Submitted:
09 November 2025
Posted:
10 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Research Strategy
3. Etiology and Assessment of Insulin Resistance
3.1. Causes of Insulin Resistance
3.2. Assessment of Insulin Resistance in Clinics and in Epidemiological Studies
3.3. Assessment of Insulin Resistance in the Diabetes Research Setting
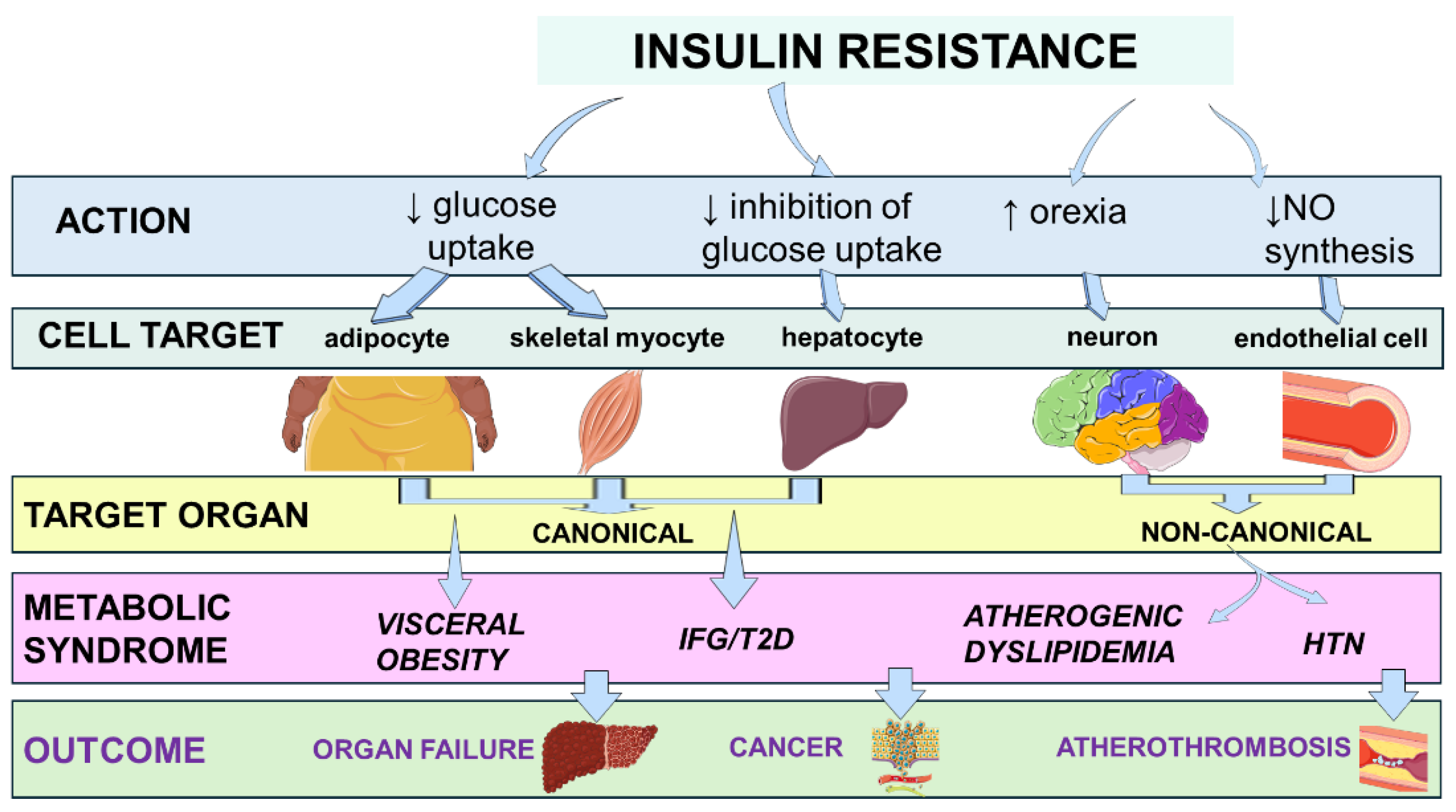
4. Molecular Physiology of Insulin Signaling and Mechanisms of Insulin Resistance
4.1. Inter-Organ Crosstalk: Hepatokines, Myokines, and Adipokines
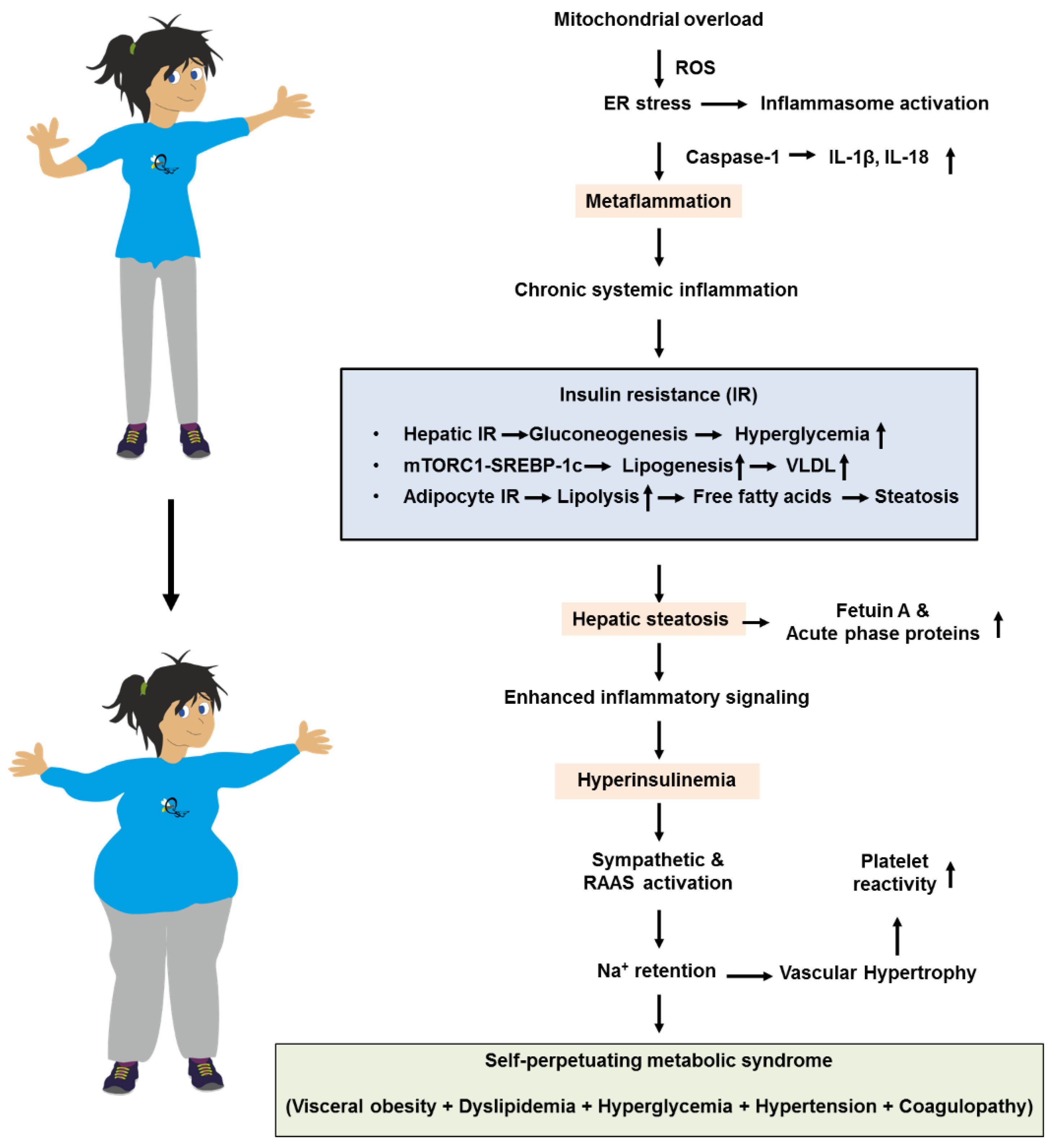
4.2. Lipotoxicity and the Self-Expanding Network of the Metabolic Syndrome
4.3. Drug-Induced Derailment of Insulin signaling
5. Pathobiology and Clinical Science Associating Ethnicity Insulin Resistance, Sex and Aging
5.1. Pathobiology Associating Insulin Resistance, Ethnicity Sex and Aging
5.2. Clinical Science Associating Insulin Resistance, Ethnicity, Sex, and Aging
6. Pathobiology and Clinical Science Associating Insulin Resistance and Metabolic Inflammation
6.1. Pathobiology Associating Insulin Resistance and Metabolic Inflammation
6.2. Clinical Science Associating Insulin Resistance and Metabolic Inflammation
7. Pathobiology and Clinical Science Associating Insulin Resistance and Cardiovascular Disease
7.1. Pathobiology Associating Insulin Resistance and Cardiovascular Disease
7.2. Clinical Science Associating Insulin Resistance and Cardiovascular Disease
8. Pathobiology and Clinical Science Associating Insulin Resistance and Organ Failure
8.1. Pathobiology Associating Insulin Resistance and Organ Failure
8.2. Clinical Science Associating Insulin Resistance and Organ Failure
9. Pathobiology and Clinical Science Associating Insulin Resistance and Cancer
9.1. Pathobiology Associating Insulin Resistance and Cancer
9.2. Clinical Science Associating Insulin Resistance and Cancer
10. Principles of Treatment of Insulin Resistance
11. Article Highlights
- Whenever supranormal insulin concentrations are required to elicit a quantitatively normal response in target tissues, IR is the underlying pathogenic defect.
- IR serves as a strategic point of intersection among the trajectories of clinically heterogeneous non-communicable diseases that are a significant public health issue globally.
- Organ failure (such as T2D, MASLD-cirrhosis, CKD, dementia, and heart failure), major adverse cardiovascular events, and cancer all share a common pathobiological denominator in IR.
- IR is strongly associated with perturbed cellular and sub-cellular components homeostasis, eventually leading to cell death and the activation of pro-fibrogenic pathways that may result in organ failure.
- Weight loss, regardless of how it is achieved, and insulin-sensitizing drugs may improve IR and potentially reverse T2D.
12. Conclusions and Research Agenda
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| BMI | body mass index |
| CKD | chronic kidney disease |
| CI | confidence interval |
| CPT1 | carnitine O-palmitoiltransferase 1 |
| CVD | cardiovascular disease |
| GLP-1RA | glucagon-like peptide receptor 1 agonist(s) |
| HOMA-IR | homeostasis model of insulin resistance |
| IL-6 | interleukin-6 |
| IR | insulin resistance |
| IRR | incidence rate ratio |
| ISR1/2 | insulin receptor substrate 1/2) |
| IST | insulin tolerance test |
| MACE | major adverse cardiovascular events |
| MASLD | metabolic dysfunction-associated steatotic liver disease |
| MASH | metabolic dysfunction-associated steatohepatitis |
| OR | odds ratio |
| PCOS | polycystic ovary syndrome |
| QUICKI | quantitative insulin sensitivity check index |
| ROS | reactive oxygen species |
| SGLT2 | sodium-glucose transporter 2 |
| SHBG | sex hormone binding globulin |
| SRRI | selective serotonin reuptake inhibitors |
| THR-β | thyroid hormone receptor-β |
| T2D | type 2 diabetes |
| TLR4 | toll-like receptor 4 |
| TyG | triglyceride-glucose index |
References
- Yalow, R.S.; Berson, S.A. Immunoassay of endogenous plasma insulin in man. J Clin Invest. 1960, 39, 1157–1175. [Google Scholar] [CrossRef]
- Yalow, R.S.; Berson, S.A. Plasma insulin concentrations in nondiabetic and early diabetic subjects. Determinations by a new sensitive immuno-assay technic. Diabetes 1960:254-60. [CrossRef]
- Aronis, K.N.; Mantzoros, C.S. A brief history of insulin resistance: from the first insulin radioimmunoassay to selectively targeting protein kinase C pathways. Metabolism 2012, 61, 445–449. [Google Scholar] [CrossRef]
- Kahn, C.R.; Neville DMJr Roth, J. Insulin-receptor interaction in the obese-hyperglycemic mouse. A model of insulin resistance. J Biol Chem. 1973, 248, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Goldfine, I.D.; Kahn, C.R.; Neville DMJr Roth, J.; Garrison, M.M.; Bates, R.W. Decreased binding of insulin to its receptors in rats with hormone induced insulin resistance. Biochem Biophys Res Commun. 1973, 53, 852–857. [Google Scholar] [CrossRef] [PubMed]
- Kahn, C.R.; Flier, J.S.; Bar, R.S.; Archer, J.A.; Gorden, P.; Martin, M.M.; Roth, J. The syndromes of insulin resistance and acanthosis nigricans. Insulin-receptor disorders in man. N Engl J Med. 1976, 294, 739–745. [Google Scholar] [CrossRef]
- Reaven, G.M. Banting lecture 1988. Role of insulin resistance in human disease. Diabetes 1988, 37, 1595–1607. [Google Scholar] [CrossRef]
- Muniyappa R, Chen H, Montagnani M, Sherman A, Quon MJ: Endothelial dysfunction due to selective insulin resistance in vascular endothelium: insights from mechanistic modeling. Am J Physiol Endocrinol Metab 2020, 319:E629–E646. [CrossRef]
- Batista TM, Haider N, Kahn CR: Defining the underlying defect in insulin action in type 2 diabetes. Diabetologia 2021, 64, 994–1006. [CrossRef]
- Andreadi, A.; Bellia, A.; Di Daniele, N.; Meloni, M.; Lauro, R.; Della-Morte, D.; Lauro, D. The molecular link between oxidative stress, insulin resistance, and type 2 diabetes: A target for new therapies against cardiovascular diseases. Curr Opin Pharmacol. 2022, 62, 85–96. [Google Scholar] [CrossRef]
- Giangregorio, F.; Mosconi, E.; Debellis, M.G.; Provini, S.; Esposito, C.; Garolfi, M.; et al. A Systematic Review of Metabolic Syndrome: Key Correlated Pathologies and Non-Invasive Diagnostic Approaches. J. Clin. Med. 2024, 13, 5880. [Google Scholar] [CrossRef] [PubMed]
- Zhan, Z.Q.; Chen, Y.Z.; Huang, Z.M.; Luo, Y.H.; Zeng, J.J.; Wang, Y.; Tan, J.; Chen, Y.X.; Fang, J.Y. Metabolic syndrome, its components, and gastrointestinal cancer risk: a meta-analysis of 31 prospective cohorts and Mendelian randomization study. J Gastroenterol Hepatol. 2024, 39, 630–641. [Google Scholar] [CrossRef]
- Hamooya, B.M.; Siame, L.; Muchaili, L.; Masenga, S.K.; Kirabo, A. Metabolic syndrome: epidemiology, mechanisms, and current therapeutic approaches. Front Nutr. 2025, 12, 1661603. [Google Scholar] [CrossRef] [PubMed]
- Lonardo, A.; Ballestri, S.; Marchesini, G.; Angulo, P.; Loria, P. Nonalcoholic fatty liver disease: a precursor of the metabolic syndrome. Dig Liver Dis. 2015, 47, 181–190. [Google Scholar] [CrossRef]
- Kosmas, C.E.; Bousvarou, M.D.; Kostara, C.E.; Papakonstantinou, E.J.; Salamou, E.; Guzman, E. Insulin resistance and cardiovascular disease. J Int Med Res. 2023, 51, 3000605231164548. [Google Scholar] [CrossRef] [PubMed]
- Subedi, B.K.; Bhimineni, C.; Modi, S.; Jahanshahi, A.; Quiza, K.; Bitetto, D. The Role of Insulin Resistance in Cancer. Curr Oncol. 2025, 32, 477. [Google Scholar] [CrossRef] [PubMed]
- Nzobokela, J.; Muchaili, L.; Mwambungu, A.; Masenga, S.K.; Kirabo, A. Pathophysiology and emerging biomarkers of cardiovascular-renal-hepato-metabolic syndrome. Front. Cardiovasc. Med. 2025, 12, 1661563. [Google Scholar] [CrossRef]
- Lonardo, A. The heterogeneity of metabolic syndrome presentation and challenges this causes in its pharmacological management: a narrative review focusing on principal risk modifiers. Expert Rev Clin Pharmacol. 2023, 16, 891–911. [Google Scholar] [CrossRef]
- Abdullah, M.Y.; Alruwaili, Y.M.; Alsairra, M.N.; Almnaa, S.A.; Mosly, A.F.; Lasuwaidan, D.T.; et al. The Economic and Social Burden of Insulin Resistance in Obesity. Journal of Healthcare Sciences 2024, 4, JOHS2024000873. [Google Scholar] [CrossRef]
- Sandri, E.; Piredda, M.; Sguanci, M.; Mancin, S. What Factors Influence Obesity in Spain? A Multivariate Analysis of Sociodemographic, Nutritional, and Lifestyle Factors Affecting Body Mass Index in the Spanish Population. Healthcare (Basel) 2025, 13, 386. [Google Scholar] [CrossRef]
- Freeman, A.M.; Acevedo, L.A.; Pennings, N. Insulin Resistance. [Updated 2023 Aug 17]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK507839/].
- Medscape. Updated Nov, 2024. https://emedicine.medscape.com/article/122501-overview. Last accessed 6 November, 2025.
- Ramos de Mendonça, C.; Cristina Monteiro Galindo, L.; Karoline Alves de Melo Silva, B.; Hilary Avelino de Vasconcelos, B.; de Araújo Bandeira, V.C.; Massao Hirabara, S.; et al. Maternal obesogenic diet causes insulin resistance by modulating insulin signaling pathways in peripheral tissues of offspring: a systematic review. Life Sci. 2025, 380, 123947. [Google Scholar] [CrossRef]
- Kim, T.W.; Jeong, J.H.; Hong, S.C. The impact of sleep and circadian disturbance on hormones and metabolism. Int J Endocrinol. 2015, 2015, 591729. [Google Scholar] [CrossRef]
- Guaraldi, G.; Lonardo, A.; Ballestri, S.; Zona, S.; Stentarelli, C.; Orlando, G.; Carli, F.; Carulli, L.; Roverato, A.; Loria, P. Human immunodeficiency virus is the major determinant of steatosis and hepatitis C virus of insulin resistance in virus-associated fatty liver disease. Arch Med Res. 2011, 42, 690–697. [Google Scholar] [CrossRef]
- Pedro, M.N.; Rocha, G.Z.; Guadagnini, D.; Santos, A.; Magro, D.O.; Assalin, H.B.; Oliveira, A.G.; Pedro, R.J.; Saad, M.J.A. Insulin Resistance in HIV-Patients: Causes and Consequences. Front Endocrinol (Lausanne) 2018, 9, 514. [Google Scholar] [CrossRef] [PubMed]
- Eliasson, B.; Smith, U. Cigarrettrökning ger insulinresistens. Nya rön om tobakens metabola effekter [Cigarette smoking causes insulin resistance. New findings on metabolic effects of tobacco]. Lakartidningen. 1995, 92, 731–733. [Google Scholar]
- Swarup, S.; Ahmed, I.; Grigorova, Y.; et al. Metabolic Syndrome. [Updated 2024 Mar 7]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK459248/.
- Amisi, C.A. Markers of insulin resistance in Polycystic ovary syndrome women: An update. World J Diabetes 2022, 13, 129–149. [Google Scholar] [CrossRef]
- Shukla, A.; Rasquin, L.I.; Anastasopoulou, C. Polycystic Ovarian Syndrome. [Updated 2025 Jul 7]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK459251/.
- Madan R, Varghese RT; Ranganath. Assessing Insulin Sensitivity and Resistance in Humans. [Updated 2024 Oct 16]. In: Feingold KR, Ahmed SF, Anawalt B, et al., editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK278954/.
- Vaidya, R.A.; Desai, S.; Moitra, P.; Salis, S.; Agashe, S.; Battalwar, R.; et al. Hyperinsulinemia: an early biomarker of metabolic dysfunction. Front Clin Diabetes Healthc. 2023, 4, 1159664. [Google Scholar] [CrossRef]
- Jarvis, P.R.E.; Cardin, J.L.; Nisevich-Bede, P.M.; McCarter, J.P. Continuous glucose monitoring in a healthy population: understanding the post-prandial glycemic response in individuals without diabetes mellitus. Metabolism 2023, 146, 155640. [Google Scholar] [CrossRef]
- Zubair, M.; Launico, M.V. Glucose Tolerance Test. [Updated 2025 Sep 15]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK532915/.
- Sun, Y.; Ji, H.; Sun, W.; An, X.; Lian, F. Triglyceride glucose (TyG) index: A promising biomarker for diagnosis and treatment of different diseases. Eur J Intern Med. 2025, 131, 3–14. [Google Scholar] [CrossRef]
- Ozturk, D.; Sivaslioglu, A.; Bulus, H.; Ozturk, B. TyG index is positively associated with HOMA-IR in cholelithiasis patients with insulin resistance: Based on a retrospective observational study. Asian J Surg. 2024, 47, 2579–2583. [Google Scholar] [CrossRef]
- Avagimyan, A.; Pogosova, N.; Fogacci, F.; Aghajanova, E.; Djndoyan, Z.; Patoulias, D.; et al. Triglyceride-glucose index (TyG) as a novel biomarker in the era of cardiometabolic medicine. Int J Cardiol. 2025, 418, 132663. [Google Scholar] [CrossRef] [PubMed]
- Salazar, J.; Bermúdez, V.; Calvo, M.; Olivar, L.C.; Luzardo, E.; Navarro, C.; et al. Optimal cutoff for the evaluation of insulin resistance through triglyceride-glucose index: A cross-sectional study in a Venezuelan population. F1000Res. 2017, 6, 1337. [Google Scholar] [CrossRef] [PubMed]
- de Cassia da Silva, C.; Zambon, M.P.; Vasques, A.C.J.; Camilo, D.F.; de Góes Monteiro Antonio, M.Â.R.; Geloneze, B. The threshold value for identifying insulin resistance (HOMA-IR) in an admixed adolescent population: A hyperglycemic clamp validated study. Arch Endocrinol Metab. 2023, 67, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Moon, S.; Park, J.H.; Jang, E.J.; Park, Y.K.; Yu, J.M.; Park, J.S.; et al. The Cut-off Values of Surrogate Measures for Insulin Sensitivity in a Healthy Population in Korea according to the Korean National Health and Nutrition Examination Survey (KNHANES) 2007-2010. J Korean Med Sci. 2018, 33, e197. [Google Scholar] [CrossRef]
- Beaudry, M.; Bissonnette, S.; Lamantia, V.; Devaux, M.; Faraj, M. Sex-Specific Models to Predict Insulin Secretion and Sensitivity in Subjects with Overweight and Obesity. Int J Mol Sci. 2023, 24, 6130. [Google Scholar] [CrossRef]
- Tam, C.S.; Xie, W.; Johnson, W.D.; Cefalu, W.T.; Redman, L.M.; Ravussin, E. Defining insulin resistance from hyperinsulinemic-euglycemic clamps. Diabetes Care 2012, 35, 1605–1610. [Google Scholar] [CrossRef]
- Mari, A.; Pacini, G.; Brazzale, A.R.; Ahrén, B. Comparative evaluation of simple insulin sensitivity methods based on the oral glucose tolerance test. Diabetologia 2005, 48, 748–751. [Google Scholar] [CrossRef] [PubMed]
- Bo, T.; Gao, L.; Yao, Z.; Shao, S.; Wang, X.; Proud, C.G.; Zhao, J. Hepatic selective insulin resistance at the intersection of insulin signaling and metabolic dysfunction-associated steatotic liver disease. Cell Metab. 2024, 36, 947–968. [Google Scholar] [CrossRef] [PubMed]
- Iglesias, P. The endocrine role of hepatokines: implications for human health and disease. Front Endocrinol (Lausanne) 2025, 16, 1663353. [Google Scholar] [CrossRef]
- Calcaterra, V.; Magenes, V.C.; Bianchi, A.; Rossi, V.; Gatti, A.; Marin, L.; Vandoni, M.; Zuccotti, G. How Can Promoting Skeletal Muscle Health and Exercise in Children and Adolescents Prevent Insulin Resistance and Type 2 Diabetes? Life (Basel) 2024, 14, 1198. [Google Scholar] [CrossRef]
- Consitt, L.A.; Clark, B.C. The Vicious Cycle of Myostatin Signaling in Sarcopenic Obesity: Myostatin Role in Skeletal Muscle Growth, Insulin Signaling and Implications for Clinical Trials. J Frailty Aging 2018, 7, 21–27. [Google Scholar] [CrossRef]
- Choi, H.M.; Doss, H.M.; Kim, K.S. Multifaceted Physiological Roles of Adiponectin in Inflammation and Diseases. Int J Mol Sci. 2020, 21, 1219. [Google Scholar] [CrossRef]
- Schon, H.T.; Weiskirchen, R. Exercise-Induced Release of Pharmacologically Active Substances and Their Relevance for Therapy of Hepatic Injury. Front Pharmacol. 2016, 7, 283. [Google Scholar] [CrossRef]
- Chait, A.; den Hartigh, L.J. Adipose Tissue Distribution, Inflammation and Its Metabolic Consequences, Including Diabetes and Cardiovascular Disease. Front Cardiovasc Med. 2020, 7, 22. [Google Scholar] [CrossRef]
- Choi, W.; Woo, G.H.; Kwon, T.H.; Jeon, J.H. Obesity-Driven Metabolic Disorders: The Interplay of Inflammation and Mitochondrial Dysfunction. Int J Mol Sci. 2025, 26, 9715. [Google Scholar] [CrossRef]
- Chandrasekaran, P.; Weiskirchen, R. The Role of SCAP/SREBP as Central Regulators of Lipid Metabolism in Hepatic Steatosis. Int J Mol Sci. 2024, 25, 1109. [Google Scholar] [CrossRef]
- Masson, W.; Lobo, M.; Siniawski, D.; Huerín, M.; Molinero, G.; Valéro, R.; Nogueira, J.P. Therapy with cholesteryl ester transfer protein (CETP) inhibitors and diabetes risk. Diabetes Metab. 2018, 44, 508–513. [Google Scholar] [CrossRef] [PubMed]
- da Silva, A.A.; do Carmo, J.M.; Li, X.; Wang, Z.; Mouton, A.J.; Hall, J.E. Role of Hyperinsulinemia and Insulin Resistance in Hypertension: Metabolic Syndrome Revisited. Can J Cardiol. 2020, 36, 671–682. [Google Scholar] [CrossRef] [PubMed]
- Kuo, T.; McQueen, A.; Chen, T.C.; Wang, J.C. Regulation of Glucose Homeostasis by Glucocorticoids. Adv Exp Med Biol. 2015, 872, 99–126. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Yao, H.; Sun, Z.; Wang, Y.; Zhao, Y.; Wang, Z.; Li, L. Role of NFAT in the Progression of Diabetic Atherosclerosis. Front Cardiovasc Med. 2021, 8, 635172. [Google Scholar] [CrossRef]
- Chakkera, H.A.; Mandarino, L.J. Calcineurin inhibition and new-onset diabetes mellitus after transplantation. Transplantation. 2013, 95, 647–652. [Google Scholar] [CrossRef]
- Murata, H.; Hruz, P.W.; Mueckler, M. The mechanism of insulin resistance caused by HIV protease inhibitor therapy. J Biol Chem. 2000, 275, 20251–20254. [Google Scholar] [CrossRef]
- Hertel, J.; Struthers, H.; Horj, C.B.; Hruz, P.W. A structural basis for the acute effects of HIV protease inhibitors on GLUT4 intrinsic activity. J Biol Chem. 2004, 279, 55147–55152. [Google Scholar] [CrossRef]
- Xu, H.; Zhuang, X. Atypical antipsychotics-induced metabolic syndrome and nonalcoholic fatty liver disease: a critical review. Neuropsychiatr Dis Treat. 2019, 15, 2087–2099. [Google Scholar] [CrossRef]
- Li, M.; Chi, X.; Wang, Y.; Setrerrahmane, S.; Xie, W.; Xu, H. Trends in insulin resistance: insights into mechanisms and therapeutic strategy. Signal Transduct Target Ther. 2022, 7, 216. [Google Scholar] [CrossRef]
- Parvaresh Rizi, E.; Teo, Y.; Leow, M.K.; Venkataraman, K.; Khoo, E.Y.; Yeo, C.R.; Chan, E.; Song, T.; Sadananthan, S.A.; Velan, S.S.; Gluckman, P.D.; Lee, Y.S.; Chong, Y.S.; Tai, E.S.; Toh, S.A.; Khoo, C.M. Ethnic Differences in the Role of Adipocytokines Linking Abdominal Adiposity and Insulin Sensitivity Among Asians. J Clin Endocrinol Metab. 2015, 100, 4249–4256. [Google Scholar] [CrossRef] [PubMed]
- Chávez-Guevara, I.A.; Amaro-Gahete, F.J.; Osuna-Prieto, F.J.; Labayen, I.; Aguilera, C.M.; Ruiz, J.R. The role of sex in the relationship between fasting adipokines levels, maximal fat oxidation during exercise, and insulin resistance in young adults with excess adiposity. Biochem Pharmacol. 2023, 216, 115757. [Google Scholar] [CrossRef]
- Weiskirchen, R.; Lonardo, A. Sex Hormones and Metabolic Dysfunction-Associated Steatotic Liver Disease. Int J Mol Sci. 2025, 26, 9594. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh Pahlavani, H.; Laher, I.; Knechtle, B.; Zouhal, H. Exercise and mitochondrial mechanisms in patients with sarcopenia. Front Physiol. 2022, 13, 1040381. [Google Scholar] [CrossRef]
- Raygor, V.; Abbasi, F.; Lazzeroni, L.C.; Kim, S.; Ingelsson, E.; Reaven, G.M.; Knowles, J.W. Impact of race/ethnicity on insulin resistance and hypertriglyceridaemia. Diab Vasc Dis Res. 2019, 16, 153–159. [Google Scholar] [CrossRef]
- Vasishta, S.; Ganesh, K.; Umakanth, S.; Joshi, M.B. Ethnic disparities attributed to the manifestation in and response to type 2 diabetes: insights from metabolomics. Metabolomics 2022, 18, 45. [Google Scholar] [CrossRef] [PubMed]
- Palmer, A.K.; Jensen, M.D. Metabolic changes in aging humans: current evidence and therapeutic strategies. J Clin Invest. 2022, 132, e158451. [Google Scholar] [CrossRef] [PubMed]
- Shou, J.; Chen, P.J.; Xiao, W.H. Mechanism of increased risk of insulin resistance in aging skeletal muscle. Diabetol Metab Syndr. 2020, 12, 14. [Google Scholar] [CrossRef] [PubMed]
- Ciarambino, T.; Crispino, P.; Guarisco, G.; Giordano, M. Gender Differences in Insulin Resistance: New Knowledge and Perspectives. Curr Issues Mol Biol. 2023, 45, 7845–7861. [Google Scholar] [CrossRef]
- Lonardo, A.; Jamalinia, M.; Weiskirchen, R. Sex differences in MASLD. [CrossRef]
- Zhao, J.; Fan, H.; Wang, T.; Yu, B.; Mao, S.; Wang, X.; et al. TyG index is positively associated with risk of CHD and coronary atherosclerosis severity among NAFLD patients. Cardiovasc Diabetol. 2022, 21, 123. [Google Scholar] [CrossRef]
- El Assar, M.; Angulo, J.; Carnicero, J.A.; Molina-Baena, B.; García-García, F.J.; Sosa, P.; Rodríguez-Mañas, L. Gender-specific capacity of insulin resistance proxies to predict functional decline in older adults. J Nutr Health Aging 2024, 28, 100376. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Wu, D.; Qiu, Y. Adipose tissue macrophage in obesity-associated metabolic diseases. Front Immunol. 2022, 13, 977485. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Leung, J.C.K.; Chan, L.Y.Y.; Yiu, W.H.; Tang, S.C.W. A global perspective on the crosstalk between saturated fatty acids and Toll-like receptor 4 in the etiology of inflammation and insulin resistance. Prog Lipid Res. 2020, 77, 101020. [Google Scholar] [CrossRef]
- Misceo, D.; Mocciaro, G.; D'Amore, S.; Vacca, M. Diverting hepatic lipid fluxes with lifestyles revision and pharmacological interventions as a strategy to tackle steatotic liver disease (SLD) and hepatocellular carcinoma (HCC). Nutr Metab (Lond). 2024, 21, 112. [Google Scholar] [CrossRef]
- Pal, D.; Dasgupta, S.; Kundu, R.; Maitra, S.; Das, G.; Mukhopadhyay, S.; Ray, S.; Majumdar, S.S.; Bhattacharya, S. Fetuin-A acts as an endogenous ligand of TLR4 to promote lipid-induced insulin resistance. Nat Med. 2012, 18, 1279–1285. [Google Scholar] [CrossRef]
- Balakrishnan, R.; Thurmond, D.C. Mechanisms by Which Skeletal Muscle Myokines Ameliorate Insulin Resistance. Int J Mol Sci. 2022, 23, 4636. [Google Scholar] [CrossRef]
- Crasan, I.-M.; Tanase, M.; Delia, C.E.; Gradisteanu-Pircalabioru, G.; Cimpean, A.; Ionica, E. Metaflammation’s Role in Systemic Dysfunction in Obesity: A Comprehensive Review. Int. J. Mol. Sci. 2025, 26, 10445. [Google Scholar] [CrossRef]
- Khanna, D.; Khanna, S.; Khanna, P.; Kahar, P.; Patel, B.M. Obesity: A Chronic Low-Grade Inflammation and Its Markers. Cureus. 2022, 14, e22711. [Google Scholar] [CrossRef]
- Kirichenko, T.V.; Markina, Y.V.; Bogatyreva, A.I.; Tolstik, T.V.; Varaeva, Y.R.; Starodubova, A.V. The Role of Adipokines in Inflammatory Mechanisms of Obesity. Int J Mol Sci. 2022, 23, 14982. [Google Scholar] [CrossRef]
- Janssen, J.A.M.J.L. The Pivotal Role of the Western Diet, Hyperinsulinemia, Ectopic Fat, and Diacylglycerol-Mediated Insulin Resistance in Type 2 Diabetes. Int J Mol Sci. 2025, 26, 9191. [Google Scholar] [CrossRef]
- Hossain, I.A.; Akter, S.; Bhuiyan, F.R.; Shah, M.R.; Rahman, M.K.; Ali, L. Subclinical inflammation in relation to insulin resistance in prediabetic subjects with nonalcoholic fatty liver disease. BMC Res Notes 2016, 9, 266. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Li, S.; Liu, Y.; Bazzano, L.; He, J.; Mi, J.; Chen, W. Temporal relationship between inflammation and insulin resistance and their joint effect on hyperglycemia: the Bogalusa Heart Study. Cardiovasc Diabetol. 2019, 18, 109. [Google Scholar] [CrossRef] [PubMed]
- Symons, J.D.; McMillin, S.L.; Riehle, C.; Tanner, J.; Palionyte, M.; Hillas, E.; Jones, D.; Cooksey, R.C.; Birnbaum, M.J.; McClain, D.A.; Zhang, Q.J.; Gale, D.; Wilson, L.J.; Abel, E.D. Contribution of insulin and Akt1 signaling to endothelial nitric oxide synthase in the regulation of endothelial function and blood pressure. Circ Res. 2009, 104, 1085–1094. [Google Scholar] [CrossRef]
- De Pascali, F.; Hemann, C.; Samons, K.; Chen, C.A.; Zweier, J.L. Hypoxia and reoxygenation induce endothelial nitric oxide synthase uncoupling in endothelial cells through tetrahydrobiopterin depletion and S-glutathionylation. Biochemistry 2014, 53, 3679–3688. [Google Scholar] [CrossRef] [PubMed]
- Gao, F.; Lucke-Wold, B.P.; Li, X.; Logsdon, A.F.; Xu, L.C.; Xu, S.; LaPenna, K.B.; Wang, H.; Talukder, M.A.H.; Siedlecki, C.A.; Huber, J.D.; Rosen, C.L.; He, P. Reduction of Endothelial Nitric Oxide Increases the Adhesiveness of Constitutive Endothelial Membrane ICAM-1 through Src-Mediated Phosphorylation. Front Physiol. 2018, 8, 1124. [Google Scholar] [CrossRef]
- Park, Y.M.; RKashyap, S.; AMajor, J.; Silverstein, R.L. Insulin promotes macrophage foam cell formation: potential implications in diabetes-related atherosclerosis. Lab Invest. 2012, 92, 1171–1180. [Google Scholar] [CrossRef]
- Cerf, M.E. Cardiac Glucolipotoxicity and Cardiovascular Outcomes. Medicina (Kaunas) 2018, 54, 70. [Google Scholar] [CrossRef]
- Sorop, O.; van de Wouw, J.; Chandler, S.; Ohanyan, V.; Tune, J.D.; Chilian, W.M.; Merkus, D.; Bender, S.B.; Duncker, D.J. Experimental animal models of coronary microvascular dysfunction. Cardiovasc Res. 2020, 116, 756–770. [Google Scholar] [CrossRef]
- D'Amario, D.; Migliaro, S.; Borovac, J.A.; Restivo, A.; Vergallo, R.; Galli, M.; Leone, A.M.; Montone, R.A.; Niccoli, G.; Aspromonte, N.; Crea, F. Microvascular Dysfunction in Heart Failure With Preserved Ejection Fraction. Front Physiol. 2019, 10, 1347. [Google Scholar] [CrossRef] [PubMed]
- Thorp, A.A.; Schlaich, M.P. Relevance of Sympathetic Nervous System Activation in Obesity and Metabolic Syndrome. J Diabetes Res. 2015, 2015, 341583. [Google Scholar] [CrossRef] [PubMed]
- Shan, P.F.; Li, Q.; Khamaisi, M.; Qiang, G.F. Type 2 Diabetes Mellitus and Macrovascular Complications. Int J Endocrinol. 2017, 2017, 4301461. [Google Scholar] [CrossRef]
- Di Pino, A.; DeFronzo, R.A. Insulin Resistance and Atherosclerosis: Implications for Insulin-Sensitizing Agents. Endocr Rev. 2019, 40, 1447–1467. [Google Scholar] [CrossRef]
- Salvia, M.G. The Look AHEAD Trial: Translating Lessons Learned Into Clinical Practice and Further Study. Diabetes Spectr. 2017, 30, 166–170. [Google Scholar] [CrossRef]
- Balducci, S.; Sacchetti, M.; Haxhi, J.; Orlando, G.; Zanuso, S.; Cardelli, P.; et al. The Italian Diabetes and Exercise Study 2 (IDES-2): a long-term behavioral intervention for adoption and maintenance of a physically active lifestyle. Trials 2015, 16, 569. [Google Scholar] [CrossRef]
- Carbone, S.; Dixon, D.L.; Buckley, L.F.; Abbate, A. Glucose-Lowering Therapies for Cardiovascular Risk Reduction in Type 2 Diabetes Mellitus: State-of-the-Art Review. Mayo Clin Proc. 2018, 93, 1629–1647. [Google Scholar] [CrossRef] [PubMed]
- English, W.J.; Spann, M.D.; Aher, C.V.; Williams, D.B. Cardiovascular risk reduction following metabolic and bariatric surgery. Ann Transl Med. 2020, 8 (Suppl. S1), S12. [Google Scholar] [CrossRef]
- Joseph, J.J.; Deedwania, P.; Acharya, T.; Aguilar, D.; Bhatt, D.L.; Chyun, D.A.; et al. Comprehensive Management of Cardiovascular Risk Factors for Adults With Type 2 Diabetes: A Scientific Statement From the American Heart Association. Circulation 2022, 145, e722–e759. [Google Scholar] [CrossRef]
- Prentki, M.; Peyot, M.L.; Masiello, P.; Madiraju, S.R.M. Nutrient-Induced Metabolic Stress, Adaptation, Detoxification, and Toxicity in the Pancreatic β-Cell. Diabetes 2020, 69, 279–290. [Google Scholar] [CrossRef]
- Yu, T.; Robotham, J.L.; Yoon, Y. Increased production of reactive oxygen species in hyperglycemic conditions requires dynamic change of mitochondrial morphology. Proc Natl Acad Sci U S A 2006, 103, 2653–2658. [Google Scholar] [CrossRef]
- Lu, X.; Xie, Q.; Pan, X.; Zhang, R.; Zhang, X.; Peng, G.; Zhang, Y.; Shen, S.; Tong, N. Type 2 diabetes mellitus in adults: pathogenesis, prevention and therapy. Signal Transduct Target Ther. 2024, 9, 262. [Google Scholar] [CrossRef]
- Tang, S.P.; Mao, X.L.; Chen, Y.H.; Yan, L.L.; Ye, L.P.; Li, S.W. Reactive Oxygen Species Induce Fatty Liver and Ischemia-Reperfusion Injury by Promoting Inflammation and Cell Death. Front Immunol. 2022, 13, 870239. [Google Scholar] [CrossRef]
- Menendez-Montes, I.; Abdisalaam, S.; Xiao, F.; Lam, N.T.; Mukherjee, S.; Szweda, L.I.; Asaithamby, A.; Sadek, H.A. Mitochondrial fatty acid utilization increases chromatin oxidative stress in cardiomyocytes. Proc Natl Acad Sci U S A 2021, 118, e2101674118. [Google Scholar] [CrossRef]
- Mackesy, D.Z.; Goalstone, M.L. Insulin augments tumor necrosis factor-alpha stimulated expression of vascular cell adhesion molecule-1 in vascular endothelial cells. J Inflamm (Lond). 2011, 8, 34. [Google Scholar] [CrossRef] [PubMed]
- Kasuga, M. Insulin resistance and pancreatic beta cell failure. J Clin Invest. 2006, 116, 1756–1760. [Google Scholar] [CrossRef] [PubMed]
- Mezza, T.; Cinti, F.; Cefalo, C.M.A.; Pontecorvi, A.; Kulkarni, R.N.; Giaccari, A. β-Cell Fate in Human Insulin Resistance and Type 2 Diabetes: A Perspective on Islet Plasticity. Diabetes 2019, 68, 1121–1129. [Google Scholar] [CrossRef]
- Gloyn, A.L.; Ibberson, M.; Marchetti, P.; Powers, A.C.; Rorsman, P.; Sander, M.; Solimena, M. Every islet matters: improving the impact of human islet research. Nat Metab. 2022, 4, 970–977. [Google Scholar] [CrossRef]
- Prentki, M.; Nolan, C.J. Islet beta cell failure in type 2 diabetes. J Clin Invest. 2006, 116, 1802–1812. [Google Scholar] [CrossRef] [PubMed]
- Alejandro, E.U.; Gregg, B.; Blandino-Rosano, M.; Cras-Méneur, C.; Bernal-Mizrachi, E. Natural history of β-cell adaptation and failure in type 2 diabetes. Mol Aspects Med. 2015, 42, 19–41. [Google Scholar] [CrossRef]
- Lonardo, A.; Weiskirchen, R. Liver and obesity: a narrative review. Explor Med. 2025, 6, 1001334. [Google Scholar] [CrossRef]
- Lonardo, A.; Weiskirchen, R. PPARs in molecular pathogenesis and drug treatment of type 2 diabetes-related MASLD. Explor Dig Dis. 2025, 4, 100590. [Google Scholar] [CrossRef]
- Ioannou, G.N.; Weiss, N.S.; Kowdley, K.V.; Dominitz, J.A. Is obesity a risk factor for cirrhosis-related death or hospitalization? A population-based cohort study. Gastroenterology 2003, 125, 1053–1059. [Google Scholar] [CrossRef] [PubMed]
- Ahn, S.B.; Powell, E.E.; Russell, A.; Hartel, G.; Irvine, K.M.; Moser, C.; Valery, P.C. Type 2 Diabetes: A Risk Factor for Hospital Readmissions and Mortality in Australian Patients With Cirrhosis. Hepatol Commun. 2020, 4, 1279–1292. [Google Scholar] [CrossRef] [PubMed]
- Pinto, K.R.D.; Feckinghaus, C.M.; Hirakata, V.N. Obesity as a predictive factor for chronic kidney disease in adults: systematic review and meta-analysis. Braz J Med Biol Res. 2021, 54, e10022. [Google Scholar] [CrossRef]
- Fenta, E.T.; Eshetu, H.B.; Kebede, N.; et al. Prevalence and predictors of chronic kidney disease among type 2 diabetic patients worldwide, systematic review and meta-analysis. Diabetol Metab Syndr 2023, 15, 245. [Google Scholar] [CrossRef]
- Lonardo, A.; Mantovani, A.; Targher, G.; Baffy, G. Nonalcoholic Fatty Liver Disease and Chronic Kidney Disease: Epidemiology, Pathogenesis, and Clinical and Research Implications. Int. J. Mol. Sci. 2022, 23, 13320. [Google Scholar] [CrossRef]
- Lonardo, A. Association of NAFLD/NASH, and MAFLD/MASLD with chronic kidney disease: an updated narrative review. Metab Target Organ Damage 2024, 4, 16. [Google Scholar] [CrossRef]
- Yoon, J.H.; Hwang, J.; Son, S.U.; Choi, J.; You, S.W.; Park, H.; Cha, S.Y.; Maeng, S. How Can Insulin Resistance Cause Alzheimer's Disease? Int J Mol Sci. 2023, 24, 3506. [Google Scholar] [CrossRef]
- Wang, F.; Huang, W.; Wang, Z.; Zhang, D.; Qiao, H.; Chen, N.; Ni, X.; Cheng, J.; Ni, W. Insulin resistance surrogates predict major adverse cardiovascular events in patients with heart failure with preserved ejection fraction and chronic kidney disease: a retrospective cohort study. Lipids Health Dis. 2025, 24, 349. [Google Scholar] [CrossRef]
- Ni, W. , Jiang, R., Xu, D. et al. Association between insulin resistance indices and outcomes in patients with heart failure with preserved ejection fraction. Cardiovasc Diabetol 2025, 24, 32. [Google Scholar] [CrossRef]
- Targher, G.; Byrne, C.D.; Lonardo, A.; Zoppini, G.; Barbui, C. Non-alcoholic fatty liver disease and risk of incident cardiovascular disease: A meta-analysis. J Hepatol. 2016, 65, 589–600. [Google Scholar] [CrossRef]
- Jamalinia, M.; Zare, F.; Lonardo, A. Liver fibrosis and risk of incident dementia in the general population: Systematic review with meta-analysis. American Journal of Preventive Medicine, in press.
- Fazio, S.; Bellavite, P.; Affuso, F. Chronically Increased Levels of Circulating Insulin Secondary to Insulin Resistance: A Silent Killer. Biomedicines 2024, 12, 2416. [Google Scholar] [CrossRef] [PubMed]
- Stanciu, S.M.; Jinga, M.; Miricescu, D.; Stefani, C.; Nica, R.I.; Stanescu-Spinu, I.I.; Vacaroiu, I.A.; Greabu, M.; Nica, S. mTOR Dysregulation, Insulin Resistance, and Hypertension. Biomedicines 2024, 12, 1802. [Google Scholar] [CrossRef]
- Boucher, J.; Kleinridders, A.; Kahn, C.R. Insulin receptor signaling in normal and insulin-resistant states. Cold Spring Harb Perspect Biol. 2014, 6, a009191. [Google Scholar] [CrossRef] [PubMed]
- Januszewski, A.S.; Rankin, W.A.; O'Neal, D.N.; Wittert, G.A.; Jenkins, A.J. Sex hormone binging globulin as an indicator of insulin resistance in type 1 diabetes. Diabetes Res Clin Pract. 2025, 229, 112935. [Google Scholar] [CrossRef] [PubMed]
- Nigam, M.; Punia, B.; Dimri, D.B.; Mishra, A.P.; Radu, A.F.; Bungau, G. Reactive Oxygen Species: A Double-Edged Sword in the Modulation of Cancer Signaling Pathway Dynamics. Cells 2025, 14, 1207. [Google Scholar] [CrossRef]
- Milani, I.; Codini, M.; Guarisco, G.; Chinucci, M.; Gaita, C.; Leonetti, F.; Capoccia, D. Hepatokines and MASLD: The GLP1-Ras-FGF21-Fetuin-A Crosstalk as a Therapeutic Target. Int J Mol Sci. 2024, 25, 10795. [Google Scholar] [CrossRef]
- Chen, Y.Q.; Pottanat, T.G.; Siegel, R.W.; Ehsani, M.; Qian, Y.W.; Zhen, E.Y.; Regmi, A.; Roell, W.C.; Guo, H.; Luo, M.J.; Gimeno, R.E.; Van't Hooft, F.; Konrad, R.J. Angiopoietin-like protein 8 differentially regulates ANGPTL3 and ANGPTL4 during postprandial partitioning of fatty acids. J Lipid Res. 2020, 61, 1203–1220. [Google Scholar] [CrossRef]
- Garstka, M.A.; Kedzierski, L.; Maj, T. Diabetes can impact cellular immunity in solid tumors. Trends Immunol. 2025, 46, 295–309. [Google Scholar] [CrossRef]
- Lonardo, F. Cancer in individuals with obesity and metabolic disorders. A preventable epidemic? Metab Target Organ Damage 2022, 2, 20. [Google Scholar] [CrossRef]
- Pan, S.Y.; Johnson, K.C.; Ugnat, A.M.; Wen, S.W.; Mao, Y.; Canadian Cancer Registries Epidemiology Research Group. Association of obesity and cancer risk in Canada. Am J Epidemiol. 2004, 159, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Abonyi-Tóth, Z.; Rokszin, G.; Sütő, G.; Fábián, I.; Kiss, Z.; Jermendy, G.; Kempler, P.; Lengyel, C.; Wittmann, I.; Molnár, G.A. Incident Cancer Risk of Patients with Prevalent Type 2 Diabetes Mellitus in Hungary (Part 2). Cancers 2024, 16, 2414. [Google Scholar] [CrossRef] [PubMed]
- Lonardo, A. Is liver fibrosis a risk factor for gynecological cancers? Metab Target Organ Damage 2024, 4, 7. [Google Scholar] [CrossRef]
- Mantovani, A.; Lonardo, A.; Stefan, N.; Targher, G. Metabolic dysfunction-associated steatotic liver disease and extrahepatic gastrointestinal cancers. Metabolism 2024, 160, 156014. [Google Scholar] [CrossRef]
- Lonardo, A.; Stefan, N.; Mantovani, A. Widening research horizons on metabolic dysfunction-associated steatotic liver disease and cancer. Trends Endocrinol Metab. 2025, 36, 610–613. [Google Scholar] [CrossRef]
- Wu, C.; Targher, G.; Byrne, C.D.; Mao, Y.; Cheung, T.T.; Yilmaz, Y.; et al. Global, Regional, and National Burden of Primary Liver Cancer Attributable to Metabolic Risks: An Analysis of the Global Burden of Disease Study 1990-2021. Am J Gastroenterol. 2025, 120, 2280–2290. [Google Scholar] [CrossRef]
- Bril, F.; Elbert, A. Metabolic dysfunction-associated steatotic liver disease and urinary system cancers: Mere coincidence or reason for concern? Metabolism 2025, 162, 156066. [Google Scholar] [CrossRef]
- Schenk, S.; Harber, M.P.; Shrivastava, C.R.; Burant, C.F.; Horowitz, J.F. Improved insulin sensitivity after weight loss and exercise training is mediated by a reduction in plasma fatty acid mobilization, not enhanced oxidative capacity. J Physiol. 2009, 587 Pt 20, 4949–4961. [Google Scholar] [CrossRef]
- Flynn, C.R.; Tamboli, R.A.; Antoun, J.; Sidani, R.M.; Williams, B.; Spann, M.D.; English, W.J.; Welch, E.B.; Sundaresan, S.; Abumrad, N.N. Caloric Restriction and Weight Loss Are Primary Factors in the Early Tissue-Specific Metabolic Changes After Bariatric Surgery. Diabetes Care 2022, 45, 1914–1916. [Google Scholar] [CrossRef] [PubMed]
- Pareek, M.; Schauer, P.R.; Kaplan, L.M.; Leiter, L.A.; Rubino, F.; Bhatt, D.L. Metabolic Surgery: Weight Loss, Diabetes, and Beyond. J Am Coll Cardiol. 2018, 71, 670–687. [Google Scholar] [CrossRef]
- Hallberg, S.J.; Gershuni, V.M.; Hazbun, T.L.; Athinarayanan, S.J. Reversing Type 2 Diabetes: A Narrative Review of the Evidence. Nutrients 2019, 11, 766. [Google Scholar] [CrossRef]
- Ram Sohan, P.; Mahakalkar, C.; Kshirsagar, S.; Bikkumalla, S.; Reddy, S.; Hatewar, A.; Dixit, S. Long-Term Effectiveness and Outcomes of Bariatric Surgery: A Comprehensive Review of Current Evidence and Emerging Trends. Cureus 2024, 16, e66500. [Google Scholar] [CrossRef]
- Herman, R.; Kravos, N.A.; Jensterle, M.; Janež, A.; Dolžan, V. Metformin and Insulin Resistance: A Review of the Underlying Mechanisms behind Changes in GLUT4-Mediated Glucose Transport. Int J Mol Sci. 2022, 23, 1264. [Google Scholar] [CrossRef] [PubMed]
- Mesquita, L.A.; Spiazzi, B.F.; Piccoli, G.F.; Nogara, D.A.; da Natividade, G.R.; Garbin, H.I.; Wayerbacher, L.F.; Wiercinski, V.M.; Baggio, V.A.; Zingano, C.P.; Schwartsmann, G.; Lopes, G.; Petrie, J.R.; Colpani, V.; Gerchman, F. Does metformin reduce the risk of cancer in obesity and diabetes? A systematic review and meta-analysis. Diabetes Obes Metab. 2024, 26, 1929–1940. [Google Scholar] [CrossRef] [PubMed]
- Said, A.; Akhter, A. Meta-Analysis of Randomized Controlled Trials of Pharmacologic Agents in Non-alcoholic Steatohepatitis. Ann Hepatol. 2017, 16, 538–547. [Google Scholar] [CrossRef]
- Eggleton, J.S.; Jialal, I. Thiazolidinediones. [Updated 2023 Feb 20]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK551656/.
- Weiskirchen, R.; Lonardo, A. How 'miracle' weight-loss semaglutide promises to change medicine but can we afford the expense? Br J Pharmacol. 2025, 182, 1651–1670. [Google Scholar] [CrossRef]
- Weiskirchen, R.; Lonardo, A. Semaglutide from Bench to Bedside: The Experimental Journey Towards a Transformative Therapy for Diabetes, Obesity and Metabolic Liver Disorders. [CrossRef]
- Karim, G.; Bansal, M.B. Resmetirom: An Orally Administered, Smallmolecule, Liver-directed, β-selective THR Agonist for the Treatment of Non-alcoholic Fatty Liver Disease and Non-alcoholic Steatohepatitis. touchREV Endocrinol. 2023, 19, 60–70. [Google Scholar] [CrossRef]
- Tiwari, A.; Sharma, A.; Kumar, H.; Gupta, V.; Deshpande, V.; Mupparaju, J.S.; Mishra, T.; Bharadwaj, H.R.; Dahiya, D.S.; Jain, V. Resmetirom for MASH: A Comprehensive Review of a Novel Therapeutic Frontier. Biomedicines 2025, 13, 2079. [Google Scholar] [CrossRef]
- Shakeel, L.; Shaukat, A.; Akilimali, A. Resmetirom: A Breakthrough in the Treatment of Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD). Health Sci Rep. 2025, 8, e70920. [Google Scholar] [CrossRef]
- Lonardo, A.; Weiskirchen, R. Tirzepatide in metabolic dysfunction-associated steatotic liver disease and steatohepatitis: a novel star on the horizon? Explor Drug Sci. 2025, 3, 1008128. [Google Scholar] [CrossRef]
- Lonardo, A.; Zheng, M.H.; Weiskirchen, R. Food insecurity is an emerging risk factor for liver disease: a scoping review. Expert Rev Gastroenterol Hepatol. 2025, 19, 1033–1049. [Google Scholar] [CrossRef] [PubMed]
- Hill-Briggs, F.; Adler, N.E.; Berkowitz, S.A.; Chin, M.H.; Gary-Webb, T.L.; Navas-Acien, A.; Thornton, P.L.; Haire-Joshu, D. Social Determinants of Health and Diabetes: A Scientific Review. Diabetes Care 2020, 44, 258–279. [Google Scholar] [CrossRef] [PubMed]
- Lonardo, A.; Bellentani, S.; Ratziu, V.; Loria, P. Insulin resistance in nonalcoholic steatohepatitis: necessary but not sufficient - death of a dogma from analysis of therapeutic studies? Expert Rev Gastroenterol Hepatol. 2011, 5, 279–289. [Google Scholar] [CrossRef] [PubMed]
| Physiologic | Reference | ||
|---|---|---|---|
| Fasting | [22] | ||
| Strenuous exercise | [22] | ||
| Puberty | [22] | ||
| Pregnancy | [23] | ||
| Aging | [21] | ||
| Pathologic | |||
| Obesity | [21] | ||
| Physical inactivity | [21] | ||
| Altered sleep-wake cycle | [24] | ||
| Stressful conditions | Trauma, surgery, diabetes ketoacidosis, severe infection, uremia, and liver cirrhosis | [21] | |
| High-sodium diets | |||
| HIV | [25,26] | ||
| Endocrinological | PCOS, Cushing syndrome, acromegaly | [21] | |
| Smoking | [27] | ||
| Hepatic Cirrhosis | [22] | ||
| Uremia | [22] | ||
| Drug-induced | Long-term glucocorticoids, cyclosporine, niacin, anti-adrenergic, protease inhibitors, SRRI, atypical antipsychotics, androgen-deprivation therapy, insulin therapy | [21,22] | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




