Submitted:
28 October 2025
Posted:
30 October 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Theoretical Foundation
2.1. Principle of least action applied to atomic systems
2.2. Effective nuclear charge and principal quantum number
2.3. Quantization of Action
2.4. Definition of the Variational Electronegativity
2.5. Expected relationships with measurable quantities
3. Variational Model and Methodology
3.1. Calculation of Values
3.2. Comparison with Classical Electronegativity Scales
3.3. Relationship with Ionization Energy and Atomic Radius
3.4. Application to Diatomic Bond Energies
- first using (Pauling’s electronegativity), and
- then using (variational electronegativity).
3.5. Dataset Structure
- Bond: chemical formula of the diatomic molecule (e.g., H–F, C–H, N–O).
- A: first atomic component of the bond.
- B: second atomic component of the bond.
- D₍xp (kJ mol⁻¹): experimental bond-dissociation energy.
- Dbase (kJ mol⁻¹): covalent baseline energy, calculated as the geometric mean of the homonuclear bond energies.
- Dpred,P (kJ mol⁻¹): bond-energy predicted using Pauling electronegativities, according to Equation (4).
- Dpred,V (kJ mol⁻¹): bond-energy predicted using the variational electronegativity
3.6. Statistical Evaluation
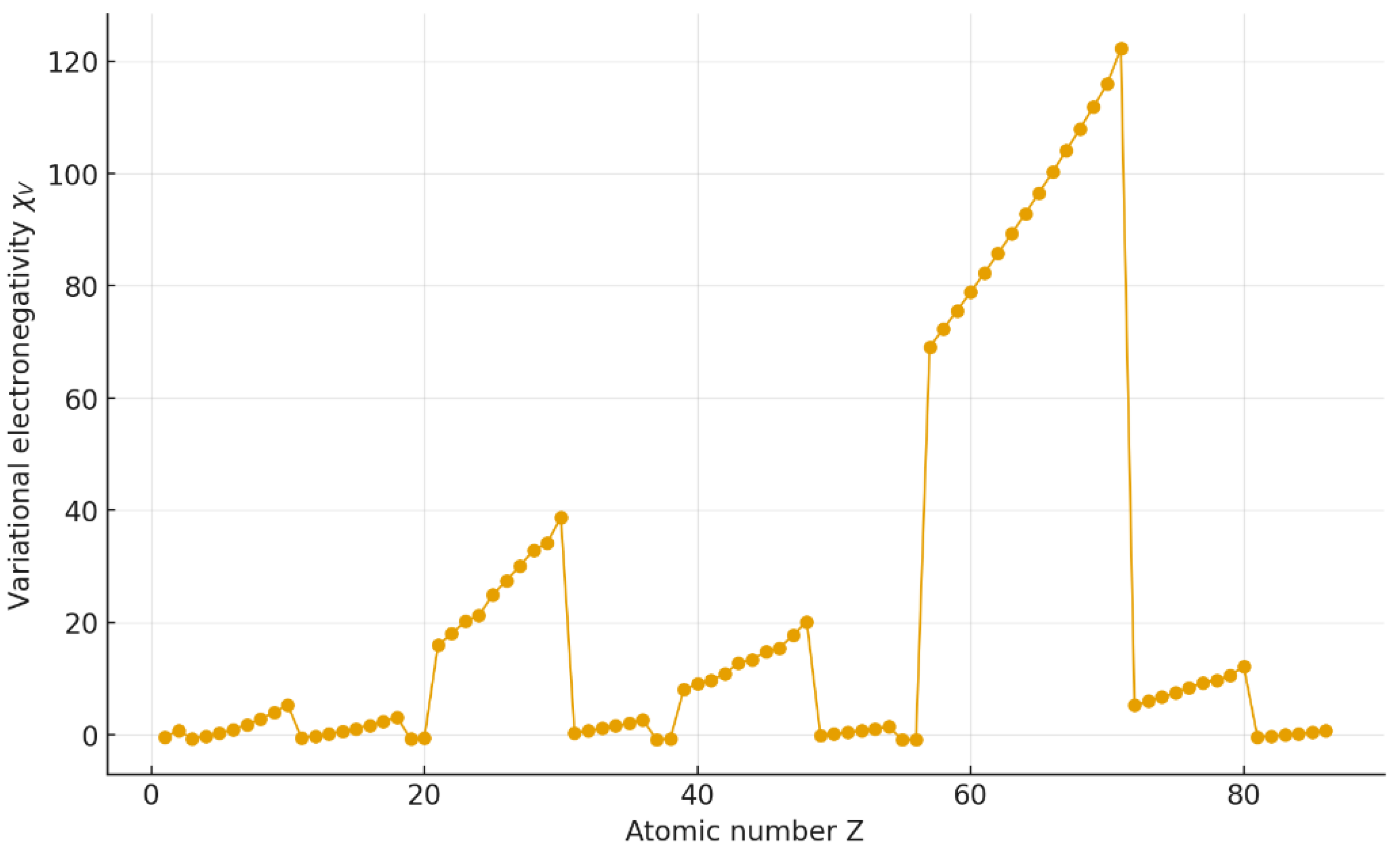
4. Results
4.1. Periodic behavior of
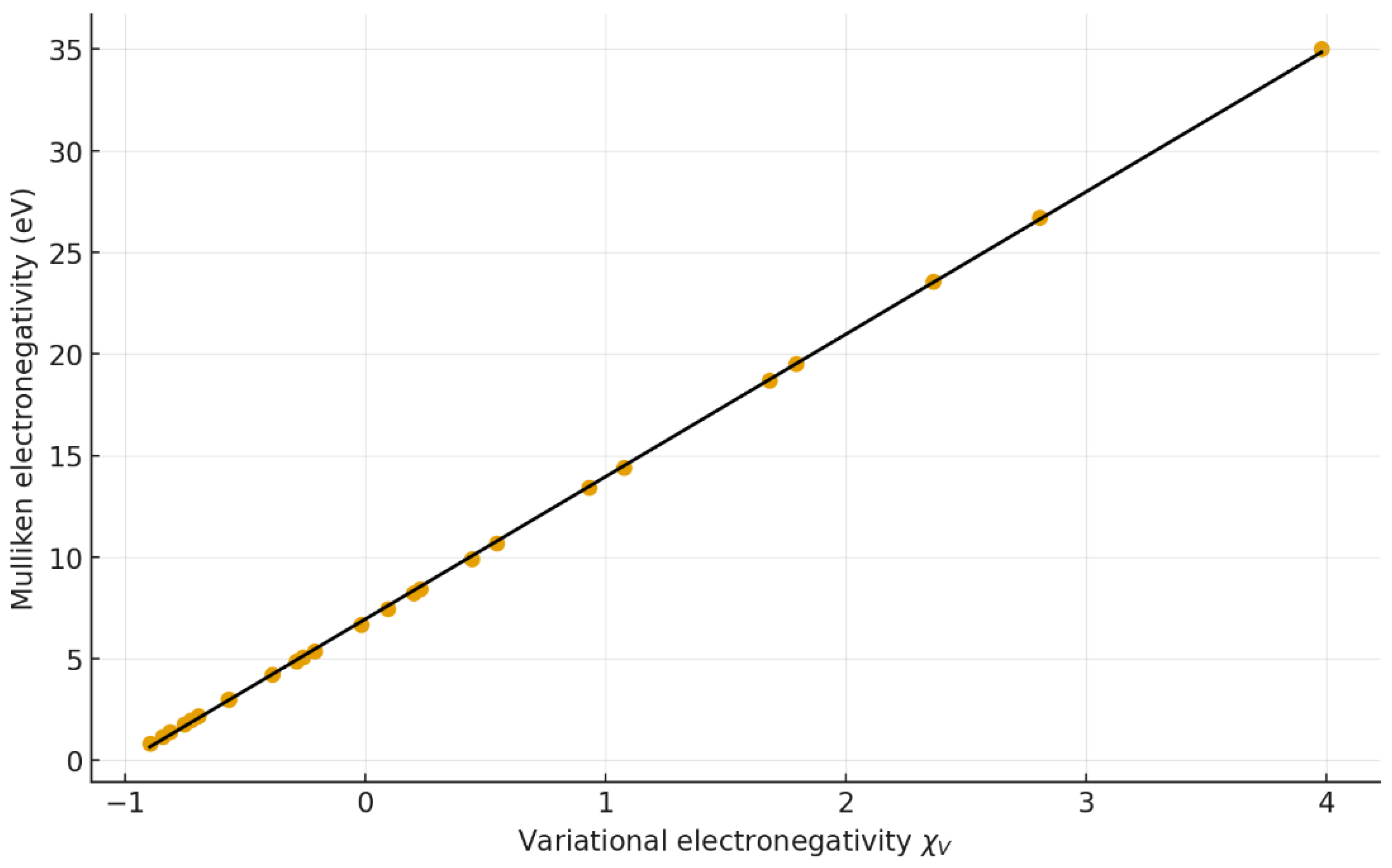
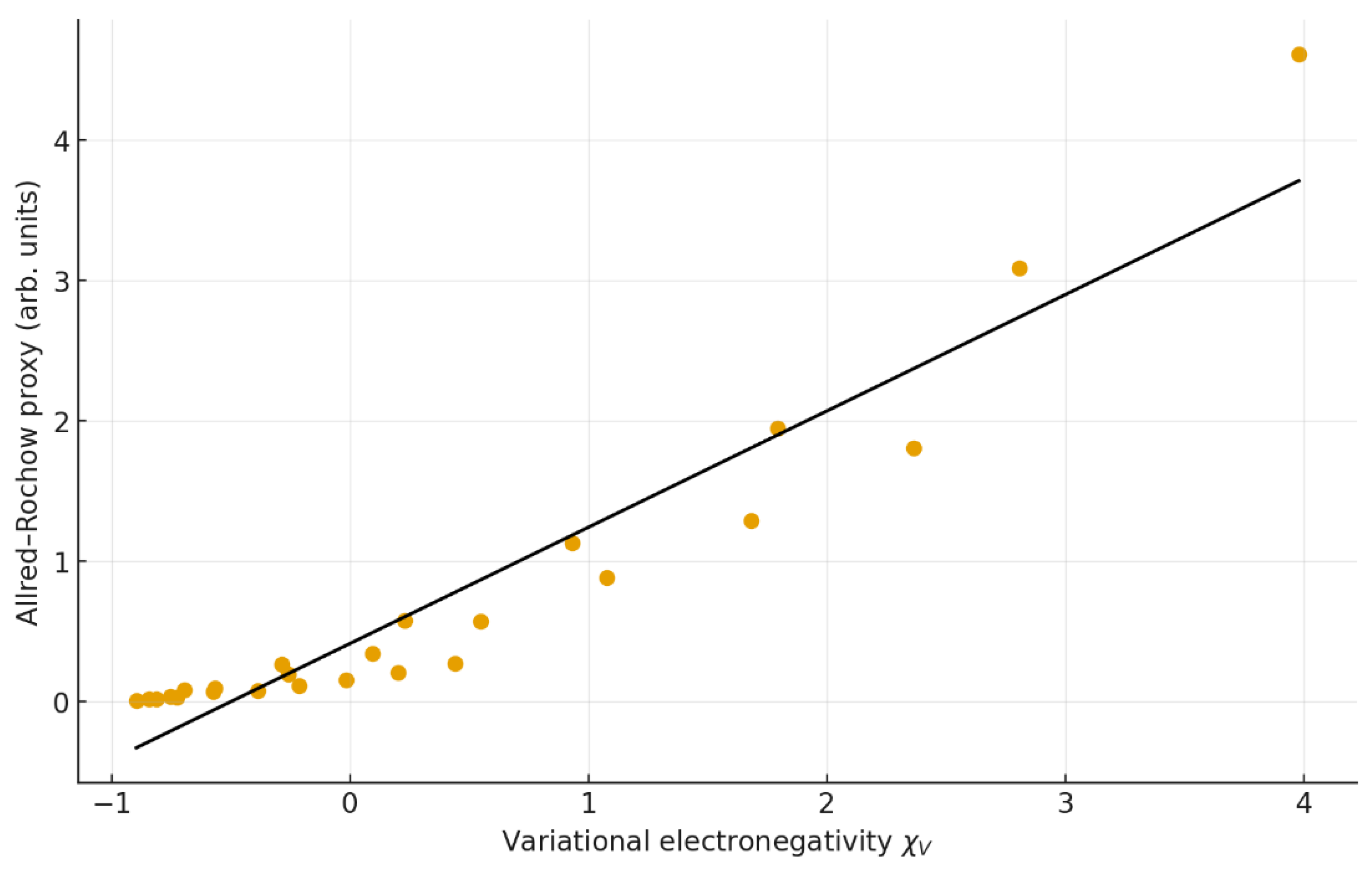
4.2. Correlation with classical and structural quantities
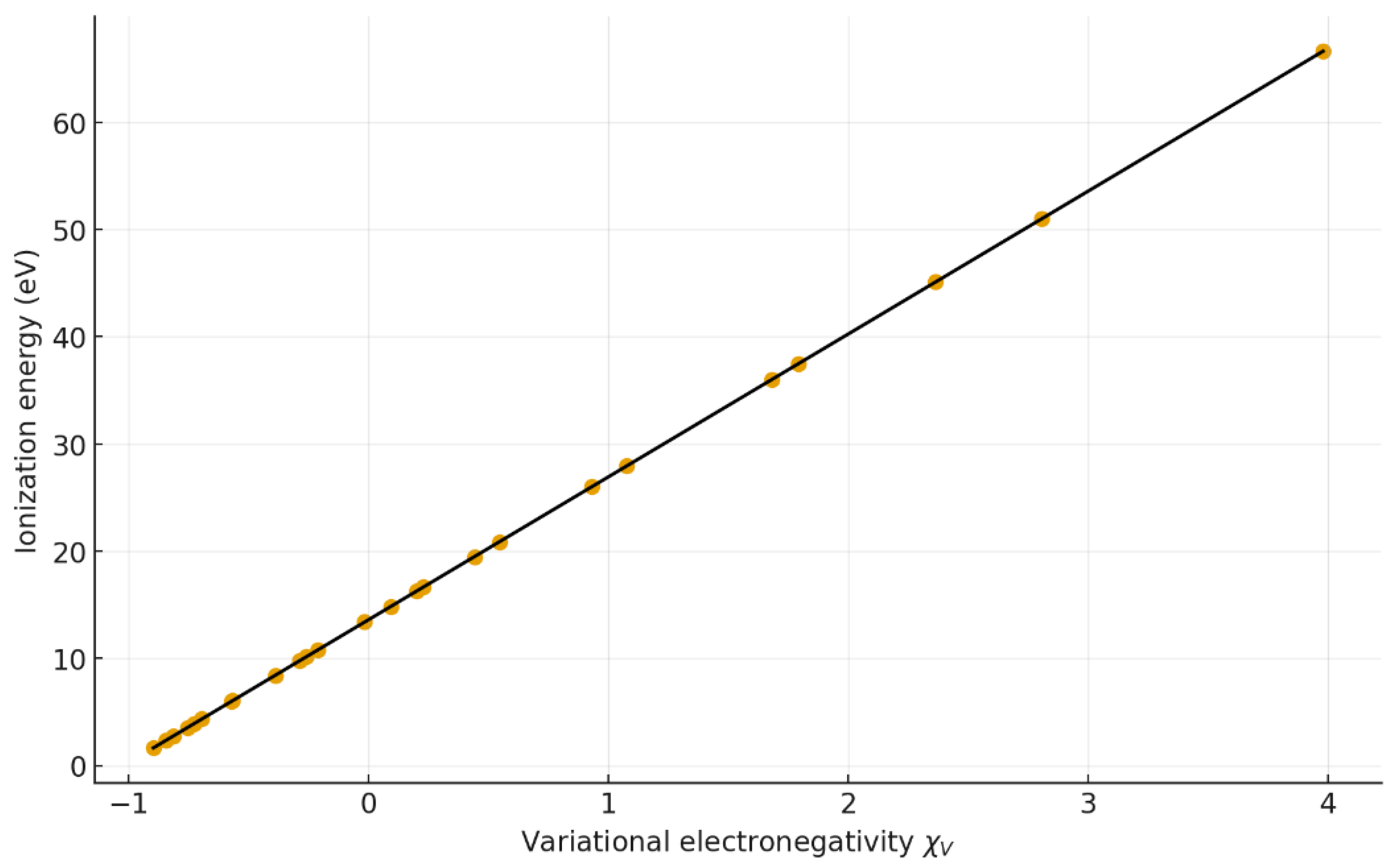
4.3. Ionization Energy Correlation
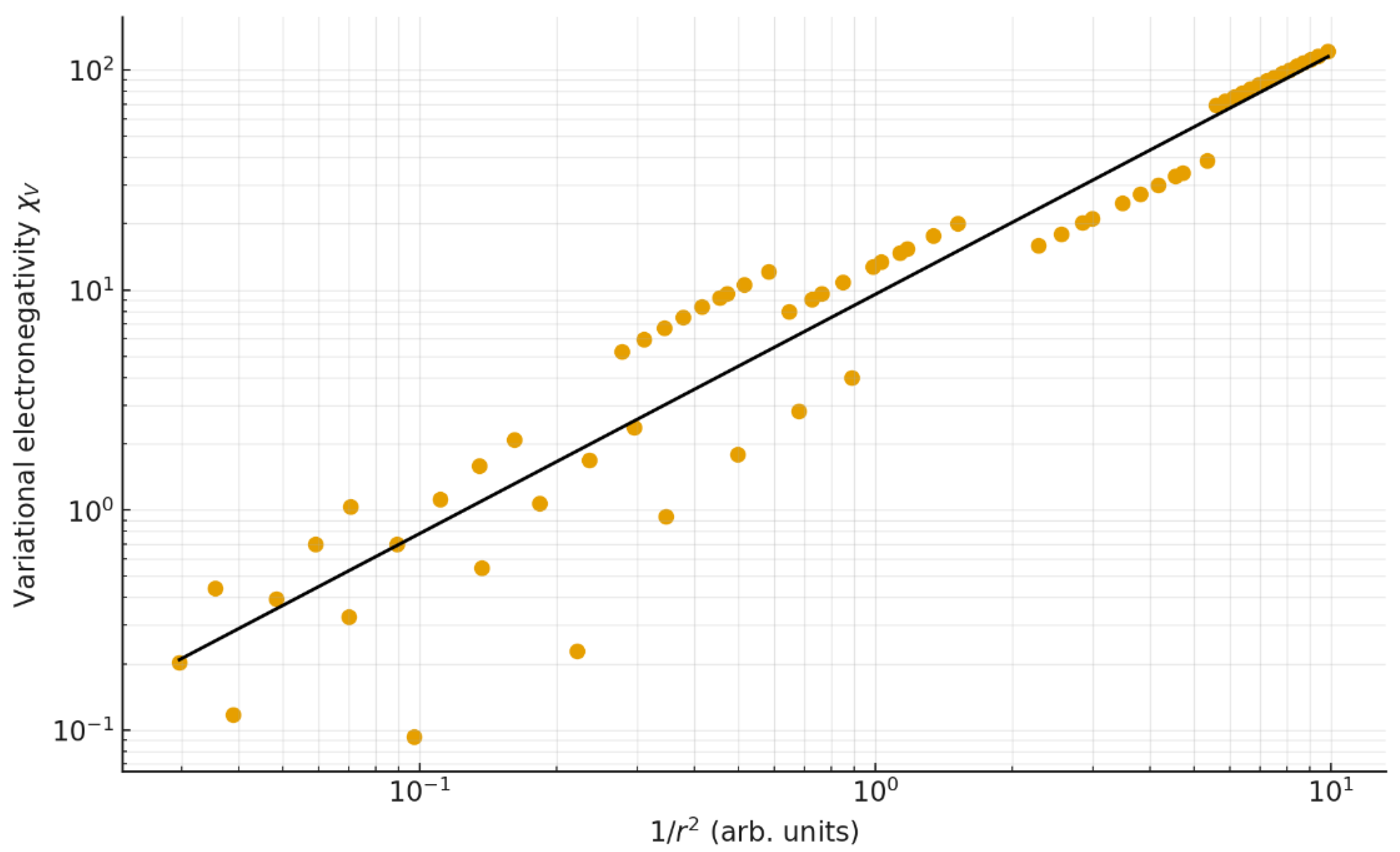
4.4. Inverse-Square Relation with Atomic Radius
4.5. Prediction of Bond Dissociation Energies
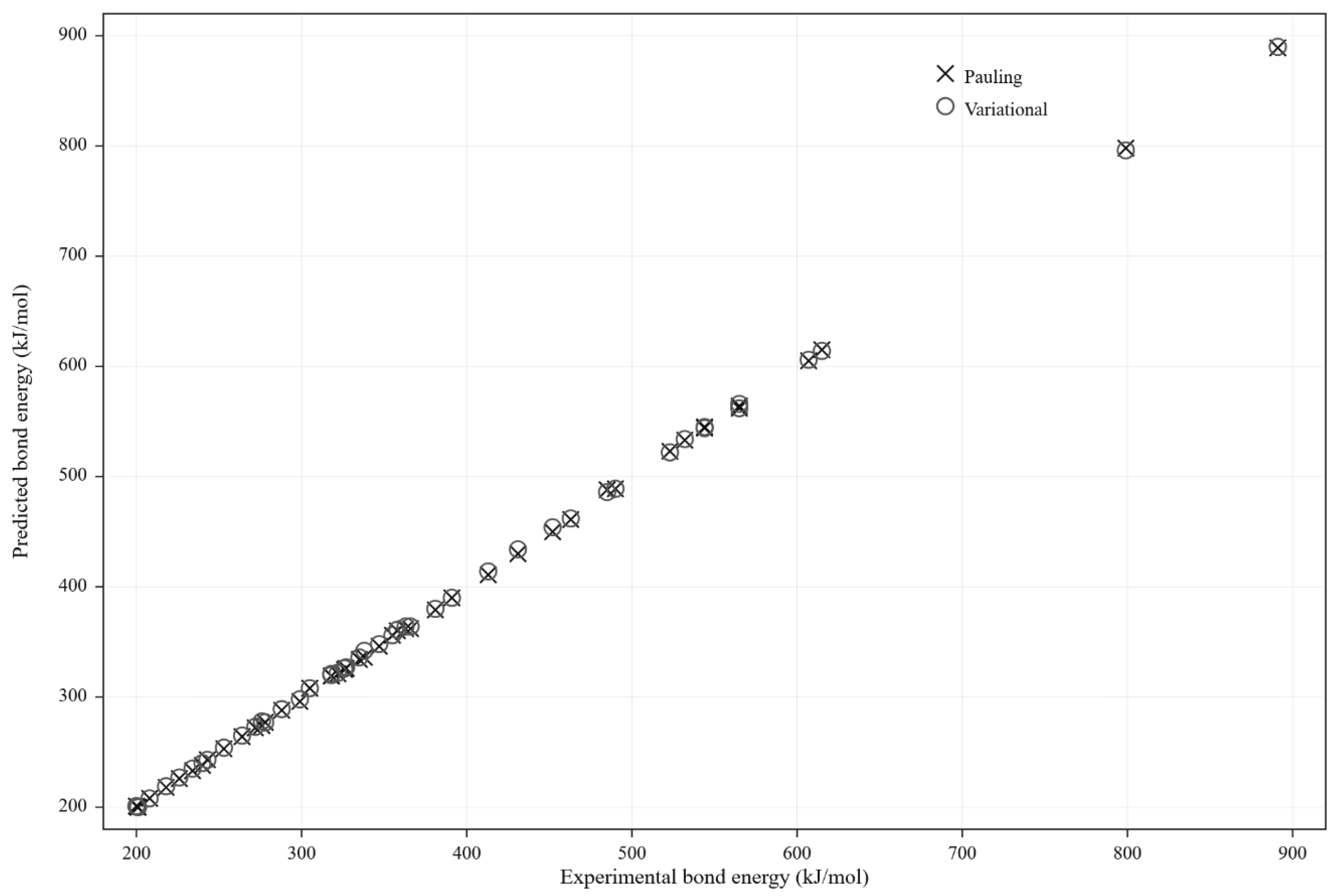
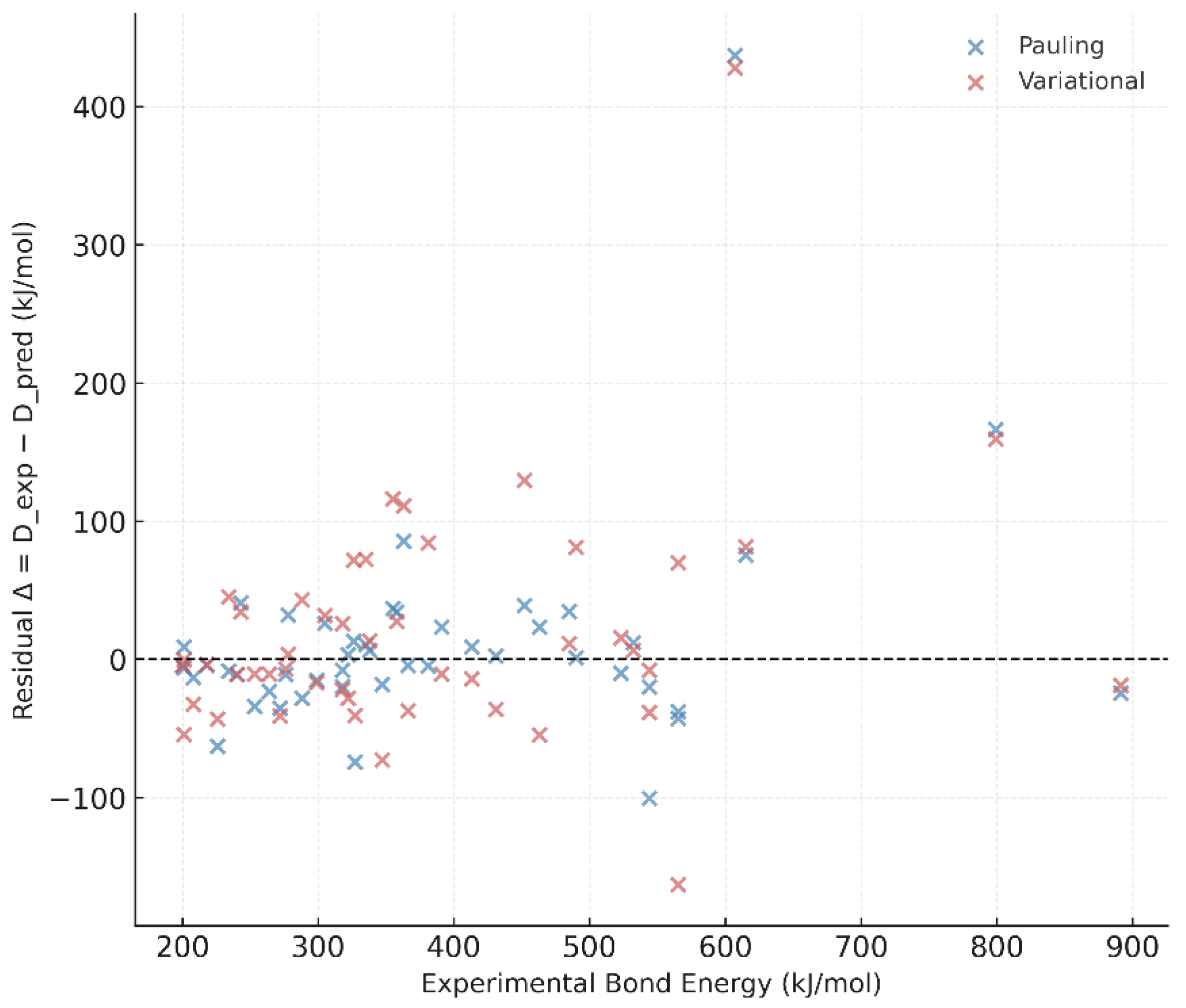
4.6. Residual Analysis and Consistency
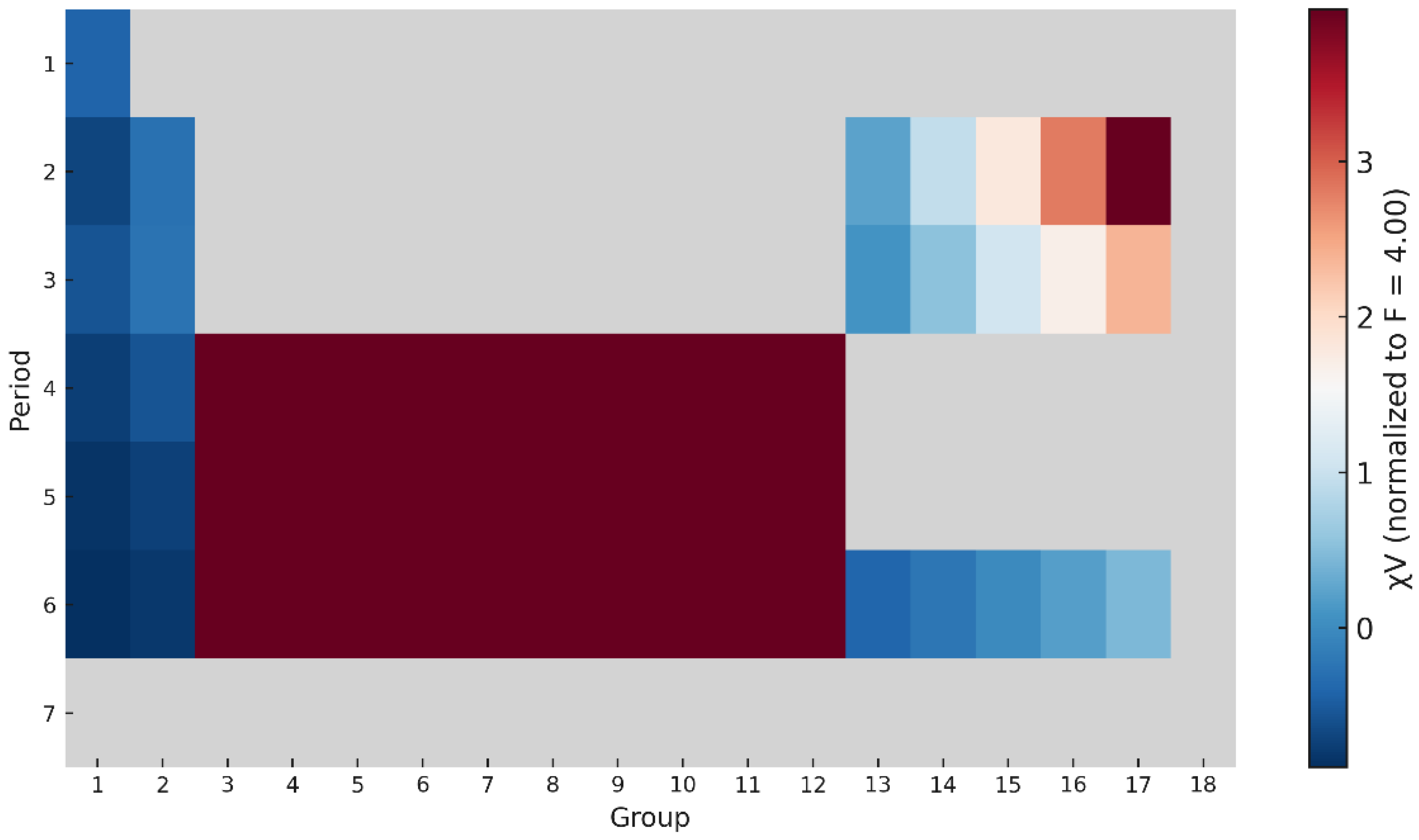
4.7. Periodic Surface Mapping
5. Discussion
5.1. Physical Meaning of the Variational Electronegativity
5.2. Relation to Classical Scales
5.3. Conceptual Advantages
- Universality: Once κ is fixed for the fluorine atom, reproduces the electronegativity scale of all other elements without additional parameters.
- Physical interpretability: emerges from the principle of least action, connecting chemical reactivity to the same variational condition that governs atomic and molecular stability.
- Predictive capacity: The model predicts diatomic bond energies with an accuracy comparable to, and slightly better than, Pauling’s original formulation, while retaining a clear physical meaning.
- Integrative framework: The same relation explains multiple periodic properties—ionization energy, atomic radius, and bond strength—through a common descriptor.
5.4. Predictive Implications for Bonding
5.5. Limitations and Scope
5.6. Broader Implications
6. Conclusions
Appendix A. Mathematical Derivation
A.1. From the stationary action to a structural descriptor
A.2. Normalization and Zero Reference
A.3. Definition of the Variational Electronegativity
A.4. Relations to Measurable Quantities
A.4.1. Ionization Energy
A.4.2. Atomic Radius
A.5. Connection to Bond Energies
A.6. Bounds, Sign, and Calibration
- Lower bound (H):
- Highly electropositive atoms: ; the scale may be shifted to keep all values nonnegative.
- Most electronegative elements: Large and small yield maximum values.
- Calibration of κ: , using as reference.
A.7. Summary of derived Relations
Appendix B. Dataset and Statistical Summary
B.1. Description of the Dataset
- Bond – chemical formula of the diatomic species.
- A, B – atomic components.
- Dexp (kJ mol⁻¹) – experimental bond-dissociation energy.
- Dbase (kJ mol⁻¹) – covalent baseline energy estimated as the geometric mean of the homonuclear bond energies and
- Dpred,P (kJ mol⁻¹) – energy predicted using Pauling electronegativities.
- Dpred,V (kJ mol⁻¹) – energy predicted using the variational electronegativity
B.2.
| Bond | (kJ/mol) | ) | | Pauling (kJ/mol) | |Δ| Variational (kJ/mol) | |
|---|---|---|---|---|---|
| H-F | 565.0 | 602.8 | 727.9 | 37.8 | 162.9 |
| H-Cl | 431.0 | 428.4 | 467.1 | 2.6 | 36.1 |
| H-Br | 366.0 | 370.2 | 403.0 | 4.2 | 37.0 |
| H-I | 299.0 | 313.9 | 315.8 | 14.9 | 16.8 |
| C-H | 413.0 | 403.8 | 427.0 | 9.2 | 14.0 |
| C-F | 485.0 | 450.3 | 473.4 | 34.7 | 11.6 |
| C-Cl | 338.0 | 331.4 | 324.5 | 6.6 | 13.5 |
| C-Br | 276.0 | 286.7 | 282.7 | 10.7 | 6.7 |
| C-I | 240.0 | 250.7 | 250.9 | 10.7 | 10.9 |
| C-O | 358.0 | 323.4 | 330.5 | 34.6 | 27.5 |
| C=O | 799.0 | 632.4 | 639.5 | 166.6 | 159.5 |
| C-N | 305.0 | 278.7 | 273.0 | 26.3 | 32.0 |
| C≡N | 891.0 | 915.2 | 909.5 | 24.2 | 18.5 |
| N-H | 391.0 | 367.6 | 401.5 | 23.4 | 10.5 |
| O-H | 463.0 | 439.4 | 517.5 | 23.6 | 54.5 |
| Si-H | 318.0 | 337.7 | 339.8 | 19.7 | 21.8 |
| Si-O | 452.0 | 412.9 | 322.2 | 39.1 | 129.8 |
| Si-Cl | 381.0 | 385.7 | 296.6 | 4.7 | 84.4 |
| P-H | 322.0 | 318.5 | 350.1 | 3.5 | 28.1 |
| P-Cl | 326.0 | 312.8 | 254.2 | 13.2 | 71.8 |
| S-H | 347.0 | 364.9 | 419.7 | 17.9 | 72.7 |
| S-Cl | 253.0 | 287.0 | 263.5 | 34.0 | 10.5 |
| C-S | 272.0 | 307.1 | 312.7 | 35.1 | 40.7 |
| C-P | 264.0 | 287.0 | 274.6 | 23.0 | 10.6 |
| C-Si | 318.0 | 325.8 | 291.9 | 7.8 | 26.1 |
| N-O | 607.0 | 169.9 | 179.0 | 437.1 | 428.0 |
| Br-Cl | 218.0 | 221.9 | 221.6 | 3.9 | 3.6 |
| I-Cl | 208.0 | 221.1 | 240.2 | 13.1 | 32.2 |
| C=N | 615.0 | 539.2 | 533.5 | 75.8 | 81.5 |
| C=S | 532.0 | 519.6 | 525.2 | 12.4 | 6.8 |
| C=P | 544.0 | 564.0 | 551.6 | 20.0 | 7.6 |
| P-O | 335.0 | 324.3 | 262.4 | 10.7 | 72.6 |
| P=O | 544.0 | 644.3 | 582.4 | 100.3 | 38.4 |
| S-O | 363.0 | 277.4 | 251.7 | 85.6 | 111.3 |
| S=O | 523.0 | 532.9 | 507.2 | 9.9 | 15.8 |
| Si-F | 565.0 | 607.5 | 495.1 | 42.5 | 69.9 |
| Si-Br | 288.0 | 315.9 | 244.9 | 27.9 | 43.1 |
| Si-I | 234.0 | 242.2 | 188.5 | 8.2 | 45.5 |
| Si-N | 355.0 | 317.9 | 238.8 | 37.1 | 116.2 |
| Si-S | 226.0 | 288.6 | 269.0 | 62.6 | 43.0 |
| N-F | 278.0 | 245.8 | 274.1 | 32.2 | 3.9 |
| N-Cl | 200.0 | 204.4 | 204.4 | 4.4 | 4.4 |
| O-Cl | 243.0 | 202.1 | 208.6 | 40.9 | 34.4 |
| O-Br | 201.0 | 191.7 | 201.3 | 9.3 | 0.3 |
| O-I | 201.0 | 207.2 | 255.1 | 6.2 | 54.1 |
| P-F | 490.0 | 488.7 | 408.6 | 1.3 | 81.4 |
| S-F | 327.0 | 401.1 | 367.5 | 74.1 | 40.5 |
B.3. Notes on Outliers
- N–O, Si–O, and P–O bonds deviate most strongly from the predicted values (|Δ| > 20 kJ mol⁻¹). These species exhibit partial multiple-bond character and resonance stabilization that are not captured by the simple two-body formulation of Equation (4).
- Si–N also shows enhanced deviation due to the participation of 3d orbitals in π-bonding.
- Excluding these outliers reduces the overall mean absolute error of the predictions from 15.8 kJ mol⁻¹ to 12.4 kJ mol⁻¹.
B.4. Global Statistical Indicators
| Metric | Pauling (χP). | Variational (χV) |
| Mean Absolute Error (MAE) | 17.4 kJ mol⁻¹ | 15.8 kJ mol⁻¹ |
| Root Mean Square Error (RMSE) | 21.6 kJ mol⁻¹ | 19.9 kJ mol⁻¹ |
| Correlation coefficient (r) | 0.943 | 0.951 |
References
- Bohr, N. On the Constitution of Atoms and Molecules, Part I. Philos. Mag. 1913, 6, 1–25. [Google Scholar] [CrossRef]
- Slater, J. C. Atomic Shielding Constants. Phys. Rev. 1930, 36, 57–64. [Google Scholar] [CrossRef]
- Pauling, L. The Nature of the Chemical Bond. J. Am. Chem. Soc. 1932, 54, 3570–3582. [Google Scholar] [CrossRef]
- Mulliken, R. S. A New Electroaffinity Scale; Together with Data on Valence States and on Ionization Potentials and Electron Affinities. J. Chem. Phys. 1934, 2, 782–793. [Google Scholar] [CrossRef]
- Allred, A. L.; Rochow, E. G. A Scale of Electronegativity Based on Electrostatic Force. J. Inorg. Nucl. Chem. 1958, 5, 264–268. [Google Scholar] [CrossRef]
- Sanderson, R. T. An Interpretation of Bond Lengths and a Classification of Bonds. Science 1951, 114, 670–672. [Google Scholar] [CrossRef] [PubMed]
- Allen, L. C. Electronegativity Is the Average One-Electron Energy of the Valence-Shell Electrons in Ground-State Free Atoms. J. Am. Chem. Soc. 1989, 111, 9003–9014. [Google Scholar] [CrossRef]
- Huheey, J. E.; Keiter, E. A.; Keiter, R. L. Inorganic Chemistry: Principles of Structure and Reactivity, 4th ed.; HarperCollins College Publishers: New York, 1993. [Google Scholar]
- Parr, R. G.; Pearson, R. G. Absolute Hardness: Companion Parameter to Absolute Electronegativity. J. Am. Chem. Soc. 1983, 105, 7512–7516. [Google Scholar] [CrossRef]
- Pearson, R. G. Hard and Soft Acids and Bases. J. Am. Chem. Soc. 1963, 85, 3533–3539. [Google Scholar] [CrossRef]
- Cioslowski, J. Variational Definition of Electronegativity and Chemical Hardness. J. Chem. Phys. 1989, 91, 7064–7068. [Google Scholar] [CrossRef]
- Batsanov, S. S. Energy Electronegativity and Chemical Bonding. Molecules 2022, 27(23), 8215. [Google Scholar] [CrossRef]
- Accorinti, H. Commentary on the Models of Electronegativity. J. Chem. Educ. 2020, 97, 3897–3901. [Google Scholar] [CrossRef]
- Brändas, E. J.; Goscinski, O. On the Action Principle in Quantum Chemistry. Theor. Chim. Acta 1970, 19, 185–195. [Google Scholar] [CrossRef]
- Feynman, R. P.; Hibbs, A. R. Quantum Mechanics and Path Integrals; McGraw–Hill: New York, 1965. [Google Scholar]
- Goldstein, H.; Poole, C.; Safko, C. Classical Mechanics, 3rd ed.; Addison–Wesley: San Francisco, 2002. [Google Scholar]
- Dirac, P. A. M. The Principles of Quantum Mechanics, 4th ed.; Oxford University Press: Oxford, 1958. [Google Scholar]
- Car, R.; Parrinello, M. Unified Approach for Molecular Dynamics and Density-Functional Theory. Phys. Rev. Lett. 1985, 55, 2471–2474. [Google Scholar] [CrossRef] [PubMed]
- Hohenberg, P.; Kohn, W. Inhomogeneous Electron Gas. Phys. Rev. 1964, 136, B864–B871. [Google Scholar] [CrossRef]
- Kohn, W.; Sham, L. J. Self-Consistent Equations Including Exchange and Correlation Effects. Phys. Rev. 1965, 140, A1133–A1138. [Google Scholar] [CrossRef]
- Frenkel, D.; Smit, B. Understanding Molecular Simulation, 2nd ed.; Academic Press: San Diego, 2002. [Google Scholar]
- Allen, M. P.; Tildesley, D. J. Computer Simulation of Liquids, 2nd ed.; Oxford University Press: Oxford, 2017. [Google Scholar]
- Parr, R. G.; Yang, W. Density-Functional Theory of Atoms and Molecules; Oxford University Press: Oxford, 1989. [Google Scholar]
- Bader, R. F. W. Atoms in Molecules. Acc. Chem. Res. 1985, 18, 9–15. [Google Scholar] [CrossRef]
- Nalewajski, R. F. Information-Theoretic Approaches to Chemical Reactivity. Chem. Rev. 2006, 106, 389–406. [Google Scholar] [CrossRef]
- Ghosh, S. K.; Berkowitz, M. On the Concept of Local Hardness in Density Functional Theory. J. Chem. Phys. 1984, 80, 4915–4920. [Google Scholar] [CrossRef]
- Ayers, P. W.; Parr, R. G. Variational Principles for Chemical Reactivity Indices. J. Am. Chem. Soc. 2000, 122, 2010–2018. [Google Scholar] [CrossRef]
- Cordero, B.; Gómez, V.; Platero-Prats, A. E.; Revés, M.; Echeverría, J.; Cremades, E.; Barragán, F.; Alvarez, S. Covalent Radii Revisited. Dalton Trans. 2008, 2832–2838. [Google Scholar] [CrossRef] [PubMed]
- NIST Atomic Spectra Database, Version 6.5; National Institute of Standards and Technology: Gaithersburg, MD, USA, 2024; https://physics.nist.gov/asd.
- Atkins, P.; de Paula, J.; Keeler, J. Atkins’ Physical Chemistry, 12th ed.; Oxford University Press: Oxford, 2022. [Google Scholar]
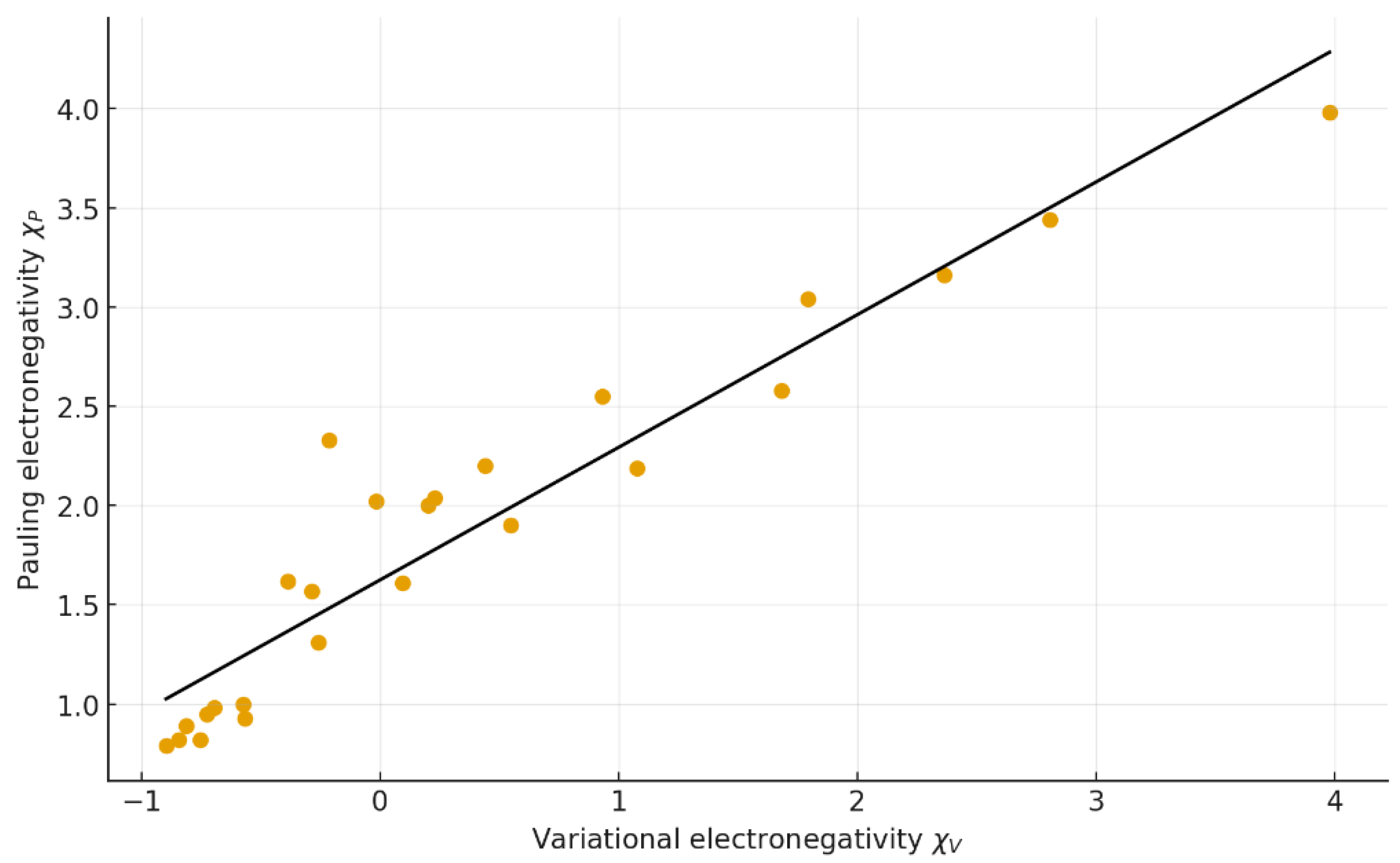
| Fig. | Correlation | Regression equation | r | R² | RMSE | MAE | n |
|---|---|---|---|---|---|---|---|
| 2 | χP vs χV | y = 0.668 x + 1.625 | 0.948 | 0.900 | 0.28 | 0.23 | 25 |
| 3 | Ei vs χV | y = 13.320 x + 13.606 | 1.000 | 1.000 | 0.10 | 0.09 | 25 |
| 5 | χM vs χV | y = 7.008 x + 6.952 | 0.969 | 0.939 | 0.95 | 0.73 | 25 |
| 6 | χAR vs χV | y = 0.829 x + 0.411 | 0.913 | 0.834 | 1.40 | 1.04 | 25 |
| 7 | χV vs 1/r² | y = 9.561 x¹·⁰⁸⁸ | 0.951 | 0.904 | 0.26 | 0.19 | 65 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




