Submitted:
05 November 2025
Posted:
06 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Data Source and Preparation
2.2. Disproportionality Analysis

3. Results
3.1. Disproportionality Signals for Cardiac Adverse Events
| Adverse Event Preferred Term (PT) | ADR | Number of Drug Events | Number of reports for AE in Full database (all drugs) | ROR (95% Cl) | |
|---|---|---|---|---|---|
| 1 | Cardiac failure | Yes | 552 | 45633 | 3.183 (2.92 5- 3.463) |
| 2 | Atrial fibrillation | Yes | 529 | 52383 | 2.649 (2.43 - 2.887) |
| 3 | Cardiomyopathy | Yes | 97 | 9014 | 2.81 (2.3 - 3.433) |
| 4 | Cardiac amyloidosis | Yes | 80 | 661 | 35.578 (28.158 - 44.953) |
| 5 | Atrial flutter | Yes | 68 | 4115 | 4.339 (3.414 - 5.515) |
| 6 | Myocarditis | Yes | 57 | 6611 | 2.245 (1.73 - 2.914) |
| 7 | Left Ventricular dysfunction | Yes | 55 | 3393 | 4.254 (3.259 - 5.554) |
| 8 | Cardiac Failure acute | Yes | 48 | 3437 | 3.657 (2.75 -4.862) |
| 9 | Supraventricular tachycardia | Yes | 43 | 5150 | 2.173 (1.61 - 2.934) |
| 10 | Right ventricular failure | Yes | 33 | 3740 | 2.297 (1.631 - 3.237) |
| 11 | Congestive cardiomyopathy | Yes | 36 | 2910 | 3.233 (2.327 - 4.492) |
| 12 | Hypertrophic cardiomyopathy | Yes | 12 | 912 | 3.44 (1.946 - 6.081) |
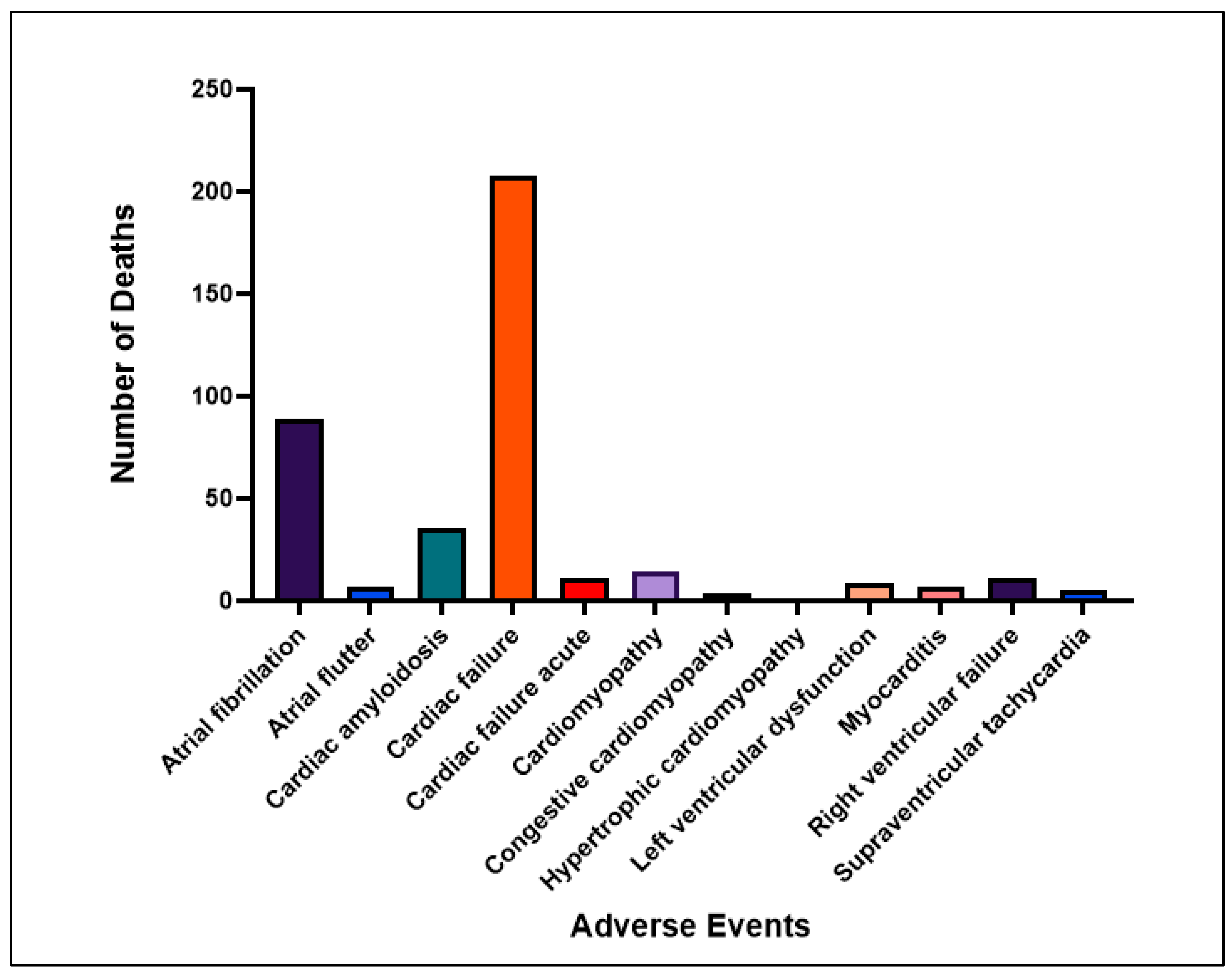
3.2. Clinical Severity and Mortality Outcomes
| Adverse Event | Hospitalization | Life-threatening | Death | Other Outcomes |
|---|---|---|---|---|
| Atrial fibrillation | 216 | 32 | 89 |
192 |
| Atrial flutter | 39 | 9 | 7 | 13 |
| Cardiac amyloidosis | 7 | 6 | 36 | 31 |
| Cardiac failure | 197 | 45 | 208 | 104 |
| Cardiac failure acute | 24 | 5 | 11 | 6 |
| Cardiomyopathy | 24 | 6 | 14 | 53 |
| Congestive cardiomyopathy | 18 | 2 | 4 | 12 |
| Hypertrophic cardiomyopathy | 10 | 0 | 1 | 1 |
| Left ventricular dysfunction | 22 | 6 | 9 | 18 |
| Myocarditis | 20 | 6 | 6 | 25 |
| Right ventricular failure | 4 | 7 | 21 |
1 |
| Supraventricular tachycardia | 21 | 10 | 4 | 8 |
3.3. Suspected Attribution and Causality Patterns
| Cardiac Failure | Atrial Fibrilation | Atrial Flutter | Cardiac Failure Acute | Cardiomyopathy | Congestive Cardiomyopathy | Hypertrophic Cardiomyopathy | Left Ventricular Dysfunction | Myocarditis | Right Ventricular Failure | Supraventricular Tachycardia | Cardiac Amyloidosis | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Suspect Attribution | # of Unique cases | 552 | 529 | 68 | 48 | 97 | 36 | 12 | 55 | 57 | 33 | 43 | 80 |
| Other | 61 (11.1%) | 89 (16.8%) | 12 (17.6%) | 2 (4.2%) | 6 (6.2%) | 1 (2.8%) | 1 (8.3%) | 6 (10.9%) | 1 (1.8%) | 4 (12.1%) | 2 (4.7%) | 5 (6.2%) | |
| Primary Suspect | 237 (42.9%) | 219 (41.4%) | 22 (32.4%) | 29 (60.4%) | 66 (68.0%) | 18 (50.0%) | 5 (41.7%) | 28 (50.9%) | 37 (64.9%) | 16 (48.5%) | 23 (53.5%) | 39 (48.8%) | |
| Secondary Suspect | 254 (46.0%) | 221 (41.4%) | 34 (50.0%) | 17 (35.4%) | 25 (25.8%) | 17 (47.2%) | 6 (50.0%) | 21 (38.18%) | 17 (29.8% | 13 (39.4%) | 18 (41.9%) | 36 (45.0%) | |
| - | |||||||||||||
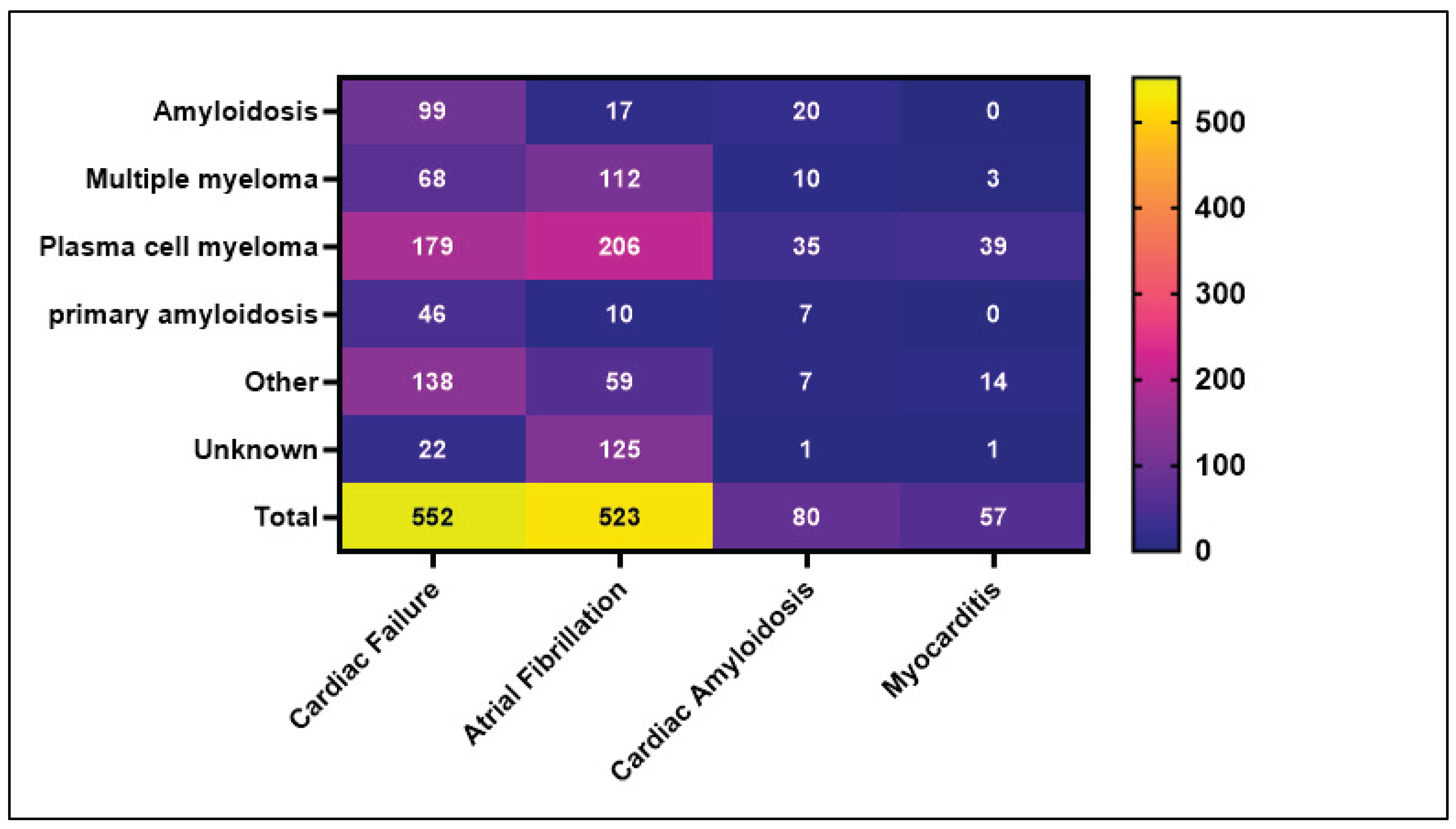
| Indication Subtype | Amyloidosis | 99 (17.9%) | 17 (3.2%) | 4 (5.9%) | 3 (6.25%) | 2 (2.1%) | 1 (2.8%) | 0 (0.0%) | 0 (0.00%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 20 (25%) |
| Multiple myeloma | 68 (12.3%) | 112 (21.2%) | 17 (25.0%) | 6 (12.5%) | 31 (32.0%) | 4 (11.1%) | 2 (16.7%) | 7 (12.73%) | 3 (5.2%) | 2 (6.1%) | 17 (39.5%) | 10 (12.5%) | |
| Plasma cell myeloma | 179 (32.4%) | 206 (38.9%) | 34 (50.0%) | 28 (58.33%) | 35 (36.1%) | 19 (52.8%) | 8 (66.7%) | 19 (34.55%) | 39 (67.2%) | 8 (24.2%) | 14 (32.6%) | 35 (43.8%) | |
| Primary amyloidosis | 46 (8.33%) | 10 (1.9%) | 0 (0.0%) | 3 (6.25%) | 3 (3.1%) | 0 (0.0%) | 0 (0.0%) | 0 (0.00%) | 0 (0.0%) | 4 (12.1%) | 0 (0.0%) | 7 (8.8%) | |
| Other | 138 (25.0%) | 59 (11.2%) | 11 (16.2%) | 4 (8.33%) | 20 (20.6%) | 12 (33.3%) | 1 (8.3%) | 21 (38.18%) | 15 (24.6%) | 17 (51.5%) | 8 (18.6%) | 7 (8.8%) | |
| Unknown | 22 (3.9%) | 125 (23.6%) | 2 (2.9%) | 4 (8.33%) | 6 (6.2%) | 0 (0.0%) | 1 (8.3%) | 8 (14.55%) | 1 (1.7%) | 2 (6.1%) | 4 (9.3%) | 1 (1.2%) | |
| - | |||||||||||||
| Age Subtype | <18 | 19 (3.4%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 1 (1.0%) | 0 (0.0%) | 0 (0.0%) | 6 (10.91%) | 2 (3.5%) | 2 (6.1%) | 0 (0.0%) | 0 (0.0%) |
| 18-44 | 18 (3.3%) | 1 (0.2%) | 0 (0.0%) | 1 (2.1%) | 6 (6.2%) | 0 (0.0%) | 0 (0.0%) | 4 (7.27%) | 7 (12.3%) | 10 (30.3%) | 0 (0.0%) | 1 (1.2%) | |
| 45-64 | 113 (20.5%) | 102 (19.3%) | 15 (22.1%) | 13 (27.1%) | 31 (32.0%) | 18 (50.0%) | 1 (8.3%) | 20 (36.36%) | 19 (33.3%) | 12 (36.4%) | 15 (34.9%) | 31 (38.8%) | |
| 65+ | 230 (41.7%) | 262 (49.5%) | 33 (48.5%) | 23 (47.9%) | 26 (26.8%) | 12 (33.3%) | 11 (91.7%) | 13 (23.64%) | 21 (36.8%) | 4 (12.1%) | 13 (30.2%) | 26 (32.5%) | |
| Unknown | 172 (31.2%) | 164 (31.0%) | 20 (29.4%) | 11 (22.9%) | 33 (34.0%) | 6 (16.7%) | 0 (0.0%) | 12 (21.82%) | 8 (14.0%) | 5 (15.2%) | 15 (34.9%) | 22 (28.7%) | |
| - | |||||||||||||
| Reporter Country | United States | 100 (18.1%) | 218 (41.2%) | 29 (42.6%) | 2 (4.17%) | 39 (40.2%) | 2 (5.6%) | 5 (41.7%) | 18 (32.73%) | 27 (47.4%) | 13 (39.4%) | 12 (27.9%) | 5 (6.25%) |
| France (FR) | 80 (14.5%) | 45 (8.5%) | 1 (1.5%) | 4 (8.33%) | 2 (2.1%) | 17 (47.2%) | 5 (41.7%) | 1 (1.82%) | 5 (8.8%) | 0 (0.0%) | 1 (2.3%) | 12 (15.0%) | |
| Japan (JP) | 66 (12.0%) | 18 (3.4%) | 5 (5.9%) | 10 (20.83%) | 4 (4.1%) | 1 (2.8%) | 0 (0.0%) | 0 (0.00%) | 0 (0.0%) | 2 (6.1%) | 7 (16.3%) | 12 (15.0%) | |
| Germany(DE) | 76 (13.8%) | 30 (5.7%) | 8 (11.8%) | 0 (0.00%) | 2 (2.1%) | 0 (0.0%) | 0 (0.0%) | 2 (3.64%) | 2 (3.5%) | 1 (3.05%) | 2 (4.7%) | 0 (0.0%) | |
| United Kingdom (GB) | 11 (2.0%) | 45 (8.5%) | 6 (8.8%) | 3 (6.25%) | 1 (1.0%) | 0 (0.0%) | 0 (0.0%) | 0 (0.00%) | 2 (3.5%) | 0 (0.0%) | 1 (2.3%) | 9 (11.25%) | |
| Other Countries | 215 (38.9%) | 164 (31.0%) | 20 (29.4%) | 29 (60.42%) | 3 (3.1%) | 1 (2.8%) | 1 (8.3%) | 34 (61.82%) | 21 (36.8%) | 17 (51.5%) | 16 (37.2%) | 39 (48.75%) | |
| Unknown | 4 (0.7%) | 9 (1.7%) | 0 (0.0%) | 0 (0.00%) | 46 (47.4%) | 15 (41.7%) | 1 (8.3%) | 0 (0.00%) | 0 (0.0%) | 0 (0.0%) | 4 (0.09%) | 3 (3.75%) |
3.4. Demographic and Treatment Context
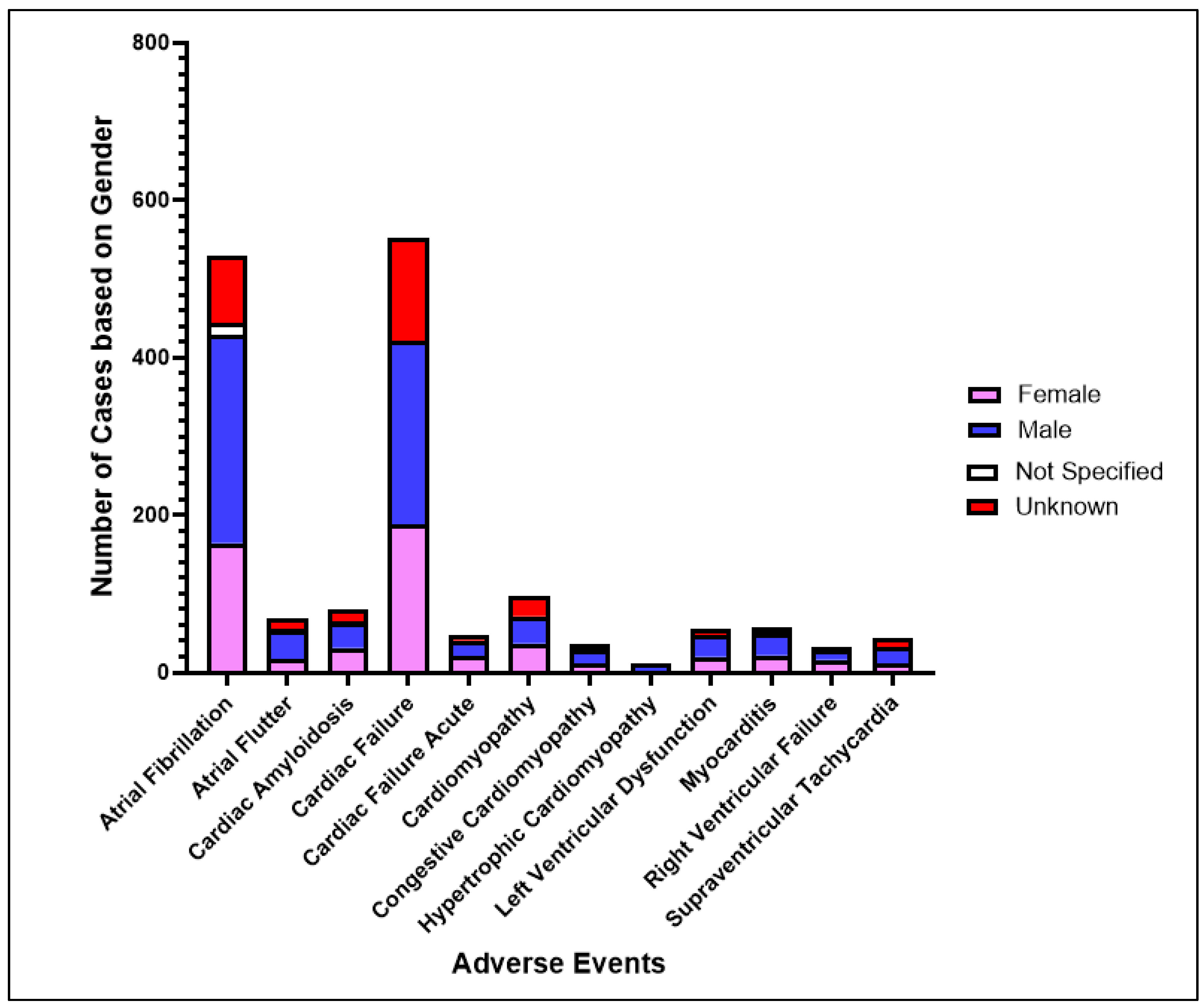
3.5. Disease-Specific Patterns and Subgroup Risk
4. Discussion
5. Conclusion
6. Study Limitations
References
- Richardson, P.G., et al., A phase 2 study of bortezomib in relapsed, refractory myeloma. N Engl J Med, 2003. 348(26): p. 2609-17. [CrossRef]
- Moreau, P., et al., Subcutaneous versus intravenous administration of bortezomib in patients with relapsed multiple myeloma: a randomised, phase 3, non-inferiority study. Lancet Oncol, 2011. 12(5): p. 431-40. [CrossRef]
- Kistler, K.D., et al., Incidence and Risk of Cardiac Events in Patients With Previously Treated Multiple Myeloma Versus Matched Patients Without Multiple Myeloma: An Observational, Retrospective, Cohort Study. Clin Lymphoma Myeloma Leuk, 2017. 17(2): p. 89-96 e3. [CrossRef]
- Ling, Y.H., et al., Reactive oxygen species generation and mitochondrial dysfunction in the apoptotic response to Bortezomib, a novel proteasome inhibitor, in human H460 non-small cell lung cancer cells. J Biol Chem, 2003. 278(36): p. 33714-23. [CrossRef]
- Kuroda, K. and H. Liu, The proteasome inhibitor, bortezomib, induces prostate cancer cell death by suppressing the expression of prostate-specific membrane antigen, as well as androgen receptor. Int J Oncol, 2019. 54(4): p. 1357-1366. [CrossRef]
- Garcia, M., et al., Cardiovascular Disease in Women: Clinical Perspectives. Circ Res, 2016. 118(8): p. 1273-93.
- Christoffersen, M., et al., Transthyretin Tetramer Destabilization and Increased Mortality in the General Population. JAMA Cardiol, 2025. 10(2): p. 155-163. [CrossRef]
- Herrmann, J., et al., Vascular Toxicities of Cancer Therapies: The Old and the New--An Evolving Avenue. Circulation, 2016. 133(13): p. 1272-89.
- Gandolfi, S., et al., The proteasome and proteasome inhibitors in multiple myeloma. Cancer Metastasis Rev, 2017. 36(4): p. 561-584. [CrossRef]
- Munoz, M.A., et al., Sociodemographic Characteristics of Adverse Event Reporting in the USA: An Ecologic Study. Drug Saf, 2024. 47(4): p. 377-387. [CrossRef]
- Sakaeda, T., et al., Data mining of the public version of the FDA Adverse Event Reporting System. Int J Med Sci, 2013. 10(7): p. 796-803. [CrossRef]
- Bate, A. and S.J. Evans, Quantitative signal detection using spontaneous ADR reporting. Pharmacoepidemiol Drug Saf, 2009. 18(6): p. 427-36. [CrossRef]
- Chari, A., et al., Analysis of carfilzomib cardiovascular safety profile across relapsed and/or refractory multiple myeloma clinical trials. Blood Adv, 2018. 2(13): p. 1633-1644. [CrossRef]
- Movila, D.E., et al., Cardiac Amyloidosis: A Narrative Review of Diagnostic Advances and Emerging Therapies. Biomedicines, 2025. 13(5). [CrossRef]
- Vogel, J., et al., Current Therapies and Future Horizons in Cardiac Amyloidosis Treatment. Curr Heart Fail Rep, 2024. 21(4): p. 305-321. [CrossRef]
- Zheng, Y., et al., Cardiovascular Toxicity of Proteasome Inhibitors in Multiple Myeloma Therapy. Curr Probl Cardiol, 2023. 48(3): p. 101536. [CrossRef]
- Xiang, C., et al., Infection-related adverse events comparison of bortezomib, carfilzomib and ixazomib: a pharmacovigilance study based on FAERS. Expert Opin Drug Saf, 2025: p. 1-10. [CrossRef]
- Chen, Q., et al., Coexisting atrial fibrillation and cancer: time trends and associations with mortality in a nationwide Dutch study. Eur Heart J, 2024. 45(25): p. 2201-2213. [CrossRef]
- Georgiopoulos, G., et al., Cardiovascular Toxicity of Proteasome Inhibitors: Underlying Mechanisms and Management Strategies: JACC: CardioOncology State-of-the-Art Review. JACC CardioOncol, 2023. 5(1): p. 1-21.
- Armenian, S.H., C. Lacchetti, and D. Lenihan, Prevention and Monitoring of Cardiac Dysfunction in Survivors of Adult Cancers: American Society of Clinical Oncology Clinical Practice Guideline Summary. J Oncol Pract, 2017. 13(4): p. 270-275. [CrossRef]
- Jang, B., et al., Real-world incidence and risk factors of bortezomib-related cardiovascular adverse events in patients with multiple myeloma. Blood Res, 2024. 59(1): p. 3. [CrossRef]
- Mitra, A.K., et al., A gene expression signature distinguishes innate response and resistance to proteasome inhibitors in multiple myeloma. Blood Cancer J, 2017. 7(6): p. e581. [CrossRef]
- Ruckrich, T., et al., Characterization of the ubiquitin-proteasome system in bortezomib-adapted cells. Leukemia, 2009. 23(6): p. 1098-105. [CrossRef]
- Curigliano, G., et al., Management of cardiac disease in cancer patients throughout oncological treatment: ESMO consensus recommendations. Ann Oncol, 2020. 31(2): p. 171-190. [CrossRef]
- Caponetti, A.G., et al., Screening approaches to cardiac amyloidosis in different clinical settings: Current practice and future perspectives. Front Cardiovasc Med, 2023. 10: p. 1146725. [CrossRef]
- Sundaravel, S.H., et al., Bortezomib-Induced Reversible Cardiomyopathy: Recovered With Guideline-Directed Medical Therapy. Cureus, 2021. 13(12): p. e20295. [CrossRef]
- Hedhli, N. and C. Depre, Proteasome inhibitors and cardiac cell growth. Cardiovasc Res, 2010. 85(2): p. 321-9. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




