer Coefficients in Bubble Column Contactors: An Interpretative Critical Review Study. Reviews in Chemical Engineering 2021, 37, 585–617. [Google Scholar] [CrossRef]
- Rosso, D.; Huo, D.L.; Stenstrom, M.K. Effects of Interfacial Surfactant Contamination on Bubble Gas Transfer. Chem Eng Sci 2006, 61, 5500–5514. [Google Scholar] [CrossRef]
- Tagawa, Y.; Takagi, S.; Matsumoto, Y. Surfactant Effect on Path Instability of a Rising Bubble. J Fluid Mech 2014, 738, 124–142. [Google Scholar] [CrossRef]
- Loubière, K.; Hébrard, G. Influence of Liquid Surface Tension (Surfactants) on Bubble Formation at Rigid and Flexible Orifices. Chemical Engineering and Processing 2004, 43, 1361–1369. [Google Scholar] [CrossRef]
- Li, Y.; Zhu, T.; Liu, Y.; Tian, Y.; Wang, H. Effects of Surfactant on Bubble Hydrodynamic Behavior under Flotation-Related Conditions in Wastewater. Water Science and Technology 2012, 65, 1060–1066. [Google Scholar] [CrossRef] [PubMed]
- Painmanakul, P.; Loubière, K.; Hébrard, G.; Mietton-Peuchot, M.; Roustan, M. Effect of Surfactants on Liquid-Side Mass Transfer Coefficients. Chem Eng Sci 2005, 60, 6480–6491. [Google Scholar] [CrossRef]
- Sardeing, R.; Painmanakul, P.; Hébrard, G. Effect of Surfactants on Liquid-Side Mass Transfer Coefficients in Gas-Liquid Systems: A First Step to Modeling. Chem Eng Sci 2006, 61, 6249–6260. [Google Scholar] [CrossRef]
- Zhang, B.; Wang, Z.; Luo, Y.; Guo, K.; Zheng, L.; Liu, C. A Mathematical Model for Single CO2 Bubble Motion with Mass Transfer and Surfactant Adsorption/Desorption in Stagnant Surfactant Solutions. Sep Purif Technol 2023, 308. [Google Scholar] [CrossRef]
- Lebrun, G.; Clergerie, N.; Hébrard, G.; Dietrich, N. Modeling Oxygen Mass Transfer in Surfactant Solutions Considering Hydrodynamics and Physico-Chemical Phenomena. Chem Eng Sci 2025, 304. [Google Scholar] [CrossRef]
- Luo, Y.; Wang, Z.; Zhang, B.; Guo, K.; Zheng, L.; Xiang, W.; Liu, H.; Liu, C. Experimental Study of the Effect of the Surfactant on the Single Bubble Rising in Stagnant Surfactant Solutions and a Mathematical Model for the Bubble Motion. Ind Eng Chem Res 2022, 61, 9514–9527. [Google Scholar] [CrossRef]
- Zheng, K.; Li, C.; Yan, X.; Zhang, H.; Wang, L. Prediction of Bubble Terminal Velocity in Surfactant Aqueous Solutions. Canadian Journal of Chemical Engineering 2020, 98, 607–615. [Google Scholar] [CrossRef]
- Babu, R.; Das, M.K. Effects of Surface-Active Agents on Bubble Growth and Detachment from Submerged Orifice. Chem Eng Sci 2018, 179, 172–184. [Google Scholar] [CrossRef]
- García-Abuín, A.; Gómez-Díaz, D.; Navaza, J.M.; Sanjurjo, B. Effect of Surfactant Nature upon Absorption in a Bubble Column. Chem Eng Sci 2010, 65, 4484–4490. [Google Scholar] [CrossRef]
- Vasconcelos, J.M.T.; Rodrigues, J.M.L.; Orvalho, S.C.P.; Alves, S.S.; Mendes, R.L.; Reis, A. Effect of Contaminants on Mass Transfer Coefficients in Bubble Column and Airlift Contactors. Chem Eng Sci 2003, 58, 1431–1440. [Google Scholar] [CrossRef]
- Ahmia, A.C.; Idouhar, M.; Wongwailikit, K.; Dietrich, N.; Hébrard, G. Impact of Cellulose and Surfactants on Mass Transfer of Bubble Columns. Chem Eng Technol 2019, 42, 2465–2475. [Google Scholar] [CrossRef]
- Mcclure, D.D.; Lee, A.C.; Kavanagh, J.M.; Fletcher, D.F.; Barton, G.W. Impact of Surfactant Addition on Oxygen Mass Transfer in a Bubble Column. Chem Eng Technol 2015, 38, 44–52. [Google Scholar] [CrossRef]
- Jia, X.; Hu, W.; Yuan, X.; Yu, K. Effect of Surfactant Type on Interfacial Area and Liquid Mass Transfer for CO2 Absorption in a Bubble Column. Chin J Chem Eng 2015, 23, 476–481. [Google Scholar] [CrossRef]
- Lu, J.; Corvalan, C.M.; Chew, Y.M.J.; Huang, J.Y. Coalescence of Small Bubbles with Surfactants. Chem Eng Sci 2019, 196, 493–500. [Google Scholar] [CrossRef]
- Asgharpour, M.; Mehrnia, M.R.; Mostoufi, N. Effect of Surface Contaminants on Oxygen Transfer in Bubble Column Reactors. Biochem Eng J 2010, 49, 351–360. [Google Scholar] [CrossRef]
- Abadie, T.; al Ma Awali, S.M.; Brennan, B.; Briciu-Burghina, C.; Tajparast, M.; Passos, T.M.; Durkan, J.; Holland, L.; Lawler, J.; Nolan, K.; et al. Oxygen Transfer of Microbubble Clouds in Aqueous Solutions – Application to Wastewater. Chem Eng Sci 2022, 257. [Google Scholar] [CrossRef]
- Chen, X.; Liu, G. hua; Fan, H.; Li, M.; Luo, T.; Qi, L.; Wang, H. Effects of Surfactant Contamination on Oxygen Mass Transfer in Fine Bubble Aeration Process. Korean Journal of Chemical Engineering 2013, 30, 1741–1746. [Google Scholar] [CrossRef]
- Gómez-Díaz, D.; Navaza, J.M.; Sanjurjo, B. Interfacial Area Evaluation in a Bubble Column in the Presence of a Surface-Active Substance. Comparison of Methods. Chemical Engineering Journal 2008, 144, 379–385. [Google Scholar] [CrossRef]
- Herrmann-Heber, R.; Oleshova, M.; Reinecke, S.F.; Meier, M.; Taş, S.; Hampel, U.; Lerch, A. Population Balance Modeling-Assisted Prediction of Oxygen Mass Transfer Coefficients with Optical Measurements. Journal of Water Process Engineering 2024, 64. [Google Scholar] [CrossRef]
- Frössling, N. The Evaporation of Falling Drops. Gerlands Beitr. Geophys. 1938, 52, 170–216. [Google Scholar]
- Higbie, R. The Rate of Absorption of a Pure Gas into a Still Liquid during Short Periods of Exposure. Transactions of the AIChE, 1935, 31, 365–389. [Google Scholar]
- Liu, Y.; Wang, B.; Zhao, D.; Jin, W.; Xu, F.; Gao, Y.; Shi, W.; Ren, H. Investigation of Surfactant Effect on Ozone Bubble Motion and Mass Transfer Characteristics. J Environ Chem Eng 2023, 11. [Google Scholar] [CrossRef]
- Campbell, K.; Wang, J. New Insights into the Effect of Surfactants on Oxygen Mass Transfer in Activated Sludge Process. J Environ Chem Eng 2020, 8. [Google Scholar] [CrossRef]
- Machado, V.C.; Lafuente, J.; Baeza, J.A. Activated Sludge Model 2d Calibration with Full-Scale WWTP Data: Comparing Model Parameter Identifiability with Influent and Operational Uncertainty. Bioprocess Biosyst Eng 2014, 37, 1271–1287. [Google Scholar] [CrossRef] [PubMed]
- American Society of Civil Engineers. Measurement of Oxygen Transfer in Clean Water; The Society, 1993; ISBN 087262885X.
- Shah, Y.T.; Kelkar, B.G.; Godbole, S.P.; Deckwer, W.-D. Design Parameters Column Reactors Estimations for Bubble Column Reactors; 1982; Vol. 28.
- van’t Riet, K.; Tramper, J. Basic Bioreactor Design; 1st ed.; CRC Press: New York, 1991.
- Watcharasing, S.; Kongkowit, W.; Chavadej, S. Motor Oil Removal from Water by Continuous Froth Flotation Using Extended Surfactant: Effects of Air Bubble Parameters and Surfactant Concentration. Sep Purif Technol 2009, 70, 179–189. [Google Scholar] [CrossRef]
- Li, X.; Gao, Y.; Li, M.; Jiang, H.; Xie, J.; Xiang, G. Comparison and Mechanism Analysis of Three-Phase Contact Formation onto Hydrophilic/Hydrophobic Mineral Surfaces in the Presence of Cationic/Anionic Surfactants during Flotation Process. Minerals 2022, 12. [Google Scholar] [CrossRef]
- Moraveji, M.K.; Pasand, M.M.; Davarnejad, R.; Chisti, Y. Effects of Surfactants on Hydrodynamics and Mass Transfer in a Split-Cylinder Airlift Reactor. Canadian Journal of Chemical Engineering 2012, 90, 93–99. [Google Scholar] [CrossRef]
- Mcclure, D.D.; Deligny, J.; Kavanagh, J.M.; Fletcher, D.F.; Barton, G.W. Impact of Surfactant Chemistry on Bubble Column Systems. Chem Eng Technol 2014, 37, 652–658. [Google Scholar] [CrossRef]
- Lebrun, G.; Xu, F.; Le Men, C.; Hébrard, G.; Dietrich, N. Gas-Liquid Mass Transfer around a Rising Bubble: Combined Effect of Rheology and Surfactant. Fluids 2021, 6. [Google Scholar] [CrossRef]
- Li, S.; Huang, S.; Fan, J. Effect of Surfactants on Gas Holdup in Shear-Thinning Fluids. International Journal of Chemical Engineering 2017, 2017. [Google Scholar] [CrossRef]
- Tomiyama, A.; Kataoka, I.; Zun, I.; Sakaguchi, T. Drag Coefficients of Single Bubbles under Normal and Micro Gravity Conditions. JSME International Journal, Series B: Fluids and Thermal Engineering 1998, 41, 472–479. [Google Scholar] [CrossRef]
- Calderbank, P.H.; Moo-Young, M.B. THE CONTINUOUS PHASE HEAT AND MASS TRANSFER PROPERTIES OF DISPERSIONS. Chem Eng Sci 1961, 16, 36–54. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



