Submitted:
04 November 2025
Posted:
05 November 2025
You are already at the latest version
Abstract
White mold (Sclerotinia sclerotiorum) affects potato production and quality in Sinaloa, Mexico. This study aimed to determine the in vitro efficacy of Trichoderma azevedoi, T. afroharzianum, T. asperellum and T. asperelloides in inhibiting the mycelial growth and sclerotia production of S. sclerotiorum. Field studies were also conducted to evaluate the effectiveness of a Trichoderma spp. combination in controlling the disease and reducing sclerotia production, as well as increasing crop yield in semi-commercial plots. In parallel, the alternating use of the antagonist combination with synthetic fungicides was assessed; an additional treatment involved using synthetic fungicides alone. The in vitro tests demonstrated the efficacy of all four Trichoderma species against the pathogen. The Trichoderma combination also significantly controlled white mold under field conditions. The alternating application of Trichoderma species with synthetic fungicides was similarly effective, while the fungicides-alone treatment was less effective than the two aforementioned treatments. The results highlight the potential of using a mixture of these four Trichoderma species to control potato white mold in Sinaloa, which could help reduce the reliance on synthetic fungicides for disease management.
Keywords:
1. Introduction
2. Results
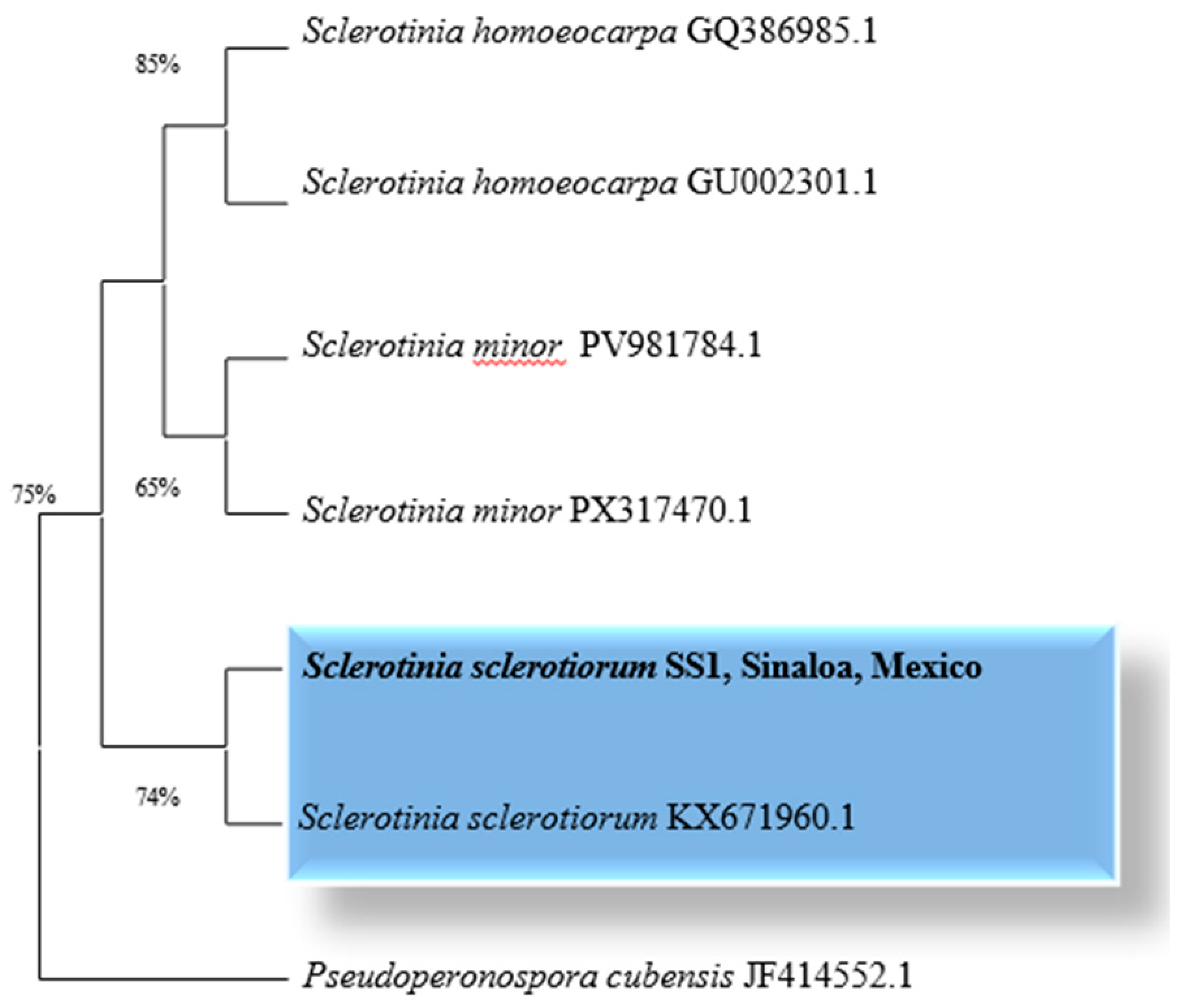
2.1. Molecular Identification of Sclerotinia sclerotiorum
2.2. In Vitro Inhibition of Sclerotinia sclerotiorum by Four Trichoderma Species
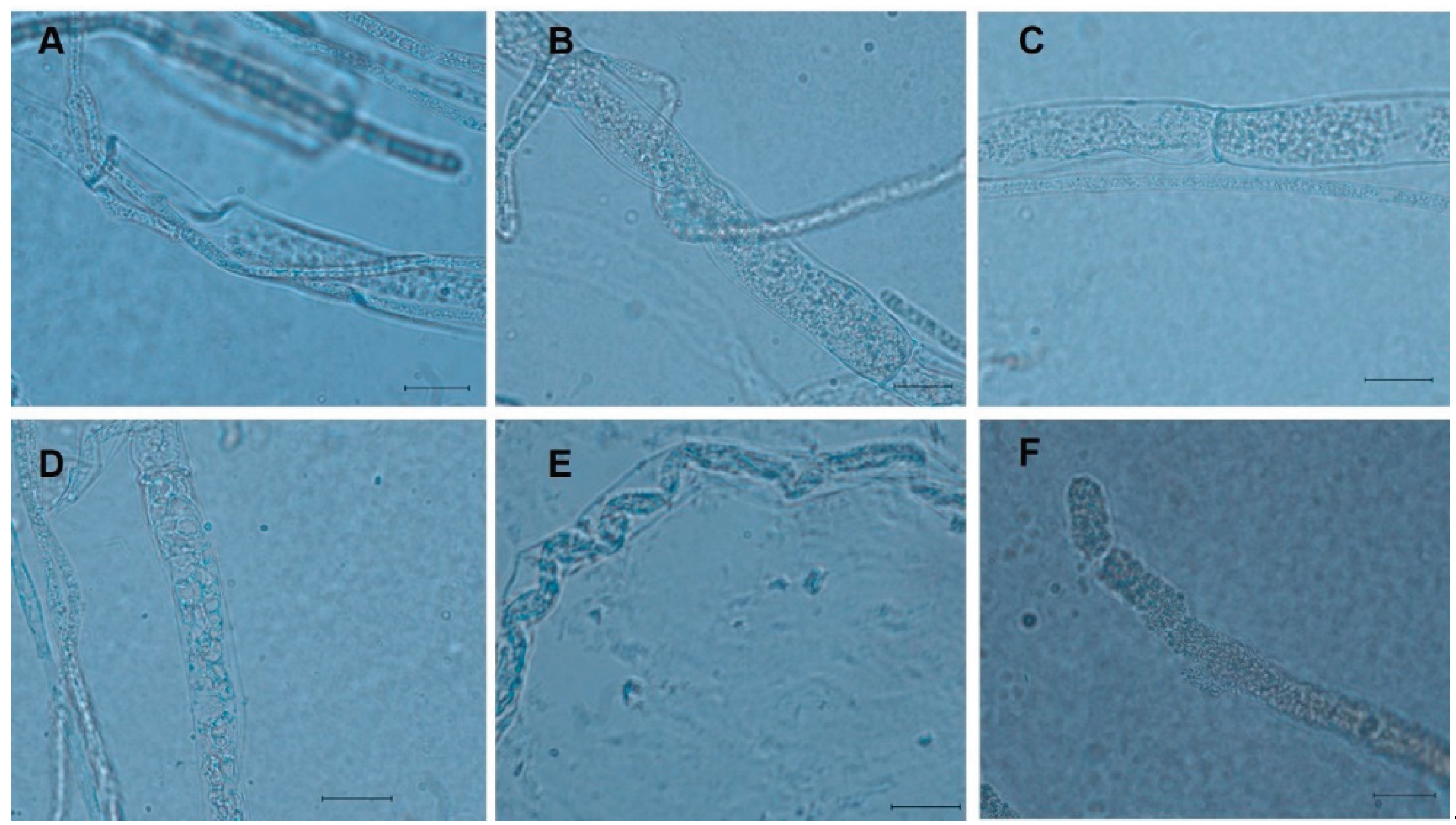
2.3. Hyphal Interactions Between Four Trichoderma spp. and Sclerotinia sclerotiorum
2.4. Efficacy of Trichoderma spp., Synthetic Fungicides and Their Alternate Application for Controlling Potato White Mold Under Field Conditions
2.5. Effectiveness of Four Trichoderma Species, Synthetic Fungicides and Their Alternate Use on Sclerotia Production by Sclerotinia sclerotiorum in Potato Plants Under Field Conditions
2.6. Effectiveness of Four Trichoderma spp., Synthetic Fungicides and Their Alternate Use in Controlling White Mold on Potato Tubers Under Field Conditions
2.7. Production of Tubers in Plots Sprayed with a Combination of Four Trichoderma spp., Synthetic Fungicides or the Alternate Use of These Treatments
3. Discussion
4. Materials and Methods
4.1. Obtaining Trichoderma Isolates and Molecular Identification of Sclerotinia sclerotiorum
| Species/ isolate | Locality / georeference | Year of collection | Code in Gen Bank |
|---|---|---|---|
| S. sclerotiorum / SS1 | Ahome, Sinaloa/ 25.819501 -108.955445 | 2021 | PX471991.1 |
| T. asperelloides / TES24 | Caborca, Sonora/31.06666 -112.338333 | 2020 | OR521164 |
| T. azevedoi / TAI73 | Ahome, Sinaloa/25.818885 -108.956014 | 2021 | OR521181 |
| T. afroharzianum / TAF75 | Ahome, Sinaloa/25.491445 -108.571659 | 2021 | OR521183 |
| T. asperellum / TAM74 | Ahome, Sinaloa/ 25.491445 -108.571659 | 2021 | OR521182 |
4.2. In Vitro Antagonism of Trichoderma spp. Against Sclerotinia sclerotiorum and Sclerotia Formation
4.3. Effect of Volatile Metabolites from Trichoderma spp. on Mycelial Growth and Sclerotia Formation
4.4. Field Experiments
4.5. Disease Incidence, Severity and Sclerotia Production in Plants
4.6. Tuber Disease Evaluation and Yield
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of interest
References
- FAO. 2023. FAOSTAT. Crops. https://www.fao.org/faostat/en/#data/QCL.
- SIAP. Anuario Estadístico de la Producción Agrícola. 2024. https://nube.siap.gob.mx/cierre_agricola/.
- Johnson, D.A.; Dung, J.K.S. Verticillium wilt of potato-the pathogen, disease and management. Can. J. Plant Pathol. 2010, 32, 58–67. [CrossRef]
- Atkinson, D.; Thornton, M.K.; Miller, J.S. Development of Rhizoctonia solani on stems, stolons and tubers of potato II. efficacy of chemical applications. Am. J. Potato Res. 2011, 88, 96–103. [CrossRef]
- Roca, L.F.; Raya, M.C.; Luque, F.; Agustí-Brisach, C.; Romero, J.; Trapero, A. First Report of Sclerotium rolfsii causing soft rot of potato tubers in Spain. Plant Dis. 2016, 100, 2535. [CrossRef]
- Azil, N.; Stefanczyk, E.; Sobkowiak, S.; Chihat, S.; Boureghda, H.; Sliwka, J. Identification and pathogenicity of Fusarium spp. associated with tuber dry rot and wilt of potato in Algeria. Eur. J. Plant Pathol. 2021, 159, 495–509. [CrossRef]
- Zhong, L.; Li, L.; Zheng, Y.; Zhou, Y.; Zeng, Y.; Zhu, W.; Chen, F. First report of black dot caused by Colletotrichum coccodes on potato in the Tibet autonomous region of China. Plant Dis. 2022, 106, 27–46. [CrossRef]
- Felix-Gastelum, R.; Herrera-Rodriguez, G.; Avila-Alistac, N.; Leon, E. Sclerotinia sclerotiorum in beans and potatoes in Sinaloa: Etiology, epidemiology and management options. Mex. J. Plant Pathol. 2024, 42, 29. [CrossRef]
- Boland, G.J.; Hall, R. Index of plant hosts of Sclerotinia sclerotiorum. Can. J. Plant Pathol. 1994, 16, 93–108. [CrossRef]
- Islam, M.R.; Akanda, A.M.; Hossain, M.M.; Hossain, M.M. First characterization of a newly emerging phytopathogen, Sclerotinia sclerotiorum causing white mold in pea. J. Basic Microbiol. 2021, 61, 923–939. [CrossRef]
- Jahan, R.; Siddique, S.S.; Jannat, R.; Hossain, M.M. Cosmos white rot: First characterization, physiology, host range, disease resistance and chemical control. J. Basic Microbiol. 2022, 62, 911–929. [CrossRef]
- Tian, B.; Xie, J.; Fu, Y.; Cheng, J.; Li, B.; Chen, T.; Zhao, Y.; Gao, Z.; Yang, P.; Barbetti, M.J.; et al. A cosmopolitan fungal pathogen of dicots adopts an endophytic lifestyle on cereal crops and protects them from major fungal diseases. ISME J. 2020, 14, 3120–3135. [CrossRef]
- Bolton, M.D.; Thomma, B.P.H.J.; Nelson, B.D. Sclerotinia sclerotiorum (Lib.) de Bary: biology and molecular traits of a cosmopolitan pathogen. Mol. Plant Pathol. 2006, 7, 1–16. [CrossRef]
- Cebreros-Sanchez, F.; Sánchez-Castro, M.A. Enfermedades de la berenjena. In Enfermedades de las Hortalizas; Ortega, J.C., García-Estrada, R., Carrillo-Facio, A., Eds.; Universidad Autónoma de Sinaloa: Sinaloa, Mexico, 1998; pp. 161–175.
- Rodriguez-Cota, F.G.; Cortez-Mondaca, E.; Sauceda-Acosta, R.H.; Acosta-Gallegos, J.A.; Padilla-Valenzuela, I.; Miranda-Arnold, P. Technology for producing bean seeds in Sinaloa. Tech. Broch. 2022, 46, 49.
- Hooker, W.J. Compendium of Potato Diseases. American Phytopathological Society: St. Paul, MN, USA, 1981.
- Clarkson, J.P.; Staveley, J.; Phelps, K.; Young, C.S.; Whipps, J.M. Ascospore release and survival in Sclerotinia sclerotiorum. Mycol. Res. 2003, 107, 213–222. [CrossRef]
- Fernando, W.G.D.; Nakkeeran, S.; Zhang, Y. Ecofriendly methods in combating Sclerotinia sclerotiorum (Lib.) de Bary. Recent Res. Dev. Environ. Biol. 2004, 1, 237–251. [CrossRef]
- Hegedus, D.D.; Rimmer, S.R. Sclerotinia sclerotiorum: When 'to be or not to be' a pathogen? FEMS Microbiol. Lett. 2005, 251, 177–184. [CrossRef]
- Bolton, M.D.; Thomma, B.P.H.J.; Nelson, B.D. Sclerotinia sclerotiorum (Lib.) de Bary: biology and molecular traits of a cosmopolitan pathogen. Mol. Plant Pathol. 2006, 7, 1–16. [CrossRef]
- Guilger-Casagrande, M.; Germano-Costa, T.; Pasquoto-Stigliani, T.; Fernandes-Fraceto, L.; De Lima, R. Biosynthesis of silver nanoparticles employing Trichoderma harzianum with enzymatic stimulation for the control of Sclerotinia sclerotiorum. Sci. Rep. 2019, 9, 1–9. [CrossRef]
- Lane, D.; Denton-Giles, M.; Derbyshire, M. Abiotic conditions governing the myceliogenic germination of Sclerotinia sclerotiorum allowing the basal infection of Brassica napus. Australas. Plant Pathol. 2019, 48, 85–91. [CrossRef]
- Matheron, M.E.; Porchas, M. Activity of boscalid, fenhexamid, fluazinam, fludioxonil and vinclozolin on growth of Sclerotinia minor and S. sclerotiorum and development of lettuce drop. Plant Dis. 2004, 88, 665–668. [CrossRef]
- Bradley, C.A.; Lamey, H.A.; Endres, G.J.; Henson, R.A.; Hanson, B.K.; McKay, K.R.; et al. Efficacy of fungicides for control of Sclerotinia stem rot of canola. Plant Dis. 2006, 90, 1129–1134. [CrossRef]
- Wang, Y.; Duan, Y.B.; Zhou, M.G. Molecular and biochemical characterization of boscalid resistance in laboratory mutants of Sclerotinia sclerotiorum. Plant Pathol. 2015, 64, 101–108. [CrossRef]
- Hideki-Sumida, C.; Canteri, M.G.; Peitl, D.C.; Tibolla, F.; Orsini, I.P.; Araújo, F.A.; et al. Chemical and biological control of Sclerotinia stem rot in the soybean crop. Cienc. Rural 2015, 45, 760–766. [CrossRef]
- Nieto-Lopez, E.H.; Justo-Miorini, T.J.; Wulkop-Gil, C.A.; Chilvers, M.I.; Giesler, L.J.; Jackson-Ziems, T.A.; et al. Fungicide sensitivity of Sclerotinia sclerotiorum from US soybean and drybean, compared to different regions and climates. Plant Dis. 2023, 107, 2395–2406. [CrossRef]
- Piscitelli-Cavalcanti, V.; Fiusa-Araújo, N.A.; Bernardes-Machado, N.; Pedroso-Costa Júnior, P.S.; Pasqual, M.; Alves, E.; et al. Yeasts and Bacillus spp. as potential biocontrol agents of Sclerotinia sclerotiorum in garlic. Sci. Hortic. 2020, 261, 108931. [CrossRef]
- Ghanem, G.A.M.; Gebily, D.A.S.; Ragab, M.M.; Ali, A.M.; Soliman, N.E.K.; El-Moity, T.H.A. Efficacy of antifungal substances of three Streptomyces spp. against different plant pathogenic fungi. Egypt. J. Biol. Pest Control 2022, 32, 112. [CrossRef]
- Albert, D.; Zboralski, A.; Ciotola, M.; Cadieux, M.; Biessy, A.; Blom, J.; et al. Identification and genomic characterization of Pseudomonas spp. displaying biocontrol activity against Sclerotinia sclerotiorum in lettuce. Front. Microbiol. 2024, 15, 1304682. [CrossRef]
- Conrad, A.M.; Johnson, W.G.; Cruz, C.D.; Telenko, D.E.P. Integration of Sclerotinia sclerotiorum-Targeted Biofungicides Coniothyrium minitans and Bacillus amyloliquefaciens into Season-Long Soybean Pest Management Practices in Indiana. PhytoFrontiers 2023, 3, 525–534. [CrossRef]
- Baruah, P.; Tewari, A.K.; Tripathi, R.; Purohit, R. Unraveling the antagonistic potential of Trichoderma for combating Sclerotinia rot of mustard. J. Basic Microbiol. 2025, 65, 70040. [CrossRef]
- Da Silva, L.R.; Muniz, P.H.P.C.; Peixoto, G.H.S.; Luccas, B.E.G.D.; da Silva, J.B.T.; de Mello, S.C.M. Mycelial inhibition of Sclerotinia sclerotiorum by Trichoderma spp. volatile organic compounds in distinct stages of development. Pak. J. Biol. Sci. 2021, 24, 527–536. [CrossRef]
- Silva, L.G.; Camargo, R.C.; Mascarin, G.M.; Nunes, P.S.O.; Dunlap, C.; Bettiol, W. Dual functionality of Trichoderma: Biocontrol of Sclerotinia sclerotiorum and biostimulant of cotton plants. Front. Plant Sci. 2022, 13, 983127. [CrossRef]
- Smolińska, U.; Kowalska, B.; Kowalczyk, W.; Szczech, M.; Murgrabia, A. Eradication of Sclerotinia sclerotiorum sclerotia from soil using organic waste materials as Trichoderma fungi carriers. J. Hortic. Res. 2016, 24, 101–110. [CrossRef]
- Townsend, G.R.; Heuberger, J.W. Methods for estimating losses caused by disease in fungicide experiments. Plant Dis. Rep.1943, 27, 340–343.
- Kurt, S.; Uysal, A.; Kara, M.; Soylu, S. Natural infection of potato by Sclerotinia sclerotiorum causing stem rot disease in Turkey. Australas. Plant Dis. 2017, 12, 3. [CrossRef]
- Baturo-Cieśniewska, A.; Frąc, M.; Jędryczka, M. Molecular identification of Sclerotinia trifoliorum and Sclerotinia sclerotiorum (Ascomycota) as causal agents of white mould of red clover and alfalfa. Plant Dis. 2017, 101, 336–343. [CrossRef]
- Sharma, P.; Mehta, S.; Kaur, A.; Singh, G.; Khatri, I.; Bhardwaj, S.C.; Bhardwaj, V. Genetic diversity studies based on morphological and ITS sequence analysis of Sclerotinia sclerotiorum isolates from oilseed Brassica. Front. Microbiol. 2018, 9, 1169. doi.org/10.3389/fmicb.2018.01169.
- Nain, M.Z.; Khan, R.U.; Singh, V.P.; Sayyeda, S. In vitro evaluation of antifungal activity of different Trichoderma spp. and plant extracts against Sclerotinia sclerotiorum (Lib.) de Bary causing stem rot of mustard. Int. J. Plant Soil Sci. 2023, 35, 40–47. [CrossRef]
- Li, E.; Zhu, N.; Zhang, S.; Xu, B.; Liu, L.; Zhang, A. Efficacy of Trichoderma longibrachiatum SC5 fermentation filtrate in inhibiting the Sclerotinia sclerotiorum growth and development in sunflower. Int. J. Mol. Sci. 2024, 26, 201. [CrossRef]
- Ibarra-Medina, V.A.; Ferrera-Cerrato, R.; Alarcón, A.; Lara-Hernández, M.E.; Valdez-Carrasco, J.M. Isolation and screening of Trichoderma strains antagonistic to Sclerotinia sclerotiorum and Sclerotinia minor. Rev. Mex. Micol. 2010, 31, 53–63. https://www.scielo.org.mx/scielo.php?pid=S0187-31802010000100008&script=sci_abstract&tlng=en.
- Hernandez-Castillo, F.D.; Berlanga-Padilla, A.M.; Gallegos-Morales, G.; Cepeda-Siller, M.; Rodriguez-Herrera, R.; Aguilar-Gonzales, C.N.; et al. In vitro antagonist action of Trichoderma strains against Sclerotinia sclerotiorum and Sclerotium cepivorum. Am. J. Agric. Biol. Sci. 2011, 6, 410–417. [CrossRef]
- Atlagić, K.; Cvetić-Antić, T.; Lukičić, J.; Kruščić, K.; Živić, M.; Unković, N.; et al. Biocontrol potential of native Trichoderma strains toward soil-borne phytopathogenic and saprotrophic fungi. J. Fungi 2025, 11, 535. [CrossRef]
- Sridharan, A.; Thankappan, S.; Karthikeyan, G.; Sivakumar, U. Comprehensive profiling of the VOCs of Trichoderma longibrachiatum EF5 while interacting with Sclerotium rolfsii and Macrophomina phaseolina. Microbiol. Res. 2020, 236, 126436. [CrossRef]
- Iqbal-Faruk, M. Integrated Management of Sclerotinia Sclerotiorum, An Emerging Fungal Pathogen Causing White Mold Disease. J. Plant Cell Dev. 2022, 1, 1–14. [CrossRef]
- Zeng, W.; Wang, D.; Kirk, W.; Hao, J. Use of Coniothyrium minitans and other microorganisms for reducing Sclerotinia sclerotiorum. Biol. Control 2012, 60, 225–232. [CrossRef]
- Geraldine, A.M.; Lopes, F.A.C.; Carvalho, D.D.C.; Barbosa, E.T.; Rodrigues, A.R.; Brandão, R.S.; et al. Cell wall-degrading enzymes and parasitism of sclerotia are key factors on field biocontrol of white mold by Trichoderma spp. Biol. Control 2013, 67, 308–316. [CrossRef]
- Zeng, W.; Kirk, W.; Hao, J. Field management of Sclerotinia stem rot of soybean using biological control agents. Biol. Control 2012, 60, 141–147. [CrossRef]
- Troian, R.F.; Steindorff, A.S.; Ramada, M.H.S.; Arruda, W.; Ulhoa, C.J. Mycoparasitism studies of Trichoderma harzianum against Sclerotinia sclerotiorum: evaluation of antagonism and expression of cell wall-degrading enzymes genes. Biotechnol. Lett. 2014, 36, 2095–2101. [CrossRef]
- Rubayet, M.T.; Bhuiyan, M.K.A.; Hossain, M.M. Effect of soil solarization and biofumigation on stem rot disease of potato caused by Sclerotium rolfsii. Ann. Bangladesh Agric. 2017, 21, 49–59. https://gau.edu.bd/aba/wp-content/uploads/sites/320/2019/07/Manuscript-5.pdf.
- García-Crespo, R.G.; Arcia-Montesuma, M.A.; Pérez-Tortolero, M.R.; Riera-Tona, R.F. Effect of Trichoderma on potato development and Rhizoctonia biocontrol under three application start times. Trop. Agron. 2012, 62, 77–95. https://ve.scielo.org/scielo.php?script=sci_arttext&pid=S0002-192X2012000100007&lng=es&nrm=iso&tlng=es.
- Huzar-Novakowiski, J.; Paul, P. A.; Dorrance, A. E. Host resistance and chemical control for management of Sclerotinia stem rot of soybean in Ohio. Phytopathology 2017, 107, 937-949. [CrossRef]
- Altomare, C.; Norvell, W.A.; Bjorkman, T.; Harman, G.E. Solubilization of Phosphates and Micronutrients by the Plant-Growth-Promoting and Biocontrol Fungus Trichoderma harzianum Rifai 1295-22. Appl. Environ. Microbiol. 1999,65, 2926–2933. [CrossRef]
- Zhang, F.; Ge, H.; Zhang, F.; Guo, N.; Wang, Y.; Chen, L.; et al. Biocontrol potential of Trichoderma harzianum isolate T-aloe against Sclerotinia sclerotiorum in soybean. Plant Physiol. Biochem. 2016, 100, 64–74. [CrossRef]
- Tyskiewicz, R.; Nowak, A.; Ozimek, E.; Jaroszuk-Ściseł, J. Trichoderma: The current status of its application in agriculture for the biocontrol of fungal phytopathogens and plant growth stimulation. Int. J. Mol. Sci. 2022, 23, 2329. [CrossRef]
- Irazoqui-Acosta, M.B.; Felix-Gastelum, R.; Leyva-Madrigal, K.Y.; Longoria-Espinoza, R.M.; Herrera-Rodriguez, G.; Armenta-López, S.E. Caracterización de Sclerotium rolfsii y especies de Trichoderma en cultivos comerciales de papa en Sonora y Sinaloa, México. Rev. Mex. Fitopatol. 2025, 43, 85. [CrossRef]
- Sanger, F.; Nicklen, S.; Coulson, A.R. DNA sequencing with chain-terminating inhibitors. Proc. Natl. Acad. Sci. 1977, 74, 5463–5467. https://doi:10.1073/pnas.74.12.5463.
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: New York, NY, USA, 1990; pp. 315–322. https://www.researchgate.net/publication/262687766.
- Hall, TA. BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symposium. 1999. 4, 95-98. https://www.scirp.org/reference/ReferencesPapers?ReferenceID=1383440.
- Edgar, R.C. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797.
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549.
- Tamura, K. Estimation of the number of nucleotide substitutions when there are strong transition-transversion and G+C-content biases. Mol. Biol. Evol. 1992, 9, 678–687. https://doi: 10.1093/oxfordjournals.molbev.a040752. PMID: 1630306.
- Rambaut A. FigTree v. 1.4: tree figure drawing tool. 2014. http://tree.bio.ed.ac.uk/software/figtree.
- Vincent, J.M. Distortion of fungal hyphae in the presence of certain inhibitors. Nature 1947, 159, 850. [CrossRef]
- Bell, D.K.; Wells, H.D.; Markham, C.R. In vitro antagonism of Trichoderma spp. against six fungal plant pathogens. Phytopathology 1982, 72, 379–382. [CrossRef]
- Dennis, C.; Webster, J. Antagonistic properties of species-groups of Trichoderma, III Hyphal interactions. Trans. Br. Mycol. Soc. 1971, 57, 363–369. [CrossRef]
- Santos-Cervantes, M.E.; Felix-Gastelum, R.; Herrera-Rodríguez, G.; Espinoza-Mancillas, M.G.; Mora-Romero, A.G.; Leyva-López, N.E. Characterization, pathogenicity and chemical control of Streptomyces acidiscabies associated to potato common scab. Am. J. Potato Res. 2016, 94, 14–25. [CrossRef]
- Little, T.M.; Hills, F.J. Agricultural Experimentation. Design and Analysis; CABI: Wallingford, UK, 1978; p. 350.
| Dual confrontation | Volatile metabolites | ||||
|---|---|---|---|---|---|
| Trichoderma isolate | % inhibition | Scale | Number of sclerotia | % inhibition | Number of sclerotia |
| T. asperellum | 63.1 a * | 3 | 6.8 b | 93.5 a | 0.0 b |
| T. afroharzianum | 62.8 ab | 3 | 4.3 c | 94.1 a | 0.0 b |
| T. asperelloides | 61.8 b | 3 | 4.0 c | 91.0 a | 0.0 b |
| T. azevedoi | 60.1 c | 3 | 4.0 c | 90.3 a | 0.0 b |
| Control | NA | NA | 20.8 a | NA | 27.0 a |
| CV (%) | 1.4 | 10.9 | 2.7 | 3.9 | |
| Types of hyphal interactionsz | ||||||
|---|---|---|---|---|---|---|
| Trichoderma species | Adhesion | Coiling | Granulation | Vacuolization | Penetration | Lysis |
| T. asperellum | X | X | X | X | X | X |
| T. afroharzianum | X | X | X | X | X | |
| T. asperelloides | X | X | X | X | ||
| T. azevedoi | X | X | X | X | X | |
| Treatment | Experiment 2021 | Experiment 2022 | ||
|---|---|---|---|---|
| Incidence (%) | Severity (%)x | Incidence (%) | Severity (%) |
|
| Trichoderma spp.y | 66.0 b* | 27.1 b | 55.6 b | 18.8 b |
| Trichoderma spp. alternating with synthetic fungicidesz | 81.1 ab | 35.6 ab | 62.2 b | 27.0 b |
| Synthetic fungicides | 93.3 a | 61.7 a | 75.6 a | 33.8 a |
| CV (%) | 15.7 | 16.1 | 15.6 | 21.8 |
| Treatment | Sclerotia on 10 plants | |
|---|---|---|
| Experiment 2021 | Experiment 2022 | |
| Trichoderma spp.y | 32.7 b* | 14.6 b |
| Trichoderma spp. alternating with synthetic fungicides z | 40.8 b | 31.2 b |
| Synthetic fungicides | 167.7 a | 93.8 a |
| CV (%) | 36.2 | 43.1 |
| Treatment | Experiment 2021 | Experiment 2022 | ||
|---|---|---|---|---|
| Incidence (%) | Severity (%) x |
Incidence (%) |
Severity (%) x |
|
| Trichoderma spp.y | 1.6 b* | 0.4 b | 1.3 b | 0.3 b |
| Trichoderma spp . alternating with synthetic fungicides z | 4.5 ab | 1.0 ab | 3.3 ab | 0.7 ab |
| Synthetic fungicides | 7.4 a | 1.5 a | 6.2 a | 1.3 a |
| CV (%) | 62.2 | 43.1 | 57.0 | 50.9 |
| Treatment | Experiment 2021 | Experiment 2022 |
|---|---|---|
| t ha -1 | t ha -1 | |
| Trichoderma spp.y | 46.0 a* | 52.9 a |
| Trichoderma + Synthetic Fungicides | 44.1 ab | 48.3 ab |
| Synthetic fungicidesz | 42.4 b | 44.5 b |
| CV (%) | 4.06 | 12.87 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




