Submitted:
12 November 2025
Posted:
13 November 2025
You are already at the latest version
Abstract
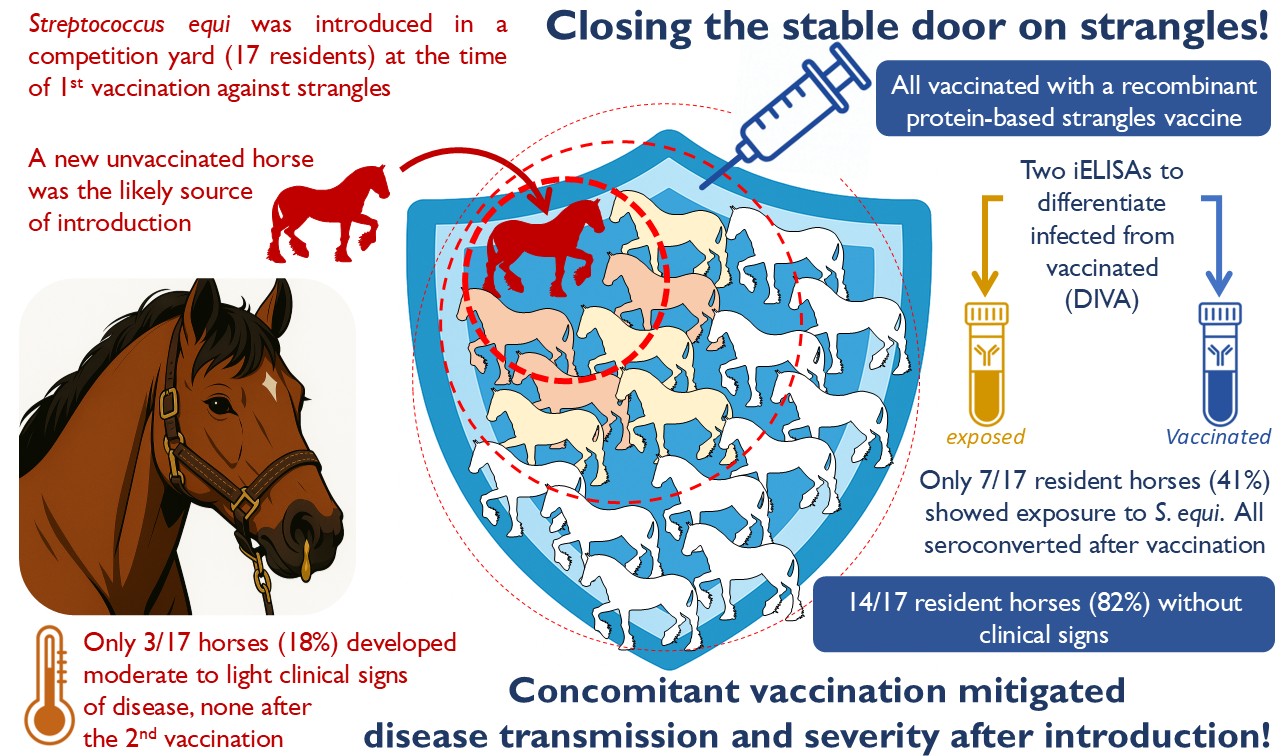
Infection of horses with Streptococcus equi subspecies equi (S. equi), the causative agent of strangles, is associated with the movement of horses from one farm to another, or the attendance of equestrian events and sales. Strangles can affect all horses on a farm, leading to the death of up to 10% of cases depending on the success of biosecurity measures and the use of vaccination. This retrospective study used ELISAs to measure the exposure of horses to S. equi at a farm that experienced an outbreak of strangles shortly after the introduction of a new horse on the same day that the majority of the 17 resident horses were vaccinated with Strangvac for the first time. One vaccinated horse, which subsequently tested positive for S. equi and EHV-4, developed a cough, elevated body temperature and nasal discharge 11 days after first vaccination. Two other horses developed fever for one day at 22 days post-first vaccination, but only one had serological evidence of exposure to S. equi. All vaccinated horses had high antibody titres to vaccine components, whilst seven of the 17 resident horses, and the new arrival, tested seropositive for exposure to S. equi. Although three out of the 17 vaccinated horses developed mild signs of disease before second vaccination, serological data support the effectiveness of vaccination in resident populations of horses to minimise the risk of strangles following the introduction of a new horse.
Keywords:
1. Introduction
2. Materials and Methods
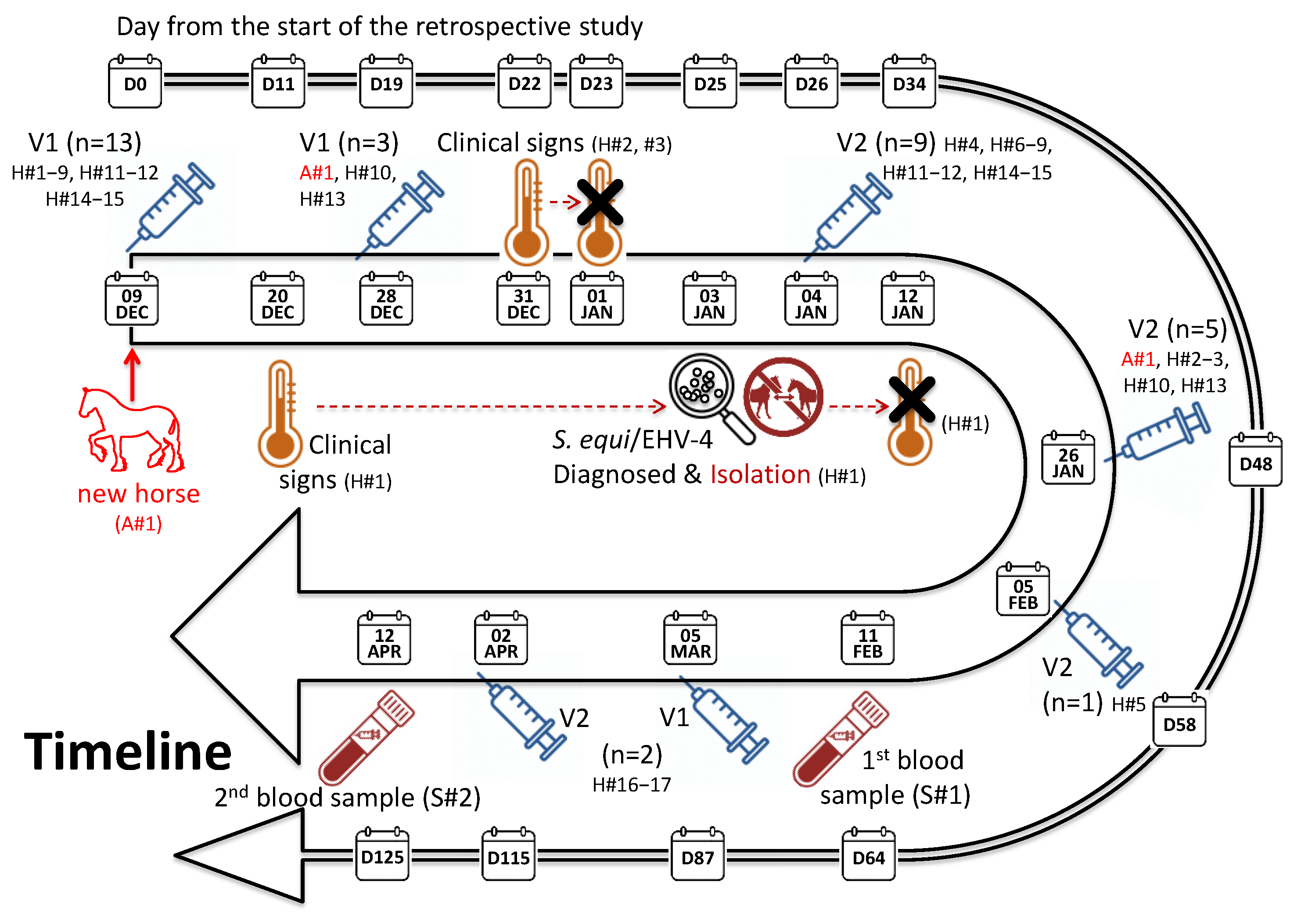
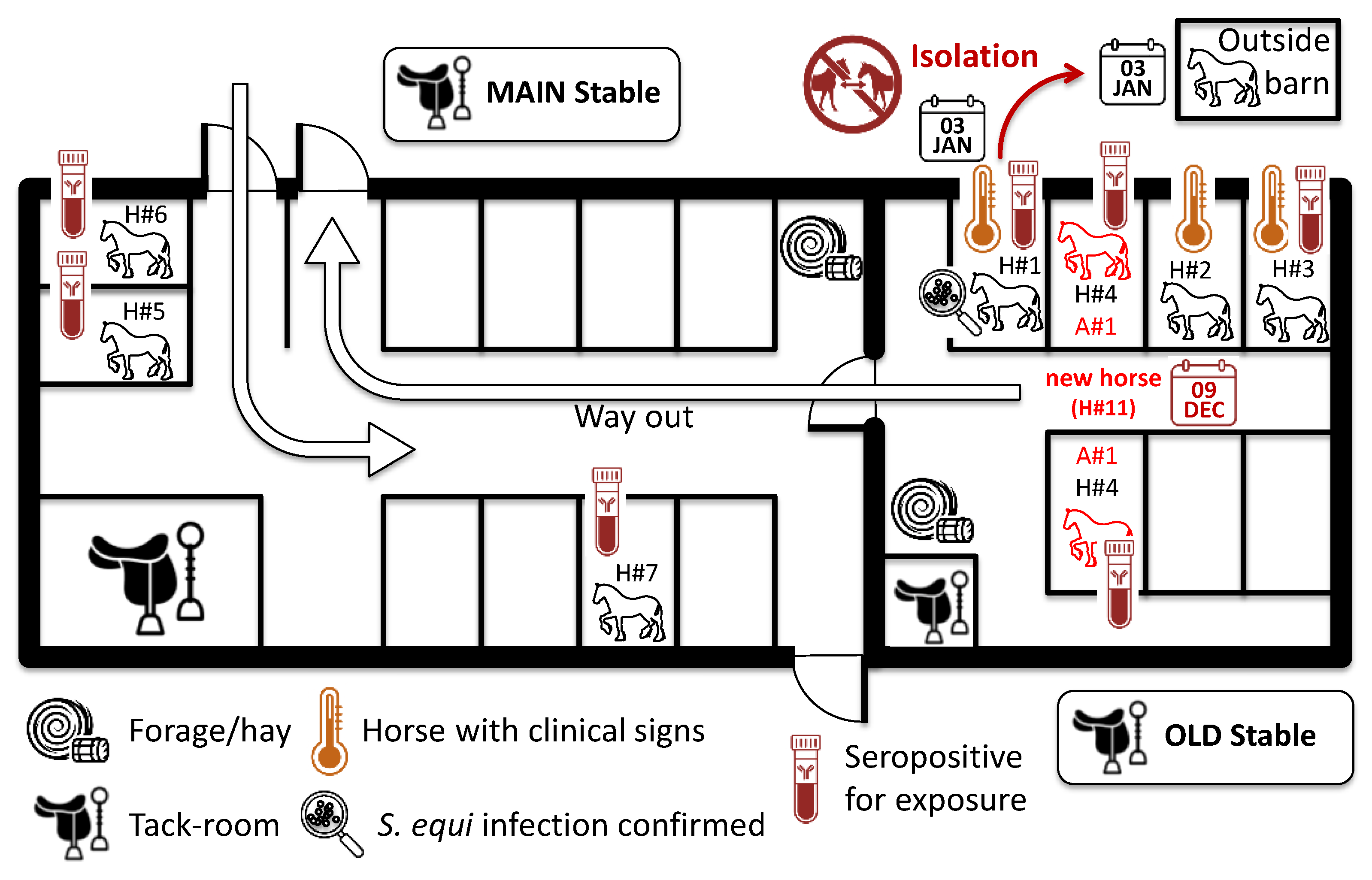
2.1. Vaccination, Outbreak Description and Sampling of Horses
2.2. CCE, Eq85 and IdeE Antigen iELISAs
2.3. Dual Antigen A/C iELISA
2.4. Statistical Methods
3. Results
3.1. Adverse Events After Immunisation
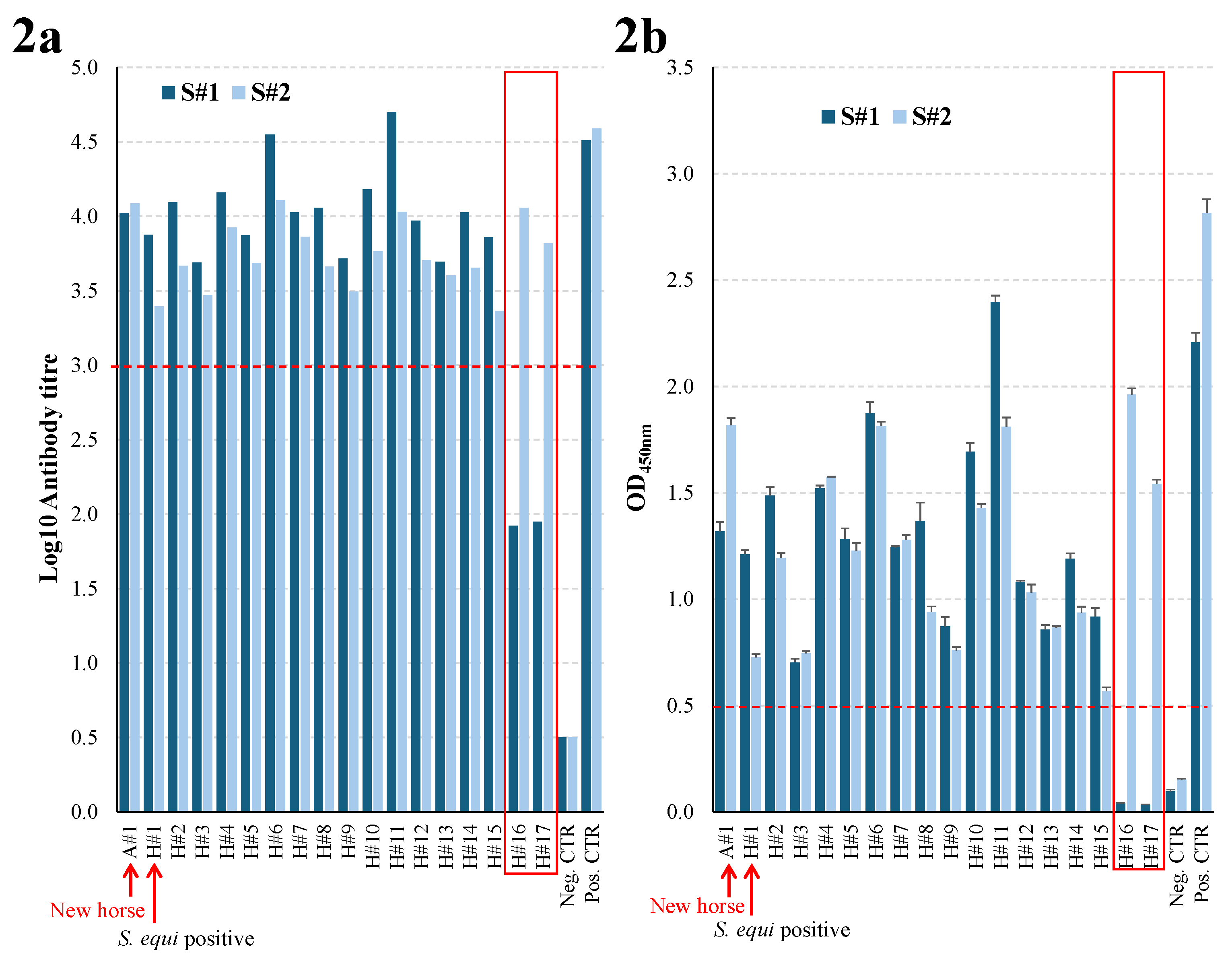
3.2. Serological Response to Vaccination
3.3. Serological Response to S. equi Exposure
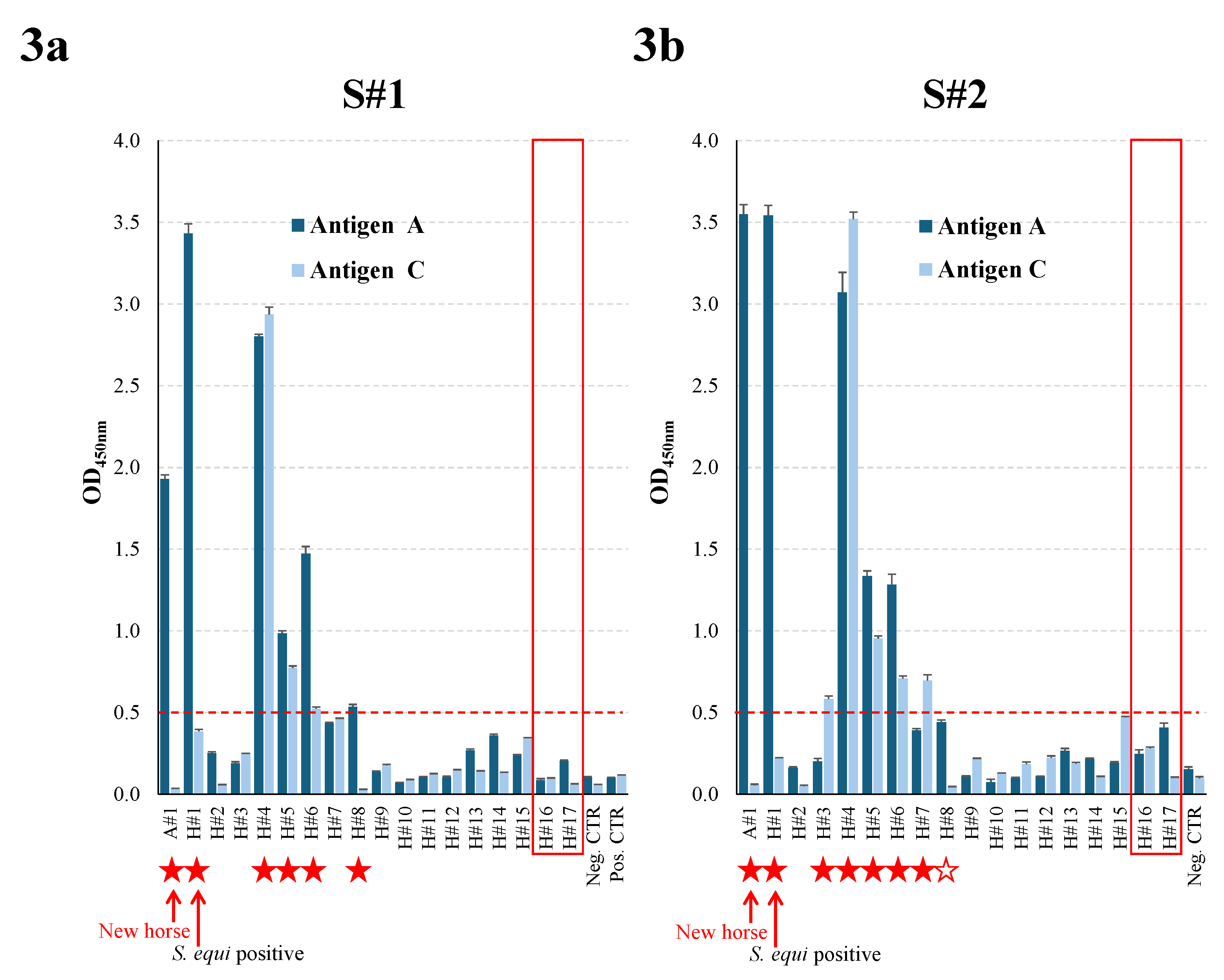
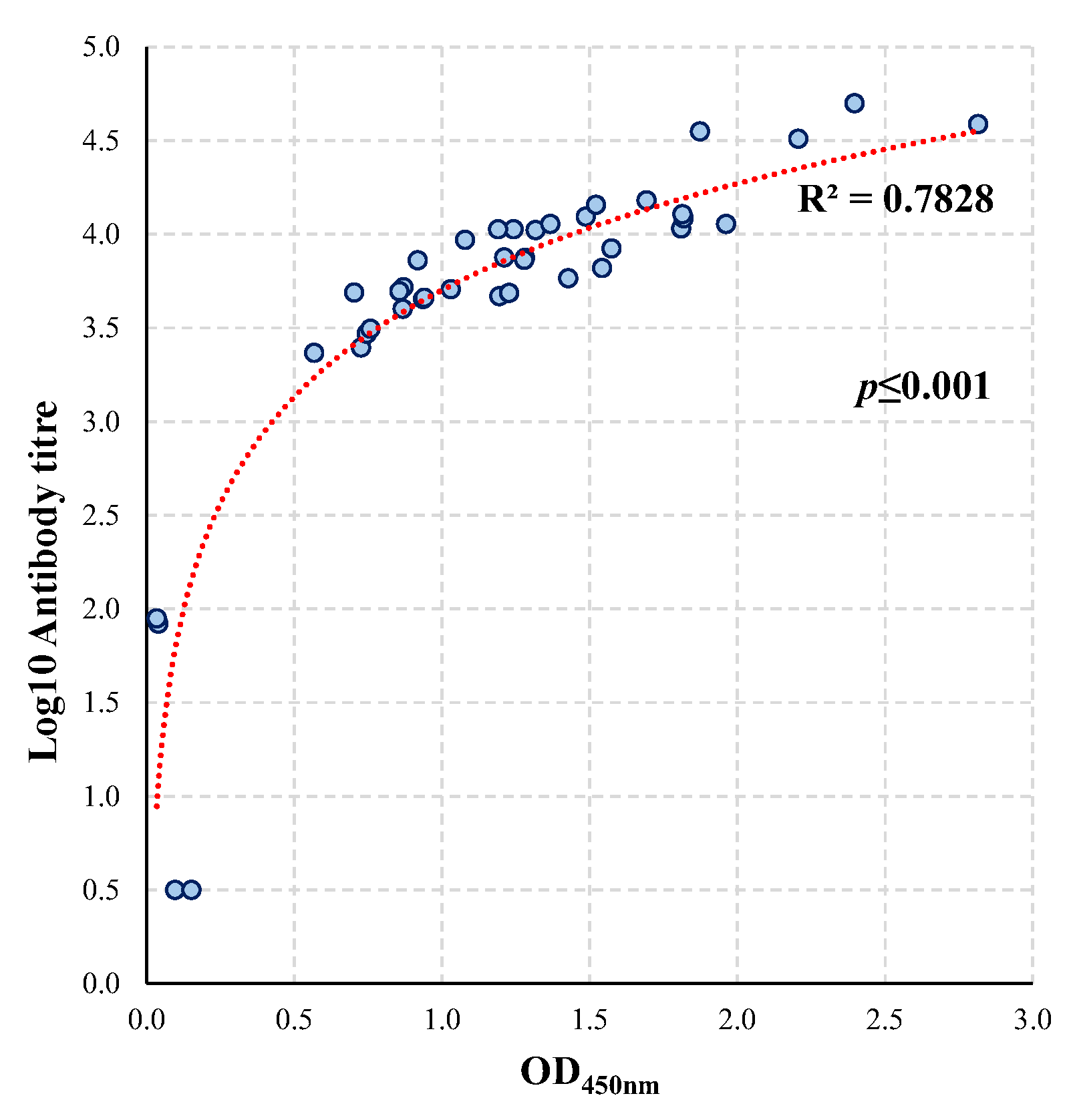
3.4. Comparison of the Conventional and Single Dilution Vaccine Antigen iELISA
4. Discussion
4.1. Possible Route of Introduction of S. equi
4.2. Transmission of S. equi in the Yard
4.3. Impact of Strangles Vaccination
4.4. Co-Circulation of S. equi and EHV-4
4.5. Comparison of the Vaccine Antigens iELISAs
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| DIVA | Differentiation of Infected from Vaccinated Animals |
| EHV-4 | Equine HerpesVirus type 4 |
| EI | Equine Influenza |
| PPID | Pituitary Pars Intermedia Dysfunction |
| S. equi | Streptococcus equi subspecies equi |
| S. zooepidemicus | Streptococcus equi subspecies zooepidemicus |
References
- EIDS (Equine Infectious Diseases Surveillance). Available online: https://equinesurveillance.org/landing/ (accessed on 22 September 2025).
- International Collating Centre (ICC). Available online: https://equinesurveillance.org/iccview/ (accessed on 22 September 2025).
- van Maanen, K.; van den Wollenberg, L.; de Haan, T.; Frippiat, T. Epidemiology of Infectious Pathogens in Horses with Acute Respiratory Disease, Abortion, and Neurological Signs: Insights Gained from the Veterinary Surveillance System for Horses in The Netherlands (SEIN). Veterinary Sciences 2025, 12, 567. [Google Scholar] [CrossRef]
- Rendle, D.; Bowen, M.; Cavalleri, J.; De Brauwere, N.; Grondahl, G.; van Maanen, K.; Newton, J.R. Strangles Vaccination: A Current European Perspective. Equine Veterinary Education 2025, 37, 90–97. [Google Scholar] [CrossRef]
- Surveillance of Equine Strangles (SES). Available online: https://equinesurveillance.org/ses/ (accessed on 24 September 2025).
- Pringle, J.; Storm, E.; Waller, A.; Riihimäki, M. Influence of Penicillin Treatment of Horses with Strangles on Seropositivity to Streptococcus Equi Ssp. Equi-Specific Antibodies. J Vet Intern Med 2020, 34, 294–299. [Google Scholar] [CrossRef]
- Boyle, A. Streptococcus Equi Subspecies Equi Infection (Strangles) in Horses. Compend Contin Educ Vet 2011, 33, E1-7, quiz E8. [Google Scholar]
- Jorm, L.R. Strangles in Horse Studs: Incidence, Risk Factors and Effect of Vaccination. Aust Vet J 1990, 67, 436–439. [Google Scholar] [CrossRef]
- Boyle, A.G.; Timoney, J.F.; Newton, J.R.; Hines, M.T.; Waller, A.S.; Buchanan, B.R. Streptococcus Equi Infections in Horses: Guidelines for Treatment, Control, and Prevention of Strangles-Revised Consensus Statement. J Vet Intern Med 2018, 32, 633–647. [Google Scholar] [CrossRef]
- Christmann, U.; Pink, C. Lessons Learned from a Strangles Outbreak on a Large Standardbred Farm. Equine Veterinary Education 2017, 29, 138–143. [Google Scholar] [CrossRef]
- Sweeney, C.R.; Whitlock, R.H.; Meirs, D.A.; Whitehead, S.C.; Barningham, S.O. Complications Associated with Streptococcus Equi Infection on a Horse Farm. J Am Vet Med Assoc 1987, 191, 1446–1448. [Google Scholar] [CrossRef] [PubMed]
- Osman, S.; Tharwat, M.; Saeed, E. An Outbreak of Strangles in Arabian Horses in Saudi Arabia: Epidemiology, Clinical Signs and Treatment Outcomes. Int J Vet Sci 2021, 10, 323–328. [Google Scholar] [CrossRef]
- Newton, J.R.; Wood, J.L.; Dunn, K.A.; DeBrauwere, M.N.; Chanter, N. Naturally Occurring Persistent and Asymptomatic Infection of the Guttural Pouches of Horses with Streptococcus Equi. Vet Rec 1997, 140, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Newton, J.R.; Verheyen, K.; Talbot, N.C.; Timoney, J.F.; Wood, J.L.; Lakhani, K.H.; Chanter, N. Control of Strangles Outbreaks by Isolation of Guttural Pouch Carriers Identified Using PCR and Culture of Streptococcus Equi. Equine Vet J 2000, 32, 515–526. [Google Scholar] [CrossRef]
- Pringle, J.; Venner, M.; Tscheschlok, L.; Bächi, L.; Riihimäki, M. Long Term Silent Carriers of Streptococcus Equi Ssp. Equi Following Strangles; Carrier Detection Related to Sampling Site of Collection and Culture versus qPCR. Vet J 2019, 246, 66–70. [Google Scholar] [CrossRef]
- Harris, S.R.; Robinson, C.; Steward, K.F.; Webb, K.S.; Paillot, R.; Parkhill, J.; Holden, M.T.G.; Waller, A.S. Genome Specialization and Decay of the Strangles Pathogen, Streptococcus Equi, Is Driven by Persistent Infection. Genome Res 2015, 25, 1360–1371. [Google Scholar] [CrossRef]
- McGlennon, A.A.; Verheyen, K.L.; Newton, J.R.; van Tonder, A.; Wilson, H.; Parkhill, J.; de Brauwere, N.; Frosth, S.; Waller, A.S. Unwelcome Neighbours: Tracking the Transmission of Streptococcus Equi in the United Kingdom Horse Population. Equine Vet J 2025. [Google Scholar] [CrossRef]
- Timoney, J.F. Strangles. Vet Clin North Am Equine Pract 1993, 9, 365–374. [Google Scholar] [CrossRef] [PubMed]
- Robinson, C.; Waller, A.S.; Frykberg, L.; Flock, M.; Zachrisson, O.; Guss, B.; Flock, J.-I. Intramuscular Vaccination with Strangvac Is Safe and Induces Protection against Equine Strangles Caused by Streptococcus Equi. Vaccine 2020, 38, 4861–4868. [Google Scholar] [CrossRef] [PubMed]
- Robinson, C.; Frykberg, L.; Flock, M.; Guss, B.; Waller, A.S.; Flock, J.-I. Strangvac: A Recombinant Fusion Protein Vaccine That Protects against Strangles, Caused by Streptococcus Equi. Vaccine 2018, 36, 1484–1490. [Google Scholar] [CrossRef] [PubMed]
- Robinson, C.; Steward, K.F.; Potts, N.; Barker, C.; Hammond, T.; Pierce, K.; Gunnarsson, E.; Svansson, V.; Slater, J.; Newton, J.R.; et al. Combining Two Serological Assays Optimises Sensitivity and Specificity for the Identification of Streptococcus Equi Subsp. Equi Exposure. Vet J 2013, 197, 188–191. [Google Scholar] [CrossRef]
- Waller, A.; Flock, M.; Smith, K.; Robinson, C.; Mitchell, Z.; Karlström, A.; Lannergård, J.; Bergman, R.; Guss, B.; Flock, J.-I. Vaccination of Horses against Strangles Using Recombinant Antigens from Streptococcus Equi. Vaccine 2007, 25, 3629–3635. [Google Scholar] [CrossRef]
- Rosenthal, J.A. Qualitative Descriptors of Strength of Association and Effect Size. Journal of Social Service Research 1996, 21, 37–59. [Google Scholar] [CrossRef]
- Boyle, A.G.; Smith, M.A.; Boston, R.C.; Stefanovski, D. A Case-Control Study Developing a Model for Predicting Risk Factors for High SeM-Specific Antibody Titers after Natural Outbreaks of Streptococcus Equi Subsp Equi Infection in Horses. J Am Vet Med Assoc 2017, 250, 1432–1439. [Google Scholar] [CrossRef] [PubMed]
- Pringle, J.; Venner, M.; Tscheschlok, L.; Waller, A.S.; Riihimäki, M. Markers of Long Term Silent Carriers of Streptococcus Equi Ssp. Equi in Horses. J Vet Intern Med 2020, 34, 2751–2757. [Google Scholar] [CrossRef]
- HBLB International Code Of Practice 2025 : Strangles - Prevention. Available online: https://codes.hblb.org.uk/index.php/page/103 (accessed on 3 November 2025).
- Ryden, A.; Fernström, L.-L.; Svonni, E.; Riihimäki, M. Effectiveness of Cleaning and Sanitation of Stable Environment and Riding Equipment Following Contamination With Streptococcus Equi Subsp. Equi. J Equine Vet Sci 2023, 121, 104204. [Google Scholar] [CrossRef] [PubMed]
- Weese, J.S.; Jarlot, C.; Morley, P.S. Survival of Streptococcus Equi on Surfaces in an Outdoor Environment. Can Vet J 2009, 50, 968–970. [Google Scholar]
- Grondahl, G.; Righetti, F.; Bjerketorp, J.; Waller, A.S. Reining in Strangles: Absence of Disease in Horses Vaccinated with a DIVA Compatible Recombinant Fusion Protein Vaccine, Following Natural Exposure to Streptococcus Equi Subspecies Equi. Equine Veterinary Journal 2024, 56, 17–18. [Google Scholar] [CrossRef]
- Reemers, S.; Sonnemans, D.; Horspool, L.; van Bommel, S.; Cao, Q.; van de Zande, S. Determining Equine Influenza Virus Vaccine Efficacy—The Specific Contribution of Strain Versus Other Vaccine Attributes. Vaccines 2020, 8, 501. [Google Scholar] [CrossRef]
- Dilai, M.; Piro, M.; El Harrak, M.; Fougerolle, S.; Dehhaoui, M.; Dikrallah, A.; Legrand, L.; Paillot, R.; Fassi Fihri, O. Impact of Mixed Equine Influenza Vaccination on Correlate of Protection in Horses. Vaccines (Basel) 2018, 6, 71. [Google Scholar] [CrossRef]
- Heldens, J.G.M.; Pouwels, H.G.W.; Derks, C.G.G.; Van de Zande, S.M.A.; Hoeijmakers, M.J.H. The First Safe Inactivated Equine Influenza Vaccine Formulation Adjuvanted with ISCOM-Matrix That Closes the Immunity Gap. Vaccine 2009, 27, 5530–5537. [Google Scholar] [CrossRef] [PubMed]
- Wilson, H.J.; Dong, J.; van Tonder, A.J.; Ruis, C.; Lefrancq, N.; McGlennon, A.; Bustos, C.; Frosth, S.; Léon, A.; Blanchard, A.M.; et al. Progressive Evolution of Streptococcus Equi from Streptococcus Equi Subsp. Zooepidemicus and Adaption to Equine Hosts. Microb Genom 2025, 11, 001366. [Google Scholar] [CrossRef]
- Tscheschlok, L.; Venner, M.; Steward, K.; Böse, R.; Riihimäki, M.; Pringle, J. Decreased Clinical Severity of Strangles in Weanlings Associated with Restricted Seroconversion to Optimized Streptococcus Equi Ssp Equi Assays. J Vet Intern Med 2018, 32, 459–464. [Google Scholar] [CrossRef]
- Kelly, C.; Bugg, M.; Robinson, C.; Mitchell, Z.; Davis-Poynter, N.; Newton, J.R.; Jolley, K.A.; Maiden, M.C.J.; Waller, A.S. Sequence Variation of the SeM Gene of Streptococcus Equi Allows Discrimination of the Source of Strangles Outbreaks. J Clin Microbiol 2006, 44, 480–486. [Google Scholar] [CrossRef]
- Cursons, R.; Patty, O.; Steward, K.F.; Waller, A.S. Strangles in Horses Can Be Caused by Vaccination with Pinnacle I. N. Vaccine 2015, 33, 3440–3443. [Google Scholar] [CrossRef]
- Mitchell, C.; Steward, K.F.; Charbonneau, A.R.L.; Walsh, S.; Wilson, H.; Timoney, J.F.; Wernery, U.; Joseph, M.; Craig, D.; van Maanen, K.; et al. Globetrotting Strangles: The Unbridled National and International Transmission of Streptococcus Equi between Horses. Microb Genom 2021, 7, mgen000528. [Google Scholar] [CrossRef]
- Ma, G.; Azab, W.; Osterrieder, N. Equine Herpesviruses Type 1 (EHV-1) and 4 (EHV-4)--Masters of Co-Evolution and a Constant Threat to Equids and Beyond. Vet Microbiol 2013, 167, 123–134. [Google Scholar] [CrossRef] [PubMed]
- Jaramillo-Morales, C.; James, K.; Barnum, S.; Vaala, W.; Chappell, D.E.; Schneider, C.; Craig, B.; Bain, F.; Barnett, D.C.; Gaughan, E.; et al. Voluntary Biosurveillance of Streptococcus Equi Subsp. Equi in Nasal Secretions of 9409 Equids with Upper Airway Infection in the USA. Veterinary Sciences 2023, 10, 78. [Google Scholar] [CrossRef] [PubMed]
- Righetti, F.; Hentrich, K.; Flock, M.; Frosth, S.; Jacobsson, K.; Bjerketorp, J.; Pathak, A.; Ido, N.; Henriques-Normark, B.; Frykberg, L.; et al. Neutralisation of the Immunoglobulin-Cleaving Activity of Streptococcus Equi Subspecies Equi IdeE by Blood Sera from Ponies Vaccinated with a Multicomponent Protein Vaccine. Vaccines (Basel) 2025, 13, 1061. [Google Scholar] [CrossRef] [PubMed]
- Rask, E.; Righetti, F.; Ruiz, A.; Bjerketorp, J.; Frosth, S.; Frykberg, L.; Jacobsson, K.; Guss, B.; Flock, J.-I.; Henriques-Normark, B.; et al. Serological Responses of Horses in Response to Vaccination in a Farm Associated with a Natural Outbreak of Strangles. In Proceedings of the Equine Veterinary Journal; 12th International Equine Infectious Diseases Conference, 30th September - 4th October 2024, Deauville, France, 2024; Vol. 56, S60, pp. 20–20.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



