Submitted:
07 November 2025
Posted:
10 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
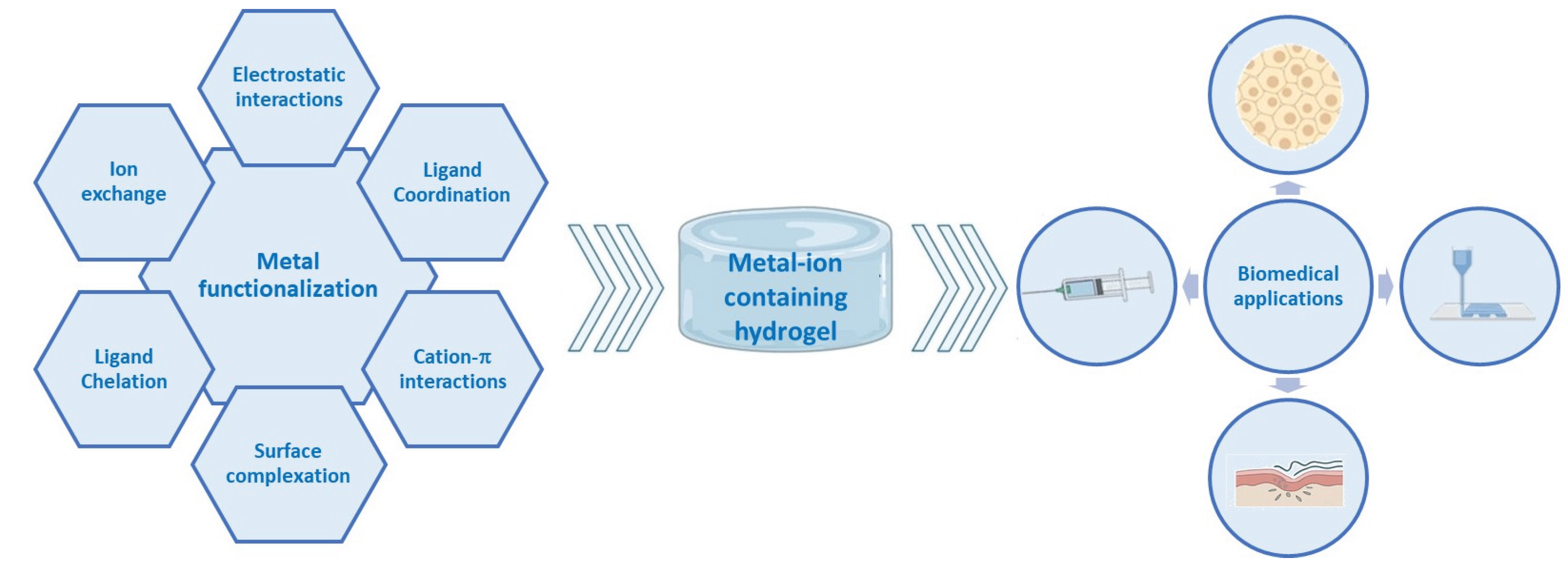
2. Drug Delivery and Anticancer Applications
| Ligand | Gelation class | Metal center(s) | Encapsulated or active drug(s) | Biomedical application | Ref. |
| 3,3’-bispyridyl-bis-amide derivative | Low-molecular-weight gelator | Pd²⁺ | Doxorubicin (DOX) | Drug delivery |
[51] |
| 1,3- dipyridin-3-ylurea | Low-molecular-weight gelator | Zn²⁺ | Ibuprofen, Naproxen, Fenoprofen, Diclofenac, Meclofenamic acid, Mefenamic acid | Drug delivery and cell-imaging |
[52] |
| nitrile-functionalized terpyridyl/ dicarboxylic acids | Low-molecular-weight gelator | Cu⁺ / Zn²⁺ | Self-delivery (no external drug) | Drug delivery |
[53] |
| N,N,N′N′-tetrakis- (2-hydroxy-ethyl)ethylenediamine | Low-molecular-weight gelator | Mg²⁺ | No external drug (intrinsic anticancer activity) | Therapy | [54] |
| 2-hydroxy-benzyl)amino aspartic acid | Low-molecular-weight gelator | Mn²⁺ | Indomethacin (IND) and Gemcitabine (GEM) | Drug delivery |
[55] |
| 3,5-bis((4-(cyanomethyl)phenyl)carbamoyl)benzoic acid | Low-molecular-weight gelator | Ru²⁺ | Self-delivery (Ru complex formed in situ) | Drug delivery |
[56] |
| 3-pyridyl amide ligand derived from a non-steroidal-anti-inflammatory-drug | Low-molecular-weight gelator | Zn²⁺ | Multi-NSAID coordination (no additional drug) | Drug delivery |
[57] |
| N-phenyl-3-pyridylamide and N-phenyl-4-pyridylamide/ non-steroidal-anti-inflammatory-drug | Low-molecular-weight gelator | Zn²⁺ | No external drug (NSAID coordinated) | Therapy | [58] |
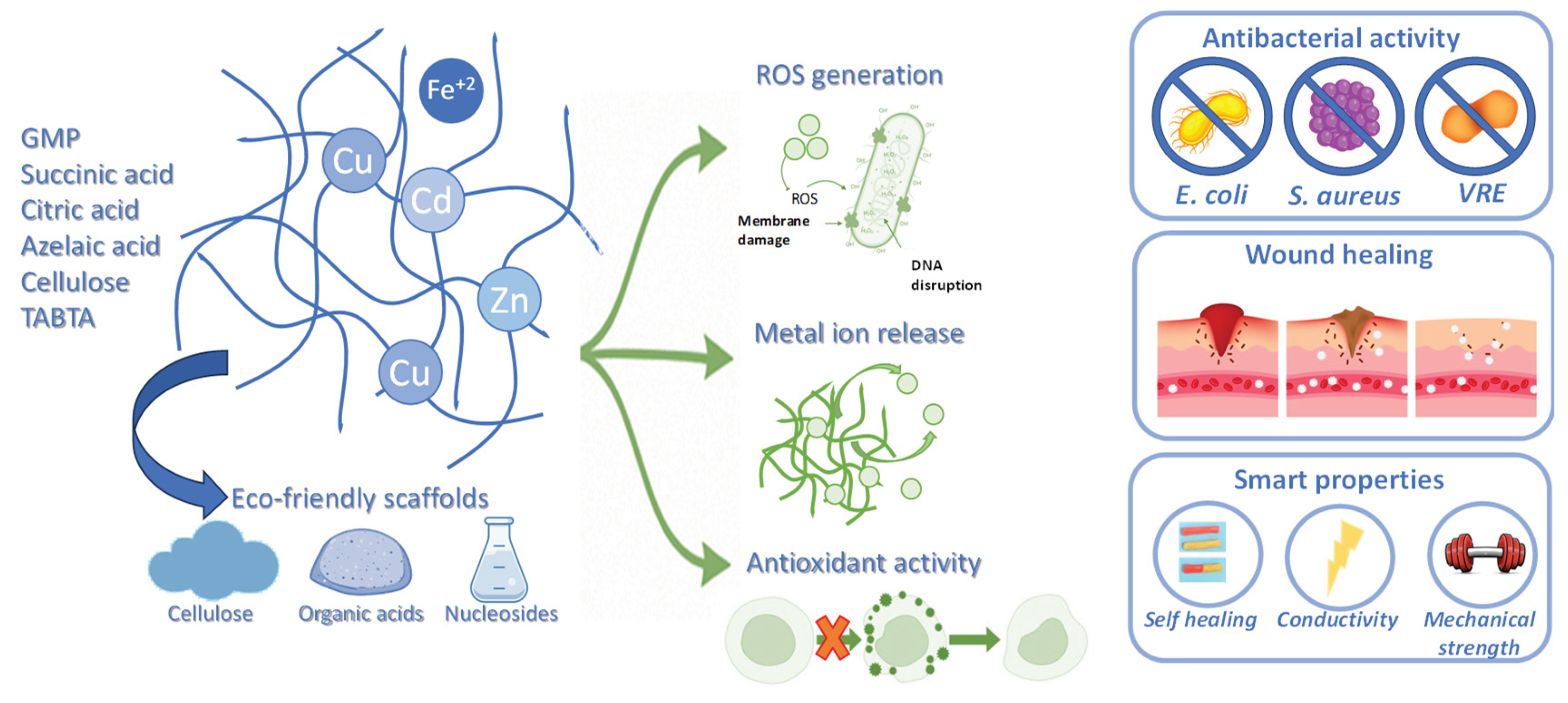
3. Antibacterial, Antioxidant, and Wound Healing Metallogels
4. Biosensing, Luminescent, and Imaging Metallogels
5. Biocompatible Scaffolds and Tissue Engineering
6. Future Perspectives and Translational Challenges
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AIE | Aggregation-Induced Emission |
| AA | Ascorbic Acid |
| ALHS(s) | Artificial Light-Harvesting System(s) |
| AzoPz | Arylazopyrazole (molecular photoswitch) |
| BAX | Bcl-2-associated X Protein (pro-apoptotic marker) |
| BCL2 | B-cell Lymphoma 2 (anti-apoptotic protein) |
| BDP / BODIPY | Boron-dipyrromethene (fluorescent dye) |
| BIPY | 2,2′-Bipyridine-4,4′-dicarboxaldehyde |
| CMCS | Carboxymethyl Chitosan |
| CuG / PdG / Ru(II)G5 | Copper / Palladium / Ruthenium-based metallogel |
| DIC | Diclofenac |
| DMF | N,N-Dimethylformamide |
| DMSO | Dimethyl Sulfoxide |
| DOX | Doxorubicin |
| ECM | Extracellular Matrix |
| EDX | Energy-Dispersive X-ray Spectroscopy |
| ESI-MS | Electrospray Ionization Mass Spectrometry |
| FESEM | Field Emission Scanning Electron Microscopy |
| FEN | Fenoprofen |
| FITC | Fluorescein Isothiocyanate |
| FLU | Flufenamic Acid |
| FT-IR | Fourier Transform Infrared Spectroscopy |
| GEM | Gemcitabine |
| GMP | Good Manufacturing Practice |
| GMP / 5′-GMP | Guanosine-5′-Monophosphate |
| G5 | Low-molecular-weight carboxylic acid gelator |
| H₆L | 5,5′,5″-(1,3,5-Triazine-2,4,6-triyl)tris(azanediyl)triisophthalate |
| HEK 293 | Human Embryonic Kidney 293 cell line |
| Hg-SA | Mercury(II)-Succinic Acid Metallogel |
| HRTEM | High-Resolution Transmission Electron Microscopy |
| IC₅₀ | Half Maximal Inhibitory Concentration |
| IBU | Ibuprofen |
| ImF | Imidazole-functionalized Phenylalanine |
| L₂ | 5-Azido-N,N′-dipyridin-3-yl-isophthalamide (ligand) |
| MCF-7 | Human Breast Cancer Cell Line |
| MEC | Meclofenamic Acid |
| MEF | Mefenamic Acid |
| Mg-Tetrakis | Mg(II)-Tetrakis(2-hydroxyethyl)ethylenediamine Hydrogel |
| MOC | Metal–Organic Cage |
| MOF | Metal–Organic Framework |
| MOG | Manganese(II)-Based Metallohydrogel |
| MOG_IND | MOG loaded with Indomethacin |
| MOG_GEM | MOG loaded with Gemcitabine |
| MTT | 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium Bromide Assay |
| NAP | Naproxen |
| Ni-AA / Co-AA | Nickel / Cobalt Adipic Acid Metallogel |
| Ni / Zn / Cu / Fe | Nickel / Zinc / Copper / Iron (metal centers) |
| NOXA | Phorbol-12-myristate-13-acetate-induced protein 1 (pro-apoptotic marker) |
| NSAIDs | Non-Steroidal Anti-Inflammatory Drugs |
| NMR | Nuclear Magnetic Resonance |
| P | Bi-benzimidazole compound (light-harvesting donor) |
| PBS | Phosphate-Buffered Saline |
| Pd(II) | Palladium(II) |
| Pd₂L₂⁴ | Palladium-based metal–organic cage |
| PCA | Principal Component Analysis |
| PXRD | Powder X-ray Diffraction |
| RAW 264.7 | Murine Macrophage Cell Line |
| Rh6G | Rhodamine 6G |
| ROS | Reactive Oxygen Species |
| Ru(II)L | Ruthenium(II) complex released from Ru(II)G5 gel |
| SH-SY5Y | Human Neuroblastoma Cell Line |
| TABTA | N(1),N(3),N(5)-tris(4-aminophenyl)benzene-1,3,5-tricarboxamide |
| TEM | Transmission Electron Microscopy |
| Th⁴⁺ / Tb³⁺ | Thorium(IV) / Terbium(III) |
| THTA-G | Tripodal Hydrogel (TH + TA co-assembly) |
| TH / TA | Tri-(isoniazid-4-yl)- and Tri-(pyridine-4-yl)-functionalized Trimesic Derivatives |
| Tetrakis | N,N,N′,N′-tetrakis(2-hydroxyethyl)ethylenediamine |
| UV–vis | Ultraviolet–Visible Spectroscopy |
| VRE | Vancomycin-Resistant Enterococcus |
| Zn(II)-NSAID | Zinc(II)-Non-Steroidal Anti-Inflammatory Drug Complex |
| Zn²⁺ / Cu²⁺ / Mn²⁺ / Mg²⁺ | Metal Ions: Zinc, Copper, Manganese, Magnesium |
| Zr⁴⁺ | Zirconium(IV) |
| XRD | X-ray Diffraction |
References
- Liu, Z.; Zhao, X.; Chu, Q.; Feng, Y. Recent Advances in Stimuli-Responsive Metallogels. Molecules 2023, 28, 2274, . [CrossRef]
- Recent Advances in Metallogels - Chemical Society Reviews (RSC Publishing). Available online: https://pubs.rsc.org/en/content/articlehtml/2013/cs/c2cs35354g (accessed on 17 October 2025). [CrossRef]
- Metallogels: Availability, Applicability, and Advanceability - Wu - 2019 - Advanced Materials - Wiley Online Library Available online: https://advanced.onlinelibrary.wiley.com/doi/full/10.1002/adma.201806204 (accessed on 17 October 2025).
- Recent Advances in Stimuli-Responsive Metallogels Available online: https://www.mdpi.com/1420-3049/28/5/2274 (accessed on 17 October 2025).
- Mikhailidi, A.; Ungureanu, E.; Belosinschi, D.; Tofanica, B.-M.; Volf, I. Cellulose-Based Metallogels—Part 3: Multifunctional Materials. Gels 2023, 9, 878, . [CrossRef]
- Metallogels as Functional Catalysts: Bridging Soft Materials with Sustainable Transformations - Mondal - 2025 - ChemCatChem - Wiley Online Library Available online: https://chemistry-europe.onlinelibrary.wiley.com/doi/full/10.1002/cctc.202500640 (accessed on 17 October 2025).
- Case, D.R.; Zubieta, J.; P. Doyle, R. The Coordination Chemistry of Bio-Relevant Ligands and Their Magnesium Complexes. Molecules 2020, 25, 3172, . [CrossRef]
- Sutar, P.; Kumar Maji, T. Recent Advances in Coordination-Driven Polymeric Gel Materials: Design and Applications. Dalton Transactions 2020, 49, 7658–7672, . [CrossRef]
- Peptide Supramolecular Self-Assembly: Regulatory Mechanism, Functional Properties, and Its Application in Foods | Journal of Agricultural and Food Chemistry Available online: https://pubs.acs.org/doi/10.1021/acs.jafc.3c09237 (accessed on 17 October 2025).
- Self-Assembly of Supramolecular Architectures by the Effect of Amino Acid Residues of Quaternary Ammonium Pillar [5]Arenes Available online: https://www.mdpi.com/1422-0067/21/19/7206 (accessed on 17 October 2025).
- Zamora-Ledezma, C.; C., D.F.C.; Medina, E.; Sinche, F.; Santiago Vispo, N.; Dahoumane, S.A.; Alexis, F. Biomedical Science to Tackle the COVID-19 Pandemic: Current Status and Future Perspectives. Molecules 2020, 25, 4620, . [CrossRef]
- Therapeutic Convergence in Neurodegeneration: Natural Products, Drug Repurposing, and Biomolecular Targets Available online: https://www.mdpi.com/2218-273X/15/9/1333 (accessed on 17 October 2025).
- Roviello, V.; Musumeci, D.; Mokhir, A.; Roviello, G.N. Evidence of Protein Binding by a Nucleopeptide Based on a Thyminedecorated L-Diaminopropanoic Acid through CD and In Silico Studies. Current Medicinal Chemistry 28, 5004–5015, . [CrossRef]
- Panja, P.; Ghosh, U.; Sil, A.; K. Patra, S. A Highly Selective Cu 2+ -Coordination Triggered Multi-Stimuli Responsive and Functional Metallogel of Bis-Terpyridyl-Based Low Molecular Weight (LMW) Gelator. Dalton Transactions 2025, 54, 14687–14700, . [CrossRef]
- Preet, K.; Kumar, R.; Kaur, L.; Roy, S.; Sahoo, S.C.; Salunke, D.B. Competition in Formation of Stimuli-Responsive Metallogel and Coordination Polymers from a Single Precursor: Synthesis, Characterization and Catalytic Application. Journal of Molecular Structure 2024, 1310, 138316, . [CrossRef]
- Precision Therapeutics Through Bioactive Compounds: Metabolic Reprogramming, Omics Integration, and Drug Repurposing Strategies Available online: https://www.mdpi.com/1422-0067/26/20/10047 (accessed on 17 October 2025).
- Fu, H.L.-K.; Yam, V.W.-W. Supramolecular Metallogels of Platinum(II) and Gold(III) Complexes. chem. Lett. 2018, 47, 605–610, . [CrossRef]
- Singha, T.; Pal, B.; Majumdar, S.; Lepcha, G.; Pal, I.; Ray, P.P.; Dey, B.; Datta, P.K. Optical Nonlinearity of Semiconducting Cd(II) Metallogel in the Femtosecond Regime with Two-, Three-, and Four-Photon Absorption. ACS Appl. Opt. Mater. 2024, 2, 474–484, . [CrossRef]
- Pal, I.; Pathak, N.K.; Majumdar, S.; Lepcha, G.; Dey, A.; Yatirajula, S.K.; Tripathy, U.; Dey, B. Solvent-Driven Variations of Third-Order Nonlinear Thermo-Optical Features of Glutaric Acid-Directed Self-Healing Supramolecular Ni(II) Metallogels. Langmuir 2023, 39, 16584–16595, . [CrossRef]
- Su, H.; Zhu, S.; Qu, M.; Liu, R.; Song, G.; Zhu, H. 1,3,5-Triazine-Based Pt(II) Metallogel Material: Synthesis, Photophysical Properties, and Optical Power-Limiting Performance. J. Phys. Chem. C 2019, 123, 15685–15692, . [CrossRef]
- Shi, H.; Li, R.; Ren, Y.; Gan, S.; Zhu, S.; Liu, R. Synthesis, Self-Healing and Optical Power Limiting Properties of Pt(II) Metallogel Based on Reversible Quadruple Hydrogen Bonding. Dyes and Pigments 2025, 242, 112980, . [CrossRef]
- Majumdar, S.; Sil, S.; Sahu, R.; Ghosh, M.; Lepcha, G.; Dey, A.; Mandal, S.; Pratim Ray, P.; Dey, B. Electronic Charge Transport Phenomena Directed Smart Fabrication of Metal-Semiconductor Based Electronic Junction Device by a Supramolecular Mn(II)-Metallogel. Journal of Molecular Liquids 2021, 338, 116769, . [CrossRef]
- Dhibar, S.; Dey, A.; Majumdar, S.; Ghosh, D.; Mandal, A.; Pratim Ray, P.; Dey, B. A Supramolecular Cd( Ii )-Metallogel: An Efficient Semiconductive Electronic Device. Dalton Transactions 2018, 47, 17412–17420, . [CrossRef]
- Sedghiniya, S.; Soleimannejad, J.; Blake, A.J. A Low Molecular Weight Zr(IV) Metallogel for Protein Delivery. Materials Today Communications 2021, 27, 102448, . [CrossRef]
- Choudhary, P.; Gaur, R.; Rambabu, D.; Dhir, A.; Gupta, A.; Pooja Copper Metallogel as Potential Drug Carrier for Anti-Inflammatory Drugs. ChemistrySelect 2021, 6, 9139–9143, . [CrossRef]
- dos Santos Batista, J.G.; Vigilato Rodrigues, M.Á.; de Freitas, L.F.; Moreira Fonseca, A.C.; de Souza Rodrigues, A.; Cunha da Cruz, C.P.; Thipe, V.C.; Lugão, A.B. Copper-Based Nanomaterials for Biologically Relevant Compounds. In Copper-Based Nanomaterials in Organic Transformations; ACS Symposium Series; American Chemical Society, 2024; Vol. 1466, pp. 305–338.
- Anchoring Drugs to a Zinc(II) Coordination Polymer Network: Exploiting Structural Rationale toward the Design of Metallogels for Drug-Delivery Applications | Inorganic Chemistry Available online: https://pubs.acs.org/doi/full/10.1021/acs.inorgchem.0c03550 (accessed on 17 October 2025).
- Afzal, S.; Maswal, M.; Lone, M.S.; Ashraf, U.; Mushtaq, U.; Dar, A.A. Metal–Ligand-Based Thixotropic Self-Healing Poly (Vinyl Alcohol) Metallohydrogels: Their Application in pH-Responsive Drug Release and Selective Adsorption of Dyes. Journal of Materials Research 2021, 36, 3293–3308, . [CrossRef]
- β-Cyclodextrin Encapsulated Platinum(II)-Based Nanoparticles: Photodynamic Therapy and Inhibition of the NF-κB Signaling Pathway in Glioblastoma | ACS Applied Bio Materials Available online: https://pubs.acs.org/doi/full/10.1021/acsabm.5c00103 (accessed on 17 October 2025).
- Xu, J.; Wang, J.; Ye, J.; Jiao, J.; Liu, Z.; Zhao, C.; Li, B.; Fu, Y. Metal-Coordinated Supramolecular Self-Assemblies for Cancer Theranostics. Advanced Science 2021, 8, 2101101, . [CrossRef]
- Dhibar, S.; Some, S.; Pal, S.; Laha, R.; Kaith, P.; Trivedi, A.; Bhattacharjee, S.; N. Nthunya, L.; O. Ajiboye, T.; Kumar Panja, S.; et al. Investigating the Potent Antimicrobial Properties of a Supramolecular Zn( Ii )-Metallogel Formed from an Isophthalic Acid-Based Low Molecular Weight Gelator. RSC Advances 2025, 15, 27544–27550, . [CrossRef]
- Dhibaris, S.; Pal, S.; Some, S.; Karmakar, K.; Saha, R.; Bhattacharjee, S.; Roy, A.; Jyoti Ray, S.; O. Ajiboye, T.; Dam, S.; et al. Efficient Antimicrobial Applications of Two Novel Supramolecular Metallogels Derived from a l (+)-Tartaric Acid Low Molecular Weight Gelator. RSC Advances 2024, 14, 26354–26361, . [CrossRef]
- Fortunato, A.; Mba, M. Metal Cation Triggered Peptide Hydrogels and Their Application in Food Freshness Monitoring and Dye Adsorption. Gels 2021, 7, 85, . [CrossRef]
- Foster, J.A.; Piepenbrock, M.-O.M.; Lloyd, G.O.; Clarke, N.; Howard, J.A.K.; Steed, J.W. Anion-Switchable Supramolecular Gels for Controlling Pharmaceutical Crystal Growth. Nat Chem 2010, 2, 1037–1043, . [CrossRef]
- Anh, H.T.P.; Huang, C.-M.; Huang, C.-J. Intelligent Metal-Phenolic Metallogels as Dressings for Infected Wounds. Sci Rep 2019, 9, 11562, . [CrossRef]
- Legrand, A.; Liu, L.-H.; Royla, P.; Aoyama, T.; Craig, G.A.; Carné-Sánchez, A.; Urayama, K.; Weigand, J.J.; Lin, C.-H.; Furukawa, S. Spatiotemporal Control of Supramolecular Polymerization and Gelation of Metal-Organic Polyhedra. J Am Chem Soc 2021, 143, 3562–3570, . [CrossRef]
- Piepenbrock, M.-O.M.; Clarke, N.; Steed, J.W. Metal Ion and Anion-Based “Tuning” of a Supramolecular Metallogel. Langmuir 2009, 25, 8451–8456, . [CrossRef]
- Lepcha, G.; Pal, B.; Majumdar, S.; Ahmed, K.T.; Pal, I.; Biswas, S.R.; Ray, P.P.; Dey, B. Ni(II) and Zn(II)-Metallogel-Based Anti-Bacterial Scaffolds for Fabricating Light-Responsive Junction-Type Semiconducting Diodes with Non-Ohmic Conduction Mechanism. Mater. Adv. 2023, 4, 2595–2603, . [CrossRef]
- Anh, H.T.P.; Huang, C.-M.; Huang, C.-J. Intelligent Metal-Phenolic Metallogels as Dressings for Infected Wounds. Sci Rep 2019, 9, 11562, . [CrossRef]
- Yee, Z.L. Human Hair Keratin Gradient Metallogel and Their Applications for Wound Healing, Nanyang Technological University, 2024.
- Puttreddy, R.; Thongrom, B.; Rautiainen, J.M.; Lahtinen, M.; Kukkonen, E.; Haag, R.; Moilanen, J.O.; Lundell, J.; Rissanen, K. Free-Standing Supramolecular Pyridine N-Oxide-Silver(I) Metallogels. Adv Mater 2025, 37, e2502818, . [CrossRef]
- Liu, Z.; Zhao, X.; Chu, Q.; Feng, Y. Recent Advances in Stimuli-Responsive Metallogels. Molecules 2023, 28, 2274, . [CrossRef]
- Dhibar, S.; Halder, S.; Pal, S.; Kumari, D.; Karmakar, K.; Saha, R.; Bhattacharjee, S.; Mohan, A.; Ajiboye, T.O.; Ray, S.J.; et al. Development of a Bioactive Supramolecular Cobalt(II)-Metallogel for Antimicrobial and Semiconducting Microelectronic Device Applications. Langmuir 2025, 41, 16777–16785, . [CrossRef]
- Singh, S.; Sharma, A.K.; Rohilla, K.; Verma, N.; Sharma, B. A Multistimuli Responsive and Self-Healing Zn(Ii)-Inosine Supramolecular Metal-Organic Gel: Phase Selective Gelation and Application as a Light-Responsive Schottky Barrier Diode. Nanoscale Adv 2025, 7, 1923–1936, . [CrossRef]
- Sarkar, K.; Dastidar, P. Rational Approach Towards Designing Metallogels From a Urea-Functionalized Pyridyl Dicarboxylate: Anti-Inflammatory, Anticancer, and Drug Delivery. Chem Asian J 2019, 14, 194–204, . [CrossRef]
- Rational Approach Towards Designing Metallogels From a Urea--Functionalized Pyridyl Dicarboxylate: Anti--inflammatory, Anticancer, and Drug Delivery - Sarkar - 2019 - Chemistry – An Asian Journal - Wiley Online Library Available online: https://aces.onlinelibrary.wiley.com/doi/10.1002/asia.201801462 (accessed on 17 October 2025).
- Roviello, G.N.; Roviello, V.; Autiero, I.; Saviano, M. Solid Phase Synthesis of TyrT, a Thymine–Tyrosine Conjugate with Poly(A) RNA-Binding Ability. RSC Adv. 2016, 6, 27607–27613, . [CrossRef]
- Li, T.-T.; Liu, S.-N.; Wu, L.-H.; Cai, S.-L.; Zheng, S.-R. Strategies for the Construction of Functional Materials Utilizing Presynthesized Metal-Organic Cages (MOCs). ChemPlusChem 2022, 87, e202200172, . [CrossRef]
- Zeng, Q.-W.; Hu, L.; Niu, Y.; Wang, D.; Kang, Y.; Jia, H.; Dou, W.-T.; Xu, L. Metal-Organic Cages for Gas Adsorption and Separation. Chem Commun (Camb) 2024, 60, 3469–3483, . [CrossRef]
- Guan, J.; Du, J.; Sun, Q.; He, W.; Ma, J.; Hassan, S.U.; Wu, J.; Zhang, H.; Zhang, S.; Liu, J. Metal-Organic Cages Improving Microporosity in Polymeric Membrane for Superior CO2 Capture. Sci Adv 2025, 11, eads0583, . [CrossRef]
- Developing Supramolecular Metallogel Derived from Pd2L4 Cage Molecule for Delivering an Anti--Cancer Drug to Melanoma Cell B16−F10 - Bera - 2024 - Chemistry – An Asian Journal - Wiley Online Library Available online: https://aces.onlinelibrary.wiley.com/doi/10.1002/asia.202400419 (accessed on 17 October 2025).
- Biswas, P.; Dastidar, P. Anchoring Drugs to a Zinc(II) Coordination Polymer Network: Exploiting Structural Rationale toward the Design of Metallogels for Drug-Delivery Applications. Inorg. Chem. 2021, 60, 3218–3231, . [CrossRef]
- Bera, S.; Datta, H.K.; Dastidar, P. Nitrile-Containing Terpyridyl Zn(II)-Coordination Polymer-Based Metallogelators Displaying Helical Structures: Synthesis, Structures, and “Druglike” Action against B16-F10 Melanoma Cells. ACS Appl. Mater. Interfaces 2023, 15, 25098–25109, . [CrossRef]
- Saha, D.; Talukdar, D.; Pal, I.; Majumdar, S.; Lepcha, G.; Sadhu, S.; Yatirajula, S.K.; Das, G.; Dey, B. Mechanically Flexible Self-Healing Mg(II)-Metallogel: Approach of Triggering the ROS-Induced Apoptosis in Human Breast Cancer Cells. Langmuir 2024, 40, 19816–19829, . [CrossRef]
- A Self-Healable Metallohydrogel for Drug Encapsulations and Drug Release - RSC Advances (RSC Publishing) Available online: https://pubs.rsc.org/en/content/articlelanding/2023/ra/d3ra00930k (accessed on 17 October 2025).
- Malviya, N.; Sonkar, C.; Ganguly, R.; Bhattacherjee, D.; Bhabak, K.P.; Mukhopadhyay, S. Novel Approach to Generate a Self-Deliverable Ru(II)-Based Anticancer Agent in the Self-Reacting Confined Gel Space. ACS Appl. Mater. Interfaces 2019, 11, 47606–47618, . [CrossRef]
- Biswas, P.; Datta, H.K.; Dastidar, P. Multi-NSAID-Based Zn(II) Coordination Complex-Derived Metallogelators/Metallogels as Plausible Multi-Drug Self-Delivery Systems. Chem. Commun. 2022, 58, 969–972, . [CrossRef]
- Mondal, S.; Dastidar, P. Designing Metallogelators Derived from NSAID-Based Zn(II) Coordination Complexes for Drug-Delivery Applications. Chemistry – An Asian Journal 2020, 15, 3558–3567, . [CrossRef]
- Ruiz-Pérez, R.; Newman-Portela, A.M.; Ruiz-Fresneda, M.A. Emerging Global Trends in Next-Generation Alternatives to Classic Antibiotics for Combatting Multidrug-Resistant Bacteria. Journal of Cleaner Production 2024, 478, 143895, . [CrossRef]
- Wang, Y.; Zhang, M.; Yan, Z.; Ji, S.; Xiao, S.; Gao, J. Metal Nanoparticle Hybrid Hydrogels: The State-of-the-Art of Combining Hard and Soft Materials to Promote Wound Healing. Theranostics 2024, 14, 1534–1560, . [CrossRef]
- Clarkin, O.M.; Wu, B.; Cahill, P.A.; Brougham, D.F.; Banerjee, D.; Brady, S.A.; Fox, E.K.; Lally, C. Novel Injectable Gallium-Based Self-Setting Glass-Alginate Hydrogel Composite for Cardiovascular Tissue Engineering. Carbohydrate Polymers 2019, 217, 152–159, . [CrossRef]
- Brady, S.A.; Fox, E.K.; Lally, C.; Clarkin, O.M. Optimisation of a Novel Glass-Alginate Hydrogel for the Treatment of Intracranial Aneurysms. Carbohydrate Polymers 2017, 176, 227–235, . [CrossRef]
- Umesh; Chandran, V.C.; Saha, P.; Nath, D.; Bera, S.; Bhattacharya, S.; Pal, A. A Hydrogel Based on Fe(II)-GMP Demonstrates Tunable Emission, Self-Healing Mechanical Strength and Fenton Chemistry-Mediated Notable Antibacterial Properties. Nanoscale 2024, 16, 13050–13060, . [CrossRef]
- Dhibar, S.; Pal, S.; Some, S.; Karmakar, K.; Saha, R.; Bhattacharjee, S.; Kumari, D.; Mohan, A.; Ajiboye, T.O.; Ray, S.J.; et al. Exploring the Efficient Antimicrobial Applications of a Novel Supramolecular Hg(II)-Metallogel Derived from Succinic Acid Acting as a Low Molecular Weight Gelator. RSC Adv. 2025, 15, 5214–5219, . [CrossRef]
- Dhibar, S.; Pal, S.; Karmakar, K.; Hafiz, S.A.; Bhattacharjee, S.; Roy, A.; Rahaman, S.K.M.; Ray, S.J.; Dam, S.; Saha, B. Two Novel Low Molecular Weight Gelator-Driven Supramolecular Metallogels Efficient in Antimicrobial Activity Applications. RSC Adv. 2023, 13, 32842–32849, . [CrossRef]
- Pal, I.; Majumdar, S.; Lepcha, G.; Ahmed, K.T.; Yatirajula, S.K.; Bhattacharya, S.; Chakravarti, R.; Bhattacharya, B.; Biswas, S.R.; Dey, B. Exploration of Variable Solvent Directed Self-Healable Supramolecular M(II)-Metallogels (M = Co, Ni, Zn) of Azelaic Acid: Investigating Temperature-Dependent Ion Conductivity and Antibacterial Efficiency. ACS Appl Bio Mater 2023, 6, 5442–5457, . [CrossRef]
- Suberic Acid-Based Supramolecular Metallogels of Ni(II), Zn(II), and Cd(II) for Anti-Pathogenic Activity and Semiconducting Diode Fabrication | Langmuir Available online: https://pubs.acs.org/doi/10.1021/acs.langmuir.3c00765 (accessed on 17 October 2025).
- Bora, A.; Munjal, R.; Chakraborty, A.; Meena, L.; Chathangad, S.N.; Sadhukhan, S.; Mukhopadhyay, S. Design and Synthesis of Ibuprofen-Based Self-Deliverable Hybrid Gel Systems: Stabilization of Silver Nanoparticles, Antibacterial and Self-Healing Properties. ACS Appl Bio Mater 2025, 8, 6159–6176, . [CrossRef]
- Some, S.; Das, P.; Pal, S.; Dhibar, S.; Kumari, D.; Bhattacharjee, S.; Ray, S.J.; Ajiboye, T.O.; Dam, S.; Ray, P.P.; et al. Development of a Semiconducting Supramolecular Copper(II)–Metallogel for Antimicrobial and Microelectronic Device Applications. RSC Adv. 2025, 15, 18392–18402, . [CrossRef]
- Bhattacharya, S.; Bhattacharyya, T.; Khanra, S.; Banerjee, R.; Dash, J. Nucleoside-Derived Metallohydrogel Induces Cell Death in Leishmania Parasites. ACS Infect Dis 2023, 9, 1676–1684, . [CrossRef]
- Mikhailidi, A.; Volf, I.; Belosinschi, D.; Tofanica, B.-M.; Ungureanu, E. Cellulose-Based Metallogels—Part 1: Raw Materials and Preparation. Gels 2023, 9, 390, . [CrossRef]
- Kuosmanen, R.; Rissanen, K.; Sievänen, E. Steroidal Supramolecular Metallogels. Chemical Society Reviews 2020, 49, 1977–1998, . [CrossRef]
- Li, X.; Jin, Y.; Zhu, N.; Yin, J.; Jin, L.Y. Recent Developments of Fluorescence Sensors Constructed from Pillar[n]Arene-Based Supramolecular Architectures Containing Metal Coordination Sites. Sensors 2024, 24, 1530, . [CrossRef]
- Panja, S.; Panja, A.; Ghosh, K. Supramolecular Gels in Cyanide Sensing: A Review. Materials Chemistry Frontiers 2021, 5, 584–602, . [CrossRef]
- Moral, R.; Das, G. An Expedition towards Designing of Luminescent Supramolecular Metallogels for Chemosensing Applications. Inorganica Chimica Acta 2025, 581, 122635, . [CrossRef]
- Mehwish, N.; Dou, X.; Zhao, Y.; Feng, C.-L. Supramolecular Fluorescent Hydrogelators as Bio-Imaging Probes. Mater. Horiz. 2019, 6, 14–44, . [CrossRef]
- Das, A.K.; Gavel, P.K. Low Molecular Weight Self-Assembling Peptide-Based Materials for Cell Culture, Antimicrobial, Anti-Inflammatory, Wound Healing, Anticancer, Drug Delivery, Bioimaging and 3D Bioprinting Applications. Soft Matter 2020, 16, 10065–10095, . [CrossRef]
- Gnaim, S.; Scomparin, A.; Eldar-Boock, A.; Bauer, C.R.; Satchi-Fainaro, R.; Shabat, D. Light Emission Enhancement by Supramolecular Complexation of Chemiluminescence Probes Designed for Bioimaging. Chem. Sci. 2019, 10, 2945–2955, . [CrossRef]
- Zhang, B.; Dong, X.; Zhou, Q.; Lu, S.; Zhang, X.; Liao, Y.; Yang, Y.; Wang, H. Carboxymethyl Chitosan‒promoted Luminescence of Lanthanide Metallogel and Its Application in Assay of Multiple Metal Ions. Carbohydrate Polymers 2021, 263, 117986, . [CrossRef]
- Zhang, Y.-M.; Zhu, W.; Zhao, Q.; Qu, W.-J.; Yao, H.; Wei, T.-B.; Lin, Q. Th4+ Tuned Aggregation-Induced Emission: A Novel Strategy for Sequential Ultrasensitive Detection and Separation of Th4+ and Hg2. Spectrochim Acta A Mol Biomol Spectrosc 2020, 229, 117926, . [CrossRef]
- Wang, Y.; Lai, Y.; Ren, T.; Tang, J.; Gao, Y.; Geng, Y.; Zhang, J.; Ma, X. Construction of Artificial Light-Harvesting Systems Based on Aggregation-Induced Emission Type Supramolecular Self-Assembly Metallogels. Langmuir 2023, 39, 1103–1110, . [CrossRef]
- Photoresponsive Zn2+-Specific Metallohydrogels Coassembled from Imidazole Containing Phenylalanine and Arylazopyrazole Derivatives - Dalton Transactions (RSC Publishing) Available online: https://pubs.rsc.org/en/content/articlelanding/2020/dt/d0dt01809k (accessed on 17 October 2025).
- Zhang, B.; Dong, X.; Xiong, Y.; Zhou, Q.; Lu, S.; Liao, Y.; Yang, Y.; Wang, H. A Heat-Set Lanthanide Metallogel Capable of Emitting Stable Luminescence under Thermal, Mechanical and Water Stimuli. Dalton Trans. 2020, 49, 2827–2832, . [CrossRef]
- Zhang, Y.-J.; Shen, F.-J.; Li, Y.-J.; Pang, X.-L.; Zhang, C.; Ren, J.-J.; Yu, X.-D. A Zr-Cluster Based Thermostable, Self-Healing and Adaptive Metallogel with Chromogenic Properties Responds to Multiple Stimuli with Reversible Radical Interaction. Chem. Commun. 2020, 56, 2439–2442, . [CrossRef]
- Dey, B.; Saha, D.; Bhattacharjee, B.; Sadhu, S.; Dey, R.; Yatirajula, S.K.; Chakrovorty, A.; Samadder, A. Self-Repairing Metallosupramolecular Soft Flexible Scaffolds with Different Gel-Immobilized Media: Study of Rheo-Reversibile Systems toward Cytochrome c and Bcl2 Protein-Mediated Apoptotic Cascades in Cancer Cells. Langmuir 2025, 41, 21107–21124, . [CrossRef]
- Picci, G.; Caltagirone, C.; Garau, A.; Lippolis, V.; Milia, J.; Steed, J.W. Metal-Based Gels: Synthesis, Properties, and Applications. Coordination Chemistry Reviews 2023, 492, 215225, . [CrossRef]
- Janarthanan, G.; Noh, I. Recent Trends in Metal Ion Based Hydrogel Biomaterials for Tissue Engineering and Other Biomedical Applications. Journal of Materials Science & Technology 2021, 63, 35–53, . [CrossRef]
- Tian, R.; Qiu, X.; Yuan, P.; Lei, K.; Wang, L.; Bai, Y.; Liu, S.; Chen, X. Fabrication of Self-Healing Hydrogels with On-Demand Antimicrobial Activity and Sustained Biomolecule Release for Infected Skin Regeneration. ACS Appl. Mater. Interfaces 2018, 10, 17018–17027, . [CrossRef]
- Yavvari, P.S.; Pal, S.; Kumar, S.; Kar, A.; Awasthi, A.K.; Naaz, A.; Srivastava, A.; Bajaj, A. Injectable, Self-Healing Chimeric Catechol-Fe(III) Hydrogel for Localized Combination Cancer Therapy. ACS Biomater. Sci. Eng. 2017, 3, 3404–3413, . [CrossRef]
- Swamy, B.Y.; Yun, Y.-S. In Vitro Release of Metformin from Iron (III) Cross-Linked Alginate–Carboxymethyl Cellulose Hydrogel Beads. International Journal of Biological Macromolecules 2015, 77, 114–119, . [CrossRef]
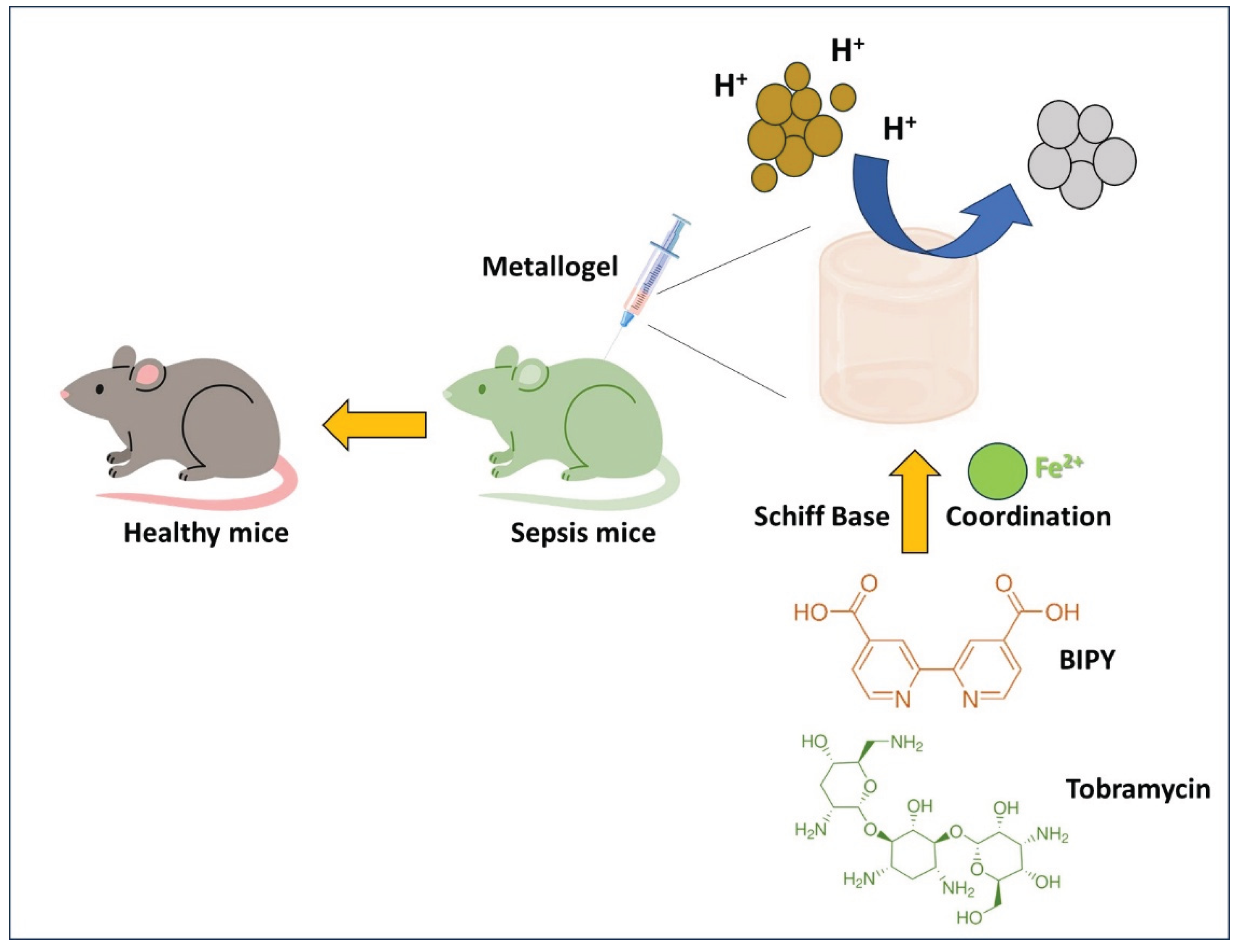
- Li, H.; Zhang, J.; Xue, H.; Li, L.; Liu, X.; Yang, L.; Gu, Z.; Cheng, Y.; Li, Y.; Huang, Q. An Injectable All-Small-Molecule Dynamic Metallogel for Suppressing Sepsis. Mater. Horiz. 2023, 10, 1789–1794, . [CrossRef]
- Su, W.-T.; Chou, W.-L.; Chou, C.-M. Osteoblastic Differentiation of Stem Cells from Human Exfoliated Deciduous Teeth Induced by Thermosensitive Hydrogels with Strontium Phosphate. Mater Sci Eng C Mater Biol Appl 2015, 52, 46–53, . [CrossRef]
|
System / Composition |
Metal center(s) | Key molecular components / mechanism | Stimuli / Functional Property and Key Application | Ref. |
| H₆L/Tb³⁺/CMCS hybrid metallogel | Tb³⁺ | 5,5′,5″-(1,3,5-triazine-2,4,6-triyl)tris(azanediyl)triisophthalate (H₆L) + carboxymethyl chitosan (CMCS) | pH-responsive luminescence (×9 quantum yield, lifetime). Photonic and biosensing metal-ion detection. | [79] |
| THTA-G / Th⁴⁺ metallogel | Th⁴⁺ / Hg²⁺ | Tripodal acylhydrazine (TH) + amide (TA) co-assembly | Th⁴⁺-triggered AIE “turn-on” fluorescence. Ion sensing and separation. | [80] |
| P–Al³⁺ metallogel (ALHS) | Al³⁺ | Bi-benzimidazole compound (P) + Rh6G / BODIPY energy acceptors | AIE and donor–acceptor energy transfer processes. Artificial Light-Harvesting Systems (ALHSs). | [81] |
| Zn²⁺-based hybrid photoresponsive metallogel | Zn²⁺ | Imidazole-functionalized phenylalanine (ImF) + arylazopyrazole (AzoPz) | UV light-triggered reversible gel–sol. Photo-responsive optical and controlled-release systems. | [82] |
| Heat-set H₆L/Tb³⁺ metallogel | Tb³⁺ | Multicarboxylate ligand H₆L | Anti-counterfeiting and optical sensing | [83] |
| Zr⁴⁺-cluster-based metallogel | Zr⁴⁺ | One-pot solvothermal synthesis; metal cluster coordination | Light-, amine-, electric-, and metal-responsiveness. Electrochromic devices and corrosion-sensing | [84] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





