Submitted:
11 December 2025
Posted:
12 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell Culture Preparation
2.2. Hippocampal Sliсes
2.3. Fluorescent [Ca2+]i Measurements
2.4. Electrophysiological Measurements
2.5. Reagents
3. Results
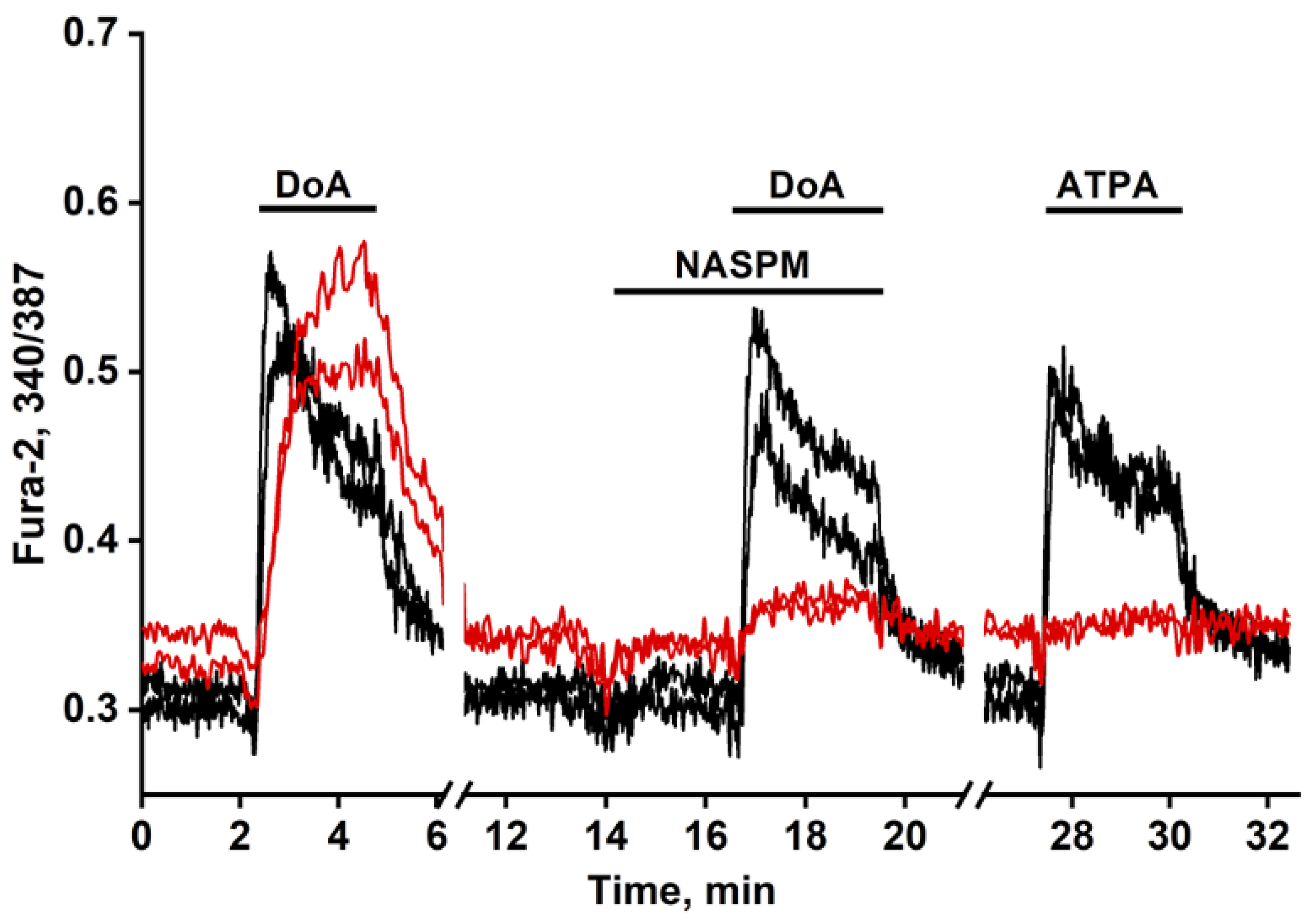
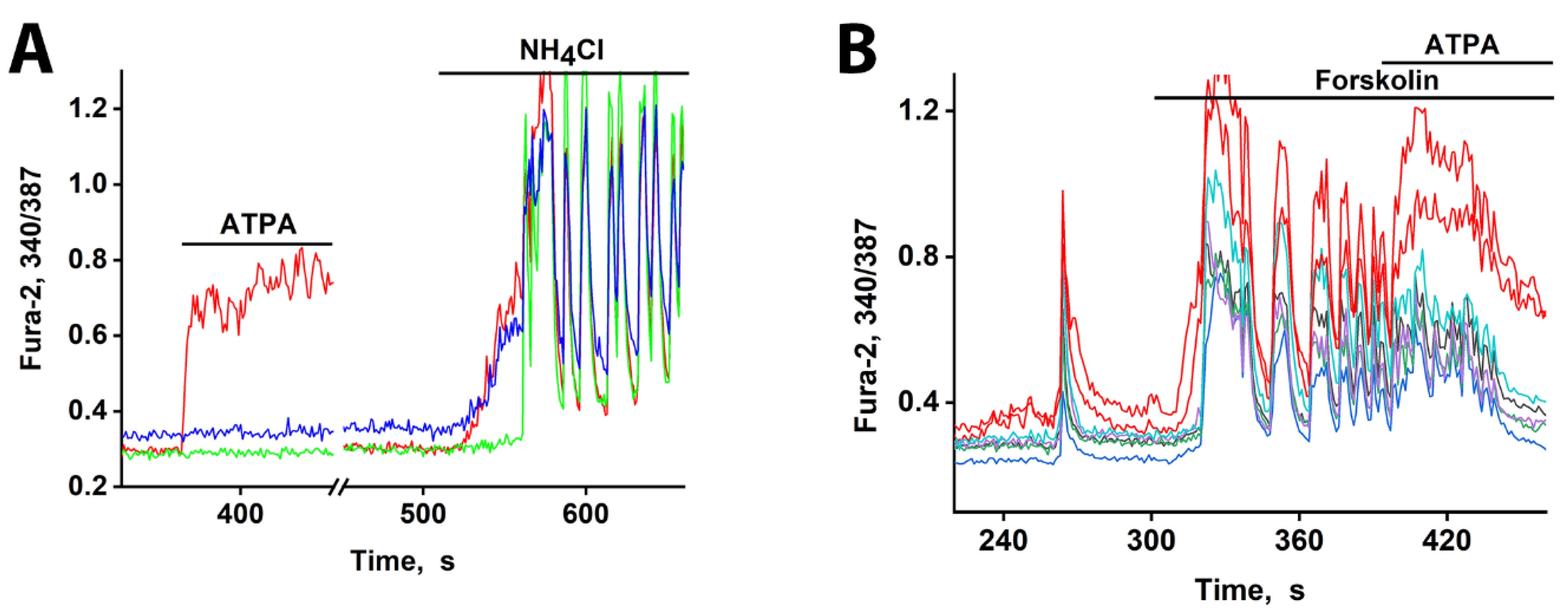
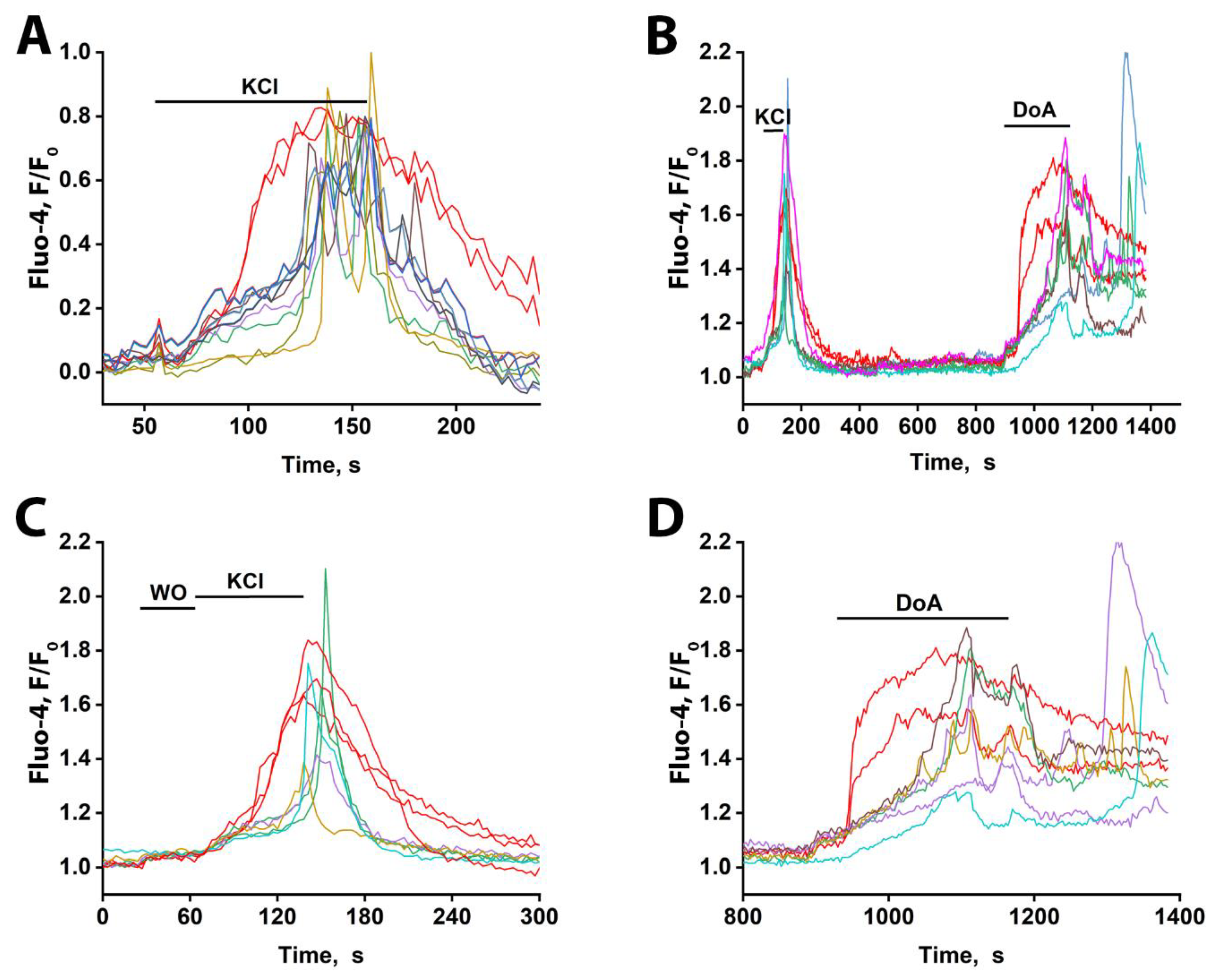
3.1. GABAergic Neurons Expressing CP-KARs and CP-AMPARs Receptors
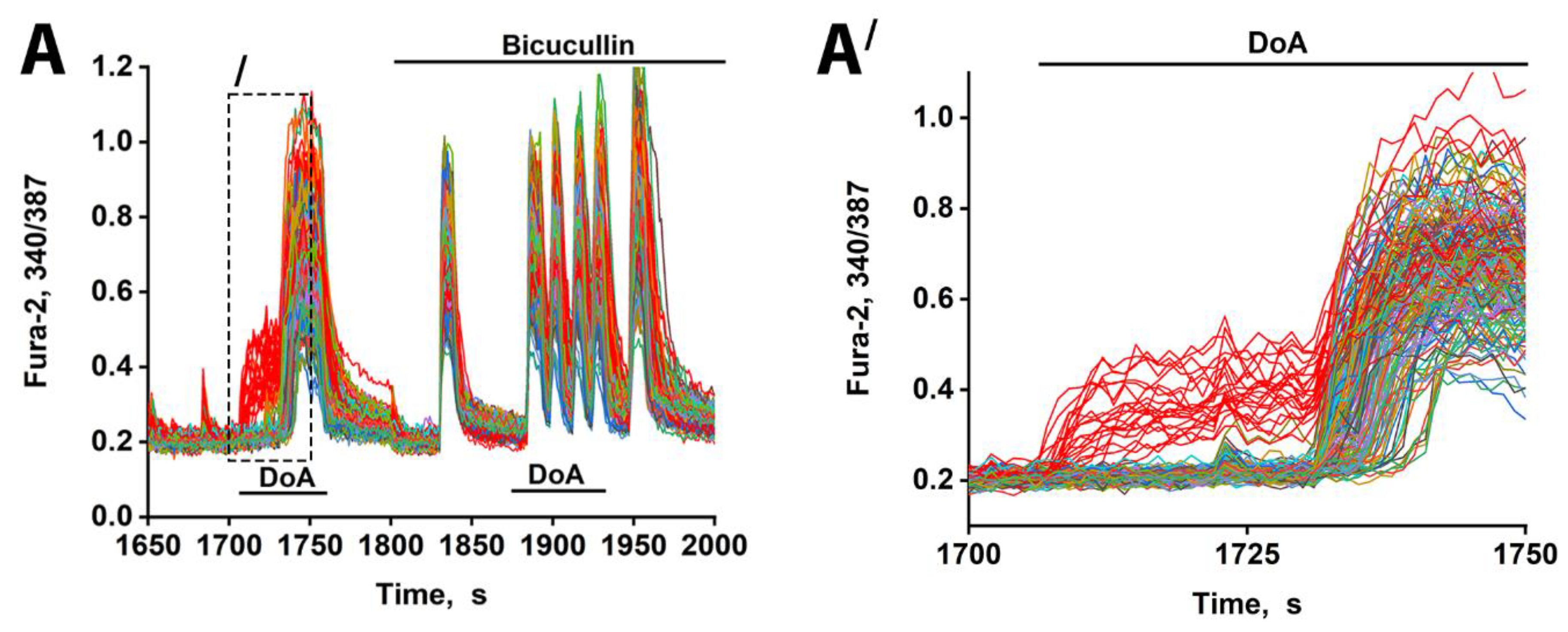
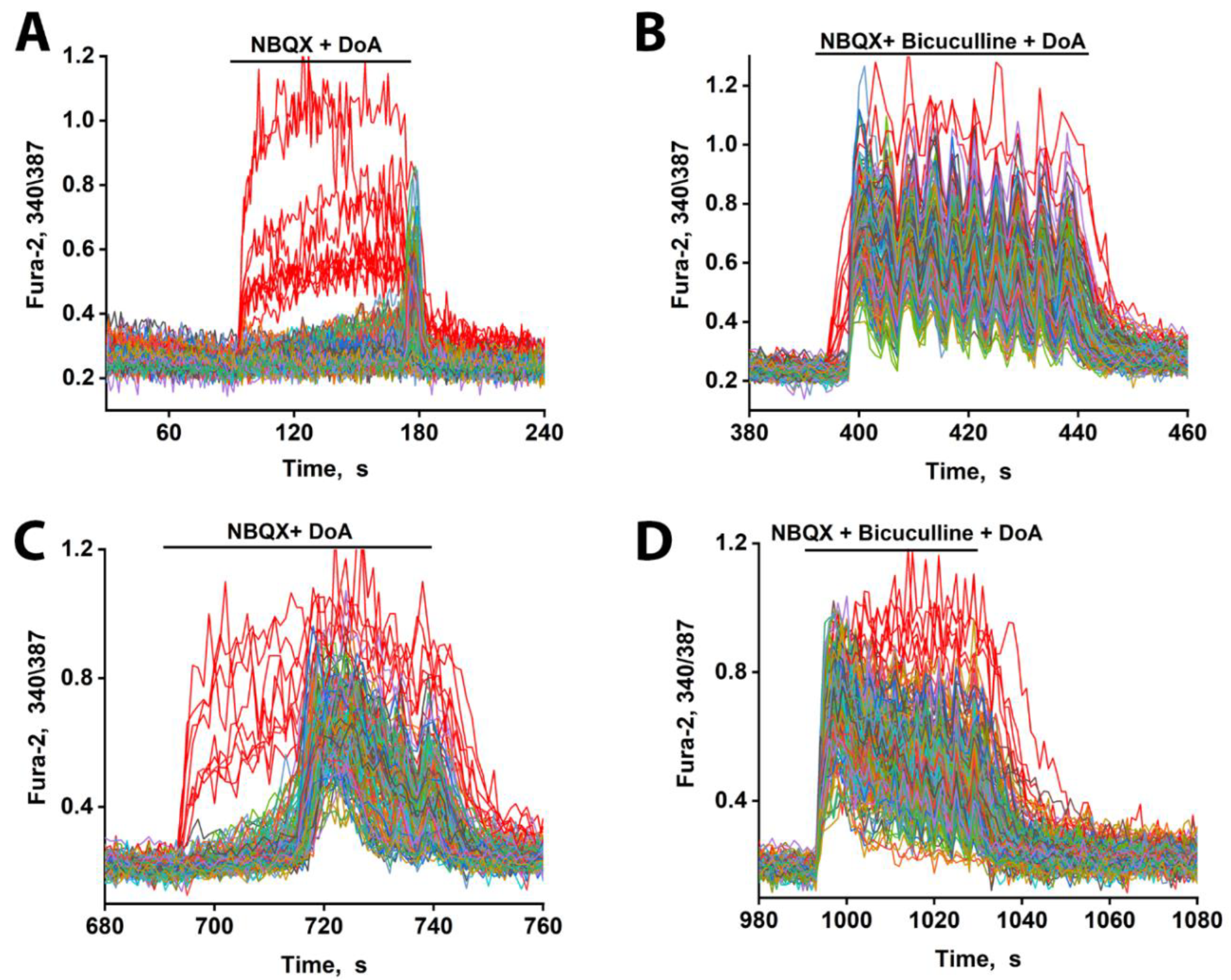
3.2. The Advancing Response of Neurons to DoA Is Eliminated by GABA(A) Receptor Inhibitors
3.3. The Excitation Delay Depends on the Concentration of the Agonist and Is Determined by CP-KARs
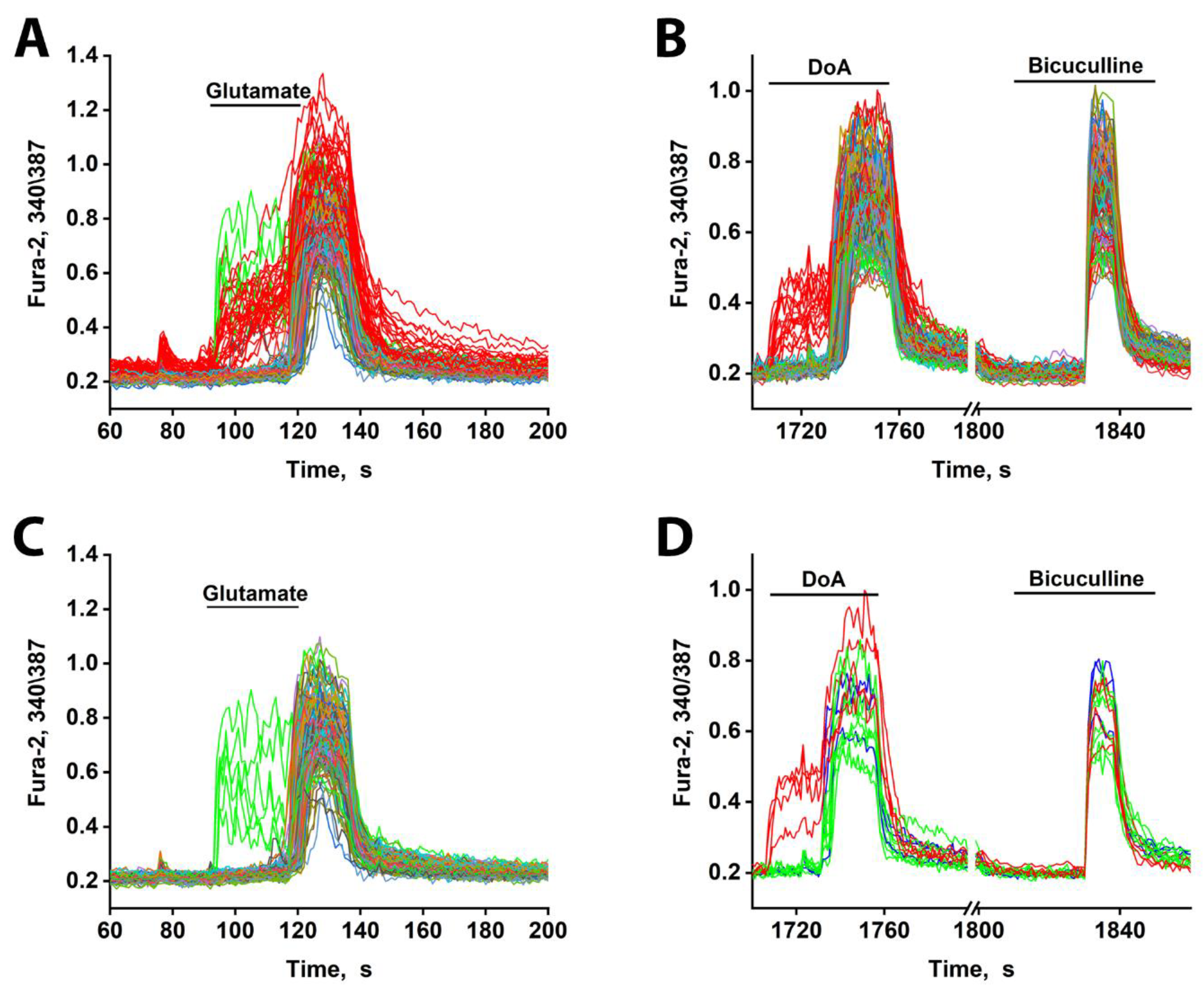
3.4. Early Response of DoA-Sensitive GABAergic Neurons to Glutamate
3.5. Early Response of DoA-Sensitive GABAergic Neurons to Depolarization
3.6. GABAergic Neurons Exhibit an Early Response to cAMP Elevation
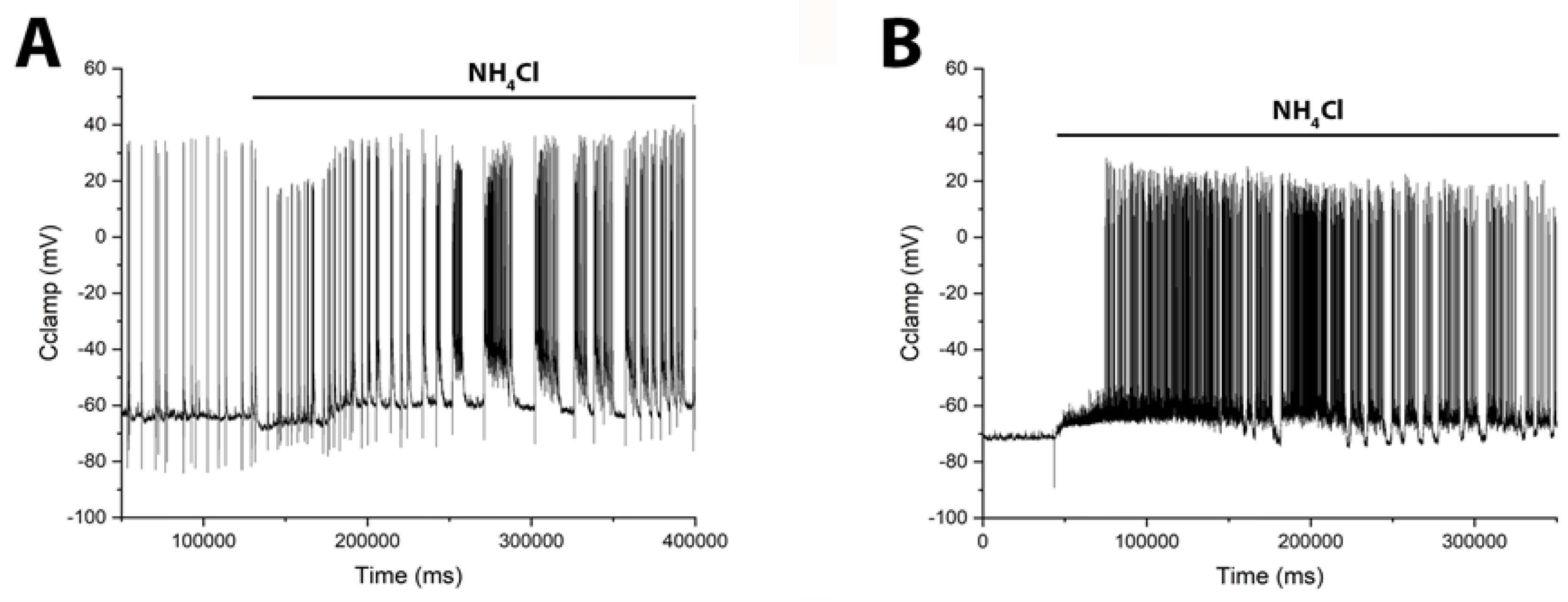
3.7. The Early GABAergic Neuron Response to Excitation in Hippocampal Slices
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Zinchenko, V.P.; Kosenkov, A.M.; Gaidin, S.G.; Sergeev, A.I.; Dolgacheva, L.P.; Tuleukhanov, S.T. Properties of GABAergic Neurons Containing Calcium-Permeable Kainate and AMPA-Receptors. Life 2021, 11, 1309. [CrossRef]
- Gaidin, S.G.; Maiorov, S.A.; Laryushkin, D.P.; Zinchenko, V.P.; Kosenkov, A.M. A novel approach for vital visualization and studying of neurons containing Ca2+-permeable AMPA receptors. J. Neurochem. 2022, 164, 583–597. [CrossRef]
- Zinchenko, V.P.; Dolgacheva, L.P.; Tuleukhanov, S.T. Calcium-permeable AMPA and kainate receptors of GABAergic neurons. Biophys. Rev. 2024, 16, 165–171. [CrossRef]
- Kosenkov, A.M.; Teplov, I.Y.; Sergeev, A.I.; Maiorov, S.A.; Zinchenko, V.P.; Gaidin, S.G. Domoic acid suppresses hyperexcitation in the network due to activation of kainate receptors of GABAergic neurons. Arch. Biochem. Biophys. 2019, 671, 52–61. [CrossRef]
- Hoo, K.; Legutko, B.; Rizkalla, G.; Deverill, M.; Hawes, C.; Ellis, G.J.; Stensbol, T.B.; Krogsgaard-Larsen, P.; Skolnick, P.; Bleakman, D. [3H]ATPA: a high affinity ligand for GluR5 kainate receptors. Neuropharmacology 1999, 38, 1811–1817. [CrossRef]
- Zinchenko, V.P.; Teplov, I.Y.; Kosenkov, A.M.; Gaidin, S.G.; Kairat, B.K.; Tuleukhanov, S.T. Participation of calcium-permeable AMPA receptors in the regulation of epileptiform activity of hippocampal neurons. Front. Synaptic Neurosci. 2024, 16, 1349984. [CrossRef]
- Kononov, A.V.; Bal’, N.V.; Zinchenko, V.P. Control of spontaneous synchronous Ca2+ oscillations in hippocampal neurons by GABAergic neurons containing kainate receptors without desensitization. Biochem. (Moscow), Suppl. Ser. A: Membr. Cell Biol. 2012, 6, 215–220. [CrossRef]
- Maiorov, S.; Zinchenko, V.; Gaidin, S.; Kosenkov, A. Potential mechanism of GABA secretion in response to the activation of GluK1-containing kainate receptors. Neurosci. Res. 2021, 171, 27–33. [CrossRef]
- Lv, Q.; Liu, Y.; Han, D.; Xu, J.; Zong, Y.-Y.; Wang, Y.; Zhang, G.-Y. Neuroprotection of GluK1 kainate receptor agonist ATPA against ischemic neuronal injury through inhibiting GluK2 kainate receptor–JNK3 pathway via GABAA receptors. Brain Res. 2012, 1456, 1–13. [CrossRef]
- Goldberg, J.H.; Lacefield, C.O.; Yuste, R. Global dendritic calcium spikes in mouse layer 5 low threshold spiking interneurones: implications for control of pyramidal cell bursting. J. Physiol. 2004, 558, 465–478. [CrossRef]
- Hewitt, L.T.; Marron, A.M.; Brager, D.H. Higher hyperpolarization-activated current in a subpopulation of interneurons in stratum oriens of area CA1 in the hippocampus of fragile X mice. J. Neurophysiol. 2025, 133, 1558–1571. [CrossRef]
- Bezaire, M.J.; Soltesz, I. Quantitative assessment of CA1 local circuits: Knowledge base for interneuron-pyramidal cell connectivity. Hippocampus 2013, 23, 751–785. [CrossRef]
- Gaidin, S.; Kosenkov, A.; Zinchenko, V.; Kairat, B.; Malibayeva, A.; Tuleukhanov, S. Identification of Neurons Containing Calcium-Permeable AMPA and Kainate Receptors Using Ca2+ Imaging. Bio-Protocol 2025, 15, e5199. [CrossRef]
- Sun, H.Y.; Bartley, A.F.; Dobrunz, L.E. Calcium-Permeable Presynaptic Kainate Receptors Involved in Excitatory Short-Term Facilitation Onto Somatostatin Interneurons During Natural Stimulus Patterns. J. Neurophysiol. 2009, 101, 1043–1055. [CrossRef]
- Caiati, M.D.; Sivakumaran, S.; Cherubini, E. In the Developing Rat Hippocampus, Endogenous Activation of Presynaptic Kainate Receptors Reduces GABA Release from Mossy Fiber Terminals. J. Neurosci. 2010, 30, 1750–1759. [CrossRef]
- Sakha, P.; Vesikansa, A.; Orav, E.; Heikkinen, J.; Kukko-Lukjanov, T.-K.; Shintyapina, A.; Franssila, S.; Jokinen, V.; Huttunen, H.J.; Lauri, S.E. Axonal Kainate Receptors Modulate the Strength of Efferent Connectivity by Regulating Presynaptic Differentiation. Front. Cell. Neurosci. 2016, 10, 3. [CrossRef]
- Cossart, R.; Tyzio, R.; Dinocourt, C.; Esclapez, M.; Hirsch, J.; Ben-Ari, Y.; Bernard, C. Presynaptic Kainate Receptors that Enhance the Release of GABA on CA1 Hippocampal Interneurons. Neuron 2001, 29, 497–508. [CrossRef]
- A Cepeda-Prado, E.; Khodaie, B.; Quiceno, G.D.; Beythien, S.; Edelmann, E.; Lessmann, V. Calcium-Permeable AMPA Receptors Mediate Timing-Dependent LTP Elicited by Low Repeat Coincident Pre- and Postsynaptic Activity at Schaffer Collateral-CA1 Synapses. Cereb. Cortex 2021, 32, 1682–1703. [CrossRef]
- Lalanne, T.; Oyrer, J.; Farrant, M.; Sjöström, P.J. Synapse Type-Dependent Expression of Calcium-Permeable AMPA Receptors. Front. Synaptic Neurosci. 2018, 10, 34. [CrossRef]
- Zinchenko, V.P.; Gaidin, S.G.; Teplov, I.Y.; Kosenkov, A.M. Inhibition of spontaneous synchronous activity of hippocampal neurons by excitation of GABAergic neurons. Biochem. (Moscow), Suppl. Ser. A: Membr. Cell Biol. 2017, 11, 261–274. [CrossRef]
- Kosenkov, A.M.; Gaidin, S.G.; Sergeev, A.I.; Teplov, I.Y.; Zinchenko, V.P. Fast changes of NMDA and AMPA receptor activity under acute hyperammonemia in vitro. Neurosci. Lett. 2018, 686, 80–86. [CrossRef]
- Allert, N.; Köller, H.; Siebler, M. Ammonia-induced depolarization of cultured rat cortical astrocytes. Brain Res. 1998, 782, 261–270. [CrossRef]
- Kilfoil, P.J.; Tipparaju, S.M.; Barski, O.A.; Bhatnagar, A. Regulation of Ion Channels by Pyridine Nucleotides. Circ. Res. 2013, 112, 721–741. [CrossRef]
- Leung, L.-W.S.; Yim, C.-Y.C. Intrinsic membrane potential oscillations in hippocampal neurons in vitro. Brain Res. 1991, 553, 261–274. [CrossRef]
- Teplov, I.Y.; Zinchenko, V.; Kosenkov, A.; Gaidin, S.; Nenov, M.; Sergeev, A. Involvement of NMDA and GABA(A) receptors in modulation of spontaneous activity in hippocampal culture: Interrelations between burst firing and intracellular calcium signal. Biochem. Biophys. Res. Commun. 2021, 553, 99–106. [CrossRef]
- Roche, K.W.; O'Brien, R.J.; Mammen, A.L.; Bernhardt, J.; Huganir, R.L. Characterization of Multiple Phosphorylation Sites on the AMPA Receptor GluR1 Subunit. Neuron 1996, 16, 1179–1188. [CrossRef]
- He K, Song L, Cummings LW, Goldman J, Huganir RL, Lee HK (2009). Stabilisation of Ca2 + -permeable AMPA receptors at periynaptic sites by GluR1-S845 phosphorylation. Proc Natl Acad Sci U S A 106: 20033–20038.
- Zinchenko, V.P.; Turovsky, E.A.; Turovskaya, M.V.; Berezhnov, A.V.; Sergeev, A.I.; Dynnik, V.V. NAD causes dissociation of neural networks into subpopulations of neurons by inhibiting the network synchronous hyperactivity evoked by ammonium ions. Biochem. (Moscow), Suppl. Ser. A: Membr. Cell Biol. 2016, 10, 118–125. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



