Submitted:
18 December 2025
Posted:
19 December 2025
Read the latest preprint version here
Abstract
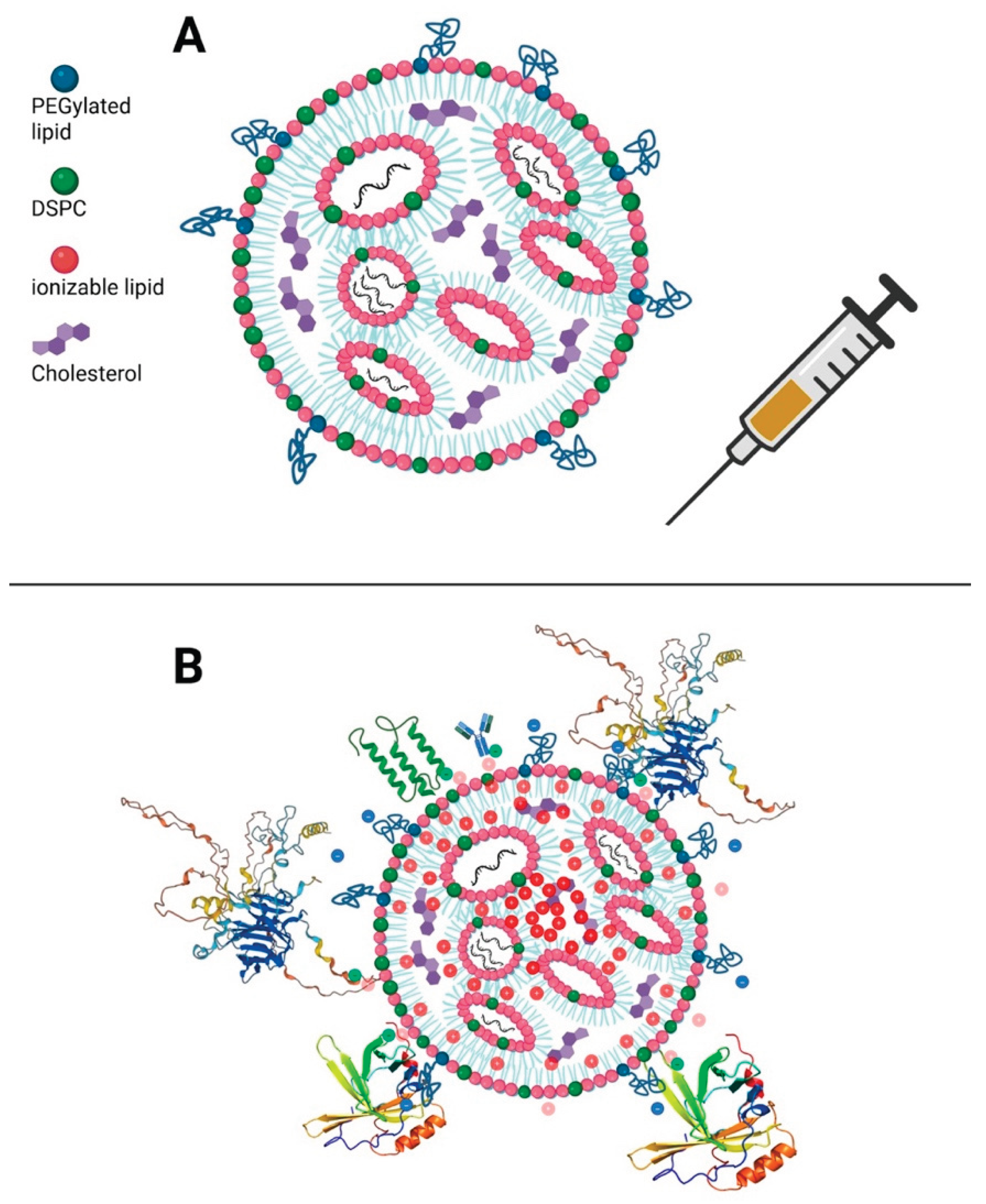
Keywords:
1. Introduction
2. Lipid Nanoparticles for mRNA Delivery: Biological Properties and Effects on Cellular Systems
2.1. Factors Influencing Nanoparticle Bioactivity
2.2. LNP Biodistribution
2.3. Mechanisms of Uptake
2.4. Endosomal Escape and Membrane Destabilization Due to Ionizable Lipids
2.5. Spread to Distant Sites via Exosomes
2.6. LNP Metabolism Leads to Oxidative Stress and Signaling Cascades
2.7. Activation of the Immune System
3. The Principles Behind How LNP-modRNA was Thought to Work
4. Omics: Evidence for Membrane Dysfunction Secondary to LNP Transfection
4.1. Ndeupen et al.—A Pioneering Omics Study
4.2. Upregulation of Multiple Inflammatory Markers
4.3. Downregulation of PPAR and AMPK Signaling
4.4. Downregulated Xenobiotic Metabolism by Cytochrome P450 Enzymes
4.5. Non-Canonical Transciptomics and Proteomic Alterations—Are the TLR4 Reactions Decoupled?
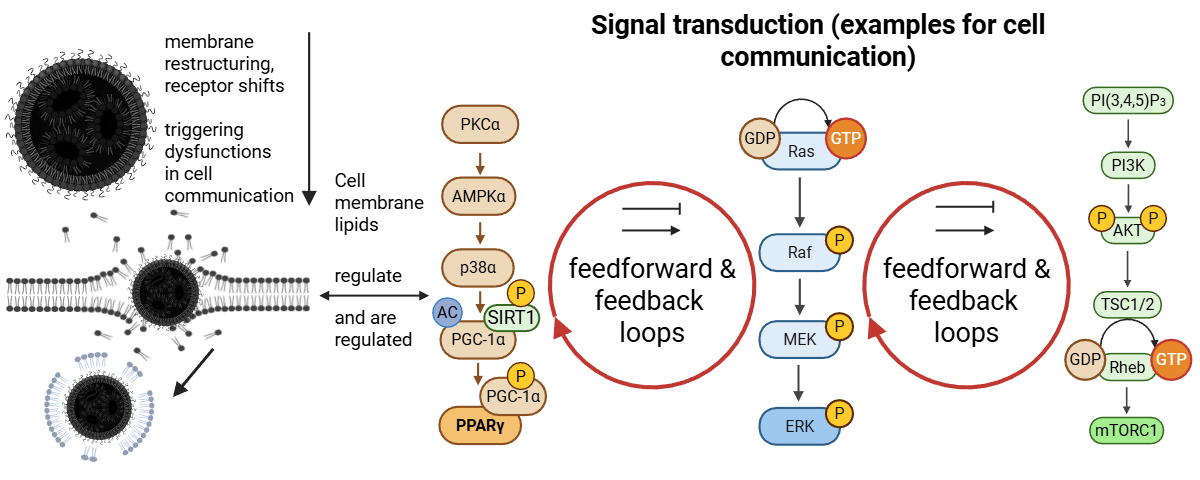
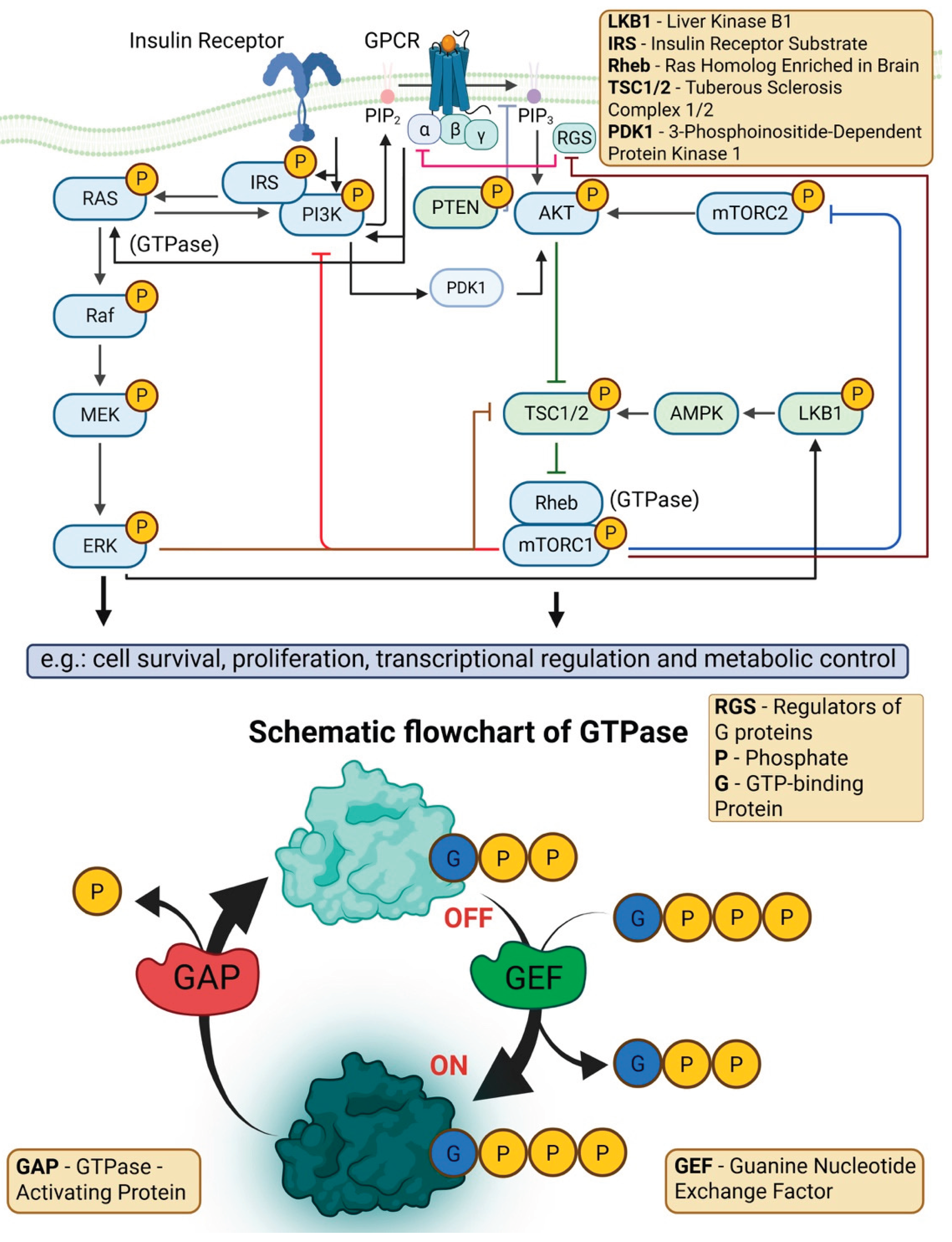
4.6. Dysregulation of MAPK/ERK, JAK-STAT, and other Signaling Pathways
4.7. Disrupted RAS Signaling
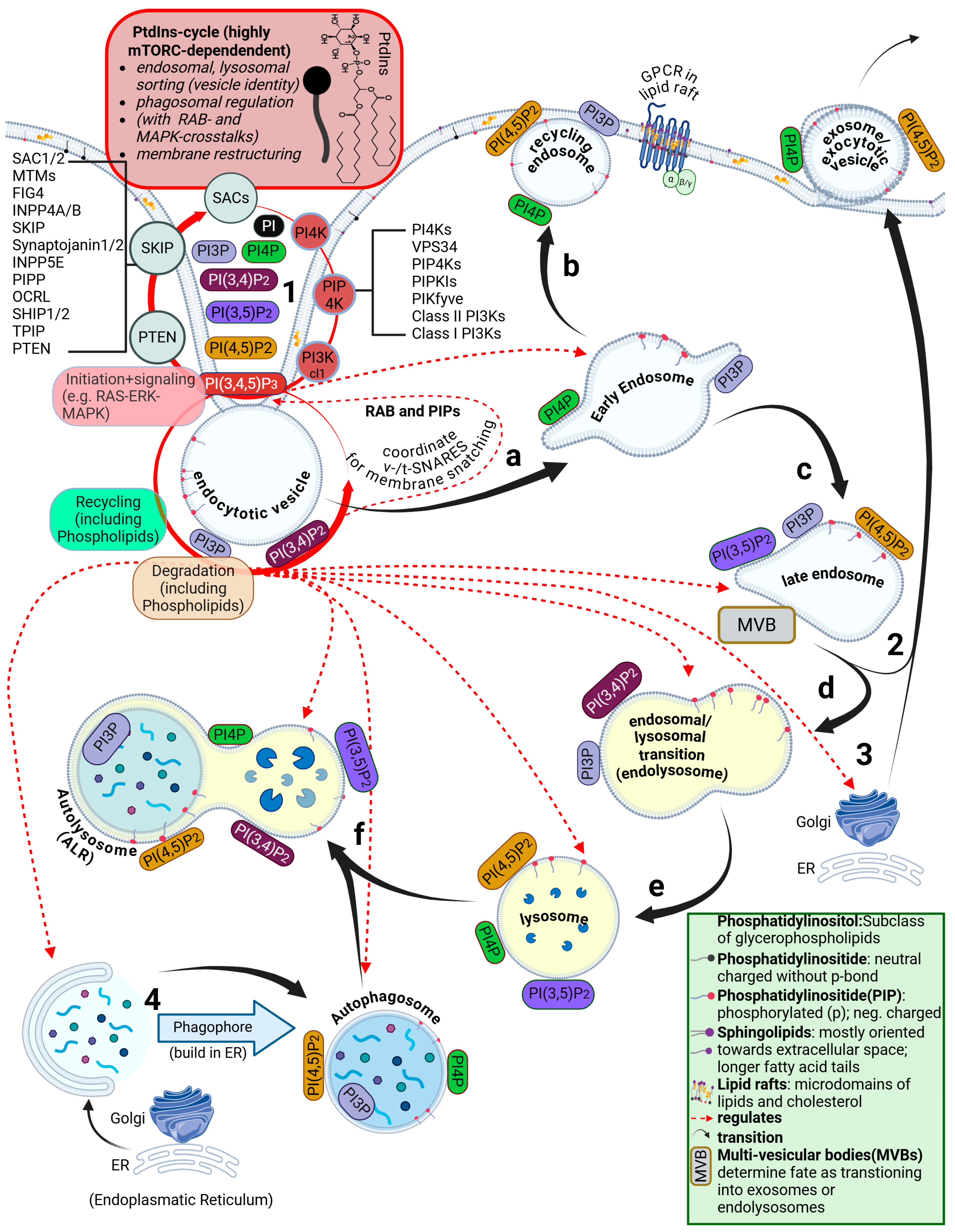
4.8. Disruption of the ESCRT Circuit and Phosphatidylinositol Signaling
4.9. Perturbations Originate at the Plasma Membrane and Disturb PtdIns Signaling Cascades
5. Breaching the Plasma Membrane: Important Roles for Phosphoinositides
5.1. Brief Overview of the Phosphatidylinositide Cycle
5.2. The Role of Lipid Rafts in LNP Uptake into Cells
5.3. Signaling through Phosphorylation States of Phosphatidylinositols
5.4. Oxysterol-Binding Proteins (OSBs) and a Role for Cholesterol
5.5. How does 1,2-distearoyl-sn-glycero-3-phosphocholine (DSPC) Affect the PI Cycle?
5.6. A Role for Lipid Impurities
5.7. Small Perturbations Can Lead to Major Shifts in PIP Signaling
6. Discussion
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Use of Generative AI
Acknowledgements
Conflicts of Interest
Abbreviations
References
- Khurana A, Allawadhi P, Khurana I, Allwadhi S, Weiskirchen R, Banothu AK, et al. Role of nanotechnology behind the success of mRNA vaccines for COVID-19. Nano Today 2021;38:101142. [CrossRef]
- Hald Albertsen C, Kulkarni JA, Witzigmann D, Lind M, Petersson K, Simonsen JB. The role of lipid components in lipid nanoparticles for vaccines and gene therapy. Adv Drug Deliv Rev 2022; 188: 114416. [CrossRef]
- Swetha K, Kotla NG, Tunki L, Jayaraj A, Bhargava SK, Hu H, et al. Recent advances in the lipid nanoparticle-mediated delivery of mRNA vaccines. Vaccines (Basel) 2023;11(3):658. [CrossRef]
- Suk JS, Xu Q, Kim N, Hanes J, Ensign LM. PEGylation as a strategy for improving nanoparticle-based drug and gene delivery. Adv Drug Deliv Rev 2016;99(Pt A):28-51. [CrossRef]
- Ju Y, Carreo JM, Simon V, Dawson K, Krammer F, Kent SJ. Impact of anti-PEG antibodies induced by SARS-CoV-2 mRNA vaccines. Nat Rev Immunol 2023;23(3):135-136. [CrossRef]
- Zhou ZH, Stone CA Jr, Jakubovic B, Phillips EJ, Sussman G, Park J, et al. Anti-PEG IgE in anaphylaxis associated with polyethylene glycol. J Allergy Clin Immunol Pract 2021;9(4):1731-1733.e3. [CrossRef]
- Kozma GT, Shimizu T, Ishida T, Szebeni J. Anti-PEG antibodies: Properties, formation, testing and role in adverse immune reactions to PEGylated nano-biopharmaceuticals. Adv Drug Deliv Rev 2020;154-155:163-175. [CrossRef]
- Khalid MB, Frischmeyer-Guerrerio PA. The conundrum of COVID-19 mRNA vaccine-induced anaphylaxis. J Allergy Clin Immunol Glob. 2023;2(1):1-13. [CrossRef]
- Zelkoski AE, Lu Z, Sukumar G, Dalgard C, Said H, Alameh MG, et al. Ionizable lipid nanoparticles of mRNA vaccines elicit NF-κB and IRF responses through toll-like receptor 4. NPJ Vaccines 2025;10(1):73. [CrossRef]
- Lee Y, Jeong M, Park J, Jung H, Lee H. Immunogenicity of lipid nanoparticles and its impact on the efficacy of mRNA vaccines and therapeutics. Exp Mol Med 2023 Oct;55(10):2085-2096. [CrossRef]
- Dilliard SA, Cheng Q, Siegwart DJ. On the mechanism of tissue-specific mRNA delivery by selective organ targeting nanoparticles. Proc Natl Acad Sci U S A 2021;118(52):e2109256118. [CrossRef]
- Back PI, Yu M, Modaresahmadi S, Hajimirzaei S, Zhang Q, Islam MR, Schwendeman AA, La-Beck NM. Immune Implications of Cholesterol-Containing Lipid Nanoparticles. ACS Nano 2024; 18(42):28480-28501. [CrossRef]
- Parrett BJ, Yamaoka S, Barry MA. Reducing off-target expression of mRNA therapeutics and vaccines in the liver with microRNA binding sites. Mol Ther Methods Clin Dev 2024;33(1):101402. [CrossRef]
- Boros LG, Kyriakopoulos AM, Brogna C, Piscopo M, McCullough PA, Seneff S. Long-lasting, biochemically modified mRNA, and its frameshifted recombinant spike proteins in human tissues and circulation after COVID-19 vaccination. Pharmacol Res Perspect 2024;12(3):e1218. [CrossRef]
- Cordes J, Zhao S, Engel CM, Stingele J. Cellular responses to RNA damage. Cell 2025;188(4):885-900. [CrossRef]
- Kaludercic N, Deshwal S, Di Lisa F. Reactive oxygen species and redox compartmentalization. Front Physiol 2014;5:285. [CrossRef]
- Kim KQ, Burgute BD, Tzeng SC, Jing C, Jungers C, Zhang J, et al. N1-methylpseudouridine found within COVID-19 mRNA vaccines produces faithful protein products. Cell Rep 2022;40(9):111300. [CrossRef]
- Röltgen K, Nielsen SCA, Silva O, Younes SF, Zaslavsky M, Costales C, et al. Immune imprinting, breadth of variant recognition, and germinal center response in human SARS-CoV-2 infection and vaccination. Cell 2022;185(6):1025-1040.e14. [CrossRef]
- Kim W, Ly NK, He Y, Li Y, Yuan Z, Yeo Y. Protein corona: Friend or foe? Co-opting serum proteins for nanoparticle delivery. Adv Drug Deliv Rev 2023;192:114635. [CrossRef]
- Suzuki Y, Ishihara H. Difference in the lipid nanoparticle technology employed in three approved siRNA (Patisiran) and mRNA (COVID-19 vaccine) drugs. Drug Metab Pharmacokinet 2021;41:100424. [CrossRef]
- Buckley M, Araínga M, Maiorino L, Pires IS, Kim BJ, Michaels KK, et al. Visualizing lipid nanoparticle trafficking for mRNA vaccine delivery in non-human primates. Mol Ther 2025;33(3):1105-1117. [CrossRef]
- Sasaki K, Sato Y, Okuda K, Iwakawa K, Harashima H. mRNA-loaded lipid nanoparticles targeting dendritic cells for cancer immunotherapy. Pharmaceutics 2022;14(8):1572. [CrossRef]
- Hassett KJ, Rajlic IL, Bahl K, White R, Cowens K, Jacquinet E, et al. mRNA vaccine trafficking and resulting protein expression after intramuscular administration. Mol Ther Nucleic Acids 2023;35(1):102083. [CrossRef]
- Maugeri M, Nawaz M, Papadimitriou A, Angerfors A, Camponeschi A, Na M, et al. Linkage between endosomal escape of LNP-mRNA and loading into EVs for transport to other cells. Nat Commun 2019; 10: 4333. [CrossRef]
- Inam W, Bhadane R, Akpolat RN, Taiseer RA, Filippov SK, Salo-Ahen OMH, et al. Interactions between polymeric nanoparticles and different buffers as investigated by zeta potential measurements and molecular dynamics simulations. View 2022; 3(4): 20210009. [CrossRef]
- Simak J, De Paoli S. The effects of nanomaterials on blood coagulation in hemostasis and thrombosis. Wiley Interdiscip Rev Nanomed Nanobiotechnol 2017;9(5). [CrossRef]
- Brooks DE, Seaman GVF. The effect of neutral polymers on the electrokinetic potential of cells and other charged particles. I. Models for the zeta potential increase. J Colloid Interf Sci 1973 ;43 (3):670-686.
- Ilinskaya AN, Dobrovolskaia MA. Nanoparticles and the blood coagulation system. Part II: safety concerns. Nanomedicine (Lond). 2013;8(6):969-81. [CrossRef]
- Omo-Lamai S, Zamora ME, Patel MN, Wu J, Nong J, Wang Z, et al. Physicochemical targeting of lipid nanoparticles to the lungs induces clotting: Mechanisms and solutions. Adv Mater 2024;36(26):e2312026. [CrossRef]
- Bekal S, Husari G, Okura M, Huang CA, Bukari MS. Thrombosis development after mRNA COVID-19 vaccine administration: A Case Series. Cureus. 2023;15(7):e41371. [CrossRef]
- Kent SJ, Li S, Amarasena TH, Reynaldi A, Lee WS, Leeming MG, et al. Blood distribution of SARS-CoV-2 lipid nanoparticle mRNA vaccine in humans. ACS Nano 2024;18(39):27077-27089. [CrossRef]
- Ren Y, Lin L, Abdallah M, Zhu X, Liu H, Fabb SA, et al. Impact of ionizable lipid type on the pharmacokinetics and biodistribution of mRNA-lipid nanoparticles after intravenous and subcutaneous injection. J Control Release 2025; 384:113945. [CrossRef]
- Luo J, Molbay M, Chen Y, Horvath I, Kadletz K, Kick B, et al. Nanocarrier imaging at single-cell resolution across entire mouse bodies with deep learning. Nat Biotechnol 2025 Jan 14. [Epub ahead of print]. [CrossRef]
- Di J, Du Z, Wu K, Jin S, Wang X, Li T, et al. Biodistribution and non-linear gene expression of mRNA LNPs affected by delivery route and particle size. Pharm Res 2022;39(1):105-114. [CrossRef]
- Dey AK, Nougarède A, Clément F, Fournier C, Jouvin-Marche E, Escudé M, et al. Tuning the immunostimulation properties of cationic lipid nanocarriers for nucleic acid delivery. Front Immunol 2021;12:722411. [CrossRef]
- Zhang W, Liu R, Chen Y, Wang M, Du J. Crosstalk between oxidative stress and exosomes. Oxid Med Cell Longev 2022;2022:3553617. [CrossRef]
- Xu X, Cui L, Zhang Y, Gu J. Deciphering the biological fate of mRNA-LNP-based biologics: A perspective from tissue to intracellular distribution. Acta Pharmaceutica Sinica 2025 Nov 19. [Epub ahead of print]. [CrossRef]
- Zhang H, Barz M. Investigating the stability of RNA-lipid nanoparticles in biological fluids: Unveiling its crucial role for understanding LNP performance. Journal of Controlled Release 2025; 381: 113559. [CrossRef]
- Balcorta HV, Mata Corral MY, Gallegos A, Chavez J, Perez J, Balivada S, et al. Development of chemical tags for universal lipid nanoparticle visualization and tracking in 2D and 3D imaging. Nano Lett 2025;25(19):7682-7689. [CrossRef]
- Yamamoto K, Mashiba T, Takano K, Suzuki T, Kami M, Takita M, et al. A case of exacerbation of subclinical hyperthyroidism after first administration of BNT162b2 mRNA COVID-19 vaccine. Vaccines (Basel) 2021;9(10):1108. [CrossRef]
- Hummel A, Oniszczuk J, Kervella D, Charbit M, Guerrot D, Testa A, et al. Idiopathic nephrotic syndrome relapse following COVID-19 vaccination: a series of 25 cases. Clin Kidney J 2022;15(8):1574-1582. [CrossRef]
- Malayala SV, Papudesi BN, Sharma R, Vusqa UT, Raza A. A case of idiopathic thrombocytopenic purpura after booster dose of BNT162b2 (Pfizer-Biontech) COVID-19 vaccine. Cureus 2021;13(10):e18985. [CrossRef]
- Shumnalieva R, Ravichandran N, Hannah J, Javaid M, Darooka N, Roy D, Gonzalez DE, et al. Characteristics of emerging new autoimmune diseases after COVID-19 vaccination: A sub-study by the COVAD group. Int J Rheum Dis 2024;27(5):e15183. [CrossRef]
- Broachwala M, Banks DW, Jevotovsky DS, Oehlermarx W, Durbhakula S. Burning mouth syndrome following Covid vaccination: A case report. Clin Case Rep 2025;13(5):e70329. [CrossRef]
- Fraiman J, Erviti J, Jones M, Greenland S, Whelan P, Kaplan RM, et al. Serious adverse events of special interest following mRNA COVID-19 vaccination in randomized trials in adults. Vaccine 2022;40(40):5798-5805. [CrossRef]
- Faksova K, Walsh D, Jiang Y, Griffin J, Phillips A, Gentile A, et al. COVID-19 vaccines and adverse events of special interest: A multinational Global Vaccine Data Network (GVDN) cohort study of 99 million vaccinated individuals. Vaccine 2024;42(9):2200-2211. [CrossRef]
- Kim HJ, Kim MH, Choi MG, Chun EM. 1-year risks of cancers associated with COVID-19 vaccination: a large population-based cohort study in South Korea. Biomark Res 2025;13(1):114. [CrossRef]
- Abbasi R, Shineh G, Mobaraki M, Doughty S, Tayebi L. Structural parameters of nanoparticles affecting their toxicity for biomedical applications: a review. J Nanopart Res 2023; 25(3): 43. [CrossRef]
- Yuan Z, Yan R, Fu Z, Wu T, Ren C. Impact of physicochemical properties on biological effects of lipid nanoparticles: Are they completely safe. Sci Total Environ 2024;927:172240. [CrossRef]
- Szebeni J, Kiss B, Bozó T, Turjeman K, Levi-Kalisman Y, Barenholz Y, et al. Insights into the structure of Comirnaty COVID-19 vaccine: A theory on soft, partially bilayer-covered nanoparticles with hydrogen bond-stabilized mRNA-lipid complexes. ACS Nano 2023;17(14):13147-13157. [CrossRef]
- Brader ML, Williams SJ, Banks JM, Hui WH, Zhou ZH, Jin L. Encapsulation state of messenger RNA inside lipid nanoparticles. Biophys J 2021;120(14):2766-2770. [CrossRef]
- Münter R, Larsen JB, Andresen TL. The vast majority of nucleic acid-loaded lipid nanoparticles contain cargo. J Colloid Interface Sci 2024;674:139-144. [CrossRef]
- Li S, Hu Y, Li A, Lin J, Hsieh K, Schneiderman Z, et al. Payload distribution and capacity of mRNA lipid nanoparticles. Nat Commun 2022;13(1):5561. [CrossRef]
- Chen X, Ye Y, Li M, Zuo T, Xie Z, Ke Y, et al. Structural characterization of mRNA lipid nanoparticles (LNPs) in the presence of mRNA-free LNPs. J Control Release 2025;386:114082. [CrossRef]
- Rampado R, Crotti S, Caliceti P, Pucciarelli S, Agostini M. Recent advances in understanding the protein corona of nanoparticles and in the formulation of "stealthy" nanomaterials. Front Bioeng Biotechnol 2020;8:166. [CrossRef]
- Sun Y, Zhou Y, Rehman M, Wang YF, Guo S. Protein corona of nanoparticles: Isolation and analysis. Chem Bio Eng 2024;1(9):757-772. [CrossRef]
- Voke E, Arral ML, Squire HJ, Lin TJ, Zheng L, Coreas R, et al. Protein corona formed on lipid nanoparticles compromises delivery efficiency of mRNA cargo. Nat Commun 2025;16(1):8699. [CrossRef]
- Sebastiani F, Yanez Arteta M, Lerche M, Porcar L, Lang C, Bragg RA, Elmore CS, et al. Apolipoprotein E binding drives structural and compositional rearrangement of mRNA-containing lipid nanoparticles. ACS Nano 2021;15(4):6709-6722. [CrossRef]
- Liu K, Nilsson R, Lázaro-Ibáñez E, Duàn H, Miliotis T, Strimfors M, et al. Multiomics analysis of naturally efficacious lipid nanoparticle coronas reveals high-density lipoprotein is necessary for their function. Nat Commun 2023;14(1):4007. [CrossRef]
- Hosseini-Kharat M, Bremmell KE, Prestidge CA. Why do lipid nanoparticles target the liver? Understanding of biodistribution and liver-specific tropism. Mol Ther Methods Clin Dev 2025;33(1):101436. [CrossRef]
- Therapeutic Goods Administration. Nonclinical Evaluation Report BNT162b2 [mRNA] COVID-19 vaccine (COMIRNATY). Health, Ed.; Department of Health and Aged Care. https://www.tga.gov.au/sites/default/files/foi-2389-06.pdf, 2021; Vol. FOI 2389.
- Neves AR, Queiroz JF, Costa Lima SA, Figueiredo F, Fernandes R, Reis S. Cellular uptake and transcytosis of lipid-based nanoparticles across the intestinal barrier: Relevance for oral drug delivery. J Colloid Interface Sci 2016;463:258-65. [CrossRef]
- Haghighi E, Abolmaali SS, Dehshahri A, Mousavi Shaegh SA, Azarpira N, Tamaddon AM. Navigating the intricate in-vivo journey of lipid nanoparticles tailored for the targeted delivery of RNA therapeutics: a quality-by-design approach. J Nanobiotechnology 2024;22(1):710. [CrossRef]
- Khare P, Edgecomb SX, Hamadani CM, Tanner EEL, S Manickam D. Lipid nanoparticle-mediated drug delivery to the brain. Adv Drug Deliv Rev 2023;197:114861. [CrossRef]
- Chen J, Xu Y, Zhou M, Xu S, Varley AJ, Golubovic A, et al. Combinatorial design of ionizable lipid nanoparticles for muscle-selective mRNA delivery with minimized off-target effects. Proc Natl Acad Sci U S A 2023;120(50):e2309472120. [CrossRef]
- Younis MA, Sato Y, Elewa YHA, Kon Y, Harashima H. Self-homing nanocarriers for mRNA delivery to the activated hepatic stellate cells in liver fibrosis. J Control Release 2023 Jan;353:685-698. [CrossRef]
- Naasani I. Establishing the pharmacokinetics of genetic vaccines is essential for maximising their safety and efficacy. Clin Pharmacokinet 2022;61(7):921-927. [CrossRef]
- European Medical Assessment. 2020. https://www.ema.europa.eu/en/documents/assessment-report/comirnaty-epar-public-assessment-report en.pdf.
- Akhter MH, Khalilullah H, Gupta M, Alfaleh MA, Alhakamy NA, Riadi Y, et al. Impact of protein corona on the biological identity of nanomedicine: Understanding the fate of nanomaterials in the biological milieu. Biomedicines 2021;9(10):1496. [CrossRef]
- Behzadi S, Serpooshan V, Tao W, Hamaly MA, Alkawareek MY, Dreaden EC, et al. Cellular uptake of nanoparticles: journey inside the cell. Chem Soc Rev 2017;46(14):4218-4244. [CrossRef]
- Lavington S, Watts A. Lipid nanoparticle technologies for the study of G protein-coupled receptors in lipid environments. Biophys Rev 2020;12(6):1287–302. [CrossRef]
- Chatterjee S, Kon E, Sharma P, Peer D. Endosomal escape: A bottleneck for LNP-mediated therapeutics. Proc Natl Acad Sci U S A. 2024;121(11):e2307800120. [CrossRef]
- Sengottiyan S, Mikolajczyk A, Jagiełło K, Swirog M, Puzyn T. Core, coating, or corona? The importance of considering protein coronas in nano-QSPR modeling of zeta potential. ACS Nano 2023;17(3):1989-1997. [CrossRef]
- Vermeulen LMP, Brans T, Samal SK, Dubruel P, Demeester J, De Smedt SC, et al. Endosomal size and membrane leakiness influence proton sponge-based rupture of endosomal vesicles. ACS Nano. 2018;12(3):2332-2345. [CrossRef]
- Johansson JM, Du Rietz H, Hedlund H, Eriksson HC, Oude Blenke E, Pote A, et al. Cellular and biophysical barriers to lipid nanoparticle mediated delivery of RNA to the cytosol. Nat Commun 2025;16(1):5354. [CrossRef]
- Ermilova I, Swenson J. Ionizable lipids penetrate phospholipid bilayers with high phase transition temperatures: perspectives from free energy calculations. Chem Phys Lipids 2023;253:105294. [CrossRef]
- Er-Rafik M, Ferji K, Combet J, Sandre O, Lecommandoux S, Schmutz M, et al. Tear of lipid membranes by nanoparticles. Soft Matter 2022; 18(17): 3318-3322. [CrossRef]
- Pilkington EH, Suys EJA, Trevaskis NL, Wheatley AK, Zukancic D, Algarni A, et al. From influenza to COVID-19: Lipid nanoparticle mRNA vaccines at the frontiers of infectious diseases. Acta Biomater 2021;131:16-40. [CrossRef]
- Müller JA, Schäffler N, Kellerer T, Schwake G, Ligon TS, Rädler JO. Kinetics of RNA-LNP delivery and protein expression. Eur J Pharm Biopharm 2024;197:114222. [CrossRef]
- Aliakbarinodehi N, Niederkofler S, Emilsson G, Parkkila P, Olsén E, Jing Y, et al. Time-resolved inspection of ionizable lipid-facilitated lipid nanoparticle disintegration and cargo release at an early endosomal membrane mimic. ACS Nano 2024;18(34):22989-23000. [CrossRef]
- Schlich M, Palomba R, Costabile G, Mizrahy S, Pannuzzo M, Peer D, et al. Cytosolic delivery of nucleic acids: The case of ionizable lipid nanoparticles. Bioeng Transl Med 2021;6(2):e10213. [CrossRef]
- Sabnis S, Kumarasinghe ES, Salerno T, Mihai C, Ketova T, Senn JJ, et al. A novel amino lipid series for mRNA delivery: Improved endosomal escape and sustained pharmacology and safety in non-human primates. Mol Ther 2018;26(6):1509-1519. [CrossRef]
- Paramasivam P, Franke C, Stöter M, Höijer A, Bartesaghi S, Sabirsh A, et al. Endosomal escape of delivered mRNA from endosomal recycling tubules visualized at the nanoscale. J Cell Biol 2022;221(2):e202110137. [CrossRef]
- Sahay G, Querbes W, Alabi C, Eltoukhy A, Sarkar S, Zurenko C, et al. Efficiency of siRNA delivery by lipid nanoparticles is limited by endocytic recycling. Nat Biotechnol 2013;31(7):653-8. [CrossRef]
- Bitounis D, Jacquinet E, Rogers MA, Amiji MM. Strategies to reduce the risks of mRNA drug and vaccine toxicity. Nat Rev Drug Discov 2024;23(4):281-300. [CrossRef]
- Moghimi SM, Simberg D. Pro-inflammatory concerns with lipid nanoparticles. Mol Ther 2022;30(6):2109-2110. [CrossRef]
- Bates SM, Munson MJ, Trovisco V, Pereira S, Miller SR, Sabirsh A, et al. The kinetics of endosomal disruption reveal differences in lipid nanoparticle induced cellular toxicity. J Control Release 2025;386:114047. [CrossRef]
- Su J, Song Y, Zhu Z, Huang X, Fan J, Qiao J, et al. Cell-cell communication: new insights and clinical implications. Signal Transduct Target Ther 2024;9(1):196. [CrossRef]
- Bansal S, Perincheri S, Fleming T, Poulson C, Tiffany B, Bremner RM, et al. Cutting edge: Circulating exosomes with COVID spike protein are induced by BNT162b2 (Pfizer-BioNTech) vaccination prior to development of antibodies: A novel mechanism for immune activation by mRNA vaccines. J Immunol 2021;207(10):2405-2410. [CrossRef]
- Kammerer USV, Steger K. BioNTech RNA-based COVID-19 injections contain large amounts of residual DNA including an SV40 promoter/enhancer Sequence. Journal of Science, Public Health Policy and the Law 2024; v5.2019-2024.
- Sahin U, Karikó K, Türeci Ö. mRNA-based therapeutics--developing a new class of drugs. Nat Rev Drug Discov 2014;13(10):759-80. [CrossRef]
- Jörgensen AM, Wibel R, Bernkop-Schnürch A. Biodegradable cationic and ionizable cationic lipids: A roadmap for safer pharmaceutical excipients. Small 2023;19(17):e2206968. [CrossRef]
- Knaggs KLM, Sun Y, Walz BA, Pang J, Khan OF. The role of excipients in lipid nanoparticle metabolism: implications for enhanced therapeutic effect. Ther Deliv 2025;16(7):687-700. [CrossRef]
- Packer M, Gyawali D, Yerabolu R, Schariter J, White P. A novel mechanism for the loss of mRNA activity in lipid nanoparticle delivery systems. Nat Commun 2021; 12(1): 6777. [CrossRef]
- Moderna. Moderna Science and Technology Day. 2022. https://s29.q4cdn.com/435878511/files/doc_presentations/2022/05/Science-Day-2022-Master-Slides-FINAL-(05.17_7am).pdf, 2022. Accessed February 28, 2023.
- USFDA. Letter to Pfizer: Children’s vaccination, authorization of formulation change. US Food and Drug Administration: https://cacmap.fda.gov/media/150386/download, 2021.
- Maelfait J, Liverpool L, Rehwinkel J. Nucleic acid sensors and programmed cell death. J Mol Biol 2020;432(2):552-568. [CrossRef]
- Fritz KS, Petersen DR. An overview of the chemistry and biology of reactive aldehydes. Free Radic Biol Med. 2013;59:85-91. [CrossRef]
- Dalleau S, Baradat M, Guéraud F, Huc L. Cell death and diseases related to oxidative stress: 4-hydroxynonenal (HNE) in the balance. Cell Death Differ 2013;20(12):1615-30. [CrossRef]
- Hashiba K, Taguchi M, Sakamoto S, Otsu A, Maeda Y, Ebe H, et al. Overcoming thermostability challenges in mRNA-lipid nanoparticle systems with piperidine-based ionizable lipids. Commun Biol 2024;7(1):556. [CrossRef]
- Wang W, Deng S, Lin J, Ouyang D. Modeling on in vivo disposition and cellular transportation of RNA lipid nanoparticles via quantum mechanics/physiologically-based pharmacokinetic approaches. Acta Pharm Sin B 2024;14(10):4591-4607. [CrossRef]
- Yu Z, Li Q, Wang J, Yu Y, Wang Y, Zhou Q, et al. Reactive oxygen species-related nanoparticle toxicity in the biomedical field. Nanoscale Res Lett 2020;15(1):115. [CrossRef]
- Atianand MK, Fitzgerald KA. Molecular basis of DNA recognition in the immune system. J Immunol 2013;190(5):1911-8. [CrossRef]
- Szebeni J, Simberg D, González-Fernández Á, Barenholz Y, Dobrovolskaia MA. Roadmap and strategy for overcoming infusion reactions to nanomedicines. Nat Nanotechnol 2018;13(12):1100-1108. [CrossRef]
- Bakos T, Mészáros T, Kozma GT, Berényi P, Facskó R, Farkas H, et al. mRNA-LNP COVID-19 vaccine lipids induce complement activation and production of proinflammatory cytokines: Mechanisms, effects of complement inhibitors, and relevance to adverse reactions. Int J Mol Sci 2024;25(7):3595. [CrossRef]
- Euopean Medicines Agency. Onpattro: European Public Assessment Report (EPAR) EMA/554262/2018. CHMP, Ed.; Amsterdam, NL, 2018.
- Wang J, Ding Y, Chong K, Cui M, Cao Z, Tang C, et al. Recent advances in lipid nanoparticles and their safety concerns for mRNA delivery. Vaccines (Basel) 2024;12(10):1148. [CrossRef]
- Zhang L, Ren F, Zhang X, Wang X, Shi H, Zhou L, et al. Peroxisome proliferator-activated receptor alpha acts as a mediator of endoplasmic reticulum stress-induced hepatocyte apoptosis in acute liver failure. Dis Model Mech 2016;9(7):799-809. [CrossRef]
- Popovics H, Mikone K, Mozes M, Kwon J-Y, Hansmann G, Kokeny G. P0721 PPAR-gamma activation inhibits TGF-beta induced renal complement and galectin-3 expression in vivo and in vitro, Nephrology Dialysis Transplantation 2020; 35(Suppl 3): gfaa142.P0721. [CrossRef]
- Kong Y, Wang N, Tong Z, Wang D, Wang P, Yang Q, et al. Role of complement factor D in cardiovascular and metabolic diseases. Front Immunol 2024;15:1453030. [CrossRef]
- Dai W, Xing M, Sun L, Lv L, Wang X, Wang Y, et al. Lipid nanoparticles as adjuvant of norovirus VLP vaccine augment cellular and humoral immune responses in a TLR9- and type I IFN-dependent pathway. J Virology 2024; 98(12): e0169924. [CrossRef]
- Chaudhary N, Kasiewicz LN, Newby AN, Arral ML, Yerneni SS, Melamed JR, et al. Amine headgroups in ionizable lipids drive immune responses to lipid nanoparticles by binding to the receptors TLR4 and CD1d. Nat Biomed Eng 2024;8(11):1483-1498. [CrossRef]
- Means N, Elechalawar CK, Chen WR, Bhattacharya R, Mukherjee P. Revealing macropinocytosis using nanoparticles. Mol Aspects Med 2022;83:100993. [CrossRef]
- Baimanov D, Wang J, Liu Y, Zheng P, Yu S, Liu F, et al. Identification of cell receptors responsible for recognition and binding of lipid nanoparticles. Journal of the American Chemical Society 2025; 147(9): 7604-7616. [CrossRef]
- Paunovska K, Da Silva Sanchez AJ, Lokugamage MP, Loughrey D, Echeverri ES, Cristian A, et al. The extent to which lipid nanoparticles require apolipoprotein E and low-density lipoprotein receptor for delivery changes with ionizable lipid structure. Nano Lett 2022;22(24):10025-10033. [CrossRef]
- Karikó K, Ni H, Capodici J, Lamphier M, Weissman D. mRNA is an endogenous ligand for Toll-like receptor 3. J Biol Chem 2004;279(13):12542-50. [CrossRef]
- Karikó K, Muramatsu H, Welsh FA, Ludwig J, Kato H, Akira S, et al. Incorporation of pseudouridine into mRNA yields superior nonimmunogenic vector with increased translational capacity and biological stability. Mol Ther 2008;16(11):1833-40. [CrossRef]
- Karikó K, Buckstein M, Ni H, Weissman D. Suppression of RNA recognition by Toll-like receptors: the impact of nucleoside modification and the evolutionary origin of RNA. Immunity 2005;23(2):165-75. [CrossRef]
- Monroe J, Eyler DE, Mitchell L, Deb I, Bojanowski A, Srinivas P, et al. N1-Methylpseudouridine and pseudouridine modifications modulate mRNA decoding during translation. Nat Commun 2024;15(1):8119. [CrossRef]
- Guo X, Wang H, Li Y, Leng X, Huang W, Ma Y, et al. Transfection reagent Lipofectamine triggers type I interferon signaling activation in macrophages. Immunol Cell Biol 2019 Jan;97(1):92-96. Erratum in: Immunol Cell Biol 2020 Jan;98(1):88. https://doi.org/10.1111/imcb.12300. [CrossRef]
- Przybylski S, Gasch M, Marschner A, Ebert M, Ewe A, Helmig G, et al. Influence of nanoparticle-mediated transfection on proliferation of primary immune cells in vitro and in vivo. PLoS One 2017;12(5):e0176517. [CrossRef]
- Choe U, Yu L, Wang TTY. Selected lipid-based transfection reagents activate NF-κB and MAP kinases signaling pathways, induced cytokines mRNA expression in human THP-1 macrophage. Anal Biochem 2019;573:73-76. [CrossRef]
- Connors J, Joyner D, Mege NJ, Cusimano GM, Bell MR, Marcy J, et al. Lipid nanoparticles (LNP) induce activation and maturation of antigen presenting cells in young and aged individuals. Commun Biol 2023;6(1):188. Erratum in: Commun Biol 2025;8(1):285. https://doi.org/10.1038/s42003-025-07603-0. [CrossRef]
- Korzun T, Moses AS, Jozic A, Grigoriev V, Newton S, Kim J, et al. Lipid nanoparticles elicit reactogenicity and sickness behavior in mice via toll-like receptor 4 and myeloid differentiation protein 88 axis. ACS Nano 2024;18(36):24842-24859. [CrossRef]
- Kawai T, Akira S. Toll-like receptors and their crosstalk with other innate receptors in infection and immunity. Immunity 2011;34(5):637-50. [CrossRef]
- Ding X, Jin S, Tong Y, Jiang X, Chen Z, Mei S, et al. TLR4 signaling induces TLR3 up-regulation in alveolar macrophages during acute lung injury. Sci Rep 2017; 7: 34278. [CrossRef]
- Nouri-Shirazi M, Tamjidi S, Nourishirazi E, Guinet E. Combination of TLR8 and TLR4 agonists reduces the degrading effects of nicotine on DC-NK mediated effector T cell generation. Int Immunopharmacol 2018;61:54-63. [CrossRef]
- Kim HJ, Kim H, Lee JH, Hwangbo C. Toll-like receptor 4 (TLR4): new insight immune and aging. Immun Ageing 2023;20(1):67. [CrossRef]
- Alhamdan F, Bayarsaikhan G, Yuki K. Toll-like receptors and integrins crosstalk. Front Immunol 2024;15:1403764. [CrossRef]
- Ghosh TK, Mickelson DJ, Solberg JC, Lipson KE, Inglefield JR, Alkan SS. TLR-TLR cross talk in human PBMC resulting in synergistic and antagonistic regulation of type-1 and 2 interferons, IL-12 and TNF-alpha. Int Immunopharmacol 2007;7(8):1111-21. [CrossRef]
- Xu XH, Shah PK, Faure E, Equils O, Thomas L, Fishbein MC, et al.Toll-like receptor-4 is expressed by macrophages in murine and human lipid-rich atherosclerotic plaques and upregulated by oxidized LDL. Circulation 2001;104(25):3103-8. [CrossRef]
- Hovland A, Jonasson L, Garred P, Yndestad A, Aukrust P, Lappegård KT, et al. The complement system and toll-like receptors as integrated players in the pathophysiology of atherosclerosis. Atherosclerosis 2015;241(2):480-94. [CrossRef]
- Luo L, Wall AA, Tong SJ, Hung Y, Xiao Z, Tarique AA, et al. TLR crosstalk activates LRP1 to recruit Rab8a and PI3Kγ for suppression of inflammatory responses. Cell Rep 2018;24(11):3033-3044. [CrossRef]
- Akinc A, Querbes W, De S, Qin J, Frank-Kamenetsky M, Jayaprakash KN, et al. Targeted delivery of RNAi therapeutics with endogenous and exogenous ligand-based mechanisms. Mol Ther. 2010;18(7):1357-64. [CrossRef]
- Köberlin MS, Heinz LX, Superti-Furga G. Functional crosstalk between membrane lipids and TLR biology. Curr Opin Cell Biol 2016;39:28-36. [CrossRef]
- Kornilov FD, Shabalkina AV, Lin C, Volynsky PE, Kot EF, Kayushin AL, et al. The architecture of transmembrane and cytoplasmic juxtamembrane regions of Toll-like receptors. Nat Commun. 2023;14(1):1503. [CrossRef]
- Ndeupen S, Qin Z, Jacobsen S, Bouteau A, Estanbouli H, Igyártó BZ. The mRNA-LNP platform's lipid nanoparticle component used in preclinical vaccine studies is highly inflammatory. iScience 2021;24(12):103479. [CrossRef]
- Finney DJ. The median lethal dose and its estimation. Arch Toxicol. 1985;56(4):215-8. [CrossRef]
- Avila AM, Bebenek I, Bonzo JA, Bourcier T, Davis Bruno KL, Carlson DB, et al. An FDA/CDER perspective on nonclinical testing strategies: Classical toxicology approaches and new approach methodologies (NAMs). Regul Toxicol Pharmacol 2020;114:104662. [CrossRef]
- Li GM. Mechanisms and functions of DNA mismatch repair. Cell Res 2008 Jan;18(1):85-98. [CrossRef]
- Molnár T., Mázló A., Tslaf V., Szöllősi A.G., Emri G., Koncz G. Current translational potential and underlying molecular mechanisms of necroptosis. Cell Death Dis 2019;10 . [CrossRef]
- Park BK, Pirmohamed M, Kitteringham NR. The role of cytochrome P450 enzymes in hepatic and extrahepatic human drug toxicity. Pharmacol Ther 1995;68(3):385-424. [CrossRef]
- Guo Q, Jin Y, Chen X, Ye X, Shen X, Lin M, et al. NF-κB in biology and targeted therapy: new insights and translational implications. Signal Transduct Target Ther 2024;9(1):53. [CrossRef]
- Parameswaran N, Patial S. Tumor necrosis factor-α signaling in macrophages. Crit Rev Eukaryot Gene Expr 2010;20(2):87-103. [CrossRef]
- Mills KHG. IL-17 and IL-17-producing cells in protection versus pathology. Nat Rev Immunol 2023 Jan;23(1):38-54. [CrossRef]
- Wicherska-Pawłowska K, Wróbel T, Rybka J. Toll-like receptors (TLRs), NOD-like receptors (NLRs), and RIG-I-like receptors (RLRs) in innate immunity. TLRs, NLRs, and RLRs ligands as immunotherapeutic agents for hematopoietic diseases. Int J Mol Sci 2021;22(24):13397. [CrossRef]
- Mohan GS, Kumar R. Impairment of tricarboxylic acid cycle (TCA) cycle in Alzheimer's disease: Mechanisms, implications, and potential therapies. Aging Dis 2025;16(5):2553-2574. [CrossRef]
- Lee Y. Roles of circadian clocks in cancer pathogenesis and treatment. Exp Mol Med 2021; 53: 1529–1538. [CrossRef]
- Dunn WG, McLoughlin MA, Vassiliou GS. Clonal hematopoiesis and hematological malignancy. J Clin Invest 2024;134(19):e180065. [CrossRef]
- Zheng Y, Sun L, Jiang T, Zhang D, He D, Nie H. TNFα promotes Th17 cell differentiation through IL-6 and IL-1β produced by monocytes in rheumatoid arthritis. J Immunol Res 2014;2014:385352. [CrossRef]
- Marasco MR, Conteh AM, Reissaus CA, Cupit JE 5th, Appleman EM, Mirmira RG, et al. Interleukin-6 reduces β-cell oxidative stress by linking autophagy with the antioxidant response. Diabetes 2018;67(8):1576-1588. [CrossRef]
- Deretic V, Saitoh T, Akira S. Autophagy in infection, inflammation and immunity. Nat Rev Immunol 2013;13(10):722-37. [CrossRef]
- Chi DS, Fitzgerald SM, Pitts S, Cantor K, King E, Lee SA, et al. MAPK-dependent regulation of IL-1- and beta-adrenoreceptor-induced inflammatory cytokine production from mast cells: implications for the stress response. BMC Immunol 2004;;5:22. [CrossRef]
- Yang HT, Cohen P, Rousseau S. IL-1beta-stimulated activation of ERK1/2 and p38alpha MAPK mediates the transcriptional up-regulation of IL-6, IL-8 and GRO-alpha in HeLa cells. Cell Signal 2008;20(2):375-80. [CrossRef]
- Tengesdal IW, Dinarello A, Powers NE, Burchill MA, Joosten LAB, Marchetti C, et al. Tumor NLRP3-derived IL-1β drives the IL-6/STAT3 axis resulting in sustained MDSC-mediated immunosuppression. Front Immunol 2021;12:661323. [CrossRef]
- Li H, Wu M, Zhao X. Role of chemokine systems in cancer and inflammatory diseases. MedComm (2020) 2022;3(2):e147. [CrossRef]
- Unver N. Macrophage chemoattractants secreted by cancer cells: Sculptors of the tumor microenvironment and another crucial piece of the cancer secretome as a therapeutic target. Cytokine Growth Factor Rev 2019;50:13-18. [CrossRef]
- Nakatsumi H, Matsumoto M, Nakayama KI. Noncanonical pathway for regulation of CCL2 expression by an mTORC1-FOXK1 axis promotes recruitment of tumor-associated macrophages. Cell Rep 2017;21(9):2471-2486. [CrossRef]
- Huda N, Khambu B, Liu G, Nakatsumi H, Yan S, Chen X, et al. Senescence connects autophagy deficiency to inflammation and tumor progression in the liver. Cell Mol Gastroenterol Hepatol 2022;14(2):333-355. [CrossRef]
- Forster III J, Nandi D, Kulkarni A. mRNA-carrying lipid nanoparticles that induce lysosomal rupture activate NLRP3 inflammasome and reduce mRNA transfection efficiency. Biomater Sci 2022;10(19):5566-5582. [CrossRef]
- Shin JN, Rao L, Sha Y, Abdel Fattah E, Hyser J, Eissa NT. p38 MAPK activity is required to prevent hyperactivation of NLRP3 inflammasome. J Immunol 2021;207(2):661-670. [CrossRef]
- Lv JM, Gao YL, Wang LY, Li BD, Shan YL, Wu ZQ, et al. Inhibition of the P38 MAPK/NLRP3 pathway mitigates cognitive dysfunction and mood alterations in aged mice after abdominal surgery plus sevoflurane. Brain Res Bull 2024;217:111059. [CrossRef]
- Sozio MS, Lu C, Zeng Y, Liangpunsakul S, Crabb DW. Activated AMPK inhibits PPAR-α and PPAR-γ transcriptional activity in hepatoma cells. Am J Physiol Gastrointest Liver Physiol 2011;301(4):G739-47. [CrossRef]
- Sun H, Zhu X, Lin W, Zhou Y, Cai W, Qiu L. Interactions of TLR4 and PPARγ, dependent on AMPK signalling pathway contribute to anti-inflammatory effects of vaccariae hypaphorine in endothelial cells. Cell Physiol Biochem 2017;42(3):1227-1239. [CrossRef]
- Harada K, Isse K, Kamihira T, Shimoda S, Nakanuma Y. Th1 cytokine-induced downregulation of PPARγ in human biliary cells relates to cholangitis in primary biliary cirrhosis. Hepatology 2005;41(6):1329-38. [CrossRef]
- Yin R, Dong YG, Li HL. PPARγ phosphorylation mediated by JNK MAPK: a potential role in macrophage-derived foam cell formation. Acta Pharmacol Sin 2006;27(9):1146-52. [CrossRef]
- Su AC, Zhang LY, Zhang JG, Hu YY, Liu XY, Li SC, et al. The regulation of autophagy by p38 MAPK-PPARγ signaling during the brain ischemic tolerance induced by cerebral ischemic preconditioning. DNA Cell Biol 2022;41(9):838-849. [CrossRef]
- Ballav S, Biswas B, Sahu VK, Ranjan A, Basu S. PPARγ partial agonists in disease-fate decision with special reference to cancer. Cells 2022;11(20):3215. [CrossRef]
- Zhao M, Ma J, Li M, Zhang Y, Jiang B, Zhao X, et al. Cytochrome P450 enzymes and drug metabolism in humans. Int J Mol Sci 2021;22(23):12808. [CrossRef]
- Liu F, Aulin LBS, Manson ML, Krekels EHJ, van Hasselt JGC. Unraveling the effects of acute inflammation on pharmacokinetics: A model-based analysis focusing on renal glomerular filtration rate and cytochrome P450 3A4-mediated metabolism. Eur J Drug Metab Pharmacokinet 2023;48(6):623-631. [CrossRef]
- Stanke-Labesque F, Gautier-Veyret E, Chhun S, Guilhaumou R; French Society of Pharmacology and Therapeutics. Inflammation is a major regulator of drug metabolizing enzymes and transporters: Consequences for the personalization of drug treatment. Pharmacol Ther 2020;215:107627. [CrossRef]
- Bayraktar İ, Yalçın N, Demirkan K. The potential interaction between COVID-19 vaccines and clozapine: A novel approach for clinical trials. Int J Clin Pract 2021;75(8):e14441. [CrossRef]
- Imai T, Ochiai S, Ishimaru T, Daitoku H, Miyagawa Y, Fukuhara R, et al. A case report: Clozapine-induced leukopenia and neutropenia after mRNA COVID-19 vaccination. Neuropsychopharmacol Rep 2022;42(2):238-240. [CrossRef]
- Thompson D, Delorme CM, White RF, Honer WG. Elevated clozapine levels and toxic effects after SARS-CoV-2 vaccination. J Psychiatry Neurosci 2021;46(2):E210-E211. [CrossRef]
- Eiermann B, Engel G, Johansson I, Zanger UM, Bertilsson L. The involvement of CYP1A2 and CYP3A4 in the metabolism of clozapine. Br J Clin Pharmacol 1997;44(5):439-46. [CrossRef]
- Villemure S, Trenaman SC, Goralski KB. The impact of COVID-19 infection on cytochrome P450 3A4-mediated drug metabolism and drug interactions. Expert Opin Drug Metab Toxicol 2023;19(6):329-332. [CrossRef]
- Lim SYM, Al Bishtawi B, Lim W. Role of cytochrome P450 2C9 in COVID-19 treatment: Current status and future directions. Eur J Drug Metab Pharmacokinet 2023;48(3):221-240. [CrossRef]
- Szychowski KA, Skóra B, Wójtowicz AK. Engagement of peroxisome proliferator-activated receptor γ (PPARγ) and mammalian target of rapamycin (mTOR) in the triclosan-induced disruption of Cyp450 enzyme activity in an in vitro model of mouse embryo fibroblasts (3T3-L1). Toxicology 2025;511:154031. [CrossRef]
- Hossam Abdelmonem B, Abdelaal NM, Anwer EKE, Rashwan AA, Hussein MA, Ahmed YF, et al. Decoding the role of CYP450 enzymes in metabolism and disease: A comprehensive review. Biomedicines 2024;12(7):1467. [CrossRef]
- Koozi H, Lengquist M, Frigyesi A. C-reactive protein as a prognostic factor in intensive care admissions for sepsis: A Swedish multicenter study. J Crit Care 2020;56:73-79. [CrossRef]
- Lenoir C, Rollason V, Desmeules JA, Samer CF. Influence of inflammation on cytochromes P450 activity in adults: A systematic review of the literature. Front Pharmacol 2021;12:733935. [CrossRef]
- Yang M, Lu Y, Piao W, Jin H. The Translational Regulation in mTOR Pathway. Biomolecules 2022;12(6):802. [CrossRef]
- Shveygert M, Kaiser C, Bradrick SS, Gromeier M. Regulation of eukaryotic initiation factor 4E (eIF4E) phosphorylation by mitogen-activated protein kinase occurs through modulation of Mnk1-eIF4G interaction. Mol Cell Biol 2010;30(21):5160-7. [CrossRef]
- Lin Y, Bian L, Zhu G, Zhang B. Vitronectin promotes proliferation and metastasis of cervical cancer cells via the epithelial-mesenchymal transition. Front Oncol 2024;14:1466264. [CrossRef]
- Gao P, Lu Y, Tang K, Wang W, Wang T, Zhu Y, et al. Ficolin-1 ameliorates pulmonary fibrosis via directly binding to TGF-β1. J Transl Med 2024;22(1):1051. [CrossRef]
- Knabl L, Lee HK, Wieser M, Mur A, Zabernigg A, Knabl L Sr, et al. BNT162b2 vaccination enhances interferon-JAK-STAT-regulated antiviral programs in COVID-19 patients infected with the SARS-CoV-2 Beta variant. Commun Med (Lond) 2022;2(1):17. [CrossRef]
- Burotto M, Chiou VL, Lee JM, Kohn EC. The MAPK pathway across different malignancies: a new perspective. Cancer 2014;120(22):3446-56. [CrossRef]
- Panwar V, Singh A, Bhatt M, Tonk RK, Azizov S, Raza AS, et al. Multifaceted role of mTOR (mammalian target of rapamycin) signaling pathway in human health and disease. Signal Transduct Target Ther 2023;8(1):375. [CrossRef]
- Yu H, Lin L, Zhang Z, Zhang H, Hu H. Targeting NF-κB pathway for the therapy of diseases: mechanism and clinical study. Signal Transduct Target Ther 2020;5(1):209. [CrossRef]
- McCubrey JA, Steelman LS, Chappell WH, Abrams SL, Montalto G, Cervello M, et al. Mutations and deregulation of Ras/Raf/MEK/ERK and PI3K/PTEN/Akt/mTOR cascades which alter therapy response. Oncotarget 2012;3(9):954-87. [CrossRef]
- Nigam M, Punia B, Dimri DB, Mishra AP, Radu AF, Bungau G. Reactive oxygen species: A double-edged sword in the modulation of cancer signaling pathway dynamics. Cells 2025;14(15):1207. [CrossRef]
- Cui D, Qu R, Liu D, Xiong X, Liang T, Zhao Y. The cross talk between p53 and mTOR pathways in response to physiological and genotoxic stresses. Front Cell Dev Biol 2021;9:775507. [CrossRef]
- Bassermann F, Eichner R, Pagano M. The ubiquitin proteasome system - implications for cell cycle control and the targeted treatment of cancer. Biochim Biophys Acta 2014 Jan;1843(1):150-62. [CrossRef]
- Chen Y, Gong W, Dai W, Jiang H, Xu X. E2F1/2/4 mRNA is associated with immune infiltration and are potential biomarkers for the prognosis of human gastric carcinoma. Transl Cancer Res 2021;10(6):2801-2811. [CrossRef]
- Daigh LH, Saha D, Rosenthal DL, Ferrick KR, Meyer T. Uncoupling of mTORC1 from E2F activity maintains DNA damage and senescence. Nat Commun 2024; 15: 9181. [CrossRef]
- Zhang Y, Hao X, Han G, Lu Y, Chen Z, Zhang L, et al. E2F1-mediated GINS2 transcriptional activation promotes tumor progression through PI3K/AKT/mTOR pathway in hepatocellular carcinoma. Am J Cancer Res 2022;12(4):1707-1726.
- Calzone L, Gelay A, Zinovyev A, Radvanyi F, Barillot E. A comprehensive modular map of molecular interactions in RB/E2F pathway. Mol Syst Biol 2008;4:173. [CrossRef]
- Bertonnier-Brouty L, Andersson J, Kaprio T, Hagström J, Bsharat S, Asplund O, et al. E2F transcription factors promote tumorigenicity in pancreatic ductal adenocarcinoma. Cancer Med 2024;13(9):e7187. [CrossRef]
- Wasserman D, Nachum S, Cohen M, Enrico TP, Noach-Hirsh M, Parasol J, et al. Cell cycle oscillators underlying orderly proteolysis of E2F8. Mol Biol Cell 2020;31(8):725-740. [CrossRef]
- Timmers C, Sharma N, Opavsky R, Maiti B, Wu L, Wu J, Orringer D, et al. E2f1, E2f2, and E2f3 control E2F target expression and cellular proliferation via a p53-dependent negative feedback loop. Mol Cell Biol. 2007 Jan;27(1):65-78. Erratum in: Mol Cell Biol 2012;32(9):1758. [CrossRef]
- Spitschak A, Dhar P, Singh KP, Casalegno Garduño R, Gupta SK, Vera J, et al. E2F1-induced autocrine IL-6 inflammatory loop mediates cancer-immune crosstalk that predicts T cell phenotype switching and therapeutic responsiveness. Front Immunol 2024;15:1470368. [CrossRef]
- Li J, Ran C, Li E, Gordon F, Comstock G, Siddiqui H, et al. Synergistic function of E2F7 and E2F8 is essential for cell survival and embryonic development. Dev Cell 2008 Jan;14(1):62-75. [CrossRef]
- Zheng J, Huang J, Xia J, Zhou W, Dai L, Lin S, et al. Transcription factor E2F8 is a therapeutic target in the basal-like subtype of breast cancer. Front Oncol 2023;13:1038787. [CrossRef]
- Vigo E, Müller H, Prosperini E, Hateboer G, Cartwright P, Moroni MC, et al. CDC25A phosphatase is a target of E2F and is required for efficient E2F-induced S phase. Mol Cell Biol 1999;19(9):6379-95. [CrossRef]
- Delgado M, Washam CL, Urbaniak A, Heflin B, Storey AJ, Lan RS, et al. Phosphoproteomics provides novel insights into the response of primary acute lymphoblastic leukemia cells to microtubule depolymerization in G1 phase of the cell cycle. ACS Omega 2021;6(38):24949-24959. [CrossRef]
- Hickey TE, Mudunuri U, Hempel HA, Kemp TJ, Roche NV, Talsania K, et al. Proteomic and serologic assessments of responses to mRNA-1273 and BNT162b2 vaccines in human recipient sera. Front Immunol 2025;15:1502458. [CrossRef]
- Savelsbergh A, Mohr D, Wilden B, Wintermeyer W, Rodnina MV. Stimulation of the GTPase activity of translation elongation factor G by ribosomal protein L7/12. J Biol Chem 2000;275(2):890-4. [CrossRef]
- Chang EC, Philips MR. Spatial segregation of Ras signaling: New evidence from fission yeast. Cell Cycle 2006;5(17):1936-9. [CrossRef]
- Fadhal E. The significance of key proteins in the RAS signaling pathway: Implications for cancer and therapeutic targets. OBM Genetics 2024; 8(2): 236; [CrossRef]
- Lin Y, Parajón E, Yuan Q, Ye S, Qin G, Deng Y, et al. Ras-mediated dynamic and biphasic regulation of cell migration. Proc Natl Acad Sci U S A 2025;122(30):e2503847122. [CrossRef]
- Chippalkatti R, Parisi B, Kouzi F, Laurini C, Ben Fredj N, Abankwa DK. RAS isoform specific activities are disrupted by disease associated mutations during cell differentiation. Eur J Cell Biol 2024;103(2):151425. [CrossRef]
- Wang YF, Dong ZK, Jin WL. Hijacking homeostasis: the brain-body neural circuitry in tumor pathogenesis and emerging therapeutic frontiers. Mol Cancer 2025;24(1):206. [CrossRef]
- Peng Q, Li X, Fang C, Zhu C, Wang T, Yin B, et al. Disrupting calcium homeostasis and glycometabolism in engineered lipid-based pharmaceuticals propel cancer immunogenic death. Acta Pharm Sin B 2025;15(3):1255-1267. [CrossRef]
- Gulluni F, Martini M, Hirsch E. Cytokinetic Abscission: Phosphoinositides and ESCRTs direct the final cut. J Cell Biochem 2017;118(11):3561-3568. Erratum in: J Cell Biochem 2024;125(8):e30626. https://doi.org/10.1002/jcb.30626. [CrossRef]
- Tan X, Thapa N, Choi S, Anderson RA. Emerging roles of PtdIns(4,5)P2 -- beyond the plasma membrane. J Cell Sci 2015;128(22):4047-56. [CrossRef]
- Ajazi A, Bruhn C, Shubassi G, Lucca C, Ferrari E, Cattaneo A, et al. Endosomal trafficking and DNA damage checkpoint kinases dictate survival to replication stress by regulating amino acid uptake and protein synthesis. Dev Cell 2021;56(18):2607-2622.e6. [CrossRef]
- Hurley JH, Coyne AN, Miączyńska M, Stenmark H. The expanding repertoire of ESCRT functions in cell biology and disease. Nature 2025; 642: 877–888. [CrossRef]
- Zhu L, Jorgensen JR, Li M, Chuang YS, Emr SD. ESCRTs function directly on the lysosome membrane to downregulate ubiquitinated lysosomal membrane proteins. Elife 2017;6:e26403. [CrossRef]
- Jia S, Yang Y, Liu J, Wang Z, Bai L. PPARγ controls ESCRT-dependent fibroblast-like synoviocyte exosome biogenesis and alleviates chondrocyte osteoarthritis mediated by exosomal ANXA1. J Orthop Translat 2025;53:187-205. [CrossRef]
- Simonsen JB. A perspective on bleb and empty LNP structures. J Control Release 2024;373:952-961. [CrossRef]
- Schoenmaker L, Witzigmann D, Kulkarni JA, Verbeke R, Kersten G, Jiskoot W, et al. mRNA-lipid nanoparticle COVID-19 vaccines: Structure and stability. Int J Pharm 2021;601:120586. [CrossRef]
- Liao S, Wang S, Wadhwa A, Birkenshaw A, Fox K, Cheng MHY, et al. Transfection potency of lipid nanoparticles containing mRNA depends on relative loading levels. ACS Appl Mater Interfaces 2025;17(2):3097-3105. [CrossRef]
- Lonez C, Vandenbranden M, Ruysschaert JM. Cationic liposomal lipids: From gene carriers to cell signaling. Prog Lipid Res 2008;47(5):340-7. [CrossRef]
- Lonez C, Vandenbranden M, Ruysschaert JM. Cationic lipids activate intracellular signaling pathways. Adv Drug Deliv Rev 2012;64(15):1749-58. [CrossRef]
- Qin Z, Bouteau A, Herbst C, Igyártó BZ. Pre-exposure to mRNA-LNP inhibits adaptive immune responses and alters innate immune fitness in an inheritable fashion. PLoS Pathog 2022;18(9):e1010830. [CrossRef]
- Sellegounder D, Ferrucci L, Anbazhagan R, Basisty N. Editorial: Molecular crosstalk between endocrine factors, paracrine signals, and the immune system during aging. Front Endocrinol (Lausanne) 2023;14:1203755. [CrossRef]
- Panariti A, Miserocchi G, Rivolta I. The effect of nanoparticle uptake on cellular behavior: disrupting or enabling functions? Nanotechnol Sci Appl 2012;5:87-100. [CrossRef]
- Voigt J, Christensen J, Shastri VP. Differential uptake of nanoparticles by endothelial cells through polyelectrolytes with affinity for caveolae. Proc Natl Acad Sci U S A 2014;111(8):2942-7. [CrossRef]
- Wang T, Bai J, Jiang X, Nienhaus GU. Cellular uptake of nanoparticles by membrane penetration: a study combining confocal microscopy with FTIR spectroelectrochemistry. ACS Nano 2012;6(2):1251-9. [CrossRef]
- Gerelli Y. Chapter Three: Exploring interactions between lipid membranes and nanoparticles through neutron and X-ray reflectometry techniques. In A. Iglič, M. Rappolt, & P. Losada-Pérez (Eds.), Advances in Biomembranes and Lipid Self-Assembly 2023; 38: 37–61. Academic Press . [CrossRef]
- Lavagna E, Barnoud J, Rossi G, Monticelli L. Size-dependent aggregation of hydrophobic nanoparticles in lipid membranes. Nanoscale 2020; 12: 9452–9461. [CrossRef]
- Cooper GM. The Cell: A Molecular Approach. 2nd edition. Sunderland (MA): Sinauer Associates; 2000.
- Hammond GRV, Burke JE. Novel roles of phosphoinositides in signaling, lipid transport, and disease. Curr Opin Cell Biol 2020;63:57-67. [CrossRef]
- Skotland T, Sandvig K. The role of PS 18:0/18:1 in membrane function. Nat Commun. 2019;10(1):2752. [CrossRef]
- Heimburg T. The excitable fluid mosaic. Biochim Biophys Acta Biomembr 2023;1865(3):184104. [CrossRef]
- Lupyan D, Mezei M, Logothetis DE, Osman R. A molecular dynamics investigation of lipid bilayer perturbation by PIP2. Biophys J 2010;98(2):240-7. [CrossRef]
- Pike LJ. Lipid rafts: bringing order to chaos. J Lipid Res 2003;44(4):655-67. [CrossRef]
- Levin R, Grinstein S, Schlam D. Phosphoinositides in phagocytosis and macropinocytosis. Biochim Biophys Acta 2015;1851(6):805-23. [CrossRef]
- Weiner OD, Neilsen PO, Prestwich GD, Kirschner MW, Cantley LC, Bourne HR. A PtdInsP(3)- and Rho GTPase-mediated positive feedback loop regulates neutrophil polarity. Nat Cell Biol 2002;4(7):509-13. [CrossRef]
- Guan K, Curtis ER, Lew DJ, Elston TC. Particle-based simulations reveal two positive feedback loops allow relocation and stabilization of the polarity site during yeast mating. PLoS Comput Biol 2023;19(10):e1011523. [CrossRef]
- Fallahi-Sichani M, Linderman JJ. Lipid raft-mediated regulation of G-protein coupled receptor signaling by ligands which influence receptor dimerization: a computational study. PLoS One 2009;4(8):e6604. [CrossRef]
- Wang X, Shi X, Wang R. Regulating mRNA endosomal escape through lipid rafts: A review. Int J Pharm 2025;675:125571. [CrossRef]
- Posor Y, Jang W, Haucke V. Phosphoinositides as membrane organizers. Nat Rev Mol Cell Biol 2022;23(12):797-816. [CrossRef]
- van Meer G, Voelker DR, Feigenson GW. Membrane lipids: where they are and how they behave. Nat Rev Mol Cell Biol 2008;9(2):112-24. [CrossRef]
- Lolicato F, Nickel W, Haucke V, Ebner M. Phosphoinositide switches in cell physiology - From molecular mechanisms to disease. J Biol Chem 2024;300(3):105757. [CrossRef]
- Balla T. Phosphoinositides: Tiny lipids with giant impact on cell regulation. Physiol Rev 2013;93(3):1019-137. [CrossRef]
- Eramo MJ, Mitchell CA. Regulation of PtdIns(3,4,5)P3/Akt signalling by inositol polyphosphate 5-phosphatases. Biochem Soc Trans 2016;44(1):240-52. [CrossRef]
- Xu S, Cao B, Xuan G, Xu S, An Z, Zhu C, et al. Function and regulation of Rab GTPases in cancers. Cell Biol Toxicol 2024;40(1):28. [CrossRef]
- Koike S, Jahn R. Rab GTPases and phosphoinositides fine-tune SNAREs dependent targeting specificity of intracellular vesicle traffic. Nat Commun 2024;15(1):2508. [CrossRef]
- Nishimura T, Gecht M, Covino R, Hummer G, Surma MA, Klose C, et al. Osh proteins control nanoscale lipid organization necessary for PI(4,5)P2 synthesis. Mol Cell 2019;75(5):1043-1057.e8. [CrossRef]
- Heckle LA, Kozminski KG. Osh-dependent and -independent regulation of PI4P levels during polarized growth of Saccharomyces cerevisiae. Molecular Biology of the Cell 2023; 34(11). [Epub ahead of print].
- Raychaudhuri S, Prinz WA. The diverse functions of oxysterol-binding proteins. Annu Rev Cell Dev Biol 2010;26:157-77. [CrossRef]
- Omo-Lamai S, Wang Y, Patel MN, Milosavljevic A, Zuschlag D, Poddar S, et al. Limiting endosomal damage sensing reduces inflammation triggered by lipid nanoparticle endosomal escape. Nat Nanotechnol 2025;20(9):1285-1297. [CrossRef]
- Cheng Y, Zhao E, Yang X, Luo C, Zi G, Wang R, et al. Entrapment of lipid nanoparticles in peripheral endosomes but not lysosomes impairs intracellular trafficking and endosomal escape. Int J Pharm 2025;669:125024. [CrossRef]
- Puranik A, Lenehan PJ, Silvert E, Niesen MJM, Corchado-Garcia J, O'Horo JC, et al. Comparative effectiveness of mRNA-1273 and BNT162b2 against symptomatic SARS-CoV-2 infection. Med 2022;3(1):28-41.e8. [CrossRef]
- Granados-Riveron JT, Aquino-Jarquin G. Engineering of the current nucleoside-modified mRNA-LNP vaccines against SARS-CoV-2. Biomed Pharmacother 2021;142:111953. [CrossRef]
- Harvey RD, Ara N, Heenan RK, Barlow DJ, Quinn PJ, Lawrence MJ. Stabilization of distearoylphosphatidylcholine lamellar phases in propylene glycol using cholesterol. Mol Pharm 2013;10(12):4408-17. [CrossRef]
- Skotland T, Kavaliauskiene S, Sandvig K. The role of lipid species in membranes and cancer-related changes. Cancer Metastasis Rev 2020;39(2):343-360. [CrossRef]
- Li J, Wang X, Zhang T, Wang C, Huang Z, Luo X, Deng Y. A review on phospholipids and their main applications in drug delivery systems. Asian J Pharm Sci 2015; 10(2): 81-98. [CrossRef]
- McMaster CR. From yeast to humans - roles of the Kennedy pathway for phosphatidylcholine synthesis. FEBS Lett 2018;592(8):1256-1272. [CrossRef]
- Cummings R, Parinandi N, Wang L, Usatyuk P, Natarajan V. Phospholipase D/phosphatidic acid signal transduction: role and physiological significance in lung. Mol Cell Biochem 2002;234-235(1-2):99-109.
- Wagner K, Brezesinski G. Phospholipase D activity is regulated by product segregation and the structure formation of phosphatidic acid within model membranes. Biophys J 2007;93(7):2373-83. [CrossRef]
- Bruntz RC, Lindsley CW, Brown HA. Phospholipase D signaling pathways and phosphatidic acid as therapeutic targets in cancer. Pharmacol Rev 2014;66(4):1033-79. [CrossRef]
- Semenkovich CF, Goldberg AC, Goldberg IJ. Disorders of Lipid Metabolism. Chapter 37 in Melmed S, Polonsky KS, Larsen PR, Kronenberg HM, Eds. Williams Textbook of Endocrinology (Thirteenth Edition). Elsevier 2016: 1660-1700. [CrossRef]
- Blunsom NJ, Cockcroft S. Phosphatidylinositol synthesis at the endoplasmic reticulum. Biochim Biophys Acta Mol Cell Biol Lipids 2020 Jan;1865(1):158471. [CrossRef]
- Pearce B, Jakobson K, Morrow C, Murphy S. Phosphatidic acid promotes phosphoinositide metabolism and DNA synthesis in cultured cortical astrocytes. Neurochem Int 1994;24(2):165-71. [CrossRef]
- Quick J, Santos ND, Cheng MHY, Chander N, Brimacombe CA, Kulkarni J, et al. Lipid nanoparticles to silence androgen receptor variants for prostate cancer therapy. J Control Release 2022;349:174-183. [CrossRef]
- Jeschek D, Lhota G, Wallner J, Vorauer-Uhl K. A versatile, quantitative analytical method for pharmaceutical relevant lipids in drug delivery systems. J Pharm Biomed Anal 2016;119:37-44. [CrossRef]
- Rezaei S, Blick EE, Mineart KP, Kelley EG. Chapter Three - Linking chemical degradation and physical instability of lipid vesicles. Advances in Biomembranes and Lipid Self-Assembly 2025; 41: 47-64. [CrossRef]
- Sarkar S, Carroll B, Buganim Y, Maetzel D, Ng AH, Cassady JP, et al. Impaired autophagy in the lipid-storage disorder Niemann-Pick type C1 disease. Cell Rep 2013;5(5):1302-15. [CrossRef]
- Dall'Armi C, Devereaux KA, Di Paolo G. The role of lipids in the control of autophagy. Curr Biol 2013;23(1):R33-45. [CrossRef]
- Zhang S, Peng X, Yang S, Li X, Huang M, Wei S, et al. The regulation, function, and role of lipophagy, a form of selective autophagy, in metabolic disorders. Cell Death Dis 2022;13(2):132. [CrossRef]
- Jarocki M, Turek K, Saczko J, Tarek M, Kulbacka J. Lipids associated with autophagy: mechanisms and therapeutic targets. Cell Death Discov 2024;10(1):460. [CrossRef]
- Karim M, Mishra M, Lo CW, Saul S, Cagirici HB, Gourdelier M, et al. PIP4K2C inhibition reverses autophagic flux impairment induced by SARS-CoV-2. Nat Commun 2025;16(1):6397. [CrossRef]
- Haucke V, Kozlov MM. Membrane remodeling in clathrin-mediated endocytosis. J Cell Sci 2018;131(17):jcs216812. [CrossRef]
- Rigby RE, Rehwinkel J. RNA degradation in antiviral immunity and autoimmunity. Trends Immunol. 2015;36(3):179-88. [CrossRef]
- Acevedo-Whitehouse K, Bruno R. Potential health risks of mRNA-based vaccine therapy: A hypothesis. Med Hypotheses 2023;171:111015. [CrossRef]
- Fung SYS, Xǔ XJ, Wu M. Nonlinear dynamics in phosphoinositide metabolism. Curr Opin Cell Biol 2024;88:102373. [CrossRef]
- Thiemicke A, Neuert G. Rate thresholds in cell signaling have functional and phenotypic consequences in non-linear time-dependent environments. Front Cell Dev Biol 2023;11:1124874. [CrossRef]
- Hou X, Chen Y, Carrillo ND, Cryns VL, Anderson RA, Sun J, et al. Phosphoinositide signaling at the cytoskeleton in the regulation of cell dynamics. Cell Death Dis 2025;16(1):296. [CrossRef]
- Adhikari H, Cullen PJ. Role of phosphatidylinositol phosphate signaling in the regulation of the filamentous-growth mitogen-activated protein kinase pathway. Eukaryot Cell 2015;14(4):427-40. [CrossRef]
- Wang L, Eghtesad S, Clemens PR. Migration of dendritic cells from murine skeletal muscle. Immunobiology 2011;216(1-2):195-9. [CrossRef]
- Woodland DL, Kohlmeier JE. Migration, maintenance and recall of memory T cells in peripheral tissues. Nat Rev Immunol 2009;9(3):153-61. [CrossRef]
- Zhang Y, Garcia-Ibanez L, Ulbricht C, Lok LSC, Pike JA, Mueller-Winkler J, et al. Recycling of memory B cells between germinal center and lymph node subcapsular sinus supports affinity maturation to antigenic drift. Nat Commun 2022;13(1):2460. [CrossRef]
- Chazaud B. Inflammation and skeletal muscle regeneration: Leave it to the macrophages! Trends Immunol 2020;41(6):481-492. [CrossRef]
- Sackstein R, Schatton T, Barthel SR. T-lymphocyte homing: an underappreciated yet critical hurdle for successful cancer immunotherapy. Lab Invest 2017;97(6):669-697. [CrossRef]
- Simonis A, Theobald SJ, Koch AE, Mummadavarapu R, Mudler JM, Pouikli A, et al. Persistent epigenetic memory of SARS-CoV-2 mRNA vaccination in monocyte-derived macrophages. Mol Syst Biol 2025;21(4):341-360. [CrossRef]
- Ochoa D, Bradley D, Beltrao P. Evolution, dynamics and dysregulation of kinase signalling. Curr Opin Struct Biol 2018;48:133-140. [CrossRef]
- Pelley JW. Membranes and Intracellular Signal Transduction. Chapter 5 in Pelley JW, Ed. Elsevier's Integrated Biochemistry. Mosby, 2007, 37-46. [CrossRef]
- Grecco HE, Schmick M, Bastiaens PI. Signaling from the living plasma membrane. Cell 2011;144(6):897-909. [CrossRef]
- Szischik CL, Reves Szemere J, Balderrama R, Sánchez de la Vega C, Ventura AC. Transient frequency preference responses in cell signaling systems. npj Syst Biol Appl 2024; 10: 86. [CrossRef]
- Cobb MH, Goldsmith EJ. Dimerization in MAP-kinase signaling. Trends Biochem Sci 2000 Jan;25(1):7-9. [CrossRef]
- Fossett N. Signal transduction pathways, intrinsic regulators, and the control of cell fate choice. Biochim Biophys Acta 2013;1830(2):2375-84. [CrossRef]
- Landry BD, Clarke DC, Lee MJ. Studying Cellular Signal Transduction with OMIC Technologies. J Mol Biol 2015;427(21):3416-40. [CrossRef]
- Vijay S, Gujral TS. Non-linear deep neural network for rapid and accurate prediction of phenotypic responses to kinase inhibitors. iScience 2020;23(5):101129. [CrossRef]
- Jiang Y, Akhavan Aghdam Z, Li Y, Zid BM, Hao N. A protein kinase A-regulated network encodes short- and long-lived cellular memories. Sci Signal 2020;13(632):eaay3585. [CrossRef]
- Ghomlaghi M, Hart A, Hoang N, Shin S, Nguyen LK. Feedback, Crosstalk and competition: ingredients for emergent non-linear behaviour in the PI3K/mTOR signalling network. Int J Mol Sci 2021;22(13):6944. [CrossRef]
- Veres T, Kerestély M, Kovács BM, Keresztes D, Schulc K, Seitz E, et al. Cellular forgetting, desensitisation, stress and ageing in signalling networks. When do cells refuse to learn more? Cell Mol Life Sci 2024;81(1):97. [CrossRef]
- Rauch N, Rukhlenko OS, Kolch W, Kholodenko BN. MAPK kinase signalling dynamics regulate cell fate decisions and drug resistance. Curr Opin Struct Biol 2016;41:151-158. [CrossRef]
- Lavoie H, Gagnon J, Therrien M. ERK signalling: a master regulator of cell behaviour, life and fate. Nat Rev Mol Cell Biol 2020; 21: 6070632. [CrossRef]
- Parres-Gold J, Levine M, Emert B, Stuart A, Elowitz MB. Contextual computation by competitive protein dimerization networks. Cell 2025;188(7):1984-2002.e17. [CrossRef]
- Avraham R, Yarden Y. Feedback regulation of EGFR signalling: decision making by early and delayed loops. Nat Rev Mol Cell Biol 2011;12(2):104-17. [CrossRef]
- Wen PJ, Osborne SL, Meunier FA. Phosphoinositides in neuroexocytosis and neuronal diseases. Curr Top Microbiol Immunol 2012;362:87-98. PMID: 23086414. [CrossRef]
- Tsuruta F. New insights into the functions of PtdIns(3,5)P2 in the pathogenisis of neurodegenerative disorders. Neural Regen Res 2016;11(2):240-1. [CrossRef]
- Vater M, Möckl L, Gormanns V, Schultz Fademrecht C, Mallmann AM, Ziegart-Sadowska K, et al. New insights into the intracellular distribution pattern of cationic amphiphilic drugs. Sci Rep 2017;7:44277. [CrossRef]
- Kazmi F, Hensley T, Pope C, Funk RS, Loewen GJ, Buckley DB, et al. Lysosomal sequestration (trapping) of lipophilic amine (cationic amphiphilic) drugs in immortalized human hepatocytes (Fa2N-4 cells). Drug Metab Dispos 2013;41(4):897-905. [CrossRef]
- Van Rooijen LA, Bazan NG. Cationic amphiphilic drugs perturb the metabolism of inosititides and phosphatidic acid in photoreceptor membranes. Biochem Biophys Res Commun 1986;134(1):378-85. [CrossRef]
- Houthaeve G, De Smedt SC, Braeckmans K, De Vos WH. The cellular response to plasma membrane disruption for nanomaterial delivery. Nano Converg 2022;9(1):6. [CrossRef]
| Pathway | Direction of NES | Consequences/Biological Function |
| Mismatch repair | Downregulated | DNA repair inefficiency; Promotion of tumorigenesis [140] |
| Phagosome | Upregulated | Induction of phagocytosis and autophagy |
| Necroptosis | Upregulated | Inflammatory form of cell death associated with many human diseases [141] |
| Apoptosis | Upregulated | Induction of programmed cell death |
| Metabolism of xenobiotics by cytochrome P450 | Downregulated | Impaired cytochrome P450 activity in the liver may lead to increased drug toxicity [142] |
| NF-κB | Upregulated | A key regulator of the immune system, inflammation, cell survival, and stress responses [143] |
| TNF | Upregulated | A powerful pro-inflammatory agent that regulates many facets of macrophage function [144] |
| IL-17 | Upregulated | Promotes proinflammatory cytokine production, neutrophil recruitment, tissue remodeling, and antimicrobial defenses [145] |
| Toll-like receptors, RIG-1-like receptors, Nod-like receptors | Upregulated | These receptors activate inflammatory and immune responses [146] |
| TCA cycle | Downregulated | Impaired TCA cycle is a feature of Alzheimer’s disease [147] |
| Circadian rhythm | Downregulated | Dysregulation of circadian rhythms increases cancer susceptibility [148] |
| Hematopoetic cell lineage | Upregulated | Increased clonal hematopoiesis leads to hematological malignancy [149] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




