Submitted:
04 November 2025
Posted:
05 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
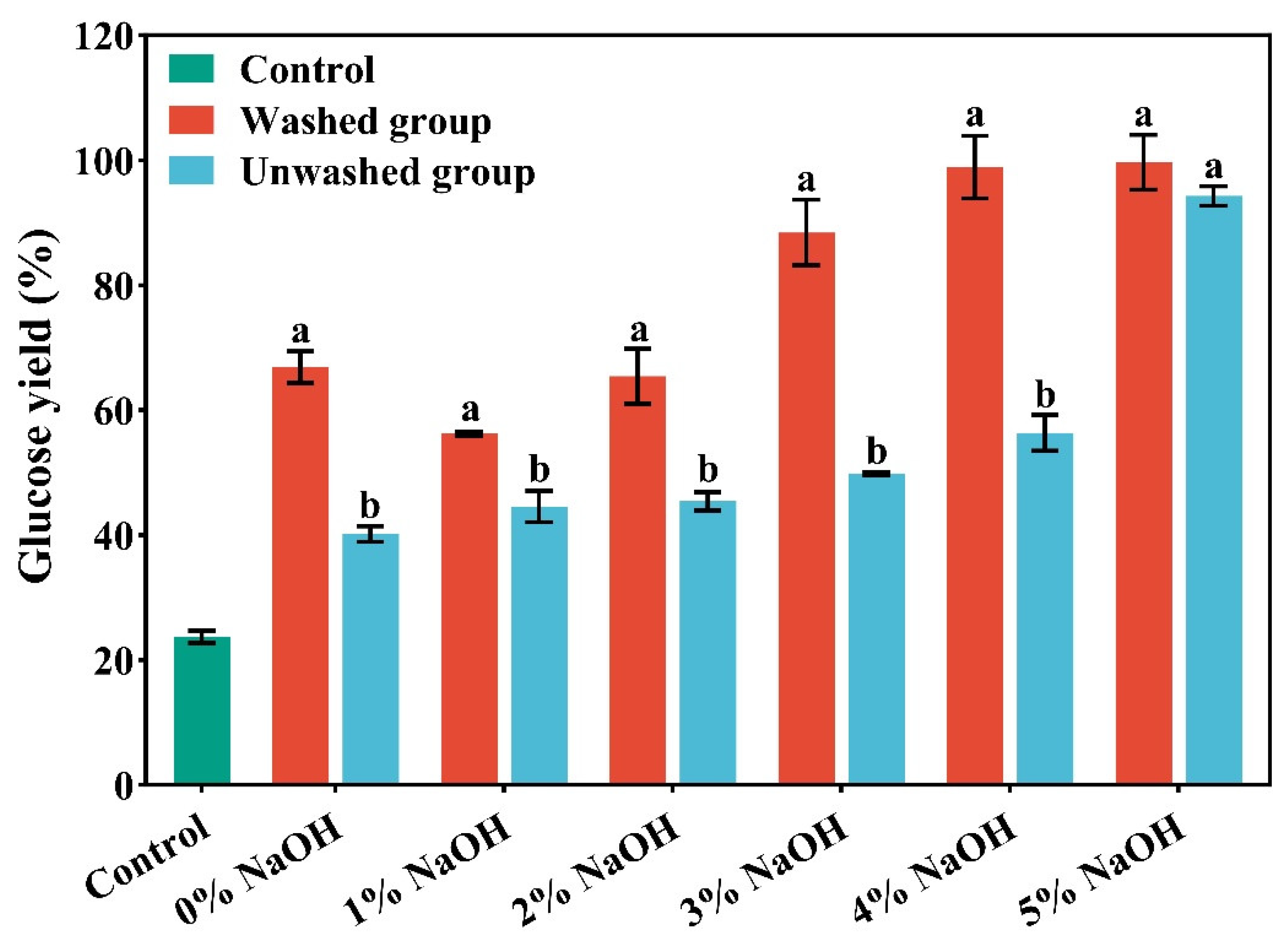
2.1. The Effect of the Concentration of NaOH on the Components and the Enzymatic Hydrolysis Performance of Corn Stover Pretreated by Low-Alkali Assisted ICSE
2.2. Washed vs Unwashed at 4% NaOH Assisted ICSE: Evidence for Lignin-Derived Water-Soluble Anti-Enzymatic Factors
2.3. Role of Alkali-Soluble Lignin as a Water-Insoluble Anti-Enzymatic Factor
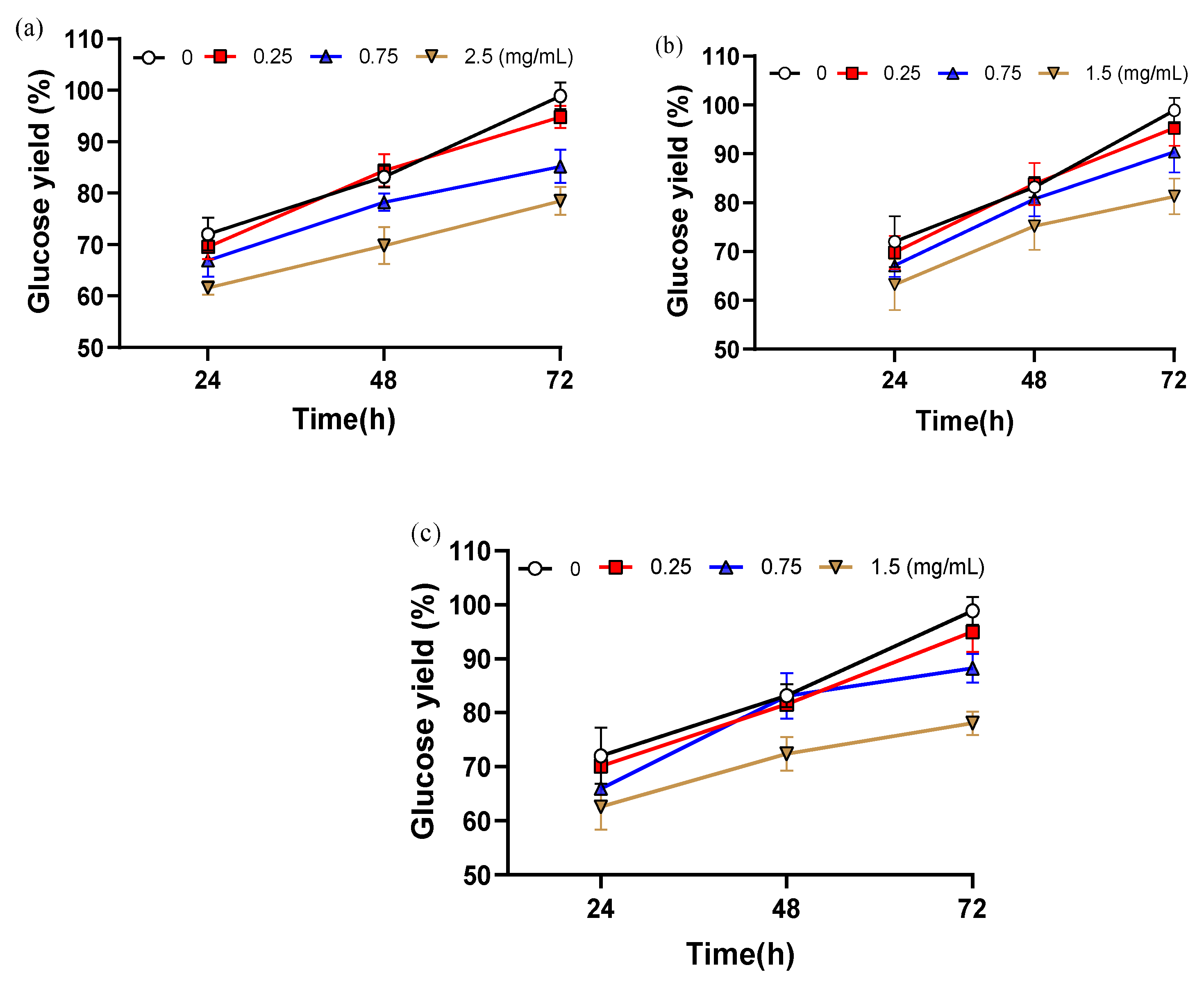
2.4. Impact of Water-Soluble Lignin on Enzymatic Hydrolysis Performance Under Varying Alkali Severities
2.5. Mechanism Analysis of Improving Enzymatic Hydrolysis Efficiency by Low-Alkali Assisted ICSE
2.5.1. Influence of Water-Soluble Lignin on Enzymatic Hydrolysis Efficiency
2.5.2. Analysis of Surface Morphology
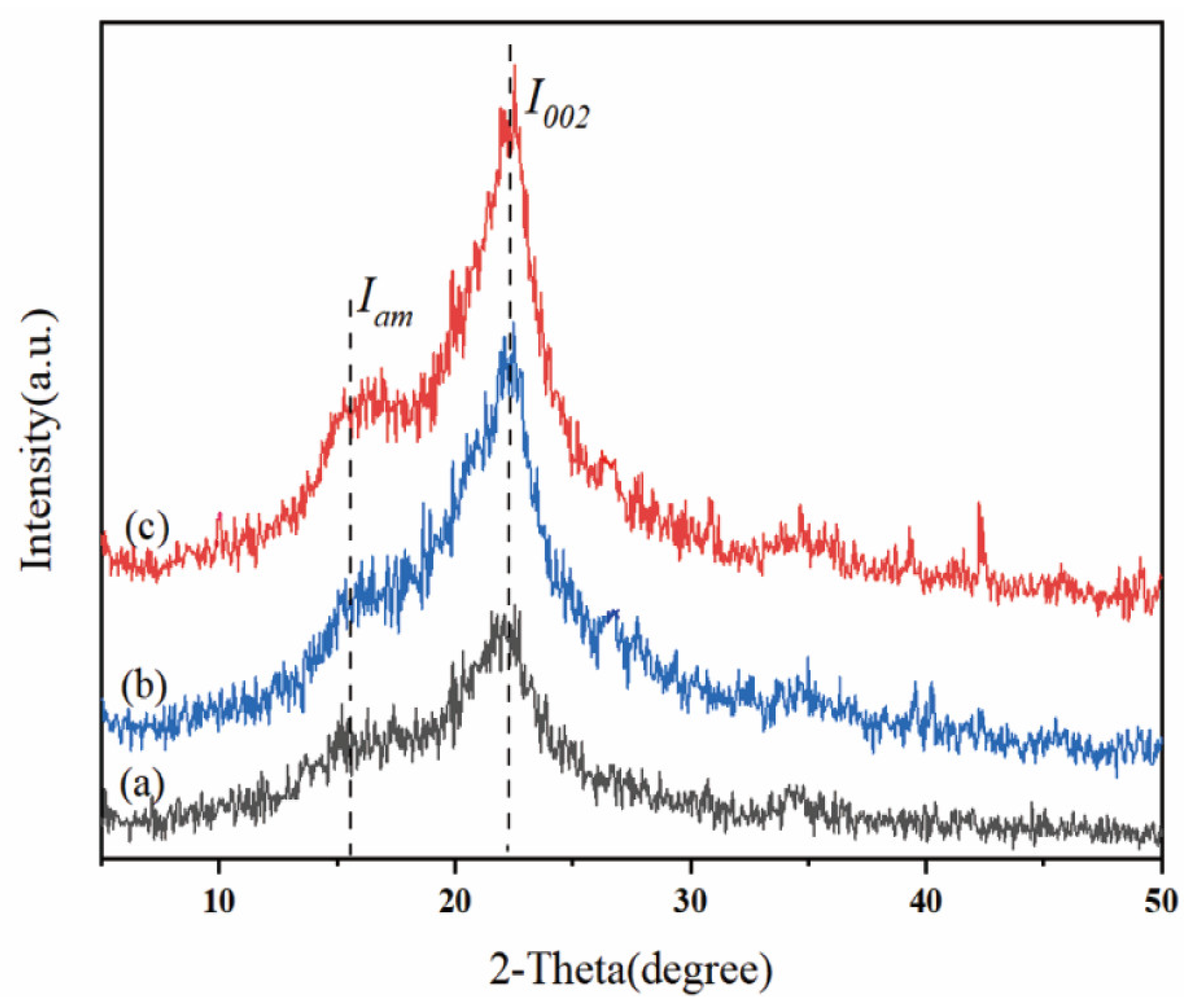
2.5.3. Analysis of X-Ray Diffraction
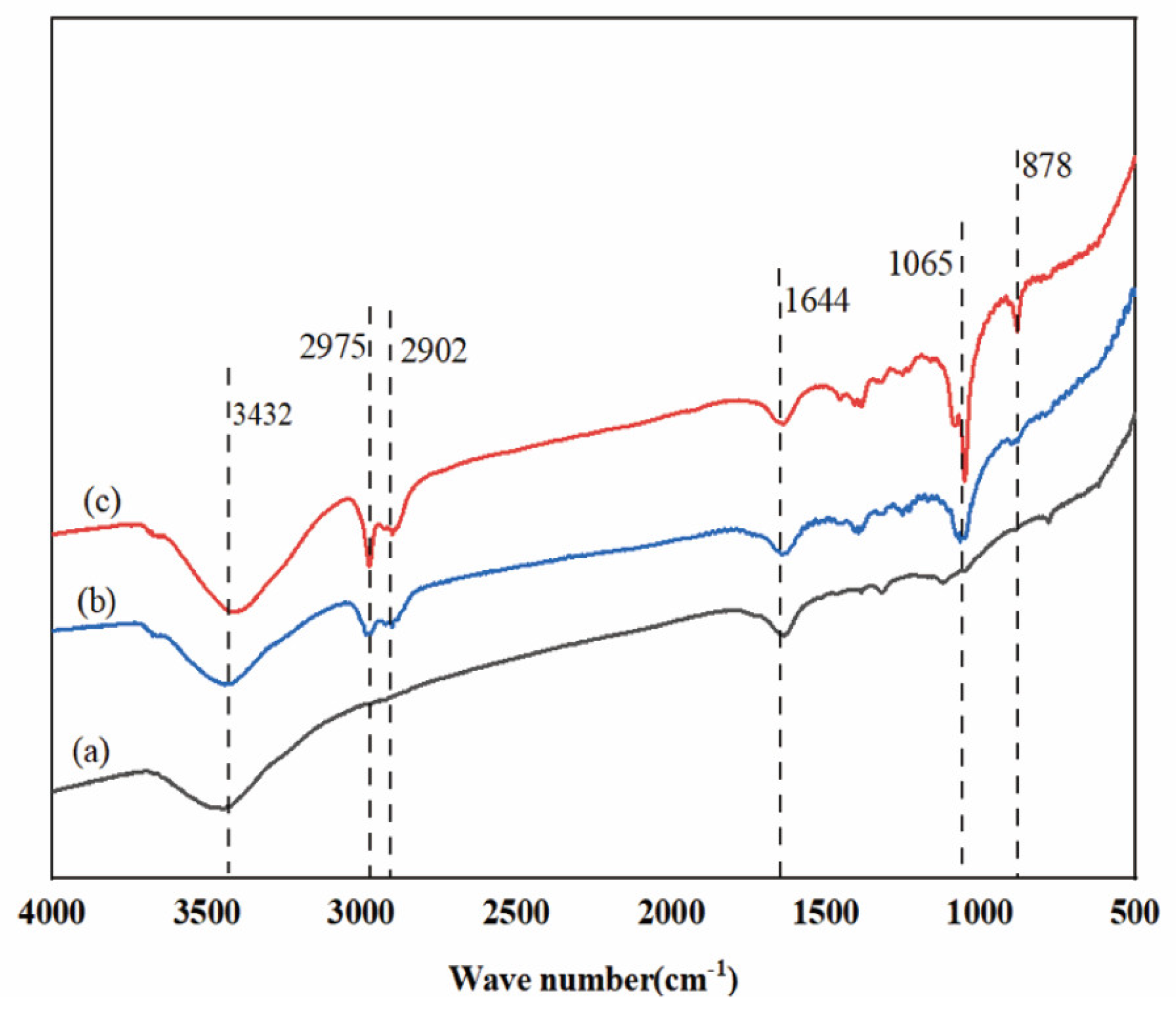
2.5.4. Analysis of Infrared Spectroscopy
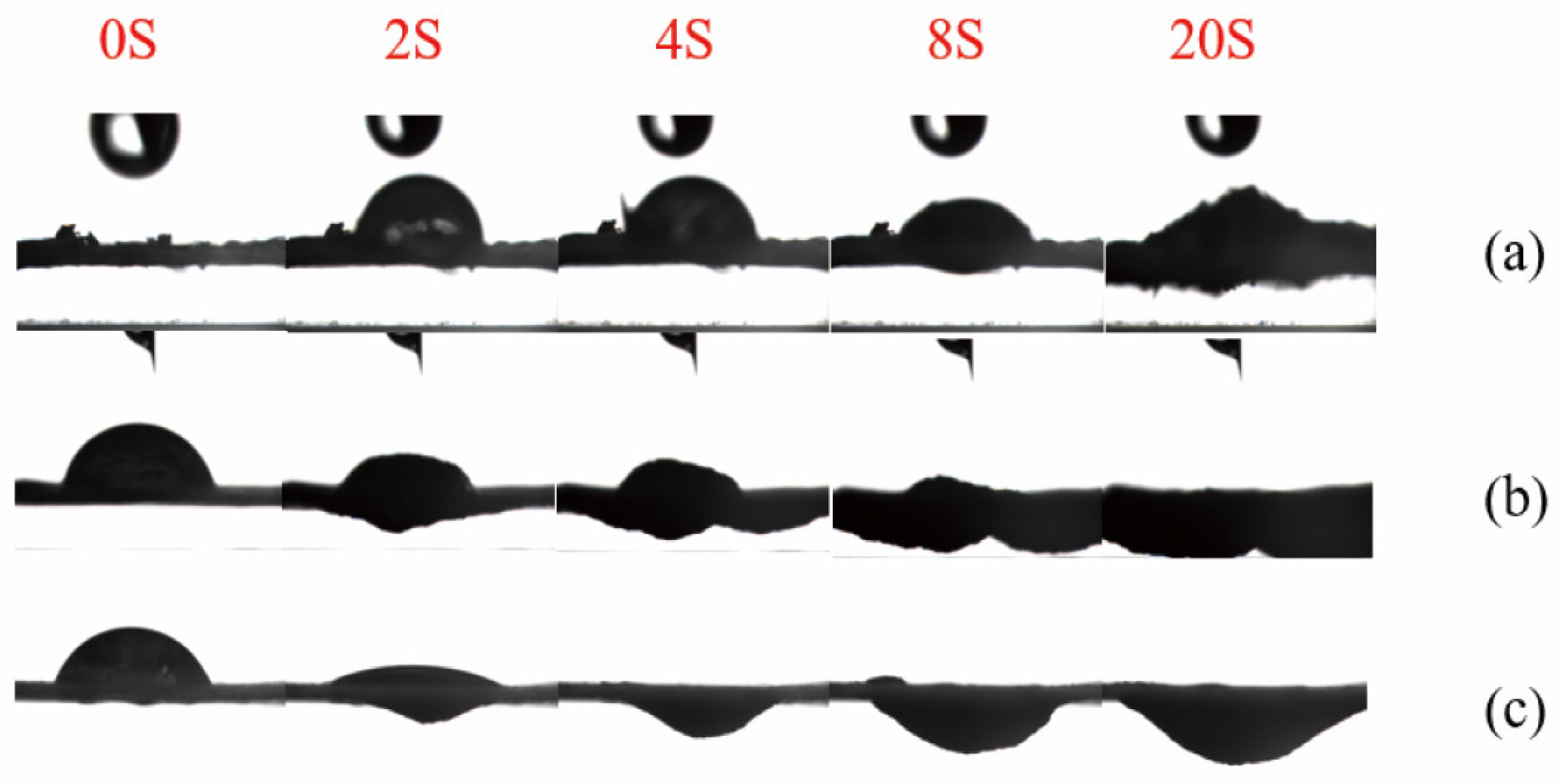
2.5.5. Surface Hydrophilicity and Interfacial Behavior by Contact Angle
3. Materials and Methods
3.1. Materials
3.2. Pretreatment of Corn Stover
3.3. Determination Methods of Components in Lignocellulosic Raw Materials
3.4. Enzymatic Hydrolysis Conditions and Determination Method of Glucose
3.5. Extraction and Analysis Methods of Water-Soluble Anti-Enzymatic Hydrolysis Factors
3.6. Quantitative Analysis Methods for Vanillin, 2-Carboxybenzaldehyde, and Syringaldehyde in Corn Stover Hydrolysate
3.7. Extraction Method of Alkali-Soluble Lignin Solid
3.8. Anti-Enzymatic Hydrolysis Factor Backfill Test Method
3.8.1. Lignin-Derived Water-Soluble Inhibitors Backfill Test Method
- (1)
- Vanillin: 0.25 mg/mL, 0.75 mg/mL, 2.5 mg/mL;
- (2)
- Syringaldehyde: 0.25 mg/mL, 0.75 mg/mL, 1.5 mg/mL;
- (3)
- 2-Carboxybenzaldehyde: 0.25 mg/mL, 0.75 mg/mL, 1.5 mg/mL.
3.8.2. Alkali-Soluble Lignin Solid Backfill Test Method
3.9. Determination Method of Water-Soluble Lignin
3.10. Analysis Method of Scanning Electron Microscope (SEM)
3.11. Analysis Method of X-Ray Diffraction (XRD)
3.12. Analysis Method of Infrared Spectroscopy
3.13. Analysis Method of Contact Angle
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Raghavi S, Sindhu R, Binod P, Gnansounou E, Pandey A. Development of a novel sequential pretreatment strategy for the production of bioethanol from sugarcane trash. Bioresour. Technol. 2016, 199, 202-210. [CrossRef]
- Zhu JY, Wang GS, Pan XJ, Gleisner R. Specific surface to evaluate the efficiencies of milling and pretreatment of wood for enzymatic saccharification. Chem. Eng. Sci. 2008, 64(3), 474-485. [CrossRef]
- Sun Y, Cheng JY. Hydrolysis of lignocellulosic materials for ethanol production: a review. Bioresour. Technol. 2002, 83(1), 1-11. [CrossRef]
- Himmel EM, Ding S, Johnson KD, Adney SW, Nimlos RM, Brady WJ. Biomass Recalcitrance: Engineering Plants and Enzymes for Biofuels Production. Science 2007, 315(5813), 804-807. [CrossRef]
- Jönsson JL, Martín C. Pretreatment of lignocellulose: Formation of inhibitory by-products and strategies for minimizing their effects. Bioresour. Technol. 2016, 199, 103-112. [CrossRef]
- Liu C, Xiao Y, Xia X, Zhao X, Peng LC, Srinophakun P, Bai F. Cellulosic ethanol production: Progress, challenges and strategies for solutions. Biotechnol. Adv. 2019, 37(3), 491-504. [CrossRef]
- Liu W, Wu R, Hu Y, Ren Q, Hou Q, Ni Y. Improving enzymatic hydrolysis of mechanically refined poplar branches with assistance of hydrothermal and Fenton pretreatment. Bioresour. Technol. 2020, 316, 123920. [CrossRef]
- Chang VS, Holtzapple MT. Fundamental factors affecting biomass enzymatic reactivity. Appl. Biochem. Biotechnol. 2000, 84-86, 35-37. [CrossRef]
- Wu Q, Zhang Y, Zhao L, Wei Z, Song C, Pang C, Pang X. Strategies for efficient degradation of lignocellulose from straw: Synergistic effects of acid-base pretreatment and functional microbial agents in composting. Chem. Eng. J. 2025, 508, 161048. [CrossRef]
- Dong LF, Gao YH, Jing XL, Guo HP, Zhang HS, Lai Q, Diao QY. Pretreatments of Broussonetia papyrifera: In vitro assessment on gas and methane production, fermentation characteristic, and methanogenic archaea profile. Asian-Australas. J. Anim. Sci. 2020, 35(9). [CrossRef]
- Chen A, Zhang B, Bao J. Adaptive evolution of Paecilomyces variotii enhanced the biodetoxification of high-titer inhibitors in pretreated lignocellulosic feedstock. Bioresour. Technol. 2024, 411, 131351. [CrossRef]
- Yu ZD, Zhang BL,Yu FQ, Xu GZ, Song AD. A real explosion: The requirement of steam explosion pretreatment. Bioresour. Technol. 2012, 121, 335-341. [CrossRef]
- Wang Q, Wang W, Tan X, Zahoor, Chen XY, Guo Y, Yuan ZH, Zhuang XS Low-temperature sodium hydroxide pretreatment for ethanol production from sugarcane bagasse without washing process. Bioresour. Technol. 2019, 291, 121844. [CrossRef]
- Vinuthana VH, Govindaraj O, Subramaniam S, Gnanachitra M, Uthandi S. Harnessing lignocellulosic biomass: Insights into pre-treatment strategies and hydrolytic enzyme production. Ind. Crops. Prod. 2025, 229, 120986. [CrossRef]
- Alvira P, Tomás-Pejó E, Ballesteros M, Negro MJ. Pretreatment technologies for an efficient bioethanol production process based on enzymatic hydrolysis: A review. Bioresour. Technol. 2009, 101(13), 4851-4861. [CrossRef]
- Durot N, Gaudard F, Kurek B. The unmasking of lignin structures in wheat straw by alkali. Phytochemistry 2003, 63(5), 617-623. [CrossRef]
- Yang H, Yoo GC, Meng X, Pu YQ, Muchero W, Tuskan GA, Tschaplinski TJ, Ragauskas AJ, Yao L. Structural changes of lignins in natural Populus variants during different pretreatments. Bioresour. Technol. 2020, 295, 122240. [CrossRef]
- Liu C, Liu L, Zi L, Zhao X, Xu Y, Bai F. Assessment and regression analysis on instant catapult steam explosion pretreatment of corn stover. Bioresour. Technol. 2014, 166, 368-372. [CrossRef]
- Kumar V, Yadav KS, Kumar J, Ahluwalia V. A critical review on current strategies and trends employed for removal of inhibitors and toxic materials generated during biomass pretreatment. Bioresour. Technol. 2020, 299, 122633. [CrossRef]
- Bhatia KS, Jagtap SS, Bedekar, AA, Bhatia RK, Patel AK, Pant D, Banu J, Rao CV, Kim Y, Yang Y. Recent developments in pretreatment technologies on lignocellulosic biomass: Effect of key parameters, technological improvements, and challenges. Bioresour. Technol. 2020, 300, 122724. [CrossRef]
- Saini KJ, Patel KA, Adsul M, Singhania RR. Cellulase adsorption on lignin: A roadblock for economic hydrolysis of biomass. Renew. Energy 2016, 98, 29-42. [CrossRef]
- Wang Z, Jönsson JL. Comparison of catalytically non-productive adsorption of fungal proteins to lignins and pseudo-lignin using isobaric mass tagging. Bioresour. Technol.2018, 268, 393-401. [CrossRef]
- Li X, Zheng Y. Lignin-enzyme interaction: Mechanism, mitigation approach, modeling, and research prospects. Biotechnol. Adv. 2017, 35(4), 466-489. [CrossRef]
- Jin Y, Chen H, Wu W, Wei W. Investigations of the effect of water-soluble lignin on enzymatic hydrolysis of lignocellulose. J. For. Eng. 2020, 5(04), 12-19. [CrossRef]
- Xie JX, Cheng Z, Zhu SY, Wang B. Phosphotungstic acid assisted with neutral deep eutectic solvent boost corn straw pretreatment for enzymatic saccharification and lignin extraction. Ind. Crops. Prod. 2021, 172, 114058. [CrossRef]
- Qi LT, Liu JK, Peng JM, Yang GH, Li FF, Xue Y, Chen JC. The Dual Effect of Ionic Liquid Pretreatment on the Eucalyptus Kraft Pulp during Oxygen Delignification Process. Polymers 2021, 13(10), 1600. [CrossRef]
- Zhao Z, Chen XC, Ali MF, Abdeltawab AA, Yakout SM, Yu GR. Pretreatment of wheat straw using basic ethanolamine-based deep eutectic solvents for improving enzymatic hydrolysis. Bioresour. Technol. 2018, 263, 325-333. [CrossRef]
- Sluiter A, Hames B, Ruiz R, Scarlata C, Sluiter J, Templeton D, Crocker D. Determination of Structural Carbohydrates and Lignin in Biomass. Technical Report NREL/TP-510-42618. Golden: National Renewable Energy Laboratory, 2012.
- Zhang H, Liu G, Zhang J, Bao J. Fermentative production of high titer gluconic and xylonic acids from corn stover feedstock by Gluconobacter oxydans and techno-economic analysis. Bioresour. Technol. 2016, 219, 123-131. [CrossRef]
- Chen SF, Mowery RA, Scarlata CJ, Chambliss CK. Compositional analysis of water-soluble materials in corn stover. J. Agric. Food Chem. 2007, 55(15), 5912-5918. [CrossRef]
- Jakab BEV, Savoy A, Saulnier KB, Singh SP, Hodge DB. Extraction, recovery, and characterization of lignin from industrial corn stover lignin cake. Bioresour. Technol. 2024, 399, 130610. [CrossRef]
- Chen L, Ren F, Yu XP, Zhang ZP, Xu DJ Tong QY. Pasting investigation, SEM observation and the possible interaction study on rice starch-pullulan combination. Int. J. Biol. Macromol. 2015, 73, 45-48. [CrossRef]
- Vydrina I, Malkov A, Vashukova K, Tyshkunova I, Mayer L, Faleva A, Shestakov S, Novozhilov E, Chukhchin D. A new method for determination of lignocellulose crystallinity from XRD data using NMR calibration. Carbohydr. Polym. Tech. 2023, 5, 100305. [CrossRef]
- Boukir A, Fellak S, Doumenq P. Structural characterization of Argania spinosa Moroccan wooden artifacts during natural degradation progress using infrared spectroscopy (ATR-FTIR) and X-Ray diffraction (XRD). Heliyon 2019, 5(9), e02477. [CrossRef]
- Rbihi S, Aboulouard A, Laallam L, Jouaiti A. Contact Angle Measurements of Cellulose based Thin Film composites: wettability, surface free energy and surface hardness. Surf. Interfaces 2020, 21, 100708. [CrossRef]

| Preprocessing conditions | Cellulose (%) | Hemicellulose (%) | Lignin (%) |
|---|---|---|---|
| Raw corn stover | 34.27 ± 0.60 | 19.41 ± 0.29 | 28.51 ± 0.17 |
| Control (washed) | 37.67 ± 0.13 | 17.77 ± 0.58 | 27.47 ± 0.87 |
| 1% NaOH (washed) | 39.67 ± 0.15 | 15.02 ± 0.15 | 25.70 ± 0.47 |
| 2% NaOH (washed) | 39.89 ± 0.47 | 16.38 ± 0.11 | 24.75 ± 1.70 |
| 3% NaOH (washed) | 40.21 ± 0.05 | 15.47 ± 0.69 | 22.70 ± 0.52 |
| 4% NaOH (washed) | 41.99 ± 0.99 | 15.39 ± 0.29 | 20.69 ± 0.06 |
| 5% NaOH (washed) | 40.42 ± 0.61 | 15.03 ± 0.04 | 20.11 ± 1.17 |
| Control (unwashed) | 35.45 ± 0.60 | 17.03 ± 0.11 | 28.27 ± 1.76 |
| 1% NaOH (unwashed) | 35.64 ± 0.26 | 16.38 ± 0.19 | 26.21 ± 0.91 |
| 2% NaOH (unwashed) | 35.09 ± 0.42 | 16.81 ± 0.06 | 25.91 ± 0.66 |
| 3% NaOH (unwashed) | 35.14 ± 0.72 | 17.46 ± 0.30 | 22.67 ± 0.52 |
| 4% NaOH (unwashed) | 36.11 ± 0.33 | 17.70 ± 0.21 | 22.38 ± 0.63 |
| 5% NaOH (unwashed) | 35.52 ± 2.56 | 17.08 ± 1.73 | 22.18 ± 0.19 |
| Addition amount of Alkali-soluble Lignin solid (g) | Glucose Yield (%) |
|---|---|
| 0 | 98.88±4.97 |
| 0.05 | 93.71±3.75 |
| 0.075 | 91.17±2.88 |
| 0.1 | 89.74±3.14 |
| Pretreatment System | Content of Water-soluble Lignin (mg/g) | Yield of Alkali-soluble Lignin (%) | Glucose Yield (%) |
|---|---|---|---|
| 4% NaOH (unwashed) | 21.14±0.84 | 43.28±0.97 | 56.34±2.85 |
| 5% NaOH (unwashed) | 32.09±0.74 | 38.22±1.62 | 94.26±1.56 |
| Pretreatment system | CrI (%) | Crystalline portion (%) |
|---|---|---|
| Raw corn stover | 33.65 | 58.62 |
| 4% NaOH (unwashed) | 50.00 | 66.70 |
| 5% NaOH (unwashed) | 53.24 | 68.57 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




