Submitted:
12 November 2025
Posted:
13 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Comparative Analysis of Green Extraction Techniques of Bioactive Compounds
2.1. Ultrasound-Assisted Extraction
2.2. Pulsed Electric Field
2.3. Microwave-Assisted Extraction
2.4. Enzyme-Assisted Extraction
2.5. Subcritical Water Extraction
2.6. Natural Deep Eutectic Solvents
2.7. Hydrodynamic Cavitation
2.8. Summary of Extraction Techniques
3. Bioactive Compounds: In Vivo, Ex Vivo and Clinical Evidence
3.1. Orange Peel Extracts
3.2. Pomegranate Peel Extracts
3.3. Abies Alba Extracts
4. Direct Blending of DPI with HC-Based Bioactive Extracts
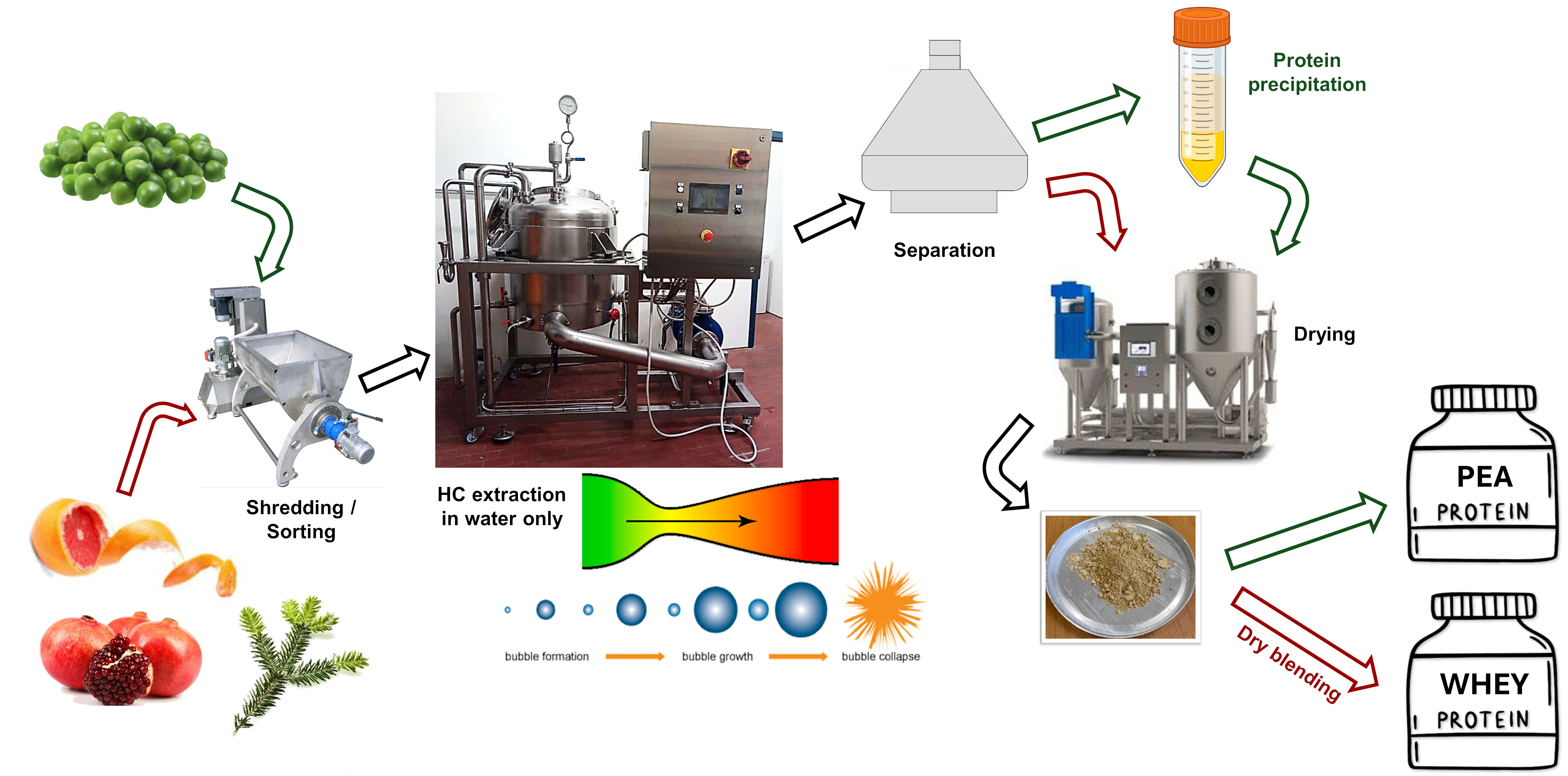
5. HC-Based Extraction of Vegetable Proteins
- Tuning pH/ionic strength to tackle phytate without harming digestibility.
- Operating at moderate temperatures and solid loadings that preserve proteins while protecting flavor and lipids.
- Designing cascaded HC lines that co-valorize proteins, polysaccharides and fibers from the same feedstock.
6. HC-Based Proteins-Polyphenols Conjugation
6.1. Early Evidence of HC-Driven Protein–Polyphenol Conjugation
6.2. Added Functionality of Protein–Polyphenol Conjugates
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ANF | Anti-nutritional factors |
| COGS | Cost of goods sold |
| CPW | Citrus peel waste |
| DPI | Dry protein isolate |
| EAE | Enzyme-assisted extraction |
| GEP | Green extraction principles |
| HC | Hydrodynamic cavitation |
| HPP | High-pressure processing |
| MAE | Microwave-assisted extraction |
| NADES | Natural Deep Eutectic Solvents |
| OPEX | Operating expenditure |
| PEF | Pulsed electric field |
| SPI | Soy protein isolate |
| SWE | Subcritical water extraction |
| TBARS | Thiobarbituric Acid-Reactive Substance |
| UAE | Ultrasound-assisted extraction |
| WPI | Whey protein isolate |
References
- Andrade, M.A.; Barbosa, C.H.; Shah, M.A.; Ahmad, N.; Vilarinho, F.; Khwaldia, K.; Silva, A.S.; Ramos, F. Citrus By-Products: Valuable Source of Bioactive Compounds for Food Applications. Antioxidants 2023, 12, 38. [Google Scholar] [CrossRef]
- Meneguzzo, F.; Brunetti, C.; Fidalgo, A.; Ciriminna, R.; Delisi, R.; Albanese, L.; Zabini, F.; Gori, A.; Nascimento, L.B. dos S.; Carlo, A. De; et al. Real-Scale Integral Valorization of Waste Orange Peel via Hydrodynamic Cavitation. Processes 2019, 7, 581. [Google Scholar] [CrossRef]
- Cano-Lamadrid, M.; Martínez-Zamora, L.; Castillejo, N.; Artés-Hernández, F. From Pomegranate Byproducts Waste to Worth: A Review of Extraction Techniques and Potential Applications for Their Revalorization. Foods 2022, 11. [Google Scholar] [CrossRef]
- Tienaho, J.; Liimatainen, J.; Myllymäki, L.; Kaipanen, K.; Tagliavento, L.; Ruuttunen, K.; Rudolfsson, M.; Karonen, M.; Marjomäki, V.; Hagerman, A.E.; et al. Pilot Scale Hydrodynamic Cavitation and Hot-Water Extraction of Norway Spruce Bark Yield Antimicrobial and Polyphenol-Rich Fractions. Sep Purif Technol 2024, 360, 130925. [Google Scholar] [CrossRef]
- Šafranko, S.; Šubarić, D.; Jerković, I.; Jokić, S. Citrus By-Products as a Valuable Source of Biologically Active Compounds with Promising Pharmaceutical, Biological and Biomedical Potential. Pharmaceuticals 2023, 16, 1081. [Google Scholar] [CrossRef]
- Flori, L.; Albanese, L.; Calderone, V.; Meneguzzo, F.; Pagliaro, M.; Ciriminna, R.; Zabini, F.; Testai, L. Cardioprotective Effects of Grapefruit IntegroPectin Extracted via Hydrodynamic Cavitation from By-Products of Citrus Fruits Industry: Role of Mitochondrial Potassium Channels. Foods 2022, 11, 2799. [Google Scholar] [CrossRef] [PubMed]
- Nuzzo, D.; Scordino, M.; Scurria, A.; Giardina, C.; Giordano, F.; Meneguzzo, F.; Mudò, G.; Pagliaro, M.; Picone, P.; Attanzio, A.; et al. Protective, Antioxidant and Antiproliferative Activity of Grapefruit Integropectin on Sh-Sy5y Cells. Int J Mol Sci 2021, 22, 9368. [Google Scholar] [CrossRef]
- Vesci, L.; Martinelli, G.; Liu, Y.; Tagliavento, L.; Dell’Agli, M.; Wu, Y.; Soldi, S.; Sagheddu, V.; Piazza, S.; Sangiovanni, E.; et al. The New Phytocomplex AL0042 Extracted from Red Orange By-Products Inhibits the Minimal Hepatic Encephalopathy in Mice Induced by Thioacetamide. Biomedicines 2025, 13, 686. [Google Scholar] [CrossRef] [PubMed]
- Pozzo, L.; Raffaelli, A.; Ciccone, L.; Zabini, F.; Vornoli, A.; Calderone, V.; Testai, L.; Meneguzzo, F. Conifer By-Products Extracted Using Hydrodynamic Cavitation as a Convenient Source of Phenolic Compounds and Free Amino Acids with Antioxidant and Antimicrobial Properties. Molecules 2025, 30, 2722. [Google Scholar] [CrossRef]
- Yusuf, H.; Fors, H.; Galal, N.M.; Elhabashy, A.E.; Melkonyan, A.; Harraz, N. Barriers to Implementing Circular Citrus Supply Chains: A Systematic Literature Review. J Environ Manage 2025, 373, 123963. [Google Scholar] [CrossRef]
- Hussain, A.; Gulbadan Dar, N.; Paracha, G.M.; Akhter, S. Evaluation of Different Techniques for Extraction of Antioxidants as Bioactive Compounds from Citrus Peels (Industrial by Products). J. Agric. & Environ. Sci 2015, 15, 676–682. [Google Scholar]
- Moreira, M.M.; Morais, S.; Delerue-Matos, C. Environment-Friendly Techniques for Extraction of Bioactive Compounds From Fruits. In Soft Chemistry and Food Fermentation; Elsevier, 2017; pp. 21–47.
- Arya, S.S.; More, P.R.; Ladole, M.R.; Pegu, K.; Pandit, A.B. Non-Thermal, Energy Efficient Hydrodynamic Cavitation for Food Processing, Process Intensification and Extraction of Natural Bioactives: A Review. Ultrason Sonochem 2023, 98, 106504. [Google Scholar] [CrossRef] [PubMed]
- Meneguzzo, F.; Zabini, F. Industrialization of Hydrodynamic Cavitation in Plant Resource Extraction. Curr Opin Chem Eng 2025, 48, 101140. [Google Scholar] [CrossRef]
- Novel Food Available online: https://www.efsa.europa.eu/en/topics/topic/novel-food.
- Chemat, F.; Rombaut, N.; Sicaire, A.-G.; Meullemiestre, A.; Fabiano-Tixier, A.-S.; Abert-Vian, M. Ultrasound Assisted Extraction of Food and Natural Products. Mechanisms, Techniques, Combinations, Protocols and Applications. A Review. Ultrason Sonochem 2017, 34, 540–560. [Google Scholar] [CrossRef]
- Nayak, B.; Dahmoune, F.; Moussi, K.; Remini, H.; Dairi, S.; Aoun, O.; Khodir, M. Comparison of Microwave, Ultrasound and Accelerated-Assisted Solvent Extraction for Recovery of Polyphenols from Citrus Sinensis Peels. Food Chem 2015, 187, 507–516. [Google Scholar] [CrossRef]
- Peiró, S.; Luengo, E.; Segovia, F.; Raso, J.; Almajano, M.P. Improving Polyphenol Extraction from Lemon Residues by Pulsed Electric Fields. Waste Biomass Valorization 2019, 10, 889–897. [Google Scholar] [CrossRef]
- Meneguzzo, F.; Albanese, L. Intensification of the Dimethyl Sulfide Precursor Conversion Reaction: A Retrospective Analysis of Pilot-Scale Brewer’s Wort Boiling Experiments Using Hydrodynamic Cavitation. Beverages 2025, 11, 22. [Google Scholar] [CrossRef]
- Ciriminna, R.; Di Liberto, V.; Albanese, L.; Li Petri, G.; Valenza, C.; Angellotti, G.; Meneguzzo, F.; Pagliaro, M. Citrus IntegroPectin: A Family of Bioconjugates With Large Therapeutic Potential. ChemFoodChem 2025. [Google Scholar] [CrossRef]
- Benedetti, G.; Flori, L.; Spezzini, J.; Miragliotta, V.; Lazzarini, G.; Pirone, A.; Meneguzzo, C.; Tagliavento, L.; Martelli, A.; Antonelli, M.; et al. Improved Cardiovascular Effects of a Novel Pomegranate Byproduct Extract Obtained through Hydrodynamic Cavitation. Nutrients 2024, 16, 506. [Google Scholar] [CrossRef]
- Vojvodić, S.; Kobiljski, D.; Srđenović Čonić, B.; Torović, L. Landscape of Herbal Food Supplements: Where Do We Stand with Health Claims? Nutrients 2025, 17, 1571. [Google Scholar] [CrossRef]
- Fernandes, F.A.; Carocho, M.; Prieto, M.A.; Barros, L.; Ferreira, I.C.F.R.; Heleno, S.A. Nutraceuticals and Dietary Supplements: Balancing out the Pros and Cons. Food Funct 2024, 15, 6289–6303. [Google Scholar] [CrossRef]
- Liu, C.; Wang, R.; Wang, T.; Gu, C.; Zhang, L.; Meng, D.; Pan, M.; Yang, R. The Whey-Plant Protein Heteroprotein Systems with Synergistic Properties and Versatile Applications. J Agric Food Chem 2025, 73, 4440–4454. [Google Scholar] [CrossRef] [PubMed]
- Dong, G.; Hu, Z.; Tang, J.; Das, R.S.; Sun, D.W.; Tiwari, B.K. Reducing Anti-Nutritional Factors in Pea Protein Using Advanced Hydrodynamic Cavitation, Ultrasonication, and High-Pressure Processing Technologies. Food Chem 2025, 488, 144834. [Google Scholar] [CrossRef]
- de Souza Mesquita, L.M.; Contieri, L.S.; e Silva, F.A.; Bagini, R.H.; Bragagnolo, F.S.; Strieder, M.M.; Sosa, F.H.B.; Schaeffer, N.; Freire, M.G.; Ventura, S.P.M.; et al. Path2Green: Introducing 12 Green Extraction Principles and a Novel Metric for Assessing Sustainability in Biomass Valorization. Green Chemistry 2024, 26, 10087–10106. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Cabal, J.; Avilés-Betanzos, K.A.; Cauich-Rodríguez, J.V.; Ramírez-Sucre, M.O.; Rodríguez-Buenfil, I.M. Recent Developments in Citrus Aurantium L.: An Overview of Bioactive Compounds, Extraction Techniques, and Technological Applications. Processes 2025, 13, 120. [Google Scholar] [CrossRef]
- Ge, M.; Zhang, G.; Petkovšek, M.; Long, K.; Coutier-Delgosha, O. Intensity and Regimes Changing of Hydrodynamic Cavitation Considering Temperature Effects. J Clean Prod 2022, 338, 130470. [Google Scholar] [CrossRef]
- Acciardo, E.; Tabasso, S.; Cravotto, G.; Bensaid, S. Process Intensification Strategies for Lignin Valorization. Chemical Engineering and Processing - Process Intensification 2022, 171, 108732. [Google Scholar] [CrossRef]
- Chemat, F.; Rombaut, N.; Sicaire, A.-G.; Meullemiestre, A.; Fabiano-Tixier, A.-S.; Abert-Vian, M. Ultrasound Assisted Extraction of Food and Natural Products. Mechanisms, Techniques, Combinations, Protocols and Applications. A Review. Ultrason Sonochem 2017, 34, 540–560. [Google Scholar] [CrossRef]
- Papoutsis, K.; Pristijono, P.; Golding, J.B.; Stathopoulos, C.E.; Bowyer, M.C.; Scarlett, C.J.; Vuong, Q. V. Screening the Effect of Four Ultrasound-Assisted Extraction Parameters on Hesperidin and Phenolic Acid Content of Aqueous Citrus Pomace Extracts. Food Biosci 2018, 21, 20–26. [Google Scholar] [CrossRef]
- Vathsala, V.; Singh, S.P.; Bishnoi, M.; Varghese, E.; Saurabh, V.; Khandelwal, A.; Kaur, C. Ultrasound-Assisted Extraction (UAE) and Characterization of Citrus Peel Pectin: Comparison between Pummelo (Citrus Grandis L. Osbeck) and Sweet Lime (Citrus Limetta Risso). Sustain Chem Pharm 2024, 37. [Google Scholar] [CrossRef]
- Kaur, S.; Panesar, P.S.; Chopra, H.K. Citrus Processing By-Products: An Overlooked Repository of Bioactive Compounds. Crit Rev Food Sci Nutr 2023, 63, 67–86. [Google Scholar] [CrossRef]
- Peiró, S.; Luengo, E.; Segovia, F.; Raso, J.; Almajano, M.P. Improving Polyphenol Extraction from Lemon Residues by Pulsed Electric Fields. Waste Biomass Valorization 2019, 10, 889–897. [Google Scholar] [CrossRef]
- Chatzimitakos, T.; Athanasiadis, V.; Kalompatsios, D.; Mantiniotou, M.; Bozinou, E.; Lalas, S.I. Pulsed Electric Field Applications for the Extraction of Bioactive Compounds from Food Waste and By-Products: A Critical Review. Biomass 2023, 3, 367–401. [Google Scholar] [CrossRef]
- Hwang, H.J.; Kim, H.J.; Ko, M.J.; Chung, M.S. Recovery of Hesperidin and Narirutin from Waste Citrus Unshiu Peel Using Subcritical Water Extraction Aided by Pulsed Electric Field Treatment. Food Sci Biotechnol 2021, 30, 217–226. [Google Scholar] [CrossRef]
- Luengo, E.; Álvarez, I.; Raso, J. Improving the Pressing Extraction of Polyphenols of Orange Peel by Pulsed Electric Fields. Innovative Food Science and Emerging Technologies 2013, 17, 79–84. [Google Scholar] [CrossRef]
- Carpentieri, S.; Režek Jambrak, A.; Ferrari, G.; Pataro, G. Pulsed Electric Field-Assisted Extraction of Aroma and Bioactive Compounds From Aromatic Plants and Food By-Products. Front Nutr 2022, 8. [Google Scholar] [CrossRef] [PubMed]
- García-Martín, J.F.; Feng, C.H.; Domínguez-Fernández, N.M.; Álvarez-Mateos, P. Microwave-Assisted Extraction of Polyphenols from Bitter Orange Industrial Waste and Identification of the Main Compounds. Life 2023, 13. [Google Scholar] [CrossRef]
- Nayak, B.; Dahmoune, F.; Moussi, K.; Remini, H.; Dairi, S.; Aoun, O.; Khodir, M. Comparison of Microwave, Ultrasound and Accelerated-Assisted Solvent Extraction for Recovery of Polyphenols from Citrus Sinensis Peels. Food Chem 2015, 187, 507–516. [Google Scholar] [CrossRef]
- Alvi, T.; Asif, Z.; Iqbal Khan, M.K. Clean Label Extraction of Bioactive Compounds from Food Waste through Microwave-Assisted Extraction Technique-A Review. Food Biosci 2022, 46. [Google Scholar] [CrossRef]
- Patra, A.; Abdullah, S.; Pradhan, R.C. Review on the Extraction of Bioactive Compounds and Characterization of Fruit Industry By-Products. Bioresour Bioprocess 2022, 9. [Google Scholar] [CrossRef]
- Li, B.B.; Smith, B.; Hossain, M.M. Extraction of Phenolics from Citrus Peels: II. Enzyme-Assisted Extraction Method. Sep Purif Technol 2006, 48, 189–196. [Google Scholar] [CrossRef]
- Marić, M.; Grassino, A.N.; Zhu, Z.; Barba, F.J.; Brnčić, M.; Rimac Brnčić, S. An Overview of the Traditional and Innovative Approaches for Pectin Extraction from Plant Food Wastes and By-Products: Ultrasound-, Microwaves-, and Enzyme-Assisted Extraction. Trends Food Sci Technol 2018, 76, 28–37. [Google Scholar] [CrossRef]
- Chávez-González, M.L.; López-López, L.I.; Rodríguez-Herrera, R.; Contreras-Esquivel, J.C.; Aguilar, C.N. Enzyme-Assisted Extraction of Citrus Essential Oil. Chemical Papers 2016, 70, 412–417. [Google Scholar] [CrossRef]
- Durmus, N.; Kilic-Akyilmaz, M. Bioactivity of Non-Extractable Phenolics from Lemon Peel Obtained by Enzyme and Ultrasound Assisted Extractions. Food Biosci 2023, 53. [Google Scholar] [CrossRef]
- Brezo-borjan, T.; Švarc-gaji, J.; Rodrigues, F. Chemical and Biological Characterisation of Orange. Processes 2023, 11, 1766. [Google Scholar] [CrossRef]
- Costa, J.M.; Strieder, M.M.; Saldaña, M.D.A.; Rostagno, M.A.; Forster-Carneiro, T. Recent Advances in the Processing of Agri-Food By-Products by Subcritical Water. Food Bioproc Tech 2023, 16, 2705–2724. [Google Scholar] [CrossRef]
- Ko, M.J.; Kwon, H.L.; Chung, M.S. Pilot-Scale Subcritical Water Extraction of Flavonoids from Satsuma Mandarin (Citrus Unshiu Markovich) Peel. Innovative Food Science and Emerging Technologies 2016, 38, 175–181. [Google Scholar] [CrossRef]
- Kim, D.S.; Lim, S. Bin Kinetic Study of Subcritical Water Extraction of Flavonoids from Citrus Unshiu Peel. Sep Purif Technol 2020, 250. [Google Scholar] [CrossRef]
- Lachos-Perez, D.; Baseggio, A.M.; Mayanga-Torres, P.C.; Maróstica, M.R.; Rostagno, M.A.; Martínez, J.; Forster-Carneiro, T. Subcritical Water Extraction of Flavanones from Defatted Orange Peel. Journal of Supercritical Fluids 2018, 138, 7–16. [Google Scholar] [CrossRef]
- Lachos-Perez, D.; Baseggio, A.M.; Torres-Mayanga, P.C.; Ávila, P.F.; Tompsett, G.A.; Marostica, M.; Goldbeck, R.; Timko, M.T.; Rostagno, M.; Martinez, J.; et al. Sequential Subcritical Water Process Applied to Orange Peel for the Recovery Flavanones and Sugars. Journal of Supercritical Fluids 2020, 160. [Google Scholar] [CrossRef]
- Panić, M.; Andlar, M.; Tišma, M.; Rezić, T.; Šibalić, D.; Cvjetko Bubalo, M.; Radojčić Redovniković, I. Natural Deep Eutectic Solvent as a Unique Solvent for Valorisation of Orange Peel Waste by the Integrated Biorefinery Approach. Waste Management 2021, 120, 340–350. [Google Scholar] [CrossRef]
- Mayanin, I. Evaluation of Polyphenol Profile from Citrus Peel Obtained by Natural Deep Eutectic Solvent / Ultrasound Extraction. Processes 2024, 12, 1–19. [Google Scholar] [CrossRef]
- Gómez-Urios, C.; Viñas-Ospino, A.; Puchades-Colera, P.; López-Malo, D.; Frígola, A.; Esteve, M.J.; Blesa, J. Sustainable Development and Storage Stability of Orange By-Products Extract Using Natural Deep Eutectic Solvents. Foods 2022, 11. [Google Scholar] [CrossRef]
- Ciriminna, R.; Scurria, A.; Pagliaro, M. Natural Product Extraction via Hydrodynamic Cavitation. Sustain Chem Pharm 2023, 33, 101083. [Google Scholar] [CrossRef]
- Arya, S.S.; More, P.R.; Ladole, M.R.; Pegu, K.; Pandit, A.B. Non-Thermal, Energy Efficient Hydrodynamic Cavitation for Food Processing, Process Intensification and Extraction of Natural Bioactives: A Review. Ultrason Sonochem 2023, 98, 106504. [Google Scholar] [CrossRef]
- Meneguzzo, F.; Brunetti, C.; Fidalgo, A.; Ciriminna, R.; Delisi, R.; Albanese, L.; Zabini, F.; Gori, A.; Nascimento, L.B. dos S.; Carlo, A. De; et al. Real-Scale Integral Valorization of Waste Orange Peel via Hydrodynamic Cavitation. Processes 2019, 7, 581. [Google Scholar] [CrossRef]
- Scurria, A.; Sciortino, M.; Garcia, A.R.; Pagliaro, M.; Avellone, G.; Fidalgo, A.; Albanese, L.; Meneguzzo, F.; Ciriminna, R.; Ilharco, L.M. Red Orange and Bitter Orange IntegroPectin: Structure and Main Functional Compounds. Molecules 2022, 27. [Google Scholar] [CrossRef] [PubMed]
- Ciriminna, R.; Angellotti, G.; Li Petri, G.; Meneguzzo, F.; Riccucci, C.; Carlo, G. Di; Pagliaro, M. Cavitation as a Zero-Waste Circular Economy Process to Convert Citrus Processing Waste into Biopolymers in High Demand. Journal of Bioresources and Bioproducts 2024, 9, 486–494. [Google Scholar] [CrossRef]
- Scurria, A.; Sciortino, M.; Albanese, L.; Nuzzo, D.; Zabini, F.; Meneguzzo, F.; Alduina, R.; Presentato, A.; Pagliaro, M.; Avellone, G.; et al. Flavonoids in Lemon and Grapefruit IntegroPectin. ChemistryOpen 2021, 10, 1055–1058. [Google Scholar] [CrossRef] [PubMed]
- Scurria, A.; Sciortino, M.; Presentato, A.; Lino, C.; Piacenza, E.; Albanese, L.; Zabini, F.; Meneguzzo, F.; Nuzzo, D.; Pagliaro, M.; et al. Volatile Compounds of Lemon and Grapefruit IntegroPectin. Molecules 2021, 26, 51. [Google Scholar] [CrossRef]
- Rubio-Senent, F.; Bermúdez-Oria, A.; Rodríguez-Gutiérrez, G.; Lama-Muñoz, A.; Fernández-Bolaños, J. Structural and Antioxidant Properties of Hydroxytyrosol-Pectin Conjugates: Comparative Analysis of Adsorption and Free Radical Methods and Their Impact on in Vitro Gastrointestinal Process. Food Hydrocoll 2025, 162, 110954. [Google Scholar] [CrossRef]
- Al Jitan, S.; Scurria, A.; Albanese, L.; Pagliaro, M.; Meneguzzo, F.; Zabini, F.; Al Sakkaf, R.; Yusuf, A.; Palmisano, G.; Ciriminna, R. Micronized Cellulose from Citrus Processing Waste Using Water and Electricity Only. Int J Biol Macromol 2022, 204, 587–592. [Google Scholar] [CrossRef]
- Iyer, G.; Pandit, A.B. Bridging Ingenuity and Utility in Cavitation─A Pioneer’s Predicament. Ind Eng Chem Res 2024, 63, 12265–12276. [Google Scholar] [CrossRef]
- Meneguzzo, F.; Ciriminna, R.; Zabini, F.; Pagliaro, M. Review of Evidence Available on Hesperidin-Rich Products as Potential Tools against COVID-19 and Hydrodynamic Cavitation-Based Extraction as a Method of Increasing Their Production. Processes 2020, 8, 549. [Google Scholar] [CrossRef]
- Minutolo, A.; Gismondi, A.; Chirico, R.; Di Marco, G.; Petrone, V.; Fanelli, M.; D’Agostino, A.; Canini, A.; Grelli, S.; Albanese, L.; et al. Antioxidant Phytocomplexes Extracted from Pomegranate (Punica Granatum L.) Using Hydrodynamic Cavitation Show Potential Anticancer Activity In Vitro. Antioxidants 2023, 12, 1560. [Google Scholar] [CrossRef] [PubMed]
- Albanese, L.; Bonetti, A.; D’Acqui, L.P.; Meneguzzo, F.; Zabini, F. Affordable Production of Antioxidant Aqueous Solutions by Hydrodynamic Cavitation Processing of Silver Fir (Abies Alba Mill.) Needles. Foods 2019, 8, 65. [Google Scholar] [CrossRef]
- Félix, J.; Díaz-Del Cerro, E.; Baca, A.; López-Ballesteros, A.; Gómez-Sánchez, M.J.; De la Fuente, M. Human Supplementation with AM3, Spermidine, and Hesperidin Enhances Immune Function, Decreases Biological Age, and Improves Oxidative–Inflammatory State: A Randomized Controlled Trial. Antioxidants 2024, 13, 1391. [Google Scholar] [CrossRef]
- Putri, D.D.P.; Maran, G.G.; Kusumastuti, Y.; Susidarti, R.A.; Meiyanto, E.; Ikawati, M. Acute Toxicity Evaluation and Immunomodulatory Potential of Hydrodynamic Cavitation Extract of Citrus Peels. J Appl Pharm Sci 2022, 12, 136–145. [Google Scholar] [CrossRef]
- Waheed, Z.A.; Aboud, H.K.; Al-Awadi, J.H.H.; Al-Mousawy, A.M.J.; Alzubaidi, khudhair R. khudhair Inhibition of α-Synuclein Aggregation by Hesperidin as a Potent Anti-Amyloidogenic Polyphenol: A Computational Approach and MM-PBSA /ADMET Analysis. Biochem Biophys Rep 2025, 44, 102318. [CrossRef]
- Rodrigues, C. V.; Pintado, M. Hesperidin from Orange Peel as a Promising Skincare Bioactive: An Overview. Int J Mol Sci 2024, 25, 1890. [Google Scholar] [CrossRef]
- Chakkalakal, M.; Nadora, D.; Gahoonia, N.; Dumont, A.; Burney, W.; Pan, A.; Chambers, C.J.; Sivamani, R.K. Prospective Randomized Double-Blind Placebo-Controlled Study of Oral Pomegranate Extract on Skin Wrinkles, Biophysical Features, and the Gut-Skin Axis. J Clin Med 2022, 11, 6724. [Google Scholar] [CrossRef]
- Grabež, M.; Škrbić, R.; Stojiljković, M.P.; Rudić-Grujić, V.; Paunović, M.; Arsić, A.; Petrović, S.; Vučić, V.; Mirjanić-Azarić, B.; Šavikin, K.; et al. Beneficial Effects of Pomegranate Peel Extract on Plasma Lipid Profile, Fatty Acids Levels and Blood Pressure in Patients with Diabetes Mellitus Type-2: A Randomized, Double-Blind, Placebo-Controlled Study. J Funct Foods 2020, 64. [Google Scholar] [CrossRef]
- Eghbali, S.; Askari, S.F.; Avan, R.; Sahebkar, A. Therapeutic Effects of Punica Granatum (Pomegranate): An Updated Review of Clinical Trials. J Nutr Metab 2021, 2021, 5297162. [Google Scholar] [CrossRef]
- Vallarino, G.; Salis, A.; Lucarini, E.; Turrini, F.; Olivero, G.; Roggeri, A.; Damonte, G.; Boggia, R.; Di Cesare Mannelli, L.; Ghelardini, C.; et al. Healthy Properties of a New Formulation of Pomegranate-Peel Extract in Mice Suffering from Experimental Autoimmune Encephalomyelitis. Molecules 2022, 27, 914. [Google Scholar] [CrossRef]
- Ikeda, Y.; Nasu, M.; Bruxer, J.; Díaz-Puertas, R.; Martínez-Godfrey, J.; Bulbiankova, D.; Herranz-López, M.; Micol, V.; Álvarez-Martínez, F.J. Photoprotective, Antioxidant and Anti-Inflammatory Effects of Aged Punica Granatum Extract: In Vitro and In Vivo Insights. Food Sci Nutr 2025, 13. [Google Scholar] [CrossRef]
- Gull, H.; Ikram, A.; Khalil, A.A.; Ahmed, Z.; Nemat, A. Assessing the Multitargeted Antidiabetic Potential of Three Pomegranate Peel-Specific Metabolites: An in Silico and Pharmacokinetics Study. Food Sci Nutr 2023, 11, 7188–7205. [Google Scholar] [CrossRef]
- Maqsood, M.; Saeed, R.A.; Rahman, H.U.U.; Khan, M.I.; Khalid, N. Pomegranate Punicalagin: A Comprehensive Review of Various In Vitro and In Vivo Biological Studies. ACS Food Science & Technology 2025, 5, 2064–2085. [Google Scholar] [CrossRef]
- Drevenšek, G.; Lunder, M.; Benković, E.T.; Mikelj, A.; Štrukelj, B.; Kreft, S. Silver Fir (Abies Alba) Trunk Extract Protects Guinea Pig Arteries from Impaired Functional Responses and Morphology Due to an Atherogenic Diet. Phytomedicine 2015, 22, 856–861. [Google Scholar] [CrossRef]
- Drevenšek, G.; Lunder, M.; Benković, E.T.; Štrukelj, B.; Kreft, S. Cardioprotective Effects of Silver Fir ( Abies Alba ) Extract in Ischemic-Reperfused Isolated Rat Hearts. Food Nutr Res 2016, 60, 29623. [Google Scholar] [CrossRef]
- Pozzo, L.; Raffaelli, A.; Ciccone, L.; Zabini, F.; Vornoli, A.; Calderone, V.; Testai, L.; Meneguzzo, F. Conifer By-Products Extracted Using Hydrodynamic Cavitation as a Convenient Source of Phenolic Compounds and Free Amino Acids with Antioxidant and Antimicrobial Properties. Molecules 2025, 30, 2722. [Google Scholar] [CrossRef] [PubMed]
- Tavčar Benković, E.; Žigon, D.; Mihailović, V.; Petelinc, T.; Jamnik, P.; Kreft, S. Identification, in Vitro and in Vivo Antioxidant Activity, and Gastrointestinal Stability of Lignans from Silver Fir (Abies Alba) Wood Extract. Journal of Wood Chemistry and Technology 2017, 37, 467–477. [Google Scholar] [CrossRef]
- Geana, E.I.; Ciucure, C.T.; Tamaian, R.; Marinas, I.C.; Gaboreanu, D.M.; Stan, M.; Chitescu, C.L. Antioxidant and Wound Healing Bioactive Potential of Extracts Obtained from Bark and Needles of Softwood Species. Antioxidants 2023, 12, 1383. [Google Scholar] [CrossRef]
- Quin, O.; Bertrand, M.; Gerardin, P.; Gerardin, P.; Gerardin-Charbonnier, C.; Landon, C.; Pichon, C. Antioxidant Impact of Soft Knotwood Extracts on Human Keratinocytes Shown by NMR Metabolomic Analysis. J Proteome Res 2025, 24, 1745–1756. [Google Scholar] [CrossRef]
- Nuzzo, D.; Scurria, A.; Picone, P.; Guiducci, A.; Pagliaro, M.; Giuseppe; Pantaleo; Albanese, L.; Meneguzzo, F.; Ciriminna, R. A Gluten-Free Biscuit Fortified with Lemon IntegroPectin. ChemistrySelect 2022, 7, e202104247. [CrossRef]
- Breschi, C.; D’Agostino, S.; Meneguzzo, F.; Zabini, F.; Chini, J.; Lovatti, L.; Tagliavento, L.; Guerrini, L.; Bellumori, M.; Cecchi, L.; et al. Can a Fraction of Flour and Sugar Be Replaced with Fruit By-Product Extracts in a Gluten-Free and Vegan Cookie Recipe? Molecules 2024, 29, 1102. [Google Scholar] [CrossRef]
- Parenti, O.; Albanese, L.; Guerrini, L.; Zanoni, B.; Zabini, F.; Meneguzzo, F. Whole Wheat Bread Enriched with Silver Fir Needles (Abies Alba Mill.) Extract: Technological and Antioxidant Properties. J Sci Food Agric. [CrossRef]
- Fidelis, M.; Tienaho, J.; Meneguzzo, F.; Pihlava, J.-M.; Rudolfsson, M.; Järvenpää, E.; Imao, H.; Hellström, J.; Liimatainen, J.; Kilpeläinen, P.; et al. Spruce, Pine and Fir Needles as Sustainable Ingredients for Whole Wheat Bread Fortification: Enhancing Nutritional and Functional Properties. LWT 2024, 117055. [Google Scholar] [CrossRef]
- Kerksick, C.M.; Arent, S.; Schoenfeld, B.J.; Stout, J.R.; Campbell, B.; Wilborn, C.D.; Taylor, L.; Kalman, D.; Smith-Ryan, A.E.; Kreider, R.B.; et al. International Society of Sports Nutrition Position Stand: Nutrient Timing. J Int Soc Sports Nutr 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.; Jacob, S. A Simple Practice Guide for Dose Conversion between Animals and Human. J Basic Clin Pharm 2016, 7, 27. [Google Scholar] [CrossRef]
- Debeljak, J.; Ferk, P.; Čokolič, M.; Zavratnik, A.; Tavčar Benković, E.; Kreft, S.; Štrukelj, B. Randomised, Double Blind, Cross-over, Placebo and Active Controlled Human Pharmacodynamic Study on the Influence of Silver Fir Wood Extract (Belinal) on Post-Prandial Glycemic Response. Pharmazie 2016, 71, 566–569. [Google Scholar] [CrossRef] [PubMed]
- Drevenšek, G.; Lunder, M.; Benković, E.T.; Mikelj, A.; Štrukelj, B.; Kreft, S. Silver Fir (Abies Alba) Trunk Extract Protects Guinea Pig Arteries from Impaired Functional Responses and Morphology Due to an Atherogenic Diet. Phytomedicine 2015, 22, 856–861. [Google Scholar] [CrossRef]
- Vek, V.; Keržič, E.; Poljanšek, I.; Eklund, P.; Humar, M.; Oven, P. Wood Extractives of Silver Fir and Their Antioxidant and Antifungal Properties. Molecules 2021, 26. [Google Scholar] [CrossRef]
- Schoss, K.; Benedetič, R.; Kreft, S. The Phenolic Content, Antioxidative Properties and Extractable Substances in Silver Fir (Abies Alba Mill.) Branches Decrease with Distance from the Trunk. Plants 2022, 11, 333. [Google Scholar] [CrossRef]
- Tang, J.; Zhu, X.; Dong, G.; Hannon, S.; Santos, H.M.; Sun, D.W.; Tiwari, B.K. Comparative Studies on Enhancing Pea Protein Extraction Recovery Rates and Structural Integrity Using Ultrasonic and Hydrodynamic Cavitation Technologies. Lwt 2024, 200, 116130. [Google Scholar] [CrossRef]
- Tang, J.; Goksen, G.; Islam, M.S.; Ranade, V.; Hannon, S.; Sun, D.W.; Tiwari, B.K. Large-Scale Protein Extraction from Oat Hulls Using Two Hydrodynamic Cavitation Techniques: A Comparison of Extraction Efficiency and Protein Nutritional Properties. Food Chem 2025, 471, 142724. [Google Scholar] [CrossRef]
- Suchintita Das, R.; Zhu, X.; Hannon, S.; Mullins, E.; Alves, S.; Garcia-Vaquero, M.; Tiwari, B.K. Exploring Osborne Fractionation and Laboratory/Pilot Scale Technologies (Conventional Extraction, Ultrasound-Assisted Extraction, High-Pressure Processing and Hydrodynamic Cavitation) for Protein Extraction from Faba Bean (Vicia Faba L.). Innovative Food Science and Emerging Technologies 2023, 89, 103487. [Google Scholar] [CrossRef]
- Faraloni, C.; Albanese, L.; Zittelli, G.C.; Meneguzzo, F.; Tagliavento, L.; Zabini, F. New Route to the Production of Almond Beverages Using Hydrodynamic Cavitation. Foods 2023, 12, 935. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, S.; Bashir, I.; Wani, S.M.; Sofi, S.A.; Amin, T.; Malik, A.R.; Khan, F.; Murtaza, I.; Khan, I.; Ayaz, Q.; et al. Protein Extraction from Apple Seeds for Waste Valorization for Sustainable Food Systems. Sustainable Food Technology 2025. [Google Scholar] [CrossRef]
- Tagliavento, L.; Nardin, T.; Chini, J.; Vighi, N.; Lovatti, L.; Testai, L.; Meneguzzo, F.; Larcher, R.; Zabini, F. Sustainable Exploitation of Apple By-Products: A Retrospective Analysis of Pilot-Scale Extraction Tests Using Hydrodynamic Cavitation. Foods 2025, 14, 1915. [Google Scholar] [CrossRef]
- Wei, F.; Ren, X.; Huang, Y.; Hua, N.; Wu, Y.; Yang, F. Hydrodynamic Cavitation Induced Fabrication of Soy Protein Isolate–Polyphenol Complexes: Structural and Functional Properties. Curr Res Food Sci 2025, 10, 100969. [Google Scholar] [CrossRef]
- Butera, V.; Ciriminna, R.; Valenza, C.; Petri, G.L.; Angellotti, G.; Barone, G.; Meneguzzo, F.; Di Liberto, V.; Bonura, A.; Pagliaro, M. Citrus IntegroPectin: A Computational Insight. Discover Molecules 2025, 2, 6. [Google Scholar] [CrossRef]
- Sano, C. Di; D’Anna, C.; Petri, G.L.; Angellotti, G.; Meneguzzo, F.; Ciriminna, R.; Pagliaro, M. Citrus Flavonoid-Pectin Conjugates: Towards Broad Scope Therapeutic Agents. Food Hydrocolloids for Health 2025, 8, 100246. [Google Scholar] [CrossRef]
- Hua, N.; Ren, X.; Yang, F.; Huang, Y.; Wei, F.; Yang, L. The Effect of Hydrodynamic Cavitation on the Structural and Functional Properties of Soy Protein Isolate–Lignan/Stilbene Polyphenol Conjugates. Foods 2024, 13, 3609. [Google Scholar] [CrossRef]
- Filla, J.M.; Hinrichs, J. Processing of Whey Protein-Pectin Complexes: Upscaling from Batch Lab Scale Experiments to a Continuous Technical Scale Process. J Food Eng 2023, 347, 111437. [Google Scholar] [CrossRef]
- Qin, X.; Di, X.; Li, Y.; Wang, Q.; Harold, C.; Liu, G. Bioactivity of Proanthocyanidin-Whey Protein Isolate Stabilized Pickering Emulsion with Encapsulated β-Carotene. Food Chem 2025, 493, 145756. [Google Scholar] [CrossRef]
- Manochai, T.; Kamthai, S.; Siriwoharn, T. Comparative Study of Free Radical Grafting and Alkaline Conjugation for Enhanced Resveratrol Incorporation and Whey Protein Functionalities. Foods 2025, 14, 2596. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Huang, F.; Xie, B.; Sun, Z.; McClements, D.J.; Deng, Q. Fabrication and Characterization of Whey Protein Isolates- Lotus Seedpod Proanthocyanin Conjugate: Its Potential Application in Oxidizable Emulsions. Food Chem 2021, 346, 128680. [Google Scholar] [CrossRef]
- Tang, C.; Tan, B.; Sun, X. Elucidation of Interaction between Whey Proteins and Proanthocyanidins and Its Protective Effects on Proanthocyanidins during In-Vitro Digestion and Storage. Molecules 2021, 26. [Google Scholar] [CrossRef] [PubMed]
| Method | Advantages | Drawbacks |
|---|---|---|
| UAE | Water can be the only solvent; Low working temperature, fast, low energy consumption. |
Scaling beyond pilot is challenging due to acoustic field attenuation; Preservation of bioactive compounds sensitive to working temperature, amplitude, frequency and power. |
| MAE | Low working temperature; Low energy consumption; High extraction yield; Preservation of bioactive compounds including volatiles. |
Scalability not proven; Generally a pre-treatment, needs further extraction technique downstream; High cost of equipment at the real scale. |
| PEF | Water as the only solvent; Very short processing time. |
Generally a pre-treatment, needs further extraction technique downstream. |
| SWE | Water as the only solvent; Selective extraction; continuous flow of operation; short time. |
Difficult cleaning; Possible degradation of bioactive compounds due to high temperature and pressure; High cost of equipment; Energy intensive. |
| EAE | High quality of recovered pectin; As a pretreatment, allows UAE to increase the extraction yield of phenolic compounds. |
Lower recovery of phenolic compounds compared with conventional Soxhlet technique; Selectivity of enzymes; Long process time; Difficult to scale up; High cost of enzymes at the real scale. |
| NADES | High selectivity of extracted bioactive compounds; Low working temperature; Simple equipment. |
Scalability not proven; High cost of NADES; NADES residues in the end product. |
| HC | Water as the only solvent; Low working temperature, fast, low energy consumption; Creation of new stably conjugated, water-soluble phytocomplexes with higher bioavailability compared to individual compounds; Insoluble residues with high technical value; Straightforwardly scalable. |
Non-standard equipment; Critical dependence of performance on construction details, hence the need for new skills. |
| GEP | HC | UAE | MAE | PEF | SWE h | EAE | NADES |
|---|---|---|---|---|---|---|---|
| 1. Use water/safe solvents a | ✓ | ✓ | ✓ | ✓ | ✓ | ✗ | ✗ |
| 2. Non-denaturing conditions | ✓ | ✓ | ✓ | ✓ | ✗ | ✓ | ✓ |
| 3. Minimize biomass pre-treatment | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| 4. Minimize energy consumption | ✓ | ✓ | ✓ | ✓ | ✗ | ✓ | ✓ |
| 5. Renewable energy sources b | ✓ | ✓ | ✓ | ✓ | ✗ | ✓ | ✓ |
| 6. Minimize unit operations | ✓ | ✓ | ✗ | ✗ | ✓ | ✓ | ✓ |
| 7. Integration with downstream c | ✓ | ✗ | ✗ | ✗ | ✗ | ✗ | ✗ |
| 8. Predictability and scalability | ✓ | ✗ | ✗ | ✓ | ✓ | ✗ | ✗ |
| 9. Automation d | ✓ | ✓ | ✓ | ✓ | ✓ | ✗ | ✗ |
| 10. Safety and hygiene e | ✓ | ✓ | ✓ | ✓ | ✗ | ✗ | ✗ |
| 11. Valorize all byproducts f | ✓ | ✓ | ✗ | ✗ | ✗ | ✗ | ✗ |
| 12. Carbon footprint reduction g | ✓ | ✓ | ✓ | ✓ | ✗ | ✗ | ✗ |
| Compliance Rate (%) | 100% | 83% | 67% | 75% | 42% | 42% | 42% |
| Raw resource | Moisturea (%) |
Daily amount (mg) |
Reference molecule (amount in mg) | Yieldb (%) |
Fresh raw material (g wet basis) |
|---|---|---|---|---|---|
| Red orange waste peel | 75 | 200 | Hesperidin (5) | 30 | 2668 |
| Pomegranate waste peel | 72 | 250-1100 | Punicalagin (35-75) | 13 | 6868-30.221 |
| Abies alba byproducts | 30c | 150-200 | Lignans (9-24)d | 11c | 1949-2597 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




