Introduction
Optical properties of plants and plant pigments as their key determinants remain in the focus of intensive research for more than 50 years (Gates et al., 1965; Jacquemoud and Ustin, 2019). There are many reasons for this: on one hand, the interaction of light and plants is at the core of plant functioning, hence it is central to productivity of photosynthesis. On the other hand, our understanding on plant optics is so far limited, despite the significant progress in this field, advent of powerful remote sensing methods and boom of proximal sensing of vegetation (Adamchuk et al., 2018; Gong et al., 2024; Xue and Su, 2017). Thus, the optical spectra of photosynthetic pigments dissolved in organic solvents or embedded in other optically thin system like a diluted chloroplast suspension shows that the absorption of main chlorophyll (Chl) of plants, namely Chl
a, beyond 700 nm is negligible (Lichtenthaler, 1987; Merzlyak et al., 2009). There are exceptions like long-wave Chl
d and recently discovered Chl
f (Allakhverdiev et al., 2016) but their distribution among photosynthetic organisms is rather narrow, so they are not discussed here. By contrast, the analysis of optical properties of intact (non-stressed) plants with mature, well-formed photosynthetic apparatus frequently displays the presence of sizeable absorption in the far red, FR (700–750 nm) range, in addition to the strong absorption in the “classical” visible or PAR (photosynthetically active radiation) range, 400–700 nm (Fig. 1). This spectral feature, termed “Red Edge,” is deemed to be the most important hallmark of organic life on the Earth as well in Space and is widely used in remote sensing of vegetation (Horler et al., 1983). The existence of FR absorption in plants elicited a lot of curiosity since it was known from the early studies of action spectra of photosynthesis that the FR quanta
per se are less efficient drivers of photoevolution of oxygen and carbon fixation in comparison with PAR quanta. At the same time, addition of FR quanta to PAR during irradiation of plans significantly stimulated photosynthesis as was discovered by Emerson (Emerson and Rabinowitch, 1960).
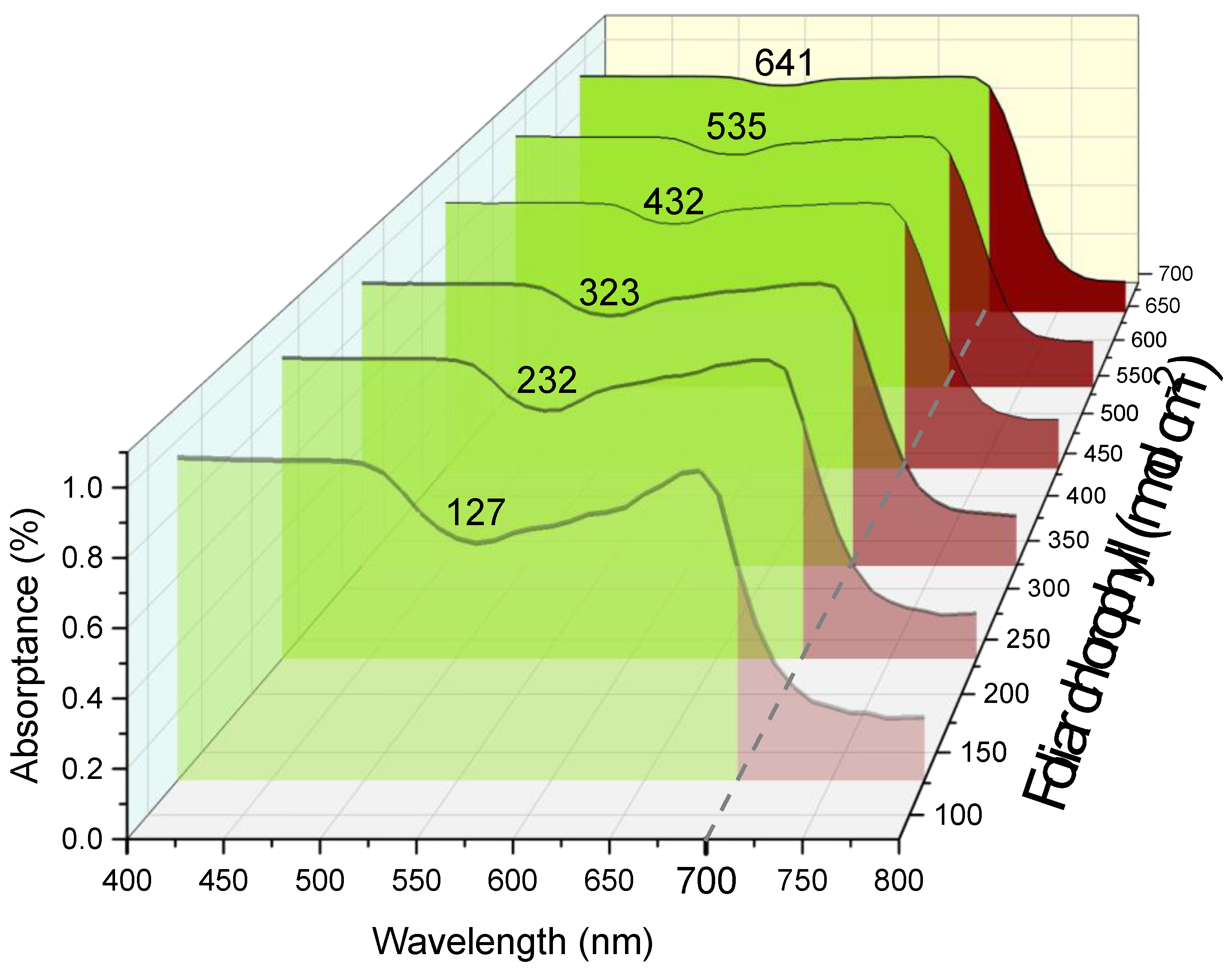
Figure 1.
Variation of absorptance spectra of beech (Fagus sylvatica L.) leaves with moderate-to-high chlorophyll content (nmol cm-2, indicated above the curves). PAR spectral range is highlighted in green, and FR is in red. Note the increase in the FR radiation relative to PAR along with increase in foliar chlorophyll. Absorptance was calculated as A% = (1–R–T)/(1–R), see (Merzlyak and Gitelson, 1995) and Online Supplementary.
Figure 1.
Variation of absorptance spectra of beech (Fagus sylvatica L.) leaves with moderate-to-high chlorophyll content (nmol cm-2, indicated above the curves). PAR spectral range is highlighted in green, and FR is in red. Note the increase in the FR radiation relative to PAR along with increase in foliar chlorophyll. Absorptance was calculated as A% = (1–R–T)/(1–R), see (Merzlyak and Gitelson, 1995) and Online Supplementary.
According to the common understanding, current models of photosynthesis simplify incident light intensity as the integrated intensity over the 400-700 nm waveband known as photosynthetic active radiation, PAR. This conventional approach is embedded into many large-scale models hence they do not account for the effect of FR light. Still, FR light (which is actually in the Near Infra-Red, NIR) range also drives photosynthesis when supplied to plants along with PAR radiation. Estimations of potential FR contribution to carbon assimilation, experimental and theoretical, appeared only recently (Jans et al., 2025; Wang et al., 2025).
While experimental evidence of FR-driven photosynthesis has been accumulating, several hypotheses have been proposed to explain this effect. One of them ascribes the pigment absorption in FR to the long-wave forms of Chl a, presumably existing in form of aggregates or other complexes with proteins of photosynthetic apparatus (Santabarbara et al., 2020). Still, it is hard to explain the large FR absorption by plants by the presence of the long-wave Chl a forms since they (i) generally do not absorb beyond 710 nm and (ii) represent a minor fraction of the pigment pool (Karapetyan et al., 2006; Santabarbara et al., 2020). It is also not possible to dismiss the FR absorption as apparent spectral features stemming from different types of light scattering since even after sophisticated corrections for scattering influence the FR absorption clearly stood out in the spectra of many plant systems (Merzlyak et al., 2009; Merzlyak et al., 2004). So, the questions about the source of the FR absorption of living plants and its possible significance for plants and potential applications in practice are repeatedly asked by many researchers. During recent decades, these questions were revisited several times and, as a result, clarified to varying degrees.
The relationship between Chl content and productivity of plants remains in the focus of research for a long time. Mature, fully functional leaves contain a lot more Chl than necessary to capture light energy in the PAR range sufficient to drive photosynthesis (see below). Illustrious examples include but not limited Schwarzwald broadleaf forest and lower storeys of equatorial rain forests, although this “overaccumulation” of Chl was documented in many common species including crops such as soybean (Gitelson et al., 1996; Gitelson et al., 2025). This “extra Chl” is not only a potential waste of metabolic resources but also a serious risk of photodamage, especially under unfavorable environmental conditions when the capability of the dark reactions (and other metabolic process) for the consumption of ATP and reducing power lags behind the ability of photosynthetic antenna to harvest light energy. On the other hand, it is well known that the relationship between Chl content and absorbed PAR fails in the upper range of Chl content e.g. when a significant part of Chl becomes inefficient in terms of “classical” PAR absorption e.g. due to self-shading and non-linear optical effects (Gitelson et al., 2021; Gitelson, 2013).
Role of FR in light harvesting
The physiological significance of FR absorption repeatedly gained attention of photosynthesis researchers, incenting numerous hypotheses. An important role of FR absorption under high-light conditions is off-loading PS II over-excitation hereby lowering the risk of photodamage (Fig. 2). It is long known that FR (up to 780 nm) can
per se drive photosynthesis (Pettai et al., 2005; Yoon et al., 2020; Zhen et al., 2019) but it is less efficient than PAR due to under-excitation of PS II. If FR is the sole energy source for photosynthesis, the gap between the energy of FR and the energy required for the electron donation is thought to be closed by thermal energy (Jennings et al., 2003). Since it was observed in many plant species, the FR-driven photosynthesis is considered as a general feature of green plants (Pettai et al., 2005).
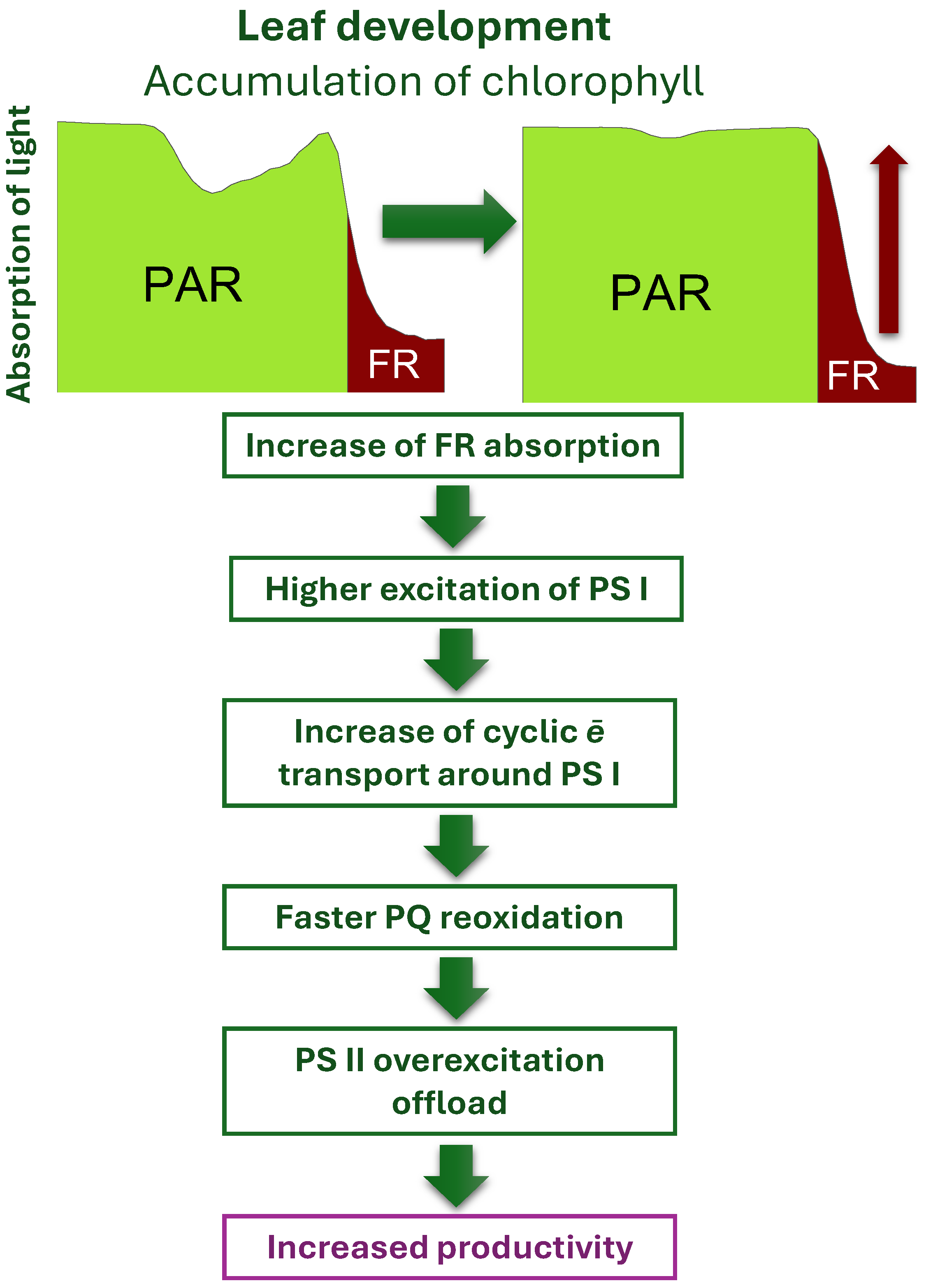
Figure 2.
Schematic summary of the current hypotheses of beneficial effects of FR radiation supplementation on photosynthetic productivity of plants. Increased flux of FR quanta facilitates offloading of photosynthetic electron transfer chain lowering PQ pool overreductions, hence mitigating the risk of photodamage and increasing the efficiency of overall light energy photochemical utilization. For more information, see text and Tan et al. (2022).
Figure 2.
Schematic summary of the current hypotheses of beneficial effects of FR radiation supplementation on photosynthetic productivity of plants. Increased flux of FR quanta facilitates offloading of photosynthetic electron transfer chain lowering PQ pool overreductions, hence mitigating the risk of photodamage and increasing the efficiency of overall light energy photochemical utilization. For more information, see text and Tan et al. (2022).
However, the most spectacular effect is observed when FR radiation is supplemented to PAR radiation (as is normally the case in Sun spectrum reaching plants on Earth surface). Briefly, the augmentation of photosynthetic apparatus by FR includes (i) upregulating the expression of Lhcb1, Lhcb2, and Lhcb4 gene increasing the content of Chl-protein complexes in the PSII reaction center; (ii) preferential excitation of PS I accelerating the oxidation of the plastoquinone pool and promotion of light energy distribution towards PS II, and (iii) acclimatory reshaping the structure of photosynthetic apparatus. Specifically, FR light supplementation may increase the number of grana formed by thylakoid stacks, leading to a high PS II/PS I ratio (see (Tan et al., 2022) for review).
The effect of FR light can be important not only to promote photosynthetic electron transport in fluctuating light environment (Zhang et al., 2025), but also to make the whole volume of canopy/stand working efficiently, both in natural ecosystems and in the crop plants with dense canopy boosting their productivity. (Here, we do not mean the phytochrome-mediated morphological effects on leaf and canopy by R/FR balance-induced change of differential gene expression e.g. alteration of leaf angle, increase in internodal length, plant height, and leaf area.) In the lower leaves of forest canopies, FR light may make up 40% of the total photon capture and usage (Rivadossi et al., 1999; Yoon et al., 2020) At the same time, the absorbed FR light energy is less affected by non-photochemical quenching than the energy acquired by absorbing PAR quanta (Pettai et al., 2005). This is especially important since current crops performance is insufficient to meet the future food demand despite the use of novel approaches such as speed breeding based on marker-assisted selection and high-throughput phenotyping (Kumar et al., 2024; Watt et al., 2020).
The “redundant chlorophyll” and its possible function
Collectively, the considerations presented above strongly support the importance of FR radiation for productivity and environmental fitness of plants. Here, we would like to link the optical features of plants determined by the “redundant” Chl and the capacity of plant to intercept FR quanta. Towards this end, it is essential to develop a consensus about the notion of “redundant” Chl and its relation to far red absorption.
One of the ways to do this is to define the “redundant Chl” as the pigment accumulated in the assimilatory tissues of plants without significant increase in plant’s capacity to capture light and photo-assimilate carbon. With plant development, leaf Chl content and leaf area increase synchronously; as a result, the fraction of PAR absorbed by photosynthetically active vegetation (fAPAR
PS) increases (Fig. 3).
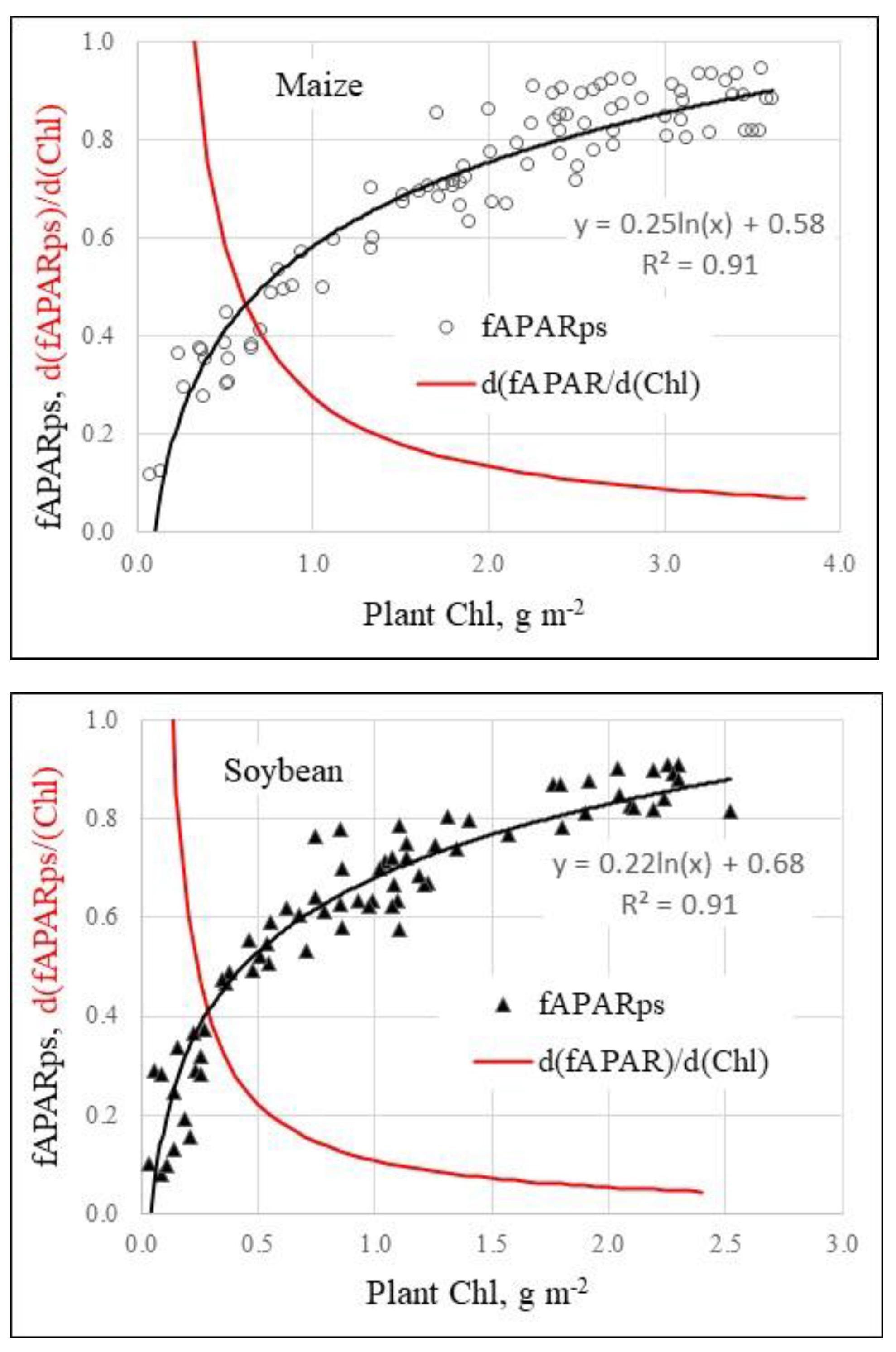
Figure 3.
Accumulation of chlorophyll brings about an increase in PAR absorption but at the same time the efficiency of chlorophyll declines. Fraction of radiation absorbed by photosynthetically active vegetation in PAR (400–700 nm) region and first derivative of fraction of PAR absorbed by plants per unit plant chlorophyll content in maize (a) and in soybean (b) are shown on the plots. The first derivative (red curves) is a measure of Chl efficiency in absorbing PAR.
Figure 3.
Accumulation of chlorophyll brings about an increase in PAR absorption but at the same time the efficiency of chlorophyll declines. Fraction of radiation absorbed by photosynthetically active vegetation in PAR (400–700 nm) region and first derivative of fraction of PAR absorbed by plants per unit plant chlorophyll content in maize (a) and in soybean (b) are shown on the plots. The first derivative (red curves) is a measure of Chl efficiency in absorbing PAR.
However, efficiency of Chl absorbing light in PAR spectral region sharply decreases as Chl content reaches about 1 g·m
–2 ); this pattern is convincingly illustrated by the first derivative of the corresponding function, d(fAPARps)/d(Chl), shown in Fig. 3. The decrease in the efficiency of light absorption by Chl is due to high absorption coefficient of this pigment in PAR. It shortens dramatically the light path inside the leaf and plant canopy which is inversely related to the absorption coefficient (Merzlyak and Gitelson, 1995). As a result, PAR is readily absorbed by the upper storeys of plant canopy shading the lower canopy storeys significantly limiting availability of PAR to the latter. The decrease of PAR absorption efficiency in moderate-to-high plant density (leaf area above 1.5–2 m
2 m
–2) is dramatic—more than 10-fold in maize and 14-fold in soybean (Fig. 3). The relationships “GPP/PAR
in vs. Chl” are quite close to linear (Fig. 4a). Decrease in absorbed light also cause the decrease of Chl efficiency of primary production defined as
where ChlE is the “efficiency” of Chl in planta, GPP is gross primary production, PAR
in is incident radiation in PAR spectral region (400–700 nm), and Chl
plant is plant Chl content calculated as the product of leaf Chl content, Chl
leaf, and Leaf Area Index (LAI).
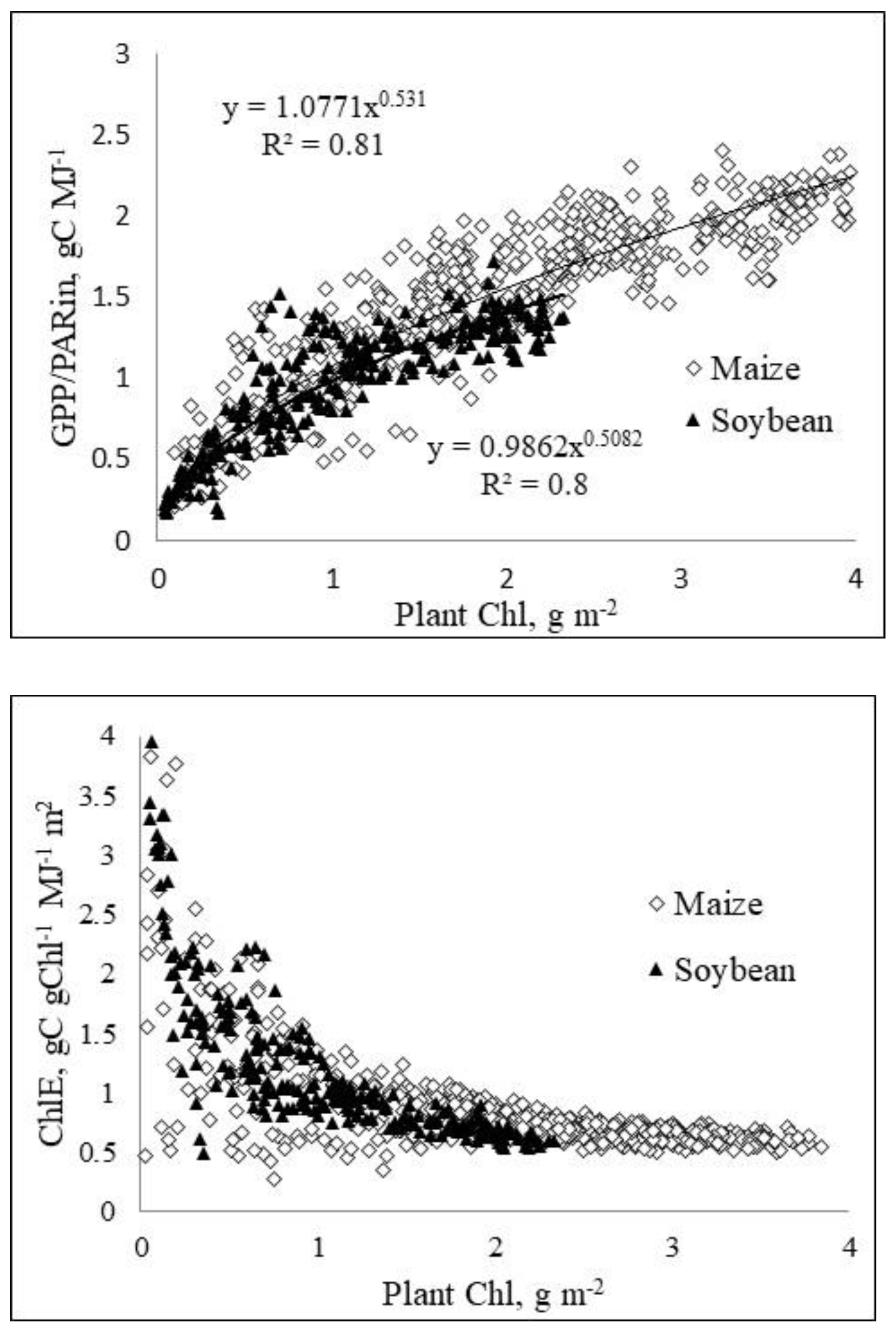
Figure 4.
Relationships of (a) GPP/PARin and (b) efficiency of the chlorophyll regarding primary production, ChlE vs. plant chlorophyll content. The slope of GPP/PAR vs. Chl relationship decreases as plant Chl exceeds 1 g m–2. However, these relationships are much closer to linearity than fAPARPS vs Chl relationships (Fig. 3). ChlE = GPP/(Chlplant×PARin).
Figure 4.
Relationships of (a) GPP/PARin and (b) efficiency of the chlorophyll regarding primary production, ChlE vs. plant chlorophyll content. The slope of GPP/PAR vs. Chl relationship decreases as plant Chl exceeds 1 g m–2. However, these relationships are much closer to linearity than fAPARPS vs Chl relationships (Fig. 3). ChlE = GPP/(Chlplant×PARin).
The ChlE parameter convincingly demonstrates the decline of Chl efficiency regarding carbon assimilation along with an increase in Chl content (Fig. 4b). Importantly, Chl efficiency in primary production is higher than Chl efficiency in PAR absorption (Fig. 3). While ChlE per unit GPP decreases ca. 5-fold, the decrease of Chl efficiency in PAR absorption is more than 10-fold.
An increase in Chl brings about an increase FR absorption without limitation due to the small absorption coefficient of Chl in this range. Indeed, the absorption coefficient of dense crops assessed in situ does not exceed 3–5% of that in the PAR range; therefore, the pathway of FR light in leaves and canopy is more than 10-time longer than that of PAR quanta (Gitelson et al., 2021; Gitelson et al., 2019). So, our hypothesis is that the productivity of vegetation possessing moderate-to-high Chl content is boosted by an increase of FR absorption on the background of a high PAR absorption (which is already saturated). To test this hypothesis, we assessed the efficiency of Chl regarding its spectral contribution to GPP employing the concepts of light absorption coefficient, α, and its spectral response to GPP. An important advantage of this approach is that α can be remotely sensed and inferred from plant reflectance.
Spectral light absorption coefficient was defined as (Gitelson et al., 2019)
where
is light absorption coefficient at wavelength
,
is reflectance in the near-infrared (NIR) spectral region, and
is reflectance at wavelength λ (in the PAR and FR regions). Notably,
α turned to be a very useful parameter empowering one to assess spectral contributions of different pigment groups, estimate precisely Chl content at the leaf (Gitelson and Solovchenko, 2017; Gitelson et al., 2020), plant and canopy levels (Gitelson et al., 2019); assessing phenology (Peng et al., 2024), photosynthetic capacity (Muramatsu et al., 2024), crop and conifer forest GPP (Peng et al., 2011; Wang et al., 2020) and quantum yield of photosynthesis (Gitelson et al., 2022). Accordingly,
α is instrumental for the assessment of the efficiency of plants in converting the absorbed radiation into photosynthetic carbon uptake.
The spectral response parameter was previously introduced as a quantitative measure for estimation of contribution of Chl absorption in different spectral ranges to GPP (Gitelson et al., 2021; Gitelson et al., 2019). In turn, the spectral response function of
α to GPP per unit of incoming PAR (
) was defined as the ratio of the first derivative of the best-fit function relating
α to the
in and the root mean square error (RMSE) of this relationship:
The magnitude of is governed by (i) the sensitivity of to i.e., slope of the relationship, and (ii) the RMSE of the same relationship showing how close this relationship is to linearity. The closer the relationship is to linearity (i.e., the smaller the RMSE) and the higher the sensitivity of αλ to GPP, the higher is the response function.
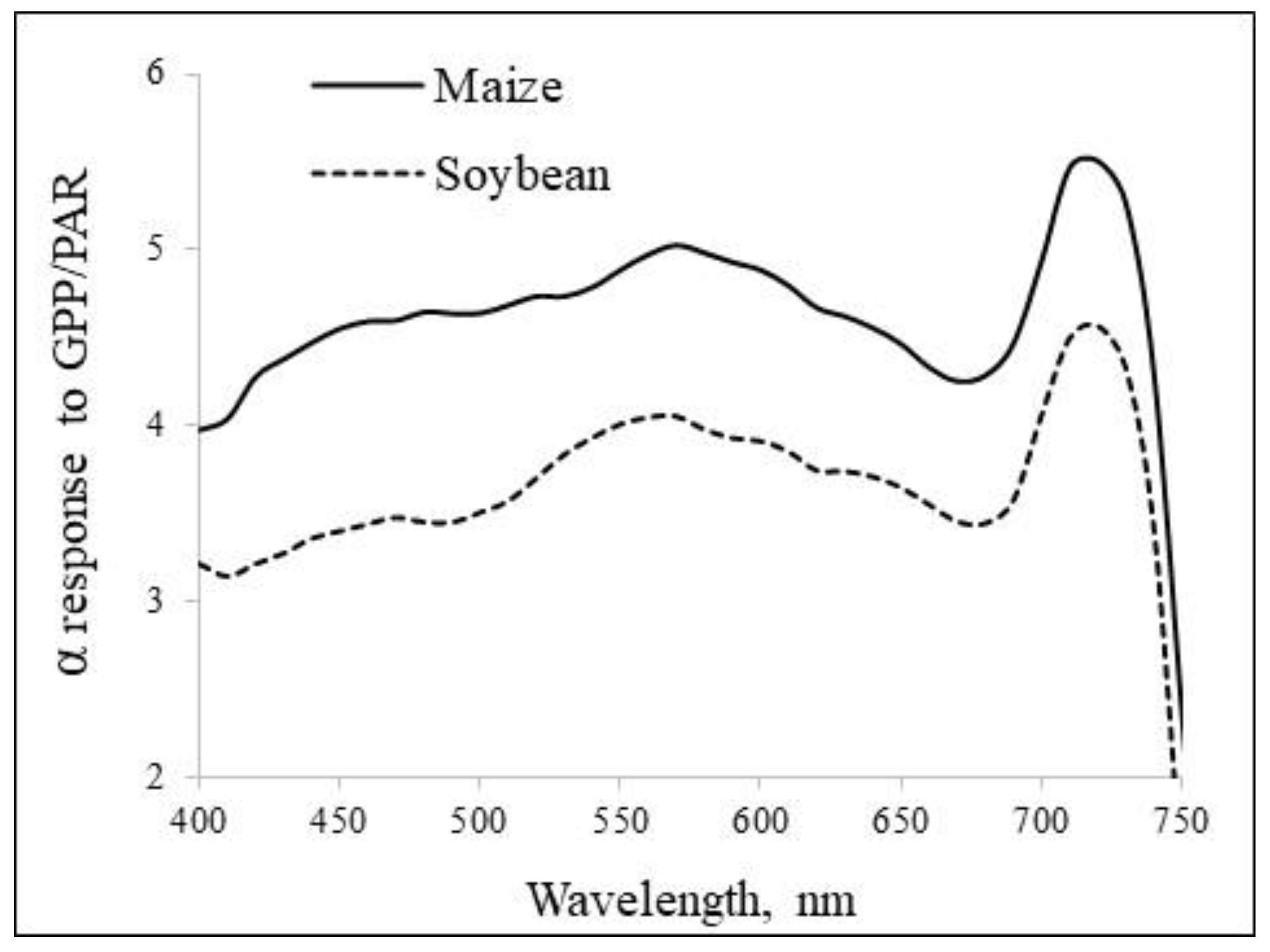
Spectral response of α in the PAR and FR regions is tightly related to GPP in plants with widely varying leaf Chl content, leaf area index, structural and functional traits (see e.g. Fig. 5 and Gitelson et al, 2025). Clearly, only absorption of FR increases with an increase in Chl content and plant leaf area. At the peak of Chl content during the plant ontogenesis, contribution of FR to the primary production can be as high as 20% or even 30% of absorbed PAR (in soybean and maize, respectively, (Gitelson et al., 2025)).
Figure 5.
Spectral response of absorption coefficient to GPP. Note the peaks of the response of to GPP in the FR region of the spectrum (Gitelson et al., 2025).
Figure 5.
Spectral response of absorption coefficient to GPP. Note the peaks of the response of to GPP in the FR region of the spectrum (Gitelson et al., 2025).
Strong positive relationships of GPP vs. Chl (Fig. 4A) confirm our hypothesis about contribution of FR light absorption to GPP. While fAPARPH vs. Chl relation is essentially non-linear (Fig. 3), GPP vs. Chl relation is quite close to linear (Fig. 4a). When Chl content increases from moderate to high, fAPARPS becomes insensitive to Chl (Fig. 3), however, absorption in the FR increases linearly with Chl increase and it increases slope of GPP/PARin vs. Chl relationship. Thus, increase in FR absorption compensates, to some degree, the decrease in Chl efficiency in PAR absorption and facilitates the “safe” transformation of the light energy captured in the PAR region by balancing of the electron flow in the chloroplast membranes (see above).
Taken together, the findings presented above support the hypothesis about important functional role of the seemingly redundant Chl making a significant contribution to the overall energy budget and hence to the photosynthetic productivity of plants by intercepting FR radiation via its tailing long-wave absorption.
Practical considerations
One of the key ongoing efforts of scientists and practitioners is to boost the efficiency of photosynthesis in crops to approach its theoretical maximum. This problem was tackled with bioengineering methods e.g. by transgenic up-regulation of specific photosynthetic enzymes leading to an increase in photosynthesis and yields of rice and soybean, also in the field (Yoon et al., 2020). However, the use of transgenic crops is associated with diverse issues related to legal regulation, customer acceptance, and potential risks which are difficult to foresee. Therefore, alternative approaches to increasing crop photosynthetic productivity are coming to foreground. How can one harness the “FR boost”? The most obvious avenue is FR light supplementation in glasshouses. Modern glasshouses are increasingly fitted with LED lamps instead of high-pressured sodium lamps resulting in dramatic decline of the IR component in the glasshouse illumination environment. In contrast to sunlight, artificial light sources such as LEDs often lack FR, resulting in a PSS (phytochrome steady state, Pfr/(Pr+Pfr) value higher than that under solar light (∼0.70). This resulted in a strongly reduced fruit production due to reduced fruit growth associated with reduced source strength and delayed flowering. This was demonstrated e.g. in tomato plants grown under artificial light without FR during the light period (Kalaitzoglou et al., 2019). Notably, it was impossible to compensate for the adverse effects of the absence of FR component in the spectrum of light in the glasshouse by a simple short-time end-of-day treatment. This finding indicates that the energetic role of FR can be as important as the R/FR balance. Overall, enhancement of FR absorption increases yield, improves nutrient utilization and plant architecture translating into an increase in net productivity.
There is a great potential in using FR radiation to improve the field crop performance as well, e.g. to improve radiation use efficiency in strip intercropping by supplementing FR light from any direction (see (Tan et al., 2022; Zhang et al., 2021) and references therein). The benefits of FR absorption in crops were estimated recently in soybean, the expected productivity boost was as high as 25% (Wang et al., 2025). However, in this work it was suggested to achieve the mentioned increase in FR quanta interception by genetically engineering the plants to produce and accumulate Chl species non-native to green plants.
Concluding remarks
Here, we argue that the “physiologically redundant Chl” is beneficial for plant since it enhances absorption of FR and its contribution to primary production, GPP. Our hypothesis is supported by estimations of plant light capture capacity, α, and its relationship with plant GPP. The analysis of α response to GPP reveals that in the case of moderate to high Chl content FR absorption greatly contributes to total absorption of light and hence to GPP (whereas response of α to GPP in the PAR region declines significantly). It is no surprise that this role was so far overlooked since it is hardly observable in “optically thin systems” traditionally used in photobiology and photosynthesis studies.
On one hand, this hypothesis aligns well with the widespread, if not ubiquitous presence of sizeable FR absorption in the spectra of healthy, mature vegetation rich in Chl. On the other hand, it is supported by the multifaceted physiological role of FR quanta in the functioning of photosynthetic apparatus in optically complex light environment of plant canopies under fluctuation environmental conditions. Importantly, plants do not need peculiar biochemical adaptations such as “exotic” forms of Chl to leverage the light energy of FR. In other words, the ability to put the FR quanta to work is inherent to many, if not all studied plant species. The mechanism for harnessing FR relies upon efficient harvesting FR quanta which arises at the levels of individual plants and canopy level. From the practical standpoint, the “FR productivity boost” can be leveraged using the leaf/canopy-level optical features of native plants and/or adjustment of illumination environment so the plant can capture more FR photons (Zhang and Zhang, 2019) cancelling largely the need for engineering of its photosynthetic apparatus.