Submitted:
17 February 2026
Posted:
06 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
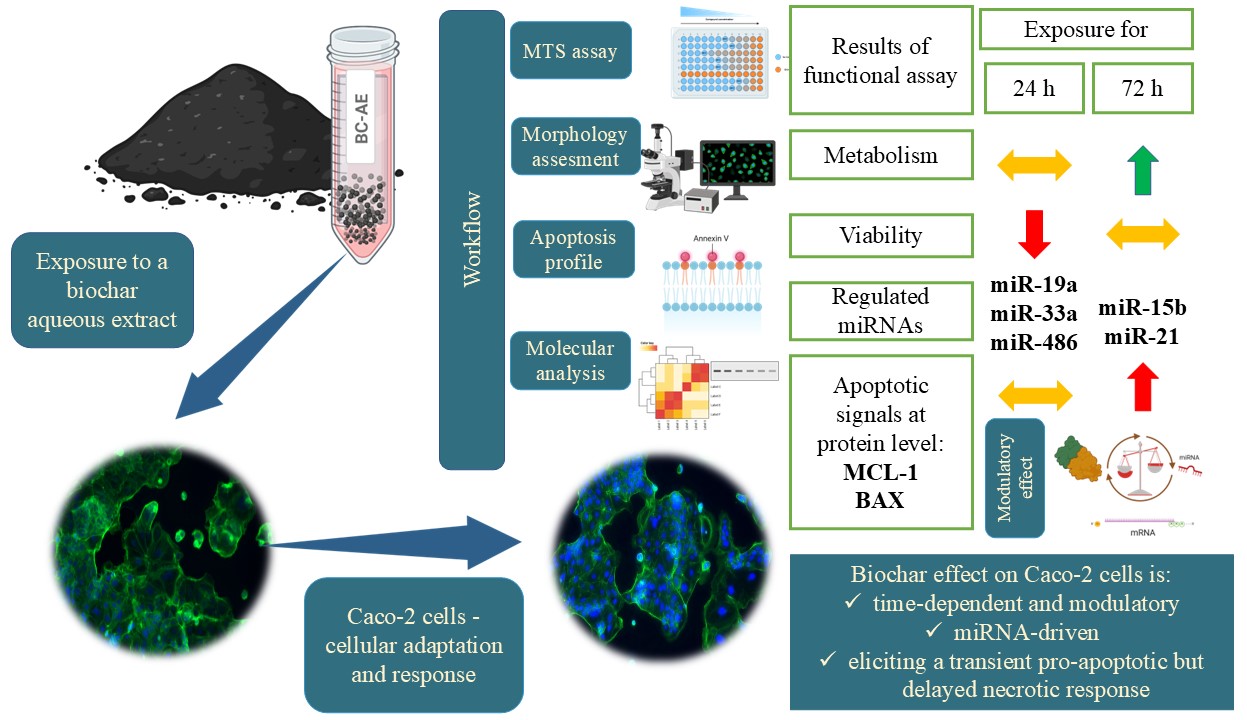
2. Results
2.1. Biochar and BC-AE Characterization
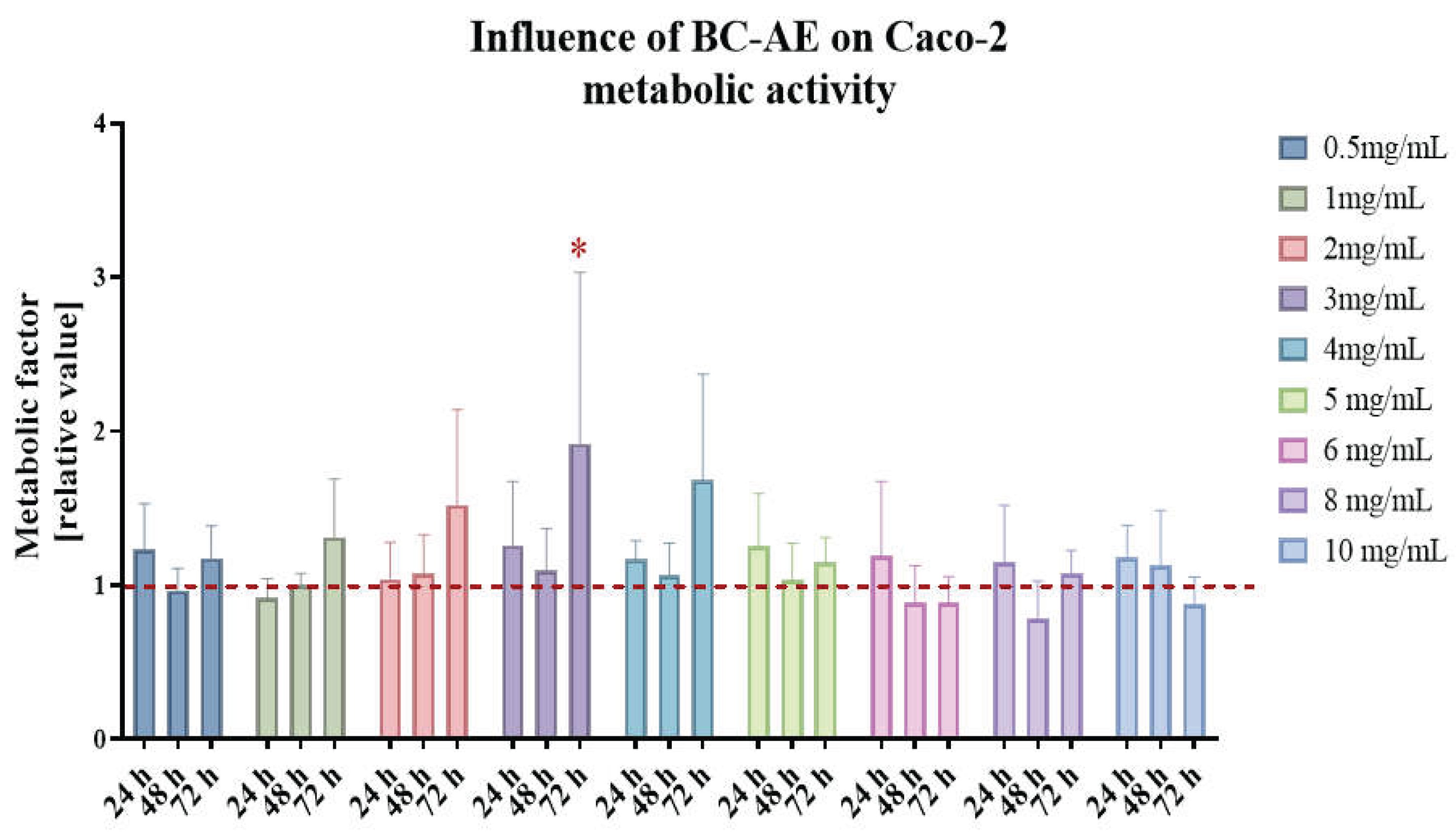
2.2. BC-AE Shows Low Cytotoxicity Toward Caco-2 Cells in a Screening Assay
2.3. Functional Assays – Analysis of Effects of a 3 mg/mL Biochar Aqueous Extract (BC-AE) on Caco-2 Activity
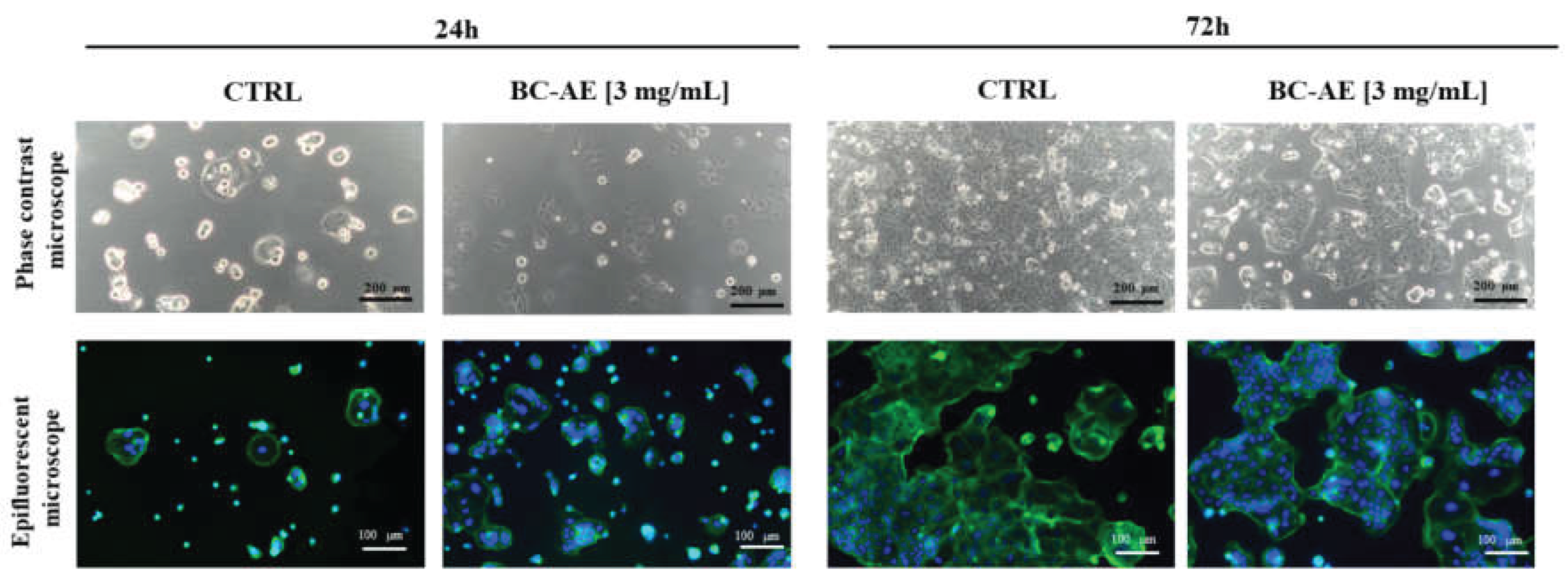
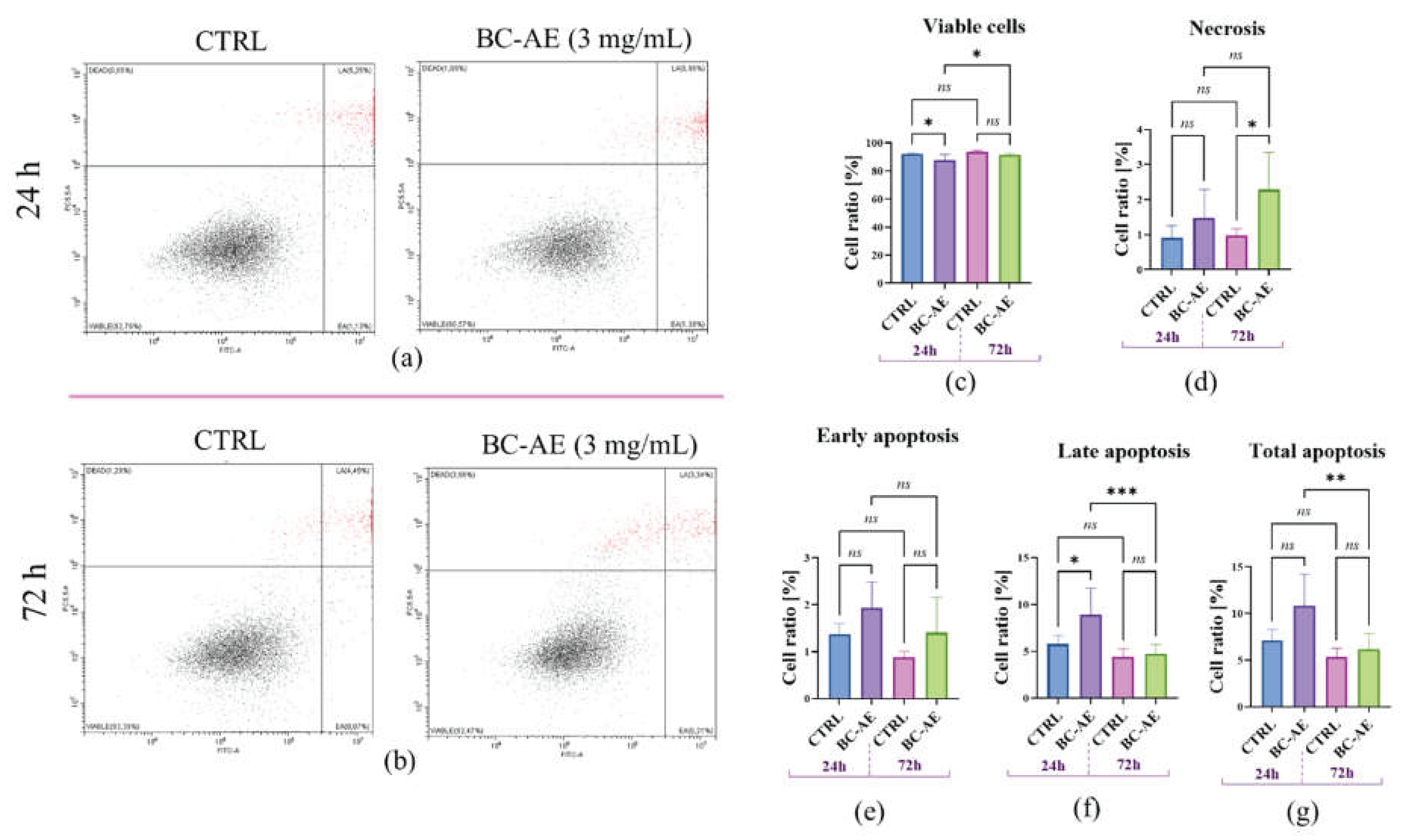
2.4. BC-AE Temporarily Regulates the Dynamics of Apoptosis and Necrosis in Caco-2 cells
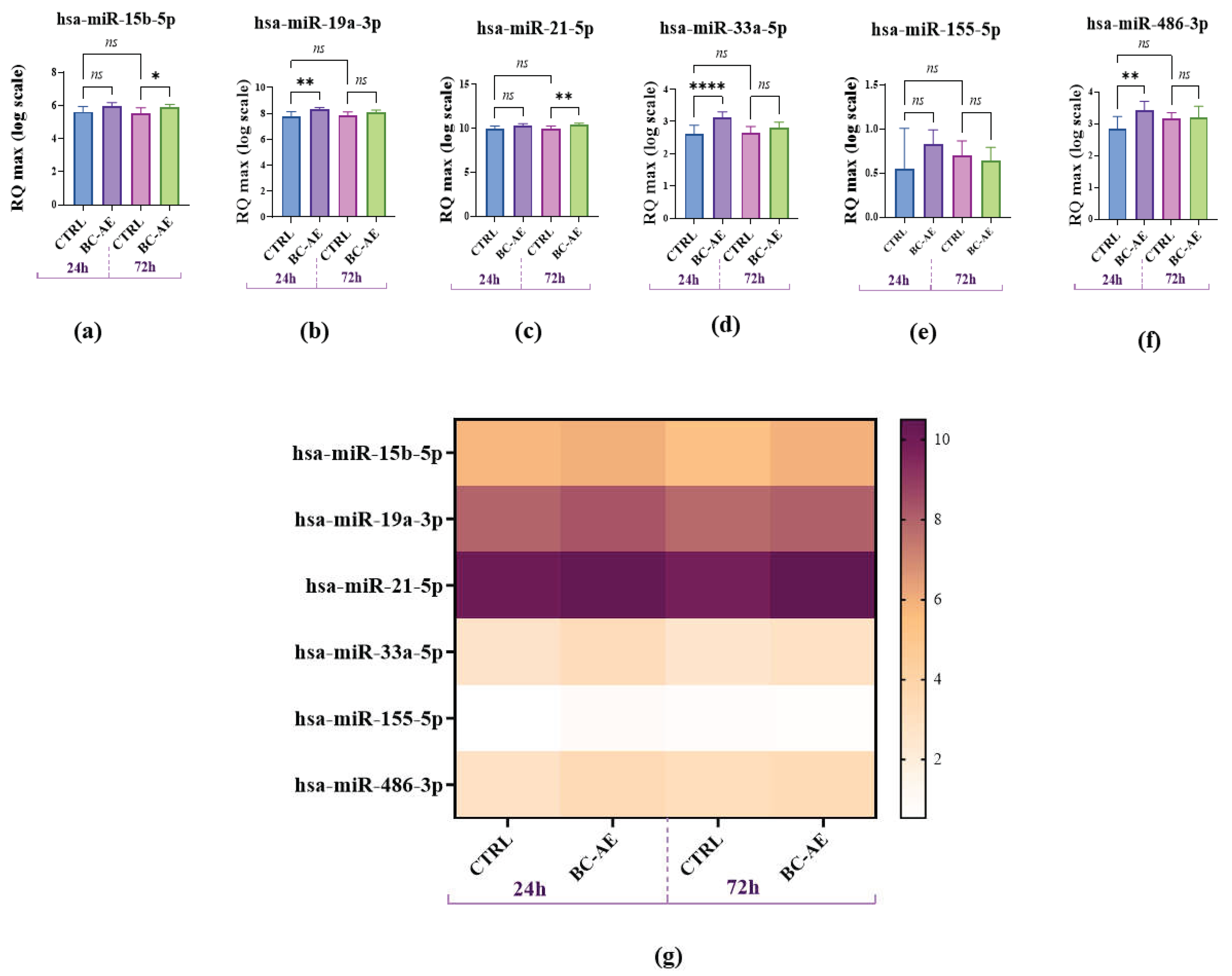
2.5. BC-AE Modulates the Endogenous Levels of survival-Related miRNAs
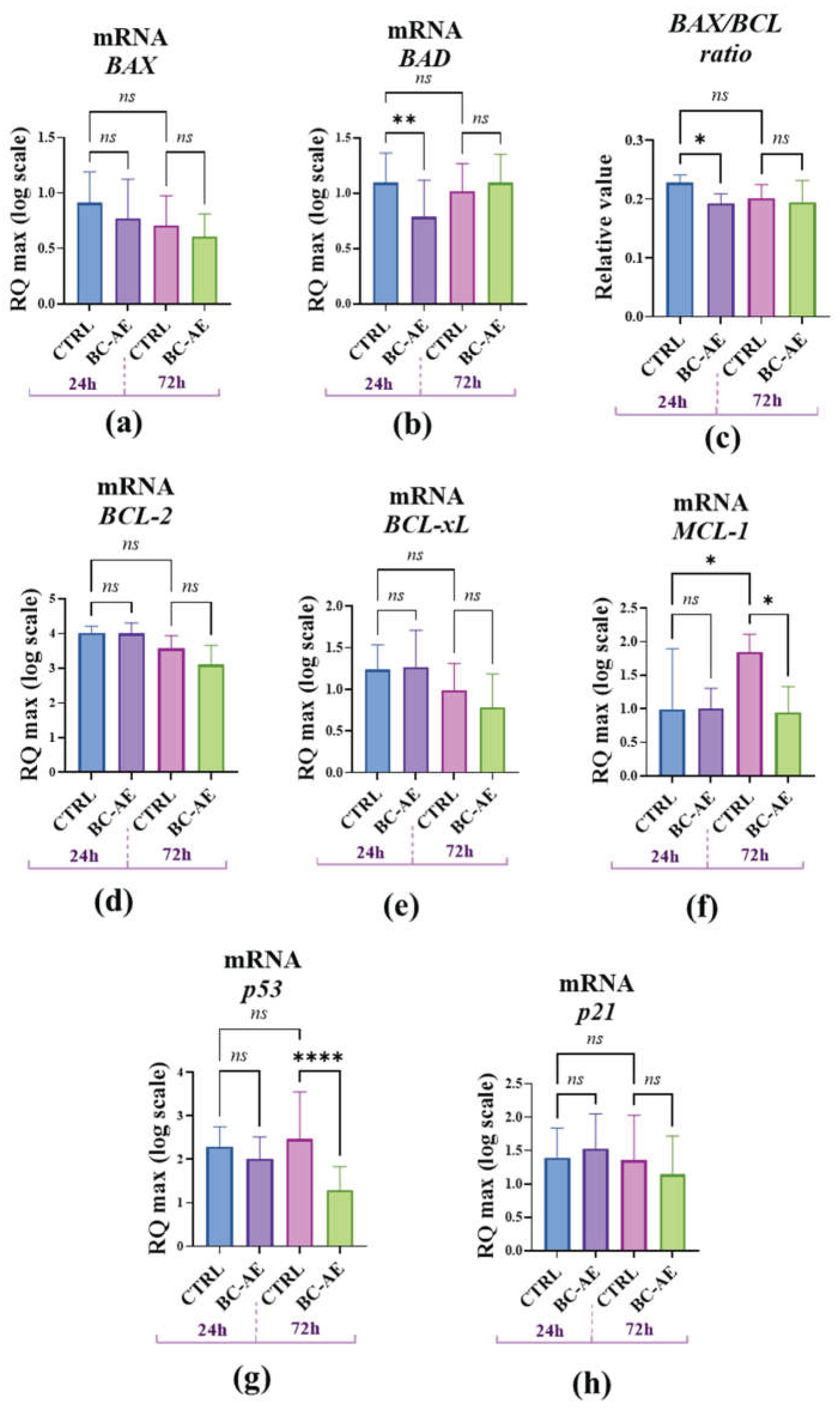
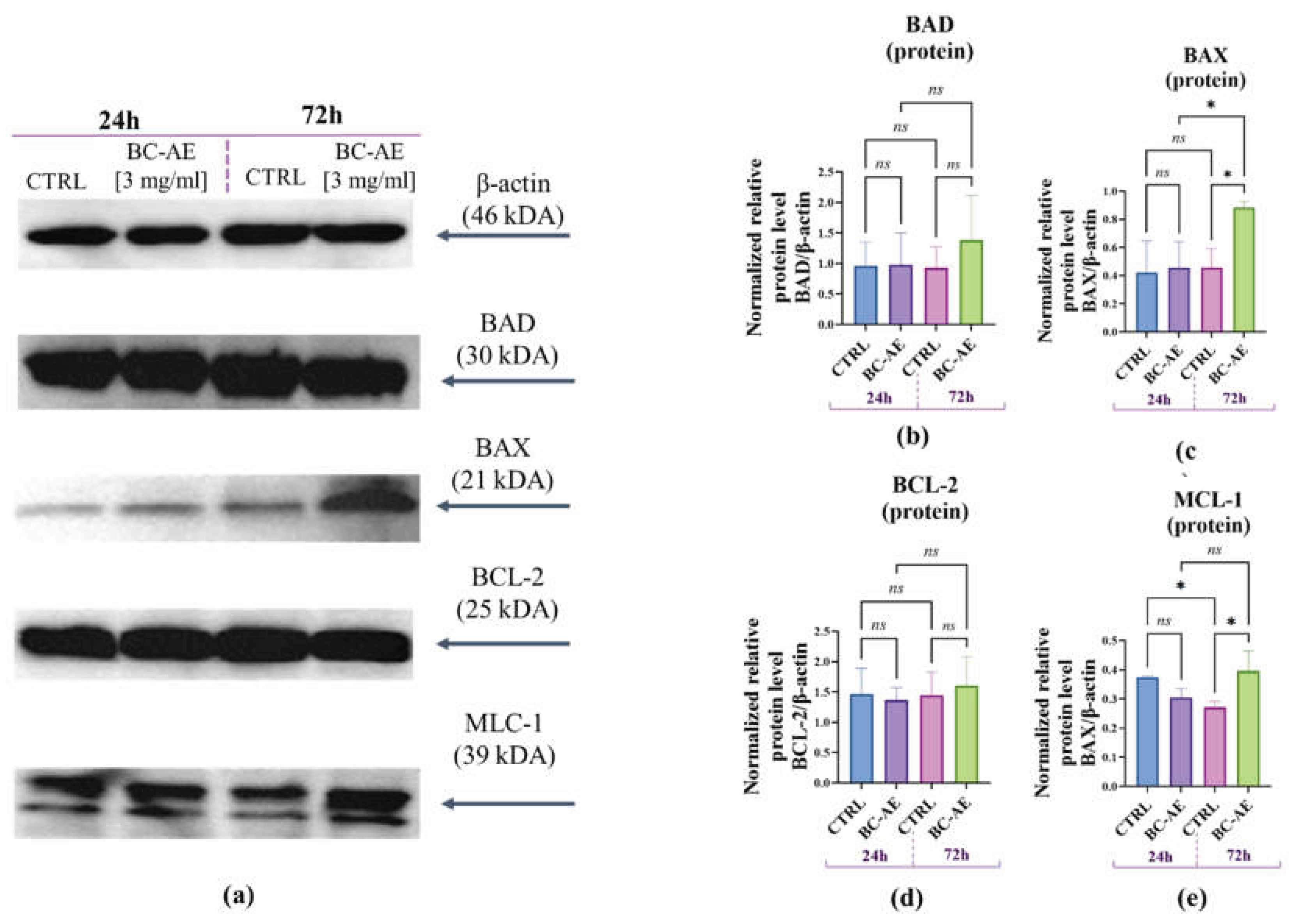
2.6. BC-AE may Trigger Anti-Apoptotic Signals at the mRNA and Protein Levels
3. Discussion
4. Materials and Methods
4.1. Cell Line
4.2. Preparation of Biochar Aqueous Extracts (BC-AE)
4.3. Multielemental Analysis
4.4. Fourier Transform Infrared Analyses
4.5. PAHs Analyses
4.6. In vitro Evaluation of Biochar Activity Using the Caco-2 Cell Model
4.7. Functional In Vitro Assay – In-Depth Evaluation of BC-AE Influence on Caco-2 Viability
4.8. Evaluation of Gene Expression Patterns Modulated by BC-AE
4.9. Evaluation of Cellular Morphology Modulated by BC-AE
4.10. Evaluation of Apoptotic Activity Modulated by BC-AE
4.11. Evaluation of Protein Expression Modulated by BC-AE
4.12. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Zhao, M.; Zhong, S.; Zhou, X.; Yu, Z. Biochar Derived from Animal and Plant Facilitates Synergistic Transformation of Heavy Metals and Phosphorus in Sewage Sludge Composting. Environ Pollut 2024, 357, 124396. [Google Scholar] [CrossRef]
- Schmidt, H.-P.; Hagemann, N.; Draper, K.; Kammann, C. The Use of Biochar in Animal Feeding. PeerJ 2019, 7, e7373. [Google Scholar] [CrossRef]
- Osman, A.I.; Fawzy, S.; Farghali, M.; El-Azazy, M.; Elgarahy, A.M.; Fahim, R.A.; Maksoud, M.I.A.A.; Ajlan, A.A.; Yousry, M.; Saleem, Y.; et al. Biochar for Agronomy, Animal Farming, Anaerobic Digestion, Composting, Water Treatment, Soil Remediation, Construction, Energy Storage, and Carbon Sequestration: A Review. Environ Chem Lett 2022, 20, 2385–2485. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, A.K.; Alkan, N.; Elad, Y.; Sela, N.; Philosoph, A.M.; Graber, E.R.; Frenkel, O. Molecular Insights into Biochar-Mediated Plant Growth Promotion and Systemic Resistance in Tomato against Fusarium Crown and Root Rot Disease. Sci Rep 2020, 10, 13934. [Google Scholar] [CrossRef]
- Yang, Y.; Ahmed, W.; Wang, G.; Ye, C.; Li, S.; Zhao, M.; Zhang, J.; Wang, J.; Salmen, S.H.; Wu, L.; et al. Transcriptome Profiling Reveals the Impact of Various Levels of Biochar Application on the Growth of Flue-Cured Tobacco Plants. BMC Plant Biol 2024, 24, 655. [Google Scholar] [CrossRef]
- Millar, A.A.; Waterhouse, P.M. Plant and Animal microRNAs: Similarities and Differences. Funct Integr Genomics 2005, 5, 129–135. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, T.; Yin, Y.; Zhang, C.-Y.; Zhang, Y.-L. Dietary microRNA-A Novel Functional Component of Food. Adv Nutr 2019, 10, 711–721. [Google Scholar] [CrossRef]
- Lopez-Escalera, S.; Wellejus, A. Evaluation of Caco-2 and Human Intestinal Epithelial Cells as in Vitro Models of Colonic and Small Intestinal Integrity. Biochem Biophys Rep 2022, 31, 101314. [Google Scholar] [CrossRef] [PubMed]
- Ran, R.; Muñoz Briones, J.; Jena, S.; Anderson, N.L.; Olson, M.R.; Green, L.N.; Brubaker, D.K. Detailed Survey of an in Vitro Intestinal Epithelium Model by Single-Cell Transcriptomics. iScience 2024, 27, 109383. [Google Scholar] [CrossRef] [PubMed]
- Lea, T. Caco-2 Cell Line. In The Impact of Food Bioactives on Health: in vitro and ex vivo models; Verhoeckx, K., Cotter, P., López-Expósito, I., Kleiveland, C., Lea, T., Mackie, A., Requena, T., Swiatecka, D., Wichers, H., Eds.; Springer: Cham (CH), 2015 ISBN 978-3-319-15791-7.
- Zhou, X.; Ren, M.; Yang, J.; Pan, H.; Yu, M.; Ji, F. Curcumin Improves Epithelial Barrier Integrity of Caco-2 Monolayers by Inhibiting Endoplasmic Reticulum Stress and Subsequent Apoptosis. Gastroenterol Res Pract 2021, 2021, 5570796. [Google Scholar] [CrossRef]
- Kaldas, M.I.; Walle, U.K.; Walle, T. Resveratrol Transport and Metabolism by Human Intestinal Caco-2 Cells. J Pharm Pharmacol 2003, 55, 307–312. [Google Scholar] [CrossRef]
- Amasheh, M.; Schlichter, S.; Amasheh, S.; Mankertz, J.; Zeitz, M.; Fromm, M.; Schulzke, J.D. Quercetin Enhances Epithelial Barrier Function and Increases Claudin-4 Expression in Caco-2 Cells. J Nutr 2008, 138, 1067–1073. [Google Scholar] [CrossRef]
- Agbu, P.; Carthew, R.W. MicroRNA-Mediated Regulation of Glucose and Lipid Metabolism. Nat Rev Mol Cell Biol 2021, 22, 425–438. [Google Scholar] [CrossRef]
- Yu, B.; Liu, J.; Cai, Z.; Mu, T.; Zhang, D.; Feng, X.; Gu, Y.; Zhang, J. MicroRNA-19a Regulates Milk Fat Metabolism by Targeting SYT1 in Bovine Mammary Epithelial Cells. Int J Biol Macromol 2023, 253, 127096. [Google Scholar] [CrossRef] [PubMed]
- Chodur, G.M.; Steinberg, F.M. Human MicroRNAs Modulated by Diet: A Scoping Review. Adv Nutr 2024, 15, 100241. [Google Scholar] [CrossRef]
- Latack, B.C.; Carvalho, P.H.V.; Moura, G.A.B.; Zinn, R.A. Influence of Biochar Supplementation on Comparative Growth Performance and Carcass Characteristics of Angus × Holstein vs. Charolais × Holstein Calf-Fed Steers. J Anim Sci 2025, 103, skaf113. [Google Scholar] [CrossRef]
- Śmieszek, A.; Giezek, E.; Chrapiec, M.; Murat, M.; Mucha, A.; Michalak, I.; Marycz, K. The Influence of Spirulina Platensis Filtrates on Caco-2 Proliferative Activity and Expression of Apoptosis-Related microRNAs and mRNA. Mar Drugs 2017, 15, 65. [Google Scholar] [CrossRef]
- Wolański, B.; Świechowski, K.; Kierzek, K.; Opaliński, S. Pyrolysis conditions and physico-chemical properties of plant-based biochar = Warunki pyrolizy a właściwości fizyczno-chemiczne biowęgla pochodzenia roślinnego. In Przemysł Chemiczny : miesięcznik poświęcony sprawom polskiego przemysłu chemicznego, wydawany staraniem Instytutu Badań Naukowych i Technicznych Metan we Lwowie; 2024.
- Valentin, M.T.; Siedlecka, A.; Demeshkant, V.; Kosiorowska, K.E.; Świechowski, K.; Wisniewski, M.; Wiercik, P.; Rasaq, W.A.; Białowiec, A. Influence of biochar particle size on biomethane production from glucose. Biomass and Bioenergy 2025, vol. 200, 108016. [Google Scholar] [CrossRef]
- Keiluweit, M.; Nico, P. S.; Johnson, M. G.; Kleber, M. Dynamic molecular structure of plant biomass-derived black carbon (biochar). Environ. Sci. Technol. 2010, 44, 1247–1253. [Google Scholar] [CrossRef]
- Martínez-Gómez, Á.; Andrés, M.F.; Barón-Sola, Á.; Díaz-Manzano, F.E.; Yousef, I.; Mena, I.F.; Díaz, E.; Gómez-Torres, Ó.; González-Coloma, A.; Hernández, L.E.; et al. Biochar from Grape Pomace, a Waste of Vitivinicultural Origin, Is Effective for Root-Knot Nematode Control. Biochar 2023, 5, 30. [Google Scholar] [CrossRef]
- Chomczynski, P.; Sacchi, N. Single-Step Method of RNA Isolation by Acid Guanidinium Thiocyanate-Phenol-Chloroform Extraction. Anal Biochem 1987, 162, 156–159. [Google Scholar] [CrossRef]
- Smieszek, A.; Kornicka, K.; Szłapka-Kosarzewska, J.; Androvic, P.; Valihrach, L.; Langerova, L.; Rohlova, E.; Kubista, M.; Marycz, K. Metformin Increases Proliferative Activity and Viability of Multipotent Stromal Stem Cells Isolated from Adipose Tissue Derived from Horses with Equine Metabolic Syndrome. Cells 2019, 8, 80. [Google Scholar] [CrossRef]
- Smieszek, A.; Marcinkowska, K.; Pielok, A.; Sikora, M.; Valihrach, L.; Carnevale, E.; Marycz, K. Obesity Affects the Proliferative Potential of Equine Endometrial Progenitor Cells and Modulates Their Molecular Phenotype Associated with Mitochondrial Metabolism. Cells 2022, 11, 1437. [Google Scholar] [CrossRef]
- Shoudho, K.N.; Khan, T.H.; Ara, U.R.; Khan, M.R.; Shawon, Z.B.Z.; Hoque, M.E. Biochar in Global Carbon Cycle: Towards Sustainable Development Goals. Current Research in Green and Sustainable Chemistry 2024, 8, 100409. [Google Scholar] [CrossRef]
- Reggi, S.; Frazzini, S.; Fusi, E.; Guagliano, M.; Cristiani, C.; Onelli, E.; Moscatelli, A.; Rossi, L. Biochar’s Adsorption of Escherichia Coli and Probiotics Lactiplantibacillus Plantarum and Limosilactobacillus Reuteri and Its Impact on Bacterial Growth Post In Vitro Digestion. Applied Sciences 2025, 15, 5090. [Google Scholar] [CrossRef]
- Sigmund, G.; Huber, D.; Bucheli, T.D.; Baumann, M.; Borth, N.; Guebitz, G.M.; Hofmann, T. Cytotoxicity of Biochar: A Workplace Safety Concern? Environ. Sci. Technol. Lett. 2017, 4, 362–366. [Google Scholar] [CrossRef]
- Hao, S.; Yang, H.; Hu, J.; Luo, L.; Yuan, Y.; Liu, L. Bioactive Compounds and Biological Functions of Medicinal Plant-Derived Extracellular Vesicles. Pharmacol Res 2024, 200, 107062. [Google Scholar] [CrossRef] [PubMed]
- Ghafouri-Fard, S.; Abak, A.; Shoorei, H.; Mohaqiq, M.; Majidpoor, J.; Sayad, A.; Taheri, M. Regulatory Role of microRNAs on PTEN Signaling. Biomed Pharmacother 2021, 133, 110986. [Google Scholar] [CrossRef]
- Etzi, F.; Griñán-Lisón, C.; Fenu, G.; González-Titos, A.; Pisano, A.; Farace, C.; Sabalic, A.; Picon-Ruiz, M.; Marchal, J.A.; Madeddu, R. The Role of miR-486-5p on CSCs Phenotypes in Colorectal Cancer. Cancers (Basel) 2024, 16, 4237. [Google Scholar] [CrossRef]
- Cimmino, A.; Calin, G.A.; Fabbri, M.; Iorio, M.V.; Ferracin, M.; Shimizu, M.; Wojcik, S.E.; Aqeilan, R.I.; Zupo, S.; Dono, M.; et al. miR-15 and miR-16 Induce Apoptosis by Targeting BCL2. Proc Natl Acad Sci U S A 2005, 102, 13944–13949. [Google Scholar] [CrossRef]
- Hashemi, M.; Mirdamadi, M.S.A.; Talebi, Y.; Khaniabad, N.; Banaei, G.; Daneii, P.; Gholami, S.; Ghorbani, A.; Tavakolpournegari, A.; Farsani, Z.M.; et al. Pre-Clinical and Clinical Importance of miR-21 in Human Cancers: Tumorigenesis, Therapy Response, Delivery Approaches and Targeting Agents. Pharmacol Res 2023, 187, 106568. [Google Scholar] [CrossRef]
- Yang, Y.; Hou, N.; Wang, X.; Wang, L.; Chang, S.; He, K.; Zhao, Z.; Zhao, X.; Song, T.; Huang, C. miR-15b-5p Induces Endoplasmic Reticulum Stress and Apoptosis in Human Hepatocellular Carcinoma, Both in Vitro and in Vivo, by Suppressing Rab1A. Oncotarget 2015, 6, 16227–16238. [Google Scholar] [CrossRef]
- Liu, J.; Xu, H.; Wang, N.; Sun, M. miR-15b, a Diagnostic Biomarker and Therapeutic Target, Inhibits Oesophageal Cancer Progression by Regulating the PI3K/AKT Signalling Pathway. Exp Ther Med 2020, 20, 222. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Leong, M.H.; Harms, B.; Kennedy, G.; Chen, L. MicroRNA-21 as a Potential Colon and Rectal Cancer Biomarker. World J Gastroenterol 2013, 19, 5615–5621. [Google Scholar] [CrossRef] [PubMed]
- Kunze-Schumacher, H.; Krueger, A. The Role of MicroRNAs in Development and Function of Regulatory T Cells – Lessons for a Better Understanding of MicroRNA Biology. Front Immunol 2020, 11, 2185. [Google Scholar] [CrossRef] [PubMed]
- Gavrilas, L.I.; Ionescu, C.; Tudoran, O.; Lisencu, C.; Balacescu, O.; Miere, D. The Role of Bioactive Dietary Components in Modulating miRNA Expression in Colorectal Cancer. Nutrients 2016, 8, 590. [Google Scholar] [CrossRef]
- Thomas, L.W.; Lam, C.; Edwards, S.W. Mcl-1; the Molecular Regulation of Protein Function. FEBS Lett 2010, 584, 2981–2989. [Google Scholar] [CrossRef]
- Germain, M.; Milburn, J.; Duronio, V. MCL-1 Inhibits BAX in the Absence of MCL-1/BAX Interaction. J Biol Chem 2008, 283, 6384–6392. [Google Scholar] [CrossRef]
- Karch, J.; Molkentin, J.D. Regulated Necrotic Cell Death: The Passive Aggressive Side of Bax and Bak. Circ Res 2015, 116, 1800–1809. [Google Scholar] [CrossRef]
- Widden, H.; Placzek, W.J. The Multiple Mechanisms of MCL1 in the Regulation of Cell Fate. Commun Biol 2021, 4, 1029. [Google Scholar] [CrossRef]
- Quintanilha, B.J.; Reis, B.Z.; Duarte, G.B.S.; Cozzolino, S.M.F.; Rogero, M.M. Nutrimiromics: Role of microRNAs and Nutrition in Modulating Inflammation and Chronic Diseases. Nutrients 2017, 9, 1168. [Google Scholar] [CrossRef] [PubMed]
- D. Skrzypczak et al., “Innovative uses of biochar derived from tannery waste as a soil amendment and fertilizer,” Biomass Convers Biorefin, vol. 14, no. 5, pp. 7057–7073, Mar. 2024. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



