Submitted:
12 November 2025
Posted:
13 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Certified Systems
2.1. Integrated Production Systems
2.2. Organic Certification
3. Strawberry Cultivation in Soil and Soilless Systems
3.1. Cultivation in Soil
3.2. Soilless Cultivation
3.2.1. Benches
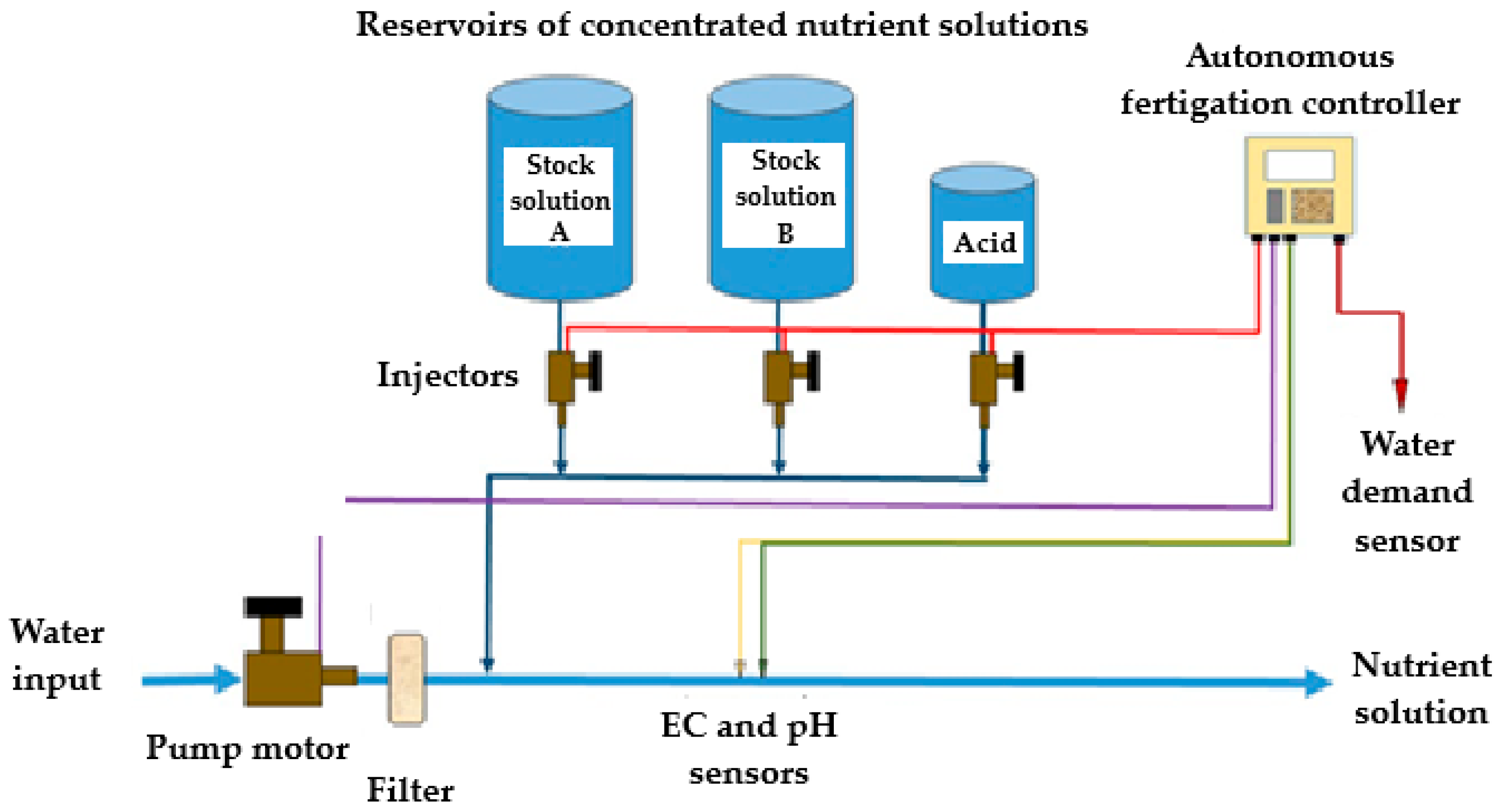
3.2.2. Basic Fertigation System
3.2.3. Substrates
3.2.4. Nutrient Solutions
- Water analysis;
- Calculation of fertilizers;
- Weighing of fertilizers;
- Fill the tank with water to approximately 80–90% of its capacity;
- Adjust the water pH to 5.8–6.2;
- Predilution of each fertilizer in a bucket before addition to the solution tank
- Add the pre-diluted macronutrients to the solution tank except calcium nitrate;
- Add the pre-diluted micronutrients to the solution tank;
- Add the pre-diluted calcium nitrate last to the solution tank;
- Top up with water to reach the final volume;
- Measure and, if necessary, readjust the solution pH to 5.8–6.2;
- Measure the EC and, if necessary, adjust it to the recommended range.
3.2.5. Management of Nutrient Solutions
- In open systems: Replenish the amount of nutrient solution equivalent to the amount used relative to the initial solution, maintaining EC at the initial level.
- In recirculating systems: Replenish the nutrient solution when the EC drops by 0.3 mS cm⁻¹, using 20% of the nutrients from the initial formulation. It is recommended to clean the reservoir and completely replace the nutrient solution at least monthly owing to differential nutrient uptake, changes in water quality, and differential accumulation of nutrients and other elements (e.g., carbonates) in the nutrient solution reservoir. Furlani et al. (2004) [26] recommended that for recirculating systems, complete renewal of the nutrient solution be carried out every 15 days.
3.3. Cultivars and Plantlets
3.4. Basic Crop Management
3.5. Protected Environments
4. General Considerations and Perspectives
Author Contributions
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Faostat. Food and Agriculture Organization of the United Nations. Crops and livestock products. https://www.fao.org/faostat/en/#data/QCL/visualize Accessed on: September 01, 2025.
- Hirakuri, M. H.; Debiasi, H.; Procópio, S. D. O.; Franchini, J. C.; Castro, C. D. Sistemas de produção: conceitos e definições no contexto agrícola. 2012. Londrina: Embrapa Soja, 14.
- Albersmeier, F.; Schulze, H.; Jahn, G.; Spiller, A. The reliability of third-party certification in the food chain: From checklists to risk-oriented auditing. Food Control 2009, 20, 927–935. [Google Scholar] [CrossRef]
- Prieto, L.B.M.; Hata, F.T.; Miguel, A.L.A.; Ferreira, N.Z.B.; Ventura, M.U.; da Costa, A.C.S. Institutionalization and legislation of organic production in Brazil, and how new regulations are impacting farmers. Nativa 2025, 13, 349–359. [Google Scholar] [CrossRef]
- Calegario, F.; Pereira, S.; Hammes, V.; Buschinelli, C.D.A.; Tanure, J.; Morandi, M.; Elisangeles Baptista de Souza, F.A.E.P. Produção integrada de morango (PIMo) como modelo de implementação de sistema de produção sustentável. In Agricultura e Meio Ambiente: A Busca pela Sustentabilidade; Morandi, M.A.B., Packer, A.P.C., Mendes, R., Tanure, J.P.M., Andrade, C.A., Menezes, C., Eds.; Embrapa Meio Ambiente: Brasília, DF, Brazil, 2024; pp. 169–207. [Google Scholar]
- Menezes Júnior, F.O.G. As boas práticas agrícolas e a produção integrada: Diferenças e indicações gerais de procedimentos das BPAs. In: Menezes Júnior, F.O.G.; Marcuzzo, L.L. (Eds.) Manual de Boas Práticas Agrícolas: Guia para a Sustentabilidade das Lavouras de Cebola do Estado de Santa Catarina; Epagri: Florianópolis, Brazil, 2016; 143 p.
- Galina, J.; Ilha, L.; Pagnoncelli, J. Cultivo orgânico do morangueiro em substrato. Cad. Agroecol. 2013, 8, 1–5. [Google Scholar]
- Hata, F.T.; Ventura, M.U.; de Souza, M.S.D.J.; de Sousa, N.V.; Oliveira, B.G.; da Silva, J.B. Mineral and organic fertilization affects Tetranychus urticae, pseudofruit production and leaf nutrient content in strawberry. Phytoparasitica 2019, 47, 513–521. [Google Scholar] [CrossRef]
- Hata, F.T.; Ventura, M.U.; Carvalho, M.G.; Miguel, A.L.; Souza, M.S.; Paula, M.T.; Zawadneak, M.A. Intercropping garlic plants reduces Tetranychus urticae in strawberry crop. Exp. Appl. Acarol. 2016, 69, 311–321. [Google Scholar] [CrossRef] [PubMed]
- Hata, F.T.; Ventura, M.U.; Béga, V.L.; Camacho, I.M.; de Paula, M.T. Chinese chives and garlic in intercropping in strawberry high tunnels for Neopamera bilobata Say (Hemiptera: Rhyparochromidae) control. Bull. Entomol. Res. 2019, 109, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Araujo, E.S.; Benatto, A.; Mirás-Avalos, J.M.; Rogoski, T.; Oelke, S.F.; Schussler, M.; Zawadneak, M.A.C. Acarofauna present in organic strawberry fields and associated weed species in southern Brazil. Exp. Appl. Acarol. 2022, 86, 91–115. [Google Scholar] [CrossRef] [PubMed]
- Hata, F.T.; Togni, P.H.; Ventura, M.U.; da Silva, J.E.P.; Ferreira, N.Z.; Constantino, L. Diverse non-crop vegetation assemblages as banker plants for predatory mites in strawberry crop. Bull. Entomol. Res. 2022, 112, 389–398. [Google Scholar] [CrossRef]
- Busuulwa, A.; Revynthi, A.M.; Liburd, O.E.; Lahiri, S. Banker plant efficacy to boost natural predators for management of field populations of Scirtothrips dorsalis Hood (Thysanoptera: Thripidae) in strawberries. Insects 2024, 15, 776. [Google Scholar] [CrossRef]
- Brasil. Ministério da Agricultura, Pecuária e Abastecimento. Portaria Nº 52, de 15 de março de 2021. Estabelece o Regulamento Técnico para os Sistemas Orgânicos de Produção e as listas de substâncias e práticas para o uso nos Sistemas Orgânicos de Produção; MAPA: Brasília, DF, Brazil, 2021.
- Resh, H.M. Hydroponic Food Production, 5th ed.; Woodbridge Press: Santa Barbara, CA, USA, 1996; 527 p. [Google Scholar]
- Furlani, P.R.; Bolonhezi, D.; Silveira, L.C.P.; Faquin, V. Nutrição mineral de hortaliças, preparo e manejo de soluções nutritivas. Informe Agropecuário. 1999, 20, 90–98.
- Menezes Júnior, F.O.G.; Vieira Neto, J.; Sousa Resende, R. Produção de cultivares de morangueiro em sistema semihidropônico sob diferentes substratos e densidades populacionais. Rev. Thema 2018, 15, 79–92. [Google Scholar] [CrossRef]
- Menezes Júnior, F.O.G.; Souza, A.G.; Saraiva, F.R.S. Qualidade dos frutos de cultivares de morangueiro submetidos a pulverização com nutrientes em sistema semi-hidropônico. Vértices 2023, 1, 1–20.
- Abad, M.B.; Noguera, P.M. Sustratos para el cultivo sin suelo y fertirrigación. In Cadahia, C. (Ed.) Fertirrigación; Mundi-Prensa: Madrid, Spain, 1998; pp. 287–342. [Google Scholar]
- Menezes Júnior, F.O.G. Caracterização de Diferentes Substratos e Seu Efeito na Produção de Mudas de Alface e Couve-flor em Ambiente Protegido; M.Sc. Dissertation, Universidade Federal de Pelotas (UFPel), Pelotas, Brazil, 1998; p. 142. [Google Scholar]
- Abad, M.B.; Martínez, P.F.; Martínez, M.D.; Martínez, J. Evaluación agronómica de los sustratos de cultivo. Actas de Horticultura 1993, 11, 141–154. [Google Scholar]
- Martinez, H.E.P. Formulação de Soluções Nutritivas para Cultivos Hidropônicos Comerciais; FUNEP: Jaboticabal, Brazil, 1997; 31 p. [Google Scholar]
- Kämpf, A.N. Seleção de materiais para uso como substrato. In Substratos para Plantas: A Base da Produção Vegetal em Recipientes; Kämpf, A.N., Fermino, M.H., Eds.; Gênesis: Porto Alegre, Brazil, 2000; pp. 139–145. [Google Scholar]
- Lejeune, J.P.; Balestrazzi, E. L’importanza dell’acqua nella coltura idroponica. L’informatore Agrario 1992, 34, 71–75.
- Böhme, M. Effects of closed systems in substrate culture for vegetable production in greenhouses. Acta Hortic. 1995, 396, 45–54. [Google Scholar] [CrossRef]
- Furlani, P.R.; Fernandes Júnior, F. Cultivo hidropônico de morango em ambiente protegido. In Raseira, M.C.B.; Antunes, L.E.C., Trevisan, R., Eds.; Dias, E.G. (Eds.) 2º Simpósio Nacional do Morango – Palestras; Embrapa Clima Temperado: Pelotas, Brazil, 2004; pp. 102–114. [Google Scholar]
- Moraes, C.A.G.; Furlani, P.R. Cultivo de hortaliças de pseudofruto em hidroponia. Informe Agropecuário 1999, 20, 105–113.
- Gonçalves, M.A.; Vignolo, G.K.; Antunes, L.E.C.; Reisser Júnior, C. Produção de morango fora do solo. Embrapa Clima Temperado: Pelotas, Brazil, 2016; 32 p. (Documentos No. 410).
- Moraes, C.A.G. Hidroponia: Como cultivar tomates em sistema NFT; DISQ Editora: Jundiaí, Brazil, 1997; p. 147. [Google Scholar]
- Menezes Júnior, F.O.G.; Neto, J.V. Avaliação de cultivares de morangueiro dias neutros ‘Albion’ e ‘San Andreas’ sob cultivo semi-hidropônico no Alto Vale do Itajaí–SC. Rev. Thema 2019, 16(4), 845–854. [Google Scholar]
- Bernard, M. F.; Schiavon, A. V.; Alves, A. da S.; Bonow, S.; Herter, F. G.; Cantillano, R. F. F.; Antunes, L. E. C.. Effect of planting date on the yield and quality of Brazilian strawberry cultivars. Semina: Ciências Agrárias, 2025 (in press).
- Aragón-Ramírez, L.A.; Castillo-González, A.M.; Valdez-Aguilar, L.A.; del Rosario García-Mateos, M.; Ybarra-Moncada, M.C.; Avitia-García, E.; Fitz-Rodríguez, E. Quality of strawberry (Fragaria × ananassa Duch.) fruits from plants cultivated under colored shade nets. Agrociencia 2025, 59, 1–14.
- Wang, F.; Wang, J.; Ji, G.; Kang, X.; Li, Y.; Hu, J.; Wang, S. Regulation of anthocyanins and quality in strawberries based on light quality. Horticulturae 2025, 11, 377. [Google Scholar] [CrossRef]
- Menzel, C.M. A review of strawberry under protected cultivation: yields are higher under tunnels than in the open field. J. Hortic. Sci. Biotechnol. 2025, 100, 286–313. [Google Scholar] [CrossRef]
- Cui, M.; Pham, M. D.; Hwang, H.; Chun, C. Flower development and fruit malformation in strawberries after short-term exposure to high or low temperature. Sci. Hortic. 2021, 288, 110308. [Google Scholar] [CrossRef]
- Sierra-Almeida, A.; Morales, L. V.; Guerrero, D.; Hasbún, R. J.; Retamal, L.; Garrido-Bigotes, A.; Tamburrino, I.; Maruri, A. Thermal Vulnerability and Potential Cultivation Areas of Four Day-Neutral Strawberries in Chile: Implications for Climate Adaptation. Plants 2025, 14, 3205. [Google Scholar] [CrossRef]
- Nascimento, D. A.; Gomes, G. C.; de Oliveira, L. V. B.; de Paula Gomes, G. F.; Ivamoto-Suzuki, S. T.; Ziest, A. R.; Mariguele, K. H.; Roberto, S. R.; de Resende, J. T. V. Adaptability and stability analyses of improved strawberry genotypes for tropical climate. Horticulturae 2023, 9, 643. [Google Scholar] [CrossRef]
- Aarif, K.O., M.; Alam, A.; Hotak, Y. Smart sensor technologies shaping the future of precision agriculture: Recent advances and future outlooks. J. Sens. 2025, 2025, 2025, 2460098. [Google Scholar]
- Shahab, H.; Naeem, M.; Iqbal, M.; Aqeel, M.; Ullah, S. S. IoT-driven smart agricultural technology for real-time soil and crop optimization. Smart Agr. Technol. 2025, 10, 100847. [Google Scholar]
- Soussi, A.; Zero, E.; Sacile, R.; Trinchero, D.; Fossa, M. Smart sensors and smart data for precision agriculture: a review. Sensors 2024, 24, 2647. [Google Scholar] [CrossRef] [PubMed]
- Fuentes-Peñailillo, F.; Gutter, K.; Vega, R.; Silva, G. C. New generation sustainable technologies for soilless vegetable production. Horticulturae 2024, 10, 49. [Google Scholar] [CrossRef]




| Physical characteristic | Reference value |
|---|---|
| Dry density | 350 to 500 kg m-3 |
| Wet density | 500 to 750 kg m-3 |
| Total porosity or total pore space | 85 % |
| Aeration space | 20 to 30 % |
| Available water | 24 to 40 % |
| Readily available water | 20 to 30 % |
| Reserve or buffering water | 4 to 10 % |
| Residual or hardly available water | 20 to 30 % |
| Determination | Suitable (mg L-1) |
Maximum Value (mg L-1) |
|---|---|---|
| Ca | 260.7 | 561.4 |
| Mg | < 12 | 12 |
| Na | 20 | 60 |
| SO4 | 26.6 | 66.8 |
| Carbonate | < 244 | 244 |
| Bicarbonate | 19.2 | 79.2 |
| Chloride | 40.5 | 101.5 |
| Fe | <1.12 | 1.12 |
| B | <0.27 | 0.27 |
| Zn | <0.32 | 0.32 |
| Cu | <0.06 | 0.06 |
| Mn | <0.24 | 0.24 |
| F | <0.47 | 0.47 |
| Cl | < 5 | 5 |
| Salt concentration | 1.12 | 200 or 350* |
| Electrical Conductivity (mS cm-1) | <0.75 | 0.5 a 2 |
| pH | 6.5 | 7.5 |
| Variable | Reference Level Range | ||||
|---|---|---|---|---|---|
| EC of irrigation water (dS m-1) | 0-0.5 | 0,5-1.0 | 1,0-2.5 | 2,5-3.5 | >3.5 |
| Recommended drainage (%) | 10-20 | 20-35 | 25-40 | 40-70 | >70 |
| Salt or Fertilizer | Supplied Nutrient | Concentration | EC (0.1% solution) |
Amount Required to Prepare 1 mg L⁻¹ of Each Nutrient | Equivalent Weight |
|---|---|---|---|---|---|
| % | mS | g 1,000 L-1 | |||
| Potassium nitrate (13-00-44) | K N-NO3 |
36.5 | 1.30 | 2.74 7.69 |
101 |
| Calcium nitrate | Ca N-NO3 N -NH4 |
19 14.5 1 |
≈1.20 | 5.26 6.90 100 |
118 |
| Magnesium nitrate | Mg N-NO3 |
9 11 |
0.9 | 11.1 9.1 |
128 |
| Monoammonium phosphate (MAP) | N -NH4 P |
11-12 26 |
≈1.00 | 9.09 – 9.92 3.91 |
115 |
| Ammonium nitrate | N-NO3 N -NH4 |
16.5 16.5 |
1.50 | 6.06 6.06 |
80 |
| Monopotassium phosphate (MKP) | K P |
29 23 |
0.70 | 3.45 4.35 |
136 |
| Potassium chloride (white) | K Cl |
52 47 |
1.70 | 1.92 2.13 |
75 |
| Potassium sulfate | K S |
41 17 |
1.20 | 2.44 5.88 |
87 |
| Magnesium sulfate | Mg S |
10 13 |
0.90 | 10 7.69 |
118 |
| Phosphoric acid 85%, D=1.70 | P | 27 | 1.00 | 3.7 (2.18 mL) | |
| Nitric acid 53%, D=1.33 | N-NO3 | 15.6 | 1.0 | 6.4 mL | |
| Ammonium sulfate | N-NH4 | 20 | 2.1 | 5.0 | 66 |
| Calcium chloride | Ca | 24 | 1.3 | 4.2 | 56 |
| Salt or Fertilizer | Supplied Nutrient | Concentration | Amount required to prepare 1 mg L⁻¹ of each nutrient |
|---|---|---|---|
| % | g 1000 L-1 | ||
| Fe-EDTA | Fe | 13 | 0.77 |
| Fe-EDDHA | Fe | 6 | 1.67 |
| Fe-EDDHMA | Fe | 6 | 1.67 |
| Fe-DTPA | Fe | 11 | 0.91 |
| Boric acid | B | 17 | 0.59 |
| Borax | B | 11 | 0.91 |
| Copper sulfate | Cu | 23 | 0.43 |
| Cu-EDTA | Cu | 14.5 | 0.69 |
| Manganese sulfate | Mn | 26 | 0.38 |
| Manganese chloride | Mn | 27 | 0.37 |
| Mn-EDTA | Mn | 13 | 0.77 |
| Zinc sulfate | Zn | 22 | 0.45 |
| Zinc chloride | Zn | 45 | 0.22 |
| Zn-EDTA | Zn | 14 | 0.71 |
| Sodium molybdate | Mo | 39 | 0.26 |
| Ammonium molybdate | Mo | 54 | 0.19 |
| Molybdic acid | Mo | 66 | 0.15 |
| Fertilizers | Vegetative stage | Fruiting stage |
|---|---|---|
| Fertilizer amount (g) for 1,000 liters of water | ||
| Calcium nitrate (15.5-0-0) | 900 | 1,060 |
| Potassium nitrate (12-0-45) | 650 | 800 |
| Monoammonium phosphate (11-60-0) | 240 | 240 |
| Monopotassium phosphate (0-51-33) | 0 | 0 |
| Magnesium sulfate | 400 | 400 |
| Boric acid | 1.5 | 3.5 |
| Copper sulfate | 0.25 | 0.25 |
| Manganese sulfate | 2.5 | 2.5 |
| Zinc sulfate | 1.0 | 1.0 |
| Sodium molybdate | 0.1 | 0.1 |
| Ferrous sulfate | 8.0 | 8.0 |
| Substrate with pH 5.2–5.5 | Substrate with pH above 7.0 | |||
|---|---|---|---|---|
| Fertilizer1 | Vegetative stage | Fruiting stage | Vegetative stage | Fruiting stage |
| Calcium nitrate (15.5-00-00) | 480 g | 480 g | 480 g | 480 g |
| Potassium nitrate (12-00-45) | 300 g | 300 g | 300 g | 180 g |
| Monoammonium phosphate (11-60-00) | 90 g | - | - | - |
| Monopotassium phosphate (00-51-33) | 108 g | 216 g | - | - |
| Magnesium sulfate (00-00-00-09) | 360 g | 360 g | 360 g | 360 g |
| Ammonium sulfate (20-00-00) | - | - | 50 g | 70 g |
| Potassium sulfate (00-00-50) | - | - | 70 g | 260 g |
| Phosphoric acid (85%) | - | - | 110 mL | 110 mL |
| Boric acid (17%B) | 1.8 g | 1.8 g | 1.8 g | 1.8 g |
| Copper sulfate (25%Cu) | 0.18 g | 0.18 g | 0.18 g | 0.18 g |
| Manganese sulfate (25%Mn) | 1.2 g | 1.2 g | 1.2 g | 1.2 g |
| Zinc sulfate (20%Zn) | 0.6 g | 0.6 g | 0.6 g | 0.6 g |
| Sodium molybdate (39%Mo) | 0.18 g | 0.18 g | 0.18 g | 0.18 g |
| Chelated iron (6% Fe) | 36 g | 36 g | 36 g | 36 g |
| Salts or fertilizers | A | B | C |
|---|---|---|---|
| g per 10 L of water | |||
| Calcium nitrate | 1,600 | 0 | 0 |
| Potassium nitrate | 0 | 1,000 | 1,000 |
| Monoammonium phosphate | 0 | 300 | 0 |
| Monopotassium phosphate | 0 | 360 | 720 |
| Magnesium sulfate | 0 | 1,200 | 1,200 |
| Boric acid | 6.0 | 0 | |
| Copper sulfate | 0.6 | 0 | |
| Manganese sulfate | 4.0 | 0 | |
| Zinc sulfate | 2.0 | 0 | |
| Sodium molybdate | 0.6 | 0 | |
| Iron chelate (6% Fe) | 120 | 0 | |
| Plant stage | Concentrated solution | ||
|---|---|---|---|
| A | B | C | |
| liters per 1,000 liters of water | |||
| Vegetative stage | 3.0 | 3.0 | - |
| Fruiting stage | 3.0 | - | 3.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




